Metal–Organic Frameworks as Multifunctional Platforms for Chemical Sensors: Advances in Electrochemical and Optical Detection of Emerging Contaminants
Abstract
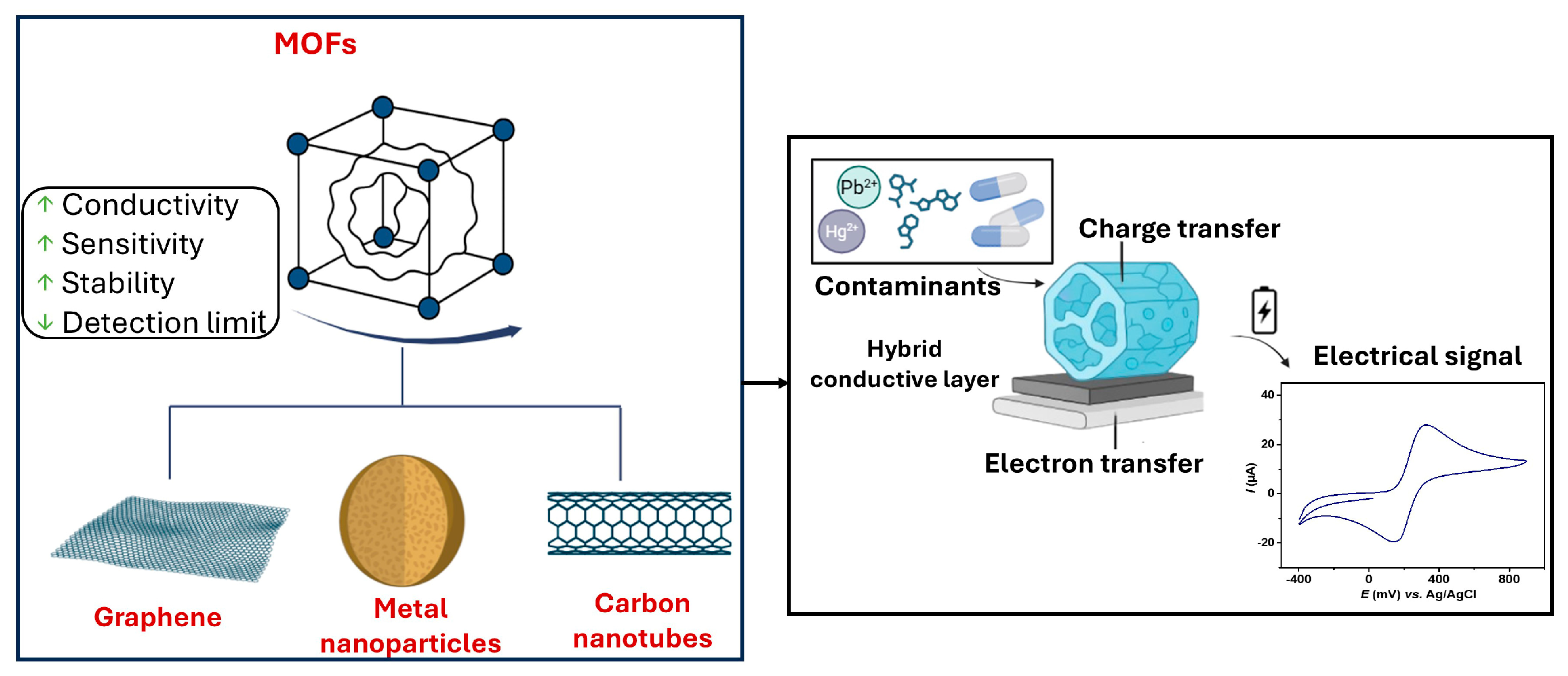
1. Introduction
2. Synthesis and Properties of Metal–Organic Frameworks
2.1. Synthesis and Optimization of Metal–Organic Frameworks Design for Chemical Sensors
2.2. Organic Linkers Used in the Synthesis of Metal–Organic Frameworks
2.3. Main Characteristics of Metal–Organic Frameworks for Sensing
2.3.1. Selectivity and Sensitivity
2.3.2. Stability
2.3.3. Fast Response Time
2.3.4. Reusability
2.4. Design of MOF-Based Sensing Platforms
3. Signal Transduction Strategy
3.1. MOFs for Electrochemical Sensors
3.2. Integration of MOFs with Conducting Nanomaterials
3.3. MOFs for Colorimetric and Optical Sensors
Principles of Optical Response: Quenching, Colorimetric, Luminescence
3.4. Optical Sensors Applied to the Monitoring of Metal Ions and Emerging Contaminants
4. Current Challenges and Outlook
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Wiart, M.; Tavakoli, C.; Hubert, V.; Hristovska, I.; Dumot, C.; Parola, S.; Lerouge, F.; Chauveau, F.; Canet-Soulas, E.; Pascual, O.; et al. Use of metal-based contrast agents for in vivo MR and CT imaging of phagocytic cells in neurological pathologies. J. Neurosci. Methods 2023, 383, 109729. [Google Scholar] [CrossRef]
- Aduko, J.; Yakubu, M.A.; Anokye, K. Assessing the environmental impacts of urban sprawl on vegetation cover and ecosystem integrity in Wa municipality, Ghana. World Dev. Sustain. 2025, 6, 100225. [Google Scholar] [CrossRef]
- Estrada-Almeida, A.G.; Castrejón-Godínez, M.L.; Mussali-Galante, P.; Tovar-Sánchez, E.; Rodríguez, A. Pharmaceutical Pollutants: Ecotoxicological Impacts and the Use of Agro-Industrial Waste for Their Removal from Aquatic Environments. J. Xenobiot. 2024, 14, 1465–1518. [Google Scholar] [CrossRef] [PubMed]
- Pharmaceutical Residues in Freshwater; OECD Publishing: Paris, France, 2019. [CrossRef]
- UN-Water. Progress on Wastewater Treatment—2024 Update; UN-Water: New York, NY, USA, 2024. [Google Scholar]
- Osuoha, J.O.; Anyanwu, B.O.; Ejileugha, C. Pharmaceuticals and personal care products as emerging contaminants: Need for combined treatment strategy. J. Hazard. Mater. Adv. 2023, 9, 100206. [Google Scholar] [CrossRef]
- Li, J.; Sun, D.; Wen, Y.; Chen, X.; Wang, H.; Li, S.; Song, Z.; Liu, H.; Ma, J.; Chen, L. Molecularly imprinted polymers and porous organic frameworks based analytical methods for disinfection by-products in water and wastewater. Environ. Pollut. 2024, 356, 124249. [Google Scholar] [CrossRef]
- Eramma, N.; Shreeja; Naidu, S.B.K.; Patil, S.J. Monitoring and Management of Pollutants in the Environment. In Biotechnology for Environmental Sustainability; Springer: Berlin/Heidelberg, Germany, 2025; pp. 673–684. [Google Scholar] [CrossRef]
- Vasilachi, I.; Asiminicesei, D.; Fertu, D.; Gavrilescu, M. Occurrence and Fate of Emerging Pollutants in Water Environment and Options for Their Removal. Water 2021, 13, 181. [Google Scholar] [CrossRef]
- Li, X.; Shen, X.; Jiang, W.; Xi, Y.; Li, S. Comprehensive review of emerging contaminants: Detection technologies, environmental impact, and management strategies. Ecotoxicol. Environ. Saf. 2024, 278, 116420. [Google Scholar] [CrossRef]
- Nishmitha, P.S.; Akhilghosh, K.A.; Aiswriya, V.P.; Ramesh, A.; Muthuchamy, M.; Muthukumar, A. Understanding emerging contaminants in water and wastewater: A comprehensive review on detection, impacts, and solutions. J. Hazard. Mater. Adv. 2025, 18, 100755. [Google Scholar] [CrossRef]
- Harrison, S.; Barnett, C.; Short, S.; Uluseker, C.; Silva, P.V.; Pavlaki, M.D.; Roberts, S.; Vieira, M.; Lofts, S.; Loureiro, S.; et al. Continuous improvement towards environmental protection for pharmaceuticals: Advancing a strategy for Europe. Environ. Sci. Eur. 2025, 37, 128. [Google Scholar] [CrossRef]
- Alqarni, A.M. Analytical Methods for the Determination of Pharmaceuticals and Personal Care Products in Solid and Liquid Environmental Matrices: A Review. Molecules 2024, 29, 3900. [Google Scholar] [CrossRef]
- Ribeiro, I.S.; Andrade, T.d.A.; Silva, T.A.; de Jesus, J.R. Integration of Mesoporous Copper Coordination Polymer in Electrochemical Platform for Detection of Fluoroquinolone Antibiotic in Biological and Food Samples. ACS Omega 2025, 10, 51466–51478. [Google Scholar] [CrossRef] [PubMed]
- Soares Ribeiro, I.; de Araujo Andrade, T.; Almeida Silva, T.; da Silva, M.J.; de Jesus, J.R. Electrochemical sensor based on coordination compound [Co(BDC)]n allows detection of catecholamine neurotransmitter associated with neurological disorder from biological samples. Microchem. J. 2024, 206, 111571. [Google Scholar] [CrossRef]
- de Araujo Andrade, T.; Ribeiro, I.S.; Silva, T.A.; de Souza, L.K.A.; Coltro, W.K.T.; Borges, L.P.; Silva, D.M.R.R.; Garcia, P.T.; de Jesus, J.R. Diagnosis of viral infectious diseases through sensitive detection of human serum antibodies using a modified label-free electrochemical biosensor. Anal. Bioanal. Chem. 2024, 416, 6345–6355. [Google Scholar] [CrossRef]
- Qian, S.; Cui, Y.; Cai, Z.; Li, L. Applications of smartphone-based colorimetric biosensors. Biosens. Bioelectron. X 2022, 11, 100173. [Google Scholar] [CrossRef]
- Anchidin-Norocel, L.; Gutt, G.; Tătăranu, E.; Amariei, S. Electrochemical sensors and biosensors: Effective tools for detecting heavy metals in water and food with possible implications for children’s health. Int. J. Electrochem. Sci. 2024, 19, 100643. [Google Scholar] [CrossRef]
- Dhahi, T.h.S.; Yousif Dafhalla, A.K.; Tayfour, O.E.; Mubarakali, A.; Alqahtani, A.S.; Tayfour Ahmed, A.E.; Elobaid, M.E.; Adam, T.; Gopinath, S.C.B. Advances in nano sensors for monitoring and optimal performance enhancement in photovoltaic cells. IScience 2024, 27, 109347. [Google Scholar] [CrossRef]
- Hosseinikebria, S.; Khazaei, M.; Dervisevic, M.; Judicpa, M.A.; Tian, J.; Razal, J.M.; Voekcker, N.H.; Nilghaz, A. Electrochemical biosensors: The beacon for food safety and quality. Food Chem. 2025, 475, 143284. [Google Scholar] [CrossRef]
- Pimpilova, M. A brief review on methods and materials for electrode modification: Electroanalytical applications towards biologically relevant compounds. Discov. Electrochem. 2024, 1, 12. [Google Scholar] [CrossRef]
- Nandi, S.; Dalapati, R. Fluorometric Sensing of Arsenic in Water: Recent Developments in Metal-Organic Framework-Based Sensors. Processes 2025, 13, 923. [Google Scholar] [CrossRef]
- El Hammoudani, Y.; Achoukhi, I.; Haboubi, K.; El Youssfi, A.; Benaissa, C.; Bourjila, A.; Touzani, A.; Ahmadi, K.E.; Allaoui, H.; Kasmi, A.E.; et al. Graphene-Based Metal–Organic Frameworks for Advanced Wastewater Treatment: A Review of Synthesis, Characterization, and Micropollutant Removal. Processes 2025, 14, 117. [Google Scholar] [CrossRef]
- de Jesus, J.R.; de Sousa Pereira, M.V.; Ribeiro, I.S.; de Araujo Andrade, T.; de Carvalho, J.P.; de Tarso Garcia, P.; Junior, C.A.L. Multifunctional and eco-friendly nanohybrid materials as a green strategy for analytical and bioanalytical applications: Advances, potential and challenges. Microchem. J. 2023, 194, 109331. [Google Scholar] [CrossRef]
- Pereira, M.V.S.; Ribeiro, I.S.; da Silva, A.R.V.; Silva, J.P.P.; Aquino, W.C.P.; Lopes Júnior, C.A.; de Jesus, J.R. Sample preparation for environmental monitoring under the perspective of white analytical chemistry: Green, sustainable and, effective. Microchem. J. 2025, 215, 114444. [Google Scholar] [CrossRef]
- Aquino, W.C.P.; Ribeiro, I.S.; Pereira, M.V.S.; Guimarães, L.M.; da Silva-Junior, G.R.; da Silva, R.C.; de Jesus, J.R. Multivariate Optimization of an Eco-Friendly Zinc-Based MOF for Adsorptive Removal of Emerging Contaminants in Food Samples. ACS Omega 2025, 10, 42901–42912. [Google Scholar] [CrossRef]
- de Jesus, J.R.; Soares Ribeiro, I.; Passos de Carvalho, J.; Luiz Alves de Oliveira, K.; Pereira Lopes Moreira, R.; Chagas da Silva, R. Successful synthesis of eco-friendly Metal-Organic framework ([Ni(BDC)]n) allows efficient extraction of multiresidues pesticides and dyes from fish samples. Microchem. J. 2024, 201, 110592. [Google Scholar] [CrossRef]
- Kumar, R.; Shafique, M.S.; Chapa, S.O.M.; Madou, M.J. Recent Advances in MOF-Based Materials for Biosensing Applications. Sensors 2025, 25, 2473. [Google Scholar] [CrossRef] [PubMed]
- Kidanemariam, A.; Cho, S. Metal–Organic-Framework-Based Optical Biosensors: Recent Advances in Pathogen Detection and Environmental Monitoring. Sensors 2025, 25, 5081. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, Y.; Chen, L.; Xie, X.; Sun, J. Gel-state MOFs for environmental decontamination: Synthesis, application and optimization. Chem. Eng. J. 2024, 499, 156241. [Google Scholar] [CrossRef]
- Zhou, H.; Baig, M.M.; Gu, S.; Zou, W.; Zhang, J.; Dou, F.; Xu, J.; Li, L.; Zhang, Y.; Pang, H.; et al. Design, synthesis, and optimization of MXene- and MOF-based materials for 3D printed energy storage devices and beyond. Chin. Chem. Lett. 2025, 111489. [CrossRef]
- Aslam, M.; Ali, M.Z.; Akram, M.U.; Akram, A.; Laiba, L.; Naz, S.; Verpoort, F. MOFs for (bio)chemical sensor devices. J. Ind. Eng. Chem. 2025, 155, 52–88. [Google Scholar] [CrossRef]
- Sheokand, A.; Sindhu, S.; Bulla, M.; Devi, R.; Dahiya, R.; Sanger, A.; Sisodia, A.K.; Mishra, A.K.; Kumar, V. Transformative progress in MOF-based hybrid materials for supercapacitors and gas sensors: Multifunctional platforms for sustainable technologies. Coord. Chem. Rev. 2026, 549, 217213. [Google Scholar] [CrossRef]
- Kukkar, P.; Kukkar, D. Recent breakthroughs in the solvothermal assisted synthesis of luminescent metal-organic frameworks. Inorg. Chem. Commun. 2025, 178, 114631. [Google Scholar] [CrossRef]
- Ali, H.; Orooji, Y.; Al Alwan, B.; Jery, A.E.L.; Alhashmialameer, D.; Abu-Dief, A.M.; Guo, S.R.; Hayat, A.; Pei, L. Unlocking the potential of MOFs-based heterojunction Photocatalysts: Synthesis, modification, and performance enhancement. Renew. Sustain. Energy Rev. 2026, 226, 116359. [Google Scholar] [CrossRef]
- Song, N.; Geng, Z.; Zhu, Z.; Zhang, S.; Wang, Y.; Xu, Y.; Yang, L.; Zhang, D.; Zhou, Z. Post-synthesis of Tb@Zn-MOF composite as a multi-emission sensor for efficient detection of DCNA. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2025, 343, 126548. [Google Scholar] [CrossRef]
- Ding, J.; Wang, Y.; Qu, Y.; Chen, H.; Fu, H. Hydrothermal synthesis of ZnO/In2O3 nanocomposites derived from dual MOFs to enhance the gas sensing performance for ethanolamine. Sens. Actuators A Phys. 2026, 397, 117198. [Google Scholar] [CrossRef]
- Iniyan, S.; Ren, J.; Deshmukh, S.; Rajeswaran, K.; Jegan, G.; Hou, H.; Suryanarayanan, V.; Murugadoss, V.; Kathiresan, M.; Xu, B.B.; et al. An Overview of Metal-Organic Framework Based Electrocatalysts: Design and Synthesis for Electrochemical Hydrogen Evolution, Oxygen Evolution, and Carbon Dioxide Reduction Reactions. Chem. Rec. 2023, 23, e202300317. [Google Scholar] [CrossRef]
- Bedia, J.; Muelas-Ramos, V.; Peñas-Garzón, M.; Gómez-Avilés, A.; Rodríguez, J.; Belver, C. A Review on the Synthesis and Characterization of Metal Organic Frameworks for Photocatalytic Water Purification. Catalysts 2019, 9, 52. [Google Scholar] [CrossRef]
- Phan, P.T.; Hong, J.; Tran, N.; Le, T.H. The Properties of Microwave-Assisted Synthesis of Metal–Organic Frameworks and Their Applications. Nanomaterials 2023, 13, 352. [Google Scholar] [CrossRef]
- Sujithkrishnan, E.; Elumalai, P.; Akremi, A.; Alshahrani, T.; Rajaji, U.; Hung, K.-Y. Facile microwave-assisted solvothermal synthesis of Cu-MOF/Graphitic carbon nitridemodified electrode for the electrochemical detection of nitrofurantoin. Microchem. J. 2025, 215, 114102. [Google Scholar] [CrossRef]
- Priecel, P.; Lopez-Sanchez, J.A. Advantages and Limitations of Microwave Reactors: From Chemical Synthesis to the Catalytic Valorization of Biobased Chemicals. ACS Sustain. Chem. Eng. 2019, 7, 3–21. [Google Scholar] [CrossRef]
- Głowniak, S.; Szczęśniak, B.; Choma, J.; Jaroniec, M. Mechanochemical Synthesis of MOF-303 and Its CO2 Adsorption at Ambient Conditions. Molecules 2024, 29, 2698. [Google Scholar] [CrossRef] [PubMed]
- Kaur, G.; Anthwal, A.; Kandwal, P.; Sud, D. Mechanochemical synthesis and theoretical investigations of Fe (II) based MOF containing 4,4′-bipyridine with ordained intercalated p-aminobenzoic acid: Application as fluoroprobe for detection of carbonyl group. Inorganica Chim. Acta 2023, 545, 121248. [Google Scholar] [CrossRef]
- Ismail, K.M.; Hassan, S.S.; Medany, S.S.; Hefnawy, M.A. A facile sonochemical synthesis of the Zn-based metal–organic framework for electrochemical sensing of paracetamol. Mater. Adv. 2024, 5, 5870–5884. [Google Scholar] [CrossRef]
- Deffo, G.; Tamo, A.K.; Fotsop, C.G.; Tchoumi, H.H.B.; Talla, D.E.N.; Wabo, C.G.; Deussi, M.C.N.; Temgoua, R.C.T.; Doungmo, G.; Njanja, E.; et al. Metal-organic framework-based materials: From synthesis and characterization routes to electrochemical sensing applications. Coord. Chem. Rev. 2025, 536, 216680. [Google Scholar] [CrossRef]
- Liu, Y.; Wei, Y.; Liu, M.; Bai, Y.; Wang, X.; Shang, S.; Chen, J.; Liu, Y. Electrochemical Synthesis of Large Area Two-Dimensional Metal–Organic Framework Films on Copper Anodes. Angew. Chem. Int. Ed. 2021, 60, 2887–2891. [Google Scholar] [CrossRef]
- Hussain, I.; Mahmud, A.A.; Amna, R.; Pato, A.H.; Sajjad, U.; Ajmal, Z.; Zhang, K. Interface and surface engineering: The nexus of MXenes, MOFs, and AI in hybrid material design for energy storage/conversion. Mater. Today 2025, 89, 344–373. [Google Scholar] [CrossRef]
- Tong, H.; Zhu, A.; Chen, G.; Tong, Z.; Wang, J. Review on the synthesis, construction and photocatalytic applications of double-ligand MOFs. Chem. Eng. J. 2025, 508, 161068. [Google Scholar] [CrossRef]
- Wang, S.; Hu, W.; Ru, Y.; Shi, Y.; Guo, X.; Sun, Y.; Pang, H. Synthesis Strategies and Electrochemical Research Progress of Nano/Microscale Metal–Organic Frameworks. Small Sci. 2022, 2, 2200042. [Google Scholar] [CrossRef] [PubMed]
- Hasani, F.; Raoof, J.B.; Ojani, R.; Ghani, M. High-performance electrochemical sensor based on Pt-Ag@Cu-BDC MOF composite modified glassy carbon electrode for detection of imidacloprid in citrus juice and water samples. Heliyon 2025, 11, e42438. [Google Scholar] [CrossRef]
- Xie, L.S.; Skorupskii, G.; Dincă, M. Electrically Conductive Metal–Organic Frameworks. Chem. Rev. 2020, 120, 8536–8580. [Google Scholar] [CrossRef]
- Molina, M.A.; Rodríguez-Campa, J.; Flores-Borrell, R.; Blanco, R.M.; Sánchez-Sánchez, M. Sustainable Synthesis of Zeolitic Imidazolate Frameworks at Room Temperature in Water with Exact Zn/Linker Stoichiometry. Nanomaterials 2024, 14, 348. [Google Scholar] [CrossRef]
- Liu, J.; Shi, L.; Shi, Y.; Zeng, Q.; Yang, Y. Glucose sensing performance of bimetallic MOFs CoFe-ZIF/CC for enzyme-free saliva sensor applications. J. Alloys Compd. 2024, 999, 174733. [Google Scholar] [CrossRef]
- Dong, R.; Shen, Z.; Li, H.; Cheng, J.; Fu, Y. Research progress in fluorescent gas sensors based on MOFs. J. Mater. Chem. C Mater. 2024, 12, 12692–12707. [Google Scholar] [CrossRef]
- Hua, Y.; Kukkar, D.; Brown, R.J.C.; Kim, K.-H. Recent advances in the synthesis of and sensing applications for metal-organic framework-molecularly imprinted polymer (MOF-MIP) composites. Crit. Rev. Environ. Sci. Technol. 2023, 53, 258–289. [Google Scholar] [CrossRef]
- Shafique, B.; Fu, Z.; Wang, H.; Xie, J.; Ding, Z. Multifunctional metal-organic framework based nanozymes: From synthesis to fluorescent and colorimetric dual mode sensing in food safety. Food Control 2025, 178, 111497. [Google Scholar] [CrossRef]
- Huang, J.; Dong, H.; Geng, L.; Xu, R.; Liu, M.; Guo, Z.; Sun, J.; Sun, X.; Guo, Y. Rational design and controlled synthesis of metal-organic frameworks to meet the needs of electrochemical sensors with different sensing characteristics: An overview. Compos. B Eng. 2024, 281, 111536. [Google Scholar] [CrossRef]
- Wang, X.; Ma, T.; Ma, J.-G.; Cheng, P. Integration of devices based on metal–organic frameworks: A promising platform for chemical sensing. Coord. Chem. Rev. 2024, 518, 216067. [Google Scholar] [CrossRef]
- Kayani, K.F.; Shatery, O.B.A.; Mohammed, S.J.; Ahmed, H.R.; Hamarawf, R.F.; Mustafa, M.S. Synthesis and applications of luminescent metal organic frameworks (MOFs) for sensing dipicolinic acid in biological and water samples: A review. Nanoscale Adv. 2025, 7, 13–41. [Google Scholar] [CrossRef]
- Shu, Y.; Ye, Q.; Dai, T.; Guan, J.; Ji, Z.; Xu, Q.; Hu, X. Incorporation of perovskite nanocrystals into lanthanide metal-organic frameworks with enhanced stability for ratiometric and visual sensing of mercury in aqueous solution. J. Hazard. Mater. 2022, 430, 128360. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Liu, L.; Chen, Y.; Deng, Y.; Li, J.; Liu, X.; Zou, Y.; Wu, L.; Xie, W. Elaborately designed intelligent responsive sensing materials for development of flexible gas sensors. Prog. Mater. Sci. 2026, 156, 101565. [Google Scholar] [CrossRef]
- Javaheri, S.; Hallajzadeh, J.; Saadat, M.; Azadnia, A. Colorimetric detection and quantification of glutathione in human serum using the peroxidase-like activity of MOF-808FA frameworks. Curr. Pharm. Anal. 2025, 21, 375–383. [Google Scholar] [CrossRef]
- Miklaszewska, P.; Wagner-Wysiecka, E. The color code: MOFs unlocking new horizons in chemical sensing. Appl. Mater. Today 2025, 47, 102966. [Google Scholar] [CrossRef]
- Zhao, D.L.; Feng, F.; Shen, L.; Huang, Z.; Zhao, Q.; Lin, H.; Chung, T.S. Engineering metal–organic frameworks (MOFs) based thin-film nanocomposite (TFN) membranes for molecular separation. Chem. Eng. J. 2023, 454, 140447. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, J.; Xu, C.; Zhang, S.; Gao, X. Synthesis and characterization of Ce-MOF-based flexible electrode materials for uric acid sensing and oxygen evolution reaction. J. Rare Earths 2026, 44, 616–629. [Google Scholar] [CrossRef]
- Hussain Shah, J.; Sharif, S.; Şahin, O.; Shahbaz, M.; Azeem, W.; Ahmad, S. A dual-emitting Rhodamine B-encapsulated Zn-based MOF for the selective sensing of Chromium(VI). Spectrochim. Acta A Mol. Biomol. Spectrosc. 2024, 310, 123899. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Wang, Z.; Wang, H.; Wu, Y. Are MOFs ready for environmental applications: Assessing stability against natural stressors? Coord. Chem. Rev. 2025, 526, 216361. [Google Scholar] [CrossRef]
- Luo, W.; Zhang, Z.; Zhu, G.; Zhang, X.; Huang, G.; Zhou, T.; Lu, X. Precise pore regulation strategies for constructing high-performance Metal–Organic Framework (MOF) membranes for gas capture: Frontier advances. J. Environ. Chem. Eng. 2025, 13, 118229. [Google Scholar] [CrossRef]
- Feng, Y.; Chen, P.; Ye, R.; Lin, X.; Guo, H. Ultrasensitive and Wide-Range Electrochemical Sensor for Acetaminophen Based on a “Three-In-One” Engineered MOF (Fe)@Fe3S4/CNF Platform. Appl. Organomet. Chem. 2025, 39, e70388. [Google Scholar] [CrossRef]
- Jiang, P.; Li, L.; Zuo, Y.; Zhou, A. Thiol-functional MOF-integrated SPR nanosensor for ultrasensitive and selective Hg2+ detection. Microchem. J. 2025, 216, 114768. [Google Scholar] [CrossRef]
- Farzin, M.A.; Naghib, S.M.; Rabiee, N. Emerging metal-organic framework (MOF)-based biosensors with high potential for point-of-care determination of biomarkers: Mechanisms and applications. TrAC Trends Anal. Chem. 2025, 191, 118345. [Google Scholar] [CrossRef]
- Elashery, S.E.A.; Attia, N.F.; El Badry Mohamed, M. Exploitation of 2D Mn-MOF nanosheets for developing rapid, sensitive, and selective sensor for determination of Mn(II) ions in food and biological samples. Talanta 2025, 294, 128217. [Google Scholar] [CrossRef] [PubMed]
- Shang, M.; Hu, J.; Shi, M.; Fan, Y.; Zou, Y.; Wang, Y.; Guo, J.; Mao, C. Zr-MOF encapsulated-magnetic beads-based fast and efficient conjugation strategy for sensitive immunoassays and early diagnosis of rheumatoid arthritis. Microchem. J. 2025, 219, 115926. [Google Scholar] [CrossRef]
- Shen, Y.; Tissot, A.; Serre, C. Recent progress on MOF-based optical sensors for VOC sensing. Chem. Sci. 2022, 13, 13978–14007. [Google Scholar] [CrossRef]
- Li, J.; Qi, W.; Li, H.; Yang, L.; Li, Y.; Zhang, X.; Cao, X.; Fu; Wei, X.; Liu, Y.; et al. Advanced three-dimensional porous materials for uranium extraction: Strategies for enhancing adsorption performance and optimizing reusability, anti-fouling property. Coord. Chem. Rev. 2026, 547, 217134. [Google Scholar] [CrossRef]
- Magnuson, Z.; Wolfe, B.; Mayers, J.M.; Wojtas, L.; Zhang, W.; Ma, S.; Larsen, R.W. Novel zinc Metalloporphyrin based metal-organic framework for Quantitative & Reusable Sensing of nitrobenzene. Inorganica Chim. Acta 2025, 586, 122785. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, X.; Luo, Y.; Li, Q.; Qin, Y.; Wang, G.; Yang, S.; Liu, Z. An electrochemical sensor based on the composite UiO-66-NH2/rGO for trace detection of Pb(II) and Cu(II). Chem. Phys. Lett. 2023, 830, 140825. [Google Scholar] [CrossRef]
- Chen, D.-H.; Gliemann, H.; Wöll, C. Layer-by-layer assembly of metal-organic framework thin films: Fabrication and advanced applications. Chem. Phys. Rev. 2023, 4, 011305. [Google Scholar] [CrossRef]
- Mohammadzadeh Jahani, P.; Tajik, S.; Dourandish, Z. Electrochemical Sensor Based on Ce-MOF Modified Screen Printed Electrode for Metronidazole Determination. Chem. Methodol. 2024, 8, 123–132. [Google Scholar] [CrossRef]
- Harilal, C.C.; Uwaya, G.E.; Kanchi, S.; Bisetty, K. Engineered MOF-199 Modified Electrodes for Enhanced Electrochemical Immunosensing of Lactoferrin via Signal Amplification. J. Electrochem. Soc. 2025, 172, 126503. [Google Scholar] [CrossRef]
- Bao, W.; Aodeng, G.; Ga, L.; Ai, J. Aptamer-based electrochemical biosensors: Signal transduction mechanisms, application progress, and future trends. Sens. Actuators Rep. 2025, 10, 100366. [Google Scholar] [CrossRef]
- Salinas Domínguez, R.A.; Águila Rosas, J.; Martínez Tolibia, S.E.; Lima, E.; Dutt, A. Opportunities in functionalized metal-organic frameworks (MOFs) with open. metal sites for optical biosensor application. Adv. Colloid Interface Sci. 2025, 344, 103598. [Google Scholar] [CrossRef] [PubMed]
- Shubhangi; Nandi, I.; Rai, S.K.; Chandra, P. MOF-based nanocomposites as transduction matrices for optical and electrochemical sensing. Talanta 2024, 266, 125124. [Google Scholar] [CrossRef]
- Zhao, H.; Tan, X.; Chai, H.; Hu, L.; Li, H.; Qu, L.; Zhang, X.; Zhang, G. Recent advances in conductive MOF-based electrochemical sensors. Chin. Chem. Lett. 2025, 36, 110571. [Google Scholar] [CrossRef]
- Saha, R.; Gómez García, C.J. Extrinsically conducting MOFs: Guest-promoted enhancement of electrical conductivity, thin film fabrication and applications. Chem. Soc. Rev. 2024, 53, 9490–9559. [Google Scholar] [CrossRef]
- Diamond, B.G.; Payne, L.I.; Hendon, C.H. Ligand field tuning of d-orbital energies in metal-organic framework clusters. Commun. Chem. 2023, 6, 67. [Google Scholar] [CrossRef]
- Lei, X.; Deng, Z.; Zeng, Y.; Huang, S.; Yang, Y.; Wang, H.; Guo, L.; Li, L. A novel composite of conductive metal organic framework and molecularly imprinted poly (ionic liquid) for highly sensitive electrochemical detection of bisphenol A. Sens. Actuators B Chem. 2021, 339, 129885. [Google Scholar] [CrossRef]
- Li, P.; Cui, Z.; Wang, M.; Yang, J.; Hu, M.; Cheng, Q.; Wang, S. Electrochemical (Bio) Sensors Based on Metal–Organic Framework Composites. Electrochem. 2025, 6, 28. [Google Scholar] [CrossRef]
- Li, X.-M.; Liu, D.; Jia, J.; Chen, B.; Gao, J. Stimuli-responsive proton conduction in crystalline porous materials. Coord. Chem. Rev. 2025, 544, 216953. [Google Scholar] [CrossRef]
- Li, Z.; Zeng, W.; Li, Y. Recent Progress in MOF-Based Electrochemical Sensors for Non-Enzymatic Glucose Detection. Molecules 2023, 28, 4891. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Guo, J.; Sun, Q.; Wang, Y.; Du, M.; Wang, A.; Yu, X.; Ding, L. Enhanced electrochemical sensor based on Uio-66-NH2/carbon nanotubes hybrid for selective detection of ofloxacin. Mater. Today Chem. 2024, 42, 102441. [Google Scholar] [CrossRef]
- Finelli, V.; Rojas-Buzo, S.; Signorile, M.; Bonino, F.; Bordiga, S. Exploring Ce(IV)-MOFs redox behavior for catalysis by spectroscopies. Nano Mater. Sci. 2024, 7, 761–772. [Google Scholar] [CrossRef]
- Shaheen, I.; Chiu, W.-H.; Chen, S.-H.; Lee, K.-M. MOF- & COF-integrated composite separators/membranes: Innovations for sustainable and high-performance redox flow batteries. Sep. Purif. Technol. 2025, 376, 134157. [Google Scholar] [CrossRef]
- Yang, Y.; Tan, H.; Wang, Y.; Wang, X.; Liu, X.; Huo, C.; Zhu, C. A novel electrochemical sensor for sensitive detection of carbendazim based on zeolitic-imidazolate-framework-67/carbon nanohorns nanocomposite. Mater. Res. Bull. 2024, 179, 112975. [Google Scholar] [CrossRef]
- Wachholz Junior, D.; Hryniewicz, B.M.; Tatsuo Kubota, L. Advanced Hybrid materials in electrochemical sensors: Combining MOFs and conducting polymers for environmental monitoring. Chemosphere 2024, 352, 141479. [Google Scholar] [CrossRef]
- Ntuli, L.M.; Nyembwe, A.; Mwengula, G.; Munonde, T.; Mulopo, J. Recent advances in metal oxide, bimetallic nanocomposites, metal-organic frameworks (MOFs), and non-invasive technologies for biofluid-based glucose sensors: A comprehensive review. Int. J. Electrochem. Sci. 2025, 20, 101184. [Google Scholar] [CrossRef]
- Naghib, S.M.; Khorasani, M.A.; Fateminia, F.S.; Sharifianjazi, F.; Tavamaishvili, K. From design to detection: MOF-functional nanomaterial hybrids for ultrasensitive electrochemical monitoring pharmaceutical contaminants in the aquatic environment. Trends Environ. Anal. Chem. 2025, 48, e00284. [Google Scholar] [CrossRef]
- Gautam, S.; Rialach, S.; Paul, S.; Goyal, N. MOF/graphene oxide based composites in smart supercapacitors: A comprehensive review on the electrochemical evaluation and material development for advanced energy storage devices. RSC Adv. 2024, 14, 14311–14339. [Google Scholar] [CrossRef]
- Zhou, F.; Lim, H.N.; Ibrahim, I.; Liu, Y.; Qin, D. Au@MOF-199 functionalized graphene oxide nanocomposite for simultaneous electrochemical detection of dopamine and uric acid. Talanta 2026, 298, 128968. [Google Scholar] [CrossRef]
- Liang, Q.; Zhou, D.; Zhang, K.; Camarada, M.B.; Xiong, J.; Liao, X. A novel portable electrochemical sensor based on post-synthetic modified Cu-MOF for highly sensitive electrochemical detection of pefloxacin in food samples. Microchem. J. 2025, 209, 112895. [Google Scholar] [CrossRef]
- Dhayanithi, C.A.; Palpandi, K.; Raman, N.; Babu, S.G. Development of amine-based transition metal MOFs as efficient electrochemical sensors for the detection of chloramphenicol in food and pharmaceutical samples. Electrochim. Acta 2023, 470, 143358. [Google Scholar] [CrossRef]
- Chen, H.; Chen, L.; Zhou, Y. A flower-like tri-metal MOF coupled with carbon nanomaterials for electrochemical detection of antibiotic Tinidazole. Microchem. J. 2025, 218, 115634. [Google Scholar] [CrossRef]
- Dhayanithi, C.A.; Maheshwaran, S.; Liu, T.-Y.; Ganesh Babu, S. Strategic interface engineering of nickel rods/Ni-MOF for the electrochemical detection of gatifloxacin in environmental samples. J. Environ. Chem. Eng. 2025, 13, 118189. [Google Scholar] [CrossRef]
- Nikfarjam, F.; Dehghan, G.; Khataee, S.; Rashtbari, S.; Amini, M. A dual-mode Co-doped Cu-MOF sensor for simultaneous colorimetric and electrochemical detection of acetaminophen and tramadol. Microchem. J. 2025, 215, 114499. [Google Scholar] [CrossRef]
- Zhang, Z.; Shi, Y.; Li, Z.; Han, J.; Chen, Z.; Ma, F.; Qi, Y.; Liu, Y. Bimetallic and dual-ligands MOFs modified electrochemical sensor for fast and highly sensitive miR-21 detection. Electrochim. Acta 2025, 540, 147229. [Google Scholar] [CrossRef]
- Jia, X.; Zhang, N.; Xu, Z.; Wang, H.; Zhang, W.; Luo, C. Fabrication of high-performance electrochemical sensors using MXene-SWCNHs-Cu@β-CD-MOF for chlorothalonil pesticide detection. Microchem. J. 2025, 219, 115983. [Google Scholar] [CrossRef]
- Dong, J.; Wang, G.; Geng, L.; Wang, H.; Li, C.; Xu, R.; Liu, J.; Liu, J.; Darwish, I.A.; Sun, X.; et al. Electrochemical aptamer sensor based on bimetallic CuZr-MOF and AuNPs for ultrasensitive detection of organophosphorus pesticides in vegetables. J. Food Compos. Anal. 2025, 144, 107761. [Google Scholar] [CrossRef]
- Habr, J.J.; Alem, F.E.; Kanagavalli, P.; Gandra, U.R.; Varghese, A.M.; Saraierh, H.; Yildiz, I.; Assen, A.H.; Price, G.; Eissa, S.; et al. Electrochemical sensor for the detection of imidacloprid using novel Cu-tetrazole MOF. Sens. Actuators Rep. 2025, 10, 100377. [Google Scholar] [CrossRef]
- Zhang, Y.; Jin, S.; Liu, R.; Liu, Z.; Gong, L.; Zhang, L.; Zhao, T.; Chen, S.; Niu, L.; Fa, H.; et al. A new portable electrochemical detection sensor based on molecularly imprinted polymer-modified MOF-808/AB for the highly sensitive and selective determination of dimethoate. Microchem. J. 2024, 206, 111436. [Google Scholar] [CrossRef]
- Khosropour, H.; Keramat, M.; Primpray, V.; Karuwan, C.; Tasca, F.; Laiwattanapaisal, W. An electrochemical aptamer-based biosensor for rapid and ultrasensitive detection of carbaryl by red blood cell-like MOFs. Alex. Eng. J. 2025, 124, 1–11. [Google Scholar] [CrossRef]
- Javed, S.; Sherin, L.; Riaz, S.; Ashfaq, A. A novel (Bi S)n MOF@MXene hybrid-integrated electrochemical sensor for simultaneous and sensitive detection of multiple trace-level heavy metal ions in drinking water and food samples. Microchem. J. 2025, 219, 116038. [Google Scholar] [CrossRef]
- Shao, Z.; Di, K.; Ding, L.; You, F.; Fan, C.; Wang, K. Amino-enriched Zn-MOFs with self-reduction for energy-free simultaneous removal and electrochemical detection of heavy metal ions in the aquatic environment. Anal. Chim. Acta 2025, 1333, 343408. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Dong, F.; Rodas-Gonzalez, A.; Wang, G.; Yang, L.; Chen, S.; Zheng, H.B.; Wang, S. Simultaneous detection of heavy metal ions in food samples using a hypersensitive electrochemical sensor based on APTES-incubated MXene-NH2@CeFe-MOF-NH2. Food Chem. 2025, 475, 143362. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, Y.; Ma, Y.; Luo, H.; Hou, J.; Hou, C.; Huo, D. Ultra-sensitive electrochemical sensors through self-assembled MOF composites for the simultaneous detection of multiple heavy metal ions in food samples. Anal. Chim. Acta 2024, 1289, 342155. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Chen, Y.; Noureen, B.; Ma, Y.; Zheng, A.; Zhou, L.; Liu, M.; Zhao, Y.; Du, L.; Zhang, W.; et al. An electrochemical sensor using nickel-based metal-organic framework towards highly-sensitive detection of multiple heavy metal ions. Sens. Actuators B Chem. 2025, 444, 138352. [Google Scholar] [CrossRef]
- Sharma, P.; Siddiqui, K.A. Metal–organic frameworks as fluorescent and colorimetric sensors for antibiotic tracing. Discov. Chem. 2025, 2, 199. [Google Scholar] [CrossRef]
- Pal, T.K. Metal–organic framework (MOF)-based fluorescence “turn-on” sensors. Mater. Chem. Front. 2023, 7, 405–441. [Google Scholar] [CrossRef]
- Taghavi-Kahagh, A.; Behboodi-Sadabad, F.; Salami-Kalajahi, M. Multi-emission metal–organic framework (MOF)-based composites: A comprehensive review of synthesis strategies and luminescence applications. J. Ind. Eng. Chem. 2026, 154, 101–135. [Google Scholar] [CrossRef]
- Lv, R.; Chen, Z.; Fu, X.; Yang, B.; Li, H.; Su, J.; Gu, W.; Liu, X. A highly selective and fast-response fluorescent probe based on Cd-MOF for the visual detection of Al3+ ion and quantitative detection of Fe3+ ion. J. Solid. State Chem. 2018, 259, 67–72. [Google Scholar] [CrossRef]
- Dalfen, I.; Borisov, S.M. Porous matrix materials in optical sensing of gaseous oxygen. Anal. Bioanal. Chem. 2022, 414, 4311–4330. [Google Scholar] [CrossRef]
- Liu, X.; Chen, H.; Hu, X.; Liu, J.; Zhong, Y.; Li, P.; Zhu, X.; Zhang, Y.; Liu, M.; Yao, S. Traffic lights inspired colorimetric sensing platform for cadmium/lead ions: PO43− induced structural change of porphyrin metal-organic framework. Sens. Actuators B Chem. 2024, 420, 136455. [Google Scholar] [CrossRef]
- Peng, B.; Li, Y.; Qin, J.; Qiao, Z.; Li, Y.; Yang, J.; Zhou, P.; Zhao, X.; Kuang, Y.; Wu, K.; et al. Structure-engineered colorimetric sensors for volatile amine detection: Advances in freshness monitoring of meat and aquatic products. TrAC Trends Anal. Chem. 2026, 196, 118661. [Google Scholar] [CrossRef]
- Scurti, S.; Racchi, O.; Spinoso, V.; Bussolari, A.; D’Altri, G.; Gualandi, I.; Di Maiolo, F.; Sissa, C.; Caretti, D. Symmetric azobenzene-substituted diketopyrrolopyrroles dyes as acid-base switchable molecular-probe for colorimetric paper-based sensors. Dye. Pigment. 2026, 245, 113288. [Google Scholar] [CrossRef]
- Motora, K.G.; Wu, C.-M.; Li, K.-C. Effective Azo-functionalized metal-organic framework colorimetric and ratiometric sensor for detection of heavy metals in aqueous solution by the naked eye. J. Environ. Chem. Eng. 2023, 11, 110144. [Google Scholar] [CrossRef]
- Cao, L.; Chen, W.; Kang, W.; Lei, C.; Nie, Z. Engineering stimuli-responsive CRISPR-Cas systems for versatile biosensing. Anal. Bioanal. Chem. 2025, 417, 1699–1711. [Google Scholar] [CrossRef]
- Xu, X.; Ma, M.; Sun, T.; Zhao, X.; Zhang, L. Luminescent Guests Encapsulated in Metal–Organic Frameworks for Portable Fluorescence Sensor and Visual Detection Applications: A Review. Biosensors 2023, 13, 435. [Google Scholar] [CrossRef]
- Sharma, A.; Malik, P.; Kaur, G.; Sharma, R.K.; Sud, D. Recent developments in luminescence MOFs based sensing platform for lead and cadmium detection—An update. Microchem. J. 2025, 215, 114299. [Google Scholar] [CrossRef]
- Gao, Y.; Zhu, Y.; Wang, Y.; Bi, J. Water-Stable Ln-MOF as a multi-emitting luminescent sensor for the detection of metal ions and pharmaceuticals. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2024, 323, 124915. [Google Scholar] [CrossRef]
- Dong, S.-W.; Zheng, L.-N.; Liu, S.; Sun, Y.-Z.; Xue, N.; Zhao, J.; Ding, T. Novel highly stable zinc(II)-MOF based on metal-/carbonate-/5-ATZ- orientation for CO2/CH4 and C2H2/CO2 gas adsorption separation and fluorescence detection of Fe3+/Cr6+. J. Mol. Struct. 2026, 1350, 144036. [Google Scholar] [CrossRef]
- Xuan, X.; Wang, M.; Manickam, S.; Boczkaj, G.; Yoon, J.Y.; Sun, X. Metal-Organic Frameworks-Based Sensors for the Detection of Toxins in Food: A Critical Mini-Review on the Applications and Mechanisms. Front. Bioeng. Biotechnol. 2022, 10, 906374. [Google Scholar] [CrossRef] [PubMed]
- Oladipo, A.A.; Derakhshan Oskouei, S.; Gazi, M. Metal-organic framework-based nanomaterials as opto-electrochemical sensors for the detection of antibiotics and hormones: A review. Beilstein J. Nanotechnol. 2023, 14, 631–673. [Google Scholar] [CrossRef]
- Elsherbiny, A.S.; Rady, A.; Abdelhameed, R.M.; Gemeay, A.H. Efficiency and selectivity of cost-effective Zn-MOF for dye removal, kinetic and thermodynamic approach. Environ. Sci. Pollut. Res. 2023, 30, 106860–106875. [Google Scholar] [CrossRef] [PubMed]
- Guselnikova, O.; Postnikov, P.; Elashnikov, R.; Miliutina, E.; Svorcik, V.; Lyutakov, O. Metal-organic framework (MOF-5) coated SERS active gold gratings: A platform for the selective detection of organic contaminants in soil. Anal. Chim. Acta 2019, 1068, 70–79. [Google Scholar] [CrossRef]
- Yue, X.; Zhou, Z.; Li, M.; Jie, M.; Xu, B.; Bai, Y. Inner-filter effect induced fluorescent sensor based on fusiform Al-MOF nanosheets for sensitive and visual detection of nitrofuran in milk. Food Chem. 2022, 367, 130763. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Liu, Y.; Lin, H.; Xu, N.; Liu, G.; Wang, X.; Chang, Z.; Li, J. A novel cadmium metal–organic framework-based multiresponsive fluorescent sensor demonstrating outstanding sensitivities and selectivities for detecting NB, Fe3+ ions and Cr2O72− anions. CrystEngComm 2020, 22, 6626–6631. [Google Scholar] [CrossRef]
- Liu, W.; Cui, H.; Zhou, J.; Su, Z.; Zhang, Y.; Chen, X.; Yue, E. Synthesis of a Cd-MOF Fluorescence Sensor and Its Detection of Fe3+, Fluazinam, TNP, and Sulfasalazine Enteric-Coated Tablets in Aqueous Solution. ACS Omega 2023, 8, 24635–24643. [Google Scholar] [CrossRef]
- Zhu, C.; Hou, J.; Wang, X.; Wang, S.; Xu, H.; Hu, J.; Jing, L.; Wang, S. Optimizing ligand-to-metal charge transfer in metal–organic frameworks to enhance photocatalytic performance. Chem. Eng. J. 2024, 499, 156527. [Google Scholar] [CrossRef]
- Wiwasuku, T.; Chuaephon, A.; Puangmali, T.; Boonmak, J.; Ittisanronnachai, S.; Promarak, V.; Youngme, S. Multifunctional fluorescent Eu-MOF probe for tetracycline antibiotics and dihydrogen phosphate sensing and visualizing latent fingerprints. RSC Adv. 2023, 13, 10384–10396. [Google Scholar] [CrossRef]
- Wang, N.; Li, S.; Li, Z.; Gong, Y.; Li, X. A Zn(II)–Metal–Organic Framework Based on 4-(4-Carboxy phenoxy) Phthalate Acid as Luminescent Sensor for Detection of Acetone and Tetracycline. Molecules 2023, 28, 999. [Google Scholar] [CrossRef]
- Kulandaivel, S.; Lo, W.-C.; Lin, C.-H.; Yeh, Y.-C. Cu-PyC MOF with oxidoreductase-like catalytic activity boosting colorimetric detection of Cr(VI) on paper. Anal. Chim. Acta 2022, 1227, 340335. [Google Scholar] [CrossRef]
- Cai, Y.; Dong, T.; Bian, Z.; Liu, H.; Liu, X.; Liu, A. Metal-organic frameworks based fluorescent sensing: Mechanisms and detection applications. Coord. Chem. Rev. 2025, 529, 216470. [Google Scholar] [CrossRef]
- Rasheed, T. Water stable MOFs as emerging class of porous materials for potential environmental applications. Chemosphere 2023, 313, 137607. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Zhou, Z.; Zhao, Z.; Liu, J.; Sun, Y.; Zhang, Y.; Gao, E.; Zhu, M.; Wu, S. Synthesis of multi-emission MOF composites for multi-dimensional sensing application. Microchem. J. 2024, 196, 109660. [Google Scholar] [CrossRef]
- Pramanik, B.; Sahoo, R.; Das, M.C. pH-stable MOFs: Design principles and applications. Coord. Chem. Rev. 2023, 493, 215301. [Google Scholar] [CrossRef]
- Ali, S.; Zuhra, Z.; Ali, S.; Han, Q.; Ahmad, M.; Wang, Z. Ultra-deep removal of Pb by functionality tuned UiO-66 framework: A combined experimental, theoretical and HSAB approach. Chemosphere 2021, 284, 131305. [Google Scholar] [CrossRef]
- Sonowal, S.; Prasad, R. Exploring the recent advancement of MOFs (metal-organic frameworks) in agriculture and food technology. Next Mater. 2025, 9, 101016. [Google Scholar] [CrossRef]
- Shah, S.; Hussain, S.; Din, S.U.; Ibrahim, T.K.; Zhao, X.; Manavalan, R.K.; Pan, X.; Liu, G.; Qiao, G. H2S gas sensors based on NiO@SnO2 heterojunction with SnO2 nanoparticles decorated MOF derived NiO hollowspheres. J. Environ. Chem. Eng. 2025, 13, 118997. [Google Scholar] [CrossRef]
- Ebadi Amooghin, A.; Sanaeepur, H.; Ghomi, M.; Luque, R.; Garcia, H.; Chen, B. Flexible–robust MOFs/HOFs for challenging gas separations. Coord. Chem. Rev. 2024, 505, 215660. [Google Scholar] [CrossRef]
- Zhang, Q.; Yang, H.; Zhou, T.; Chen, X.; Li, W.; Pang, H. Metal–Organic Frameworks and Their Composites for Environmental Applications. Adv. Sci. 2022, 9, e2204141. [Google Scholar] [CrossRef]
- Wang, F.; Wang, C.-C.; Yi, S. Rational design and synthesis of metal–organic frameworks derivatives: A perspective on emerging techniques. Chem. Eng. J. 2024, 495, 153398. [Google Scholar] [CrossRef]
- Borah, P.; Borah, P.; Ahmaruzzaman, M. MOF@metal-sulfide: Revolutionizing hybrid materials from structural engineering to advanced energy and environmental applications. Coord. Chem. Rev. 2026, 556, 217688. [Google Scholar] [CrossRef]
- Ouyang, Q.; Zhang, Y.; Xu, Z.; Xu, L.; Chang, B.; Lin, H.; Ma, Y.; She, Y. AI-integrated dye@MOF@MIPs odor sensors for monitoring of food freshness. Biosens. Bioelectron. 2026, 296, 118364. [Google Scholar] [CrossRef] [PubMed]
- Ji, Z.; Wang, M.; Guan, H.; Huang, H.; Guan, X.; Dong, Y. Artificial intelligence-powered MOF-incorporated thin-film nanocomposite desalination membranes for sustainable water purification. Desalination 2026, 621, 119702. [Google Scholar] [CrossRef]
- Zhang, M.; He, Y.; Tang, Z.; Qin, C.; Zhang, L.; Liu, H. Bibliometric analysis of MOF-based fluorescence detection in food safety: Development, multi-strategy optimization, and future AI integration. Food Chem. X 2025, 32, 103331. [Google Scholar] [CrossRef]
- Gaidimas, M.A.; Bai, J.; Kang, Y.; Kirlikovali, K.O.; Bernales, V.; Aspuru-Guzik, A.; Farha, O.K. Reimagining metal-organic framework discovery: Integrating experiment, computation, and artificial intelligence. Chem 2026, 102921. [Google Scholar] [CrossRef]
- Ji, Y.; Xue, L.; Luan, G.; Li, C. One-pot synthesized multifunctional Zn-MOF/HOF heterostructure sensor array assisted by machine learning for efficient capture, target discrimination and optosmart sensing of doxycycline analogs. J. Hazard. Mater. 2025, 494, 138512. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Chang, S.-J.; Chen, C.-J.; Liu, J.-T. Metal-organic frameworks for electrochemical glucose sensors: Progress and challenges. Coord. Chem. Rev. 2025, 543, 216907. [Google Scholar] [CrossRef]
- Naga Prasuna, D.; Rao, A.V.; Anjani Devi, K.; Erfan, S.K.; Reddy, P.V.S.S.S.; Babu, B.V. A review on versatile roles of MOFs: Bridging biological applications and environmental technologies. Results Chem. 2025, 17, 102567. [Google Scholar] [CrossRef]
- Liu, Z.; Li, R.; Qi, Q.; Chen, X.; Yu, J.; Wang, R.; Chen, J. Research progress and prospects of MOF-based and -derived cathode catalysts in microbial electrochemical technologies: Mechanisms, performance and applications in wastewater treatment. J. Environ. Chem. Eng. 2025, 13, 118567. [Google Scholar] [CrossRef]
- Chakraborty, S.; Björk, J.; Dahlqvist, M.; Rosen, J.; Heintz, F. A survey of AI-supported materials informatics. Comput. Sci. Rev. 2026, 59, 100845. [Google Scholar] [CrossRef]
- Zivic, F.; Malisic, A.K.; Grujovic, N.; Stojanovic, B.; Ivanovic, M. Materials informatics: A review of AI and machine learning tools, platforms, data repositories, and applications to architectured porous materials. Mater. Today Commun. 2025, 48, 113525. [Google Scholar] [CrossRef]
- Xie, E.; Wang, X.; Siepmann, J.I.; Chen, H.; Snurr, R.Q. Generative AI for design of nanoporous materials: Review and future prospects. Digit. Discov. 2025, 4, 2336–2363. [Google Scholar] [CrossRef]
- Bae, S.; Jeon, M.; Moon, H.R. Text mining in MOF research: From manual curation to large language model-based automation. Chem. Commun. 2025, 61, 11083–11094. [Google Scholar] [CrossRef] [PubMed]
- Ti, H.; Yang, L.; Yan, W.; Chen, Y.; Yang, F.; Yue, Y.; Sun, Y.; Li, X.; Niu, Y.; Li, S. Structure-based machine learning with standardized feature selection for screening MOFs with high ammonia capture capacity. Process Saf. Environ. Prot. 2025, 203, 107955. [Google Scholar] [CrossRef]
- Arjmandi, M.; Aytaç, E.; Khayet, M.; Hilal, N. Next-generation MOFs for atmospheric water harvesting: The role of machine learning techniques. Coord. Chem. Rev. 2026, 548, 217211. [Google Scholar] [CrossRef]
- Abed, H.; Sabouni, R.; Ghommem, M. MOF-based spectrophotometric sensors for cholesterol detection: Current trends and challenges. RSC Adv. 2024, 14, 39472–39497. [Google Scholar] [CrossRef] [PubMed]


| MOF | Synthesis | Analyte (Signal/Method) | Sample | Limit of Detection | Reference |
|---|---|---|---|---|---|
| Fe-MOF | Solvothermal | CAP (Current/DPV) | Milk and eyedrops | 0.011 µmol L−1 | [102] |
| f-C@FeCoNi-MOF | Solvothermal | TN (Current/DPV) | Tablet and Drugs | 7.4 × 10−10 mol L−1 | [103] |
| NiR/Ni-MOF | Solvothermal | GAT (Current/DPV) | River water, human sérum and urine | 3.8 nmol L−1 | [104] |
| Co/Cu-MOF | Solvothermal | Acp and Tra (Current/DPV) | Human blood serum | 3.8 and 3 nmol L−1 | [105] |
| CoNi@MOF | Solvothermal | MiR-21 (Current/DPV) | Human serum | 10 f mol L−1 | [106] |
| β-CD-MOF | Sonochemical | CHL (Current density/CV) | Tomato | 0.22 μg L−1 | [107] |
| Zr-MOF and CuZr-MOF | Solvothermal | OPs (Current/DPV) | Spinach and carrot | 4.6 × 10 −14 mol L−1 | [108] |
| KU-1 | Solvothermal | IMD (Current/DPV) | Rice and tomato | 0.089 μ mol L−1 | [109] |
| MOF-808 | Solvothermal | DIM (Current/DPV) | Orange and Cucumber | 43.05 pmol L−1 | [110] |
| rbMOFs | Reflux-isted solvothermal | CBA (Current/DPV) | Apple and potato | 0.4 pmol L−1 | [111] |
| (Bi-S)n MOF | Microwave-assisted | Pb2+, Cu2+ and Hg2+ (Current/SWV) | Milk and rice water | 5.4036–6.4295 n mol L−1 | [112] |
| Zn-MOF | Solution | Cu2+, Hg2+ and Pb2+ (Current/SWASV) | Tap and lake water | 0.17–0.25 μg L−1 | [113] |
| CeFe-MOF | Hydrothermal | Cd2+, Pb2+ and Hg2+ (Current/DPV) | Fish, milk, rice and corn | 0.33–0.95 nmol L−1 | [114] |
| UiO-67 and Al-MOF | Solvothermal | Cd2+, Pb2+, Cu2+ and Hg2+ (Current/SWV) | Milk, honey and black tea | 0.018–0.041 pmol L−1 | [115] |
| Ni-MOF | Solution | Pb2+ and Cu2+ (Current/ SWASV) | Tap water | 1.21 and 0.77 μg L−1 | [116] |
| MOF | Synthesis | Contaminant | Detection Type | Sample | LOD | Reference |
|---|---|---|---|---|---|---|
| Al-MOF | Hydrothermal | Nitrofurazone, nitrofurantoin, furazolidone | Fluorescence | Milk | 0.53–0.838 μmol L−1 | [135] |
| Cd-MOF | Solvothermal | Fe3+, Cr2O72−, NB | Fluorescence | Aqueous solutions | 4.2 × 10−6–5.3 × 10−5 M | [136] |
| Cd-MOF | Solvothermal | Fe3+, fluazinam, TNP | Fluorescence | Aqueous solutions | 10−5–10−6 mol L−1 | [137] |
| NH2-MIL-88B(Fe) | Solvothermal | BTEX vapors (benzene, toluene, ethylbenzene, xylene) | Colorimetry | Atmospheric | 0.22–3.70 g·m−3 | [138] |
| Eu-MOF | Hydrothermal | Tetracyclines (OTC, TC, DOX) and H2PO4− | Luminescent | Aqueous solutions and food | 78 nmol L−1–0.70 µmol L−1 | [139] |
| Zn-MOF | Hydrothermal | TC and VOC | Luminescent | Water, urine and aquaculture wastewater | 3.34 µmol L−1. | [140] |
| Cu–PyC MOF | Solvothermal | Cr (VI) | Colorimetric | Environmental samples | 0.051 µmol L−1 | [141] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Soares Ribeiro, I.; Aquino, W.C.P.; Alfredo, L.H.M.; de Jesus, J.R. Metal–Organic Frameworks as Multifunctional Platforms for Chemical Sensors: Advances in Electrochemical and Optical Detection of Emerging Contaminants. Processes 2026, 14, 886. https://doi.org/10.3390/pr14060886
Soares Ribeiro I, Aquino WCP, Alfredo LHM, de Jesus JR. Metal–Organic Frameworks as Multifunctional Platforms for Chemical Sensors: Advances in Electrochemical and Optical Detection of Emerging Contaminants. Processes. 2026; 14(6):886. https://doi.org/10.3390/pr14060886
Chicago/Turabian StyleSoares Ribeiro, Iare, Wesley C. P. Aquino, Lucas H. M. Alfredo, and Jemmyson R. de Jesus. 2026. "Metal–Organic Frameworks as Multifunctional Platforms for Chemical Sensors: Advances in Electrochemical and Optical Detection of Emerging Contaminants" Processes 14, no. 6: 886. https://doi.org/10.3390/pr14060886
APA StyleSoares Ribeiro, I., Aquino, W. C. P., Alfredo, L. H. M., & de Jesus, J. R. (2026). Metal–Organic Frameworks as Multifunctional Platforms for Chemical Sensors: Advances in Electrochemical and Optical Detection of Emerging Contaminants. Processes, 14(6), 886. https://doi.org/10.3390/pr14060886






