Optimizing Methane Production from Lignocellulosic Biomass: Low-Temperature Potassium Ferrate Pretreatment via Response Surface Methodology
Abstract
1. Introduction
2. Materials and Methods
2.1. Substrate and Inoculum
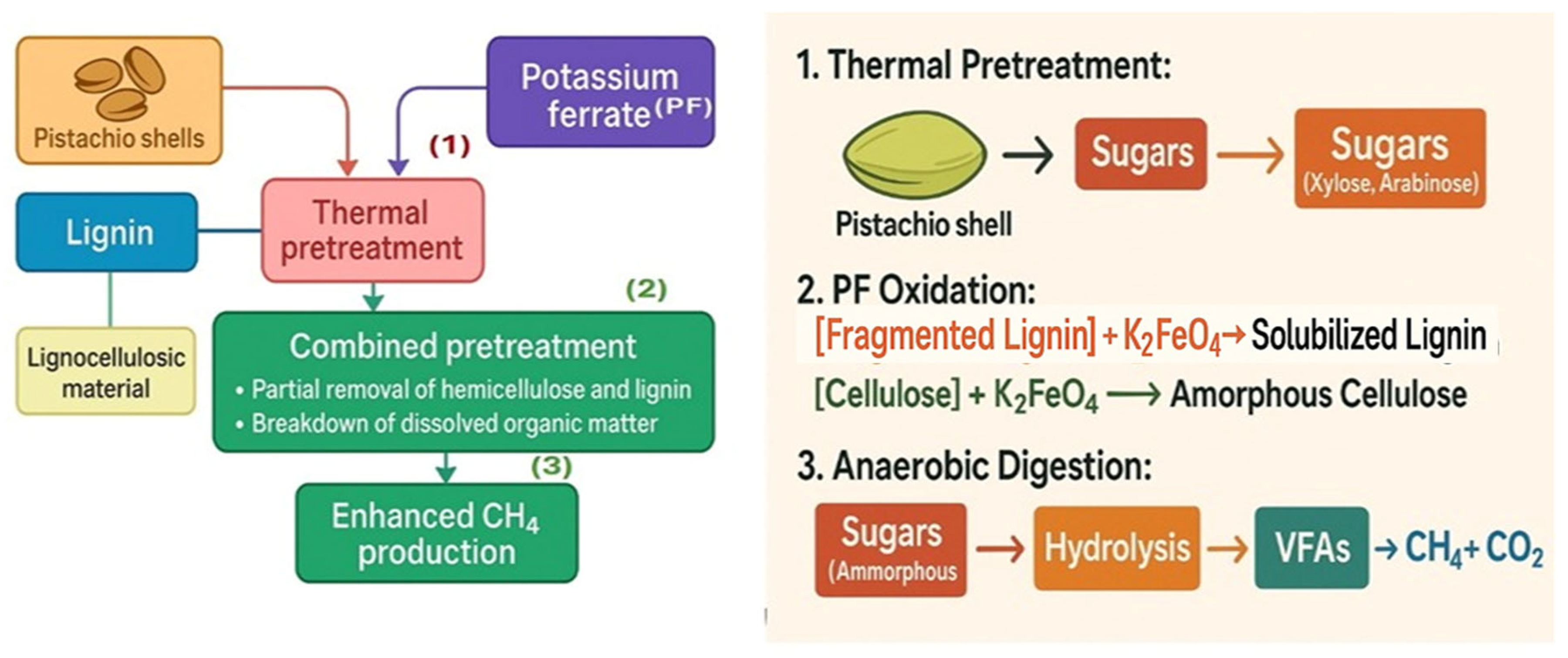
2.2. PF-Based and Thermal Pretreatment Procedure
2.3. Experimental Design and RSM-Based Optimization
2.4. Biochemical Methane (CH4) Potential (BMP) Assay
2.5. Analytical Methods
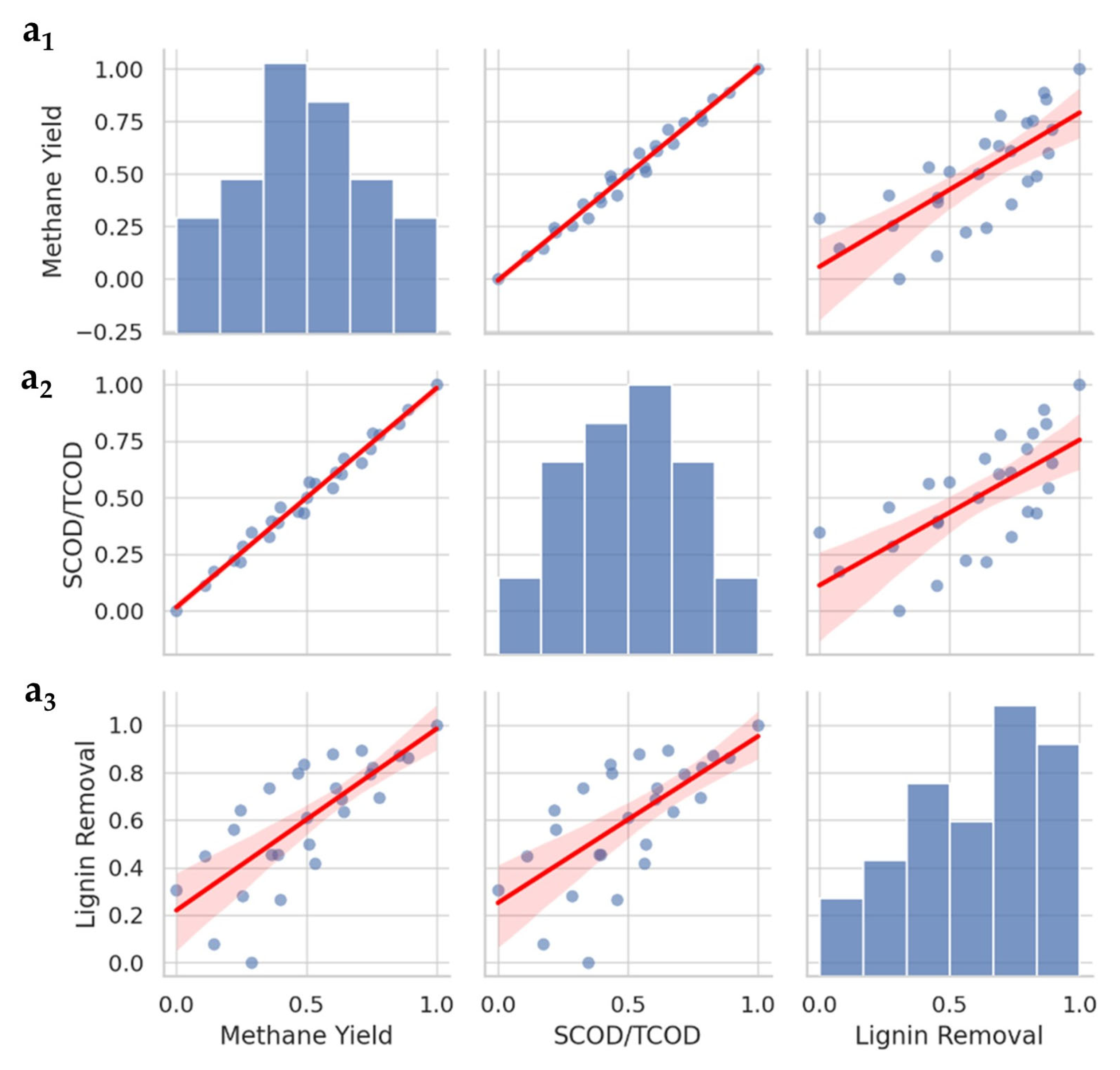
3. Results and Discussion
3.1. Physicochemical Characterization of Pistachio Shells and Inoculum
3.2. ANOVA and Model Fitting
3.3. Response Surface Plot
3.4. Model Equations
3.5. Optimization of Pretreatment Conditions
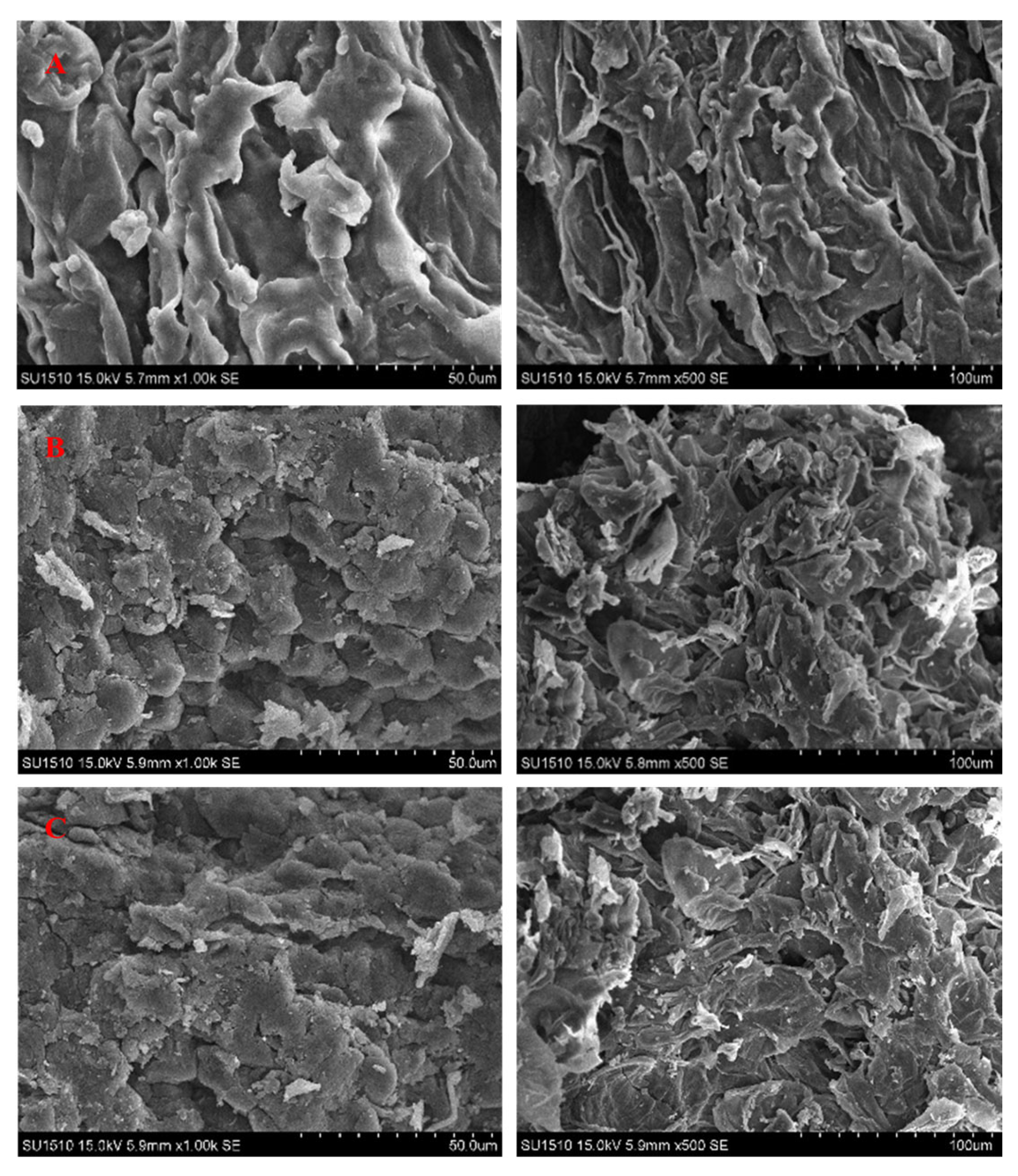
3.6. Process Mechanisms, Structural Changes, and Implications
4. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| Anaerobic digestion | AD |
| Pistachio shells | PSs |
| Potassium ferrate | PF |
| Methane | CH4 |
| Advanced oxidation processes | AOPs |
| Hydroxyl | •OH |
| Sulfate | SO4•− |
| Ferrate ions | FeO42− |
| Analysis of variance | ANOVA |
| Soluble COD | SCOD |
| Total COD | TCOD |
| Response surface methodology | RSM |
| Box–Behnken Design | BBD |
| Total solids | TSs |
| Volatile solids | VSs |
References
- Mussatto, S.I.; Dragone, G.; Guimarães, P.M.; Silva, J.P.A.; Carneiro, L.M.; Roberto, I.C.; Antonio, V.; Domingues, L.; Teixeira, A.J. Technological trends, global market, and challenges of bio-ethanol production. Biotechnol. Adv. 2010, 28, 817–830. [Google Scholar] [CrossRef]
- Xu, F.; Li, Y.; Ge, X.; Yang, L.; Li, Y. Anaerobic digestion of food waste–challenges and opportunities. Bioresour. Technol. 2018, 247, 1047–1058. [Google Scholar] [CrossRef]
- Romero-Ceron, C.; Flores-Silva, P.; Saucedo-Salazar, E.; Gallardo-Vega, C.; Hernandez-Gamez, F.; Hernandez-Hernandez, E.; Sifuentes-Nieves, I. Modified pistachio shell waste: Structure, chemical interaction, and viscoelastic performance in biopolymer composites-based starch. Polym. Compos. 2025, 46, 8025–8036. [Google Scholar] [CrossRef]
- Čolnik, M.; Irgolič, M.; Perva, A.; Skerget, M. The conversion of pistachio and walnut shell waste into valuable components with subcritical water. Processes 2024, 12, 195. [Google Scholar] [CrossRef]
- Kazemi, M. Recycling agricultural waste: Sustainable solutions for enhancing livestock nutrition. Vet. Med. Sci. 2025, 11, 71321. [Google Scholar] [CrossRef] [PubMed]
- Putro, J.N.; Soetaredjo, F.E.; Lin, S.; Ju, Y.; Ismadji, S. Pretreatment and conversion of lignocellulose biomass into valuable chemicals. RSC Adv. 2016, 6, 46834–46852. [Google Scholar] [CrossRef]
- Alsheheri, S.Z.; Salama, R.S. Sustainable nanocomposite fabrication from pistachio shells and aluminum cans: Alumina-activated carbon with silver ferrite nanoparticles for high-performance supercapacitors. J. Inorg. Organomet. Polym. Mater. 2025, 35, 5751–5766. [Google Scholar] [CrossRef]
- Di Fraia, A.; Di Fraia, S.; Massarotti, N. Role of advanced oxidation processes in lignocellulose pretreatment towards biorefinery applications: A review on emerging trends and economic considerations. Green Chem. 2024, 15, 8461–8496. [Google Scholar] [CrossRef]
- Wang, J.; Feng, K.; Lou, Y.; Lu, B.; Liu, B.; Xie, G.; Ren, N.; Xing, D. The synergistic effect of potassium ferrate and peroxymonosulfate application on biogas production and shaping microbial community during anaerobic co-digestion of a cow manure-cotton straw mixture. Bioresour. Technol. 2021, 333, 125166. [Google Scholar] [CrossRef]
- Yu, J.; Zhang, K.; Zhu, Q.; Wu, C.; Huang, S.; Zhang, Y.; Yao, S.; Pang, W. A review of research progress in the preparation and application of ferrate (VI). Water 2023, 15, 699. [Google Scholar] [CrossRef]
- Planý, M.; Czolderová, M.; Kraková, L.; Puškárová, A.; Bučková, M.; Šoltys, K.; Budiš, J.; Szemes, T.; Mackulak, T.; Wu, J.; et al. Biogas production: Evaluation of the influence of K2FeO4 pretreatment of maple leaves (Acer platanoides) on microbial consortia composition. Bioprocess Biosyst. Eng. 2019, 42, 1151–1163. [Google Scholar] [CrossRef] [PubMed]
- Ellis, J.L.; Kebreab, E.; Odongo, N.E.; McBride, B.W.; Okine, E.K.; France, J. Prediction of methane production from dairy and beef cattle. J. Dairy Sci. 2007, 90, 3456–3466. [Google Scholar] [CrossRef] [PubMed]
- Şenol, H.; Çolak, E.; Elibol, E.A.; Hassaan, M.A.; El Nemr, A. Optimisation of biochar dose in anaerobic co-digestion of green algae and cattle manure using artificial neural networks and response surface methodology. Chem. Eng. J. 2024, 493, 152750. [Google Scholar] [CrossRef]
- Bianco, F.; Şenol, H.; Papirio, S.; Zenk, H.; Kara, A.; Atasoy, S. Combined ultrasonic–hydrothermal pretreatment to improve the biomethane potential of hazelnut shell. Biomass Bioenergy 2022, 165, 106554. [Google Scholar] [CrossRef]
- Guo, B.; Hu, J.; Zhang, J.; Wu, Z.; Li, Z. Enhanced methane production from waste activated sludge by potassium ferrate combined with ultrasound pretreatment. Bioresour. Technol. 2021, 341, 125841. [Google Scholar] [CrossRef]
- Li, W.; Yu, N.; Liu, Q.; Li, Y.; Ren, N.; Xing, D. Enhancement of the sludge disintegration and nutrients release by a treatment with potassium ferrate combined with an ultrasonic process. Sci. Total Environ. 2018, 635, 699–704. [Google Scholar] [CrossRef]
- Şenol, H. Improvement of methane yield from hazelnut shells by potassium ferrate and mild ultrasonic pretreatment: Kinetics study. Fuel 2026, 403, 135989. [Google Scholar] [CrossRef]
- Aridi, R.; Yehya, A. Anaerobic biodigesters heating sources: Analysis and recommendations. Renew. Sustain. Energy Rev. 2024, 202, 114700. [Google Scholar] [CrossRef]
- Wang, X.; Wang, Y.; Tian, L.; Zheng, K.; Zhu, T.; Chen, X.; Zhao, Y.; Liu, Y. Heat-assisted potassium ferrate pretreatment enhancing short-chain fatty acids production from waste activated sludge: Performance and mechanisms. J. Clean. Prod. 2022, 380, 134989. [Google Scholar] [CrossRef]
- Córdoba, V.; Bavio, M.; Acosta, G. Biomethane production modelling from third-generation biomass. Renew. Energy 2024, 234, 121211. [Google Scholar] [CrossRef]
- Moset, V.; Al-zohairi, N.; Møller, H.B. The impact of inoculum source, inoculum to substrate ratio and sample preservation on methane potential from different substrates. Biomass Bioenergy 2015, 83, 474–482. [Google Scholar] [CrossRef]
- Bianco, F.; Şenol, H.; Papirio, S. Enhanced lignocellulosic component removal and biomethane potential from chestnut shell by a combined hydrothermal–alkaline pretreatment. Sci. Total Environ. 2021, 762, 144178. [Google Scholar] [CrossRef] [PubMed]
- A.P.H.A. Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 1926. [Google Scholar]
- Van Soest, P.v.; Robertson, J.B.; Lewis, B.A. Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J. Dairy Sci. 1991, 74, 3583–3597. [Google Scholar] [CrossRef] [PubMed]
- Rintala, K.; Kinnunen, V.; Berg, A.; Rintala, J.; Kokko, M. Biogas production from piloted wood-based bioethanol process using high-rate anaerobic treatment–focusing for inhibition and improving biodegradability. Renew. Energy. 2024, 237, 121594. [Google Scholar] [CrossRef]
- Cai, J.; He, Y.; Yu, X.; Banks, S.W.; Yang, Y.; Zhang, X.; Yu, Y.; Liu, R.; Bridgwater, A.V. Review of physicochemical properties and analytical characterization of lignocellulosic biomass. Renew. Sustain. Energy Rev. 2017, 76, 309–322. [Google Scholar] [CrossRef]
- Robles, E.; Izaguirre, N.; Martin, A.; Moschou, D.; Labidi, J. Assessment of bleached and unbleached nanofibers from pistachio shells for nanopaper making. Molecules 2021, 26, 1371. [Google Scholar] [CrossRef]
- Olatunji, K.O.; Madyira, D.M. comparative analysis of the effects of five pretreatment methods on morphological and methane yield of groundnut shells. Waste Biomass Valorization 2024, 15, 469–486. [Google Scholar] [CrossRef]
- Rahmani, A.M.; Tyagi, V.K.; Kazmi, A.A.; Ojha, C.S.P. Hydrothermal and thermal-acid pretreatments of wheat straw: Methane yield, recalcitrant formation, process inhibition, kinetic modeling. Energy 2023, 283, 129083. [Google Scholar] [CrossRef]
- Hendriks, A.; Zeeman, G. Pretreatments to enhance the digestibility of lignocellulosic biomass. Bioresour. Technol. 2009, 100, 10–18. [Google Scholar] [CrossRef]
- Yang, W.J.; He, Z.W.; Ren, Y.X.; Jin, H.Y.; Wang, R.; Tang, C.C.; Zou, A.; Liu, W.; Wang, A. Low temperature assisting potassium ferrate treatment to enhance short-chain fatty acids production from waste activated sludge. J. Environ. Chem. Eng. 2022, 10, 107821. [Google Scholar] [CrossRef]
- Pazara, M.; Provelengiadi, G.; Mantiniotou, M.; Athanasiadis, V.; Samanidis, I.; Makrygiannis, I.; Tzavellas, I.F.; Martakos, I.C.; Thomaidis, N.S.; Lalas, S.I. optimizing bioactive compound recovery from chestnut shells using pressurized liquid extraction and the box–behnken design. Processes 2025, 13, 2283. [Google Scholar] [CrossRef]
- Wang, J.; Cui, H.; Xie, G.; Liu, B.; Cao, G.; Xing, D. Co-treatment of potassium ferrate and peroxymonosulfate enhances the decomposition of the cotton straw and cow manure mixture. Sci. Total Environ. 2020, 724, 138321. [Google Scholar] [CrossRef]
- Şenol, H. Alkaline-thermal and mild ultrasonic pretreatments for improving biomethane yields: Impact on structural properties of chestnut shells. Fuel 2023, 354, 129373. [Google Scholar] [CrossRef]
- Li, W.; Yu, N.; Fang, A.; Liu, B.; Ren, N.; Xing, D. Co-treatment of potassium ferrate and ultrasonication enhances degradability and dewaterability of waste activated sludge. Chem. Eng. J. 2019, 361, 148–155. [Google Scholar] [CrossRef]

| Parameters | Pistachio Shell | Inoculum |
|---|---|---|
| %TSs a | 92.91 ± 1.21 | 15.44 ± 2.10 |
| %VSs a | 86.92 ± 1.97 | 5.45 ± 1.209 |
| TCOD (mg O2/L) | 69,244 ± 545 | 8111 ± 335 |
| SCOD (mg O2/L) | n.a. | 1798 ± 147 |
| Lignin b | 34.32 ± 2.19 | n.a. |
| Cellulose b | 23.11 ± 1.79 | n.a. |
| Hemicellulose b | 21.55 ± 1.57 | n.a. |
| pH | n.a. | 6.58 ± 0.21 |
| %C b | 49.12 ± 1.8 | 4.52 ± 0.33 |
| %H b | 5.48 ± 0.19 | n.a. |
| %O | 45.17 | n.a. |
| %N b | 0.13 ± 0.03 | 0.40 ± 0.04 |
| %S b | 0.1 ± 0.02 | n.a. |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value | Adjusted R2 | Prediction R2 | R2 | MSE | |
|---|---|---|---|---|---|---|---|---|---|---|
| CH4 yields (model) | 23,272.03 | 9 | 2585.7 | 10.30 | 0.0028 | Significant | 0.8395 | −0.1238 | 0.9297 | 103.44 |
| A—PF | 4919.33 | 1 | 4919.3 | 19.59 | 0.0031 | |||||
| B—pretreatment temperate | 2457.01 | 1 | 2457.0 | 9.78 | 0.0167 | |||||
| C—pretreatment time | 7174.82 | 1 | 7174.8 | 28.57 | 0.0011 | |||||
| AB | 136.89 | 1 | 136.9 | 0.55 | 0.4844 | |||||
| AC | 403.61 | 1 | 403.6 | 1.61 | 0.2454 | |||||
| BC | 2430.49 | 1 | 2430.5 | 9.68 | 0.0171 | |||||
| A2 | 1777.68 | 1 | 1777.7 | 7.08 | 0.0324 | |||||
| B2 | 566.81 | 1 | 566.8 | 2.26 | 0.1767 | |||||
| C2 | 2867.7 | 1 | 2867.7 | 11.42 | 0.0118 | |||||
| Residual | 1758.02 | 7 | 251.5 | |||||||
| Lack of fit | 1758.02 | 3 | 586.0 | |||||||
| Pure error | 0 | 4 | ||||||||
| Cor. total | 25,030.05 | 16 | ||||||||
| SCOD/TCOD (model) | 0.0234 | 9 | 0.0020 | 12.59 | 0.0015 | Significant | 0.8670 | 0.1882 | 0.9418 | 8.49 × 10−5 |
| A—PF | 0.0045 | 1 | 0.0040 | 21.89 | 0.0023 | |||||
| B—pretreatment temperate | 0.0028 | 1 | 0.0030 | 13.65 | 0.0077 | |||||
| C—pretreatment time | 0.0091 | 1 | 0.0090 | 44.21 | 0.0003 | |||||
| AB | 0.0004 | 1 | 0.0004 | 1.84 | 0.2165 | |||||
| AC | 0.0009 | 1 | 0.0009 | 4.51 | 0.0713 | |||||
| BC | 0.0026 | 1 | 0.0026 | 12.37 | 0.0098 | |||||
| A2 | 0.0019 | 1 | 0.0019 | 9.42 | 0.0181 | |||||
| B2 | 0.0001 | 1 | 0.0001 | 0.3309 | 0.5831 | |||||
| C2 | 0.0009 | 1 | 0.0009 | 4.58 | 0.0696 | |||||
| Residual | 0.0014 | 7 | 0.0002 | |||||||
| Lack of fit | 0.0012 | 3 | 0.0004 | 8.06 | 0.0359 | |||||
| Pure error | 0.0002 | 4 | 0.0001 | |||||||
| Cor. total | 0.0248 | 16 | ||||||||
| Lignin removal (model) | 283.49 | 7 | 40.5 | 13.63 | 0.0004 | Significant | 0.8467 | 0.4231 | 0.9138 | 1.57 |
| A—PF | 55.12 | 1 | 55.12 | 18.55 | 0.002 | |||||
| B—pretreatment temperate | 9.03 | 1 | 9.03 | 3.04 | 0.1153 | |||||
| C—pretreatment time | 117.81 | 1 | 117.81 | 39.64 | 0.0001 | |||||
| AC | 23.04 | 1 | 23.04 | 7.75 | 0.0213 | |||||
| BC | 19.8 | 1 | 19.8 | 6.66 | 0.0296 | |||||
| A2 | 26.36 | 1 | 26.36 | 8.87 | 0.0155 | |||||
| C2 | 29.06 | 1 | 29.06 | 9.78 | 0.0122 | |||||
| Residual | 26.75 | 9 | 2.97 | |||||||
| Lack of fit | 23.22 | 5 | 4.64 | 5.27 | 0.0663 | |||||
| Pure error | 3.53 | 4 | 0.882 | |||||||
| Cor. total | 310.24 | 16 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Şenol, H.; Çolak, E. Optimizing Methane Production from Lignocellulosic Biomass: Low-Temperature Potassium Ferrate Pretreatment via Response Surface Methodology. Processes 2025, 13, 2768. https://doi.org/10.3390/pr13092768
Şenol H, Çolak E. Optimizing Methane Production from Lignocellulosic Biomass: Low-Temperature Potassium Ferrate Pretreatment via Response Surface Methodology. Processes. 2025; 13(9):2768. https://doi.org/10.3390/pr13092768
Chicago/Turabian StyleŞenol, Halil, and Emre Çolak. 2025. "Optimizing Methane Production from Lignocellulosic Biomass: Low-Temperature Potassium Ferrate Pretreatment via Response Surface Methodology" Processes 13, no. 9: 2768. https://doi.org/10.3390/pr13092768
APA StyleŞenol, H., & Çolak, E. (2025). Optimizing Methane Production from Lignocellulosic Biomass: Low-Temperature Potassium Ferrate Pretreatment via Response Surface Methodology. Processes, 13(9), 2768. https://doi.org/10.3390/pr13092768






