Stability and Foam Performance Optimization of CO2-Soluble Foaming Agents: Influencing Factors and Mechanistic Analysis
Abstract
1. Introduction
2. Experimental Materials and Methods
2.1. Materials
2.2. Equipment and Instruments
2.2.1. Dynamic Foam Analyzer (DFA-100)
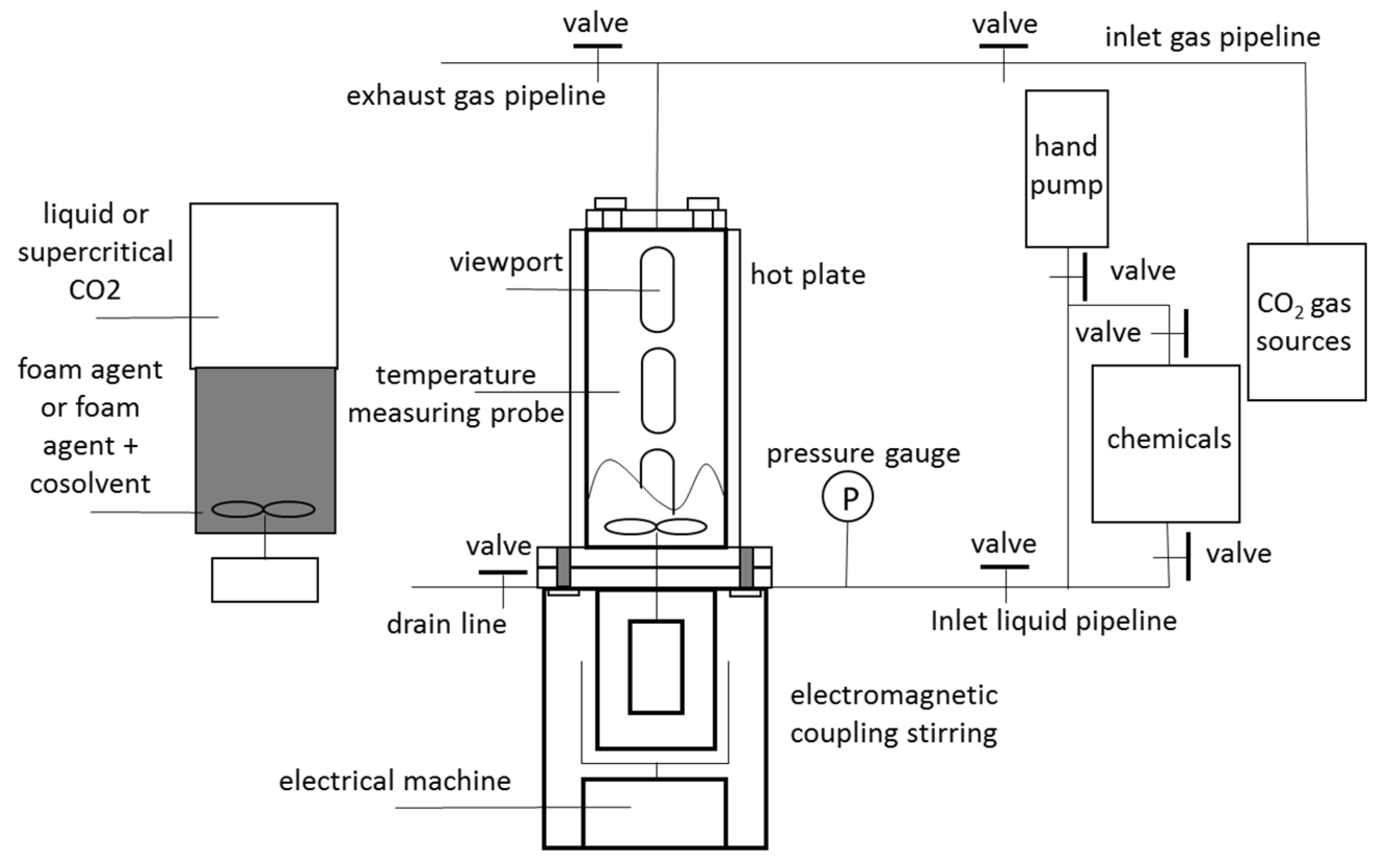
2.2.2. High-Temperature and High-Pressure Visual Foam Analyzer
2.2.3. High-Temperature and High-Pressure Interfacial Tensiometer
2.2.4. Foam Film Thickness Measurement Device
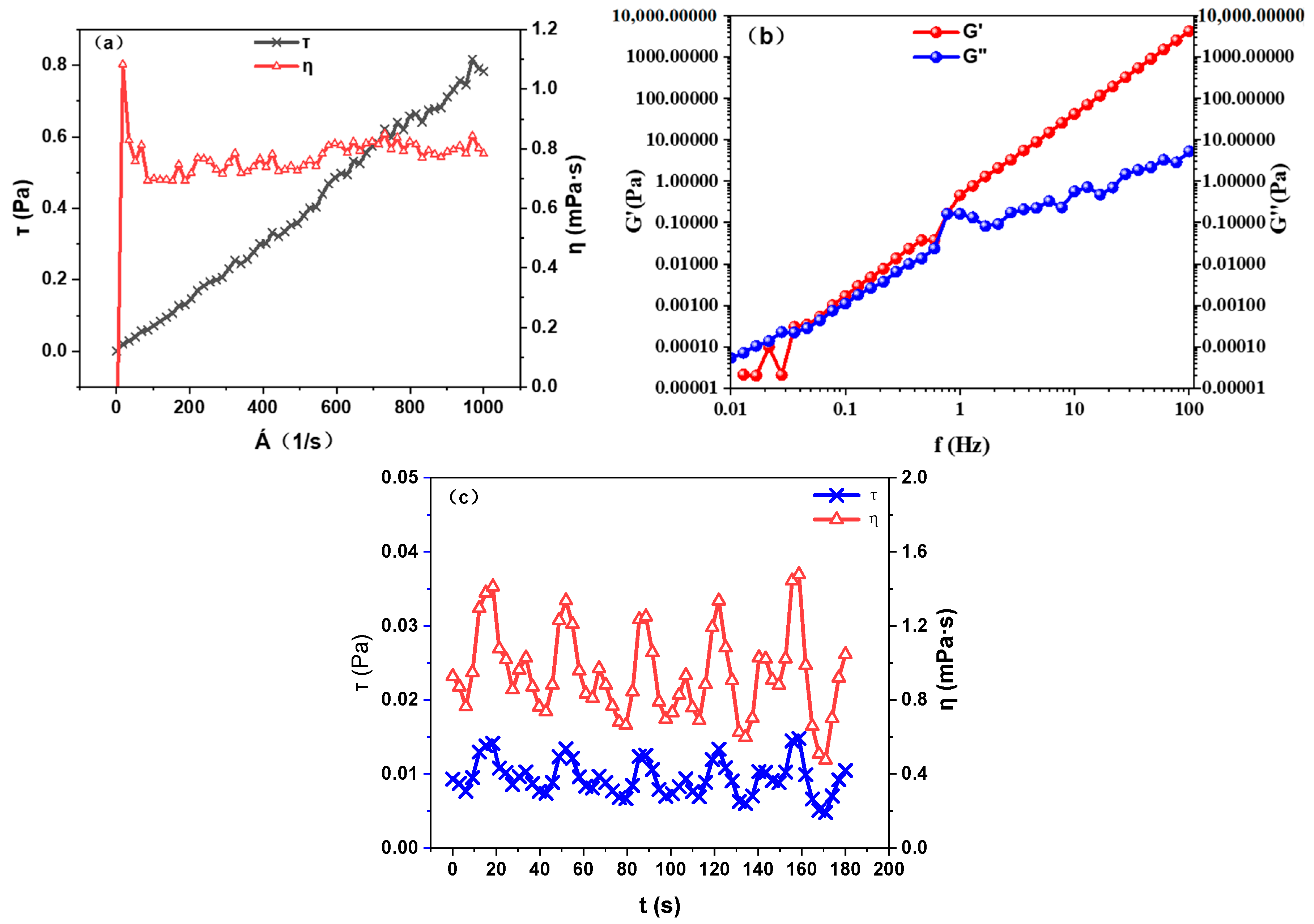
2.2.5. High-Temperature and High-Pressure Rheometer
2.3. Experimental Methods
2.3.1. Foam Performance Evaluation
2.3.2. pH Adjustment Experiments
2.3.3. Foaming Agent Concentration Experiments
2.3.4. Stabilizer Synergy Experiments
2.3.5. Effects of Temperature and Pressure
2.3.6. Liquid Film Thickness Measurement
2.3.7. Rheological and Viscoelastic Property Measurements
3. Experimental Results and Discussion
3.1. Comparative Analysis of Foam Performance Among Different Nonionic Surfactants
3.2. Influence of Chemical Factors
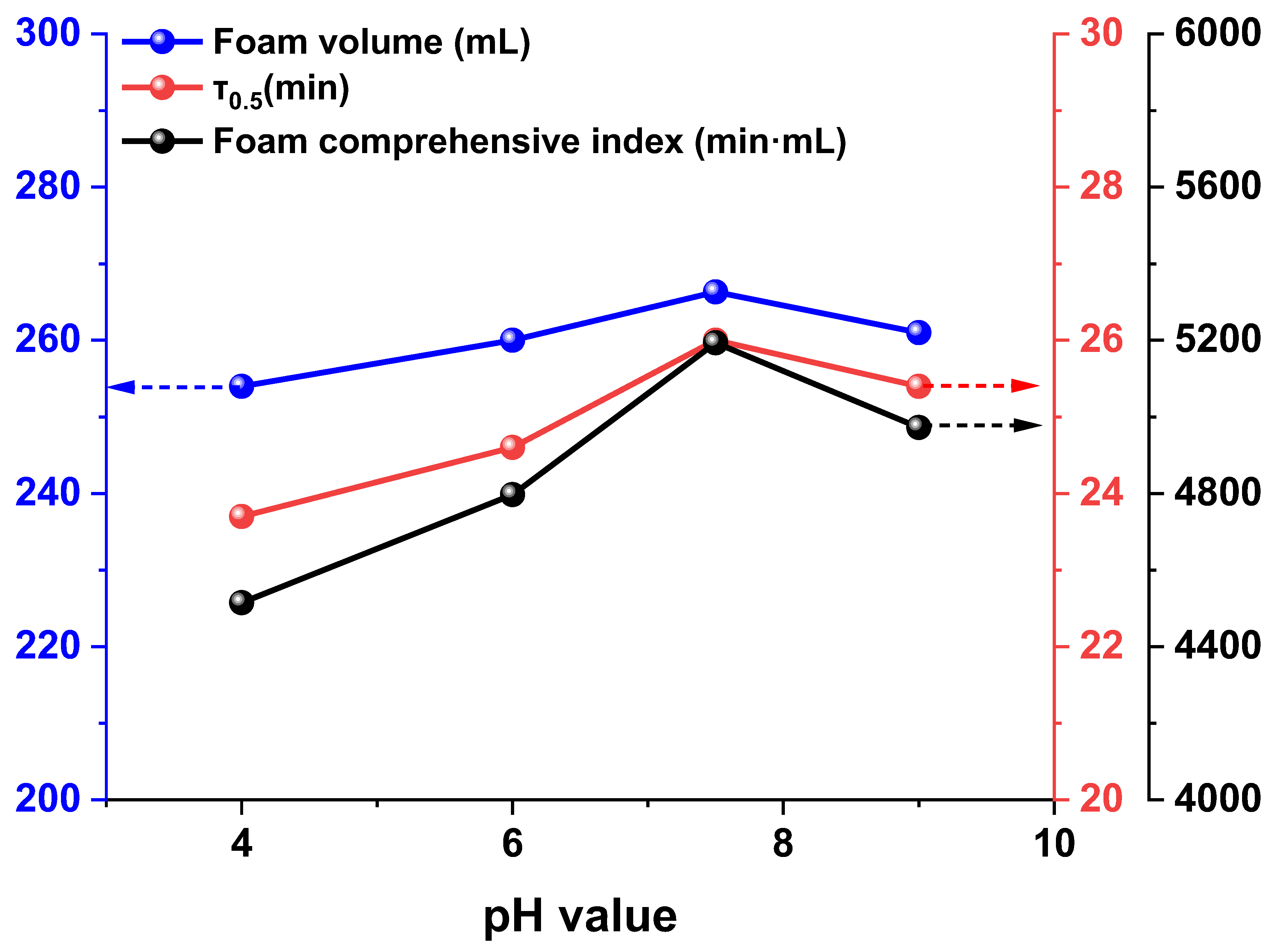
3.2.1. Effect of Solution pH
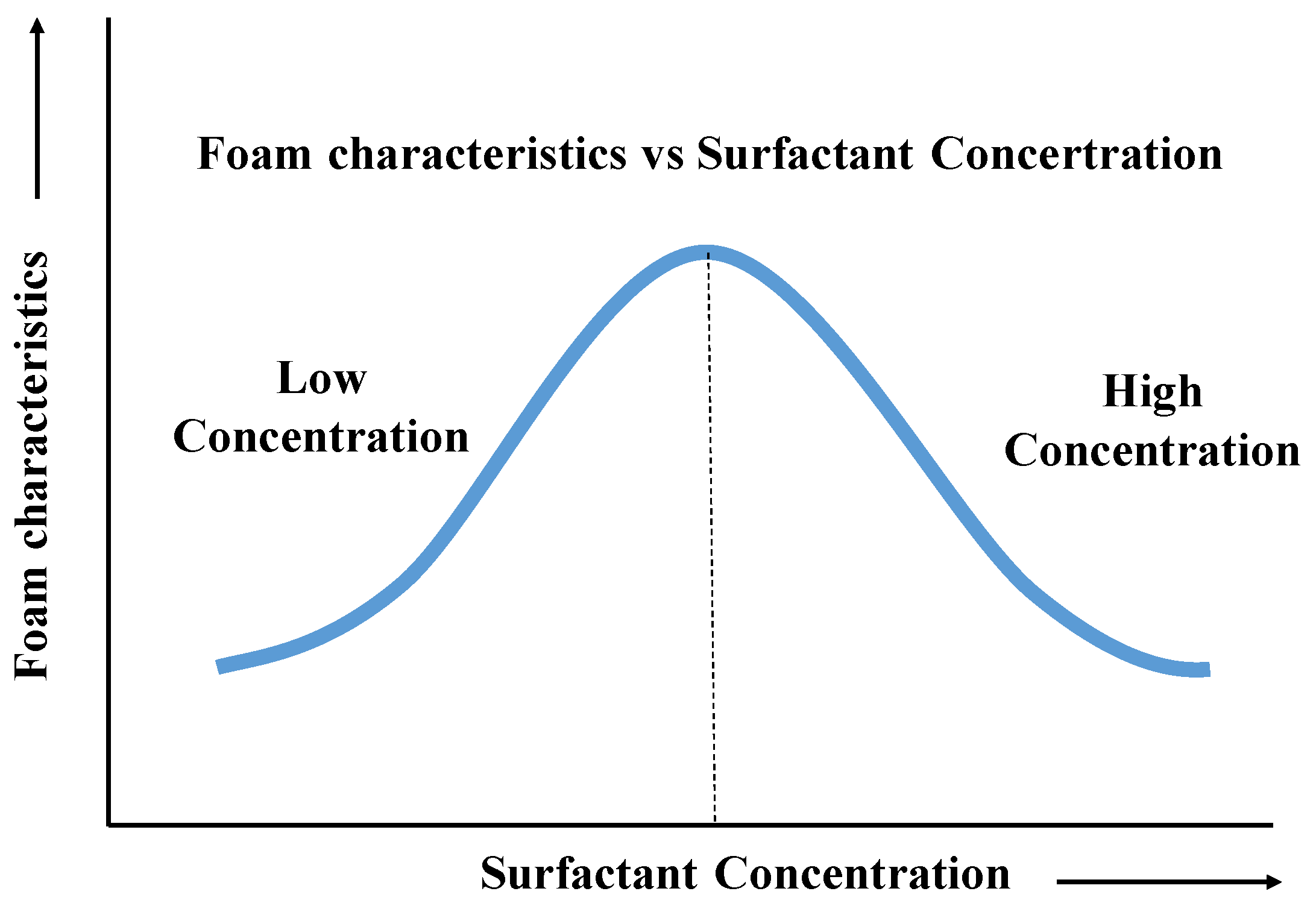
3.2.2. Effect of Foaming Agent Concentration
3.2.3. Role of Foam Stabilizers
3.3. Influence of Physical Factors
3.3.1. Effect of Temperature
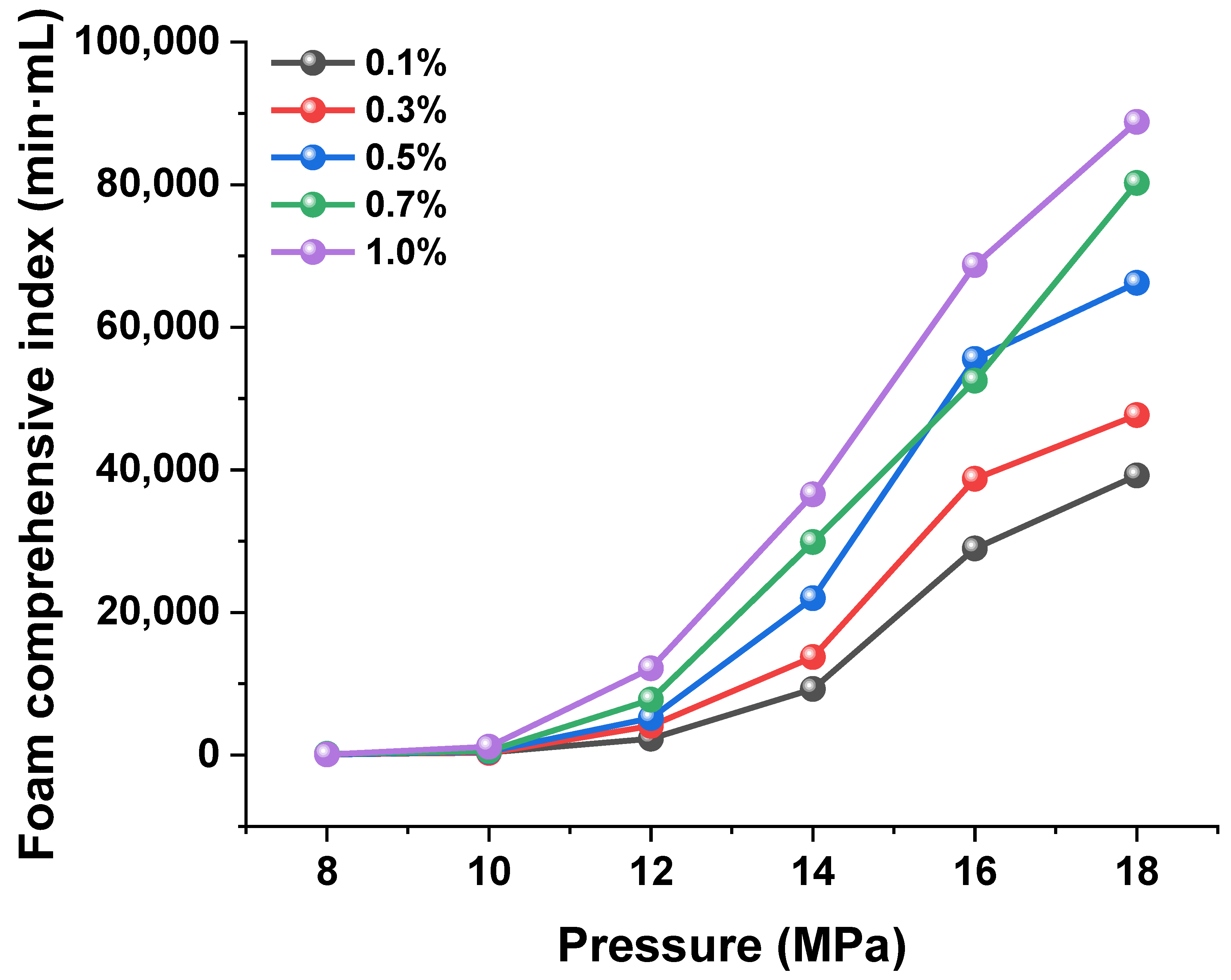
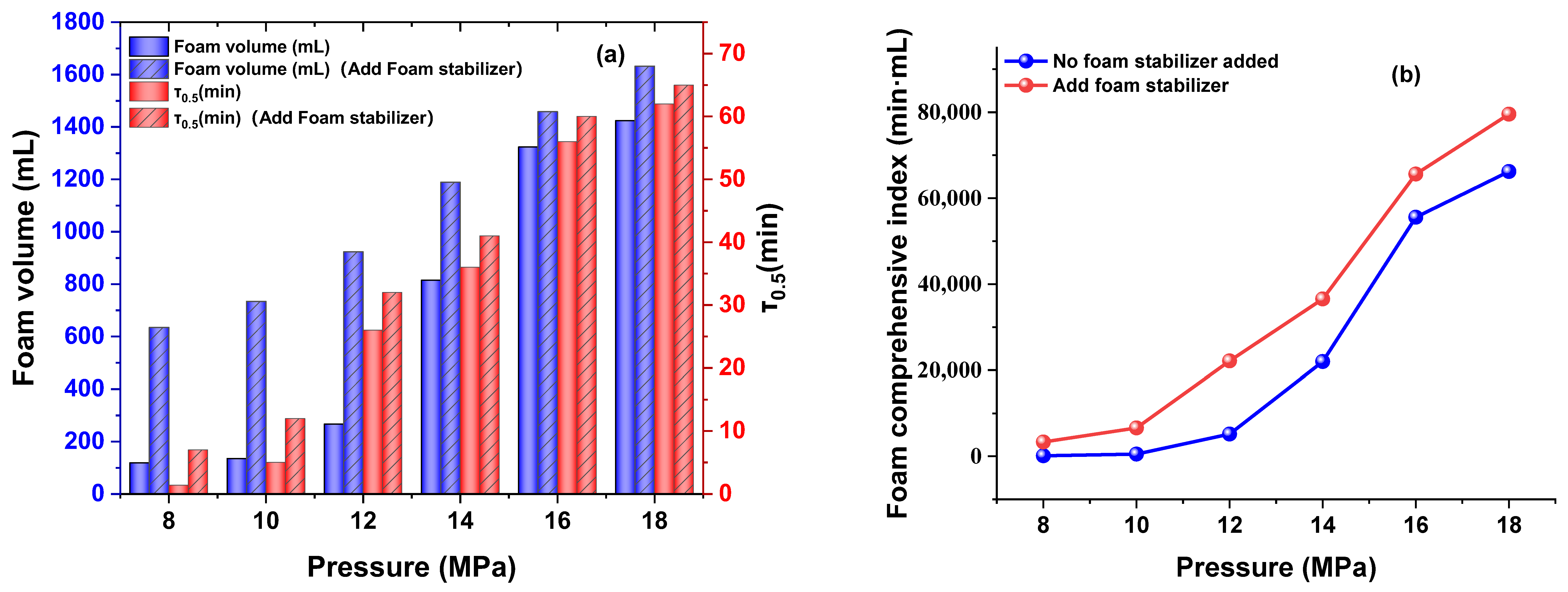
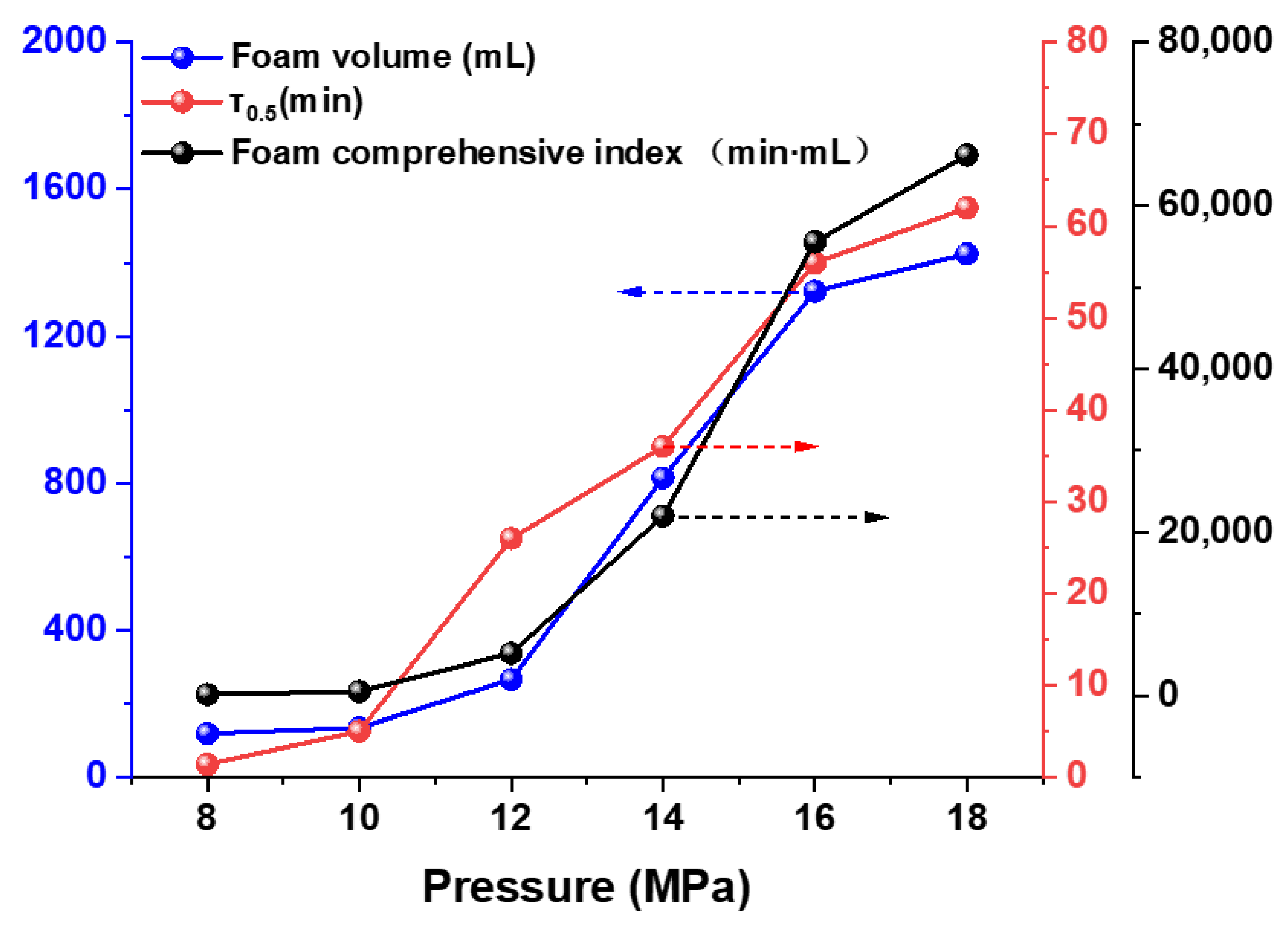
3.3.2. Effect of Pressure
3.4. Foam Stability and Interfacial Behavior
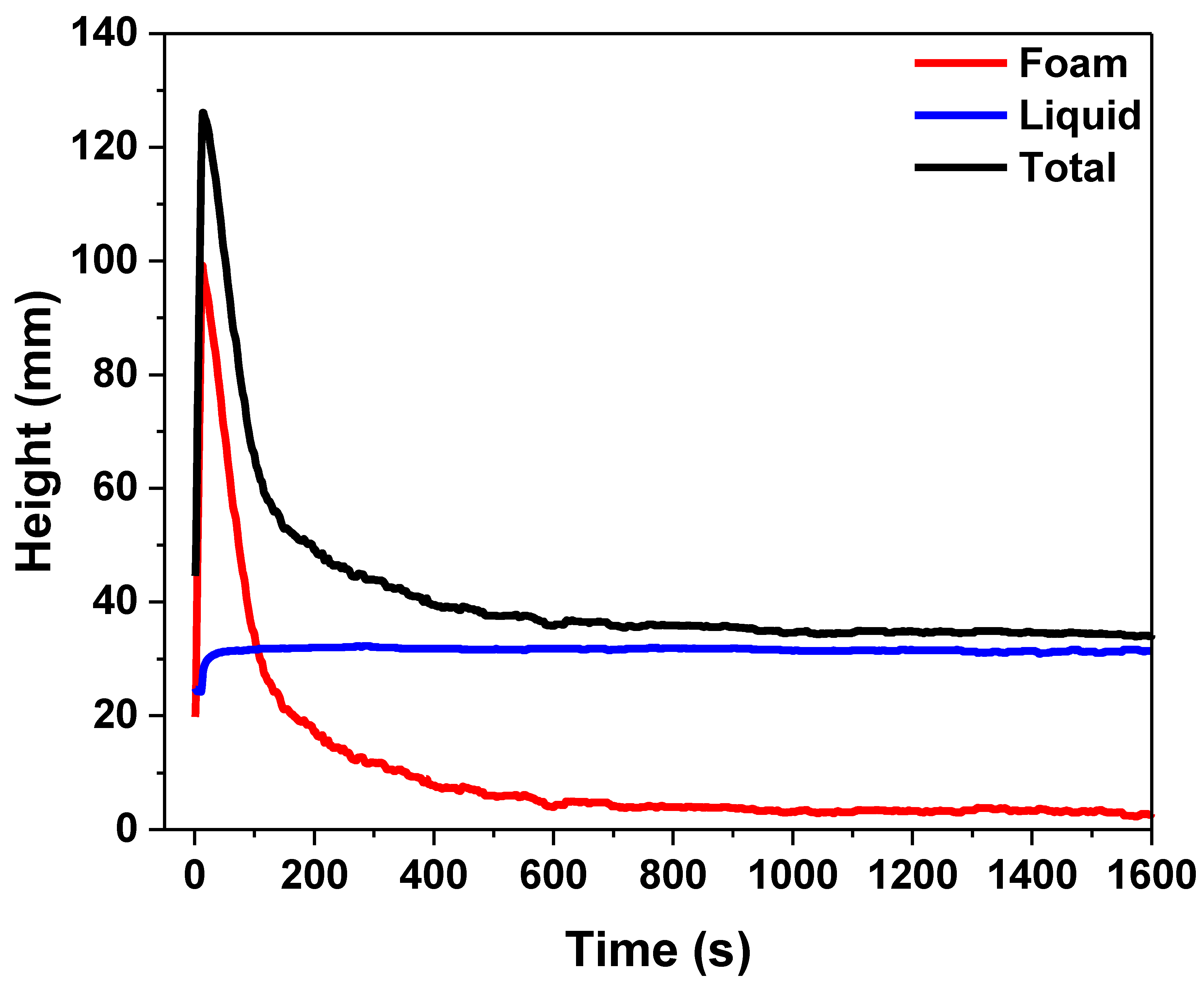
3.4.1. Liquid Film Thickness and Its Effect on Foam Longevity
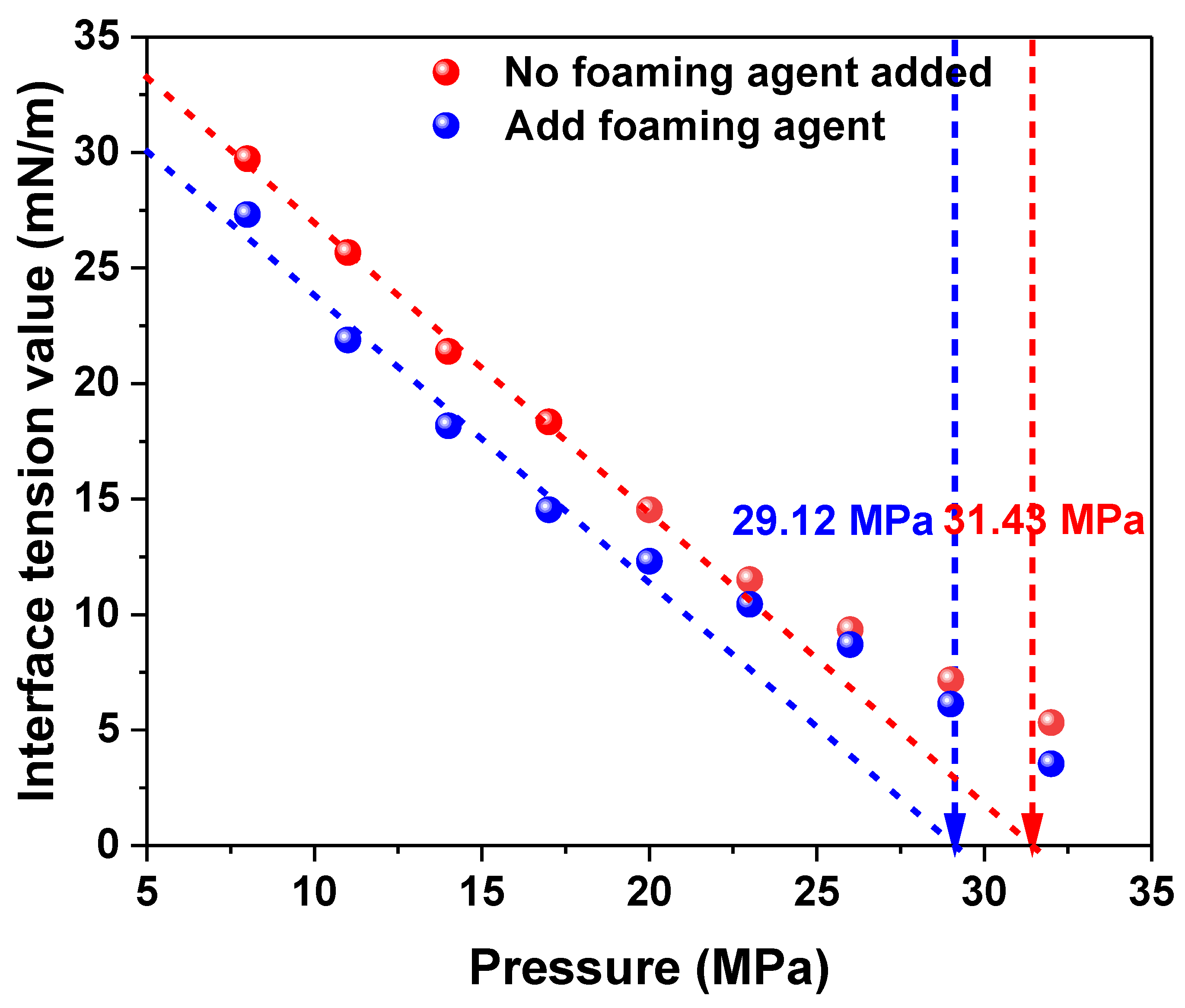
3.4.2. Liquid Drainage Rate and Interfacial Tension Regulation
4. Mechanistic Analysis of CO2-Soluble Foaming Agents
4.1. Molecular Structure and Gas–Liquid Interfacial Behavior
4.2. Concentration Control and Foam Generation Efficiency
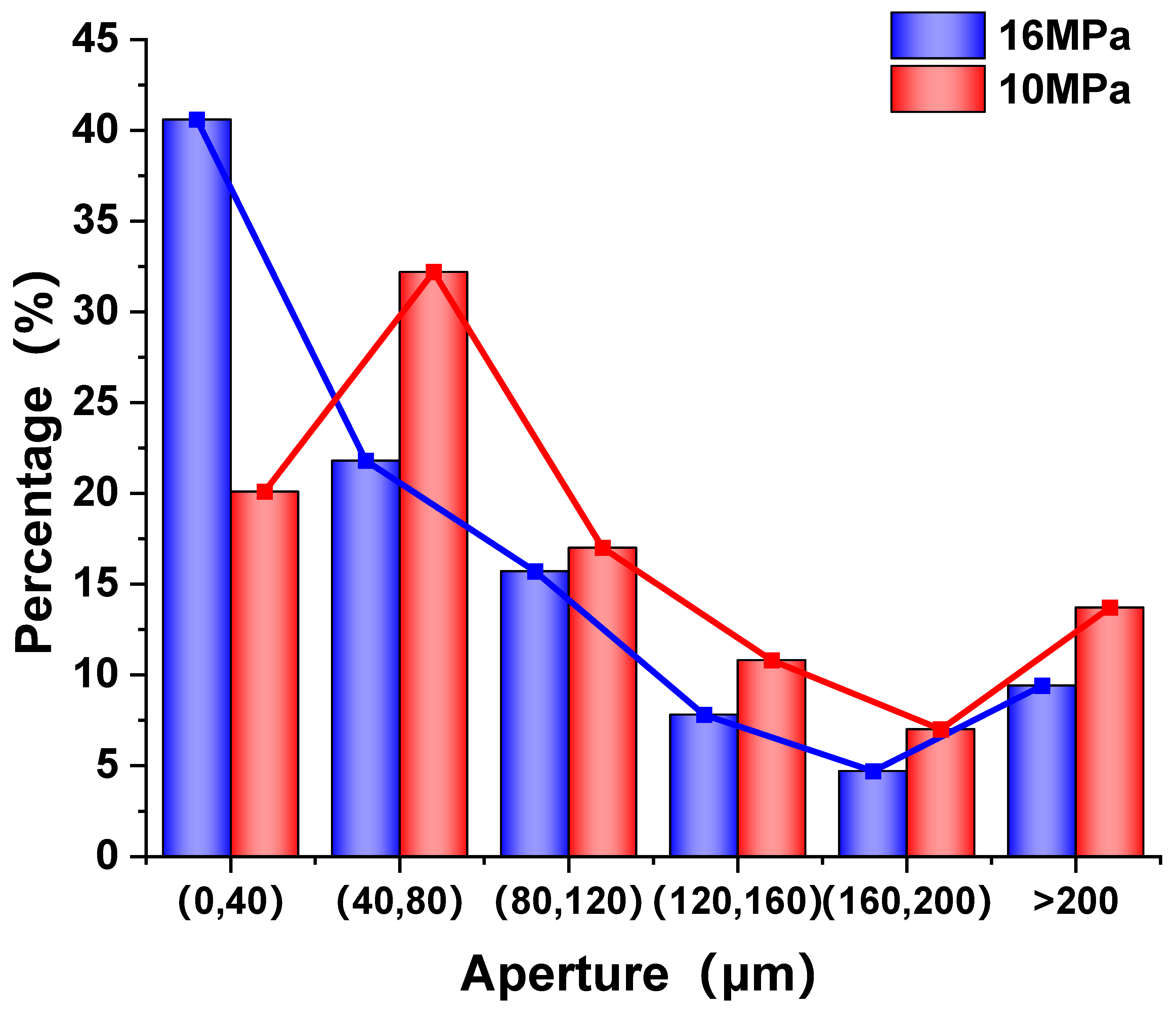
4.3. Foam Formation Mechanism in Porous Media
4.4. Critical Role of Film Stability and Interfacial Adsorption
4.5. Coupling Between Film Thickness and Foam Stability
4.6. Synergistic Role of Foam Stabilizers
5. Conclusions
- (1)
- The foam stability of CO2-soluble foaming agents is strongly dependent on the synergistic effects of chemical factors (such as pH, surfactant type, and concentration) and physical factors (such as temperature and pressure). A moderately alkaline environment, an optimal concentration around 0.5%, and high pressure are favorable for enhancing foam performance, while high temperatures accelerate film drainage and lead to rapid foam decay.
- (2)
- Incorporating nano-sized foam stabilizers such as silica (SiO2) enhances the thickness of the liquid film and improves the regulation of interfacial tension. These stabilizers form robust molecular barriers that effectively prevent foam collapse and bubble coalescence, with especially pronounced effects under high-pressure conditions.
- (3)
- Foam stability is governed by the adsorption capacity of the foaming agent at the gas–liquid interface, the rate of film drainage, and variations in film thickness. The experimental results show that film thickness decreases linearly over time, with an average drainage rate of 1.32 μm/s. Regulation of surface tension—achieved through pH and salinity adjustment—can effectively slow down the drainage process.
- (4)
- A mechanistic model for foam generation in porous media was developed, incorporating gas–liquid mixing, interfacial adsorption, bubble aggregation, and connectivity. This model reveals the directional migration and structural reorganization behavior of foaming agent molecules under CO2 flooding conditions.
- (5)
- For the large-scale application of CCUS-EOR in deep and low-permeability reservoirs, future research needs to systematically explain the synergistic stability mechanism of foam stabilizers in the real reservoir salinity temperature pressure coupled field and accordingly build a multi-functional composite foam system integrating scale prevention (chelating agent/pH buffer), corrosion inhibition (metal ion complex/inhibitor), and thermal shear bistability (thermal stable polymer/nanoparticle synergy). This system has been proven to have excellent stability under supercritical CO2 and high pressure and high temperature (>2500 m) conditions, and is demonstrating excellent flow control performance in the core displacement experiment of the national major project “CCUS Demonstration Zone in Songliao Basin”, laying a solid material foundation and data support for the industrial promotion of deep shale oil and low-permeability reservoirs.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gauglitz, P.A.; Friedmann, F.; Kam, S.I.; Rossen, W.R. Foam generation in porous media. In Proceedings of the SPE Improved Oil Recovery Conference, Tulsa, OK, USA, 13–17 April 2002; p. SPE-75177. [Google Scholar]
- Talebian, S.H.; Masoudi, R.; Tan, I.M.; Zitha, P.L.J. Foam assisted CO2-EOR: A review of concept, challenges, and future prospects. J. Pet. Sci. Eng. 2014, 120, 202–215. [Google Scholar] [CrossRef]
- Simjoo, M.; Rezaei, T.; Andrianov, A.; Zitha, P.L.J. Foam stability in the presence of oil: Effect of surfactant concentration and oil type. Colloids Surf. A Physicochem. Eng. Asp. 2013, 438, 148–158. [Google Scholar] [CrossRef]
- Der, O. Multi-Output Prediction and Optimization of CO2 Laser Cutting Quality in FFF-Printed ASA Thermoplastics Using Machine Learning Approaches. Polymers 2025, 17, 1910. [Google Scholar] [CrossRef] [PubMed]
- Ürgün, S.; Yiğit, H.; Fidan, S.; Sınmazçelik, T. Optimization of laser cutting parameters for PMMA using metaheuristic algorithms. Arab. J. Sci. Eng. 2024, 49, 12333–12355. [Google Scholar] [CrossRef]
- Kovscek, A.R.; Radke, C.J. Fundamentals of Foam Transport in Porous Media; No. DOE/BC-93000174; Lawrence Berkeley National Lab. (LBNL): Berkeley, CA, USA, 1993. [Google Scholar]
- Guo, F.; Aryana, S. An experimental investigation of nanoparticle-stabilized CO2 foam used in enhanced oil recovery. Fuel 2016, 186, 430–442. [Google Scholar] [CrossRef]
- Emrani, A.S.; Ibrahim, A.F.; Nasr-El-Din, H.A. Evaluation of mobility control with nanoparticle-stabilized CO2 foam. In Proceedings of the SPE Latin America and Caribbean Petroleum Engineering Conference, Buenos Aires, Argentina, 17–19 May 2017; p. SPE-185551. [Google Scholar]
- Singh, R.; Mohanty, K.K. Foam flow in a layered, heterogeneous porous medium: A visualization study. Fuel 2017, 197, 58–69. [Google Scholar] [CrossRef]
- Kedir, A.S.; Solbakken, J.S.; Aarra, M.G. Foamability and stability of anionic surfactant-anionic polymer solutions: Influence of ionic strength, polymer concentration, and molecular weight. Colloids Surf. A Physicochem. Eng. Asp. 2022, 632, 127801. [Google Scholar]
- Espinosa, D.; Caldelas, F.; Johnston, K.; Bryant, S.L.; Huh, C. Nanoparticle-stabilized supercritical CO2 foams for potential mobility control applications. In Proceedings of the SPE Improved Oil Recovery Conference, Tulsa, OK, USA, 24–28 April 2010; p. SPE-129925. [Google Scholar]
- Ahmed, S.; Elraies, K.A.; Hashmet, M.R.; Bt Mohd Shaifan, S.R.; Hsia, I.C.; Bahrim, R.Z. Experimental investigation and optimization of polymer enhanced CO2 foam stability and apparent viscosity. In Proceedings of the SPE Kingdom of Saudi Arabia Annual Technical Symposium and Exhibition, Dammam, Saudi Arabia, 24–27 April 2017; p. SPE 188018-MS. [Google Scholar]
- Farajzadeh, R.; Andrianov, A.; Krastev, R.; Hirasaki, G.J.; Rossen, W.R. Foam-oil interaction in porous media: Implications for foam assisted enhanced oil recovery. In Proceedings of the SPE EOR Conference at Oil and Gas West Asia, Muscat, Oman, 16–18 April 2012; p. SPE-154197. [Google Scholar]
- Jiang, N.; Yu, X.; Sheng, Y.; Zong, R.; Li, C.; Lu, S. Role of salts in performance of foam stabilized with sodium dodecyl sulfate. Chem. Eng. Sci. 2020, 216, 115474. [Google Scholar]
- Hirasaki, G.J.; Lawson, J.B. Mechanisms of foam flow in porous media: Apparent viscosity in smooth capillaries. Soc. Pet. Eng. J. 1985, 25, 176–190. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Y.; Liu, Y.; Zhang, L.; Ren, S.; Lu, J.; Wang, X.; Fan, N. The stability study of CO2 foams at high pressure and high temperature. J. Pet. Sci. Eng. 2017, 154, 234–243. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, Q.; Li, Y.; Liu, J.; Liu, Z.; Liu, S. Investigation on enhanced oil recovery and CO2 storage efficiency of temperature-resistant CO2 foam flooding. Fuel 2024, 364, 130870. [Google Scholar]
- Bertin, H.H.; Kovscek, A.R. Foam Mobility in Heterogeneous Porous Media. I: Scaling Concepts. Transp. Porous Media 2003, 52, 17–35. [Google Scholar]
- Farhadi, H.; Riahi, S.; Ayatollahi, S.; Ahmadi, H. Experimental study of nanoparticle-surfactant-stabilized CO2 foam: Stability and mobility control. Chem. Eng. Res. Des. 2016, 111, 449–460. [Google Scholar]
- Alvarenga, B.; Lopes, L.; Ribeiro, F.; Pérez-Gramatges, A. Challenges in screening nanoparticles for CO2-foam stabilization: From atmospheric to high-pressure conditions. Colloids Surf. A Physicochem. Eng. Asp. 2025, 720, 137165. [Google Scholar]
- Aroonsri, A. Nanoparticle-Stabilized Supercritical CO2 Foam for Mobility Control in CO2 Enhanced Oil Recovery. Ph.D. Dissertation, University of Texas at Austin, Austin, TX, USA, 2014. [Google Scholar]
- Koyanbayev, M.; Hazlett, R.D.; Wang, L.; Hashmet, M.R. An experimental investigation of surfactant-stabilized CO2 foam flooding in carbonate cores in reservoir conditions. Energies 2024, 17, 3353. [Google Scholar]
- Heller, J.P.; Boone, D.A.; Watts, R.J. Field test of CO2 mobility control at Rock Creek. In Proceedings of the SPE Annual Technical Conference and Exhibition, Las Vegas, NV, USA, 22–26 September 1985; p. SPE-14395. [Google Scholar]
- Yu, J.; An, C.; Mo, D.; Liu, N.; Lee, R. Foam mobility control for nanoparticle-stabilized CO2 foam. In Proceedings of the SPE Improved Oil Recovery Conference, Tulsa, OK, USA, 14–18 April 2012; p. SPE-153336. [Google Scholar]







| Number | Number of EO Units | Chemical Name | Skeleton Symbol |
|---|---|---|---|
| E-1303 | 3 | Polyoxyethylene (3) isotridecyl ether | i-C13H27O(CH2CH2O)3H |
| E-1305 | 5 | Polyoxyethylene (5) isotridecyl ether | i-C13H27O(CH2CH2O)5H |
| E-1306 | 6 | Polyoxyethylene (6) isotridecyl ether | i-C13H27O(CH2CH2O)6H |
| E-1308 | 8 | Polyoxyethylene (8) isotridecyl ether | i-C13H27O(CH2CH2O)8H |
| E-1309 | 9 | Polyoxyethylene (9) isotridecyl ether | i-C13H27O(CH2CH2O)9H |
| E-1310 | 10 | Polyoxyethylene (10) isotridecyl ether | i-C13H27O(CH2CH2O)10H |
| E-1312 | 12 | Polyoxyethylene (12) isotridecyl ether | i-C13H27O(CH2CH2O)12H |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, W.; Yang, W.; Yang, Z.; Cao, S.; Xu, Q.; Zhao, F.; Guo, T.; Sun, T. Stability and Foam Performance Optimization of CO2-Soluble Foaming Agents: Influencing Factors and Mechanistic Analysis. Processes 2025, 13, 2784. https://doi.org/10.3390/pr13092784
Sun W, Yang W, Yang Z, Cao S, Xu Q, Zhao F, Guo T, Sun T. Stability and Foam Performance Optimization of CO2-Soluble Foaming Agents: Influencing Factors and Mechanistic Analysis. Processes. 2025; 13(9):2784. https://doi.org/10.3390/pr13092784
Chicago/Turabian StyleSun, Wenjing, Wenlu Yang, Zian Yang, Sheng Cao, Quan Xu, Fajun Zhao, Tianjiao Guo, and Tianyi Sun. 2025. "Stability and Foam Performance Optimization of CO2-Soluble Foaming Agents: Influencing Factors and Mechanistic Analysis" Processes 13, no. 9: 2784. https://doi.org/10.3390/pr13092784
APA StyleSun, W., Yang, W., Yang, Z., Cao, S., Xu, Q., Zhao, F., Guo, T., & Sun, T. (2025). Stability and Foam Performance Optimization of CO2-Soluble Foaming Agents: Influencing Factors and Mechanistic Analysis. Processes, 13(9), 2784. https://doi.org/10.3390/pr13092784






