Process Design and Kinetic-Based Simulation of a Coupled Biomass Gasification and Chemical Looping Ammonia Generation System
Abstract
1. Introduction
2. Process Simulation and Model Development
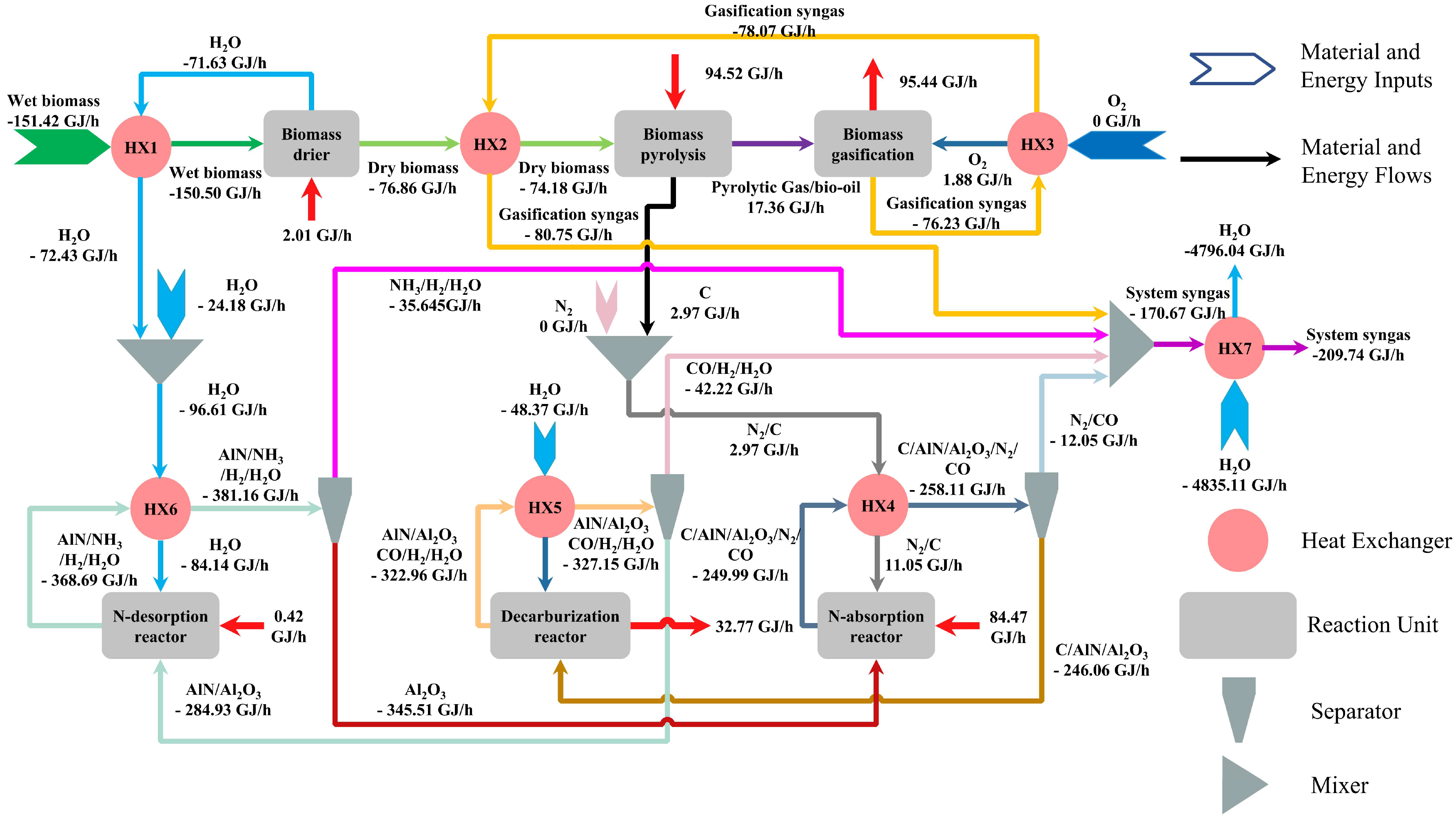
2.1. System Description and Simulation Assumptions
2.2. Model Assumptions and Simulation Constraints
- The system operates under steady-state conditions. Pressure drops across reactors, cyclones, and piping are neglected in this conceptual design phase. This simplification is adopted because detailed geometric parameters (e.g., pipe layouts and lengths) are undefined at this stage. While it is acknowledged that practical operation requires auxiliary power for blowers, preliminary order-of-magnitude analysis indicates that this mechanical energy input is substantially lower than the system’s dominant thermal energy duties (reaction enthalpy). Furthermore, this omission does not affect the fundamental comparative advantage of the CLAG process over the Haber-Bosch route, which requires massive energy for high-pressure gas compression (150–300 bar).
- Chemical reactions are modeled based on their respective governing principles: thermodynamic equilibrium is assumed for biomass gasification, while conversion levels for the CLAG process are strictly defined by experimental kinetic data. Phase separations are assumed to be ideal.
- Biomass is modeled as a non-conventional component based on its ultimate and proximate analyses. The O2/Biomass molar ratio (O2/B) in the gasification unit is normalized to the molar flow rate of carbon in the feedstock.
- The product spectrum of biomass gasification is restricted to major thermodynamic species: O2, CO, CO2, CH4, C2H6, C3H8, COS, SO2, H2S, NH3, N2, H2O, and H2 [35].
- In the decarburization unit, complete conversion of residual carbon with steam is assumed, yielding CO and H2.
- The waste heat recovery unit is designed to cool process gas streams to a discharge temperature of 120 °C to prevent acid dew point corrosion while maximizing energy recovery.
2.3. Physical Property Parameters and Model Construction
3. Results and Discussion
3.1. Performance Characteristics of Biomass Gasification
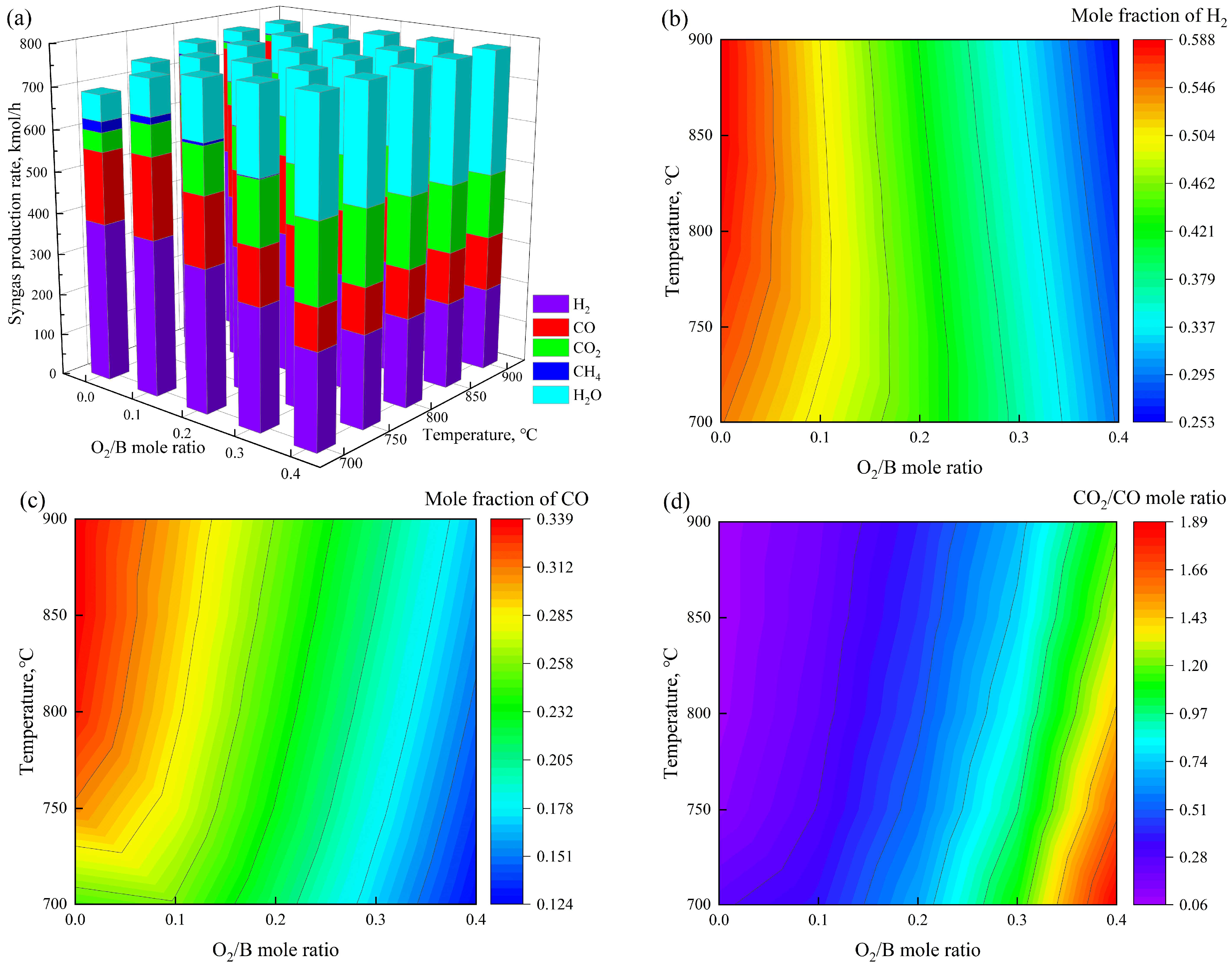
3.1.1. Syngas Yield and Compositional Analysis
3.1.2. Energy Balance and Optimal Operating Point
3.2. Optimization of the Chemical Looping Ammonia Generation (CLAG) Process
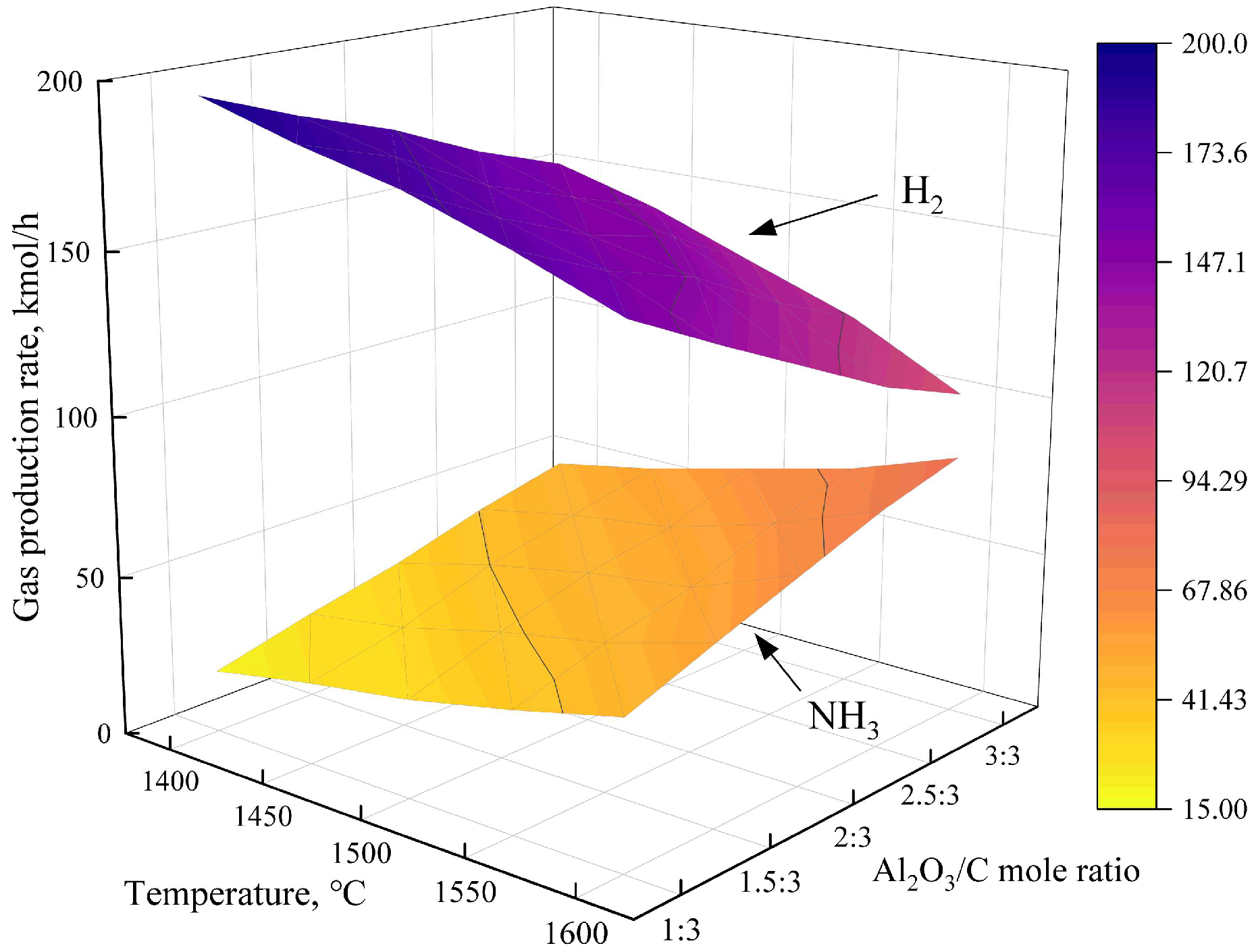
3.2.1. Ammonia and Hydrogen Yield Analysis
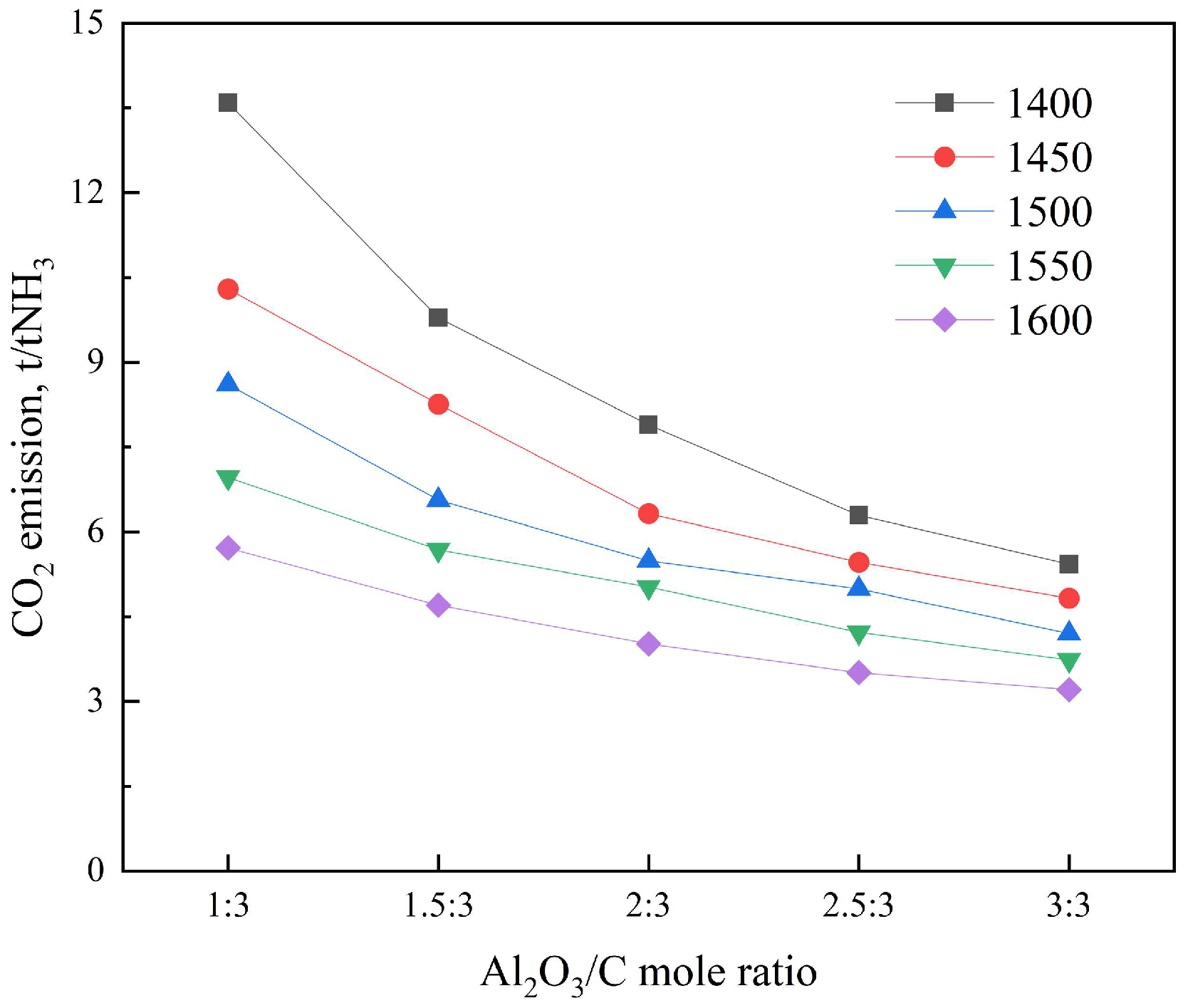
3.2.2. Carbon Footprint Analysis
3.3. System-Level Energy Efficiency Analysis
3.3.1. Heat Duty Distribution
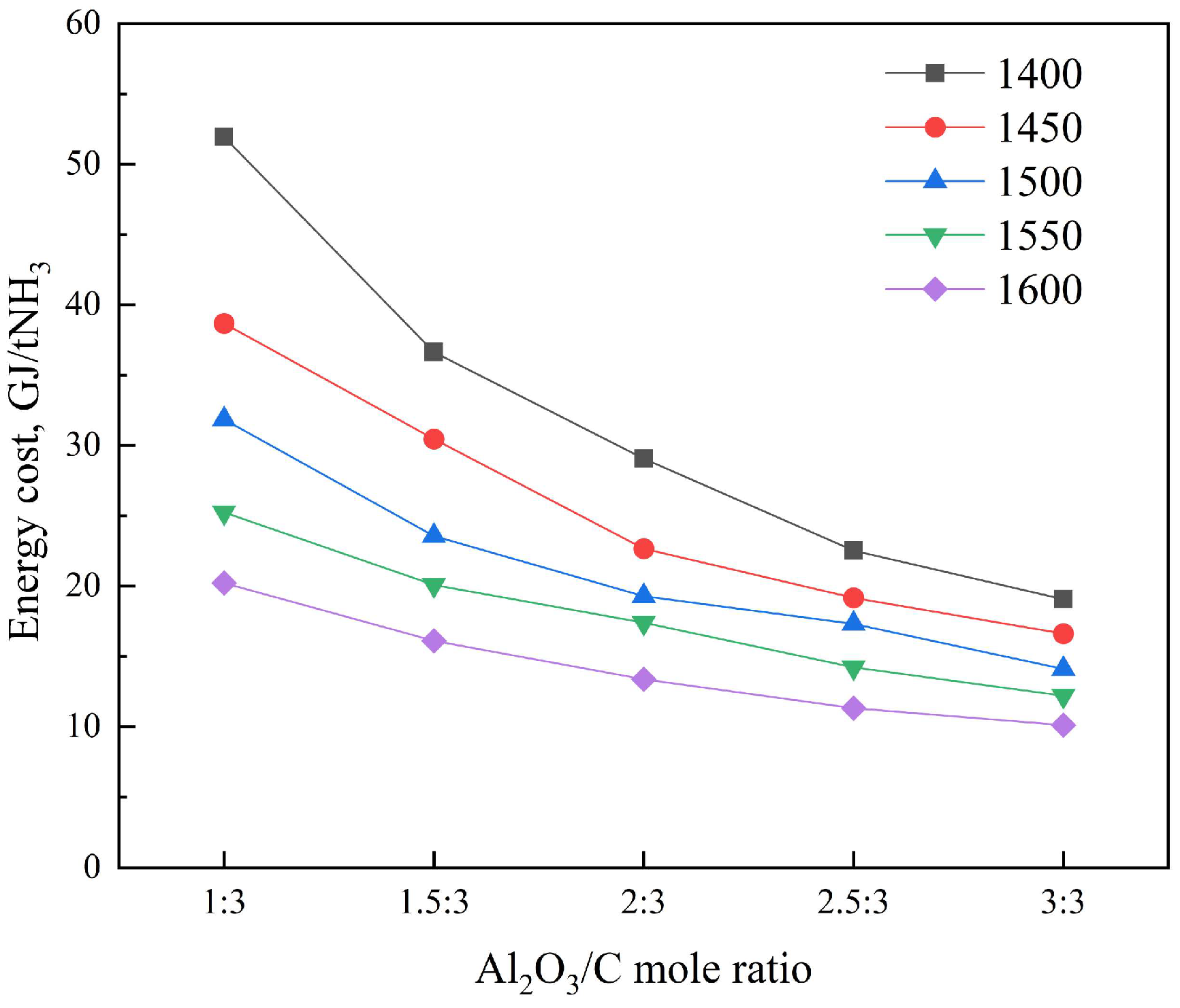
3.3.2. Specific Energy Consumption Comparison
3.4. Energy Flow and Gas Production Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Gezerman, A.O. A Critical Assessment of Green Ammonia Production and Ammonia Production Technologies. Kem. Ind. 2022, 71, 57–66. [Google Scholar] [CrossRef]
- Wang, Y.; Yao, Z.; Wang, Y.; Yan, G.; Janz, B.; Wang, X.; Zhan, Y.; Wang, R.; Zheng, X.; Zhou, M.; et al. Characteristics of annual NH(3) emissions from a conventional vegetable field under various nitrogen management strategies. J. Environ. Manag. 2023, 342, 118276. [Google Scholar] [CrossRef] [PubMed]
- Rafiqul, I.; Weber, C.; Lehmann, B.; Voss, A. Energy efficiency improvements in ammonia production—Perspectives and uncertainties. Energy 2005, 30, 2487–2504. [Google Scholar] [CrossRef]
- Chi, W.; Yang, Q.; Chen, X.; Liu, G.; Zhao, Y.; Li, L. Performance evaluation of NH3/CO2 cascade refrigeration system with ejector subcooling for low-temperature cycle. Int. J. Refrig. 2022, 136, 162–171. [Google Scholar] [CrossRef]
- Lesmana, H.; Zhang, Z.; Li, X.; Zhu, M.; Xu, W.; Zhang, D. NH3 as a transport fuel in internal combustion engines: A technical review. J. Energy Resour. Technol. 2019, 141, 070703. [Google Scholar] [CrossRef]
- Boretti, A.; Castelletto, S. NH3 Prospects in Combustion Engines and Fuel Cells for Commercial Aviation by 2030. ACS Energy Lett. 2022, 7, 2557–2564. [Google Scholar] [CrossRef]
- Zhou, M.; Wang, Y.; Chu, Y.; Tang, Y.; Tian, K.; Zheng, S.; Chen, J.; Wang, Z. Ammonia as an environmentally benign energy carrier for the fast growth of China. Energy Procedia 2019, 158, 4986–4991. [Google Scholar] [CrossRef]
- Awad, O.I.; Zhou, B.; Harrath, K.; Kadirgama, K. Characteristics of NH3/H2 blend as carbon-free fuels: A review. Int. J. Hydrogen Energy 2022, 48, 38077–38100. [Google Scholar] [CrossRef]
- Wang, Y.; Wright, L.A. A Comparative Review of Alternative Fuels for the Maritime Sector: Economic, Technology, and Policy Challenges for Clean Energy Implementation. World 2021, 2, 456–481. [Google Scholar] [CrossRef]
- Humphreys, J.; Lan, R.; Tao, S. Development and Recent Progress on Ammonia Synthesis Catalysts for Haber–Bosch Process. Adv. Energy Sustain. Res. 2020, 2, 2000043–2000066. [Google Scholar] [CrossRef]
- Klaas, L.; Guban, D.; Roeb, M.; Sattler, C. Recent progress towards solar energy integration into low-pressure green ammonia production technologies. Int. J. Hydrogen Energy 2021, 46, 25121–25136. [Google Scholar] [CrossRef]
- Vojvodic, A.; Medford, A.J.; Studt, F.; Abild-Pedersen, F.; Khan, T.S.; Bligaard, T.; Nørskov, J.K. Exploring the limits: A low-pressure, low-temperature Haber–Bosch process. Chem. Phys. Lett. 2014, 598, 108–112. [Google Scholar] [CrossRef]
- Liu, Z.; Yu, Q.; Wang, H.; Wu, J.; Tao, S. Selecting nitrogen carriers used for chemical looping ammonia generation of biomass and H2O by thermodynamic method. Int. J. Hydrogen Energy 2023, 48, 4035–4051. [Google Scholar]
- Xiang, P.-P.; Wang, J.-C.; Jiang, K.-J.; He, C.-M.; Jiang, W.-Y.; Guo, L.-Q.; Jiao, Y.-J.; Chen, S. Regional reallocation of zero-carbon ammonia production in China with carbon neutrality targets. Adv. Clim. Change Res. 2025, 16, 199–212. [Google Scholar] [CrossRef]
- Chen, J.; Guan, B.; Zhuang, Z.; Zheng, C.; Zhou, J.; Su, T.; Chen, Y.; Zhu, C.; Hu, X.; Zhao, S.; et al. Recent advances of structure-performance relationship and improvement methods of catalysts for photochemical and electrochemical reduction of nitrogen to green ammonia. Fuel 2024, 371, 131928–131970. [Google Scholar] [CrossRef]
- Wang, S.-X.; Maimaiti, H.; Xu, B.; Guo, Y.; Zhai, P.-s.; Zhang, H.-z. Fixation of Nitrogen to Ammonia with Photocatalytic on Petroleum Pitch-Based Graphene Oxide Supported Nickel/Nickel Oxide Composite Catalyst. J. Phys. Chem. C 2019, 123, 31119–31129. [Google Scholar] [CrossRef]
- Shipman, M.A.; Symes, M.D. Recent progress towards the electrosynthesis of ammonia from sustainable resources. Catal. Today 2017, 286, 57–68. [Google Scholar] [CrossRef]
- Ling, C.; Zhang, Y.; Li, Q.; Bai, X.; Shi, L.; Wang, J. New Mechanism for N2 Reduction: The Essential Role of Surface Hydrogenation. J. Am. Chem. Soc. 2019, 141, 18264–18270. [Google Scholar] [CrossRef]
- Neyts, E.C.; Bogaerts, A. Understanding plasma catalysis through modelling and simulation—A review. J. Phys. D Appl. Phys. 2014, 47, 224010–224029. [Google Scholar] [CrossRef]
- Bröer, S.; Hammer, T. Selective catalytic reduction of nitrogen oxides by combining a non-thermal plasma and a V2O5-WO3/TiO2 catalyst. Appl. Catal. B: Environ. 2000, 28, 101–111. [Google Scholar] [CrossRef]
- Li, L.; Tang, C.; Cui, X.; Zheng, Y.; Wang, X.; Xu, H.; Zhang, S.; Shao, T.; Davey, K.; Qiao, S.Z. Efficient Nitrogen Fixation to Ammonia through Integration of Plasma Oxidation with Electrocatalytic Reduction. Angew. Chem. Int. Ed. 2021, 60, 14131–14137. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Wang, T.; Zhao, Z.-J.; Yang, W.; Li, J.-F.; Li, A.; Yang, Z.; Ozin, G.A.; Gong, J. Promoted Fixation of Molecular Nitrogen with Surface Oxygen Vacancies on Plasmon-Enhanced TiO2 Photoelectrodes. Angew. Chem. Int. Ed. 2018, 57, 5278–5282. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zhao, Y.; Shi, R.; Waterhouse, G.I.N.; Zhang, T. Photocatalytic ammonia synthesis: Recent progress and future. EnergyChem 2019, 1, 100013. [Google Scholar] [CrossRef]
- Chen, J.G.; Crooks, R.M.; Seefeldt, L.C.; Bren, K.L.; Bullock, R.M.; Darensbourg, M.Y.; Holland, P.L.; Hoffman, B.; Janik, M.J.; Jones, A.K.; et al. Beyond fossil fuel-driven nitrogen transformations. Science 2018, 360, eaar6611. [Google Scholar] [CrossRef]
- Gao, W.; Guo, J.; Wang, P.; Wang, Q.; Chang, F.; Pei, Q.; Zhang, W.; Liu, L.; Chen, P. Production of ammonia via a chemical looping process based on metal imides as nitrogen carriers. Nat. Energy 2018, 3, 1067–1075. [Google Scholar] [CrossRef]
- Wang, B.; Yin, X.; Wang, P.; Shen, L. Chemical looping ammonia synthesis at atmospheric pressure benefiting from synergistic effect of Mn- and Fe-based nitrogen carriers. Int. J. Hydrogen Energy 2023, 48, 2705–2717. [Google Scholar] [CrossRef]
- Gálvez, M.; Hischier, I.; Frei, A.; Steinfeld, A. Ammonia Production via a Two-Step Al2O3/AlN Thermochemical Cycle. 3. Influence of the Carbon Reducing Agent and Cyclability. Ind. Eng. Chem. Res. 2008, 47, 2231–2237. [Google Scholar] [CrossRef]
- Feng, M.; Zhang, Q.; Wu, Y.; Liu, D. Using Coal Coke for N-Sorption with an Al-based Nitrogen Carrier during Chemical Looping Ammonia Generation. Energy Fuels 2020, 34, 12527–12534. [Google Scholar] [CrossRef]
- Liu, Z.; Yu, Q.; Gao, J.; Zhao, J.; Duan, W. Effect of pyrolysis parameters on the biochar reactivity in the N-absorption reaction of chemical looping ammonia generation. Energy 2024, 310, 133321. [Google Scholar] [CrossRef]
- Zhang, Q.; Wu, Y.; Gao, Y.; Chen, X.; Liu, D.; Fan, M. High-performance mesoporous (AlN/Al2O3) for enhanced NH3 yield during chemical looping ammonia generation technology. Int. J. Hydrogen Energy 2020, 45, 9903–9913. [Google Scholar] [CrossRef]
- Liu, Z.; Yu, Q.; Zhao, J.; Gao, J.; Duan, W. α-Al2O3-loaded promoted the N-desorption reaction and inhibited NH3 decomposition in chemical looping ammonia generation: Experiments and DFT simulations. Int. J. Hydrogen Energy 2024, 78, 481–491. [Google Scholar] [CrossRef]
- Weng, Q.; Toan, S.; Ai, R.; Sun, Z.; Sun, Z. Ammonia production from biomass via a chemical looping-based hybrid system. J. Clean. Prod. 2021, 289, 125749. [Google Scholar] [CrossRef]
- Fang, J.; Xiong, C.; Feng, M.; Wu, Y.; Liu, D. Utilization of carbon-based energy as raw material instead of fuel with low CO2 emissions: Energy analyses and process integration of chemical looping ammonia generation. Appl. Energy 2022, 312, 118809–118818. [Google Scholar] [CrossRef]
- Wen, D.; Aziz, M. Design and analysis of biomass-to-ammonia-to-power as an energy storage method in a renewable multi-generation system. Energy Convers. Manag. 2022, 261, 115611. [Google Scholar] [CrossRef]
- Kataria, G.; Sharma, A.; Joshi, J.B.; Hameed, S.; Amiri, A. A system level analysis of pyrolysis of cotton stalk biomass. Mater. Today Proc. 2022, 57, 1528–1532. [Google Scholar] [CrossRef]
- Duan, W.; Yu, Q.; Wang, K.; Qin, Q.; Hou, L.; Yao, X.; Wu, T. ASPEN Plus simulation of coal integrated gasification combined blast furnace slag waste heat recovery system. Energy Convers. Manag. 2015, 100, 30–36. [Google Scholar] [CrossRef]
- Liu, Z.; Yu, Q.; Xie, H.; Gao, J.; Zhao, J. Experimental and Kinetic Study of Biochar in N-Absorption Reaction of Chemical Looping Ammonia Generation. Processes 2024, 12, 2870. [Google Scholar] [CrossRef]
- Yan, R.; Wu, M.; Fan, J.; Sun, C.; Wang, J.; He, Y.; Liu, H.; Li, P.; Zhang, J. Process integration and thermodynamic analysis of a multi-generation system including solar-assisted biomass gasification and chemical looping ammonia generation. Energy Convers. Manag. 2024, 306, 118263. [Google Scholar] [CrossRef]


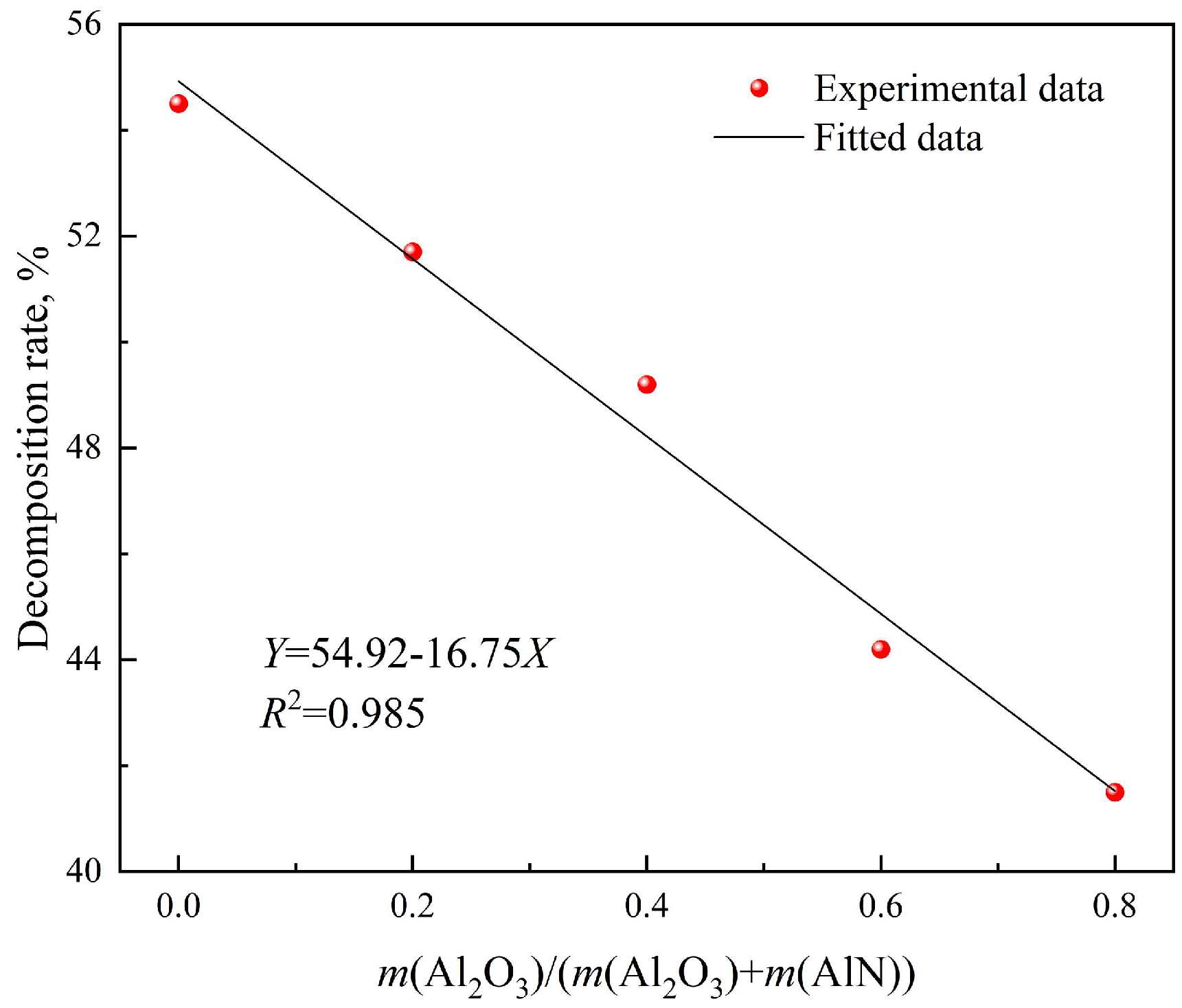





| Proximate Analysis | wt% | Ultimate Analysis (d.b.) | wt% |
|---|---|---|---|
| Fixed carbon (d.b.) | 14.48 | Carbon | 48.50 |
| Moisture (a.r.) | 4.97 | Hydrogen | 7.94 |
| Volatile matter (d.b.) | 80.46 | Oxygen (by difference) | 41.50 |
| Ash (d.b.) | 0.09 | Nitrogen | 2.05 |
| Total sulfur | 0.01 |
| Temperature, °C | α-Al2O3:C | C Conversion, % | Mass Fraction of α-Al2O3, % | NH3 Decomposition Rate, % |
|---|---|---|---|---|
| 1400 | 1:3 | 21.9 | 81.6 | 41.3 |
| 1.5:3 | 30.4 | 83.1 | 41 | |
| 2:3 | 37.5 | 84.4 | 40.8 | |
| 2.5:3 | 47.1 | 84.3 | 40.8 | |
| 3:3 | 54.5 | 84.9 | 40.7 | |
| 1450 | 1:3 | 29.6 | 74.8 | 42.4 |
| 1.5:3 | 36.3 | 79.6 | 41.6 | |
| 2:3 | 47.4 | 80 | 41.5 | |
| 2.5:3 | 54.6 | 81.6 | 41.2 | |
| 3:3 | 61.6 | 82.8 | 41.1 | |
| 1500 | 1:3 | 36.0 | 68.9 | 43.4 |
| 1.5:3 | 46.5 | 73.5 | 42.6 | |
| 2:3 | 55.2 | 76.6 | 42.1 | |
| 2.5:3 | 60.1 | 79.7 | 41.6 | |
| 3:3 | 71.3 | 79.9 | 41.5 | |
| 1550 | 1:3 | 45.7 | 59.6 | 44.9 |
| 1.5:3 | 54.6 | 68.5 | 43.4 | |
| 2:3 | 60.7 | 74.1 | 42.5 | |
| 2.5:3 | 71.9 | 75.5 | 42.3 | |
| 3:3 | 81.0 | 77.1 | 42 | |
| 1600 | 1:3 | 57.8 | 47.6 | 46.9 |
| 1.5:3 | 67.6 | 60.3 | 44.8 | |
| 2:3 | 77.7 | 66.2 | 43.8 | |
| 2.5:3 | 88.0 | 69.6 | 43.3 | |
| 3:3 | 95.5 | 72.7 | 42.7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Liu, Z.; Yu, Q.; Xie, H.; Luo, L.; Chen, Z.; Yu, G.; Wang, C. Process Design and Kinetic-Based Simulation of a Coupled Biomass Gasification and Chemical Looping Ammonia Generation System. Processes 2026, 14, 588. https://doi.org/10.3390/pr14040588
Liu Z, Yu Q, Xie H, Luo L, Chen Z, Yu G, Wang C. Process Design and Kinetic-Based Simulation of a Coupled Biomass Gasification and Chemical Looping Ammonia Generation System. Processes. 2026; 14(4):588. https://doi.org/10.3390/pr14040588
Chicago/Turabian StyleLiu, Zhongyuan, Qingbo Yu, Huaqing Xie, Lunbo Luo, Ziwen Chen, Guangming Yu, and Chen Wang. 2026. "Process Design and Kinetic-Based Simulation of a Coupled Biomass Gasification and Chemical Looping Ammonia Generation System" Processes 14, no. 4: 588. https://doi.org/10.3390/pr14040588
APA StyleLiu, Z., Yu, Q., Xie, H., Luo, L., Chen, Z., Yu, G., & Wang, C. (2026). Process Design and Kinetic-Based Simulation of a Coupled Biomass Gasification and Chemical Looping Ammonia Generation System. Processes, 14(4), 588. https://doi.org/10.3390/pr14040588






