1. Introduction
Fruits are essential components of the human diet due to their high contents of vitamins, minerals, and bioactive compounds [
1]. Brazil ranks as the third-largest fruit producer worldwide, with approximately 43 million tons produced in 2023 [
2]; however, several native species remain underexploited because of limited technological and industrial development.
In this context, the genus
Euterpe (Arecaceae) is of ecological and socioeconomic relevance in tropical America. While
Euterpe oleracea (açaí) has a well-established production chain,
Euterpe edulis (juçara), native to the Brazilian Atlantic Forest, has historically been subjected to predatory palm heart extraction, placing the species at risk of extinction [
3,
4,
5,
6,
7]. The sustainable use of its fruits has emerged as a promising conservation strategy, enabling income generation without tree removal [
3].
Juçara fruits are small, purple, and non-climacteric, with seeds representing approximately 85% of the fruit mass [
8,
9]. The pulp is rich in lipids, proteins, and dietary fiber and is particularly notable for its phenolic compounds, especially anthocyanins, as well as carotenoids, which are associated with antioxidant and other bioactive properties [
10,
11,
12]. Moreover, the fixed oil of
E. edulis is characterized by a high oleic acid content, reinforcing its nutraceutical potential [
8,
13].
Despite its considerable functional potential, the commercial viability of juçara fruits is limited by their high perishability. The elevated moisture content and rich nutrient composition favor enzymatic reactions and microbial growth, leading to rapid deterioration. The challenge is compounded by estimated annual losses of 30% along the fruit and vegetable supply chain, resulting from deficiencies in post-harvest handling, transportation, and storage, as well as the pronounced seasonality of juçara harvest, which is largely restricted to the period between March and June [
8,
13,
14]. Consequently, immediate processing of the pulp is essential not only to reduce post-harvest losses but also to mitigate seasonality and ensure year-round market availability. In addition, juçara may present emerging commercial and export potential, similar to that observed for Amazonian açaí [
15]. Although this potential has not yet been fully established, the expansion of international markets for related species highlights the importance of investigating its processing behavior and technological characteristics. Therefore, the present study may contribute to the international debate on the sustainable valorization of underexplored tropical fruit species.
Among the strategies employed to extend the shelf-life of perishable products, pulp drying represents an effective approach to reduce moisture content, inhibit microbial and enzymatic activity, and add commercial value, while also lowering logistics and transportation costs [
16]. Although several advanced drying technologies have been developed, such as freeze drying, vacuum drying, microwave drying, osmotic drying, spray drying, and fluidized bed-drying, convective drying in air-circulating ovens remains widely used due to its operational simplicity and relatively low cost [
17]. However, inappropriate combinations of drying time and temperature may lead to undesirable losses in nutritional, sensory, or biological activity properties [
18,
19,
20].
In this context, the study of drying kinetics combined with mathematical modeling constitutes an essential approach for predicting moisture removal behavior and evaluating the thermal degradation of bioactive compounds. Such tools enable process optimization and have been successfully applied to several fruit pulps [
18,
21,
22,
23,
24]. However, investigations addressing the drying kinetics and phenolic compound retention in
E. edulis pulp remain limited. Among the variables governing oven-drying processes, temperature and drying time—considered jointly as a time/temperature binomial—are the most critical parameters for achieving the target final moisture content while minimizing losses of bioactive constituents. Accordingly, different studies have sought to establish optimal operating conditions. For instance, the drying of Brazilian pequi pulp (
Caryocar brasiliense Camb.) in forced-air ovens [
23] was evaluated at 40 and 55 °C, with analyses of carotenoids, total phenolics, phenolic acids, and flavonoids. The condition of 55 °C for 6 h resulted in the highest carotenoid retention. Likewise, studies on oven-dried cupuaçu and bacuri pulps [
22] demonstrated that the time/temperature combination is decisive for process optimization, particularly in maximizing phenolic compound retention.
In powdered form, the dehydrated juçara pulp exhibits versatile application potential. In the food industry, it can be used as a natural colorant and flavoring agent, for instance in dairy beverages [
25]. In addition to imparting distinctive sensory characteristics, it enhances the nutritional value of formulations due to its high content of anthocyanins, phenolic compounds, dietary fiber, and unsaturated fatty acids [
9,
26]. Juçara extract has also been used as a natural antioxidant for broiler meat [
27].
Therefore, the aim of this study was to investigate the behavior of phenolic compounds in E. edulis pulp during convective drying in an air-circulating oven and to apply mathematical models to describe the drying kinetics under different time and temperature conditions. The ultimate goal was to identify optimal drying parameters that enable efficient moisture reduction while preserving the bioactive constituents of juçara pulp.
4. Discussion
In Brazil, there is no specific legislation regulating pulps derived from E. edulis. However, the Brazilian Health Regulatory Agency (ANVISA) recommends applying the legislation established for açaí pulp (Euterpe oleracea Mart.), according to Normative Instruction No. 01 of 7 January 2000, issued by the Brazilian Ministry of Agriculture, Livestock and Food Supply (MAPA), which defines the characteristics and quality parameters for fruit pulps. According to this regulation, thin or popular açaí pulp (Type C) is obtained by water addition followed by filtration and must present a total solids content between 8% and 11%, resulting in a product of lower density. Based on these parameters, the samples evaluated in this study exhibited moisture contents above the recommended limits, indicating possible dilution beyond acceptable levels. Therefore, the results should be interpreted in light of the relatively high water content of the samples, which may have influenced the measured properties.
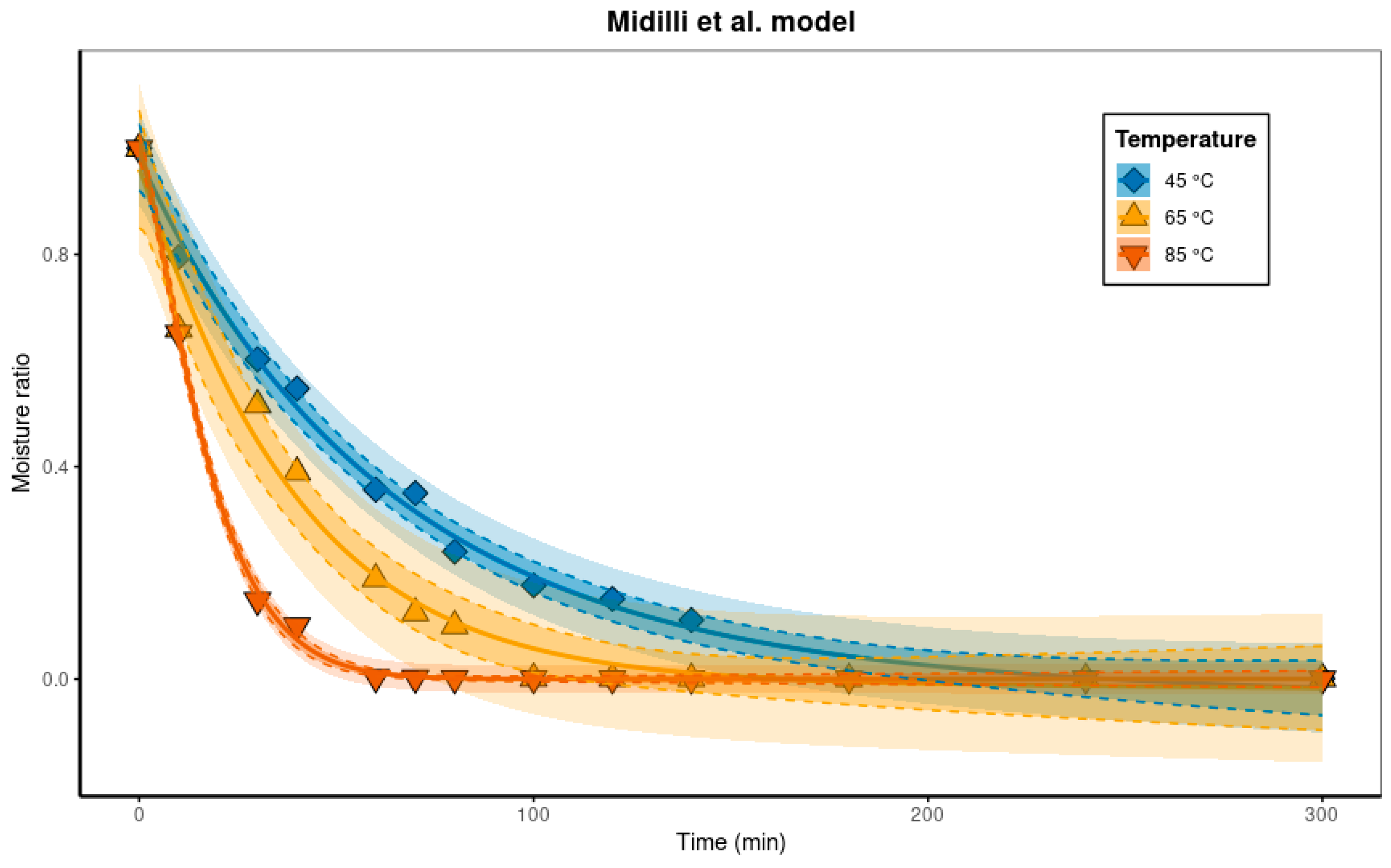
Regarding the drying process, only a slight moisture loss was observed during the first 10 min for all temperatures evaluated, with a reduction below 1.4% and no significant differences among treatments. However, after 30 min, drying at 85 °C resulted in a pronounced moisture reduction, reaching 12.8%. For drying at 65 °C, a comparable reduction (~10%) was observed only after 60 min, whereas at 45 °C, this reduction occurred only after approximately 100 min.
These results indicate that higher temperatures promote faster moisture removal. After 100 min, the two highest temperatures (85 and 65 °C) resulted in moisture reductions exceeding 90% of the initial content, while at 45 °C the reduction over the same period was only 10.6%. Drying at 45 °C achieved a moisture reduction greater than 90% only after approximately 180 min.
Mass transfer during drying is influenced by the physical properties of the material, temperature, and initial moisture content. Heat transfer, in turn, is determined by air temperature, moisture, airflow rate and direction, as well as the exposed surface area of the solid [
42]. In this study, the controlled variables were drying air temperature and exposure time.
Thus, it is confirmed that higher drying temperatures allow moisture content to be reached in shorter times. Consequently, the drying curves become steeper: at 85 °C, 60 min were required for the sample to reach a moisture content close to 10%, whereas at 65 °C this value was achieved after approximately 100 min, and at 45 °C only after 180 min.
With regard to the drying models, it was found that the two-term model generally presented higher R2 values and lower chi-square and RMSE values compared with the other models evaluated (although, in general, the adjustments for all models were very good), as already mentioned. Considering the issues of non-identifiability of the two-term model and the numerical problems in the estimation process, other models should be considered as alternatives for the present problem—especially if we consider that all models showed excellent fits to the experimental data. In this sense, both the Midilli et al. and Page models yielded similar values for the statistical performance indicators. Nevertheless, the Midilli et al. model employs four adjustable parameters, in contrast to the two parameters of the Page model. For both formulations, a strong correlation between the parameters k and n was observed, highlighting limitations in the independent interpretation of these parameters. Finally, it should be noted that parameter b in the Midilli et al. model presented extremely low values, pointing to the insignificance of the term bt in the model (obviously, for the problem in question).
During the first 10 min of drying, a relatively small reduction in total phenolics (TP), below 6%, was observed regardless of the temperature applied. This behavior suggests that, during the initial drying stage, when moisture removal was similar across the three thermal conditions, the TP values remained comparable. This result contrasts with the findings of other studies (on other fruit pulps) [
18], which reported greater initial TP losses at higher drying temperatures.
After 30 min, drying at 85 °C resulted in significantly higher losses (~30%) compared with the other temperatures, which remained below 16%. This pronounced reduction may be attributed to increased thermal stress imposed on more thermolabile phenolic compounds. For drying at 45 and 65 °C, similar losses (~30%) were observed only at later times, after 100 and 80 min, respectively, indicating greater stability of these compounds under less severe thermal conditions.
From 100 min onward, drying at 65 and 85 °C exhibited very similar losses, around 60%, which persisted up to 180 min. This convergence suggests that, after a certain degree of moisture reduction, TP degradation occurs in a similar manner, regardless of the thermal intensity applied. This behavior coincides with the point at which drying at 65 °C exhibited a sharp decrease in moisture content, reinforcing the relationship between water removal and phenolic degradation.
In contrast, drying at 45 °C resulted in TP losses exceeding 50% only after 180 min, which coincided with a marked reduction in moisture content. This finding indicates that moisture reduction exerts a more decisive influence on TP concentration than total drying time, particularly under milder temperature conditions.
Overall, these results indicate that degradation of phenolic compounds during drying is associated not only with direct thermal effects but also with structural transformations of the food matrix. During drying, four main factors contribute to the degradation of organic compounds: (i) high temperature and prolonged heat exposure accelerate degradation reactions [
43]; (ii) exposure to oxygen during drying and the high reducibility of these bioactive compounds promote oxidative degradation [
44]; (iii) removal of hydration water, which normally protects reactive molecular sites; and (iv) structural changes in the pulp that increase the exposure of reactive sites [
45]. The combined effect of these factors favors oxidative and degradation reactions, intensifying phenolic losses throughout the drying process. Studies have demonstrated that phenolic compounds are highly sensitive to thermal processing, particularly under prolonged heat exposure, which can lead to structural degradation, oxidation, and polymerization [
46,
47,
48]. The observed reduction in phenolic content during drying may also be attributed to interactions between polyphenols and other macromolecules, as well as to chemical modifications that alter their extractability and hinder their accurate quantification by conventional analytical methods. Moreover, the rate and conditions of dehydration play a critical role in determining the extent of these changes, as faster or milder drying may better preserve phenolic stability.
In general, the results of this study indicates that TP degradation is more closely related to physical and structural changes induced by moisture removal than to heat exposure time alone. Therefore, dehydration kinetics play a central role in determining the stability of phenolic compounds during the drying process.
Considering the two approaches adopted for estimating the activation energy, the values obtained were
Ea = 19.01 and
Ea = 16.35 kJ mol
−1. These values are of the same order of magnitude as those reported in the literature for the thermal degradation of phenolic compounds and anthocyanins in fruit pulps. In particular, an activation energy of 24.16 kJ mol
−1 has been reported for anthocyanin inactivation in açaí berry pulp [
49].
An important point to consider in this study is the correlation between moisture, expressed as moisture ratio, and total phenolic content, as discussed in the literature [
50].
Figure 9 shows the correlations between moisture ratio values and total phenolic content for temperatures of 45, 65, and 85 °C. It should be clear that these results must be analyzed carefully—and should not be extrapolated to other experimental conditions—since moisture (represented by the moisture ratio) and total phenolic content vary over time and depend on other factors. On the other hand, they can provide useful insights into the conditions of the experiment. In this sense, the data presented for each temperature in
Figure 9 should be understood as a “Pareto curve” (used here in a descriptive, not optimization-based, sense) or a multi-criteria compromise region, where there are two conflicting objectives—reducing moisture and maintaining phenolic content. It is clear (especially from the data at 65 °C) that at the end of the process there is an abrupt decrease in phenolic content with virtually constant moisture values. This situation can be explained by the fact that the drying process has already reached equilibrium moisture content (and is essentially complete), while the degradation of phenolics is still occurring. The results in
Figure 9 indicate that it is possible to obtain, for example, a dry pulp with
MR values close to 10
−3 while maintaining the phenolic content at around 10–12 mg/g at 65 °C. Drying values at 45 °C and 85 °C, for the same
MR value, generate lower phenolic contents. Multi-objective approaches, including Pareto curves and surfaces, have been applied to drying problems [
51].
A direct comparison with results obtained using other drying techniques is challenging due to the variability in fresh pulp characteristics and the broad range of operating conditions reported in the literature. Nevertheless, some contextual considerations can be drawn. For example, a recent study [
52] reported total phenolic retention above 80% when drum drying was applied (residence time of 20 s at 135 °C) for the production of juçara flakes. In another study [
53], the production of “pure” juçara powder (without carrier agents) using a laboratory-scale spray dryer resulted in approximately 90% retention of total phenolic compounds, with inlet and outlet air temperatures of 160 °C and 86 °C, respectively.
In the present study, considering the experiments conducted at 65 °C, approximately 50% of the total phenolics were preserved, with a final moisture content on the order of 10
−3. Although this retention is lower than the values reported for drum and spray drying, it should be interpreted in light of the distinct raw material characteristics and processing conditions. Another relevant aspect concerns process yield. Oven drying may provide higher powder yields than spray drying, particularly at laboratory-scale. For instance, in chamomile drying [
54], powder yields of around 90% were reported for oven drying, whereas spray drying under comparable laboratory conditions resulted in yields below 30%. Specifically for juçara, spray drying applied to microencapsulation of the pulp has been reported to produce yields ranging from 21.5% to 53.1% [
55], depending on formulation and operating parameters.
It is also important to note that commercial (frozen) pulp was used in the present study. There is evidence that freezing time may reduce total phenolic content by approximately 30% [
56]. Moreover, the initial moisture content of the pulp suggests that water may have been added prior to processing, which could further dilute phenolic concentrations and help explain the lower total phenolic values observed here compared with those reported in other studies [
53].
One limitation of the present study is the absence of an evaluation of the long-term storage stability of dried juçara pulp powder. Bioactive compounds such as anthocyanins and other phenolic constituents are known to be susceptible to degradation during storage, particularly as a function of temperature, oxygen availability, light exposure, and residual moisture content [
57]. In this sense, future research will include storing the dried pulp for a period of 12 months (under different conditions) to assess the maintenance of phenolic compounds and physical properties. In addition, investigations into the application of dry pulp in different food matrices, such as fortification of juices, yogurts, and baked goods, may provide information on interactions with other ingredients and retention of bioactive compounds during processing. Further investigations should evaluate different drying techniques (such as fluidized bed drying, spray drying, or vacuum drying) and pre-treatments to preserve phenolic compounds, with a special focus on anthocyanins and color stability.
It is also important to emphasize that phenolic compounds, sugars, and other constituents of juçara pulp do not exist as isolated entities, but rather as components of a complex and interactive food matrix. Chemical and physical interactions among these constituents may influence the stability and degradation behavior of bioactive compounds during the dehydration process. Considering that the present study exclusively evaluated phenolic compounds, the absence of an integrated matrix-based approach represents a limitation. Therefore, future, more comprehensive investigations may contribute to a better understanding of the interactions among pulp constituents and their effects on the stability of phenolic compounds during dehydration.