Comparative Analysis of Coalbed Methane Well Productivity in Eastern Yunnan
Abstract
1. Introduction
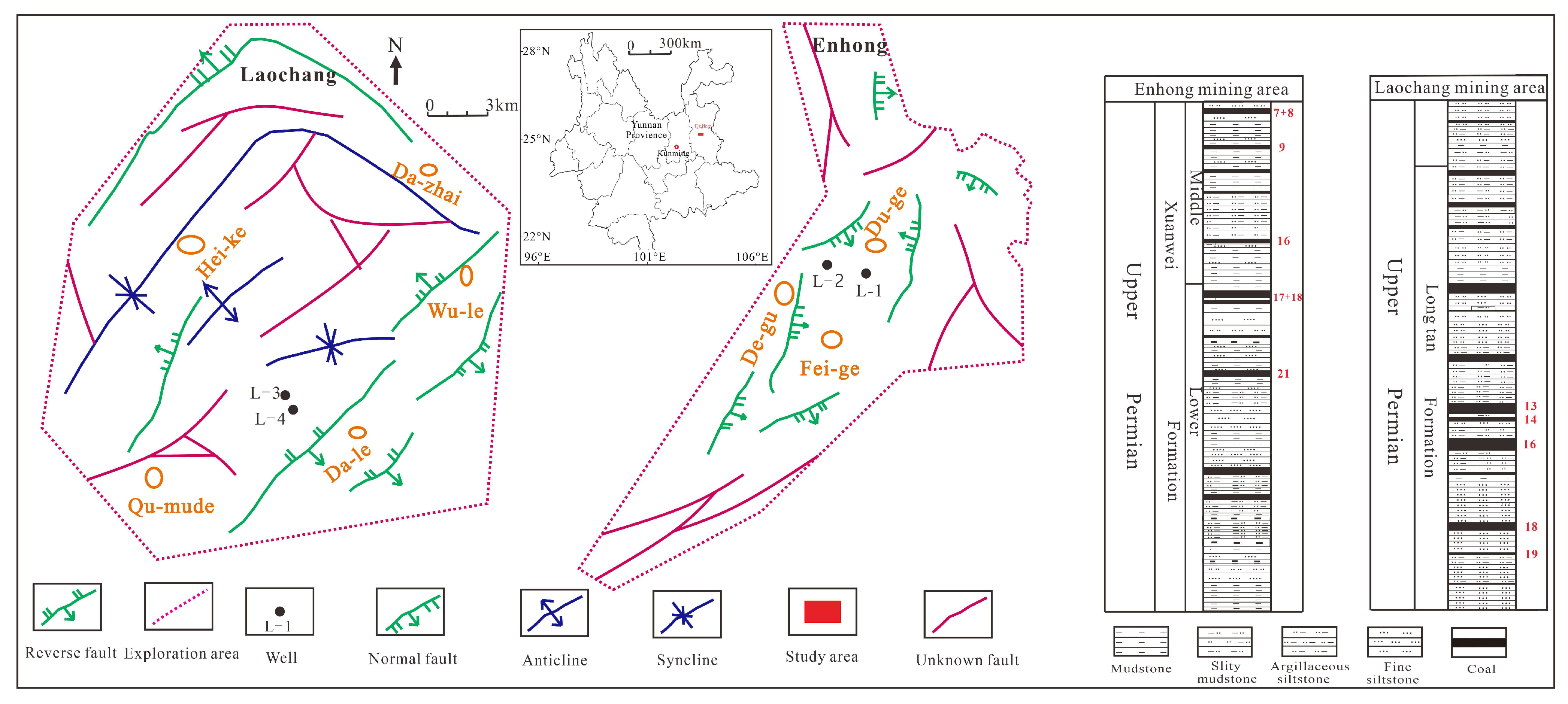
2. Geological Background
3. Test Methods and Results
4. Analysis and Discussion
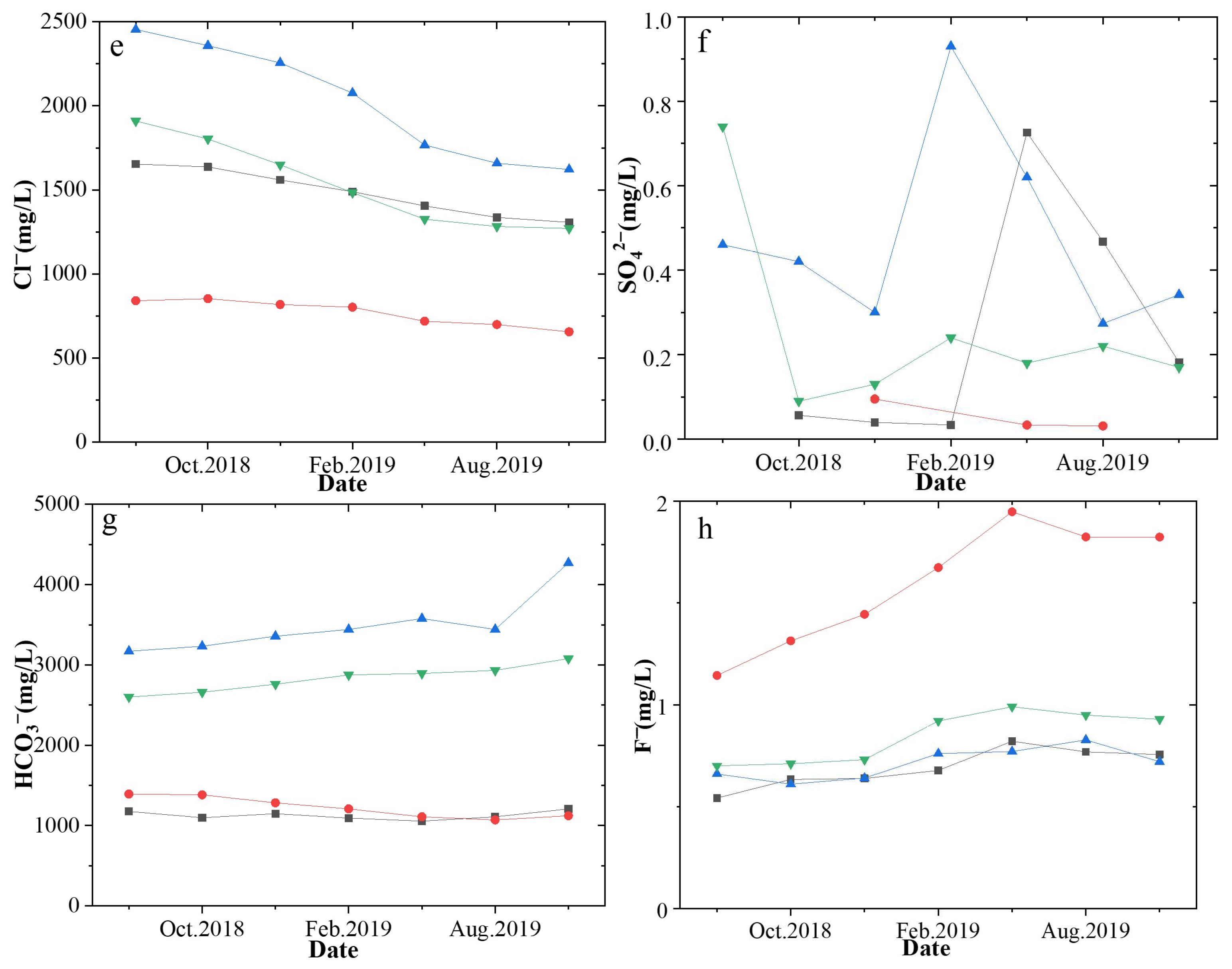
4.1. Variation in the Conventional Ion Concentrations and Their Effects on Productivity
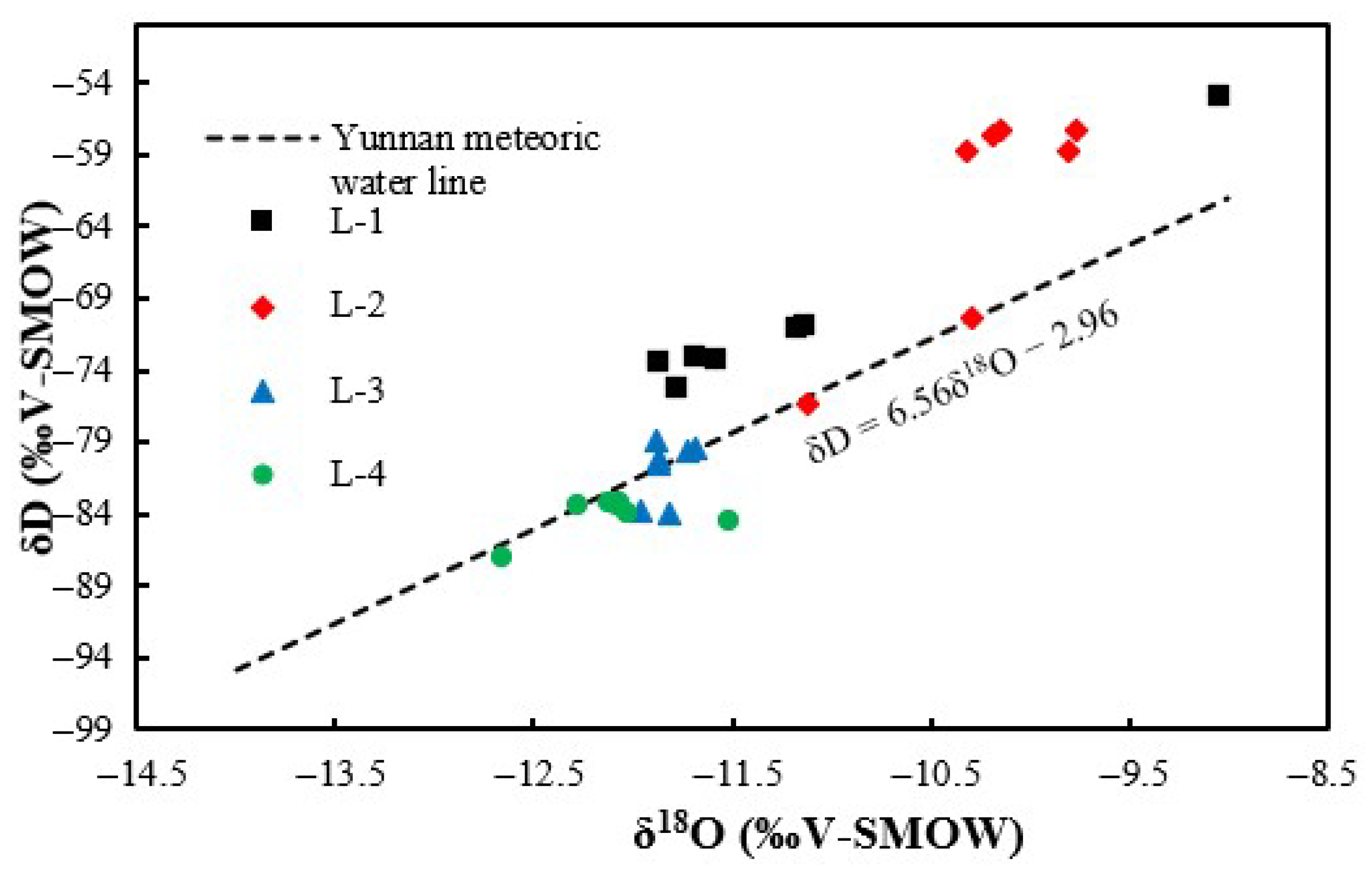
4.2. Variation in Hydrogen and Oxygen Isotopes and Their Effects on Productivity
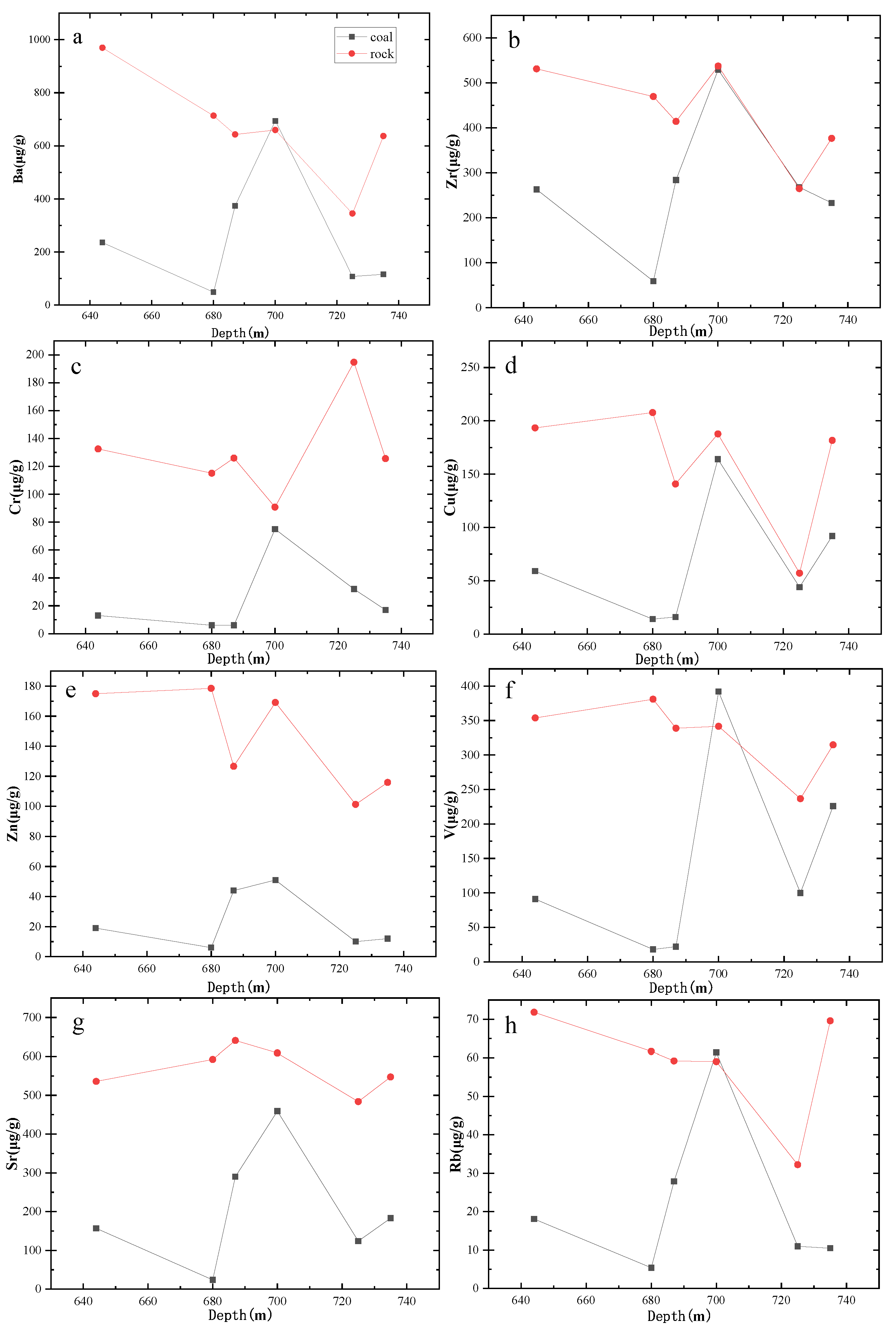
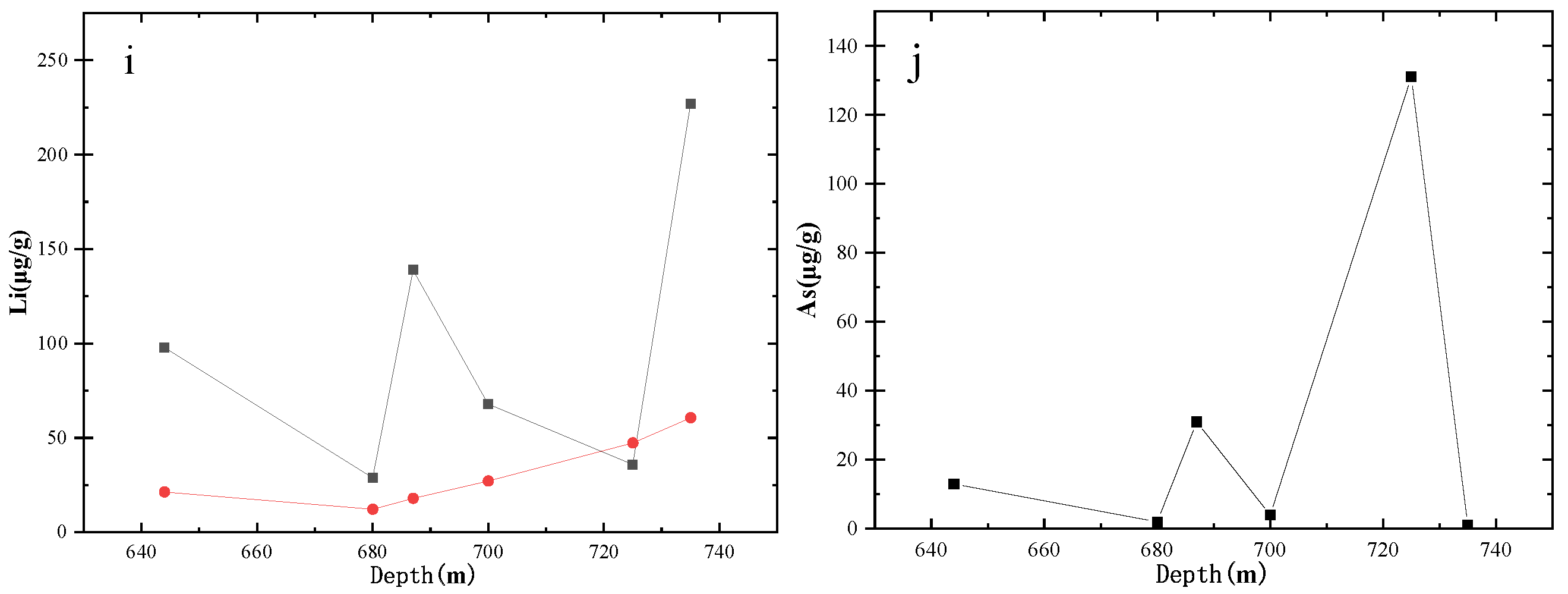
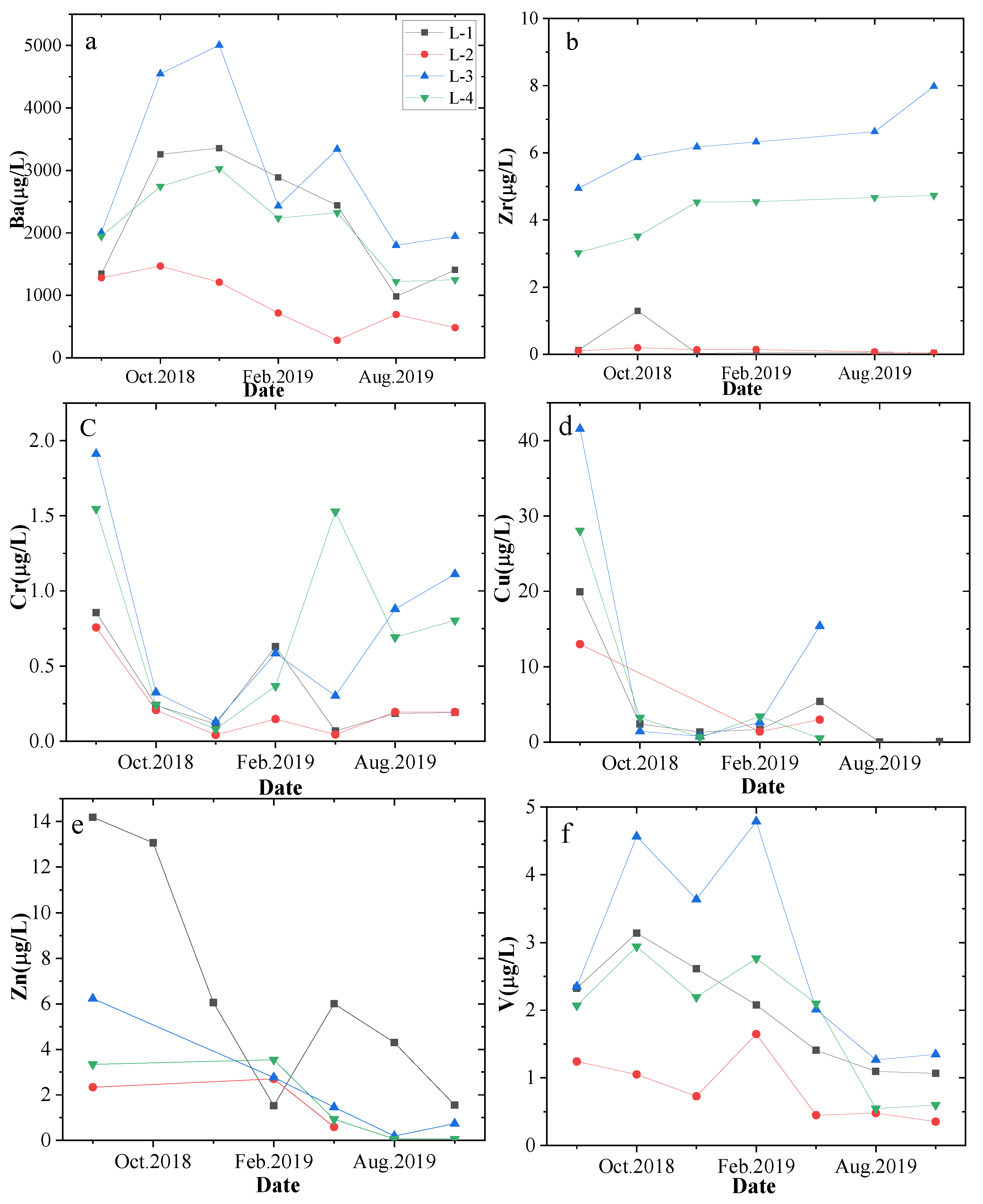
4.3. Variation in the Concentrations of Trace Elements and Their Effects on Productivity
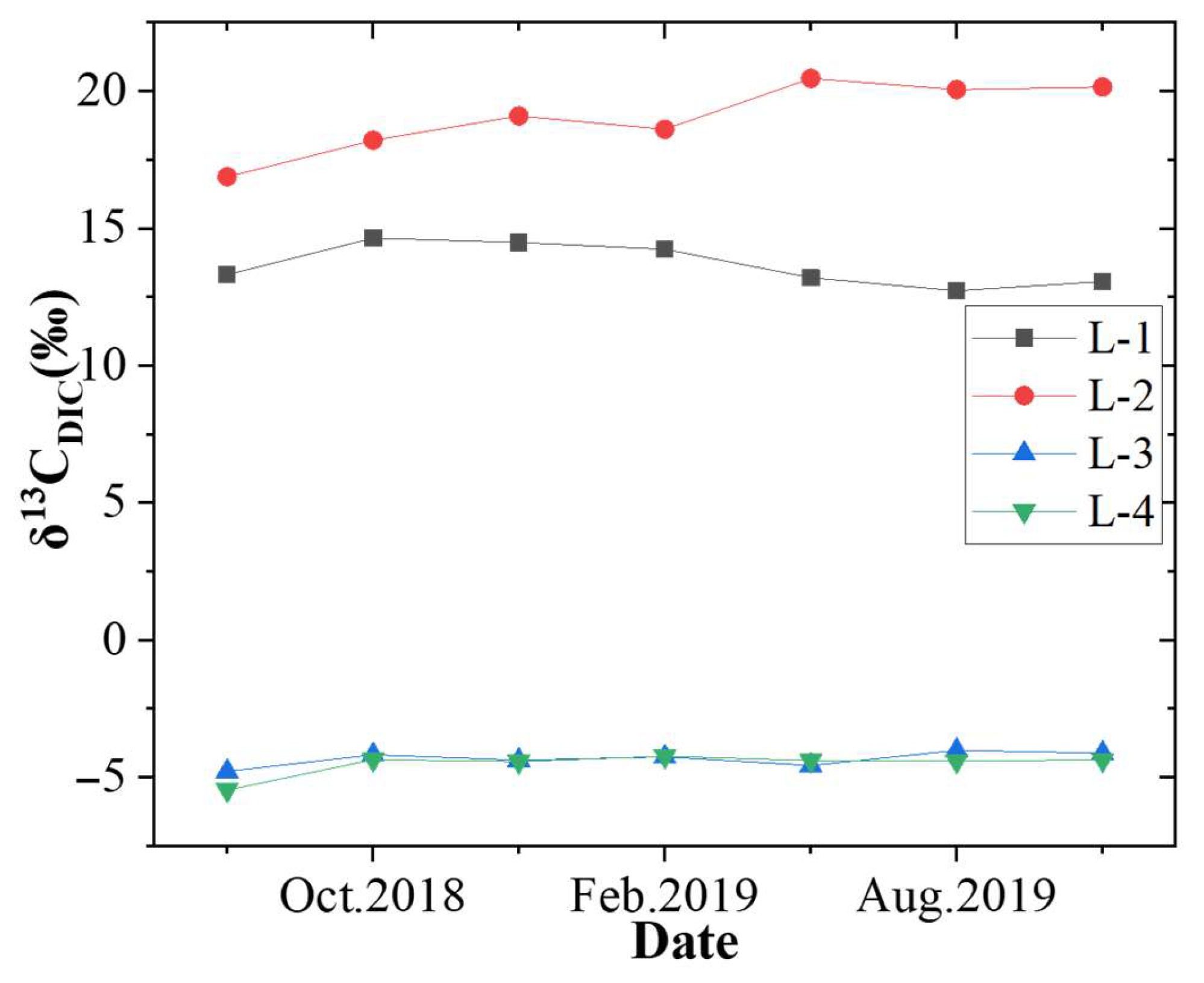
4.4. Variation in δ13CDIC and Its Effect on Productivity
5. Conclusions
- (1)
- The water from the selected four CBM wells is produced in a closed environment. The water produced from wells L-1 and L-2 is of the Na-Cl-HCO3 type, whereas the water produced from wells L-3 and L-4 is of the Na-HCO3 type. The concentration of HCO3− is positively correlated with gas production; however, when the concentration of HCO3− exceeds 2800 mg/L, the gas production decreases.
- (2)
- The isotope values of the water produced from wells L-1 and L-2 exhibit the characteristics of D drift, and the isotope values of the water produced from well L-3 and L-4 exhibit the characteristics of both D drift and 18O drift, which are indicative of good CBM production.
- (3)
- The contents of trace elements in the produced water are affected by the contents of trace elements in the coal seams and surrounding rocks, especially at the peak trace element concentrations in the rocks and coal seams.
- (4)
- The 13CDIC in the produced water is influenced by microbial methanogenesis and carbonate dissolution; when its value is approximately −4‰, gas production is high, which is beneficial for the production of CBM.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bachu, S.; Michael, K. Possible controls of hydrogeological and stress regimes on the producibility of coalbed methane in upper Cretaceous-Tertiary strata of the Alberta basin, Canada. AAPG Bull. 2003, 87, 1729–1754. [Google Scholar] [CrossRef]
- Pashin, J.; McIntyre-Redden, M.; Mann, S.; Kopaska-Merkel, D.; Varonka, M.; Orem, W. Relationships between water and gas chemistry in mature coalbed methane reservoirs of the Black Warrior Basin. Int. J. Coal Geol. 2014, 126, 92–105. [Google Scholar] [CrossRef]
- Qin, Y.; Shen, J.; Shi, R. Strategic Value and Choice on Construction of CMG Industry in China. J. China Coal Soc. 2022, 47, 371–387. [Google Scholar]
- Ye, J.; Wu, Q.; Wang, Z. Controlled characteristics of hydrogeological conditions on the coalbed methane migration and accumulation. J. China Coal Soc. 2001, 26, 459–462. [Google Scholar]
- Wei, M.; Ju, Y. Chemical characteristics and origin of produced waters from coalbed gas field in the southern of Qinshui Basin. J. China Coal Soc. 2015, 40, 629–635. [Google Scholar]
- Zhang, X.; Wu, C.; Liu, S. Characteristic analysis and fractal model of the gas-water relative permeability of coal under different confining pressures. J. Pet. Sci. Eng. 2017, 159, 488–496. [Google Scholar] [CrossRef]
- Zhang, L.; Kan, Z.; Zhang, C.; Tian, S.; Zeng, S.; Hao, D. Permeability and Stress Sensitivity of Coals with Different Fracture Directions Under Cyclic Loading-Unloading Conditions: A Case Study of the Xutuan Coal Mine in Huaibei Coalfield, China. Rock Mech. Rock Eng. 2025, 1–19. [Google Scholar] [CrossRef]
- Van Voast, W. Geochemical signature of formation waters as-sociated with coalbed methane. AAPG Bull. 2003, 87, 667–676. [Google Scholar]
- Taulis, M.; Milke, M. Chemical variability of groundwater samples collected from a coal seam gas exploration well, Maramarua, New Zealand. Water. Res. 2013, 47, 1021–1034. [Google Scholar] [CrossRef]
- Wu, C.; Yao, S.; Du, Y. Production systems optimization of a CBM well based on a time series BP neural network. J. China Univ. Min. Technol. 2015, 44, 64–69. [Google Scholar]
- Hou, X.; Zhang, J.; Liu, S.; Zhu, Y.; Hu, J. Estimation of multitype diffusion behaviors of CH4 and CO2 in coal under pore structure-induced restrictions. J. Chem. Eng. 2025, 525, 170144. [Google Scholar] [CrossRef]
- Wu, C.; Yang, Z.; Qin, Y.; Chen, J.; Zhang, Z.; Li, Y. Characteristics of Hydrogen and Oxygen Isotopes in Produced Water and Productivity Response of Coalbed Methane Wells in Western Guizhou. Energy Fuels 2018, 32, 11203–11211. [Google Scholar] [CrossRef]
- Li, C.; Tang, S.; Zhang, S.; Wang, S.; Li, Z. Chemical Characteristics and Significance of CBM well Produced Water in Shizhuang South Area, Qinshui Basin. Coal Geol. China 2013, 25, 25–29. [Google Scholar]
- Zhang, S.; Tang, S.; Li, Z.; Pan, Z.; Shi, W. Study of hydrochemical characteristics of CBM co-produced water of the Shizhuangnan Block in the southern Qinshui Basin, China, on its implication of CBM development. Int. J. Coal Geol. 2016, 159, 169–182. [Google Scholar] [CrossRef]
- Huang, H.; Sang, S.; Miao, Y.; Dong, Z.; Zhang, H. Trends of ionic concentration variations in water coproduced with coalbed methane in the Tiefa Basin. Int. J. Coal Geol. 2017, 182, 32–41. [Google Scholar] [CrossRef]
- Tao, S.; Pan, Z.; Tang, S.; Chen, S. Current status and geological conditions for the applicability of CBM drilling technologies in China: A review. Int. J. Coal Geol. 2019, 202, 95–108. [Google Scholar] [CrossRef]
- Han, J.; Xu, J.; Yi, L.; Chang, Z.; Wang, J.; Ma, H.; Zhang, B.; Jiang, H. Seasonal Interaction of River Water-Groundwater-Salt Lake Brine and Its Influence on Water-Salt Balance in the Nalenggele River Catchment in Qaidam Basin, NW China. J. Earth Sci. 2022, 33, 1298–1308. [Google Scholar] [CrossRef]
- Bao, Y.; Hu, Y.; Huang, H.; Meng, J.; Zheng, R. Evidence of Coal Biodegradation from Coalbed-Produced Water—A Case Study of Dafosi Gas Field, Ordos Basin, China. ACS Omega 2023, 8, 41885–41896. [Google Scholar] [CrossRef]
- Yang, M.; Ju, Y.; Liu, G.; Tong, L.; Kang, Y.; Hou, Q. Geochemical characters of water coproduced with coalbed gas and shallow groundwater in Liulin Coalfield of China. Acta Geol. Sin. 2013, 87, 1690–1700. [Google Scholar] [CrossRef]
- Li, Z.; Tang, S.; Wang, X.; Zheng, G.; Zhang, J. Relationship between water chemical composition and production of coalbed methane wells, Qinshui basin. J. China Univ. Min. Technol. 2011, 40, 424–429. [Google Scholar]
- Li, Z.; Luo, Z. Environmental impact of hydraulic fracturing on groundwater by isotope composition and hydrochemistry. Environ. Earth Sci. 2024, 83, 580. [Google Scholar] [CrossRef]
- Yang, Z.; Wu, C.; Zhang, Z.; Jin, J.; Zhao, L.; Li, Y. Geochemical significance of CBM produced water: A case study of developed test wells in Songhe block of Guizhou province. J. China Univ. Min. Technol. 2017, 46, 710–717. [Google Scholar]
- Yu, B. Chemical Characteristic of Produced Water of JunLian CBM Well and Its Intention. China Coalbed Methane 2015, 12, 32–35+8. [Google Scholar]
- Rice, C.; Flores, R.; Stricker, G.; Ellis, M. Chemical and stable isotopic evidence for water and rock interaction and biogenic origin of coalbed methane, Fort Union Formation, Powder River Basin, Wyoming and Montana U.S.A. Int. J. Coal Geol. 2008, 76, 76–85. [Google Scholar] [CrossRef]
- Rive, K.; Agrinier, P.; Gaillardet, J. DIC dynamics in stream water of volcanic settings: Climatic and lithology impact on the CO2 chemical weathering (Lesser Antilles, Reunion, French Massif Central and Iceland). In Proceedings of the AGU Fall Meeting Abstracts, San Francisco, CA, USA, 11–15 December 2006. [Google Scholar]
- Du, M.; Chen, S.; Wu, C. Geochemical characteristics and productivity evaluation of water produced from coalbed methane wells in the Laochang Block, Eastern Yunnan. ACS Omega 2023, 8, 47585–47594. [Google Scholar] [CrossRef]
- Dai, Q.; Zhang, L.; Zhang, K.; Chen, G.; Chen, Z.; Xue, X.; Wang, Y.; Wang, Z.; Di, C.; Zhao, F. Carbon-pricing optimization framework for oilfield carbon dioxide-water alternation with economic and environmental gains. Energy Convers. Manag. 2026, 348, 120613. [Google Scholar] [CrossRef]
- Li, Y.; Tang, S.; Zhang, S.; Xi, Z.; Wang, P. Biogeochemistry and Water-Rock Interactions of Coalbed Methane Co-Produced Water in the Shizhuangnan Block of the Southern Qinshui Basin, China. Water 2020, 12, 130. [Google Scholar] [CrossRef]
- Guo, C.; Qin, Y.; Wu, C.; Lu, L. Hydrogeological control and productivity modes of coalbed methane commingled production in multi-seam areas: A case study of the Bide-Santang Basin, western Guizhou, South China. J. Petrol. Sci. Eng. 2020, 189, 107039. [Google Scholar] [CrossRef]
- Zhang, Z.; Qin, Y.; Yang, Z.; Li, G.; You, Z. Primary Controlling Factors of Coalbed Methane Well Productivity and High Productive Well Patterns in Eastern Yunnan and Western Guizhou, China. Nat. Resour. Res. 2023, 32, 2711–2726. [Google Scholar] [CrossRef]
- Liu, S.; Zhang, D.; Lun, Z.; Zhao, C.; Wang, H. Occurrence of water within different rank coals: A review. Energy Sources Part A Recovery Util. Environ. Eff. 2024, 46, 10231–10249. [Google Scholar] [CrossRef]
- Baublys, K.; Hofmann, H.; Esterle, J.; Cendon, D.; Vink, S.; Golding, S. Geochemical influences on methanogenic groundwater from a low rank coal seam gas reservoir: Walloon Subgroup, Surat Basin. Int. J. Coal Geol. 2021, 246, 103841. [Google Scholar] [CrossRef]
- Birkle, P.; Rosillo Aragon, J. Evolution and orgin of deep reservoir water at the Activo Luna oil field, Gulf of Mexico, Mexico. AAPG Bull. 2002, 86, 457–484. [Google Scholar]
- Hu, H.; Wang, J. On Characteristics of Hydrogen and Oxygen Isotope in Precipitation in Yunnan and Analysis of Moisture Sources. J. Southwest China Norm. Univ. (Nat. Sci. Ed.) 2015, 40, 142–149. [Google Scholar]
- Chen, L.; Gui, H.; Yin, X.; Qian, J. Composing Characteristic of 18O and D and Current Field in Deep Groundwater. J. China Univ. Min. Technol. 2008, 37, 854–859. [Google Scholar]
- Mao, Q.; Wang, J.; Wang, J.; Li, W. Analysis of the Characteristics of δD and δ18O in the Meteoric Precipitation in Anshun, Guizhou Province and Beibei Chongqing. J. Southwest Univ. (Nat. Sci. Ed.) 2017, 39, 114–120. [Google Scholar]
- Bao, Y.; An, C.; Wang, C.; Guo, C.; Wang, W. Hydrogeochemical Characteristics and Water-Rock Interactions of Coalbed-Produced Water Derived from the Dafosi Biogenic Gas Field in the Southern Margin of Ordos Basin, China. Geofluids 2021, 2021, 5972497. [Google Scholar] [CrossRef]
- Ju, Y.; Xiao, L.; Wang, G.; Yang, M.; Sun, Y.; Zhang, S.; Yang, Z.; Li, Y.; Wu, H.; Wei, M.; et al. Water-gas-rock interactions and their variations in coal measures during evolution of the North China Craton Basin. GSA Bull. 2026, 138, 512–526. [Google Scholar] [CrossRef]
- Chen, P.; Kuang, H.; Tang, X. Research on the distribution and occurrence of arsenic in coal. J. China Coal Soc. 2002, 27, 259–263. [Google Scholar]
- Li, D.; Chen, K.; Deng, T.; Cheng, F.; Yang, J. Distribution and Enrichment Factors of Arsenic in Coals of Southwestern China. J. China Univ. Min. Technol. 2002, 31, 419–422. [Google Scholar]
- Wang, M.; Zheng, B.; Hu, J.; Li, S.; Wang, B. Distribution of arsenic in southwest coals. J. China Coal Soc. 2005, 30, 344–348. [Google Scholar]
- Du, M.; Zhao, M.; Wu, C. Hydrochemical characteristics and significance of coalbed water and coalbed methane well–produced water in the Suzhou mining area. Sci. Rep. 2025, 15, 43047. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Tang, S.; Chen, J.; Zhang, S.; Xi, Z. Geochemical Characteristics and Development Significances of Constant and Trace Elements from Coalbed Methane Co-Produced Water: A Case Study of the Shizhuangnan Block, the Southern Qinshui Basin. J. Earth Sci. 2024, 35, 51–61. [Google Scholar] [CrossRef]
- Lemay, T.; Konhauser, K. Water Chemistry of Coalbed Methane Reservoirs; Special Report; Alberta Energy and Utilities Board, EUB/AGS: Edmonton, AB, Canada, 2006. [Google Scholar]
- Chen, L.; Gui, H.; Yin, X. Composing characteristic and evolution law of carbon and oxygen stable isotopes in groundwater dissolved carbonate. J. China Coal Soc. 2008, 33, 537–542. [Google Scholar]
- Brinck, E.; Drever, J.; Frost, C. The geochemical evolution of water coproduced with coalbed natural gas in the Powder River Basin, Wyoming. Environ. Geosci. 2008, 15, 153–171. [Google Scholar] [CrossRef]
- McLaughlin, J.; Frost, C.; Sharma, S. Geochemical analysis of Atlantic Rim water, Carbon County, Wyoming: New applications for characterizing coalbed natural gas reservoirs. AAPG Bull. 2011, 95, 191–217. [Google Scholar] [CrossRef]
- Quillinan, S.; Frost, C. Carbon isotope characterization of powder river basin coal bed waters: Key to minimizing unnecessary water production and implications for exploration and production of biogenic gas. Int. J. Coal Geol. 2014, 126, 106–119. [Google Scholar] [CrossRef]
- Bao, Y.; Hao, Y.; Guo, Z.; Hu, Y.; Li, J.; Meng, J.; Wang, F. Hydrogeochemical Disparities and Constraints of Water Produced from Various Coal Seams in the Baode Block, Ordos Basin, China. ACS Omega 2024, 9, 4905–4919. [Google Scholar] [CrossRef]
- Guo, Z.; Bao, Y.; Wang, Y.; Yuan, Y.; Li, Z.; Wang, Y.; Xia, L.; Liu, W.; Ma, J. Hydrogeochemical characteristics and water-rock interaction mechanism of coalbed-produced water in the Linfen mining area, eastern margin of Ordos Basin, China. Front. Earth Sci. 2023, 10, 1108520. [Google Scholar] [CrossRef]

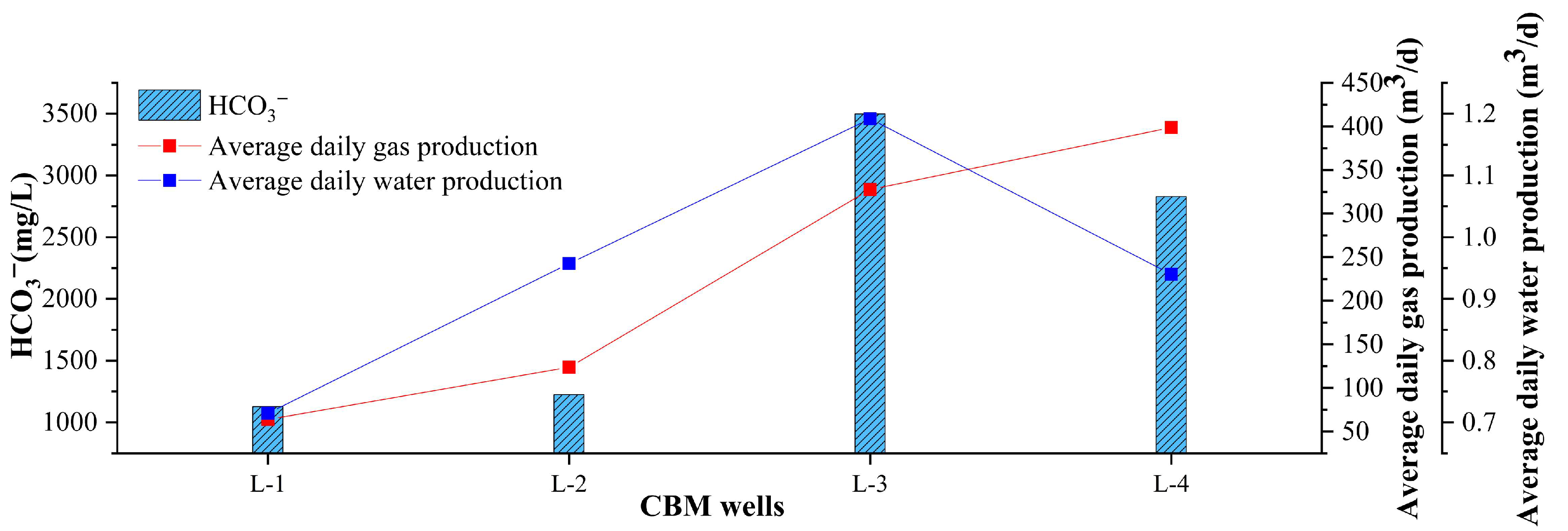




| CBM Wells | Main Coal Seam | Depth | Average Daily Gas Production | Average Daily Water Production |
|---|---|---|---|---|
| m | m3 | m3 | ||
| L-1 | 7 + 8/9/16/21 | 1182.90 | 63.90 | 0.71 |
| L-2 | 16/17 + 18 | 1031.00 | 123.77 | 0.96 |
| L-3 | 13/16/18/19 | 713.20 | 327.80 | 1.19 |
| L-4 | 14/16/18 | 745.80 | 398.81 | 0.94 |
| CBM Wells | Date | Na+ | K+ | Ca2+ | Mg2+ | Cl− | SO42− | HCO3− | F− | δ13CDIC |
|---|---|---|---|---|---|---|---|---|---|---|
| mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | ‰ | ||
| L-1 | Sep.2018 | 1490.96 | 11.72 | 15.46 | 5.88 | 1654.14 | - | 1176.83 | 0.54 | 13.31 |
| Oct.2018 | 1420.22 | 11.59 | 15.25 | 5.77 | 1637.03 | 0.06 | 1098.58 | 0.63 | 14.64 | |
| Nov.2018 | 1503.94 | 13.03 | 13.45 | 5.95 | 1559.26 | 0.04 | 1148.66 | 0.64 | 14.48 | |
| Feb.2019 | 1498.92 | 11.29 | 9.62 | 4.30 | 1489.55 | 0.03 | 1093.92 | 0.68 | 14.23 | |
| Jun.2019 | 1413.13 | 9.70 | 8.46 | 3.95 | 1405.09 | 0.73 | 1055.94 | 0.82 | 13.19 | |
| Aug.2019 | 1350.10 | 9.30 | 8.44 | 4.00 | 1336.11 | 0.47 | 1109.12 | 0.77 | 12.72 | |
| Oct.2019 | 1305.55 | 8.47 | 8.13 | 3.86 | 1306.94 | 0.18 | 1208.13 | 0.76 | 13.06 | |
| L-2 | Sep.2018 | 1057.86 | 4.49 | 5.65 | 1.59 | 841.12 | - | 1392.79 | 1.14 | 16.88 |
| Oct.2018 | 1044.70 | 4.41 | 5.52 | 1.52 | 853.38 | - | 1383.40 | 1.31 | 18.20 | |
| Nov.2018 | 1067.69 | 6.38 | 4.91 | 1.38 | 818.44 | 0.10 | 1283.24 | 1.44 | 19.09 | |
| Feb.2019 | 1056.26 | 4.67 | 4.18 | 0.91 | 802.58 | - | 1207.87 | 1.67 | 18.60 | |
| Jun.2019 | 773.43 | 3.27 | 3.14 | 0.65 | 719.18 | 0.03 | 1109.12 | 1.95 | 20.46 | |
| Aug.2019 | 926.41 | 4.04 | 3.67 | 0.85 | 699.19 | 0.03 | 1071.13 | 1.82 | 20.05 | |
| Oct.2019 | 877.18 | 3.91 | 3.88 | 0.71 | 656.30 | - | 1122.94 | 1.82 | 20.14 | |
| L-3 | Sep.2018 | 2564.84 | 206.35 | 17.49 | 5.46 | 2454.66 | 0.46 | 3170.55 | 0.66 | −4.78 |
| Oct.2018 | 2565.69 | 200.69 | 17.31 | 5.60 | 2358.30 | 0.42 | 3233.15 | 0.61 | −4.18 | |
| Nov.2018 | 2564.53 | 207.43 | 16.43 | 5.96 | 2255.89 | 0.30 | 3358.34 | 0.64 | −4.38 | |
| Feb.2019 | 2690.99 | 164.87 | 11.75 | 4.68 | 2076.53 | 0.93 | 3441.30 | 0.76 | −4.26 | |
| Jun.2019 | 2520.73 | 122.77 | 5.62 | 3.70 | 1766.93 | 0.62 | 3578.04 | 0.77 | −4.57 | |
| Aug.2019 | 2389.69 | 140.73 | 7.61 | 3.77 | 1658.57 | 0.27 | 3441.30 | 0.83 | −4.02 | |
| Oct.2019 | 2407.07 | 131.13 | 8.63 | 3.80 | 1621.63 | 0.34 | 4271.04 | 0.72 | −4.13 | |
| L-4 | Sep.2018 | 2006.94 | 197.32 | 11.07 | 4.38 | 1910.23 | 0.74 | 2600.91 | 0.70 | −5.46 |
| Oct.2018 | 2011.09 | 207.93 | 10.51 | 4.44 | 1803.25 | 0.09 | 2660.38 | 0.71 | −4.36 | |
| Nov.2018 | 2060.99 | 181.62 | 9.48 | 4.42 | 1649.12 | 0.13 | 2760.54 | 0.73 | −4.43 | |
| Feb.2019 | 2132.75 | 139.19 | 7.70 | 3.51 | 1484.26 | 0.24 | 2875.35 | 0.92 | −4.23 | |
| Jun.2019 | 2036.32 | 104.57 | 3.07 | 2.75 | 1325.50 | 0.18 | 2894.34 | 0.99 | −4.39 | |
| Aug.2019 | 2003.77 | 116.59 | 2.94 | 2.79 | 1282.47 | 0.22 | 2932.32 | 0.95 | −4.41 | |
| Oct.2019 | 1904.04 | 114.23 | 3.02 | 2.71 | 1271.20 | 0.17 | 3078.40 | 0.93 | −4.37 |
| Isotope | Date | CBM Wells | |||
|---|---|---|---|---|---|
| ‰ | L-1 | L-2 | L-3 | L-4 | |
| δ18O | Sep.2018 | −11.17 | −9.77 | −11.69 | −12.03 |
| Oct.2018 | −11.14 | −9.80 | −11.72 | −12.06 | |
| Nov.2018 | −9.04 | −10.29 | −11.82 | −11.52 | |
| Feb.2019 | −11.77 | −11.13 | −11.96 | −12.66 | |
| Jun.2019 | −11.58 | −10.18 | −11.87 | −12.12 | |
| Aug.2019 | −11.69 | −10.15 | −11.88 | −12.28 | |
| Oct.2019 | −11.86 | −10.32 | −11.87 | −12.09 | |
| δD | Sep.2018 | −71.15 | −57.37 | −79.46 | −83.96 |
| Oct.2018 | −70.89 | −58.74 | −79.57 | −83.23 | |
| Nov.2018 | −54.90 | −70.41 | −83.97 | −84.52 | |
| Feb.2019 | −75.28 | −76.35 | −83.77 | −86.98 | |
| Jun.2019 | −73.23 | −57.58 | −80.56 | −83.28 | |
| Aug.2019 | −73.16 | −57.26 | −78.87 | −83.50 | |
| Oct.2019 | −73.43 | −58.69 | −80.17 | −83.43 | |
| CBM Wells | Date | Ba | Zr | Cr | Cu | Zn | V | Sr | Rb | Li | As |
|---|---|---|---|---|---|---|---|---|---|---|---|
| μg/L | μg/L | μg/L | μg/L | μg/L | μg/L | μg/L | μg/L | μg/L | μg/L | ||
| L-1 | Sep.2018 | 1343.66 | 0.13 | 0.86 | 19.95 | 14.18 | 2.32 | 1953.35 | 38.03 | 84.43 | 1.36 |
| Oct.2018 | 3257.16 | 1.30 | 0.24 | 2.40 | 13.07 | 3.14 | 4619.41 | 41.08 | 48.83 | 0.63 | |
| Nov.2018 | 3356.30 | 0.03 | 0.12 | 1.32 | 6.06 | 2.61 | 4473.98 | 38.63 | 50.16 | 0.31 | |
| Feb.2019 | 2887.99 | 0.05 | 0.63 | 1.66 | 1.52 | 2.08 | 3582.61 | 37.14 | 47.82 | 0.57 | |
| Jun.2019 | 2439.64 | - | 0.07 | 5.37 | 6.01 | 1.41 | - | - | 36.78 | 0.90 | |
| Aug.2019 | 980.00 | 0.06 | 0.19 | 0.01 | 4.30 | 1.10 | 2601.40 | 17.87 | 38.31 | 0.93 | |
| Oct.2019 | 1405.60 | 0.04 | 0.19 | 0.07 | 1.55 | 1.07 | 2577.71 | 17.68 | 39.99 | 0.97 | |
| L-2 | Sep.2018 | 1279.94 | 0.10 | 0.76 | 12.99 | 2.33 | 1.24 | 612.50 | 7.93 | 36.97 | 1.86 |
| Oct.2018 | 1467.17 | 0.20 | 0.21 | - | - | 1.05 | 1434.71 | 9.02 | 22.05 | 1.01 | |
| Nov.2018 | 1209.30 | 0.14 | 0.04 | - | - | 0.73 | 1300.08 | 8.60 | 26.27 | 1.18 | |
| Feb.2019 | 716.25 | 0.15 | 0.15 | 1.39 | 2.70 | 1.65 | 765.41 | 6.08 | 3.65 | 2.70 | |
| Jun.2019 | 277.91 | - | 0.05 | 2.96 | 0.59 | 0.45 | - | - | 17.62 | 1.38 | |
| Aug.2019 | 692.15 | 0.07 | 0.20 | - | - | 0.48 | 814.57 | 3.80 | 18.85 | 0.73 | |
| Oct.2019 | 482.14 | 0.04 | 0.20 | - | - | 0.35 | 740.95 | 4.01 | 18.04 | 0.49 | |
| L-3 | Sep.2018 | 2004.51 | 4.94 | 1.91 | 41.54 | 6.23 | 2.35 | 5542.35 | 62.54 | 964.92 | 2.78 |
| Oct.2018 | 4546.07 | 5.86 | 0.32 | 1.43 | - | 4.56 | 13,423.98 | 78.17 | 569.54 | 1.35 | |
| Nov.2018 | 5004.39 | 6.18 | 0.13 | 0.77 | - | 3.64 | 12,929.26 | 76.13 | 751.36 | 1.38 | |
| Feb.2019 | 2429.18 | 6.33 | 0.58 | 2.62 | 2.76 | 4.79 | 6691.05 | 45.61 | 105.74 | 5.68 | |
| Jun.2019 | 3339.49 | - | 0.30 | 15.38 | 1.46 | 2.01 | - | - | 554.50 | 3.27 | |
| Aug.2019 | 1798.80 | 6.63 | 0.88 | - | 0.19 | 1.27 | 6304.88 | 41.75 | 609.04 | 2.52 | |
| Oct.2019 | 1945.22 | 7.98 | 1.11 | - | 0.74 | 1.35 | 7585.86 | 40.08 | 632.12 | 2.11 | |
| L-4 | Sep.2018 | 1941.72 | 3.02 | 1.55 | 28.05 | 3.34 | 2.07 | 4206.15 | 54.44 | 1284.98 | 1.18 |
| Oct.2018 | 2742.91 | 3.52 | 0.24 | 3.22 | - | 2.94 | 10,779.80 | 67.70 | 745.56 | 0.99 | |
| Nov.2018 | 3027.71 | 4.54 | 0.08 | 0.62 | - | 2.19 | 10,049.76 | 63.27 | 1001.60 | 0.70 | |
| Feb.2019 | 2236.59 | 4.55 | 0.37 | 3.40 | 3.54 | 2.76 | 6147.19 | 49.21 | 129.43 | 5.28 | |
| Jun.2019 | 2323.16 | - | 1.53 | 0.50 | 0.93 | 2.10 | - | - | 958.55 | 2.63 | |
| Aug.2019 | 1221.11 | 4.67 | 0.69 | - | 0.06 | 0.55 | 5177.17 | 31.45 | 803.72 | 2.98 | |
| Oct.2019 | 1248.33 | 4.73 | 0.80 | - | 0.06 | 0.60 | 5226.41 | 31.39 | 846.56 | 2.36 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Du, M.; Zhang, H.; Xia, X.; Zeng, A.; Jiang, W.; Wu, C. Comparative Analysis of Coalbed Methane Well Productivity in Eastern Yunnan. Processes 2026, 14, 270. https://doi.org/10.3390/pr14020270
Du M, Zhang H, Xia X, Zeng A, Jiang W, Wu C. Comparative Analysis of Coalbed Methane Well Productivity in Eastern Yunnan. Processes. 2026; 14(2):270. https://doi.org/10.3390/pr14020270
Chicago/Turabian StyleDu, Mingyang, Hui Zhang, Xiongfei Xia, Aiping Zeng, Wei Jiang, and Caifang Wu. 2026. "Comparative Analysis of Coalbed Methane Well Productivity in Eastern Yunnan" Processes 14, no. 2: 270. https://doi.org/10.3390/pr14020270
APA StyleDu, M., Zhang, H., Xia, X., Zeng, A., Jiang, W., & Wu, C. (2026). Comparative Analysis of Coalbed Methane Well Productivity in Eastern Yunnan. Processes, 14(2), 270. https://doi.org/10.3390/pr14020270






