Mechanism of Hydrogen Bonding at Oil–Water Interfaces on Crude Oil Migration Under Nanoconfinement
Abstract
1. Introduction
2. Materials and Methods
2.1. Construction of Molecular Models
- (1)
- Quartz Surface Model
- (2)
- Injected Water and Formation Water Models
- (3)
- Crude Oil Model
2.2. Molecular Dynamics Simulation
3. Results and Discussion
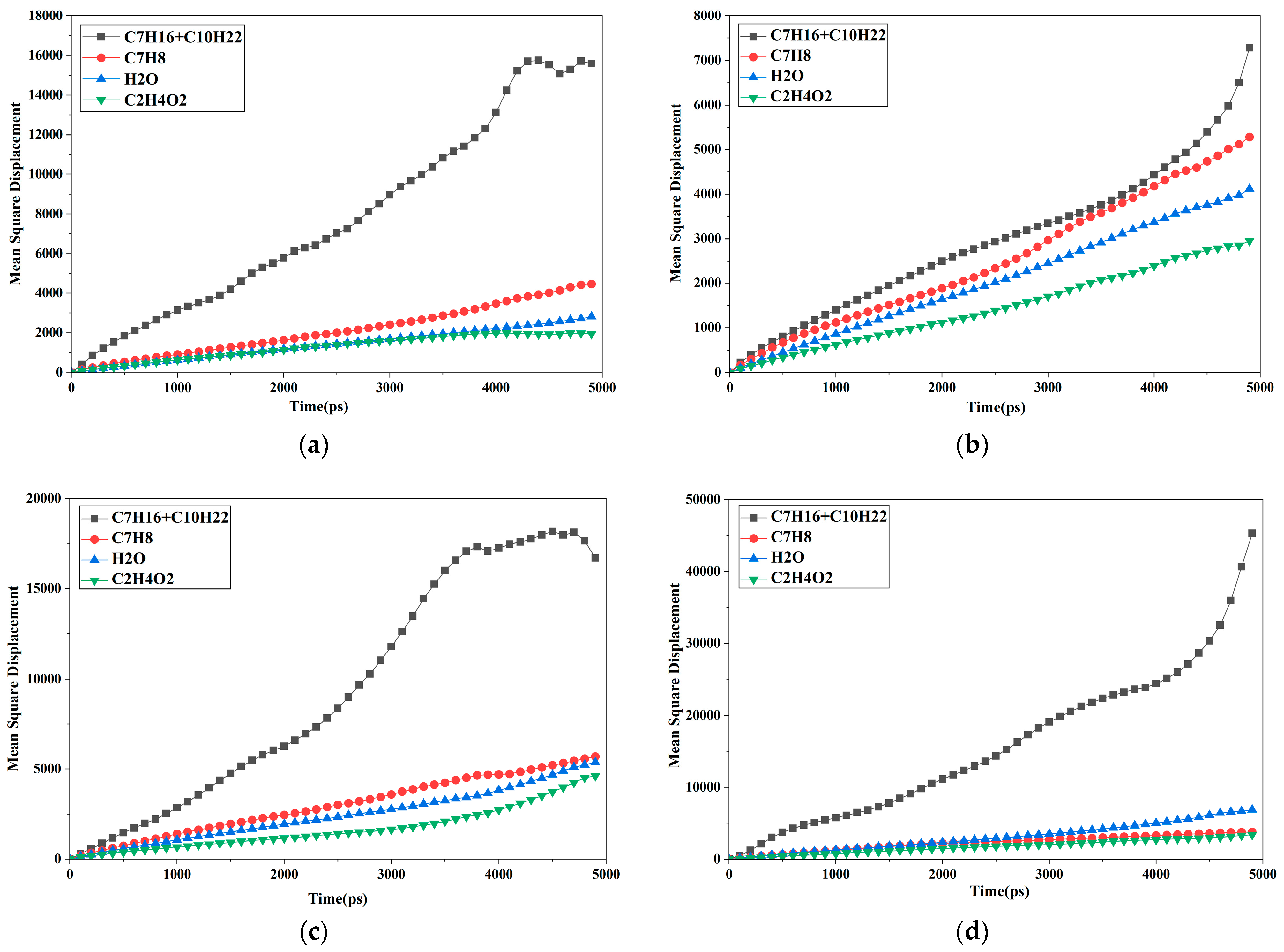
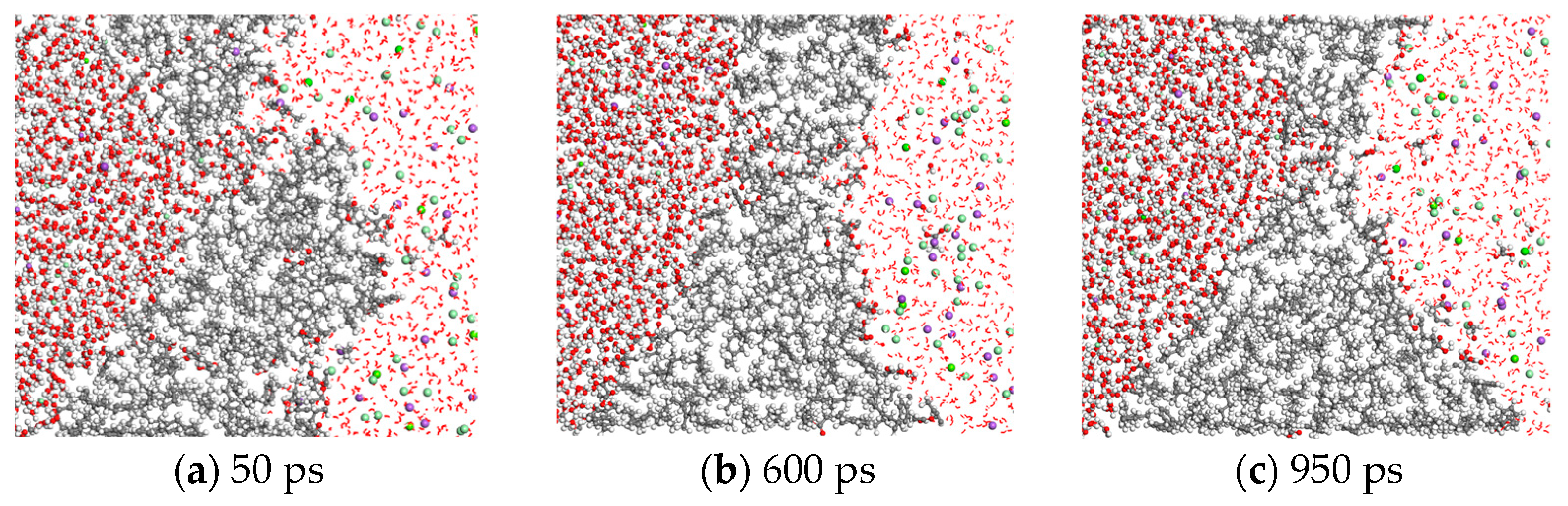
3.1. Microscopic Migration Behavior of Crude Oil Components and Water Molecules
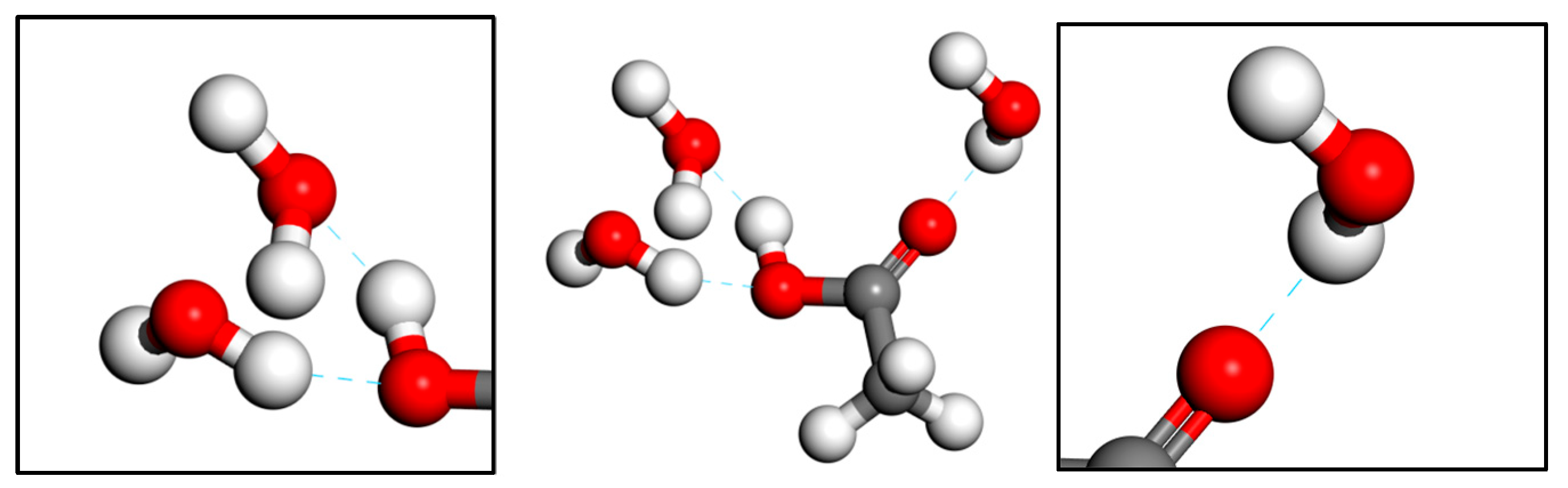
3.2. Formation and Stability of Acetic Acid–Water Hydrogen Bonds at the Oil–Water Interface
3.3. Influence of Salinity on the Hydrogen-Bonding Effect
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Dordzie, G.; Balhoff, M. A grand challenge: Update on improved recovery from tight/shale reservoirs. J. Pet. Technol. 2024, 76, 38–42. [Google Scholar] [CrossRef]
- Liu, S.; Qiu, Y.; Liu, T.; Li, X.; Chen, X.; Yang, J.; Ma, X.; Zhao, H.; Wang, Y.; Liu, J. Peeling off mechanism and influencing factors of adhesive oil film in ultra-deep tight reservoir—Insights from molecular dynamics. Chem. Eng. Sci. 2025, 309, 121515. [Google Scholar] [CrossRef]
- He, Z.; Ji, X.B.; Xie, A.; Wang, D.X.; Wang, Z.Z.; Tan, Q.; Ma, A.Y.; Abudukelimu, P.; Zhang, Y.J.; Zhao, X.D. Tight sandstone reservoir characteristics and main controlling factors of tight oil enrichment in the second member of Permian Jingjingzigou Formation, Jimsar Sag. Chin. J. Geol. 2025, 60, 33–43. [Google Scholar] [CrossRef]
- Ren, J.; Xiao, W.; Cheng, Q.; Song, P.; Bai, X.; Xie, Q.; Pu, W.; Zheng, L. Experimental study on water/CO2 flow of tight oil using HTHP microscopic visualization and NMR technology. Geoenergy Sci. Eng. 2025, 250, 213834. [Google Scholar] [CrossRef]
- Wang, W.; Li, T.; Xiao, D.; Wang, B.; Yang, Y.; Zhang, Y.; La, W.; He, J. Geological conditions and controls on accumulation of tight sandstone gas, deep part of the Shengbei sub-sag, Turpan-Hami basin, NW China. Mar. Pet. Geol. 2023, 158, 106513. [Google Scholar] [CrossRef]
- Wang, W.; Zheng, K.; Huang, S.; Wang, Q.; Feng, G.; Su, Y.; Li, J. Difference of hydrocarbon generation between argillaceous dolomite and mudstone and its influence on oil source: A case study of the Es4 in the Leijia area, Liaohe depression. Geoenergy Sci. Eng. 2024, 237, 212805. [Google Scholar] [CrossRef]
- Wang, W.; Ma, H.; Li, Y.; Yi, Z.; Li, Z.; Yan, Y. Salt-induced coil-globule transition of sulfonate-modified HPAM is affected by the branched chain length. Langmuir 2023, 39, 14969–14976. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhang, T.; Liu, S.; Ding, K.; Liu, T.; Yao, J.; Sun, H.; Yang, Y.; Zhang, L.; Wang, W.-D.; et al. Unveiling nanoscale fluid miscible behaviors with nanofluidic slim-tube. Energy Environ. Sci. 2024, 17, 9635–9651. [Google Scholar] [CrossRef]
- Wang, Z.; Liu, T.; Liu, S.; Jia, C.; Yao, J.; Sun, H.; Yang, Y.; Zhang, L.; Delshad, M.; Sepehrnoori, K.; et al. Adsorption effects on CO2-oil minimum miscibility pressure in tight reservoirs. Energy 2024, 288, 129815. [Google Scholar] [CrossRef]
- Jing, W.; Zhang, L.; Li, A.; Liu, T.; Cheng, Y.; Sun, H.; Yang, Y.; Zhu, G.; Yao, J.; Zhong, J. Phase behaviors of gas condensate at pore scale: Direct visualization via microfluidics and in-situ CT scanning. SPE J. 2024, 29, 2566–2577. [Google Scholar] [CrossRef]
- Yu, Z.; Li, Z.; Wang, S.; Zhao, Q.; Dong, X.; Li, K.; Yao, Z. Adsorption mechanism of polymers at the oil-water interfacial film and its effect on interfacial stability. Geoenergy Sci. Eng. 2026, 257, 214189. [Google Scholar] [CrossRef]
- Xue, M.; Chen, L.; Chen, H.; Fu, L.; Bai, Y.; Lv, W.; Hou, B.; Riazi, M. Hydrogen-bond-modulated flowable weak gel for EOR in ultra-high temperature and ultra-high salinity fracture-cavity ordinary heavy oil reservoirs. Colloids Surf. A Physicochem. Eng. Asp. 2025, 725, 137537. [Google Scholar] [CrossRef]
- Zhou, H.; Liu, X.; He, C.; He, L.; Sui, H. Reconfiguration of intermolecular hydrogen bond for viscosity reduction of heavy oil. Chem. Eng. Sci. 2025, 313, 121696. [Google Scholar] [CrossRef]
- Kumar, R.; Schmidt, J.R.; Skinner, J.L. Hydrogen bonding definitions and dynamics in liquid water. J. Chem. Phys. 2007, 126, 204107. [Google Scholar] [CrossRef]
- Liu, X.; Zhou, D.; Yan, L.; Liu, S.; Liu, Y. On the imbibition model for oil-water replacement of tight sandstone oil reservoirs. Geofluids 2021, 2021, 8846132. [Google Scholar] [CrossRef]
- Liu, X.; Yan, L.; Gao, Q.; Liu, Y.; Huang, H.; Liu, S. Effect of salinity on the imbibition recovery process of tight sandstone reservoirs. Processes 2022, 10, 228. [Google Scholar] [CrossRef]
- Liu, X.; Zhou, D.S.; Shi, Y.H.; Li, M.; Zhang, Z. A semi-analytical mathematical model for imbibition in tight sandstone reservoirs considering osmotic pressure. Pet. Geol. Recovery Effic. 2018, 25, 93–98. [Google Scholar] [CrossRef]
- Cai, Y.Y.; Zhao, W.C.; Ge, J.; Cong, Z.C.; Hu, S.; Gong, Y.T. Molecular Dynamics Simulation of Spontaneous Percolation of Tight Oil after Pressure. Sci. Technol. Eng. 2023, 23, 12064–12071. [Google Scholar] [CrossRef]
- Cui, F.; Jin, X.; Xia, J.; Liu, H.; Wu, H.; Wang, F. Micromechanical mechanism of oil/brine/rock interfacial interactions based on first-principles calculations. J. Mol. Liq. 2023, 386, 122502. [Google Scholar] [CrossRef]
- Lu, P.; Yan, Z.; Lai, J.; Wang, K. Molecular insight into oil displacement by CO2 flooding in water-cut dead-end nanopores. RSC Adv. 2024, 14, 25385–25392. [Google Scholar] [CrossRef]
- Li, J.; Xu, Y.F.; Wang, Z.H. Influencing factors and microscopic mechanisms of oil-water interfacial film formation in shear flow fields. Acta Pet. Sin. 2024, 45, 1244–1256. [Google Scholar] [CrossRef]
- Zhang, X.; He, C.; Mostafa, M.Y.M.; Akram, W.; He, L.; Sui, H.; Li, X. Constructing oxygen-based multiple hydrogen bonding sites and delocalized π bonds for efficient oil-water-solid interfacial separation. J. Hazard. Mater. 2025, 490, 137766. [Google Scholar] [CrossRef] [PubMed]
- Akkermans, R.L.C.; Spenley, N.A.; Robertson, S.H. COMPASS III: Automated fitting workflows and extension to ionic liquids. Mol. Simul. 2021, 47, 540–551. [Google Scholar] [CrossRef]
- Marcus, Y. Effect of ions on the structure of water: Structure making and breaking. Chem. Rev. 2009, 109, 1346–1370. [Google Scholar] [CrossRef]
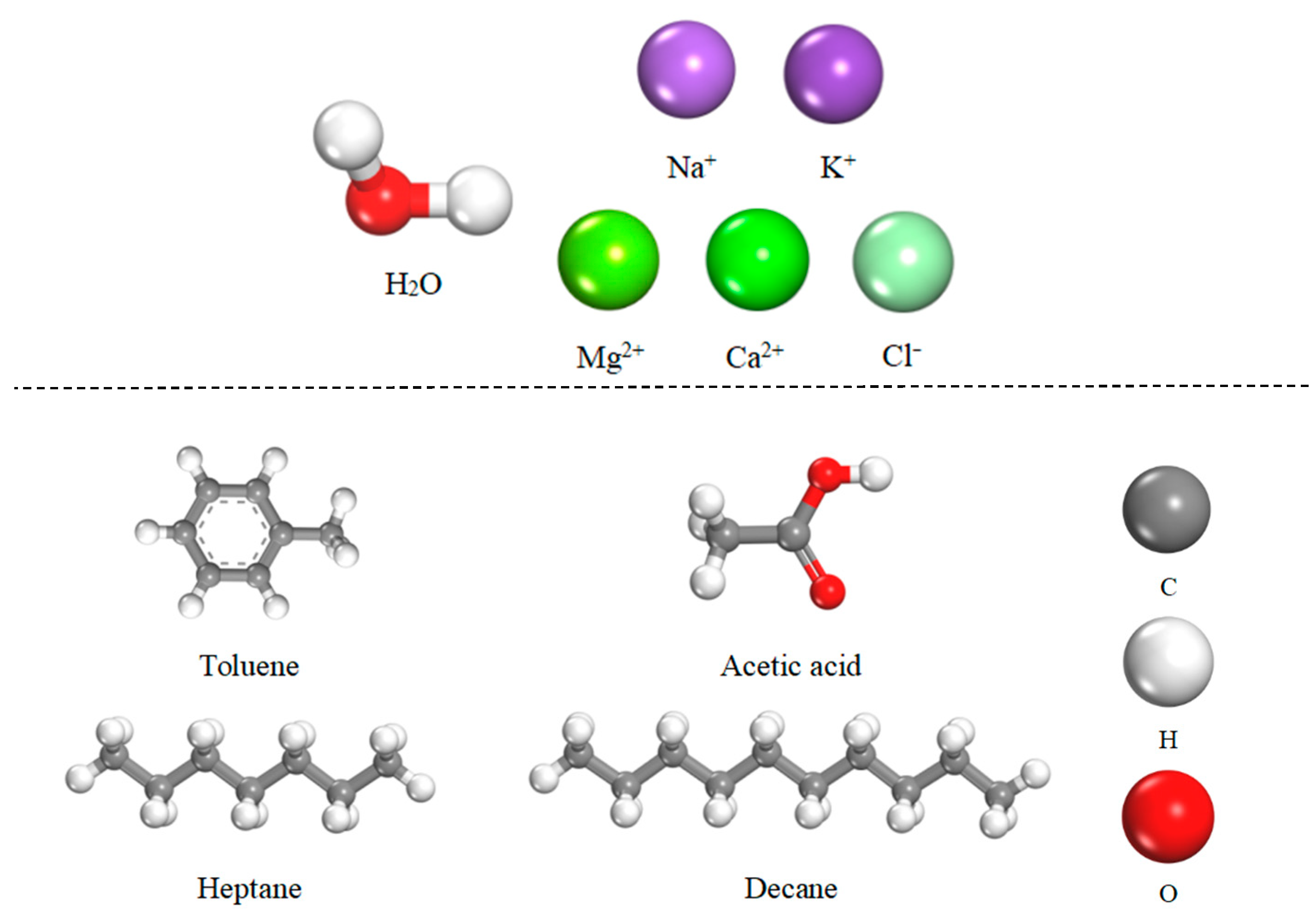







| Number | Salinity (g/L) | Number of Water Molecules and Ions in the Aqueous Phase | Water Box Dimensions () | |||||
|---|---|---|---|---|---|---|---|---|
| H2O | Na+ | K+ | Mg2+ | Ca2+ | Cl− | |||
| #1 (HW) | 246.5 | 1600 | 80 | 14 | 18 | 18 | 166 | 60 × 20 × 52.93 |
| #2 (MW) | 140.6 | 1600 | 40 | 7 | 9 | 9 | 83 | 60 × 20 × 46.41 |
| #3 (MLW) | 54.0 | 1600 | 15 | 2 | 3 | 3 | 29 | 60 × 20 × 42.16 |
| #4 (LW) | 21.9 | 1600 | 6 | 1 | 1 | 1 | 11 | 60 × 20 × 39.88 |
| SW | 33.1 | 1600 | 10 | 1 | 2 | 2 | 19 | 60 × 20 × 44.83 |
| Viscosity/mPa·s | Density/g·cm−3 | Acidity/mg·g−1 | Component Volume Fraction/% | |||
|---|---|---|---|---|---|---|
| Saturates/% | Aromatics/% | Resins/% | Asphaltenes/% | |||
| 4.21 | 0.821 | 0.435 | 82.19 | 10.73 | 6.79 | 0.29 |
| Number | Molecular Diffusion Coefficients (2/ps) | |||
|---|---|---|---|---|
| Alkanes (Heptane, Decane) | Toluene | Acetic Acid | Injected Water | |
| #1 | 0.689 | 0.180 | 0.039 | 0.093 |
| #2 | 0.259 | 0.202 | 0.112 | 0.146 |
| #3 | 0.643 | 0.198 | 0.127 | 0.154 |
| #4 | 1.650 | 0.296 | 0.108 | 0.283 |
| Number | EAcetic Acid–Water (kcal/mol) | EWater–Water (kcal/mol) | EAcetic Acid–Water/EWater–Water |
|---|---|---|---|
| #1 | −589.796 ± 15.3 | −434.982 ± 10.1 | 1.35 |
| #2 | −561.992 ± 18.6 | −473.358 ± 9.8 | 1.19 |
| #3 | −535.149 ± 14.8 | −495.543 ± 11.2 | 1.08 |
| #4 | −521.889 ± 20.1 | −518.026 ± 10.5 | 1.01 |
| Number | Maximum Number | Minimum Number | Maximum Bond Length () | Minimum Bond Length () | Maximum Bond Angle | Minimum Bond Angle |
|---|---|---|---|---|---|---|
| #1 | 169 | 122 | 3.09991 | 1.36482 | 179.6088 | 90.00097 |
| #2 | 176 | 122 | 3.09988 | 1.38348 | 179.6425 | 90.02183 |
| #3 | 168 | 123 | 3.09991 | 1.39304 | 179.7578 | 90.00211 |
| #4 | 173 | 123 | 3.09999 | 1.40139 | 179.7695 | 90.01118 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Liu, X.; Cheng, Y.; Peng, L.; Cui, Y.; Gong, Y. Mechanism of Hydrogen Bonding at Oil–Water Interfaces on Crude Oil Migration Under Nanoconfinement. Processes 2026, 14, 343. https://doi.org/10.3390/pr14020343
Liu X, Cheng Y, Peng L, Cui Y, Gong Y. Mechanism of Hydrogen Bonding at Oil–Water Interfaces on Crude Oil Migration Under Nanoconfinement. Processes. 2026; 14(2):343. https://doi.org/10.3390/pr14020343
Chicago/Turabian StyleLiu, Xiong, Yuchan Cheng, Lingxuan Peng, Yueqi Cui, and Yue Gong. 2026. "Mechanism of Hydrogen Bonding at Oil–Water Interfaces on Crude Oil Migration Under Nanoconfinement" Processes 14, no. 2: 343. https://doi.org/10.3390/pr14020343
APA StyleLiu, X., Cheng, Y., Peng, L., Cui, Y., & Gong, Y. (2026). Mechanism of Hydrogen Bonding at Oil–Water Interfaces on Crude Oil Migration Under Nanoconfinement. Processes, 14(2), 343. https://doi.org/10.3390/pr14020343






