Correlating Feed Characteristics and Catalyst Properties with Fluid Catalytic Cracking Performance
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Laboratory FCC ACE Experiments
3.2. Commercial FCC Experiments
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| A | Aromatic content, wt.% |
| ACE | Advanced catalytic evaluation |
| Basic N | Basic nitrogen |
| CA (n-d-M) | Aromatic carbon content calculated by the n-d-M method |
| CA (Total) | Aromatic carbon content calculated by the total method |
| CCR | Conradson carbon content, wt.% |
| Conv | Conversion at a catalyst-to-oil ratio of 7.5 wt/wt |
| CTO | Catalyst-to-oil ratio, wt/wt |
| D15°C | Density at 15 °C, g/cm3 |
| GC | Gas chromatography |
| FBP | Final boiling point |
| FCC | Fluid catalytic cracking |
| H-Oil | H-oil hydrocracking unit |
| H (Total) | Hydrogen content calculated by the total method |
| HCO | Heavy cycle oil |
| HDS | Hydrodesulfurization |
| HS-RGA | Hi-speed refinery gas analyzer |
| IBP | Initial boiling point |
| ICrA | Intercriteria analysis |
| IFP | Intuitionistic fuzzy pair |
| LCO | Light cycle oil |
| KW | Watson characterizing factor |
| MA | Micro-activity of a catalyst, wt.% |
| MSA | Specific area of matrix, m2/g |
| MW | Molecular weight, g/mol |
| n | Reaction order |
| N | Nitrogen content, wt.% |
| Naph | Naphthene content, wt.% |
| P | Paraffins content, wt.% |
| RE2O3 | Oxides of rare earth elements |
| RI | Refractive index |
| SA | Specific area, m2/g |
| SimDis | Simulated distillation |
| TOS | Time on stream |
| UCS | Unit sell size, Å |
| UOP | Universal oil products company |
| VGO | Vacuum gas oil |
| WHSV | Weight hourly space velocity |
| ZSA | Specific area of zeolite, m2/g |
| k | Apparent kinetic constant; h−n × frac.−1 |
| τ | Reaction time, hours |
| X | Feed conversion, wt.% |
References
- Fletcher, R.P. The History of Fluidized Catalytic Cracking: A History of Innovation: 1942–2008. In Innovations in Industrial and Engineering Chemistry; American Chemical Society: Washington, DC, USA, 2009; ACS Symposium Series; Volume 1000, pp. 189–249. [Google Scholar]
- Vogt, E.T.C.; Weckhuysen, B.M. Fluid catalytic cracking: Recent developments on the grand old lady of zeolite catalysis. Chem. Soc. Rev. 2015, 44, 7342–7370. [Google Scholar] [CrossRef]
- From the Beginning: 80 Years of FCC. Available online: https://grace.com/insights/80-years-of-fcc/ (accessed on 22 December 2025).
- Büchele, M. Heavy Residues and Pyrolysis Oils as Feedstocks in the FCC Process for a More Sustainable Production of Olefins and High Octane Gasoline. Ph.D. Thesis, TU Wien, Faculty of Mechanical and Industrial Engineering, Institute of Chemical, Environmental and Bioscience Engineering, Vienna, Austria, August 2022. [Google Scholar]
- Mammadova, T.; Abbasov, M.; Movsumov, N.; Latifova, T.; Hasanova, A.; Kocharli, Z.; Khalafova, I.; Abbasov, V. Production of diesel fractions by catalytic cracking of vacuum gas oil and its mixture with cottonseed oil under the influence of a magnetic field. Egypt. J. Pet. 2018, 27, 1029–1033. [Google Scholar] [CrossRef]
- Mizuno, T.; Yamazaki, H.; Takamiya, Y.; Hasegawa, H.; Tanaka, C.; Mitsui, T. Effects of the FCC catalyst binder type on propylene production during catalytic cracking of VGO. Appl. Catal. A Gen. 2023, 661, 119214. [Google Scholar] [CrossRef]
- Contreras, R.O.; Leirós, M.B.; Olong, N.; Brandt, S. Co-processing of chemical recycling products in FCC units. Digital Refining, March 2025; pp. 15–18. [Google Scholar]
- Fisher, L.P. Effect of feedstock variability on catalytic cracking yields. Appl. Catal. 1990, 65, 189–210. [Google Scholar] [CrossRef]
- Ng, S.H.; Rahimi, P.M. Catalytic Cracking of Canadian Nonconventional Feedstocks. 1. Cracking Characteristics of Gas Oils Derived from Coprocessing Distillate and Shale Oil. Energy Fuels 1991, 5, 595–601. [Google Scholar] [CrossRef]
- Ng, S.H.; Wang, J.; Fairbridge, C.; Zhu, Y.; Yang, L.; Ding, F.; Yui, S. Study of Canadian FCC feeds from various origins and treatments. 1. Ranking of feedstocks based on feed quality and product distribution. Energy Fuels 2004, 8, 160–171. [Google Scholar] [CrossRef]
- Zhao, Y.; Guo, X.; Fang, Y. Coprocessing of cashew nut shell liquid and phenol model compounds with VGO in a pilot-scale FCC riser. Energy 2024, 307, 132764. [Google Scholar] [CrossRef]
- Stratiev, D. Evaluation of Feedstock Characteristics Determined by Different Methods and Their Relationships to the Crackability of Petroleum, Vegetable, Biomass, and Waste-Derived Oils Used as Feedstocks for Fluid Catalytic Cracking: A Systematic Review. Processes 2025, 13, 2169. [Google Scholar] [CrossRef]
- Lappas, A.A.; Iatridis, D.K.; Vasalos, I.A. Production of reformulated gasoline in the FCC unit. Effect of feedstock type on gasoline composition. Catal. Today 1999, 50, 73–85. [Google Scholar] [CrossRef]
- Navarro, U.; Ni, M.; Orlicki, D. Understanding the potential for FCC feed to generate valuable products and how this knowledge can benefit refinery operation. PTQ/Digital Refining, March 2015; pp. 1–12. [Google Scholar]
- Schiller, R.; Chau, C. The fastest route to higher octanes. Digital Refining, May 2016.
- Moncrief, M.; Hunt, D.; Stafford, K. Worldwide FCC equilibrium catalyst trends—A ten-year review. Catalagram 2007, 102, 20–27. [Google Scholar]
- Pope, J.; Clough, M.; Shackleford, A. Lessons from FCC history. Digital Refining, April 2017.
- ASTM D4052-22; Standard Test Method for Density, Relative Density, and API Gravity of Liquids by Digital Density Meter. ASTM International: West Conshohocken, PA, USA, 2022.
- ASTM D1747-09(2019); Standard Test Method for Refractive Index of Viscous Materials. ASTM International: West Conshohocken, PA, USA, 2019.
- Goossens, A.G. Prediction of Molecular Weight of Petroleum Fractions. Ind. Eng. Chem. Res. 1996, 35, 985–988. [Google Scholar] [CrossRef]
- ASTM D4294-21; Standard Test Method for Sulfur in Petroleum and Petroleum Products by Energy Dispersive X-Ray Fluorescence Spectrometry. ASTM International: West Conshohocken, PA, USA, 2021.
- ASTM D3228-96; Standard Test Method for Total Nitrogen in Lubricating Oils and Fuel Oils by Modified Kjeldahl Method. ASTM International: West Conshohocken, PA, USA, 1996.
- ASTM UOP269-10; S Nitrogen Bases in Hydrocarbons by Potentiometric Titration. ASTM International: West Conshohocken, PA, USA, 2010.
- ASTM D445-24; Standard Test Method for Kinematic Viscosity of Transparent and Opaque Liquids (and Calculation of Dynamic Viscosity). ASTM International: West Conshohocken, PA, USA, 2024.
- ASTM D189-06(2019); Standard Test Method for Conradson Carbon Residue of Petroleum Products. ASTM International: West Conshohocken, PA, USA, 2019.
- ASTM D7169-20e1; Standard Test Method for Boiling Point Distribution of Samples with Residues Such as Crude Oils and Atmospheric and Vacuum Residues by High Temperature Gas Chromatography. ASTM International: West Conshohocken, PA, USA, 2020.
- Gharagheizi, F.; Fazeli, A. Prediction of the Watson characterization factor of hydrocarbon components from molecular properties. QSAR Comb. Sci. 2008, 227, 758–767. [Google Scholar] [CrossRef]
- Stratiev, D.; Shishkova, I.; Nikolova, R.; Tsaneva, T.; Mitkova, M.; Yordanov, D. Investigation on precision of determination of sara analysis of vacuum residual oils from different origin. Pet. Coal 2016, 58, 109–119. [Google Scholar]
- ASTM D3238-17a; Standard Test Method for Calculation of Carbon Distribution and Structural Group Analysis of Petroleum Oils by the n-d-M Method. ASTM International: West Conshohocken, PA, USA, 2017.
- Riazi, M.R.; Daubert, T.E. Prediction of molecular-type analysis of petroleum fractions and coal liquids. Ind. Eng. Chem. Process. Dev. 1986, 25, 1009–1015. [Google Scholar] [CrossRef]
- Dhulesia, H. New Correlations Predict FCC Feed Characterizing Parameters. Oil Gas J. 1986, 84, 51–54. [Google Scholar]
- ASTM D7964/D7964M-19; Standard Test Method for Determining Activity of Fluid Catalytic Cracking (FCC) Catalysts in a Fluidized Bed. ASTM International: West Conshohocken, PA, USA, 2019.
- Vieira, R.C.; Pinto, J.C.; Biscaia, E.C.; Claudia, M.L.; Baptista, A.; Cerqueira, H.S. Simulation of catalytic cracking in a fixed-fluidized-bed unit. Ind. Eng. Chem. Res. 2004, 43, 6027–6034. [Google Scholar] [CrossRef]
- Stratiev, D.; Ivanov, M.; Chavdarov, I.; Argirov, G.; Strovegli, G. Revamping Fluid Catalytic Cracking Unit, and Optimizing Catalyst to Process Heavier Feeds. Appl. Sci. 2023, 13, 2017. [Google Scholar] [CrossRef]
- Stratiev, D.; Dinkov, R.; Shiskova, I.; Nedelchev, A.; Kolev, I.; Argirov, G.; Sotirov, S.; Sotirova, E.; Bureva, V.; Atanassov, K.; et al. Feed Variability Effect on Performance of a Commercial Residue Hydrocracker. Processes 2025, 13, 3486. [Google Scholar] [CrossRef]
- Atanassov, K.; Atanassova, V.; Gluhchev, G. Intercriteria analysis: Ideas and problems. Notes Intuitionistic Fuzzy Sets 2015, 21, 81–88. [Google Scholar]
- Ancheyta, J.; Sotelo, R. Kinetic modeling of vacuum gas oil catalytic cracking. Rev. Soc. Química México 2002, 46, 38–42. [Google Scholar]
- Shayegh, F.; Farshi, A.; Dehgan, A. A kinetics lumped model for VGO catalytic cracking in a fluidized bed reactor. Pet. Sci. Technol. 2012, 30, 945–957. [Google Scholar] [CrossRef]
- Hagelberg, P.; Eilos, I.; Hiltunen, J.; Lipiäinen, K.; Niemi, V.M.; Aittamaa, J.; Krause, A.O. Kinetics of catalytic cracking with short contact times. Appl. Catal A Gen. 2002, 223, 73–84. [Google Scholar] [CrossRef]
- Khongprom, P.; Ratchasombat, S.; Wanchan, W.; Bumphenkiattikulde, P.; Limtrakul, S. Scaling of catalytic cracking fluidized bed downer reactor based on CFD simulations—Part II: Effect of reactor scale. RSC Adv. 2022, 12, 21394. [Google Scholar] [CrossRef]
- Letzsch, W. Troubleshooting low or high regenerator temperatures. Digital Refining, April 2024; pp. 57–60. [Google Scholar]
- Sadeghbeigi, R. Fluid Catalytic Cracking Handbook. An Expert Guide to the Practical Operation, Design, and Optimization of FCC Units; Elsevier: Cambridge, MA, USA, 2020. [Google Scholar]
- Oloruntoba, A.; Zhang, Y.; Hsu, C.S. State-of-the-Art Review of Fluid Catalytic Cracking (FCC) Catalyst Regeneration Intensification Technologies. Energies 2022, 15, 2061. [Google Scholar] [CrossRef]
- Voltz, S.E.; Nace, D.M.; Weekman, V.W., Jr. Application of a Kinetic Model for Catalytic Cracking. Some Correlations of Rate Constants. Ind. Eng. Chem. Process Des. Develop. 1971, 10, 538–541. [Google Scholar] [CrossRef]
- Nace, D.M.; Voltz, S.E.; Weekman, V.W., Jr. Application of a Kinetic Model for Catalytic Cracking. Effects of Charge Stocks. Ind. Eng. Chem. Process Des. Develop. 1971, 10, 530–538. [Google Scholar] [CrossRef]
- Wear, C.C. Selectivity fundamentals. Catalagram 2009, 106, 3–9. [Google Scholar]
- Caeiro, G.; Costa, A.F.; Cerqueira, H.S.; Magnoux, P.; Lopes, J.M.; Matias, P.; Ribeiro, F.R. Nitrogen poisoning effect on the catalytic cracking of gasoil. Appl. Catal. A Gen. 2007, 320, 8–15. [Google Scholar] [CrossRef]
- Stratiev, D. Catalytic Cracking of Non-Hydrotreated, Hydrotreated and Sulfuric Acid-Treated Vacuum Gas Oils. Processes 2025, 13, 1351. [Google Scholar] [CrossRef]
- Fals, J.; Ospina-Castro, M.L.; Ramos-Hernández, A.; Pacheco-Londoño, L.; Bocanegra, S. Deactivation and regeneration dynamics in hierarchical zeolites: Coke characterization and impact on catalytic cracking of vacuum gas oil. Heliyon 2024, 10, e37813. [Google Scholar] [CrossRef] [PubMed]
- Cordero-Lanzac, T.; Bilbao, J. Deactivation kinetic models for the fluid catalytic cracking (FCC). A review. J. Chem. Eng. 2025, 514, 162856. [Google Scholar] [CrossRef]
- Stratiev, D.; Shishkova, I.; Ivanov, M.; Chavdarov, I.; Yordanov, D. Dependence of Fluid Catalytic Cracking Unit Performance on H-Oil Severity, Catalyst Activity, and Coke Selectivity. Chem. Eng. Technol. 2020, 43, 2266–2276. [Google Scholar] [CrossRef]

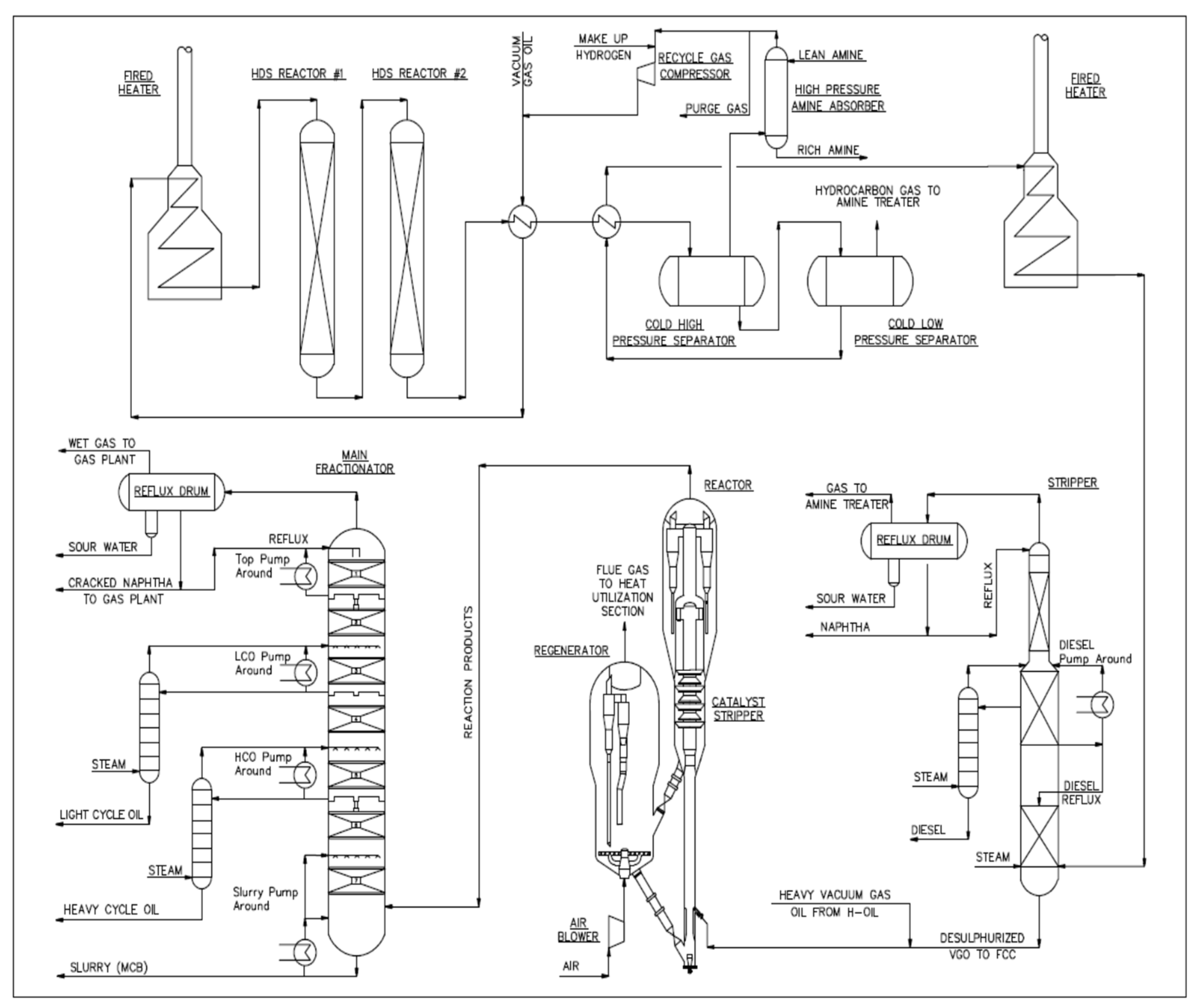
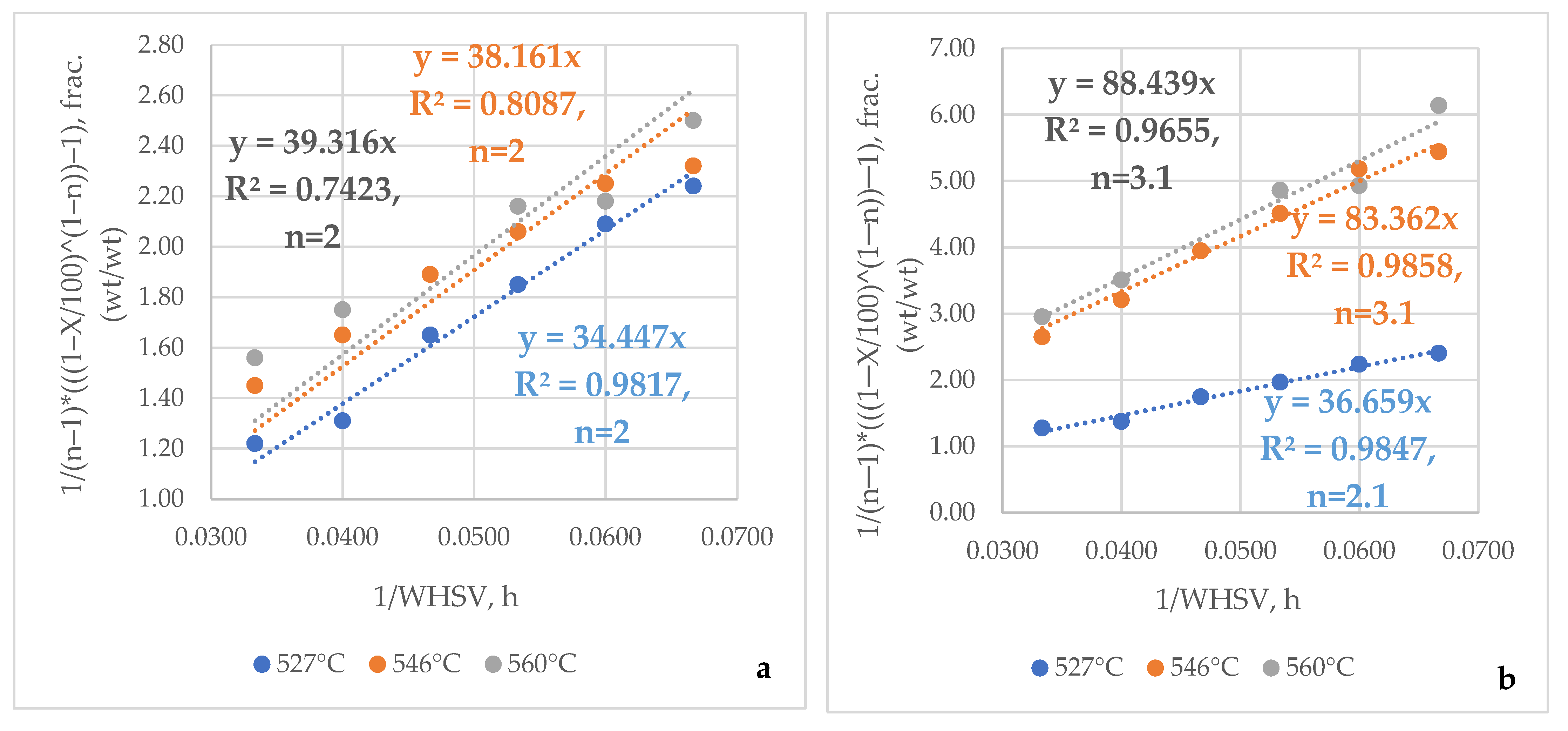






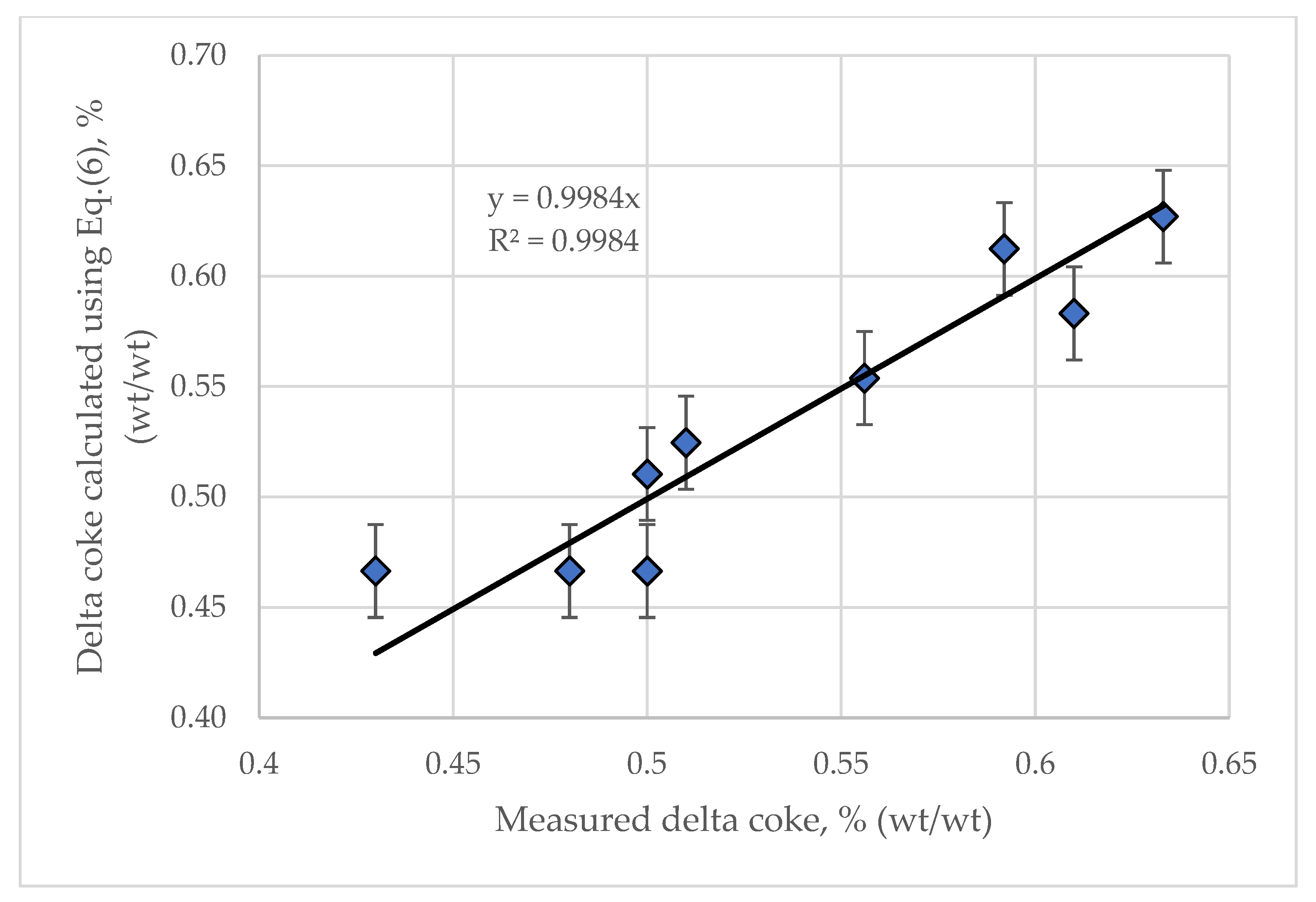

| FCC Feed Property | Method | Feed 1 | Feed 2 | Feed 3 | Feed 4 | Feed 5 | Feed 6 | Feed 7 | Feed 8 | Feed 9 | Feed 10 | Feed 11 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| D15°C, g/cm3 | ASTM D4052 [18] | 0.9355 | 0.9139 | 0.9174 | 0.9285 | 0.934 | 0.926 | 0.9181 | 0.973 | 0.901 | 0.920 | 0.914 |
| Refractive index at 20 °C | ASTM D1747 [19] | 1.5291 | 1.5143 | 1.5165 | 1.5267 | 1.5318 | 1.5282 | 1.5181 | 1.5613 | 1.5000 | 1.5106 | 1.5065 |
| Molecular Weight, g/mol | [20] | 366 | 345 | 378 | 358 | 407 | 325 | 316 | 583 | 367 | 388 | 342 |
| Sulfur, % (wt/wt) | ASTM D4294 [21] | 0.6089 | 0.26 | 0.37 | 0.5 | 0.683 | 0.612 | 0.471 | 0.985 | 0.176 | 1.695 | 1.608 |
| Nitrogen, % (wt/wt) | ASTM D3228 [22] | 0.27 | 0.20 | 0.19 | 0.27 | 0.35 | 0.36 | 0.33 | 0.25 | 0.08 | 0.15 | 0.14 |
| Basic N, ppm (wt/wt) | ASTM UOP269-10 [23] | 757 | 561 | 533 | 786 | 1099 | 1125 | 1009 | 701 | 224 | 421 | 398 |
| Viscosity (98.89 °C), cSt | ASTM D445 [24] | 9.6 | 7.1 | 8.9 | 8.5 | 12.8 | 6.8 | 6.2 | 152.4 | 7.4 | 9.7 | 7.0 |
| Concarbon, % | ASTM D189 [25] | 0.30 | 0.20 | 0.20 | 0.20 | 2.42 | 0.25 | 0.11 | 12.8 | 0.1 | 0.4 | 0.05 |
| Initial Boiling Point (IBP), °C | ASTM D7169 [26] | 299 | 310 | 310 | 308 | 338 | 255 | 277 | 224 | 282 | 311 | 303 |
| 5% Evaporated (wt/wt), °C | ASTM D7169 [26] | 345 | 341 | 343 | 341 | 383 | 310 | 336 | 463 | 335 | 356 | 343 |
| 10% Evaporated (wt/wt), °C | ASTM D7169 [26] | 365 | 356 | 360 | 357 | 402 | 331 | 357 | 534 | 357 | 377 | 361 |
| 50% Evaporated (wt/wt), °C | ASTM D7169 [26] | 450 | 429 | 453 | 442 | 476 | 417 | 408 | 573 | 441 | 460 | 427 |
| 90% Evaporated (wt/wt), °C | ASTM D7169 [26] | 525 | 510 | 518 | 516 | 540 | 524 | 475 | 614 | 528 | 541 | 482 |
| 95% Evaporated (wt/wt), °C | ASTM D7169 [26] | 541 | 527 | 529 | 529 | 555 | 544 | 499 | 681 | 550 | 562 | 496 |
| Final Boiling Point (FBP), °C | ASTM D7169 [26] | 588 | 649 | 561 | 568 | 607 | 615 | 576 | 711 | 613 | 628 | 551 |
| Kw-factor | [27] | 11.67 | 11.83 | 11.92 | 11.72 | 11.83 | 11.61 | 11.66 | 11.83 | 12.06 | 11.92 | 11.82 |
| Saturates, % (wt/wt) | In-house [28] | 47 | 58 | 56.6 | 49.0 | 44.3 | 47.3 | 48.8 | 30 | 55.7 | 50.6 | 53.1 |
| Aromatics, % (wt/wt) | In-house [28] | 47.1 | 34.3 | 41.3 | 46.4 | 53.1 | 50.2 | 49 | 63.3 | 43.5 | 46.8 | 45.3 |
| Light Aromatics, % (wt/wt) | In-house [28] | 13 | 13.8 | 14.5 | 14.0 | 15.5 | 12.7 | 19.5 | N.D. | 20.9 | 13.8 | 14.2 |
| Medium Aromatics, % (wt/wt) | In-house [28] | 10 | 13.8 | 6.7 | 8.4 | 11.3 | 8 | 7.4 | N.D. | 9.91 | 16.9 | 14.3 |
| Heavy Aromatics, % (wt/wt) | In-house [28] | 24.1 | 6.7 | 20.1 | 24.0 | 26.3 | 29.5 | 22.1 | N.D. | 12.6 | 16.1 | 16.8 |
| Resins, % (wt/wt) | In-house [28] | 5.9 | 20.1 | 2.1 | 4.6 | 2.6 | 2.5 | 2.2 | 6.7 | 0.9 | 2.6 | 1.6 |
| CA (n-d-M), % (wt/wt) | ASTM D3238 [29] | 32.0 | 26.0 | 25.9 | 32.7 | 34.6 | 36.4 | 29.3 | 47.0 | 15.3 | 22.9 | 21.6 |
| P, % (wt/wt) | [30] | 61.1 | 62.9 | 64.4 | 61.6 | 65.3 | 60.1 | 60.8 | 88.7 | 65.1 | 65.0 | 63.4 |
| Naph, % (wt/wt) | [30] | 21.9 | 23.3 | 21.7 | 22.1 | 18.4 | 23.4 | 24.3 | -4.4 | 23.7 | 22.0 | 24.0 |
| A, % (wt/wt) | [30] | 17.0 | 13.9 | 14.0 | 16.3 | 16.3 | 16.5 | 14.8 | 15.7 | 11.2 | 13.0 | 12.6 |
| CA (Total), % (wt/wt) | [31] | 25.1 | 20.4 | 20.2 | 25.3 | 25.5 | 27.6 | 22.8 | 24.4 | 13.0 | 15.1 | 15.1 |
| H (Total), % (wt/wt) | [31] | 11.8 | 12.4 | 12.3 | 12.0 | 12.0 | 11.9 | 12.2 | 12.1 | 12.8 | 12.3 | 12.4 |
| D15°C, g/cm3 | ASTM D4052 [18] | 0.907 | 0.910 | 0.916 | 0.909 | 0.964 | 0.939 | 0.978 | 0.919 | 0.922 | 0.921 | |
| Refractive Index at 20 °C | ASTM D1747 [19] | 1.5062 | 1.5111 | 1.5170 | 1.5111 | 1.5577 | 1.5393 | 1.5689 | 1.5132 | 1.5196 | 1.5190 | |
| Molecular Weight, g/mol | [20] | 362 | 356 | 365 | 354 | 295 | 268 | 401 | 366 | 348 | 385 | |
| Sulfur, % (wt/wt) | ASTM D4294 [21] | 0.164 | 0.294 | 0.269 | 0.381 | 0.480 | 0.369 | 0.666 | 0.386 | 0.321 | 0.283 | |
| Nitrogen, % (wt/wt) | ASTM D3228 [22] | 0.12 | 0.16 | 0.15 | 0.14 | 0.35 | 0.25 | 0.28 | 0.18 | 0.1657 | 0.940 | |
| Basic N, ppm (wt/wt) | ASTM UOP269-10 [23] | 313.59 | 414 | 393 | 367 | 1070 | 686 | 819 | 505 | 439 | 172 | |
| Viscosity (98.89 °C), cSt | ASTM D445 [24] | 7.4 | 7.4 | 8.1 | 7.2 | 7.1 | 5.4 | 21.1 | 8.3 | 7.6 | 9.6 | |
| Concarbon, % | ASTM D189 [25] | 0.1859 | 0.084 | 0.239 | 0.3911 | 0.34 | 0.15 | 2.67 | 0.2 | 0.07 | 0.51 | |
| Initial Boiling Point (IBP), °C | ASTM D7169 [26] | 303 | 291 | 287 | 282 | 254 | 234 | 213 | 285 | N.A. | N.A. | |
| 5% Evaporated (wt/wt), °C | ASTM D7169 [26] | 353 | 350 | 353 | 328 | 316 | 300 | 395 | 340 | 349 | 368 | |
| 10% Evaporated (wt/wt), °C | ASTM D7169 [26] | 371 | 369 | 371 | 350 | 336 | 322 | 416 | 361 | 365 | 387 | |
| 50% Evaporated (wt/wt), °C | ASTM D7169 [26] | 439 | 436 | 444 | 434 | 403 | 373 | 484 | 445 | 433 | 459 | |
| 90% Evaporated (wt/wt), °C | ASTM D7169 [26] | 517 | 514 | 529 | 518 | 479 | 433 | 541 | 526 | 513 | 564 | |
| 95% Evaporated (wt/wt), °C | ASTM D7169 [26] | 536 | 533 | 556 | 540 | 503 | 455 | 556 | 544 | 533 | 596 | |
| Final Boiling Point (FBP), °C | ASTM D7169 [26] | 584 | 588 | 607 | 555 | 517 | 595 | 598 | 576 | 688 | ||
| Kw-factor | [27] | 11.98 | 11.92 | 11.89 | 11.92 | 11.08 | 11.19 | 11.33 | 11.86 | 11.75 | 11.90 | |
| Saturates, % (wt/wt) | In-house [28] | N.D. | N.D. | N.D. | 56.4 | 35 | 45 | 31 | 53.9 | N.D. | N.D. | |
| Aromatics, % (wt/wt) | In-house [28] | N.D. | N.D. | N.D. | 42.5 | 62.2 | 52.7 | 66.5 | 43.7 | N.D. | N.D. | |
| Light Aromatics, % (wt/wt) | In-house [28] | N.D. | N.D. | N.D. | 18.0 | 10.3 | 17.1 | 13.1 | 17.6 | N.D. | N.D. | |
| Medium Aromatics, % (wt/wt) | In-house [28] | N.D. | N.D. | N.D. | 7.1 | 7.5 | 6.9 | 10.8 | 8.9 | N.D. | N.D. | |
| Heavy Aromatics, % (wt/wt) | In-house [28] | N.D. | N.D. | N.D. | 17.4 | 36.1 | 28.7 | 32.9 | 17.1 | N.D. | N.D. | |
| Resins, % (wt/wt) | In-house [28] | N.D. | N.D. | N.D. | 1.0 | 2.8 | 2.3 | 2.5 | 2.4 | N.D. | N.D. | |
| CA (n-d-M), % (wt/wt) | ASTM D3238 [29] | 25.4 | 31.4 | 37.2 | 32.5 | 75.9 | 62.7 | 82.0 | 29.0 | 38.2 | 36.7 | |
| P, % (wt/wt) | [30] | 64.5 | 64.0 | 63.8 | 64.3 | 47.9 | 52.8 | 59.7 | 63.1 | 61.7 | 64.3 | |
| Naph, % (wt/wt) | [30] | 23.2 | 22.9 | 22.1 | 22.8 | 27.3 | 27.3 | 16.0 | 23.0 | 23.2 | 21.2 | |
| A, % (wt/wt) | [30] | 12.3 | 13.1 | 14.1 | 12.9 | 24.8 | 19.9 | 24.3 | 13.9 | 15.1 | 14.5 | |
| CA (Total), % (wt/wt) | [31] | 16.2 | 18.8 | 21.2 | 19.1 | 40.8 | 33.8 | 39.9 | 18.3 | 22.3 | 20.9 | |
| H (Total), % (wt/wt) | [31] | 12.6 | 12.5 | 12.3 | 12.5 | 10.8 | 11.4 | 10.9 | 12.3 | 12.2 | 12.3 |
| Sample | Micro-Activity, % (wt/wt) | Al2O3, % (wt/wt) d.b. | RE2O3, % (wt/wt) d.b. | MgO, % (wt/wt) d.b. | P2O5, % (wt/wt) d.b. | Na2O, % (wt/wt) d.b. | SO4, % (wt/wt) | Carbon, % (wt/wt) | Ni, ppm (wt/wt) | V, ppm (wt/wt) | SA, m2/g | ZSA, m2/g | MSA, m2/g | UCS, Å |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Catalyst 1 | 73.0 | 45.2 | 1.0 | 0.5033 | 0.26 | 0.19 | N.A. | 0.13 | 99 | 208 | 138 | N.A. | N.A. | 24.28 |
| Catalyst 2 | 73.2 | 45.3 | 1.08 | 0.5033 | 0.23 | 0.19 | N.A. | 0.15 | 101 | 210 | 134 | N.A. | N.A. | 24.28 |
| Catalyst 3 | 73.8 | 45.5 | 1.36 | 0.4920 | 0.11 | 0.2 | N.A. | 0.18 | 101 | 210 | 139 | N.A. | N.A. | 24.28 |
| Catalyst 4 | 73.3 | 42.3 | 1.7 | 0.0486 | 0.07 | 0.26 | 0.06 | N.A. | N.A. | N.A. | 160 | 122 | 38 | 24.28 |
| Catalyst 5 | 73.2 | 44.3 | 1.8 | 0.0461 | 0.09 | 0.28 | 0.07 | N.A. | N.A. | N.A. | 161 | 124 | 37 | 24.282 |
| Catalyst 6 | 71.0 | 43.0 | 1.6 | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | 161 | 115 | 46 | 24.27 |
| Catalyst 7 | 75.2 | 40.5 | 2.7 | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | 187 | 158 | 29 | 24.309 |
| Catalyst 8 | 67.0 | 40.3 | 1.4 | N.A. | N.A. | 0.3 | N.A. | N.A. | N.A. | N.A. | 206 | 171 | 35 | 24.256 |
| Catalyst 9 | 71.0 | 40.6 | 1.9 | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | 192 | 155 | 37 | 24.276 |
| Catalyst 10 | 75.0 | 43.0 | 2.6 | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | 171 | 143 | 28 | 24.314 |
| Catalyst 11 | 73.5 | 42.2 | 2.5 | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | 191 | 151 | 40 | 24.282 |
| Catalyst 12 | 75.5 | 42.2 | 2.9 | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | 152 | 122 | 30 | 24.301 |
| Catalyst 13 | 76.3 | N.A. | 2.31 | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | 153 | 114 | 39 | 24.31 |
| Catalyst 14 | 72.8 | N.A. | 1.75 | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | 193 | 142 | 51 | 24.27 |
| Catalyst 15 | 75.2 | N.A. | 1.59 | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | 195 | 143 | 51 | 24.3 |
| Catalyst 16 | 74.2 | N.A. | 1.54 | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | 221 | 149 | 73 | 24.27 |
| Catalyst 17 | 70.3 | N.A. | 2.22 | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | 153 | 86 | 66 | 24.27 |
| Catalyst 18 | 73.5 | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | 24.28 |
| Catalyst 19 | 72.5 | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | 24.28 |
| Catalyst 20 | 73.3 | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. |
| Catalyst 21 | 76.0 | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. |
| μ | Conv | MA | Δ Coke | D15 | RI 20 °C | MW | N | Basic N | CCR | Kw | CA (n-d-M) | P | Naph | A | CA (Total) | H (Total) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Conv | 1.00 | 0.49 | 0.32 | 0.16 | 0.12 | 0.57 | 0.14 | 0.16 | 0.35 | 0.82 | 0.20 | 0.77 | 0.39 | 0.10 | 0.09 | 0.82 |
| MA | 0.49 | 1.00 | 0.63 | 0.49 | 0.47 | 0.40 | 0.49 | 0.50 | 0.39 | 0.38 | 0.48 | 0.36 | 0.46 | 0.49 | 0.50 | 0.40 |
| Δ Coke | 0.32 | 0.63 | 1.00 | 0.74 | 0.70 | 0.50 | 0.68 | 0.69 | 0.54 | 0.25 | 0.61 | 0.30 | 0.37 | 0.69 | 0.68 | 0.21 |
| D15 | 0.16 | 0.49 | 0.74 | 1.00 | 0.92 | 0.51 | 0.81 | 0.81 | 0.58 | 0.16 | 0.82 | 0.21 | 0.46 | 0.92 | 0.88 | 0.08 |
| RI 20 °C | 0.12 | 0.47 | 0.70 | 0.92 | 1.00 | 0.48 | 0.77 | 0.76 | 0.57 | 0.18 | 0.83 | 0.22 | 0.47 | 0.91 | 0.90 | 0.10 |
| MW | 0.57 | 0.40 | 0.50 | 0.51 | 0.48 | 1.00 | 0.43 | 0.43 | 0.62 | 0.63 | 0.44 | 0.68 | 0.08 | 0.45 | 0.42 | 0.55 |
| N | 0.14 | 0.49 | 0.68 | 0.81 | 0.77 | 0.43 | 1.00 | 0.98 | 0.49 | 0.20 | 0.70 | 0.23 | 0.54 | 0.82 | 0.82 | 0.21 |
| Basic N | 0.16 | 0.50 | 0.69 | 0.81 | 0.76 | 0.43 | 0.98 | 1.00 | 0.49 | 0.20 | 0.70 | 0.24 | 0.54 | 0.83 | 0.82 | 0.21 |
| CCR | 0.35 | 0.39 | 0.54 | 0.58 | 0.57 | 0.62 | 0.49 | 0.49 | 1.00 | 0.44 | 0.54 | 0.47 | 0.24 | 0.54 | 0.54 | 0.37 |
| Kw | 0.82 | 0.38 | 0.25 | 0.16 | 0.18 | 0.63 | 0.20 | 0.20 | 0.44 | 1.00 | 0.23 | 0.88 | 0.38 | 0.13 | 0.14 | 0.89 |
| CA (n-d-M) | 0.20 | 0.48 | 0.61 | 0.82 | 0.83 | 0.44 | 0.70 | 0.70 | 0.54 | 0.23 | 1.00 | 0.24 | 0.49 | 0.84 | 0.88 | 0.20 |
| P | 0.77 | 0.36 | 0.30 | 0.21 | 0.22 | 0.68 | 0.23 | 0.24 | 0.47 | 0.88 | 0.24 | 1.00 | 0.32 | 0.17 | 0.17 | 0.81 |
| Naph | 0.39 | 0.46 | 0.37 | 0.46 | 0.47 | 0.08 | 0.54 | 0.54 | 0.24 | 0.38 | 0.49 | 0.32 | 1.00 | 0.49 | 0.52 | 0.48 |
| A | 0.10 | 0.49 | 0.69 | 0.92 | 0.91 | 0.45 | 0.82 | 0.83 | 0.54 | 0.13 | 0.84 | 0.17 | 0.49 | 1.00 | 0.95 | 0.06 |
| CA (Total) | 0.09 | 0.50 | 0.68 | 0.88 | 0.90 | 0.42 | 0.82 | 0.82 | 0.54 | 0.14 | 0.88 | 0.17 | 0.52 | 0.95 | 1.00 | 0.11 |
| H (Total) | 0.82 | 0.40 | 0.21 | 0.08 | 0.10 | 0.55 | 0.21 | 0.21 | 0.37 | 0.89 | 0.20 | 0.81 | 0.48 | 0.06 | 0.11 | 1.00 |
| ν | Conv | MA | Δ Coke | D15 | RI 20 °C | MW | N | Basic N | CCR | Kw | CA (n-d-M) | P | Naph | A | CA (Total) | H (Total) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Conv | 0.00 | 0.44 | 0.64 | 0.79 | 0.79 | 0.36 | 0.79 | 0.79 | 0.47 | 0.11 | 0.74 | 0.14 | 0.50 | 0.84 | 0.85 | 0.12 |
| MA | 0.44 | 0.00 | 0.28 | 0.40 | 0.39 | 0.47 | 0.40 | 0.40 | 0.39 | 0.50 | 0.41 | 0.49 | 0.40 | 0.40 | 0.40 | 0.49 |
| Δ Coke | 0.64 | 0.28 | 0.00 | 0.18 | 0.19 | 0.40 | 0.24 | 0.24 | 0.27 | 0.66 | 0.31 | 0.59 | 0.50 | 0.23 | 0.24 | 0.71 |
| D15 | 0.79 | 0.40 | 0.18 | 0.00 | 0.04 | 0.47 | 0.18 | 0.18 | 0.30 | 0.82 | 0.17 | 0.72 | 0.47 | 0.06 | 0.11 | 0.90 |
| RI 20 °C | 0.79 | 0.39 | 0.19 | 0.04 | 0.00 | 0.47 | 0.18 | 0.20 | 0.27 | 0.78 | 0.11 | 0.71 | 0.46 | 0.04 | 0.06 | 0.85 |
| MW | 0.36 | 0.47 | 0.40 | 0.47 | 0.47 | 0.00 | 0.54 | 0.54 | 0.24 | 0.33 | 0.52 | 0.24 | 0.83 | 0.51 | 0.55 | 0.42 |
| N | 0.79 | 0.40 | 0.24 | 0.18 | 0.18 | 0.54 | 0.00 | 0.00 | 0.37 | 0.77 | 0.28 | 0.70 | 0.39 | 0.15 | 0.16 | 0.77 |
| Basic N | 0.79 | 0.40 | 0.24 | 0.18 | 0.20 | 0.54 | 0.00 | 0.00 | 0.37 | 0.78 | 0.29 | 0.71 | 0.40 | 0.17 | 0.18 | 0.79 |
| CCR | 0.47 | 0.39 | 0.27 | 0.30 | 0.27 | 0.24 | 0.37 | 0.37 | 0.00 | 0.42 | 0.32 | 0.35 | 0.57 | 0.33 | 0.33 | 0.49 |
| Kw | 0.11 | 0.50 | 0.66 | 0.82 | 0.78 | 0.33 | 0.77 | 0.78 | 0.42 | 0.00 | 0.73 | 0.06 | 0.54 | 0.84 | 0.84 | 0.08 |
| CA (n-d-M) | 0.74 | 0.41 | 0.31 | 0.17 | 0.11 | 0.52 | 0.28 | 0.29 | 0.32 | 0.73 | 0.00 | 0.70 | 0.44 | 0.14 | 0.11 | 0.78 |
| P | 0.14 | 0.49 | 0.59 | 0.72 | 0.71 | 0.24 | 0.70 | 0.71 | 0.35 | 0.06 | 0.70 | 0.00 | 0.59 | 0.77 | 0.78 | 0.14 |
| Naph | 0.50 | 0.40 | 0.50 | 0.47 | 0.46 | 0.83 | 0.39 | 0.40 | 0.57 | 0.54 | 0.44 | 0.59 | 0.00 | 0.44 | 0.43 | 0.45 |
| A | 0.84 | 0.4 | 0.23 | 0.06 | 0.04 | 0.51 | 0.15 | 0.17 | 0.33 | 0.84 | 0.14 | 0.8 | 0.4 | 0 | 0.04 | 0.93 |
| CA (Total) | 0.85 | 0.40 | 0.24 | 0.11 | 0.06 | 0.55 | 0.16 | 0.18 | 0.33 | 0.84 | 0.11 | 0.78 | 0.43 | 0.04 | 0.00 | 0.89 |
| H (Total) | 0.12 | 0.49 | 0.71 | 0.9 | 0.85 | 0.42 | 0.77 | 0.79 | 0.49 | 0.08 | 0.78 | 0.1 | 0.5 | 0.9 | 0.89 | 0 |
| High-Activity Catalyst | Low-Activity Catalyst | |
|---|---|---|
| Parameters | Refractory Feed (KW of 11.98 and N of 0.12% (wt/wt)) | |
| Micro-activity,% (wt/wt) | 76 | 72 |
| Δ Coke, % (wt/wt) | 0.63 | 0.55 |
| Catalyst-to-oil ratio, wt/wt | 7.71 | 8.84 |
| Conversion, % (wt/wt) | 74.7 | 73.0 |
| Coke yield, % (wt/wt) | 4.83 | 4.90 |
| Reactive feed (KW of 11.77 and N of 0.17% (wt/wt)) | ||
| Micro-activity, % (wt/wt) | 75.5 | 72 |
| Δ Coke, % (wt/wt) | 0.55 | 0.48 |
| Catalyst-to -oil ratio, wt/wt | 8.91 | 9.99 |
| Conversion, % (wt/wt) | 81.6 | 80.4 |
| Coke yield, % (wt/wt) | 4.90 | 4.80 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Stratiev, D.; Shiskova, I.; Ivanov, M.; Kolev, I.; Bureva, V.; Ribagin, S.; Atanassov, K. Correlating Feed Characteristics and Catalyst Properties with Fluid Catalytic Cracking Performance. Processes 2026, 14, 110. https://doi.org/10.3390/pr14010110
Stratiev D, Shiskova I, Ivanov M, Kolev I, Bureva V, Ribagin S, Atanassov K. Correlating Feed Characteristics and Catalyst Properties with Fluid Catalytic Cracking Performance. Processes. 2026; 14(1):110. https://doi.org/10.3390/pr14010110
Chicago/Turabian StyleStratiev, Dicho, Ivelina Shiskova, Mihail Ivanov, Iliyan Kolev, Veselina Bureva, Simeon Ribagin, and Krassimir Atanassov. 2026. "Correlating Feed Characteristics and Catalyst Properties with Fluid Catalytic Cracking Performance" Processes 14, no. 1: 110. https://doi.org/10.3390/pr14010110
APA StyleStratiev, D., Shiskova, I., Ivanov, M., Kolev, I., Bureva, V., Ribagin, S., & Atanassov, K. (2026). Correlating Feed Characteristics and Catalyst Properties with Fluid Catalytic Cracking Performance. Processes, 14(1), 110. https://doi.org/10.3390/pr14010110









