Mass Deposition Rates of Carbon Dioxide onto a Cryogenically Cooled Surface
Abstract
1. Introduction
2. Experimental Section
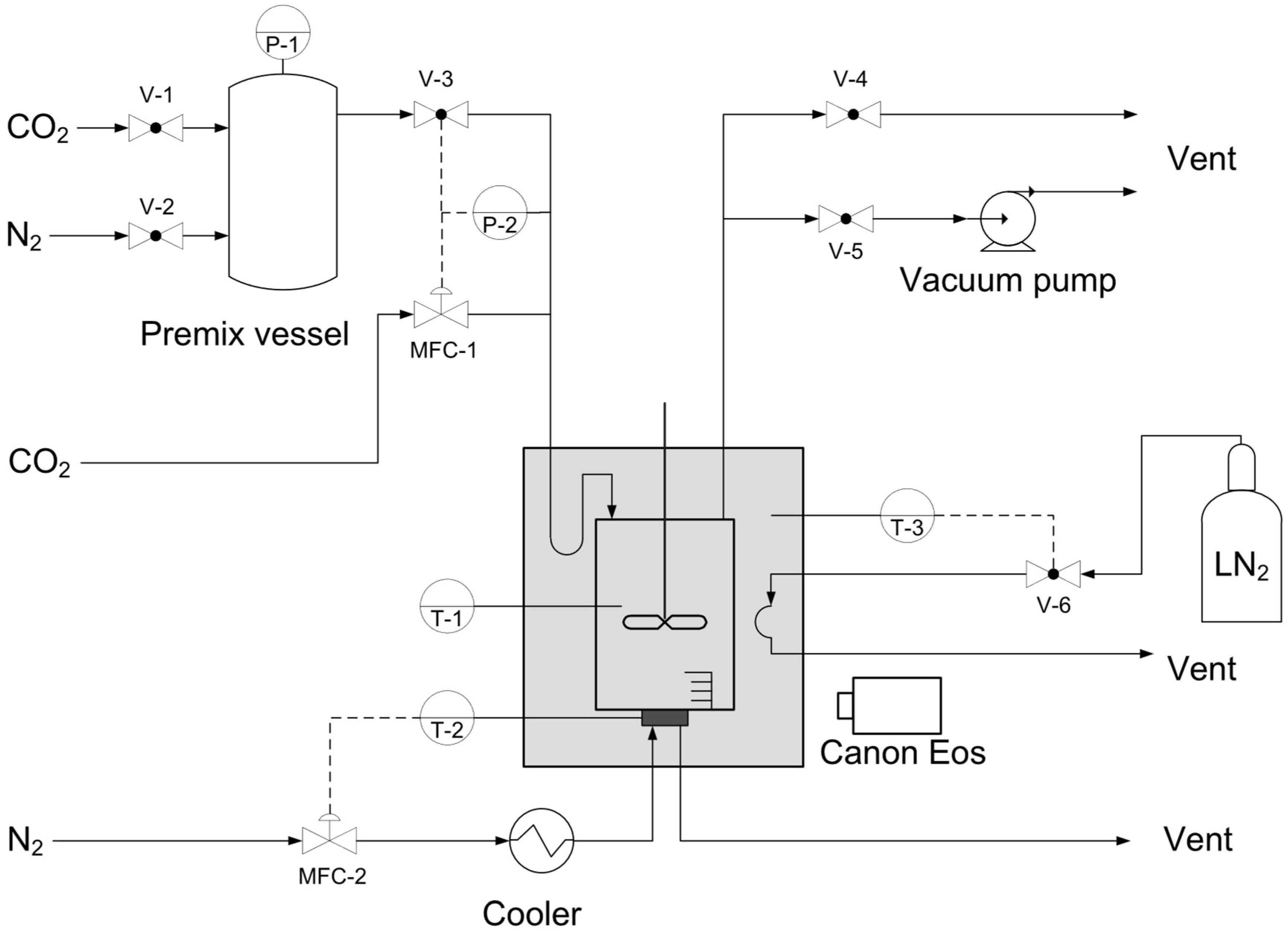
2.1. Setup
2.2. Procedure
2.3. Data Processing
3. Results
4. Development of a Frost Growth Model
4.1. Model Development
- -
- The temperature profiles within the layer are established instantly (quasi-steady state approach), meaning the frost growth is much slower than the change over time of the temperature profiles in the layer.
- -
- The desublimated CO2 at the frost surface is in equilibrium with the gas phase.
- -
- The measurements revealed that density changes over time. The formed ice layer probably has a porous structure that becomes denser as time passes. The modeling study by Lei et al. also observed a gradual transformation from a loosely structured to a densely structured solid CO2 layer [16]. The porosity, density, and consequently the layer heat conductivity might vary depending on the location within the frost layer. However, this information could not be obtained from the experiments; therefore, it is assumed that the layer has a uniform density and thermal conductivity. CO2 desublimation inside the layer has been incorporated in a lumped manner into the bulk density.
- -
- The mass and heat transfer coefficients for the transfer from the gas bulk toward the frost surface are coupled according to the Chilton–Colburn analogy.
4.2. Simulation Results
4.3. Significance for the Cryogenic Packed Bed Concept
5. Discussion and Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Nomenclature
| Area | ||
| Thermal diffusivity | ||
| Concentration | ||
| Heat capacity | ||
| Diffusion coefficient | ||
| Diameter | ||
| Particle diameter | ||
| Mass deposition rate constant | ||
| Gas solid mass transfer coefficient | ||
| Molecular weight | ||
| Molar flux | ||
| Stirrer rotations speed | ||
| Nusselt number () | ||
| Pressure | ||
| Prandtl number () | ||
| Gas constant | ||
| Reynolds number (, ) | ||
| Schmidt number () | ||
| Sherwood number () | ||
| Time | ||
| Temperature | ||
| Volume | ||
| Superficial velocity | ||
| Mole fraction | ||
| Greek letters | ||
| Gas solid heat transfer coefficient | ||
| Layer thickness | ||
| Sublimation enthalpy | ||
| Emissivity | ||
| Viscosity | ||
| Thermal conductivity | ||
| Effective conductivity frost layer | ||
| Correction factor for drift flux | ||
| Mass density | ||
| Atomic diffusion volume | ||
| Stefan–Boltzmann constant | ||
| Dimensionless factor for drift flux | ||
| Gas–solid heat flux | ||
| Subscripts and superscripts | ||
| Cold plate, initial | ||
| Gas phase | ||
| Particle | ||
| Solid phase | ||
| Total | ||
| Equilibrium | ||
References
- Intergovernmental Panel on Climate Change (IPCC). Technical Summary in Climate Change 2021—The Physical Science Basis; Cambridge University Press: Cambridge, UK, 2023; pp. 35–144. [Google Scholar] [CrossRef]
- The Paris Agreement. 2016. Available online: https://treaties.un.org/Pages/ViewDetails.aspx?src=TREATY&mtdsg_no=XXVII-7- (accessed on 1 September 2025).
- COP26. Special Report on Climate Change and Health: The Health Argument for Climate Action; WHO: Geneva, Switzerland, 2021. [Google Scholar]
- Song, C.; Liu, Q.; Deng, S.; Li, H.; Kitamura, Y. Cryogenic-Based CO2 Capture Technologies: State-of-the-Art Developments and Current Challenges; Elsevier Ltd.: Amsterdam, The Netherlands, 2019. [Google Scholar] [CrossRef]
- Song, C.F.; Kitamura, Y.; Li, S.H. Evaluation of Stirling cooler system for cryogenic CO2 capture. Appl. Energy 2012, 98, 491–501. [Google Scholar] [CrossRef]
- Clodic, D.; Younes, M. A new Method for CO2 Capture Frosting CO2 at Atmospheric Pressure. In Greenhouse Gas Control Technologies—6th International Conference; Elsevier: Kyoto, Japan, 2003; pp. 155–160. [Google Scholar] [CrossRef]
- Tuinier, M.J.; Van Sint Annaland, M.; Kuipers, J.A.M. A novel process for cryogenic CO2 capture using dynamically operated packed beds-An experimental and numerical study. Int. J. Greenh. Gas Control 2011, 5, 694–701. [Google Scholar] [CrossRef]
- Tuinier, M.J.; van Sint Annaland, M.; Kramer, G.J.; Kuipers, J.A.M. Cryogenic CO2 capture using dynamically operated packed beds. Chem. Eng. Sci. 2010, 65, 114–119. [Google Scholar] [CrossRef]
- Chang, H.-M.; Smith, J.L. Mass transfer of supersaturated contaminants in cryogenic helium heat exchangers. Int. J. Heat Mass Transf. 1990, 33, 1299–1306. [Google Scholar] [CrossRef]
- Chang, H.-M.; Chung, M.J.; Park, S.B. Cryogenic Heat-Exchanger Design for Freeze-out Removal of Carbon Dioxide from Landfill Gas. J. Therm. Sci. Technol. 2009, 4, 362–371. [Google Scholar] [CrossRef]
- Naletov, V.A.; Lukyanov, V.L.; Kulov, N.N.; Naletov, A.Y.; Glebov, M.B. An experimental study of desublimation of carbon dioxide from a gas mixture. Theor. Found. Chem. Eng. 2014, 48, 312–319. [Google Scholar] [CrossRef]
- Ogunbameru, A.N.; Brian, P.L.T.; Reid, R.C. On Carbon Dioxide Frost Formation. Ind. Eng. Chem. Fundam. 1973, 12, 385–387. [Google Scholar] [CrossRef]
- Shchelkunov, V.N.; Rudenko, N.Z.; Shostak, Y.V.; Dolganin, V.I. Surface desublimation of carbon dioxide from binary gas mixtures. J. Eng. Phys. 1986, 51, 1432–1435. [Google Scholar] [CrossRef]
- Haddad, S.; Rivera-Tinoco, R.; Bouallou, C. Modelling of CO2 Frost Formation and Growth on a Flat Plate. Chem. Eng. Trans. 2019, 76, 667–672. [Google Scholar] [CrossRef]
- Lei, T.; Luo, K.H.; Pérez, F.E.H.; Wang, G.; Yang, J.; Cano, J.R.; Im, H.G. Pore-scale study of CO2 desublimation and sublimation in a packed bed during cryogenic carbon capture. J. Fluid Mech. 2024, 990, A6. [Google Scholar] [CrossRef]
- Lei, T.; Luo, K.H.; Pérez, F.E.H.; Wang, G.; Wang, Z.; Cano, J.R.; Im, H.G. Study of CO2 desublimation during cryogenic carbon capture using the lattice Boltzmann method. J. Fluid Mech. 2023, 964, A1. [Google Scholar] [CrossRef]
- Ren, Z.Y.; Wang, B.B.; Qiu, G.D.; Bian, J.; Li, Q.Y.; Cai, W.H. Molecular dynamics study of carbon dioxide desublimation on surfaces with different hydrophobicity. Energy 2025, 318, 134972. [Google Scholar] [CrossRef]
- Wang, Y.N.; Pfotenhauer, J.M.; Qiu, L.M.; Zhi, X.Q.; Jiang, X.B. Experimental study of carbon dioxide desublimation and sublimation process on low temperature surface. In IOP Conference Series: Materials Science and Engineering; Institute of Physics Publishing: Bristol, UK, 2020. [Google Scholar] [CrossRef]
- Du, S.; Shi, Y.; Wang, H.; Qiu, G.; Li, Q.; Li, Q.; Cai, W. Experimental study on the effect of CO2 desublimation on heat transfer. Int. Commun. Heat Mass Transf. 2024, 159, 108136. [Google Scholar] [CrossRef]
- Cai, W.; Du, S.; Wang, H.; Qiu, G.; Wang, Z.; Li, Q. Desublimation characteristics of CO2 under pressurized cryogenic conditions. Appl. Therm. Eng. 2026, 282, 128845. [Google Scholar] [CrossRef]
- Du, S.; Wang, H.; Qiu, G.; Wan, H.; Wang, Z.; Li, Q.; Cai, W. The effect of CO2 desublimation on heat transfer in binary gas based on cryogenic experiment. Int. Commun. Heat Mass Transf. 2025, 169, 109834. [Google Scholar] [CrossRef]
- Wang, Y.N.; Pfotenhauer, J.M.; Zhi, X.Q.; Qiu, L.M.; Li, J.F. Transient model of carbon dioxide desublimation from nitrogen-carbon dioxide gas mixture. Int. J. Heat Mass Transf. 2018, 127, 339–347. [Google Scholar] [CrossRef]
- Le, T.T.; Trebble, M.A. Measurement of carbon dioxide freezing in mixtures of methane, ethane, and nitrogen in the solid-vapor equilibrium region. J. Chem. Eng. Data 2007, 52, 683–686. [Google Scholar] [CrossRef]
- Krishna, R.; Taylor, R. Multicomponent Mass Transfer, 1st ed.; John Wiley & Sons: New York, NY, USA, 1993. [Google Scholar]
- Winkelman, J.G.M.; Voorwinde, O.K.; Ottens, M.; Beenackers, A.A.C.M.; Janssen, L.P.B.M. Kinetics and chemical equilibrium of the hydration of formaldehyde. Chem. Eng. Sci. 2002, 57, 4067–4076. [Google Scholar] [CrossRef]
- Fuller, E.N.; Schettler, P.D.; Giddings, J.C. A new method for prediction of binary gas-phase diffusion coefficients. Ind. Eng. Chem. 1966, 58, 18–27. [Google Scholar] [CrossRef]
- Daubert, T.E.; Danner, R.P. Data Compilation Tables of Properties of Pure Compounds; American Institute of Chemical Engineers: New York, NY, USA, 1985. [Google Scholar]
- Reid, R.C.; Prausnitz, J.M.; Poling, B.E. The Properties of Gases and Liquids, 4th ed.; McGraw-Hill: Columbus, OH, USA, 1987. [Google Scholar]
- Gunn, D.J.; Misbah, M.M.A. Bayesian estimation of heat transport parameters in fixed beds. Int. J. Heat Mass Transf. 1993, 36, 2209–2221. [Google Scholar] [CrossRef]
- Carslaw, H.S.; Jaeger, J.C. Conduction of Heat in Solids, 2nd ed.; Oxford University Press: Oxford, UK, 1986. [Google Scholar]
- Tuinier, M.J.; Ververs, W.J.R.; Tešić, D.; Roghair, I.; van Sint Annaland, M. Corresponding data for ‘Mass deposition rates of carbon dioxide onto a cryogenically cooled surface’. Int. J. Greenh. Gas Control 2025, 230–254. [Google Scholar] [CrossRef]















| Experiment | xCO2 [%] | PCO2 [mbar] | PN2 [mbar] | T0 [°C] | Tg [°C] | Ns [%] |
|---|---|---|---|---|---|---|
| P100 | 100 | 100 | 0 | −130 | −30 | 100 |
| P150 | 100 | 150 | 0 | −130 | −30 | 100 |
| P200 | 100 | 200 | 0 | −130 | −30 | 100 |
| P250 | 100 | 250 | 0 | −130 | −30 | 100 |
| T0-145 | 100 | 100 | 0 | −145 | −30 | 100 |
| T0-160 | 100 | 100 | 0 | −160 | −30 | 100 |
| 50T0-130 | 50 | 100 | 100 | −130 | −30 | 100 |
| 50T0-140 | 50 | 100 | 100 | −140 | −30 | 100 |
| 50T0-150 | 50 | 100 | 100 | −150 | −30 | 100 |
| 10T0-130 | 10 | 100 | 900 | −130 | −30 | 100 |
| 10T0-140 | 10 | 100 | 900 | −140 | −30 | 100 |
| 10T0-150 | 10 | 100 | 900 | −150 | −30 | 100 |
| 10Tg-45 | 10 | 100 | 900 | −130 | −45 | 100 |
| 10Tg-60 | 10 | 100 | 900 | −130 | −60 | 100 |
| 10N75 | 10 | 100 | 900 | −130 | −30 | 75 |
| 10N50 | 10 | 100 | 900 | −130 | −30 | 50 |
| The mass transfer coefficient is calculated according to in which and are taken from Winkelman et al. [25], and is fitted to experimental results. |
| The binary diffusivity for N2/CO2 mixtures is calculated according to the Fuller–Schettler–Giddings correlation [26]: in which is the sum of atomic diffusion volumes, which is 26.9 for CO2 and 17.9 for N2. |
| The CO2 mole fraction at the interface is the equilibrium value at the surface temperature [27] is defined as follows: |
| The gas-to-solid heat transfer coefficient is coupled to the mass transfer coefficient , according to the Chilton–Colburn analogy, as follows: |
| The physical properties of the gas phase were computed at the average gas phase temperature according to Reid et al. [28], using the pure component data supplied by Daubert and Danner [27]. |
| Particle diameter | ||
| Particle material | Glass | |
| Solids material | ||
| Heat capacity | ||
| Initial particle temperature | ||
| Gas phase temperature | ||
| Gas phase mole fraction CO2 | ||
| Gas mass flow |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Tuinier, M.J.; Ververs, W.J.R.; Tešić, D.; Roghair, I.; van Sint Annaland, M. Mass Deposition Rates of Carbon Dioxide onto a Cryogenically Cooled Surface. Processes 2026, 14, 271. https://doi.org/10.3390/pr14020271
Tuinier MJ, Ververs WJR, Tešić D, Roghair I, van Sint Annaland M. Mass Deposition Rates of Carbon Dioxide onto a Cryogenically Cooled Surface. Processes. 2026; 14(2):271. https://doi.org/10.3390/pr14020271
Chicago/Turabian StyleTuinier, Martin Jan, Wout Jacob René Ververs, Danica Tešić, Ivo Roghair, and Martin van Sint Annaland. 2026. "Mass Deposition Rates of Carbon Dioxide onto a Cryogenically Cooled Surface" Processes 14, no. 2: 271. https://doi.org/10.3390/pr14020271
APA StyleTuinier, M. J., Ververs, W. J. R., Tešić, D., Roghair, I., & van Sint Annaland, M. (2026). Mass Deposition Rates of Carbon Dioxide onto a Cryogenically Cooled Surface. Processes, 14(2), 271. https://doi.org/10.3390/pr14020271







