Rational Design of ZnGa-Sebacate/Graphene Nanoribbon Synergy for Effective Anticorrosive Polyurethane Coatings
Abstract
1. Introduction
2. Experimental
2.1. Materials and Reagents
2.2. Protocol for Fabricating the Nanofillers
2.2.1. Synthesis of ZnGa- LDH
2.2.2. Synthesis of ZnGa-SB LDH
2.2.3. Synthesis of GNR
2.2.4. Synthesis of ZnGa-SB/GNR
2.3. Protocol for Fabrication of Coatings
2.3.1. Preparation of Acrylic-Coated Steel Samples
2.3.2. Preparation of Polyurethane-Coated Steel Samples
2.4. Pigments Characterisation
2.5. Anticorrosive Evaluations
2.5.1. Electrochemical Assessments
2.5.2. Neutral Salt Spray Test
3. Results and Discussion
3.1. Nanoparticles Characterisation and Microstructure
3.1.1. X-Ray Diffraction Analysis
3.1.2. FT-IR Analysis
3.1.3. Raman Spectroscopy
3.1.4. Thermogravimetric Analysis
3.1.5. FE-SEM and EDS Analysis
3.2. Corrosion Studies
3.2.1. Short-Term Evaluation of Nanofillers
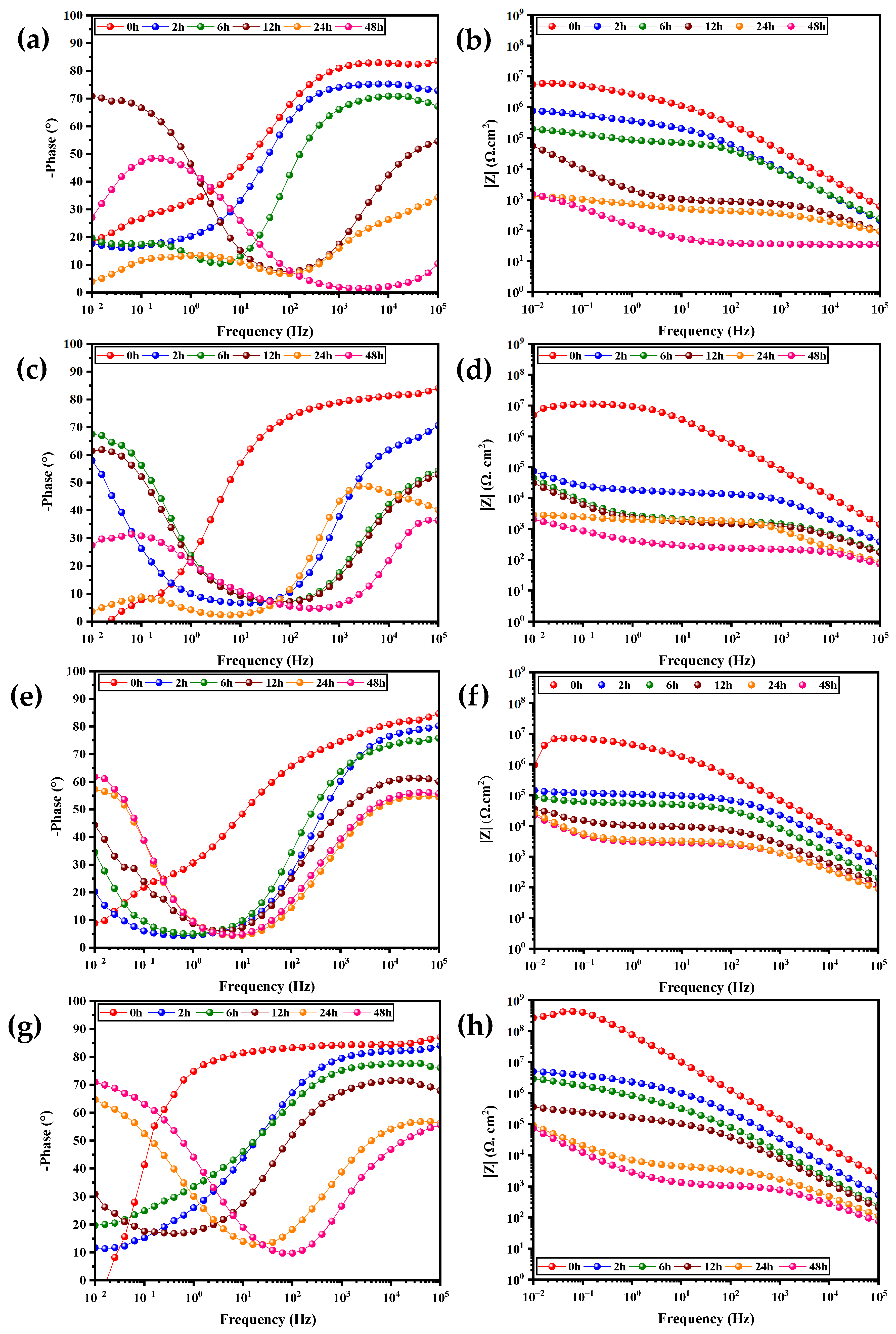
3.2.2. EIS of Intact Polyurethane Coatings
3.2.3. Neutral Salt Spray Analysis
3.2.4. Crosshatch Adhesion Test
3.2.5. Pencil Hardness Test
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nguyen, T.D.; Nguyen, A.S.; Tran, B.A.; Vu, K.O.; Tran, D.L.; Phan, T.T.; Scharnagl, N.; Zheludkevich, M.L.; To, T.X.H. Molybdate intercalated hydrotalcite/graphene oxide composite as corrosion inhibitor for carbon steel. Surf. Coat. Technol. 2020, 399, 126165. [Google Scholar] [CrossRef]
- Zhang, M.; Xu, F.; Lin, D.; Peng, J.; Zhu, Y.; Wang, H. A smart anti-corrosion coating based on triple functional fillers. Chem. Eng. J. 2022, 446, 137078. [Google Scholar] [CrossRef]
- Ji, X.; Seif, A.; Duan, J.; Rashidi, A.; Zhou, Z.; Pourhashem, S.; Mirzaee, M.; Zhai, X.; Zhao, X.; Hou, B. Experimental and DFT studies on corrosion protection performance of epoxy/graphene quantum dots@TiO2 nanotubes coatings. Constr. Build. Mater. 2022, 322, 126501. [Google Scholar] [CrossRef]
- Zehra, S.; Mobin, M.; Parveen, M.; Ahmad, R. Corrosion Fundamentals: Understanding the Science Behind the Damage. In Industrial Corrosion: Fundamentals, Failure, Analysis and Prevention; Wiley: Hoboken, NJ, USA, 2025; pp. 1–16. [Google Scholar] [CrossRef]
- Anjum, M.J.; Zhao, J.; Zahedi Asl, V.; Yasin, G.; Wang, W.; Wei, S.; Zhao, Z.; Qamar Khan, W. In-situ intercalation of 8-hydroxyquinoline in Mg-Al LDH coating to improve the corrosion resistance of AZ31. Corros. Sci. 2019, 157, 1–10. [Google Scholar] [CrossRef]
- Danaee, I.; Darmiani, E.; Rashed, G.R.; Zaarei, D. Self-healing and anticorrosive properties of Ce(III)/Ce(IV) in nanoclay–epoxy coatings. Iran. Polym. J. 2014, 23, 891–898. [Google Scholar] [CrossRef]
- Zhong, F.; He, Y.; Wang, P.; Chen, C.; Xie, P.; Li, H.; Chen, J. One-step hydrothermal synthesis of reduced graphene oxide/aspartic acid intercalated layered double hydroxide for enhancing barrier and self-healing properties of epoxy coating. React. Funct. Polym. 2019, 145, 104380. [Google Scholar] [CrossRef]
- Tabish, M.; Zhao, J.; Kumar, A.; Yan, J.; Wang, J.; Shi, F.; Zhang, J.; Peng, L.; Mushtaq, M.A.; Yasin, G. Developing epoxy-based anti-corrosion functional nanocomposite coating with CaFe-Tolyl-triazole layered double hydroxide@g-C3N4 as nanofillers on Q235 steel substrate against NaCl corrosive environment. Chem. Eng. J. 2022, 450, 137624. [Google Scholar] [CrossRef]
- Li, H.; Qiang, Y.; Zhao, W.; Zhang, S. 2-Mercaptobenzimidazole-inbuilt metal-organic-frameworks modified graphene oxide towards intelligent and excellent anti-corrosion coating. Corros. Sci. 2021, 191, 109715. [Google Scholar] [CrossRef]
- Liu, Y.; Shi, W.; Li, L.; Wei, W.; Wang, H.; Xu, Y.; Yang, X.; Dong, S. “One Stone Three Birds”: A self-healing polyurethane multifunctional composite coating with favorable photothermal effect, anti-corrosion and anti-cavitation. Appl. Surf. Sci. 2026, 718, 164948. [Google Scholar] [CrossRef]
- Kiani, S.; Haddadi-Asl, V.; Khosravi, A.; Eivaz Mohammadloo, H.; Ahmadi, H. Designing a smart polyurethane anti-corrosion coating loaded with APTES/IMZ modified halloysite nanotubes. Surf. Coat. Technol. 2024, 492, 131179. [Google Scholar] [CrossRef]
- Wang, L.; Li, S.N.; Fu, J.J. Self-healing anti-corrosion coatings based on micron-nano containers with different structural morphologies. Prog. Org. Coat. 2023, 175, 107381. [Google Scholar] [CrossRef]
- Nawaz, M.; Yusuf, N.; Habib, S.; Shakoor, R.A.; Ubaid, F.; Ahmad, Z.; Kahraman, R.; Mansour, S.; Gao, W. Development and properties of polymeric nanocomposite coatings. Polymers 2019, 11, 852. [Google Scholar] [CrossRef] [PubMed]
- Attaei, M.; Calado, L.M.; Morozov, Y.; Taryba, M.G.; Shakoor, R.A.; Kahraman, R.; Marques, A.C.; Montemor, M.F. Smart epoxy coating modified with isophorone diisocyanate microcapsules and cerium organophosphate for multilevel corrosion protection of carbon steel. Prog. Org. Coat. 2020, 147, 105864. [Google Scholar] [CrossRef]
- Ramezanzadeh, B.; Akbarian, M.; Ramezanzadeh, M.; Mahdavian, M.; Alibakhshi, E.; Kardar, P. Corrosion Protection of Steel with Zinc Phosphate Conversion Coating and Post-Treatment by Hybrid Organic-Inorganic Sol-Gel Based Silane Film. J. Electrochem. Soc. 2017, 164, C224–C230. [Google Scholar] [CrossRef]
- Zhang, Y.; Shao, Y.; Zhang, T.; Meng, G.; Wang, F. The effect of epoxy coating containing emeraldine base and hydrofluoric acid doped polyaniline on the corrosion protection of AZ91D magnesium alloy. Corros. Sci. 2011, 53, 3747–3755. [Google Scholar] [CrossRef]
- Hao, Y.; Liu, F.; Han, E. Inhibitive Behavior and Mechanism of a Ferrite Inhibition Pigment in Epoxy Paints. J. Electrochem. Soc. 2012, 159, C403–C410. [Google Scholar] [CrossRef]
- Behzadnasab, M.; Mirabedini, S.M.; Kabiri, K.; Jamali, S. Corrosion performance of epoxy coatings containing silane treated ZrO2 nanoparticles on mild steel in 3.5% NaCl solution. Corros. Sci. 2011, 53, 89–98. [Google Scholar] [CrossRef]
- Zahidah, K.A.; Kakooei, S.; Ismail, M.C.; Bothi Raja, P. Halloysite nanotubes as nanocontainer for smart coating application: A review. Prog. Org. Coat. 2017, 111, 175–185. [Google Scholar] [CrossRef]
- Hayatdavoudi, H.; Rahsepar, M. Smart inhibition action of layered double hydroxide nanocontainers in zinc-rich epoxy coating for active corrosion protection of carbon steel substrate. J. Alloys Compd. 2017, 711, 560–567. [Google Scholar] [CrossRef]
- Chen, T.; Chen, R.; Jin, Z.; Liu, J. Engineering hollow mesoporous silica nanocontainers with molecular switches for continuous self-healing anticorrosion coating. J. Mater. Chem. A 2015, 3, 9510–9516. [Google Scholar] [CrossRef]
- Blustein, G.; Di Sarli, A.R.; Jaén, J.A.; Romagnoli, R.; Del Amo, B. Study of iron benzoate as a novel steel corrosion inhibitor pigment for protective paint films. Corros. Sci. 2007, 49, 4202–4231. [Google Scholar] [CrossRef]
- Sinko, J. Challenges of chromate inhibitor pigments replacement in organic coatings. Prog. Org. Coat. 2001, 42, 267–282. [Google Scholar] [CrossRef]
- Cao, Y.; Zheng, D.; Li, X.; Lin, J.; Wang, C.; Dong, S.; Lin, C. Enhanced Corrosion Resistance of Superhydrophobic Layered Double Hydroxide Films with Long-Term Stability on Al Substrate. ACS Appl. Mater. Interfaces 2018, 10, 15150–15162. [Google Scholar] [CrossRef] [PubMed]
- Ding, C.; Tai, Y.; Wang, D.; Tan, L.; Fu, J. Superhydrophobic composite coating with active corrosion resistance for AZ31B magnesium alloy protection. Chem. Eng. J. 2019, 357, 518–532. [Google Scholar] [CrossRef]
- Dong, Y.; Wang, F.; Zhou, Q. Protective behaviors of 2-mercaptobenzothiazole intercalated Zn-Al-layered double hydroxide coating. J. Coat. Technol. Res. 2014, 11, 793–803. [Google Scholar] [CrossRef]
- Wang, Y.; Yuan, Z.; Zhang, Z.; Xin, Y.; Fujita, T.; Wei, Y. In situ one-step fabrication of superhydrophobic layered double hydroxide on Al alloys for anti-corrosion. Appl. Surf. Sci. 2022, 593, 153400. [Google Scholar] [CrossRef]
- Wu, H.; Zhang, Y.; Long, S.; Zhang, L.; Jie, X. Tribological behavior of graphene anchored Mg-Al layered double hydroxide film on Mg alloy pre-sprayed Al coating. Appl. Surf. Sci. 2020, 530, 146536. [Google Scholar] [CrossRef]
- Shen, G.; Zhang, L.; Gu, Z.; Zheng, Z.; Liu, Y.; Tan, G.; Jie, X. Zinc aluminum-layered double hydroxide(LDH)-graphene oxide(GO) lubricating and corrosion-resistant composite coating on the surface of magnesium alloy. Surf. Coat. Technol. 2022, 437, 128354. [Google Scholar] [CrossRef]
- Xue, K.; Xing, M.; Chen, T.; Xi, B.; Zhang, H.; Ye, K.; Feng, J.; Qian, W.; Qiu, J.; Liu, X. Mg-Ga layered double hydroxides coating endow magnesium with antibacterial and osteogenic properties for guided-bone regeneration application. J. Mater. 2025, 11, 101071. [Google Scholar] [CrossRef]
- Chen, R.; Xu, Y.; Xie, X.; Li, C.; Zhu, W.; Xiang, Q.; Li, G.; Wu, D.; Li, X.; Wang, L. Synthesis of TiO2 nanotubes/nickel-gallium layered double hydroxide heterostructure for highly-efficient photocathodic anticorrosion of 304 stainless steel. Surf. Coat. Technol. 2021, 424, 127641. [Google Scholar] [CrossRef]
- Akman, A.; Alberta, L.A.; Giraldo-Osorno, P.M.; Turner, A.B.; Hantusch, M.; Palmquist, A.; Trobos, M.; Calin, M.; Gebert, A. Effect of minor gallium addition on corrosion, passivity, and antibacterial behaviour of novel β-type Ti–Nb alloys. J. Mater. Res. Technol. 2023, 25, 4110–4124. [Google Scholar] [CrossRef]
- Chung, Y.; Lee, C.-W. Electrochemistry of Gallium. J. Electrochem. Sci. Technol. 2013, 4, 1–18. [Google Scholar] [CrossRef]
- Rezaeifard, S.; Shahrabi, T.; Ramezanzadeh, B. Emerging Mo-doped MgAl-LDH lamellar/carbon nanotubes as 3D sustainable nanohybrid for designing a durable smart anti-corrosion composite. Surf. Coat. Technol. 2024, 488, 131013. [Google Scholar] [CrossRef]
- Cristoforetti, A.; Rossi, S.; Deflorian, F.; Fedel, M. Unlocking the Potential of Sebacate: Investigating Its Role in the Inhibition of Filiform Corrosion on Organic Coated Steel. Metals 2024, 14, 623. [Google Scholar] [CrossRef]
- Hefter, G.T.; North, N.A.; Tan, S.H. Organic Corrosion Inhibitors in Neutral Solutions; Part 1—Inhibition of Steel, Copper, and Aluminum by Straight Chain Carboxylates. Corrosion 1997, 53, 657–667. [Google Scholar] [CrossRef]
- Rammelt, U.; Koehler, S.; Reinhard, G. Electrochemical characterisation of the ability of dicarboxylic acid salts to the corrosion inhibition of mild steel in aqueous solutions. Corros. Sci. 2011, 53, 3515–3520. [Google Scholar] [CrossRef]
- Lahem, D.; Poelman, M.; Atmani, F.; Olivier, M.G. Synergistic improvement of inhibitive activity of dicarboxylates in preventing mild steel corrosion in neutral aqueous solution. Corros. Eng. Sci. Technol. 2012, 47, 463–471. [Google Scholar] [CrossRef]
- Cristoforetti, A.; Parola, F.; Parrino, F.; Izquierdo, J.; Souto, R.M.; Rossi, S.; Deflorian, F.; Fedel, M. Sebacate intercalated Ca–Al layered double hydroxide pigments for corrosion protection of low carbon steel: Anion exchange and electrochemical properties. Appl. Clay Sci. 2024, 250, 107300. [Google Scholar] [CrossRef]
- Stimpfling, T.; Leroux, F.; Hintze-Bruening, H. Organo-modified layered double hydroxide in coating formulation to protect AA2024 from corrosion. Colloids Surf. A Physicochem. Eng. Asp. 2014, 458, 147–154. [Google Scholar] [CrossRef]
- Wang, X.; Zhu, J.; Lei, Y.; Lei, W. Synthesis and characterization of layered double hydroxides hybrid microcapsules for anticorrosion via self-healing and chloride ion adsorption. Appl. Clay Sci. 2022, 221, 106481. [Google Scholar] [CrossRef]
- Rezaeifard, S.; Shahrabi, T.; Ramezanzadeh, B. Trisodium phosphate-loaded magnesium-aluminum layered double hydroxides/oxidized-multiwalled carbon nanotubes self-assembled 2D-nanohybrid for designing a multifunctional smart epoxy nanocomposite. Appl. Clay Sci. 2024, 254, 107376. [Google Scholar] [CrossRef]
- Swamy, N.K.; Mohana, K.N.S.; Hegde, M.B.; Madhusudana, A.M. Fabrication of 1D graphene nanoribbon and malenized linseed oil-based nanocomposite: A highly impervious bio-based anti-corrosion coating material for mild steel. J. Appl. Electrochem. 2022, 52, 1133–1148. [Google Scholar] [CrossRef]
- Ansell, P.; Berry, L.; Mcgettrick, J.; Searle, J.; Wint, N. Role of Smart-Release Pigments in Preventing Corrosion Driven Cathodic Disbondment of Organically Coated Hot Dip Galvanised Steel. J. Electrochem. Soc. 2023, 170, 011502. [Google Scholar] [CrossRef]
- Ding, W.; Luo, J.-X.; Gu, Q.; Liu, Z.H. Ultrathin 2D ZnGa-borate-LDH nanosheets for boosting dye-sensitized photocatalytic coupled reaction of H2 production with pollutant degradation. Colloids Surf. A Physicochem. Eng. Asp. 2023, 657, 130575. [Google Scholar] [CrossRef]
- Kosynkin, D.V.; Higginbotham, A.L.; Sinitskii, A.; Lomeda, J.R.; Dimiev, A.; Price, B.K.; Tour, J.M. Longitudinal unzipping of carbon nanotubes to form graphene nanoribbons. Nature 2009, 458, 872–876. [Google Scholar] [CrossRef]
- ASTM B117-19; Standard Practice for Operating Salt Spray (Fog) Apparatus. ASTM International: West Conshohocken, PA, USA, 2019; pp. 1–15. [CrossRef]
- Souza, L.H.d.O.; Cristoforetti, A.; Cotting, F.; Reis da Costa Campos, W.; Rossi, S.; Fedel, M. Sebacate-Intercalated CaAl-LDH Pigments for Corrosion Protection of Aluminum Alloy. ACS Omega 2025, 10, 59771–59781. [Google Scholar] [CrossRef]
- Nguyen, D.T.; To, H.T.X.; Gervasi, J.; Paint, Y.; Gonon, M.; Olivier, M.G. Corrosion inhibition of carbon steel by hydrotalcites modified with different organic carboxylic acids for organic coatings. Prog. Org. Coat. 2018, 124, 256–266. [Google Scholar] [CrossRef]
- Caballero, D.; Beltrán-Cobos, R.; Tavares, F.; Cruz-Yusta, M.; Granados, L.S.; Sánchez-Moreno, M.; Pavlovic, I. The Inhibitive Effect of Sebacate-Modified LDH on Concrete Steel Reinforcement Corrosion. ChemEngineering 2022, 6, 72. [Google Scholar] [CrossRef]
- Huang, C.; Zhang, M.; Li, C. A novel non-enzymatic electrochemical glucose sensors based on graphene oxide nanoribbons: Tracking energy expenditure and nutritional intake in sports. Alex. Eng. J. 2024, 89, 184–194. [Google Scholar] [CrossRef]
- Habibpour, S.; Um, J.G.; Jun, Y.-S.; Bhargava, P.; Park, C.B.; Yu, A. Structural Impact of Graphene Nanoribbon on Mechanical Properties and Anti-corrosion Performance of Polyurethane Nanocomposites. Chem. Eng. J. 2021, 405, 126858. [Google Scholar] [CrossRef]
- Heiba, A.R.; Abou Shahba, R.M.; Dhmees, A.S.; Taher, F.A.; El Sawy, E.N. Graphene oxide nanoribbons (GONRs) as pH-tolerant electrodes for supercapacitors: Effect of charge carriers and loading. J. Energy Storage 2024, 83, 110762. [Google Scholar] [CrossRef]
- Heiba, A.R.; Omran, M.M.; Abou Shahba, R.M.; Dhmees, A.S.; Taher, F.A.; El Sawy, E. Compositing LaSrMnO3 perovskite and graphene oxide nanoribbons for highly stable asymmetric electrochemical supercapacitors. Mater. Sci. Energy Technol. 2025, 8, 82–95. [Google Scholar] [CrossRef]
- Wu, S.; Liang, H.; Zhang, Z.; Zhang, Q.; Han, Q.; Wang, J.; Gao, M.; Fan, H.; Yang, J.; Lang, J. The photocatalytic degradation and mechanism of rhodamine B by Zn–Al layered double hydroxide. Opt. Mater. 2022, 131, 112636. [Google Scholar] [CrossRef]
- Singh, K.S.; Sawant, S.G. Identification of CaCO3 polymorphs of shellfish by FTIR spectroscopy and evaluation of metals adsorption by powdered exoskeleton shell. Indian J. Geo-Marine Sci. 2022, 51, 304–309. [Google Scholar] [CrossRef]
- Dehghani, A.; Sanaei, Z.; Fedel, M.; Ramezanzadeh, M.; Mahdavian, M.; Ramezanzade, B. Fabrication of an intelligent anti-corrosion silane film using a MoO42− loaded Micro/mesoporous ZIF67-MOF/multi-walled-CNT/APTES core-shell nano-container. Colloids Surf. A Physicochem. Eng. Asp. 2023, 656, 130511. [Google Scholar] [CrossRef]
- Majidi, R.; Danaee, I.; Vrsalović, L.; Zarei, D. Development of a smart anticorrosion epoxy coating containing a pH-sensitive GO/MOF nanocarrier loaded with 2-mercaptobenzothiazole corrosion inhibitor. Mater. Chem. Phys. 2023, 308, 128291. [Google Scholar] [CrossRef]
- Stanimirova, T.; Piperov, N.; Petrova, N.; Kirov, G. Thermal evolution of Mg-Al-CO3 hydrotalcites. Clay Miner. 2004, 39, 177–191. [Google Scholar] [CrossRef]
- Zhou, L.; Zhang, Q.; Feng, X.; Rui, Z.; Guo, W.; Zhang, Y.; Li, W.; Li, Z. Chloride ion-responsive active protection epoxy composite coatings realized by 2-Mercaptobenzimidazole and 2-Mercaptobenzothiazole intercalated Mg-Al layered double hydroxides. Corros. Sci. 2024, 234, 112144. [Google Scholar] [CrossRef]
- Jaśkaniec, S.; Hobbs, C.; Seral-Ascaso, A.; Coelho, J.; Browne, M.P.; Tyndall, D.; Sasaki, T.; Nicolosi, V. Low-temperature synthesis and investigation into the formation mechanism of high quality Ni-Fe layered double hydroxides hexagonal platelets. Sci. Rep. 2018, 8, 4179. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Ma, R.; Wang, C.; Liang, J.; Liu, X.; Zhou, K.; Sasaki, T. A superlattice of alternately stacked Ni-Fe hydroxide nanosheets and graphene for efficient splitting of water. ACS Nano 2015, 9, 1977–1984. [Google Scholar] [CrossRef]
- Rammelt, U.; Reinhard, G. Application of electrochemical impedance spectroscopy (EIS) for characterizing the corrosion-protective performance of organic coatings on metals. Prog. Org. Coat. 1992, 21, 205–226. [Google Scholar] [CrossRef]
- Guo, H.; Yang, C.; Sun, H.; Xiang, N.; Li, C.; Wang, C. Silane coupling agent modified layered double hydroxide/graphene oxide preparation of intelligent anticorrosive coating. Surf. Coat. Technol. 2023, 467, 129728. [Google Scholar] [CrossRef]
- Su, Y.; Qiu, S.; Wei, J.; Zhu, X.; Zhao, H.; Xue, Q. Sulfonated polyaniline assisted hierarchical assembly of graphene-LDH nanohybrid for enhanced anticorrosion performance of waterborne epoxy coatings. Chem. Eng. J. 2021, 426, 131269. [Google Scholar] [CrossRef]
- Wang, J.; Cao, Y.; Xue, J.; Zhang, X.; Liang, Y.; Chen, K.; Huang, C.; Zheng, D. A comparative experimental and theoretical calculation study of CaAl-LDH modified with various aromatic inhibitors for corrosion protection study in epoxy coatings. Corros. Sci. 2024, 231, 111994. [Google Scholar] [CrossRef]
- Shoar Abouzari, M.R.; Berkemeier, F.; Schmitz, G.; Wilmer, D. On the physical interpretation of constant phase elements. Solid State Ion. 2009, 180, 922–927. [Google Scholar] [CrossRef]
- Harsha, Y.M.; Mohana, K.N.S.; Sunilkumar, M.C.; Hithesh, M.C.; Sreelakshmi, M.; Madhusudhana, A.M. Syntheses of diphenolic resin based anti-corrosion coating material and reinforce its performance through MWCNT-Ag and MWCNT-Ag/PANI nanofillers. Surf. Coat. Technol. 2024, 485, 130871. [Google Scholar] [CrossRef]
- Guan, J.; Du, X. Utilizing the synergistic effect of CNT and graphene hybrid fillers to improve the anticorrosion performance of Zn/epoxy coatings. Mater. Today Commun. 2025, 49, 114029. [Google Scholar] [CrossRef]
- AitAghzzaf, A.; Zarki, Y.; Rhouta, B.; Khalil, A.; Veys-Renaux, D.; Majdoub, H.; Rocca, E. Intercalation of decanoate anions (C10) in layered double hydroxide Mg–Al and its application as controlled-release corrosion inhibitor of steel. Surf. Coatings Technol. 2024, 489, 131055. [Google Scholar] [CrossRef]
- Li, J.; Wang, D.; Hou, L.; Zhang, S.; Wu, P.; Zhang, Y.; Liu, B.; Wei, Y. Enhancing the corrosion resistance of the epoxy coating using CaAl LDH intercalated with L-cysteine and its derivatives as a pigment on steel substrate. Prog. Org. Coat. 2024, 193, 108527. [Google Scholar] [CrossRef]
- Mukunda Murthy, K.N.; Shivaswamy, M.B.; Sanjana, M.S.; Kavyashree, M.B.; Sangamesha, M.A.; Deepak Raj, G.R.; Nagendra Prasad, H.S. CuO@Bi2MoO6 heterojunction nanocomposite-reinforced epoxy coating: Enhanced anti-corrosion performance on low-carbon steel. Mater. Today Commun. 2025, 49, 113790. [Google Scholar] [CrossRef]
- ISO 2409; Paints and Varnishes—Cross-Cut Test. ISO: Geneva, Switzerland, 2020.
- Zhang, Q.; Yu, Y.; Li, J.; Yin, C.; Tian, F.; Liu, J.; Zhou, J. Synergistic Corrosion Inhibition and UV Protection via TTA-Loaded LDH Nanocontainers in Epoxy Coatings. Coatings 2025, 15, 505. [Google Scholar] [CrossRef]
- STN EN ISO 15184; Paints and Varnishes-Determination of Film Hardness by Pencil Test. Slovak Office of Standards, Metrology and Testing: Bratislava, Slovakia, 2021.

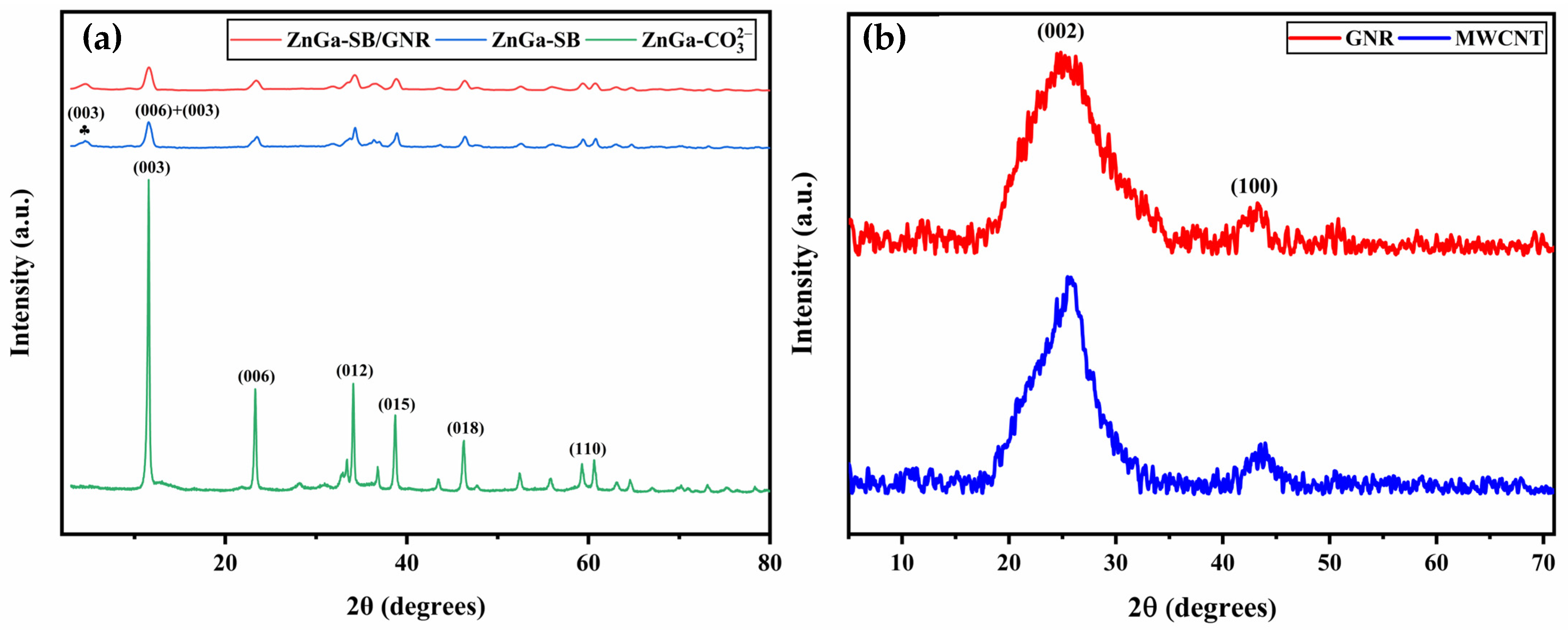












| Nanoparticle | β (Radian) | θ (Radian) | D (nm) | ε | δ (1015 m−2) | γ |
|---|---|---|---|---|---|---|
| ZnGa- | 0.0046 | 0.1007 | 30.075 | 0.0114 | 1.12 | 0.795 |
| ZnGa-SB | 0.0199 | 0.0495 | 06.970 | 0.1004 | 21.00 | 1.136 |
| 0.0124 | 0.0975 | 11.189 | 0.0318 | 08.11 | 0.808 | |
| ZnGa-SB/GNR | 0.0205 | 0.0487 | 06.767 | 0.1050 | 22.30 | 1.145 |
| 0.0138 | 0.0965 | 10.061 | 0.0357 | 10.00 | 0.812 |
| Materials | θ (Radian) | Interplanar Distance (Å) | FWHM (Radian) | τ (Å) | Lx |
|---|---|---|---|---|---|
| MWCNT | 0.224 | 3.46 | 0.1198 | 11.86 | 3.427 |
| GNR | 0.378 | 3.58 | 0.1457 | 9.73 | 2.714 |
| Materials | ID/IG | La (nm) |
|---|---|---|
| MWCNT | 1.1 | 35 |
| GNR | 1.4 | 27.5 |
| Coatings | Immersion Time (h) | CPEc (S sn cm−2) | Cc (F cm2) | Rc (Ω cm2) | CPEdl (S sn cm−2) | Cdl (F cm2) | Rct (Ω cm2) | χ2 (Chi-Square) | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Yo | n | Yo | n | |||||||
| PU | 0 | 9.57 × 10−11 | 0.96 | 8.32 × 10−10 | 3.61 × 107 | 2.70 × 10−10 | 0.71 | 1.13 × 10−9 | 1.23 × 1011 | 5.96 × 10−4 |
| 672 | 1.80 × 10−10 | 0.94 | 1.19 × 10−10 | 8.54 × 106 | 7.19 × 10−10 | 0.53 | 2.08 × 10−9 | 4.61 × 109 | 1.44 × 10−3 | |
| 1344 | 1.85 × 10−10 | 0.94 | 1.26 × 10−10 | 1.27 × 107 | 6.18 × 10−10 | 0.53 | 2.05 × 10−9 | 6.25 × 109 | 1.45 × 10−3 | |
| 2016 | 1.87 × 10−10 | 0.94 | 1.29 × 10−10 | 1.66 × 107 | 6.92 × 10−10 | 0.50 | 4.14 × 10−9 | 8.65 × 109 | 3.12 × 10−4 | |
| 2688 | 1.56 × 10−10 | 0.95 | 1.01 × 10−10 | 1.51 × 106 | 6.28 × 10−10 | 0.51 | 4.83 × 10−9 | 1.33 × 1010 | 2.85 × 10−4 | |
| PU/ZnGa-SB | 0 | 8.67 × 10−11 | 0.96 | 6.77 × 10−11 | 3.02 × 107 | 2.54 × 10−10 | 0.67 | 8.61 × 10−10 | 4.69 × 1010 | 4.45 × 10−4 |
| 672 | 1.96 × 10−10 | 0.93 | 9.50 × 10−11 | 3.40 × 105 | 3.12 × 10−9 | 0.48 | 1.08 × 10−10 | 1.44 × 107 | 3.98 × 10−3 | |
| 1344 | 8.83 × 10−11 | 1.00 | 8.83 × 10−11 | 5.52 × 104 | 4.98 × 10−9 | 0.62 | 5.00 × 10−10 | 4.72 × 106 | 6.51 × 10−4 | |
| 2016 | 2.24 × 10−10 | 0.94 | 1.13 × 10−10 | 9.91 × 104 | 5.94 × 10−9 | 0.61 | 6.17 × 10−10 | 4.87 × 106 | 3.78 × 10−3 | |
| 2688 | 3.46 × 10−10 | 0.93 | 1.43 × 10−10 | 2.40 × 104 | 1.29 × 10−8 | 0.66 | 1.16 × 10−9 | 7.21 × 105 | 2.05 × 10−3 | |
| PU/GNR | 0 | 9.39 × 10−11 | 0.96 | 7.36 × 10−11 | 3.08 × 107 | 2.83 × 10−10 | 0.69 | 1.07 × 10−9 | 6.84 × 1010 | 1.13 × 10−3 |
| 672 | 2.53 × 10−10 | 0.91 | 9.02 × 10−11 | 1.17 × 105 | 1.55 × 10−8 | 0.47 | 5.32 × 10−9 | 2.50 × 107 | 3.87 × 10−3 | |
| 1344 | 1.97 × 10−10 | 0.94 | 1.09 × 10−10 | 4.89 × 105 | 2.57 × 10−9 | 0.45 | 9.41 × 10−11 | 2.60 × 107 | 2.33 × 10−3 | |
| 2016 | 1.85 × 10−10 | 0.94 | 1.03 × 10−10 | 5.86 × 105 | 2.03 × 10−9 | 0.47 | 8.39 × 10−11 | 2.92 × 107 | 4.47 × 10−4 | |
| 2688 | 1.87 × 10−10 | 0.94 | 1.04 × 10−10 | 5.71 × 105 | 2.12 × 10−9 | 0.46 | 8.22 × 10−11 | 2.96 × 107 | 2.39 × 10−4 | |
| PU/ZnGa-SB/GNR | 0 | 8.03 × 10−11 | 0.97 | 6.72 × 10−11 | 4.01 × 107 | 2.29 × 10−10 | 0.68 | 9.50 × 10−10 | 8.97 × 1010 | 7.12 × 10−4 |
| 672 | 1.12 × 10−10 | 0.96 | 8.85 × 10−11 | 3.11 × 107 | 2.47 × 10−9 | 0.39 | 7.32 × 10−10 | 1.86 × 108 | 2.05 × 10−3 | |
| 1344 | 1.32 × 10−10 | 0.95 | 9.13 × 10−11 | 6.86 × 106 | 6.58 × 10−9 | 0.49 | 2.28 × 10−8 | 5.01 × 108 | 6.02 × 10−4 | |
| 2016 | 1.34 × 10−10 | 0.95 | 9.35 × 10−11 | 7.94 × 106 | 5.96 × 10−9 | 0.53 | 1.73 × 10−8 | 5.59 × 108 | 3.32 × 10−4 | |
| 2688 | 1.39 × 10−10 | 0.94 | 8.94 × 10−11 | 7.13 × 106 | 5.53 × 10−9 | 0.61 | 9.65 × 10−9 | 4.32 × 108 | 4.15 × 10−3 | |
| Classification | Description | Appearance |
|---|---|---|
| 0 | The edges of the cuts are completely smooth, and none of the squares of the lattice is detached. |  |
| 1 | Detachment of small flakes of the coating at the intersections of the cuts. A cross-cut area of no more than 5% is affected. |  |
| 2 | The coating has flaked along the edges and/or at the intersections of the cuts. A cross-cut area greater than 5%, but not greater than 15%, is affected. |  |
| 3 | The coating has flaked along the edges of the cuts partly or wholly in large ribbons, and/or it has flaked partly or wholly on different parts of the squares. A cross-cut area greater than 15%, but not greater than 35%, is affected. |  |
| 4 | The coating has flaked along the edges of the cuts in large ribbons, and/or some squares have partially or wholly detached. A cross-cut area greater than 35%, but not greater than 65%, is affected. |  |
| 5 | Any degree of flaking that cannot even be classified by classification 4. | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Mukkati Praveena, U.; Fedel, M.; Rossi, S. Rational Design of ZnGa-Sebacate/Graphene Nanoribbon Synergy for Effective Anticorrosive Polyurethane Coatings. Processes 2026, 14, 400. https://doi.org/10.3390/pr14030400
Mukkati Praveena U, Fedel M, Rossi S. Rational Design of ZnGa-Sebacate/Graphene Nanoribbon Synergy for Effective Anticorrosive Polyurethane Coatings. Processes. 2026; 14(3):400. https://doi.org/10.3390/pr14030400
Chicago/Turabian StyleMukkati Praveena, Ujwal, Michele Fedel, and Stefano Rossi. 2026. "Rational Design of ZnGa-Sebacate/Graphene Nanoribbon Synergy for Effective Anticorrosive Polyurethane Coatings" Processes 14, no. 3: 400. https://doi.org/10.3390/pr14030400
APA StyleMukkati Praveena, U., Fedel, M., & Rossi, S. (2026). Rational Design of ZnGa-Sebacate/Graphene Nanoribbon Synergy for Effective Anticorrosive Polyurethane Coatings. Processes, 14(3), 400. https://doi.org/10.3390/pr14030400







