The Evolution Law of Molecular Structure of Vitrain and Durain During Low–Medium Coalification
Abstract
1. Introduction
2. Materials and Methods
2.1. Coal Sample Preparation
2.2. Construction of References
2.2.1. Fourier Transform Infrared Spectroscopy Testing
2.2.2. X-Ray Diffraction Testing
2.2.3. Nuclear Magnetic Resonance Carbon-13 Spectroscopy Testing
2.2.4. X-Ray Photoelectron Spectroscopy Testing
3. Results and Discussion
3.1. FTIR Analysis
3.1.1. Aromatic Structural Changes
3.1.2. Aliphatic Structural Changes
3.1.3. Oxygen Functional Group Changes
3.1.4. Hydroxyl Functional Group Changes
3.2. XRD Analysis
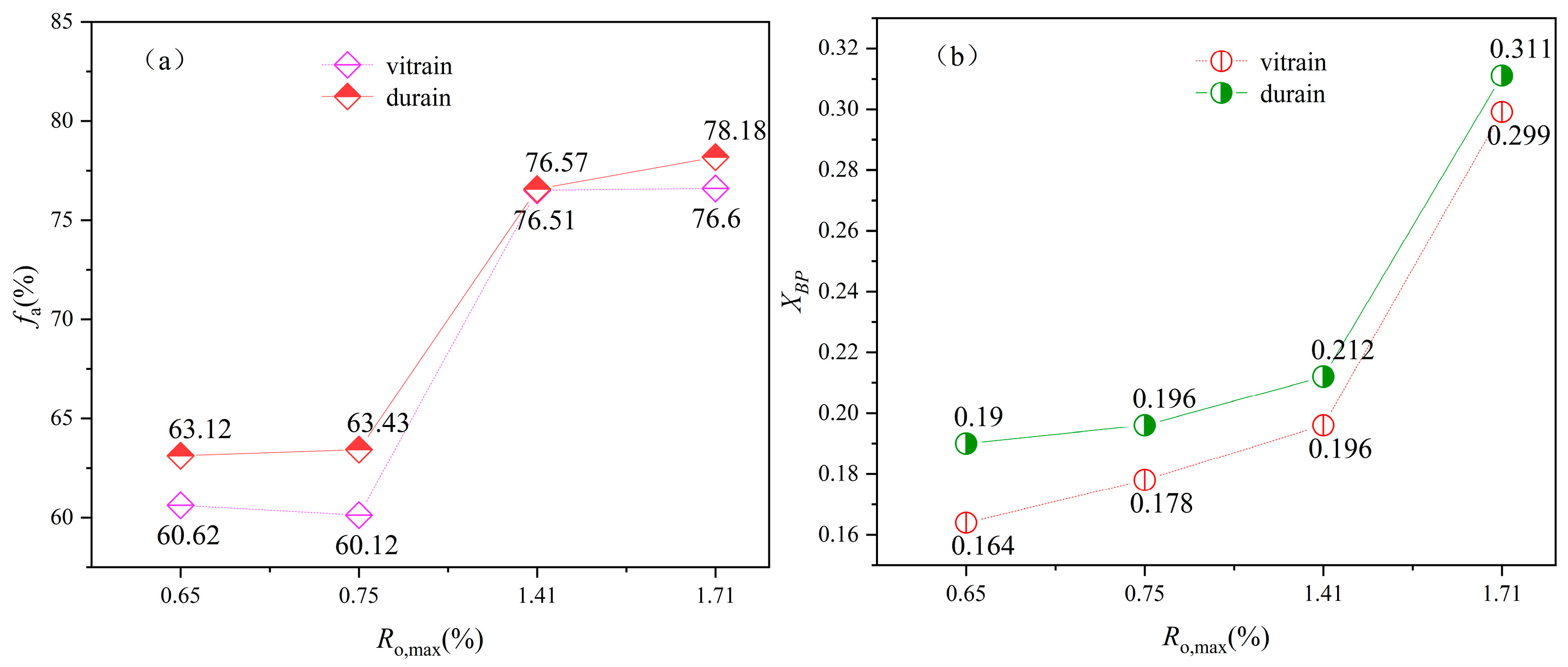
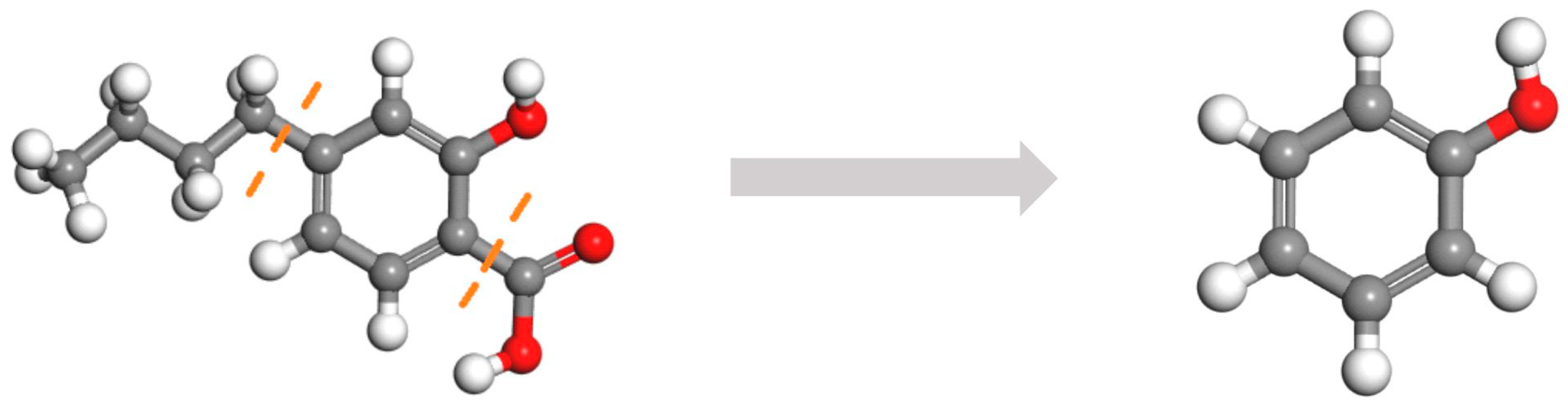
3.3. 13C-NMR Analysis
3.4. XPS Analysis
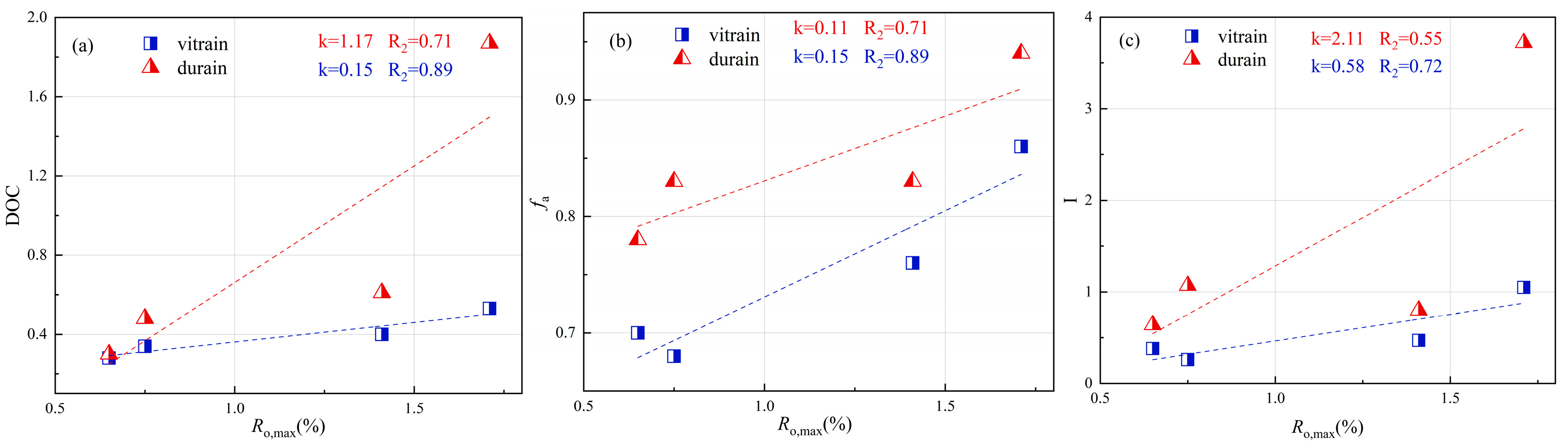
3.5. Evolution of the Coalification Stage
4. Conclusions
- (1)
- Vitrain predominantly exhibits trisubstitution, with trisubstituted aromatics comprising a larger proportion than in durain. In contrast, durain transitions from trisubstitution to pentasubstitution. Among oxygen functional groups, C–O bonds are the most abundant, while C=O and COOH groups are less common. Durain generally contains a higher concentration of oxygen functional groups compared to vitrain. The CH2/CH3 ratio in vitrain is consistently higher than in durain. Durain also displays greater aromaticity, higher condensation, and increased aromatic carbon content, suggesting that vitrain contains longer alkyl side chains, while durain is characterized by more extensively developed aromatic structures.
- (2)
- The d002 size of the vitrain decreased from 3.82 to 3.47, while that of the durain reduced from 3.52 to 3.40. Both exhibited a progressive decline, with the vitrain showing a more pronounced reduction compared to the durain. These findings suggest that the lateral extension of the microcrystalline structure in durain is more developed, indicative of a denser network of molecular interactions and tighter interconnections.
- (3)
- During the structural evolution of the carbon skeleton, progressive coal metamorphism leads to a marked reduction in the aliphatic peak area, accompanied by a gradual increase in both the area and intensity of the aromatic peak. Concurrently, the bridging carbon ratio (XBP) exhibits a systematic rise with increasing metamorphic rank. As a result, the proportion of aromatic carbon in the coal matrix steadily increases, whereas the contribution of aliphatic carbon diminishes as coal maturity advances.
- (4)
- The X-ray photoelectron spectroscopy (XPS) analysis of carbon (C), nitrogen (N), and oxygen (O) revealed that carbon predominantly exists in C-C/C-H bonding configurations, followed by C-O groups, with carboxyl groups (–COOH) being the least abundant. Notably, the content of C-C/C-H bonds in durain is typically lower than that in vitrain.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| Ro,max | Maximum Reflectance of Vitrinite |
| Mad | Moisture in the Air-Dried Basis |
| Aad | Ash in the Air-Dried Basis |
| Vad | Volatile Matter in the Air-Dried Basis |
| FCad | Fixed Carbon in the Air-Dried Basis |
| Cdaf | Carbon in the Dry Ash-Free Basis |
| Hdaf | Hydrogen in the Dry Ash-Free Basis |
| Odaf | Oxygen in the Dry Ash-Free Basis |
| Ndaf | Nitrogen in the Dry Ash-Free Basis |
References
- Wang, T.; Liu, F.; Zhao, X.; Deng, X.; Yuan, T.; Ren, S. Reflection on China’s coal resource guarantee capacity and exploration work under the background of “double carbon”. Coal Sci. Technol. 2023, 51, 1–8. [Google Scholar] [CrossRef]
- Özgen, K.C.; Ruiz, F.A.; Cotè, M.; Phipps, S.J.I.J.o.C.G. Coal mine methane: A review of capture and utilization practices with benefits to mining safety and to greenhouse gas reduction. Int. J. Coal Geol. 2011, 86, 121–156. [Google Scholar] [CrossRef]
- Qin, Y.; Moore, T.A.; Shen, J.; Yang, Z.; Shen, Y. Resources and geology of coalbed methane in China: A review. Int. Geol. Rev. 2017, 60, 777–812. [Google Scholar] [CrossRef]
- Zou, C.; Li, S.; Liu, H.; Feng, M.J.P.E. Revolution and significance of “Green Energy Transition” in the context of new quality productive forces: A discussion on theoretical understanding of “Energy Triangle”. Pet. Explor. Dev. 2024, 51, 1611–1627. [Google Scholar] [CrossRef]
- Tao, G.; Wang, Z.; Jin, Y.; Wang, H.; Xia, D.; Pan, J. Multiscale Pore–Fracture Structure Characteristics of Deep Coal Reservoirs in the Eastern Margin of the Ordos Basin, China. Nat. Resour. Res. 2025, 34, 1557–1584. [Google Scholar] [CrossRef]
- Wang, Z.; Xiong, J.; Zhang, Y.; Tao, G.; Pan, J.; Niu, Q. Investigation of Permeability Stress Induced Damage Evolution of Shallow and Deep Coal Reservoirs in the Junggar Basin, China. Rock Mech. Rock Eng. 2025. [Google Scholar] [CrossRef]
- Li, S.K.; Zhu, Y.M.; Wang, Y.; Liu, J. The Chemical and Alignment Structural Properties of Coal: Insights from Raman, Solid-State 13C NMR, XRD, and HRTEM Techniques. Acs Omega 2021, 6, 11266–11279. [Google Scholar] [CrossRef]
- Zheng, C.; Ma, D.M.; Chen, Y.; Gao, Z.; Teng, J.X. Pore structure of different macroscopically distinguished components within low-rank coals and its methane desorption characteristics. Fuel 2021, 293, 120465. [Google Scholar] [CrossRef]
- Li, X.; Zeng, F.G.; Wang, W.; Dong, K.; Cheng, L.Y. FTIR characterization of structural evolution in low-middle rank coals. J. China Coal Soc. 2015, 40, 2900–2908. [Google Scholar] [CrossRef]
- Yu, C.M.; Zhang, N.; Teng, H.P. Investigation of Different Structures of Coals Through FTIR and Raman Techniques. Spectrosc. Spectr. Anal. 2021, 41, 2050–2056. [Google Scholar]
- Li, W.; Zhu, Y.M. Structural Characteristics of Coal Vitrinite during Pyrolysis. Energy Fuels 2014, 28, 3645–3654. [Google Scholar] [CrossRef]
- Zhou, H.; Wu, C.F.; Pan, J.N.; Wang, Z.Z.; Niu, Q.H.; Du, M.Y. Research on Molecular Structure Characteristics of Vitrinite and Inertinite from Bituminous Coal with FTIR, Micro-Raman, and XRD Spectroscopy. Energy Fuels 2021, 35, 1322–1335. [Google Scholar] [CrossRef]
- Wang, Q.; Zhang, J.P.; Li, H.N.; Zhang, H.; Bai, H.C.; Guo, Q.J. Exploring molecular structure characteristics and chemical index of Qinghua bituminous coal: A comprehensive insight from single molecule of macerals to particles with various sizes. Powder Technol. 2022, 396, 36–49. [Google Scholar] [CrossRef]
- Yang, Z.Q.; Liu, Y.; Zhu, G.W.; Liu, J.H.; Xu, R.; Shi, D.J. Molecule structure and ultrasonic wave velocity of different ranking coals: An experimental perspective. Fuel 2022, 312, 122913. [Google Scholar] [CrossRef]
- Chen, Y.; Ma, D.M.; Guo, C.; Yang, F.; Mu, T.; Gao, Z. An Experimental Study on the Conductivity Changes in Coal during Methane Adsorption-Desorption and their Influencing Factors. Acta Geol. Sin.-Engl. Ed. 2019, 93, 704–717. [Google Scholar] [CrossRef]
- Jia, J.Z.; Wang, D.M.; Li, B. Study on molecular structure characteristic and optimization of molecular model construction of coal with different metamorphic grade. J. Mol. Struct. 2024, 1295, 136655. [Google Scholar] [CrossRef]
- Yin, T.T.; Liu, D.M.; Cai, Y.D.; Liu, Z.H.; Gutierrez, M. A new constructed macromolecule-pore structure of anthracite and its related gas adsorption: A molecular simulation study. Int. J. Coal Geol. 2020, 220, 103415. [Google Scholar] [CrossRef]
- GB/T 212-2008; Proximate Analysis of Coal. Standardization Administration of the People’s Republic of China: Beijing, China, 2008.
- GB/T 31391-2015; Ultimate Analysis of Coal. Standardization Administration of the People’s Republic of China: Beijing, China, 2015.
- GB/T 6948-2008; Method of Determining Microscopically the Reflectance of Vitrinite in Coal. National Technical Committee on Coal Standardization of China: Xi’an, China, 2008.
- Zhao, J.; Shi, Q.M.; Wang, S.M.; Mi, Y.C.; Zhao, H.C.; Ji, R.J. Study on molecular structural heterogeneity of tar-rich coal based on micro-FTIR. Spectrochim. Acta Part A-Mol. Biomol. Spectrosc. 2025, 330, 125749. [Google Scholar] [CrossRef]
- Gao, F.; Jia, Z.; Cui, Z.; Li, Y.D.; Jiang, H. Evolution of macromolecular structure during coal oxidation via FTIR, XRD and Raman. Fuel Process. Technol. 2024, 262, 108114. [Google Scholar] [CrossRef]
- Xu, C.; Li, H.; Lu, J.; Lu, Y.; Shi, S.; Ye, Q. An investigation into the modification of microwave-assisted oxidation in the macromolecular structure of coal via XRD and Raman spectroscopy. Fuel 2023, 338, 127192. [Google Scholar] [CrossRef]
- Feng, B.; Bhatia, S.K.; Barry, J.C. Structural ordering of coal char during heat treatment and its impact on reactivity. Carbon 2002, 40, 481–496. [Google Scholar] [CrossRef]
- Lv, T.; Fang, M.X.; Zeng, X.; Yan, J.Q.; Huang, Y.; Cen, J.M. Carbon Structure of Coal from the CP/MAS 13C NMR Spectra: Effect of Contact Time and Potential Quantitative Modification. Energy Fuels 2024, 38, 3740–3754. [Google Scholar] [CrossRef]
- Baysal, M.; Yürüm, A.; Yildiz, B.; Yürüm, Y. Structure of some western Anatolia coals investigated by FTIR, Raman, 13C solid state NMR spectroscopy and X-ray diffraction. Int. J. Coal Geol. 2016, 163, 166–176. [Google Scholar] [CrossRef]
- Zhuo, X.Y.; Su, J.S.; Xu, H.L.; Wang, L.; Wang, W.F.; Zhang, B. Synergistically effective flotation enrichment of vitrinite by Na removal for high-Na high-inertinite low-rank Zhundong coal. J. Clean. Prod. 2023, 428, 139433. [Google Scholar] [CrossRef]
- Ping, A.; Xia, W.C.; Peng, Y.L.; Xie, G.Y. Comparative filtration and dewatering behavior of vitrinite and inertinite of bituminous coal: Experiment and simulation study. Int. J. Min. Sci. Technol. 2021, 31, 233–240. [Google Scholar] [CrossRef]
- Liu, Y.; Zhu, Y.M.; Liu, S.M.; Chen, S.B.; Li, W.; Wang, Y. Molecular structure controls on micropore evolution in coal vitrinite during coalification. Int. J. Coal Geol. 2018, 199, 19–30. [Google Scholar] [CrossRef]
- Shan, C.A.; Zhang, T.S.; Liang, X.; Zhang, Z.; Wang, M.; Zhang, K. On the fundamental difference of adsorption-pores systems between vitrinite- and inertinite-rich anthracite derived from the southern Sichuan basin, China. J. Nat. Gas Sci. Eng. 2018, 53, 32–44. [Google Scholar] [CrossRef]
- Dwivedi, A.; Dwivedi, A.; Kumar, A. Qualitative surface characterization of Indian Permian coal using XPS and FTIR. Int. J. Coal Prep. Util. 2023, 43, 1152–1163. [Google Scholar] [CrossRef]
- Ju, Y.W.; Jiang, B.; Hou, Q.I.; Wang, G.I. FTIR spectroscopic study on the stress effect of compositions of macromolecular structure in tectonically deformed coals. Guang Pu Xue Yu Guang Pu Fen Xi Guang Pu 2005, 25, 1216–1220. [Google Scholar]
- Li, J.Z.; Guo, X.; Panchal, B.; Wang, J.X.; Guo, W.M.; Liu, B.J. Quantitative analysis of molecular structure characterization of different liptinite-rich coals using FTIR spectroscopy. Infrared Phys. Technol. 2024, 141, 105458. [Google Scholar] [CrossRef]
- Xu, C.F.; Hu, S.; Xiang, J.; Zhang, L.Q.; Sun, L.S.; Shuai, C. Interaction and kinetic analysis for coal and biomass co-gasification by TG-FTIR. Bioresour. Technol. 2014, 154, 313–321. [Google Scholar] [CrossRef]
- Chen, A.K.; Zhai, C.; Yu, X.; Xu, J.Z.; Zheng, Y.F.; Zhu, X.Y. Quantitative Analysis of the Effect of NaClO Stimulation in Anthracite and Bituminous Coal with FTIR and LF-NMR. Energy Fuels 2024, 38, 2018–2032. [Google Scholar] [CrossRef]
- Dong, T.; Murata, S.; Miura, M.C.; Nomura, M.; Nakamura, K.T. Computer-aided molecular design study of coal model molecules. 3. Density simulation for model structures of bituminous Akabira coal. Energy Fuels 1993, 7, 1123–1127. [Google Scholar] [CrossRef]
- Shukla, A.; Prasad, A.K.; Mishra, S.; Vinod, A.; Varma, A.K. Rapid Estimation of Sulfur Content in High-Ash Indian Coal Using Mid-Infrared FTIR Data. Minerals 2023, 13, 634. [Google Scholar] [CrossRef]
- Isah, U.A.; Rashid, M.I.; Lee, S.; Kiman, S.; Iyodo, H.M. Correlations of coal rank with the derived Fourier Transform Infra-Red (FTIR) spectroscopy structural parameters: A review. Infrared Phys. Technol. 2024, 141, 105456. [Google Scholar] [CrossRef]
- Bin, L.; Wei, Z.; Xie, Z.H.; Chen, X.J.; Yong, C. FTIR and XRD microscopic characterisation of coal samples with different degrees of metamorphism. J. Mol. Struct. 2024, 1309, 138270. [Google Scholar] [CrossRef]
- Chang, H.Z.; Zeng, F.G.; Li, W.Y.; Li, J.; Jia, J.B.; Xie, K.C. Micro-FTIR study on structure of macerals from Jurassic coals in Northwestern China. Spectrosc. Spectr. Anal. 2008, 28, 1535–1538. [Google Scholar] [CrossRef]
- Domazetis, G.; James, B.D.; Liesegang, J. Computer molecular models of low-rank coal and char containing inorganic complexes. J. Mol. Model. 2008, 14, 581–597. [Google Scholar] [CrossRef]
- Zhou, Y.; Albijanic, B.; Wang, Y.L.; Yang, J.G. Characterizing surface properties of oxidized coal using FTIR and contact angle measurements. Energy Sources Part A-Recovery Util. Environ. Eff. 2018, 40, 1559–1564. [Google Scholar] [CrossRef]
- Xia, W.; Zhang, W. Characterization of surface properties of Inner Mongolia coal using FTIR and XPS. Energy Sources Part A-Recovery Util. Environ. Eff. 2017, 39, 1190–1194. [Google Scholar] [CrossRef]
- Zhu, H.Q.; Zhao, H.R.; Wei, H.Y.; Wang, W.; Wang, H.R.; Li, K. Investigation into the thermal behavior and FTIR micro-characteristics of re-oxidation coal. Combust. Flame 2020, 216, 354–368. [Google Scholar] [CrossRef]
- Rautenbach, R.; Matjie, R.H.; Strydom, C.A.; Bunt, J.R.; Ward, C.R.; French, D. Evaluation of mineral matter transformations in low-temperature ashes of South African coal feedstock samples and their density separated cuts using high-temperature X-ray diffraction. Int. J. Coal Prep. Util. 2020, 40, 320–347. [Google Scholar] [CrossRef]
- Boral, P.; Varma, A.K.; Maity, S. X-ray diffraction studies of some structurally modified Indian coals and their correlation with petrographic parameters. Curr. Sci. 2015, 108, 384–394. Available online: https://www.jstor.org/stable/24216565 (accessed on 30 December 2025).
- Yan, J.C.; Lei, Z.P.; Li, Z.K.; Wang, Z.C.; Ren, S.B.; Kang, S.G. Molecular structure characterization of low-medium rank coals via XRD, solid state 13C NMR and FTIR spectroscopy. Fuel 2020, 268, 117038. [Google Scholar] [CrossRef]
- Ping, A.; Xia, W.C.; Peng, Y.L.; Xie, G.Y. Construction of bituminous coal vitrinite and inertinite molecular assisted by 13C NMR, FTIR and XPS. J. Mol. Struct. 2020, 1222, 128959. [Google Scholar] [CrossRef]
- Lu, Y.; Kang, Y.L.; Ramakrishna, S.; You, L.J.; Hu, Y. Enhancement of multi-gas transport process in coalbed methane reservoir by oxidation treatment: Based on the change of the interaction force between coal matrix and gas molecules and knudsen number. Int. J. Hydrogen Energy 2023, 48, 478–494. [Google Scholar] [CrossRef]
- Sun, Z.; Li, K.J.; Bu, Y.S.; Liang, Z.; Jiang, C.H.; Zhang, J.L. Molecular insights into bituminous coals pyrolysis: A combined study using spectroscopic techniques, thermogravimetric-mass spectrometry and ReaxFF molecular dynamics simulations. Energy 2025, 315, 134442. [Google Scholar] [CrossRef]
- Xia, W.C.; Yang, J.G.; Liang, C. Investigation of changes in surface properties of bituminous coal during natural weathering processes by XPS and SEM. Appl. Surf. Sci. 2014, 293, 293–298. [Google Scholar] [CrossRef]
- Jing, Z.H.; Rodrigues, S.; Strounina, E.; Li, M.R.; Wood, B.; Underschultz, J.R. Use of FTIR, XPS, NMR to characterize oxidative effects of NaClO on coal molecular structures. Int. J. Coal Geol. 2019, 201, 1–13. [Google Scholar] [CrossRef]












| Absorption Peak Assignment | Position (cm−1) | Absorption Peak Assignment | Position (cm−1) |
|---|---|---|---|
| di-substituted benzene ring | 750 cm−1 | Symmetric CH3 stretching vibration of alkanes | 2860 cm−1 |
| tri-substituted benzene ring | 800 cm−1 | CH stretching vibration in alkanes | 2900 cm−1 |
| tetra-substituted benzene ring | 840 cm−1 | Asymmetric CH2 stretching vibration in alkanes | 2920 cm−1 |
| penta-substituted benzene ring | 860 cm−1 | Asymmetric CH3 stretching vibration in alkanes | 2940 cm−1 |
| Ash content | 1100 cm−1 | Hydroxyl–nitrogen hydrogen bonding | 3100~2800 cm−1 |
| C-O in phenols, alcohols, ethers, and esters | 1100~1300 cm−1 | Cyclic tightly associated hydroxyl hydrogen bonding | 3200 cm−1 |
| CH3 and CH2 asymmetric deformation vibration | 1440 cm−1 | Hydroxyl–ether oxygen hydrogen bonding | 3300 cm−1 |
| aromatic nucleus C=C | 1600 cm−1 | Self-associated hydroxyl hydrogen bonding | 3400 cm−1 |
| C=O stretching vibration | 1650 cm−1 | Hydroxyl-π hydrogen bonding | 3516 cm−1 |
| COOH stretching vibration | 1700 cm−1 | Free hydroxyl group | 3611 cm−1 |
| Symmetric CH2 stretching vibration in alkanes | 2840 cm−1 |
| Chemical Shift (ppm) | Primary Affiliation | Symbol |
|---|---|---|
| 0~16 | aliphatic methyl, terminal methyl | fal* |
| 16~23 | methyl connected to aromatic ring | |
| 23~36 | methylene, methylidene, methylene group in saturated cycloalkane | falH |
| 36~50 | the α-position carbon on aliphatic and aromatic carbons | |
| 50~75 | methoxy group, phenoxy group, oxygen-connected methylene carbon, oxygen-connected secondary methylene carbon | falO |
| 75~90 | ring-in oxygen bonded to fatty carbon | |
| 100~129 | protonated aromatic carbon | faH |
| 129~137 | inter-ring bridging aromatic carbon | faB |
| 137~148 | alkyl-substituted aryl carbon (side-chain aryl carbon) | faS |
| 148~165 | oxygen-substituted aromatic carbon | faP |
| 165~188 | carboxyl carbon | faC |
| 188~220 | carbonyl carbon | faN |
| 129~165 | deprotonated carbon | fa’ |
| 100~165 | sp2 hybridization in aromatic rings | fa |
| 100~220 | aromatic carbon ratio | fal |
| 0~90 | fat-to-carbon ratio | faH |
| Sample | Ro,max (%) | Mad (%) | Aad (%) | Vad (%) | FCad (%) | Cdaf (%) | Hdaf (%) | Odaf (%) | Ndaf (%) | Vitrinite (%) | Inertinite (%) | Exinite (%) | Mineral (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| YZG-VC | 0.65 | 5.21 | 0.36 | 35.57 | 58.86 | 76.60 | 4.80 | 17.47 | 1.12 | 68.3 | 27.8 | 2.2 | 1.7 |
| YZG-DC | 4.31 | 1.68 | 24.97 | 69.05 | 76.67 | 4.29 | 18.03 | 1.02 | 39.4 | 54.1 | 2.1 | 4.4 | |
| HL-VC | 0.75 | 3.93 | 3.45 | 30.95 | 61.67 | 76.85 | 4.98 | 16.85 | 1.32 | 72.8 | 22.8 | 1.1 | 3.3 |
| HL-DC | 3.87 | 7.24 | 24.70 | 64.19 | 77.41 | 4.12 | 17.45 | 1.02 | 13.3 | 74.6 | 7.3 | 4.8 | |
| LL-VC | 1.41 | 0.75 | 18.29 | 13.60 | 67.36 | 83.46 | 4.53 | 9.25 | 1.76 | 73.0 | 24.0 | 0.0 | 3.0 |
| LL-DC | 0.27 | 21.33 | 12.35 | 66.05 | 85.57 | 4.02 | 9.09 | 1.32 | 26.7 | 67.3 | 2.2 | 3.8 | |
| XS-VC | 1.71 | 0.84 | 10.45 | 22.72 | 65.99 | 89.38 | 3.98 | 5.27 | 1.37 | 73.9 | 19.2 | 0.0 | 6.9 |
| XS-DC | 0.79 | 26.65 | 17.63 | 54.94 | 91.36 | 3.91 | 3.59 | 1.14 | 18.2 | 73.5 | 2.6 | 5.7 |
| Sample | faH | faN | faC | faB | faS | faP | fa’ | fa | fal* | falH | falO | fal | XBP |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| YZG-VC | 39.19 | 17.86 | 3.58 | 8.05 | 5.64 | 4.16 | 57.04 | 60.62 | 8.92 | 20.12 | 10.35 | 39.38 | 0.164 |
| YZG-DC | 41.56 | 16.38 | 5.18 | 9.24 | 4.36 | 2.77 | 57.94 | 63.12 | 8.24 | 18.79 | 9.86 | 36.88 | 0.190 |
| HL-VC | 40.07 | 17.62 | 2.43 | 8.74 | 5.24 | 3.64 | 57.69 | 60.12 | 9.74 | 21.35 | 8.78 | 39.88 | 0.178 |
| HL-DC | 43.55 | 16.13 | 3.76 | 9.8 | 3.57 | 2.76 | 59.67 | 63.43 | 8.72 | 19.82 | 8.03 | 36.57 | 0.196 |
| LL-VC | 39.62 | 17.44 | 19.46 | 9.79 | 3.44 | 4.20 | 57.06 | 76.51 | 5.96 | 5.81 | 11.72 | 23.49 | 0.207 |
| LL-DC | 42.88 | 17.72 | 15.97 | 10.59 | 4.56 | 2.58 | 60.60 | 76.57 | 3.75 | 7.72 | 11.95 | 23.43 | 0.212 |
| XS-VC | 32.28 | 22.32 | 19.01 | 12.57 | 7.45 | 2.30 | 57.60 | 76.60 | 6.80 | 8.29 | 11.31 | 23.40 | 0.299 |
| XS-DC | 38.37 | 18.60 | 21.21 | 13.54 | 2.61 | 2.44 | 56.97 | 78.18 | 4.78 | 5.31 | 11.73 | 21.82 | 0.311 |
| Sample | C-C,C-H | C-O | C=O | COOH |
|---|---|---|---|---|
| YZG-VC | 65.00 | 23.08 | 9.67 | 2.25 |
| YZG-DC | 60.46 | 27.29 | 10.05 | 2.20 |
| HL-VC | 59.15 | 29.62 | 10.08 | 1.15 |
| HL-DC | 58.37 | 32.81 | 8.83 | 0.00 |
| LL-VC | 69.00 | 18.23 | 11.51 | 1.26 |
| LL-DC | 67.12 | 21.63 | 10.05 | 1.20 |
| XS-VC | 72.47 | 17.96 | 8.80 | 0.77 |
| XS-DC | 69.24 | 13.58 | 11.10 | 0.61 |
| Sample | C-O | C=O | COO- |
|---|---|---|---|
| YZG-VC | 74.45 | 15.77 | 6.49 |
| YZG-DC | 74.59 | 14.07 | 9.68 |
| HL-VC | 74.17 | 7.68 | 17.62 |
| HL-DC | 70.12 | 16.42 | 7.79 |
| LL-VC | 58.25 | 10.79 | 22.12 |
| LL-DC | 61.43 | 11.70 | 19.56 |
| XS-VC | 78.79 | 11.14 | 4.34 |
| XS-DC | 73.17 | 7.57 | 10.19 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Chen, Y.; Liu, Z.; Wang, H.; Ji, C.; Wang, L.; Guan, P.; Wang, P.; Ma, K.; Dang, L. The Evolution Law of Molecular Structure of Vitrain and Durain During Low–Medium Coalification. Processes 2026, 14, 401. https://doi.org/10.3390/pr14030401
Chen Y, Liu Z, Wang H, Ji C, Wang L, Guan P, Wang P, Ma K, Dang L. The Evolution Law of Molecular Structure of Vitrain and Durain During Low–Medium Coalification. Processes. 2026; 14(3):401. https://doi.org/10.3390/pr14030401
Chicago/Turabian StyleChen, Yue, Zan Liu, Huaichang Wang, Changjiang Ji, Liya Wang, Pengpeng Guan, Peilin Wang, Kai Ma, and Liyuan Dang. 2026. "The Evolution Law of Molecular Structure of Vitrain and Durain During Low–Medium Coalification" Processes 14, no. 3: 401. https://doi.org/10.3390/pr14030401
APA StyleChen, Y., Liu, Z., Wang, H., Ji, C., Wang, L., Guan, P., Wang, P., Ma, K., & Dang, L. (2026). The Evolution Law of Molecular Structure of Vitrain and Durain During Low–Medium Coalification. Processes, 14(3), 401. https://doi.org/10.3390/pr14030401






