Fungal Solid-State Fermentation as a Strategy to Release Polyphenols from Orange Peel Waste
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Microorganism and Raw Material
2.3. Evaluation of the Factors That Influence the Fermentation Process
2.4. Recovery of Fermented Extracts
2.5. Determination of Hydrolyzable Tannins by Microplate
2.6. Determination of Condensed Tannins by Microplate
2.7. Analysis of Liquid Chromatography of High Resolution by Electrospray Mass Spectrometry (HPLC-ESI-MS)
2.8. Statistical Analysis
3. Results and Discussion
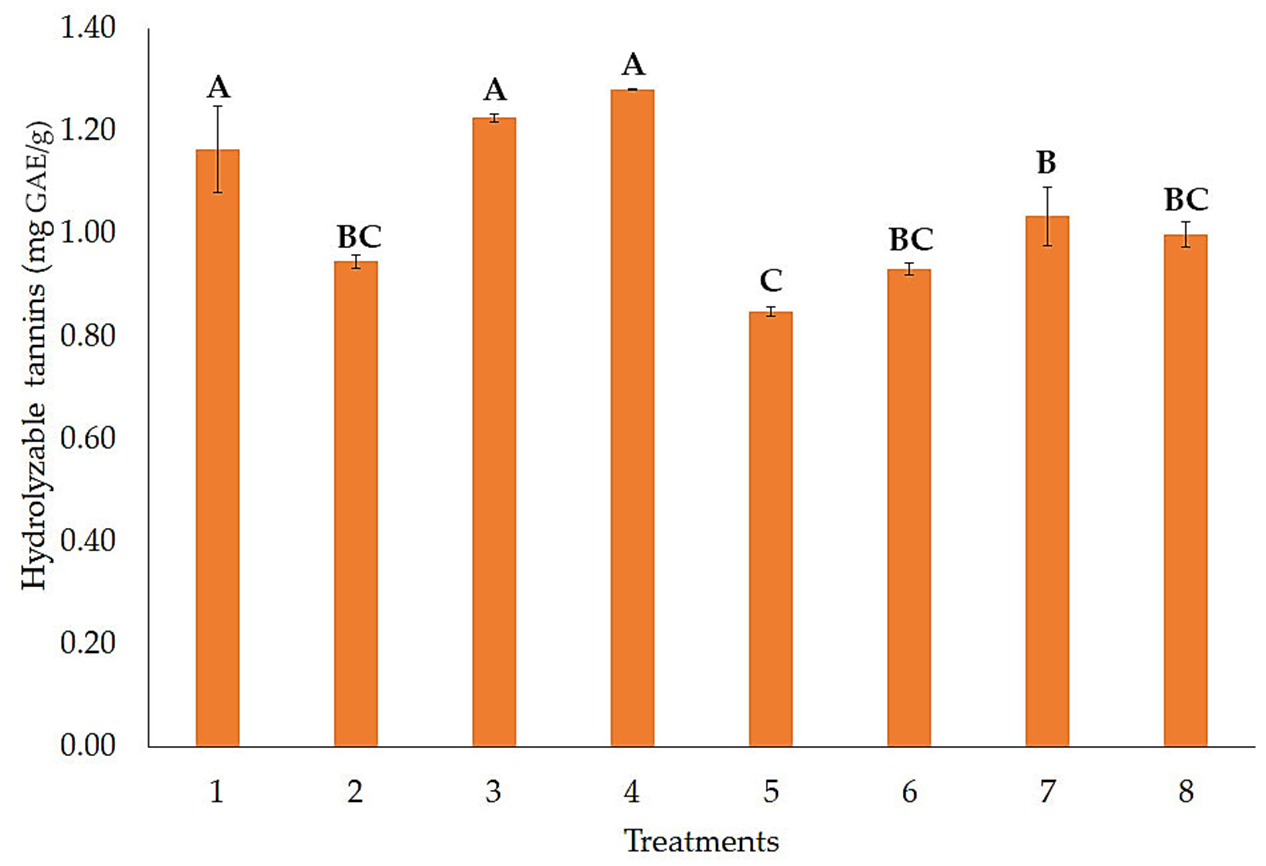
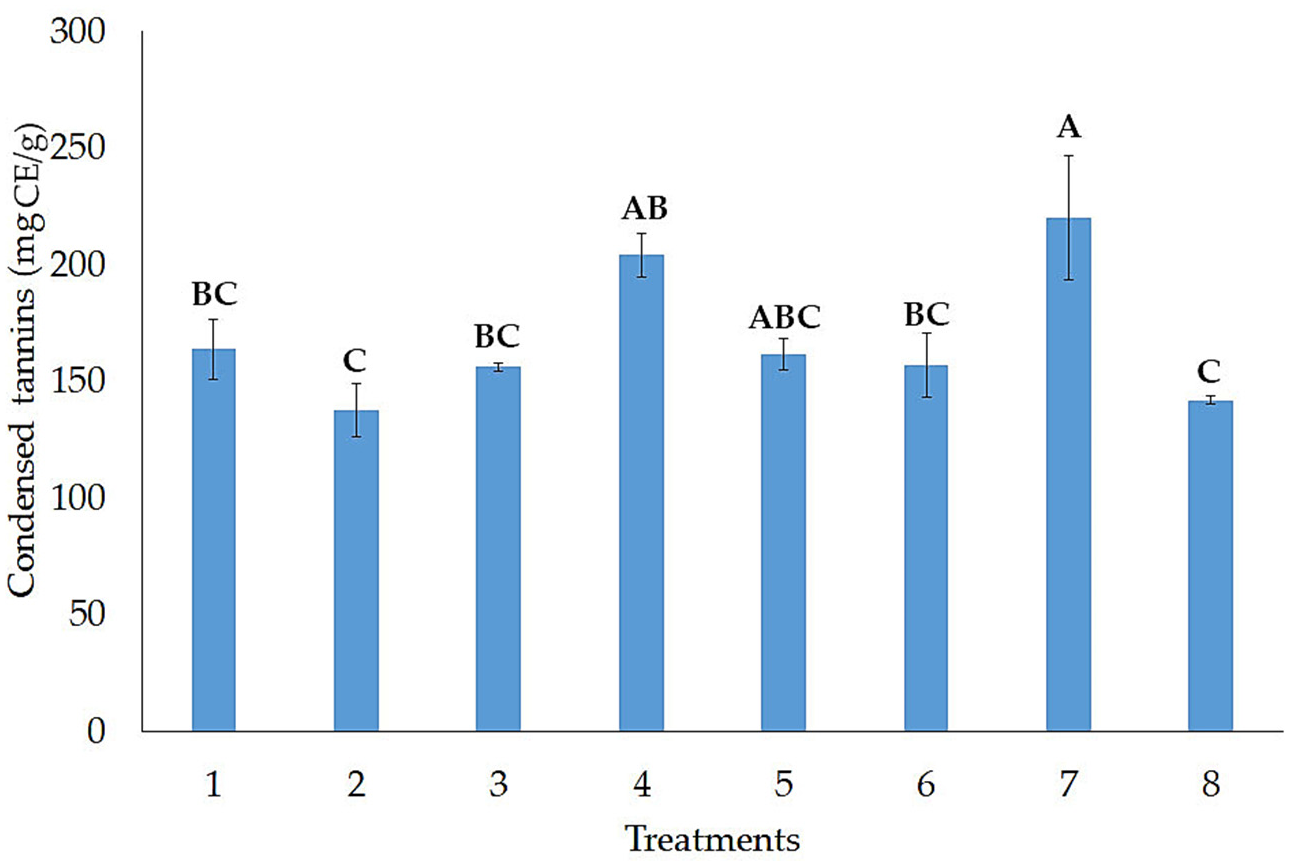
3.1. Release of Hydrolyzable and Condensed Tannins
3.2. Effect of Independent Factors on the Release of Condensed Tannins
3.3. Identification of Polyphenolic Compounds from Solid-State Fermentation by HPLC-ESI-MS
| Compound | Structure 1 | [M-H]− | MS2 | Treatment |
|---|---|---|---|---|
| Scopoletin | 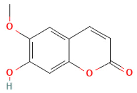 | 190.9 | 146, 111, 102 | 1 to 8 |
| Ferulic acid |  | 192.9 | 149, 134, 121 | 1 to 8 |
| p-Cumaroyl glycolic acid |  | 221.9 | 163 | 2 to 8 |
| (−)-Epicatechin |  | 288.9 | 273, 139 | 1 to 8 |
| Cirsimaritin |  | 313.0 | 299, 285 | 1 to 8 |
| 5-O-Galloylquinic acid |  | 342.9 | 191 | 1 to 8 |
| Ferulic acid 4-O-glucoside | 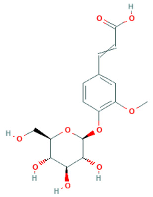 | 354.9 | 193 | 1 to 8 |
| 3-Feruloylquinic acid |  | 366.9 | 193, 191 | 1 to 8 |
| (−)-Epicatechin 3-O-gallate |  | 442.9 | 289, 169 | 7 |
| Vitisin A |  | 560.9 | 399 | 3 |
| Apigenin 6-C-glucoside 8-C-arabinoside | 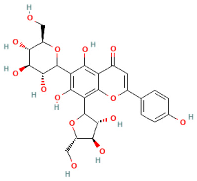 | 563.0 | 545, 503, 473, 455, 443 | 2 and 3 |
| Apigenin 6,8-di-C-glucoside |  | 593.0 | 575, 503, 473, 455, 437 | 2 to 8 |
| Delphinidin 3-O-sambubioside |  | 596.0 | 355, 327 | 1 |
| Quercetin 3-O-xylosyl-glucuronide |  | 609.0 | 479, 303, 285, 239 | 1 to 8 |
| Isorhamnetin 3-O-glucoside 7-O-rhamnoside |  | 623.0 | 477, 461, 314 | 1 to 8 |
| Petunidin 3-O-(6″-p-coumaroyl-glucoside) |  | 624.0 | 330, 478 | 4 |
| (−)-Epicatechin-(2a-7) (4a-8)-epicatechin 3-O-Galactoside | 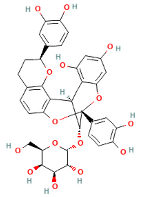 | 706.9 | 353, 289 | 3 and 7 |
| 1,2,2′-Triferuloylgentiobiose |  | 868.8 | 717, 499 | 2, 6, 7, and 8 |
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, N.; Li, X.; Zhao, P.; Zhang, X.; Qiao, O.; Huang, L.; Guo, L.; Gao, W. A Review of Chemical Constituents and Health-Promoting Effects of Citrus Peels. Food Chem. 2021, 365, 130586. [Google Scholar] [CrossRef]
- SIAP. Anuario Estadístico de la Producción Agrícula. Available online: https://nube.agricultura.gob.mx/cierre_agricola/ (accessed on 15 October 2025).
- Li, S.; Lo, C.Y.; Ho, C.T. Hydroxylated Polymethoxy-Flavones and Methylated Flavonoids in Sweet Orange (Citrus sinensis) Peel. J. Agric. Food Chem. 2006, 54, 4176–4185. [Google Scholar] [CrossRef]
- Rezzadori, K.; Benedetti, S.; Amante, E.R. Proposals for the Residues Recovery: Orange Waste as Raw Material for New Products. Food Bioprod. Process. 2012, 90, 606–614. [Google Scholar] [CrossRef]
- Shah, M.A.; Tariq, S.; Abuzar, S.M.; Ilyas, K.; Qadees, I.; Alsharif, I.; Anam, K.; Almutari, R.T.; Al-Regaiey, K.A.; Babalghith, A.O.; et al. Peel Waste of Citrus Fruits: A Valuable and Renewable Source of Polyphenols for the Treatment of Diabesity. Curr. Res. Biotechnol. 2024, 7, 100204. [Google Scholar] [CrossRef]
- Wang, L.; Wang, J.; Lianying, F.; Zheng, Z.; Zhi, D.; Wang, S.; Li, S.; Ho, C.; Zhao, H. Anticancer Activities of Citrus Peel Polymethoxyflavones Related to Angiogenesis and Others. BioMed Res. Int. 2014, 2014, 453972. [Google Scholar] [CrossRef]
- Xi, W.; Zhang, Y.; Sun, Y.; Shen, Y.; Ye, X.; Zhou, Z. Phenolic Composition of Chinese Wild Mandarin (Citrus reticulata Blanco) Pulps and Their Antioxidant Properties. Ind. Crops Prod. 2014, 52, 466–474. [Google Scholar] [CrossRef]
- Cavia-Saiz, M.; Busto, M.D.; Pilar-Izquierdo, M.C.; Ortega, N.; Pérez-Mateos, M.; Muñiz, P. Antioxidant Properties, Radical Scavenging Activity and Biomolecule Protection Capacity of Flavonoid Naringenin and Its Glycoside Naringin: A Comparative Study. J. Sci. Food Agric. 2010, 90, 1238–1244. [Google Scholar] [CrossRef] [PubMed]
- Anagnostopoulou, M.A.; Kefalas, P.; Papageorgiou, V.P.; Assimopoulou, A.N.; Boskou, D. Radical Scavenging Activity of Various Extracts and Fractions of Sweet Orange Peel (Citrus sinensis). Food Chem. 2006, 94, 19–25. [Google Scholar] [CrossRef]
- Barragán-Ocaña, A.; Silva-Borjas, P.; Olmos-Peña, S.; Polanco-Olguín, M. Biotechnology and Bioprocesses: Their Contribution to Sustainability. Processes 2020, 8, 436. [Google Scholar] [CrossRef]
- Siddiqui, S.A.; Erol, Z.; Rugji, J.; Taşçı, F.; Kahraman, H.A.; Toppi, V.; Musa, L.; Di Giacinto, G.; Bahmid, N.A.; Mehdizadeh, M.; et al. An Overview of Fermentation in the Food Industry—Looking Back from a New Perspective. Bioresour. Bioprocess. 2023, 10, 85. [Google Scholar] [CrossRef]
- Kumar, V.; Bansal, V.; Madhavan, A.; Kumar, M.; Sindhu, R.; Awasthi, M.K.; Binod, P.; Saran, S. Active Pharmaceutical Ingredient (API) Chemicals: A Critical Review of Current Biotechnological Approaches. Bioengineered 2022, 13, 4309–4327. [Google Scholar] [CrossRef] [PubMed]
- Salas-Millán, J.Á.; Aguayo, E. Fermentation for Revalorisation of Fruit and Vegetable By-Products: A Sustainable Approach Towards Minimising Food Loss and Waste. Foods 2024, 13, 3680. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; Singh, V.; Vern, P.; Panesar, P.S. From Citrus Waste to Value-Added Products: Exploring Biochemical Routes for Sustainable Valorization. Process Biochem. 2025, 158, 81–98. [Google Scholar] [CrossRef]
- Mahato, N.; Sharma, K.; Sinha, M.; Dhyani, A.; Pathak, B.; Jang, H.; Park, S.; Pashikanti, S.; Cho, S. Biotransformation of Citrus Waste-I: Production of Biofuel and Valuable Compounds by Fermentation. Processes 2021, 9, 220. [Google Scholar] [CrossRef]
- Mamy, D.; Duah Boateng, I.; Chen, X. Metabolomic Changes in Citrus Reticulata Peel After Conventional and Ultrasound-Assisted Solid-State Fermentation with Aspergillus niger: A Focus on Flavonoid Metabolism. Food Chem. 2025, 467, 142224. [Google Scholar] [CrossRef] [PubMed]
- Madeira Junior, J.V.; Teixeira, C.B.; Macedo, G.A. Biotransformation and Bioconversion of Phenolic Compounds Obtainment: An Overview. Crit. Rev. Biotechnol. 2015, 33, 191–205. [Google Scholar] [CrossRef]
- Lopez-Hernández, B.N.; Escamilla-Alvarado, C.; Albalate-Ramírez, A.; Rodríguez-Valderrama, S.; Rivas-García, P.; Paredes, M.G. Transforming Orange Peel Waste into Hydrogen: The Effect of Biocompound Extraction and Inoculum-to-Substrate Ratio on Dark Fermentation. Int. J. Hydrogen Energy 2025, 141, 1261–1270. [Google Scholar] [CrossRef]
- De la Rosa-Esteban, K.; Sepúlveda, L.; Buenrostro-Figueroa, J.J.; Chávez-González, M.L.; Sáenz-Galindo, A.; Aguilar, C.N.; Ascacio-Valdés, J.A. Sustainable Production of Ellagic Acid via Solid-State Fermentation with Saccharomyces cerevisiae in Column Reactors. Environ. Qual. Manag. 2025, 35, e70232. [Google Scholar] [CrossRef]
- Yepes-Betancur, D.P.; Márquez-Cardozo, C.J.; Cadena-Chamorro, E.M.; Martinez-Saldarriaga, J.; Torres-León, C.; Ascacio-Valdés, A.; Aguilar, C.N. Solid-State Fermentation-Assisted Extraction of Bioactive Compounds from Hass Avocado Seeds. Food Bioprod. Process. 2021, 126, 155–163. [Google Scholar] [CrossRef]
- Cerda-Cejudo, N.D.; Buenrostro-Figueroa, J.J.; Sepúlveda, L.; Estrada-Gil, L.E.; Torres-León, C.; Chávez-González, M.L.; Aguilar, C.N.; Ascacio-Valdés, J.A. Enhancing the Release of Ellagic Acid from Mexican Rambutan Peel Using Solid-State Fermentation. Biomass 2024, 4, 1005–1016. [Google Scholar] [CrossRef]
- Mantzouridou, F.; Paraskevopoulou, A.; Lalou, S. Yeast Flavour Production by Solid State Fermentation of Orange Peel Waste. Biochem. Eng. J. 2015, 101, 1–8. [Google Scholar] [CrossRef]
- Yuan, Y.; Zhou, J.; Zhang, N.; Xing, X.; Zhao, L.; Zhou, F. In Vivo Study of Orange Peel Fermentation and Improvement of Androgen Overproduction Causing Acne. Food Biosci. 2023, 56, 103124. [Google Scholar] [CrossRef]
- Gómez-Mejía, E.; Rosales-Coronado, N.; León-González, M.E.; Madrid, Y. Citrus Peels Waste as a Source of Value-Added Compounds. Food Chem. 2019, 295, 289–299. [Google Scholar] [CrossRef]
- Kumar, A.; Saranyadevi, S.; Thirumalaisamy, S.K.; Dapana Durage, T.T.; Jaiswal, S.G.; Kavitake, D.; Wei, S. Phenolic Acids in Fermented Foods: Microbial Biotransformation, Antioxidant Mechanisms, and Functional Health Implications. Front. Mol. Biosci. 2025, 12, 1678673. [Google Scholar] [CrossRef]
- Huynh, N.T.; Van Camp, J.; Smagghe, G.; Raes, K. Improved Release and Metabolism of Flavonoids by Steered Fermentation Processes: A Review. Int. J. Mol. Sci. 2014, 15, 19369–19388. [Google Scholar] [CrossRef]
- Omoba, O.S.; Obafaye, R.O.; Salawu, S.O.; Boligon, A.A.; Athayde, M.L. HPLC-DAD Phenolic Characterization and Antioxidant Activities of Ripe and Unripe Sweet Orange Peels. Antioxidants 2015, 4, 498–512. [Google Scholar] [CrossRef]
- Reyes-Moreno, C.; Cuevas-Rodríguez, E.O.; Millán-Carrillo, J.; Cárdenas-Valenzuela, O.G.; Barrón-Hoyos, J. Solid state fermentation process for producing chickpea (Cicer arietinum L.) tempeh flour. Physicochemical and nutritional characteristics of the product. J. Sci. Food Agric. 2004, 84, 271–278. [Google Scholar] [CrossRef]
- Londoño-Hernández, L.; Ruiz, H.A.; Ramírez, T.C.; Ascacio, J.A.; Rodríguez-Herrera, R.; Aguilar, C.N. Fungal Detoxification of Coffee Pulp by Solid-State Fermentation. Biocatal. Agric. Biotechnol. 2020, 23, 101417. [Google Scholar] [CrossRef]
- Dulf, F.V.; Vodnar, D.C.; Socaciu, C. Effects of Solid-State Fermentation with Two Filamentous Fungi on the Total Phenolic Contents, Flavonoids, Antioxidant Activities and Lipid Fractions of Plum Fruit (Prunus domestica L.) By-Products. Food Chem. 2016, 209, 27–36. [Google Scholar] [CrossRef] [PubMed]
- De La Rosa-Esteban, K.; Sepúlveda, L.; Buenrostro-Figueroa, J.J.; Chávez-González, M.L.; Estrada-Gil, L.E.; Saenz-Galindo, A.; Aguilar, C.N.; Ascacio-Valdés, J.A. Solid-State Fermentation: An Alternative for Continuous and Discontinuous Production of Bioactive Compounds with Biological Activity. Explor. Foods Foodomics 2025, 3, 101085. [Google Scholar] [CrossRef]
- Srivastava, N.; Srivastava, M.; Ramteke, P.W.; Mishra, P.K. Solid State Fermentation Strategy for Microbial Metabolites Production. In New and Future Developments in Microbial Biotechnology and Bioengineering; Gupta, V.K., Pandey, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 345–352. [Google Scholar]
- Roasa, J.; De Villa, R.; Mine, Y.; Tsao, R. Phenolics of cereal, pulse and oilseed processing by-products and potential effects of solid-state fermentation on their bioaccessibility, bioavailability and health benefits: A review. Trends Food Sci. Technol. 2021, 116, 954–974. [Google Scholar] [CrossRef]
- Fiorentini, D.; Cappadone, C.; Farruggia, G.; Prata, C. Magnesium: Biochemistry, Nutrition, Detection, and Social Impact of Diseases Linked to Its Deficiency. Nutrients 2021, 13, 1136. [Google Scholar] [CrossRef]
- Kumar, V.; Ahluwalia, V.; Saran, S.; Kumar, J.; Patel, A.K.; Singhania, R.R. Recent developments on solid-state fermentation for production of microbial secondary metabolites: Challenges and solutions. Bioresour. Technol. 2021, 323, 124566. [Google Scholar] [CrossRef]
- López-Gómez, J.P.; Manan, M.A.; Webb, C. Solid-State Fermentation of Food Industry Wastes. In Food Industry Wastes, 2nd ed.; Kosseva, M.R., Webb, C., Eds.; Elsevier: London, UK, 2020; pp. 135–161. [Google Scholar]
- Rodríguez-Pupo, E.C.; Pérez-Llano, Y.; Tinoco-Valencia, J.R.; Sánchez, N.S.; Padilla-Garfias, F.; Calahorra, M.; Sánchez, N.d.C.; Sánchez-Reyes, A.; Rodríguez-Hernández, M.d.R.; Peña, A.; et al. Osmolyte Signatures for the Protection of Aspergillus sydowii Cells under Halophilic Conditions and Osmotic Shock. J. Fungi 2021, 7, 414. [Google Scholar] [CrossRef]
- Markina-Iñarrairaegui, A.; Spielvogel, A.; Etxebeste, O.; Ugalde, U.; Espeso, E.A. Tolerance to Alkaline Ambient pH in Aspergillus nidulans Depends on the Activity of ENA Proteins. Sci. Rep. 2020, 10, 14325. [Google Scholar] [CrossRef]
- Mostafa, F.A.; Wehaidy, H.R.; Sharaf, S.; El-Hennawi, H.M.; Mahmoud, S.A.; Saleh, S.A. Aspergillus awamori MK788209 cellulase: Production, statistical optimization, pea peels saccharification and textile applications. Microb. Cell Factories 2024, 23, 11. [Google Scholar] [CrossRef] [PubMed]
- Madeira, J.V., Jr.; Macedo, G.A. Simultaneous Extraction and Biotransformation Process to Obtain High Bioactivity Phenolic Compounds from Brazilian Citrus Residues. Biotechnol. Prog. 2015, 31, 1273–1279. [Google Scholar] [CrossRef] [PubMed]
- Ni, H.; Chen, F.; Jiang, Z.D.; Cai, M.Y.; Yang, Y.F.; Xiao, A.F.; Cai, H.N. Biotransformation of tea catechins using Aspergillus niger tannase prepared by solid state fermentation on tea byproduct. LWT 2015, 60, 1206–1213. [Google Scholar] [CrossRef]
- Mamy, D.; Huang, Y.; Akpabli-Tsigbe, N.D.K.; Battino, M.; Chen, X. Valorization of Citrus Reticulata Peels for Flavonoids and Antioxidant Enhancement by Solid-State Fermentation Using Aspergillus niger CGMCC 3.6189. Molecules 2022, 27, 8949. [Google Scholar] [CrossRef] [PubMed]
- Cao, C.; Lin, D.; Zhou, Y.; Li, N.; Wang, Y.; Gong, W.; Zhu, Z.; Liu, C.; Yan, L.; Hu, Z.; et al. Solid-state fermentation of Apocynum venetum L. by Aspergillus niger: Effect on phenolic compounds, antioxidant activities and metabolic syndrome-associated enzymes. Front. Nutr. 2023, 10, 1125746. [Google Scholar] [CrossRef]
- Casas-Rodríguez, A.D.; Ascacio-Valdés, J.A.; Dávila-Medina, M.D.; Medina-Morales, M.A.; Londoño-Hernández, L.; Sepúlveda, L. Evaluation of Solid-State Fermentation Conditions from Pineapple Peel Waste for Release of Bioactive Compounds by Aspergillus niger spp. Appl. Microbiol. 2024, 4, 934–947. [Google Scholar] [CrossRef]
- PubChem. Available online: https://pubchem.ncbi.nlm.nih.gov/ (accessed on 31 October 2025).
- Chen, Z.T.; Chu, H.L.; Chyau, C.C.; Chu, C.C.; Duh, P.D. Protective effects of sweet orange (Citrus sinensis) peel and their bioactive compounds on oxidative stress. Food Chem. 2012, 135, 2119–2127. [Google Scholar] [CrossRef]
- Wang, Y.C.; Chuang, Y.C.; Hsu, H.W. The Flavonoid, Carotenoid and Pectin Content in Peels of Citrus Cultivated in Taiwan. Food Chem. 2008, 106, 277–284. [Google Scholar] [CrossRef]
- Escobar-Blanco, M. Extracción de Compuestos Fenólicos de las Cáscaras de Cítricos Producidos en México. Master’s Thesis, Instituto Politécnico Nacional, Ciudad de México, Mexico, 2010. [Google Scholar]
- Tenorio-Domínguez, M. Flavonoids extracted from orange peelings tangelo (Citrus reticulata × Citrus paradisi) and their application as a natural antioxidant in sacha inchi (Plukenetia volubilis) vegetable oil. Sci. Agropecu. 2016, 7, 419–431. [Google Scholar] [CrossRef]
- Araújo, L.P.; Vilela, H.; Solinho, J.; Pinheiro, R.; Belo, I.; Lopes, M. Enrichment of Fruit Peels’ Nutritional Value by Solid-State Fermentation with Aspergillus ibericus and Rhizopus oryzae. Molecules 2024, 29, 3563. [Google Scholar] [CrossRef] [PubMed]




| Treatments | Inoculum (Spores/g) | KCl (g/L) | MgSO4 (g/L) |
|---|---|---|---|
| 1 | −1 | −1 | −1 |
| 2 | 1 | −1 | 1 |
| 3 | −1 | −1 | 1 |
| 4 | 1 | −1 | −1 |
| 5 | −1 | 1 | 1 |
| 6 | 1 | 1 | −1 |
| 7 | −1 | 1 | −1 |
| 8 | 1 | 1 | 1 |
| 9 | −1 | −1 | −1 |
| 10 | 1 | −1 | 1 |
| 11 | −1 | −1 | 1 |
| 12 | 1 | −1 | −1 |
| 13 | −1 | 1 | 1 |
| 14 | 1 | 1 | −1 |
| 15 | −1 | 1 | −1 |
| 16 | 1 | 1 | 1 |
| 17 | −1 | −1 | −1 |
| 18 | 1 | −1 | 1 |
| 19 | −1 | −1 | 1 |
| 20 | 1 | −1 | −1 |
| 21 | −1 | 1 | 1 |
| 22 | 1 | 1 | −1 |
| 23 | −1 | 1 | −1 |
| 24 | 1 | 1 | 1 |
| Levels | |||
| Factors | +1 | −1 | |
| Inoculum (spores/g) | 1 × 108 | 1 × 106 | |
| KCl (g/L) | 1.56 | 0.76 | |
| MgSO4 (g/L) | 1.56 | 0.76 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Cruz-López, S.M.; Laredo-Alcalá, E.I.; Ruelas-Chacón, X.; Aguilar, C.N.; Ascacio-Valdés, J.A.; Sepúlveda, L. Fungal Solid-State Fermentation as a Strategy to Release Polyphenols from Orange Peel Waste. Processes 2026, 14, 622. https://doi.org/10.3390/pr14040622
Cruz-López SM, Laredo-Alcalá EI, Ruelas-Chacón X, Aguilar CN, Ascacio-Valdés JA, Sepúlveda L. Fungal Solid-State Fermentation as a Strategy to Release Polyphenols from Orange Peel Waste. Processes. 2026; 14(4):622. https://doi.org/10.3390/pr14040622
Chicago/Turabian StyleCruz-López, Silvia Magali, Elan I. Laredo-Alcalá, Xochitl Ruelas-Chacón, Cristóbal N. Aguilar, Juan A. Ascacio-Valdés, and Leonardo Sepúlveda. 2026. "Fungal Solid-State Fermentation as a Strategy to Release Polyphenols from Orange Peel Waste" Processes 14, no. 4: 622. https://doi.org/10.3390/pr14040622
APA StyleCruz-López, S. M., Laredo-Alcalá, E. I., Ruelas-Chacón, X., Aguilar, C. N., Ascacio-Valdés, J. A., & Sepúlveda, L. (2026). Fungal Solid-State Fermentation as a Strategy to Release Polyphenols from Orange Peel Waste. Processes, 14(4), 622. https://doi.org/10.3390/pr14040622











