Abstract
Shipyards are significant industrial sources of environmental pollution, releasing substantial amounts of heavy metals, petroleum hydrocarbons, and organic solvents into soil and groundwater during shipbuilding and maintenance operations. Such contamination not only affects the shipyard premises but also poses serious environmental threats to nearby communities, raising concerns about the long-term sustainability of the shipbuilding industry. Given the increasing global emphasis on sustainable industrial practices, addressing shipyard-related pollution has become a critical environmental challenge. This review aims to provide a comprehensive understanding of the pollution issues associated with shipyards and explore effective remediation strategies. It focuses on contamination in both soil and groundwater, and covers pollution generated throughout the shipbuilding and maintenance lifecycle. First, it examines previous studies to identify the major contaminants and pollution sources typically found at shipyard sites. Next, the paper reviews recent advances in soil and groundwater remediation technologies, including physical, chemical, and biological methods tailored to the unique challenges of shipyard environments. Finally, the review discusses current limitations in remediation practices and outlines potential directions for future research and technological development.
1. Introduction
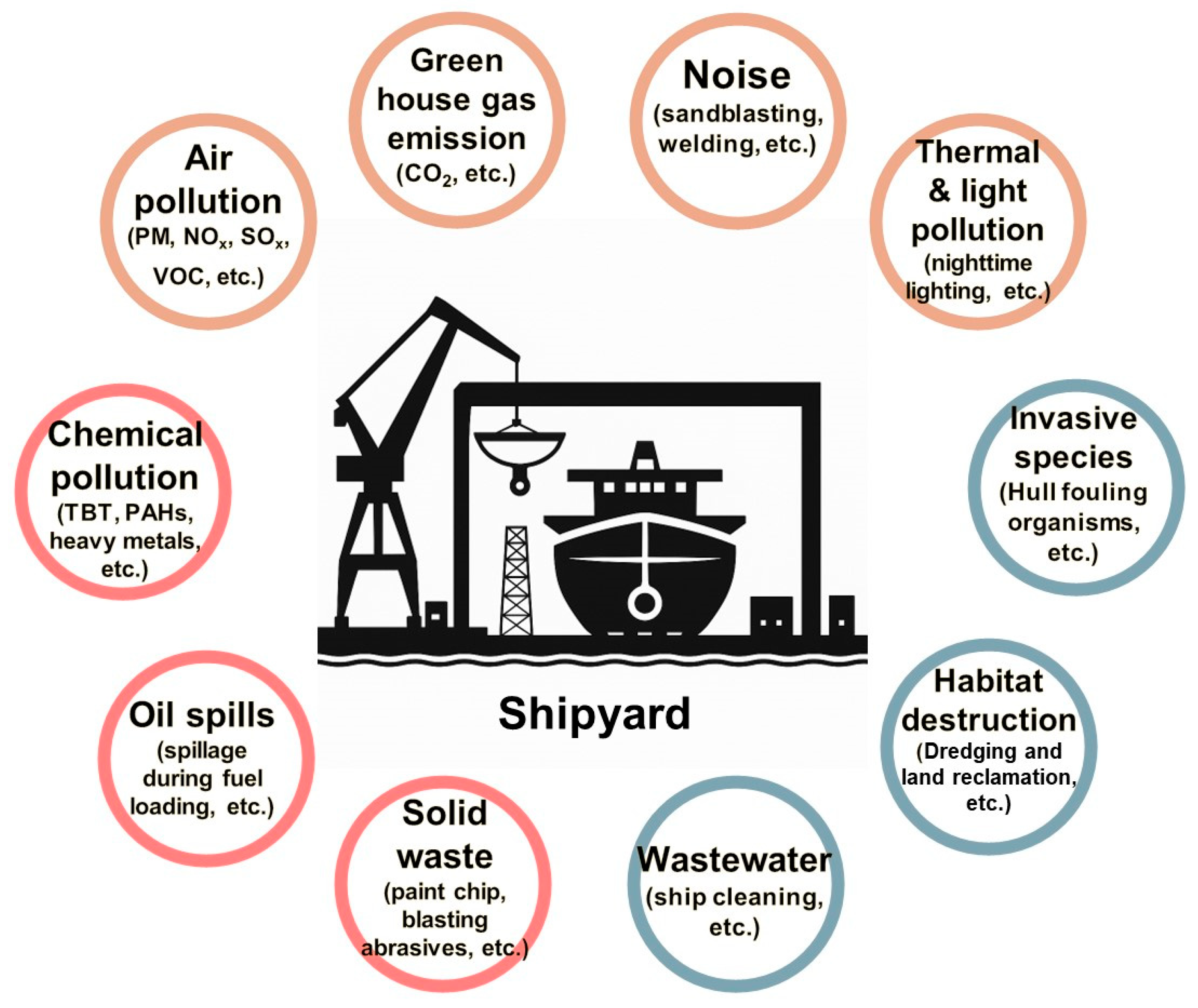
The shipbuilding industry has long played a pivotal role in the national economy and has contributed significantly to various sectors, including maritime transportation and defense [1]. This is not merely about constructing vessels; it is a complex, integrated sector that brings together technologies, materials, and human resources from a wide range of industries. Due to this multidisciplinary nature, various synthetic chemical products are extensively used at shipyard sites, resulting in the release of substantial amounts of pollutants [2] (Figure 1). Despite the significant discharge of pollutants from shipyards, the proper treatment of these contaminants has been insufficient. Although environmental regulations were strengthened in the 1990s, leading to the introduction of wastewater treatment facilities and hazardous substance management systems, large quantities of pollutants have continued to enter the soil, groundwater, and marine environments. This ongoing contamination is evidenced by persistent reports of soil, groundwater, and marine pollution in areas surrounding shipyards [2,3].
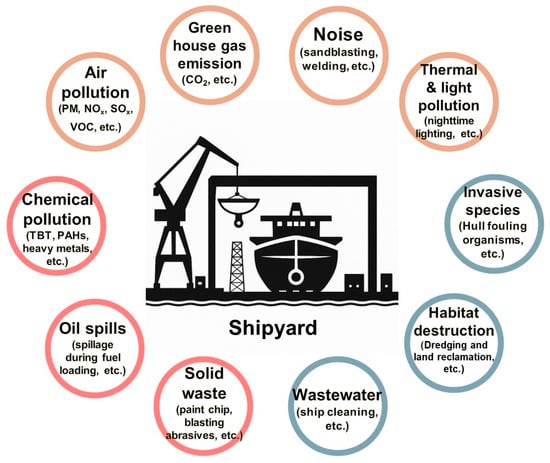
Figure 1.
Environmental impacts of shipyards.
This poses potential environmental risks to the surrounding ecosystems and local communities and may significantly undermine the long-term sustainability of the shipbuilding industry, ultimately affecting future generations [4]. Therefore, to address these issues, continuous monitoring of the environment around shipyards, the adoption of pollution prevention technologies, and the remediation of contaminated environments are essential [5,6]. Furthermore, the remediation technologies must be appropriately tailored to the specific characteristics of the shipbuilding industry.
This review is based on a wide survey of shipyard contamination cases reported across multiple global sites, and focuses on remediation technologies considered applicable to shipyard soils. To this end, we selectively reviewed key studies published over the past five years, with literature retrieved from major databases including Scopus and Web of Science. Keywords such as “shipyard soil contamination,” “remediation technologies,” “heavy metals,” and “persistent organic pollutants” were used. Recent research trends were also considered to reflect the evolving focus on technology integration and field applicability.
In this review, we explore the remediation technologies applicable to shipyard environments that have already been contaminated. To this end, we review previous studies that analyze the contamination of shipyard sites to identify common pollution trends and sources. We then examine both conventional and cutting-edge remediation techniques for restoring contaminated soil and groundwater. Real-world case studies of remediation projects in shipyard contexts are also discussed to assess practical applicability. Furthermore, we analyze the challenges and limitations of current technologies and conclude by presenting future perspectives and potential directions for overcoming these barriers.
Unlike previous reviews that addressed soil remediation in general industrial contexts, this work specifically focuses on the complex contamination conditions unique to shipyard sites and systematically compares practical case studies with emerging remediation technologies.
2. Contaminants Profile in Shipyards
2.1. Key Pollutants and Their Origins
Shipbuilding and dismantling activities have long been recognized as significant sources of hazardous substances released into surrounding soils and marine sediments [2]. These pollutants are not randomly distributed but are closely associated with specific industrial operations, such as metal cutting, surface coating, and equipment dismantling [7]. This section reviews the key contaminants commonly found in soils and sediments near shipyards, based on findings from prior studies, and explores their respective sources.
A summary of the major contaminants detected in shipyard soils, along with their primary sources, maximum concentrations, and exceedance of regulatory standards, is provided in Table 1. Polycyclic aromatic hydrocarbons (PAHs) are among the most prevalent organic pollutants detected in areas adjacent to shipyards [2,8]. These compounds typically originate from high-temperature processes such as fuel combustion and metal cutting. Numerous studies conducted near decommissioned shipyards have reported elevated concentrations of PAHs in both soil and nearby sediments, often exceeding international regulatory thresholds [2,8]. In some cases, PAH congener ratios confirmed a thermal origin, indicating a direct link to dismantling activities involving intense heat [8]. For instance, benzo[a]pyrene and fluoranthene have been detected at concentrations up to 2400 µg/kg in sediment samples, significantly exceeding U.S. EPA and Dutch sediment quality standards [2,8].
Polychlorinated biphenyls (PCBs), once widely used in industrial paints and insulating fluids, have been detected in marine sediments near ship dismantling sites [9]. Due to their chemical stability and lipophilic properties, PCBs persist in sediments over long periods. In certain regions, their concentrations have reached levels associated with potential ecological harm. Concentrations as high as 847 µg/kg have been reported, surpassing the Effects Range Median (ERM) for marine ecosystems, while even lower levels (124 ng/g) still suggest moderate risk [10].
DDT and its degradation products, despite being banned or strictly regulated in many countries, are still found in areas where antifouling paints were historically used. Soil and sediment analyses from a former shipyard in southern China revealed significant accumulations of total DDT, with concentrations far exceeding both national and international safety guidelines [11]. Specifically, levels of up to 235.6 mg/kg have been recorded in sediments, exceeding thresholds set by both Chinese and Dutch regulatory frameworks [11]. These findings underscore the long-term environmental persistence of organochlorine-based coatings.
Tributyltin (TBT), a highly toxic biocide historically used in antifouling paints, continues to be detected in sediments near former shipyard facilities, despite its global ban under the International Maritime Organization (IMO). In some locations, TBT concentrations exceed ecological threshold values, and the compound is known to cause severe endocrine disruption in marine organisms. Measured TBT levels have exceeded 36,000 ng Sn/g in some sediment samples, far above the ERM guideline, with known impacts such as imposex in gastropods and endocrine disruption in higher organisms [12]. Other organotin compounds, such as mono- and dibutyltin, have also been detected at concentrations above 5000 ng/g, exceeding WHO aquatic toxicity thresholds [13,14].
In addition to these persistent organic pollutants (POPs), several heavy metals have also been reported in shipyard environments [2,5,15]. Copper (Cu), commonly found in marine paints and electrical components, has been detected at elevated levels in soils and leachates surrounding inactive shipyards. In some cases, the concentrations exceeded the U.S. EPA’s Universal Treatment Standards (UTS) [16]. Similarly, high Cu levels have been observed in soils near shipyards in South Korea, posing risks to soil quality and ecosystem health. Cu concentrations up to 1321 mg/kg have been reported in soil samples, clearly surpassing ERM values and representing a major ecological hazard [15].
Lead (Pb) remains a key contaminant, particularly in areas where painted surfaces and electrical systems have been dismantled [15]. Pb has been found not only in the soil but also in food crops cultivated near shipbreaking yards, raising concerns about human exposure through the food chain [17]. Its strong binding affinity allows it to persist in soil over long periods, which is consistent with its historical use in paints and electronic materials [18]. Pb concentrations have reached up to 312 mg/kg in shipyard soil, exceeding both ERM and USEPA UTS guidelines [15]. Zinc (Zn), associated with galvanization and surface coatings, has been detected at concentrations exceeding ecological guidelines in multiple shipyard areas [19]. Maximum Zn levels reported include 2147 mg/kg in soil, indicating severe enrichment in contaminated zones [15]. Like Cu and Pb, Zn often co-occurs with other heavy metals, amplifying the cumulative ecological risks to both terrestrial and aquatic environments [20]. Cadmium (Cd), though typically found at lower concentrations, poses a unique risk due to its high mobility and bioaccumulative potential [21]. Soil and crop analyses near shipbreaking yards in Bangladesh revealed the presence of Cd in both soil and leafy vegetables, indicating a direct route for human exposure through diet [16]. Moreover, the slightly acidic to neutral pH of these soils may enhance cadmium mobility, further increasing its environmental impact [16,22]. While absolute Cd concentrations are often modest, their uptake by crops has led to exceedances of WHO/FAO food safety limits, especially for leafy vegetables consumed locally [21].

Table 1.
Major contaminants in shipyards with regulatory standards.
Table 1.
Major contaminants in shipyards with regulatory standards.
| Pollutant | Primary Source | Maximum Detected Concentration | Exceedance of Standards | Refs. |
|---|---|---|---|---|
| PAHs | Fuel combustion, high-temperature cutting | 2400 μg/kg (sediment) | Exceeds U.S. EPA and Dutch standards | [2] |
| DDT | Antifouling paints | 235.6 mg/kg (sediment) | Exceeds Chinese and Dutch soil quality standards | [11] |
| PCBs | Electrical equipment, industrial coatings | 124.33 ng/g (sediment) | Exceeds moderate to heavy contamination level | [23] |
| 847 μg/kg | Exceeds effects range median (ERM) | [10] | ||
| OCPs (DDT derivatives) | Residues from antifouling agents | – | Exceeds ERM and effect range low (ERL) standards | [10] |
| TBT | Antifouling paints, ship surface coatings | 36,292 ng Sn/g | Banned under IMO; exceeds ERM | [12] |
| Cu | Paint coatings, dismantling of electrical cables | 1321 mg/kg | Exceeds ERM | [15] |
| Pb | Fuel residues, corrosion of painted surfaces | 312 mg/kg | Exceeds ERM and U.S. EPA Universal Treatment Standards (UTS) | [15] |
| Zn | Metal corrosion, antifouling paints | 2147 mg/kg | Exceeds ERM | [15] |
| Cd | Welding, antifouling agents | High plant transfer factor | Exceeds WHO/FAO plant intake standards | [21] |
| Other Organotin Compounds | Antifouling paints, paint waste | 5380 ng/g (sediment) | Exceeds ERM and WHO aquatic toxicity thresholds | [13,14] |
2.2. Ecotoxicity and Human Health Risk
As previously discussed, a diverse array of contaminants, including PAHs, heavy metals, organochlorine pesticides, and organotin compounds, coexist in shipyard environments. While each of these substances poses significant individual toxicity, there is increasing concern about their potential to exert synergistic effects when present together [24,25].
Table 2 summarizes the ecological and human health effects of major contaminants commonly detected at shipyard sites. To begin with PAHs, high-molecular-weight compounds such as benzo[a]pyrene and benz[a]anthracene are well-documented for their carcinogenic and mutagenic properties. In a study conducted in Hong Kong, Chiu et al. [2] found that concentrations of these PAHs exceeded the U.S. EPA guidelines as well as the Soil Screening Levels established for Superfund Region III. Furthermore, the study reported a BOD5/COD ratio of 0.021 in leachate, indicating the presence of POPs with low biodegradability, thereby limiting the self-purification capacity of the soil. Guo et al. [11] conducted a risk assessment under a residential exposure scenario and found that DDT exposure via soil ingestion and dermal contact resulted in estimated cancer risks exceeding the threshold of 10−6 to 10−5. These findings highlight the long-term health implications of chronic exposure to organochlorine pesticides. Yılmaz et al. [10] identified high ecotoxicity in the Aliağa region of Turkey, with sediment concentrations of PAHs and PCBs surpassing the ERM. Additionally, total DDT levels exceeded the Effects Range Low (ERL) at all sampled locations, suggesting widespread ecological stress.

Table 2.
Ecotoxicity and human health risk of the pollutants detected in shipbuilding yards.
Organotin compounds, particularly TBT, are regarded as among the most persistent and toxic pollutants found in shipyard sediments [12]. Despite a global ban imposed by the IMO, Kim et al. [12] reported TBT concentrations exceeding 580 ng/g, which are known to induce imposex in gastropods and endocrine disruption in higher organisms. According to Kotrikla and Staniszewska et al. [13,14], other organotin compounds similarly exhibit severe neuroendocrine and immune system toxicity.
Heavy metals such as Cu, Pb, Zn, and Cd have also consistently exceeded ERM benchmarks in various studies [2,17,19,21]; Chowdhury and Rasid [21] observed significant accumulation of these metals, particularly Cd, in the edible parts of leafy vegetables, with levels surpassing the WHO/FAO food safety guidelines. Given their high bioavailability and mobility, these metals pose long-term risks to food safety and are associated with chronic toxicities, including renal impairment and skeletal disorders.
The interactive effects of these pollutants are further exemplified in a study by Choi and Jeon [15], who investigated a shipyard site in Busan, South Korea. They reported severe contamination (Enrichment Factor > 10) and observed combined organic and inorganic pollution. This mixed pollution scenario was found to exert significant ecotoxicological pressure on benthic organisms and to impair overall ecosystem functioning.
Therefore, minimizing contaminant emissions is essential, but comprehensive site remediation is equally critical to reduce long-term risks and support the redevelopment of former shipyard sites while minimizing adverse impacts on the surrounding environment.
3. Recent Advancement of Remediation Technologies
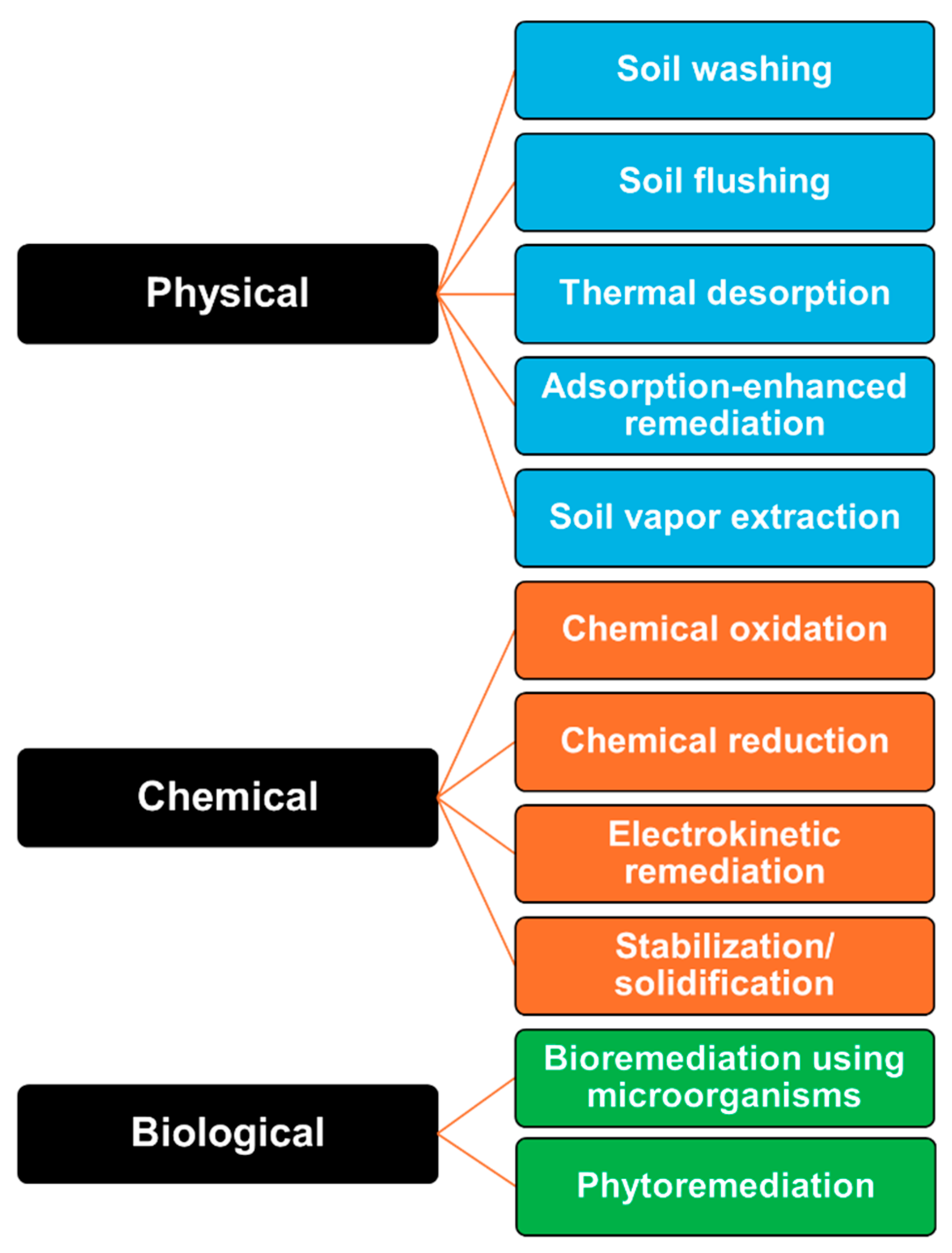
Various technologies are currently being applied to soil remediation [26,27,28]. In this section, soil remediation methods applicable for shipyard remediation are categorized into physical, chemical, and biological treatments. The fundamental principles of each approach and representative studies conducted to date are reviewed (Figure 2).
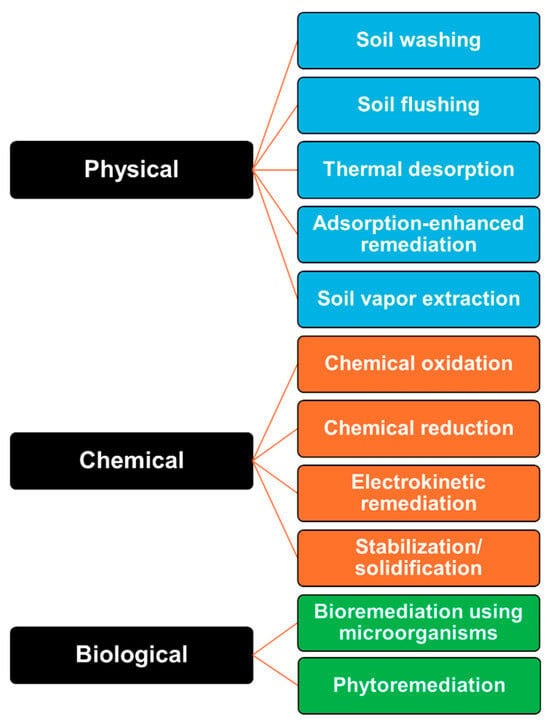
Figure 2.
Classification of remediation technologies.
3.1. Physical Remediation
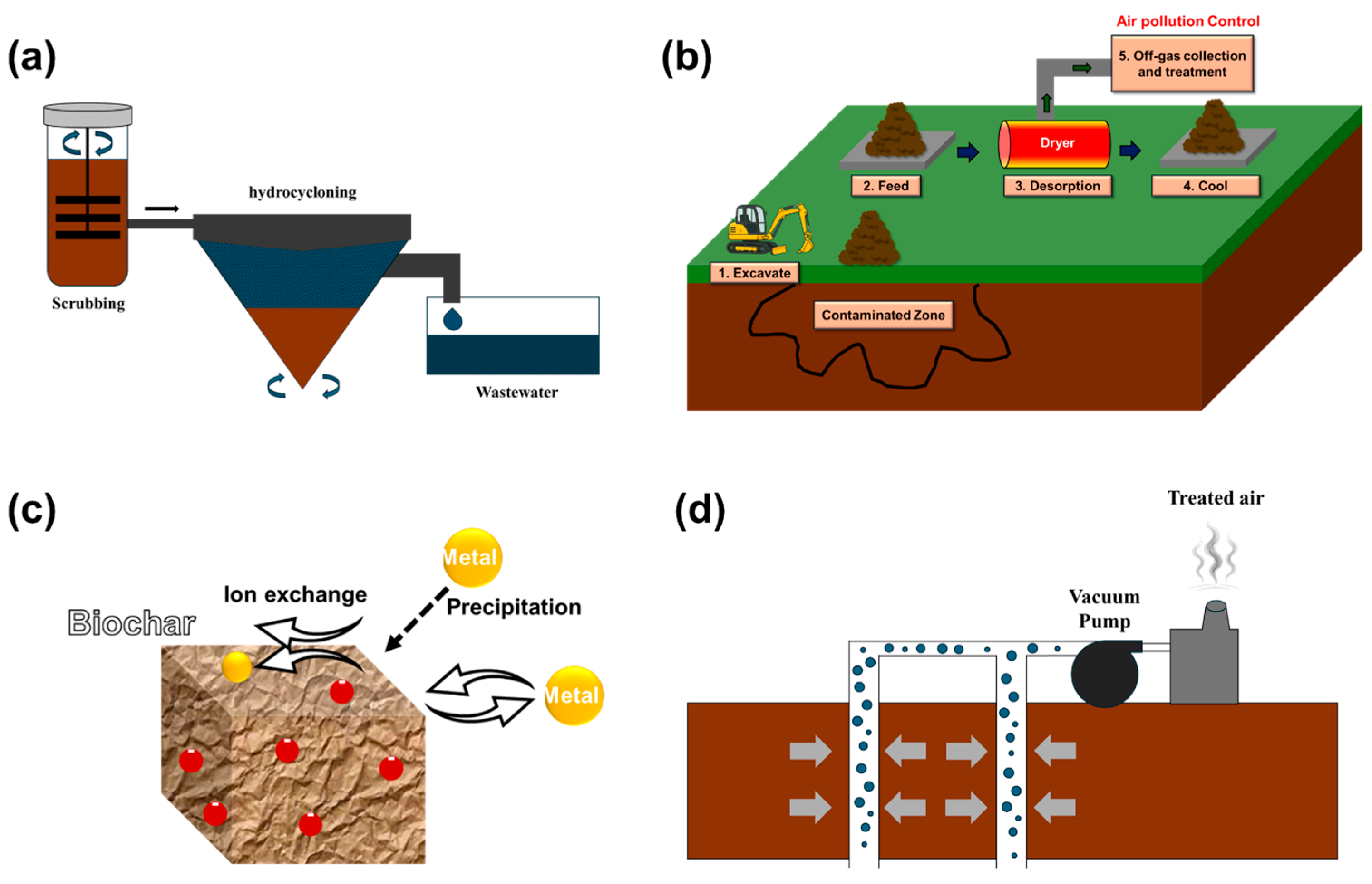
Physical remediation technologies aim to remove or isolate contaminants from polluted soil using mechanical or physical means. These methods are particularly effective in complexly contaminated environments such as shipyard sites. Key approaches include soil washing, thermal desorption, adsorption-based treatments, and soil vapor extraction (SVE). The underlying principles of each technique, along with recent representative studies, are summarized in Figure 3 and Table 3.
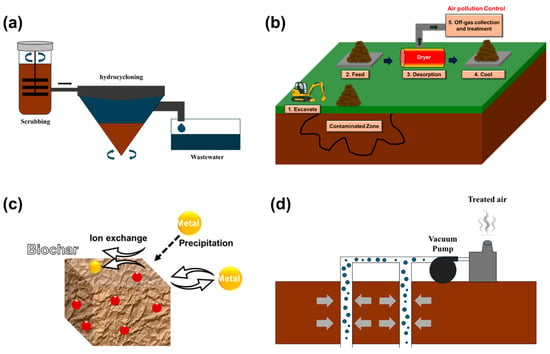
Figure 3.
Illustration of representative physical remediation techniques: (a) soil washing, (b) thermal desorption, (c) adsorption-enhanced remediation, and (d) soil vapor extraction [26,27,28,29].
3.1.1. Soil Washing
Soil washing, a representative physical remediation technique, is increasingly recognized as an effective and relatively fast solution for treating complex contamination typically found at industrial sites like shipyards [30]. The process involves excavating contaminated soil and applying washing solutions, such as acids, chelating agents, or surfactants, to separate and remove pollutants that are bound to soil particles through physical and chemical interactions (Figure 3a) [31]. This method has proven especially efficient in removing heavy metals [31,32]. In addition, it shows promising results for certain organic pollutants such as Total petroleum hydrocarbon (TPH) and PAHs when surfactant-based agents are used [33]. Given the diversity of shipyard activities, including painting, metal cutting, and machinery dismantling, contamination often includes both metals and POPs. Therefore, soil washing is particularly well-suited for sites where pollutants are concentrated in surface soil and excavation is feasible.
Soil washing has long been recognized as an effective remediation strategy for metal-contaminated soils [31,32]; however, traditional methods are often limited by high wash water demand and poor selectivity for co-existing organic pollutants [31]. Recent studies have addressed these limitations by incorporating nanomaterials and biodegradable chelating agents, thereby improving contaminant removal while minimizing secondary pollution. For instance, Grimison et al. [34] demonstrated field-scale application of soil washing for PFAS-contaminated firefighting sites, achieving an average PFOS reduction of ~90% using a recycled washing solution. Similarly, Yuan et al. [35] applied citric acid-based washing with nZVI-supported biochar for Cr(VI) removal and showed effective recovery of the washing agent, achieving 79.9% removal from soil and 98.9% from effluent. Addressing PAH contamination, Zhang et al. [33] used surfactant-enhanced washing on coking plant soil, reaching up to 50.2% removal under optimized multi-step conditions. Further, Rabieian and Taghavijeloudar [36] integrated advanced oxidation (UV/O3) into a continuous soil washing system for phenanthrene remediation, achieving over 98% removal from soil and complete effluent detoxification.
Innovative techniques have also been proposed for metal recovery from wash water, such as the electrochemical treatment of EDTA complexes by Huang et al. [37], enabling up to 95% heavy metal removal with 96.8% EDTA recovery. For aged PAH-contaminated soils, Xing et al. [38] employed thiacalix-4-arene tetrasulfonate, achieving notable removal efficiencies (up to 55.1%) while elucidating interaction mechanisms via spectroscopic analyses. Corres et al. [39] explored electrostatic separation to optimize washing conditions for ten toxic elements, revealing variable recovery efficiencies based on element type and operating voltage. Finally, Keochanh and Phenrat [40] showed that magnet-assisted nZVI significantly enhanced chlorpyrifos removal (>99.95%), demonstrating the synergy of magnetic separation with nanoscale reactive media. These recent developments collectively reflect a shift toward more selective, resource-efficient, and integrative soil washing systems, particularly relevant to industrial sites such as shipyards, where mixed contaminant profiles pose additional remediation challenges.
3.1.2. Thermal Desorption
Thermal desorption is a physical remediation technology that removes contaminants from soil by heating it to volatilize or decompose organic pollutants, and in some cases, certain heavy metals such as Cd [41,42] (Figure 3b). This method is particularly advantageous for industrial sites like shipyards where complex contamination is prevalent, due to its rapid treatment capability and ease of application [13].
Recent research has focused on improving both efficiency and ecological compatibility through optimized processes and integrated treatment approaches. For example, Liu et al. [43] investigated the thermal behavior of PAH-contaminated soil, emphasizing pollutant volatilization patterns and changes in soil functionality after treatment. Similarly, Sang et al. [44] demonstrated the effectiveness of low-temperature indirect thermal desorption for lubricating oil-contaminated soil, reporting significant contaminant removal alongside microbial community recovery and improved soil properties.
Wang et al. [45] applied thermal desorption to heavy petroleum-contaminated soil, achieving effective contaminant reduction and ecological risk mitigation while highlighting the potential for reuse of treated residues. Bykova et al. [46] evaluated treatment performance across diverse landscapes (tundra, taiga, forest-steppe), underscoring the need for site-specific operational strategies.
In terms of optimization, Li et al. [42] developed a sustainability-based evaluation tool to guide the operational design of thermal desorption systems, enhancing overall removal performance. Hu et al. [27] combined low-temperature thermal desorption with secure landfilling for oil-based drill cuttings, demonstrating pollutant control, reduced health risk, and economic feasibility.
Complementary strategies have also emerged. Ilyas et al. [47] compared the effects of pyrochar-assisted phytoremediation and thermal desorption for crude oil-contaminated soil, revealing synergistic benefits in soil recovery and contaminant reduction. Zhang et al. [48] proposed an optimized numerical model to guide thermal desorption for pyrene-contaminated soils, offering new insights into temperature–time optimization. Xue et al. [49] explored electromagnetic induction-based thermal desorption and demonstrated effective remediation of petroleum hydrocarbons at lower energy inputs. Finally, Yue et al. [50] applied biomass co-pyrolysis to Cd-contaminated soil, achieving Cd removal efficiencies exceeding 96% in laboratory soil and 92% in field soil at 550 °C under a nitrogen atmosphere. Collectively, these studies demonstrate a clear evolution of thermal desorption from simple high-temperature treatment toward more integrated and precise remediation technologies that can effectively address mixed contaminants.
3.1.3. Adsorption-Enhanced Remediation
Adsorption-enhanced remediation is a physicochemical technique that selectively removes or immobilizes heavy metals and certain organic pollutants from contaminated soils using solid-phase sorbents (Figure 3c). In recent years, significant advances have been made in the development of bio-based sorbents, many of which offer improved performance and compatibility with other remediation technologies. Zhang et al. [51] demonstrated that MnO2-coated rice husk biomass effectively removed cadmium from both soil and aqueous phases, highlighting its potential as a high-performance sorbent prepared via a simple synthesis route. Similarly, Chen et al. [52] applied carboxylated cellulose nanocrystals embedded with α-FeOOH for the simultaneous removal and immobilization of Pb(II), Cd(II), and As(V), identifying synergistic effects of adsorption and precipitation processes. Lin et al. [53] reported that biochar/g-C3N4 composites exhibited strong adsorption and degradation capacities for petroleum hydrocarbons, indicating their potential for addressing organic contaminants.
In the context of mixed contamination, Qi et al. [54] utilized biochar loaded with mixed bacteria to enhance the immobilization of uranium and cadmium in co-contaminated soils. Li et al. [55] developed polyaniline-embedded polyvinyl alcohol/sodium alginate hydrogel beads for Cr(VI) removal, extending their application from water treatment to soil remediation. Qi et al. [56] further improved adsorption efficiency for Cu2+, Cd2+, and Pb2+ using MgO-loaded nitrogen and phosphorus self-doped biochar, while Zhang et al. [57] employed ball-milled, phosphorus-loaded biochar to remediate alkaline soils contaminated with Pb and Cd.
Lastly, Li et al. [58] integrated β-cyclodextrin/hydrothermal biochar derived from waste cotton into an electrokinetic-permeable reactive barrier (EK-PRB) system, achieving both high metal immobilization efficiency and resource circularity. These studies collectively demonstrate a trend toward multifunctional, sustainable sorbents that not only enhance remediation performance but also address complex contaminant mixtures characteristic of industrial sites.
3.1.4. Soil Vapor Extraction
SVE is an in situ remediation technology that removes VOCs and aromatic hydrocarbons (e.g., BTEX) by extracting contaminated vapors from the unsaturated zone (Figure 3d) [59]. This method is particularly effective in non-cohesive soils with high porosity and is widely applied for the early-stage treatment of volatile contaminants due to its low energy demand and ease of implementation [60].
Recently, Zhang et al. [29] developed a machine learning-based predictive model to evaluate the efficiency of SVE systems, identifying contaminant concentration, soil moisture content, and temperature as critical influencing parameters. Liu et al. [61] combined pneumatic fracturing with SVE to enhance VOC removal in low-permeability clay soils, effectively overcoming mass transfer limitations caused by soil structure.
Additionally, Lyu et al. [59] proposed a sequential treatment system that integrated SVE with an internal combustion engine to utilize extracted vapors as a fuel source, thereby minimizing secondary emissions. Xu et al. [62] improved SVE efficiency in low-permeability soils by establishing pressure gradients through air sparging, which enhanced the desorption and mass transfer of volatile pollutants.
Meanwhile, de Melo Henrique et al. [63] employed an electrokinetic soil flushing-enhanced SVE system to significantly improve the removal of hexachlorocyclohexanes, highlighting its potential for treating persistent halogenated compounds. These advancements collectively indicate a shift toward hybridized SVE processes that are tailored for complex site conditions and contaminant profiles.

Table 3.
Previous studies of physical remediation techniques.
Table 3.
Previous studies of physical remediation techniques.
| Method | Soil Type | Contaminants | Experimental Conditions | Efficiency | Refs. |
|---|---|---|---|---|---|
| Soil washing | Clay-rich firefighting site | PFASs (PFOS and PFHxS, etc.) | Field-scale soil washing with agent recycling | Avg. ~90% removal (PFOS116→11 µg/L) | [34] |
| Cr-spiked agricultural | Cr | Citric acid wash + nZVI-BC for effluent | 79.9% (soil), 98.9% (effluent) | [35] | |
| Coking plant soil, low TOC | 16 PAHs (e.g., FLT, PYR, and BaP) | Single/multi-step surfactant washing | ~50.2% (best, TX100 multi-wash) | [33] | |
| Phenanthrene (PHE) | Phenanthrene-spiked loam | Ultrasound + surfactant wash + UV/O3 | 98% (soil), 100% (effluent) | [36] | |
| Soil washing effluent (not direct soil) | Cu, Pb, and Cd (as EDTA complexes) | Electrochemical AC process with graphite electrodes | 49–95% metal removal, 96.8% EDTA recovery | [37] | |
| Fine sandy clay loam, artificially spiked with PAHs (Nap, Flua, Pyr); aged 1 year | Naphthalene (Nap), Fluoranthene (Flua), and Pyrene (Pyr) | Batch soil washing using thiacalix-4-arene tetrasulfonate (TCAS) under controlled pH and temperature; interaction mechanisms studied via FTIR, NMR, UV-vis, and computational modeling | Nap: 55.1%, Flua: 47.7%, Pyr: 50.0% at 0.7% TCAS concentration | [38] | |
| Soil from a former lead mining district in Linares, Spain; granulometric fraction (1000–2000 µm) | 10 potentially toxic elements (PTEs): As, Cd, Cr, Cu, Hg, Mo, Ni, Pb, Sb, and Zn | Peak recoveries at 40 kV: Zn (83.3%), Mo (81.0%), Cu (62.2%), Ni (57.3%) Lowest for Hg (13.2%) | Electrostatic separation at 12 voltages (20–41.5 kV); optimized via a new PPAA-U (Penalized Attributive Analysis) index that balances yield and recovery while penalizing variance | [39] | |
| Silt loam from Thailand, artificially spiked with 40 mg/kg Chlorpyrifos | Chlorpyrifos (CPF) | Magnetic separation + nanoscale zero-valent iron (nZVI) assisted by activation and reductive/oxidative degradation processes | >99.95% CPF removal using activated AZ or AZ + H2O2 | [40] | |
| soil washing effluent (SWE) containing PHE, not the soil itself | Phenanthrene (PHE), one of the U.S. EPA priority PAHs | Ultrasound-activated persulfate (US/PS) oxidation with APG surfactant; reaction influenced by pH, PS dose, temperature, anions, and organic matter; mechanism confirmed by EPR, DFT, GC-MS | Up to 79.8% degradation in SWE (initial PHE = 6 mg/L, optimized US/PS conditions) | [64] | |
| Real uranium-contaminated soil with different Fe/U ratios (5:1, 2.5:1, 1.5:1); up to 160 Bq/g U(VI) | U(VI), Fe, Al, Si, Ca, Mg, K, and Na | 3-cycle soil washing with H2SO4 followed by pH-adjusted neutralization using hydrazine (N2H4) for co-precipitation; major metal behavior analyzed by ICP-OES and XRF | ≥99.4% U(VI) removal with 3-step washing using ≥0.05 M H2SO4 Clearance levels achieved: <81 ppm (1 Bq/g) | [65] | |
| Thermal desorption | Loess soil (Gansu, China) | Phenanthrene, Pyrene, and Benzo(a)pyrene (PAHs) | 50–800 °C, 2 h, argon atmosphere, tube furnace | >99% removal at 400 °C; <5% below 200 °C | [43] |
| Loam, clay, sand (China) | Waste lube oil | 200–500 °C, 60 min, indirect TD under N2 atmosphere | 93% at 350 °C | [44] | |
| Artificially contaminated clay loam soil (Shenyang, China) | Heavy petroleum (alkanes, aromatics, resins, and asphaltenes) | 100–600 °C, 5–60 min, bench-scale furnace (TD system) | 87.26–98.69% at 500–600 °C after 60 min | [45] | |
| Gleysols, Podzols, Phaeozems (Russia) | Petroleum hydrocarbons | Ex situ thermal desorption, 250 °C | Reduced from up to 24,850 mg/kg to ≤410 mg/kg | [46] | |
| Artificially diesel-contaminated soil (Baoding, China) | Diesel hydrocarbons (C10–C40) | Muffle furnace; 150 °C, 350 °C, 550 °C; treated until ≤826 mg/kg | 350 °C: Residual ≈ 280 mg/kg; 550 °C: Residual ≈ 5 mg/kg | [42] | |
| Oil-based drill cuttings (Hubei, China) | PAHs, Heavy metals (As, Cr, etc.) | Pilot rotary reactor; 250–300 °C, 10–30 min | PAHs: up to 96.27% at 300 °C / 30 min; Heavy metals: low | [27] | |
| Crude oil-spiked soil (Rawalpindi, Pakistan) | Crude oil (10%, 20%) | Thermal desorption at 500 °C, 60 min, lab scale | 19.7% degradation at 20% contamination level | [47] | |
| Sandy soil (Suqian, China) | Pyrene (PAH) | Bench-scale TD; 100–400 °C, 2–60 min; optimized temp: 200 °C | 99.1% at 400 °C/10 min, 94.9% at 200 °C/40 min | [48] | |
| Diesel-spiked sandy soil (Wuhan, China) | TPH | Electromagnetic induction LTTD; 200–300 °C, 20–60 min | 93.44–97.65% (optimum at 216 °C, 21 min) | [49] | |
| Cd-contaminated soil (spiked + field soil, Hunan, China) | Cd | Biomass co-pyrolysis (WS/soil = 0.4), 550 °C, 1 h, N2 atmosphere | Up to 96.5% (lab soil); >92% (4 field soils) | [50] | |
| Adsorption-enhanced remediation | Petroleum-contaminated soil | Cd | MnO2 modified biochar | 115.04 mg g−1 (Cd) | [51] |
| Contaminated soil | Pb, Cd, and As | Fe/cellulose nanocrystals | 126.06 (Pb), 53.07 (Cd), and 15.80 mg g−1 (As) | [52] | |
| Petroleum-contaminated soil | TPH | Biochar/g-C3N4 | 80 mg g−1 (TPH) | [53] | |
| U and Cd co-contaminated soil | U and Cd | Mixed bacteria 9 | 69% (U) and 56% (Cd) | [54] | |
| Artificially contaminated soil | Cr | Polyaniline/polyvinyl alcohol/sodium alginate composite gel bead | 83.1 mg g−1 (Cr) | [55] | |
| Heavy metal-contaminated soils | Cu, Cd, and Pb | MgO-loaded fish scale biochar | 505.8 (Cu), 327.2 (Cd), and 661.2 mg g−1 (Pb) | [56] | |
| Farmland soil | Cd and Pb | Ball-milled P-loaded biochar materials | 18.7 (Cd) and 126 mg g−1 (Pb) | [57] | |
| Heavy metal-contaminated soils | Pb and Cd | Novel β-CD/hydrothermal biochar | 50.44 (Pb) and 33.77 mg g−1 (Cd) | [58] | |
| Soil vapor extraction (SVE) | Sandy, silty, clay | Organic pollutants including VOCs (e.g., BTEX), SVOCs, and chlorinated hydrocarbons | Machine learning (XGB, SVM, ANN, RF) trained on 11 variables (e.g., time, temperature, soil structure) | Predicted with R2 = 0.9799 using XGB model; actual removal varies by conditions | [29] |
| Low-permeability clay artificially contaminated with ammonia gas (NH3); lab-scale soil column and 2D plate models | Ammonia gas | Pneumatic fracturing combined with SVE; tested air pressure (0.1–0.5 MPa), fracturing point spacing (10–25 cm), and extraction modes (continuous, intermittent, stepwise, gas-assisted) | Up to 98% removal under continuous extraction + bottom gas injection at 20 kPa; optimal vapor flow rate = 9 L/min | [61] | |
| Loam soil spiked with toluene (0.25–1 g/kg); water content adjusted (1–10%) | Toluene | Soil Vapor Extraction (SVE) + Internal Combustion Engine (ICE) for VOC off-gas destruction; optimized via Response Surface Methodology (RSM); thermal reuse of ICE exhaust applied to SVE | SVE: 100% DRE (Destruction Removal Efficiency) via ICE Desorption time optimized to 615 min at 0.3 g/kg, 10 L/min air flow, 9% moisture Pulsed operation improved efficiency and saved 1.104 kWh/kg | [59] | |
| Low-permeability soil (clayey), spiked with MTBE | MTBE (a common gasoline additive VOC) | Air sparging | Up to 28% MTBE removal at 100 kPa with enhanced air sparging; improvement over conventional by +23.7% | [62] | |
| Synthetic compacted silty loam | γ-HCH, ε-HCH, α-HCH, and δ-HCH | Up to 92.9% removal in 15 days with SVE + EKSF at 3.0 V/cm (vs. 70% with SVE only) | Combined Soil Vapor Extraction (SVE) and Electrokinetic Soil Flushing (EKSF) | [63] |
3.2. Chemical Remediation
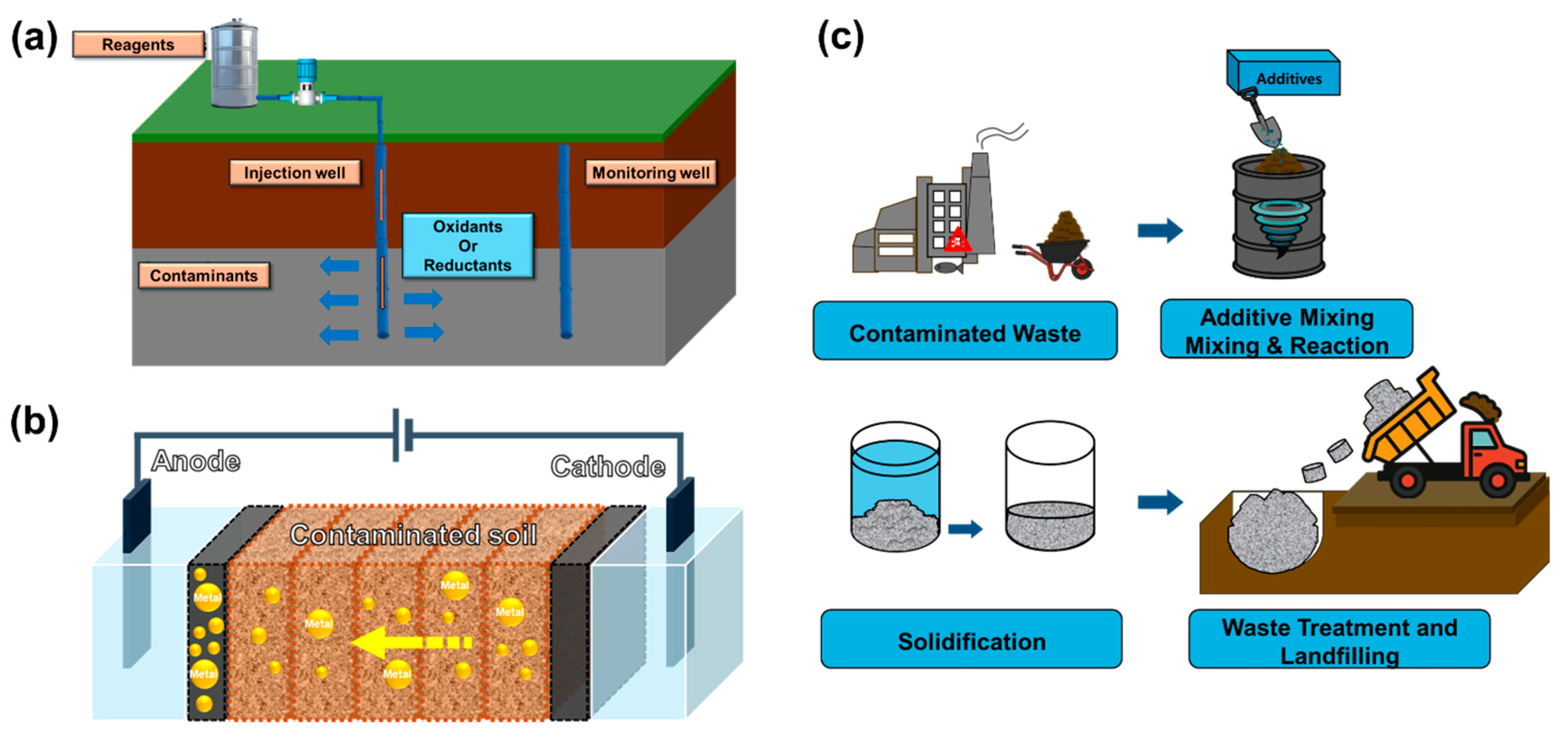
Chemical remediation encompasses a range of in situ treatment techniques that leverage redox reactions to transform or degrade contaminants directly within the subsurface [66]. It is particularly effective for managing a broad spectrum of pollutants, including organic compounds and toxic metals, which are commonly encountered in industrial sites [30]. Recent advances focus on enhancing the selectivity, sustainability, and field applicability of chemical oxidants and reductants through the integration of nanomaterials, catalysts, and bio-based activators. These technologies can be broadly categorized into three main approaches such as chemical oxidation and reduction, electrokinetic remediation, and stabilization/solidification. Each method targets specific contaminant profiles and geochemical conditions, as illustrated in Figure 4. Recent studies demonstrating their efficiency and innovation across diverse contaminated sites are systematically summarized in Table 3.
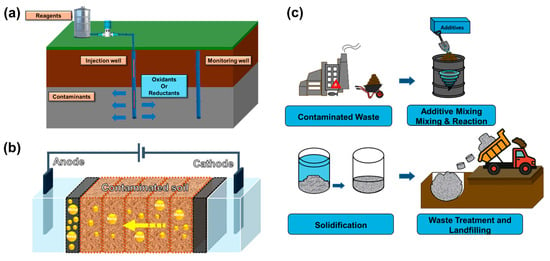
Figure 4.
Illustrations of representative chemical remediation techniques: (a) Chemical oxidation and reduction, (b) electrokinetic remediation, and (c) stabilization/solidification [67,68,69].
3.2.1. Chemical Oxidation and Reduction
Chemical oxidation and chemical reduction are widely applied in situ remediation techniques designed to treat a broad range of contaminants, including VOCs, PAHs, chlorinated solvents (such as trichloroethylene and perchloroethylene), and toxic metals (Figure 4a). These methods are particularly relevant for industrial sites, where co-contamination is common and rapid, and large-scale treatment is often necessary [70].
Recent developments have focused on combining nanomaterials, biodegradable activators, and biochar to improve treatment efficiency, sustainability, and field applicability. The evolution of these methods reflects a shift toward integrated remediation approaches that minimize secondary pollution and adapt to site-specific conditions.
For example, Zeng et al. [71] developed a pilot-scale system using biochar-supported nanoscale zero-valent iron to activate persulfate for treating soil contaminated with aromatic hydrocarbons. This approach significantly improved degradation efficiency under real field conditions.
Song et al. [72] demonstrated the effectiveness of sulfidated nanoscale zero-valent iron in treating cadmium-contaminated groundwater at a zinc smelter site. Their study confirmed high reduction efficiency and operational stability in highly acidic environments.
Wang et al. [73] introduced a novel oxidation strategy using polymetallic-doped graphitic carbon nitride to generate hydrogen peroxide directly within the contaminated zone. This in situ oxidant generation method reduces the need for external chemical inputs, lowering costs and environmental risks.
In the treatment of persistent chlorinated organics, Liu et al. [74] combined chemical reduction with enhanced bioremediation to accelerate trichloroethylene degradation in groundwater. Their integrated system showed superior performance in both speed and long-term stability. Reginatto et al. [75] explored the application of nanoscale zero-valent iron in clay-rich soils contaminated with hexavalent chromium. Laboratory-scale column tests confirmed the feasibility of applying this technology to low-permeability soils typical of shipyard environments.
Recent research has also emphasized the ecological impact of these chemical processes. Geng et al. [76] used high-resolution mass spectrometry to assess the structural transformation of soil organic matter after oxidation treatments. Meanwhile, Hong et al. [77] investigated how different oxidation methods affect microbial community structure in petroleum-contaminated soils. Rodrigues et al. [78] provided a thorough overview of field applications of chemical reduction methods, particularly focusing on the use of zero-valent metals in permeable reactive barriers to remediate chlorinated organic pollutants in complex contaminated sites such as shipyards. Lastly, Mouvet et al. [79] reported on a field-scale chemical reduction project targeting chlordecone-contaminated soils in the French Caribbean. The study not only demonstrated successful contaminant immobilization but also evaluated soil recovery and agronomic potential post-treatment.
3.2.2. Electrokinetic Remediation
Electrokinetic remediation is an in situ technique that employs low-intensity direct current to mobilize and extract contaminants from fine-grained or low-permeability soils such as clay and silt [30]. As illustrated in Figure 3b, an electric potential is applied between embedded electrodes (anode and cathode) in the contaminated soil, inducing electroosmotic flow, electromigration, and electrophoresis [80]. These mechanisms facilitate the transport of charged contaminants, such as metal ions, toward the electrodes, where they can be collected or treated [81].
Recent research has focused on optimizing electrode configurations, enhancing electrokinetic transport using surfactants or biodegradable complexing agents, and integrating electrokinetic systems with biological or chemical treatment processes to increase remediation efficiency; Kim et al. [81] compared one-dimensional and two-dimensional electrode arrangements and reported that the two-dimensional system significantly improved the removal efficiency of Pb and Zn from mine-impacted soils. Shi et al. [82] combined electrokinetics with bioremediation to treat trichloroethylene (TCE) in low-permeability soils, observing enhanced microbial activity and dehalogenation near the electrode zones. Similarly, Narenkumar et al. [83] applied a biosurfactant-assisted bio-electrokinetic system, achieving effective mobilization and removal of Cu and chromium (Cr), while maintaining microbial compatibility. Sun et al. [84] demonstrated that electrode placement plays a critical role in soft clay environments, where a vertical anode–cathode arrangement enhances electroosmotic flow. In another study, Wang et al. [85] used biodegradable complexing agents to chelate Pb and Cu, which significantly improved their mobility and removal efficiency, achieving over 75% removal.
3.2.3. Stabilization/Solidification
Stabilization/solidification is a passive in situ or ex situ remediation technique that physically immobilizes contaminants by incorporating them into a stable solid matrix, thereby reducing their leachability and environmental mobility [86]. As shown in Table 4 and illustrated in Figure 4c, Recent advances in stabilization/solidification technologies focus on low-carbon binders, industrial waste reuse, and long-term durability under environmental stress. Several studies have reported excellent performance improvements in both contaminant immobilization and mechanical integrity. Fan et al. [86] compared coal fly ash-based geopolymer with ordinary Portland cement for solidifying MSWI fly ash. The geopolymer achieved over 95% reduction in Pb leachability and demonstrated higher unconfined compressive strength (10.4 MPa) than cement (6.2 MPa), meeting both environmental and structural performance thresholds. Wang et al. [87] developed a low-alkalinity, clay-based binder to stabilize Pb-contaminated soils without cement. The treatment reduced Pb leaching by approximately 90% and maintained compressive strength of ~2 MPa after 28 days—suitable for capping or subgrade applications. Chen et al. [88] applied a composite of red mud and blast furnace slag under alkali activation to Cd-contaminated soils. Cd leachability was suppressed by over 96%, and compressive strength improved to 5.2 MPa with a 10% binder dosage. Xia et al. [89] evaluated a hydroxyapatite-based binder in field-scale applications at a Pb-Zn smelter site. The system retained strength above 3.5 MPa over 90 days and reduced Pb concentrations to <5 mg/L, remaining within regulatory leaching limits. Liu et al. [90] investigated the durability of a Zn-contaminated soil solidified with a cement-soda residue mix under freeze–thaw cycling. After six cycles, Zn leaching remained below 2 mg/L, and compressive strength was maintained at ~4.1 MPa. Zhang et al. [91] compared calcium oxide (CaO) and magnesium oxide (MgO) activated GGBS binders for Zn-contaminated clay slurry. MgO-activated binders outperformed CaO in both Zn immobilization (88%) and unconfined strength (~6.5 MPa), indicating better stability and lower long-term risk.

Table 4.
Previous studies of chemical remediation.
3.3. Biological Remediation
Biological remediation refers to the use of microorganisms, plants, or their enzymatic systems to degrade, transform, or immobilize environmental contaminants under natural or engineered conditions [30]. Compared to physicochemical approaches, biological methods are generally more sustainable and better suited for the long-term treatment of organic pollutants and selected heavy metals. These techniques are applicable to shipyard soils where mixed contamination is prevalent.
As illustrated in Figure 5 and summarized in Table 3, biological remediation can be broadly categorized into microbial and phytoremediation strategies. Each has been applied either individually or in combination with other techniques to improve remediation efficiency and ecological compatibility.
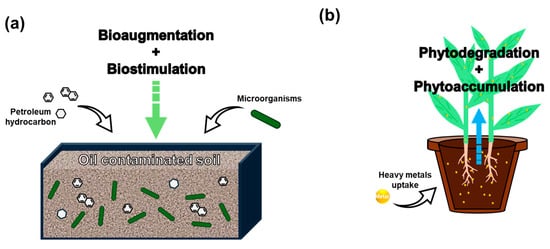
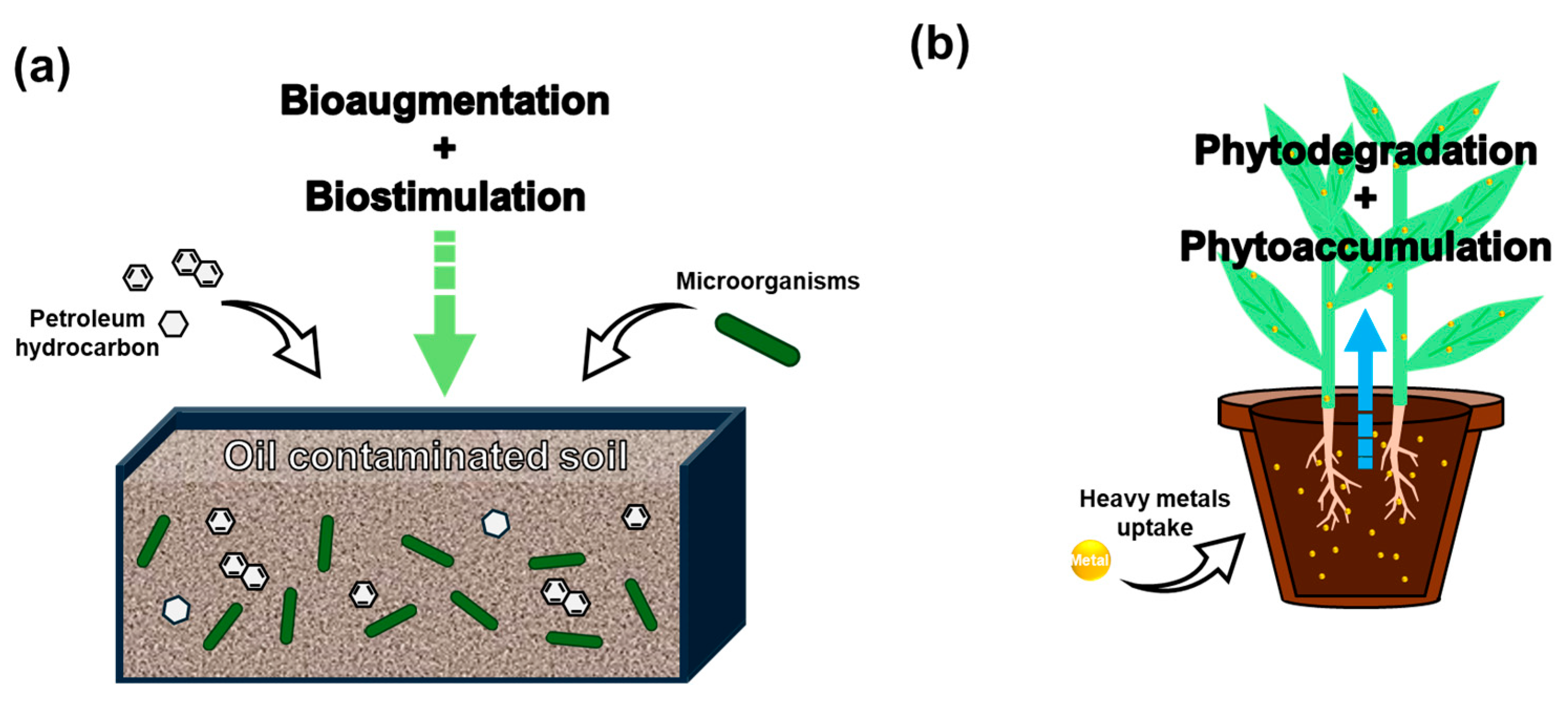
Figure 5.
Schematic illustration of (a) microbial remediation and (b) phytoremediation [111,112].
3.3.1. Microbial Remediation
Microbial remediation involves the use of indigenous or introduced microorganisms to degrade, transform, or immobilize contaminants in polluted soils. This strategy primarily includes biostimulation (enhancing native microbial activity through nutrient or surfactant addition) and bioaugmentation (introducing specific strains with high degradation capacity) [30]. As shown in Table 5, this approach has been widely used in hydrocarbon-contaminated sites. For example, Sánchez Mata et al. [113] applied a mixture of enzymes, surfactants, and vermicompost to stimulate microbial activity in automotive oil-contaminated soils, achieving a total petroleum hydrocarbon (TPH) reduction of over 70% within 28 days. Similarly, Wu et al. [114] showed that the addition of nutrients significantly accelerated hydrocarbon degradation and improved microbial diversity in oil-impacted soils. Bioaugmentation techniques have also shown high potential, especially when combined with biostimulation. In a comparative study, Yaman [111] found that combined bioaugmentation-biostimulation outperformed natural attenuation by achieving over 85% crude oil removal efficiency in 40 days. Haleyur et al. [115] revealed that bioaugmentation with PAH-degrading bacteria significantly enhanced degradation rates in aged soils and led to a shift in the microbial community structure toward hydrocarbon degraders.
Recent developments also highlight hybrid microbial systems, integrating microbial remediation with biochar or electrokinetics to overcome challenges like low pollutant bioavailability. For instance, Wang et al. [116] used Stenotrophomonas acidaminiphila supported on agricultural biochar to remediate Cr(VI)-contaminated soil, achieving over 90% reduction in Cr(VI) via coupled bioreduction and adsorption. Furthermore, Barba et al. [117] compared biostimulation and bioaugmentation under electrokinetic fields and found the hybrid system yielded faster degradation of 2,4-D herbicides and improved microbial viability around electrodes. These studies demonstrate that microbial remediation, particularly when combined with supportive amendments or physical assistance (e.g., biochar, electric fields), can offer effective, environmentally friendly solutions for legacy pollution in shipyard soils and similar industrial environments.
3.3.2. Phytoremediation
Phytoremediation refers to the use of plants and their associated rhizosphere microorganisms to remove, stabilize, or transform contaminants in soil and water [118]. It is particularly attractive for long-term and sustainable remediation at marginally contaminated or shallowly polluted sites due to its low cost, minimal invasiveness, and ecological co-benefits [119]. As illustrated in Figure 5 and summarized in Table 5, recent research emphasizes a range of mechanisms, including phytoextraction, phytostabilization, and rhizodegradation, often enhanced through soil amendments or microbial inoculation. Wiafe et al. [120] applied both organic and inorganic amendments to enhance phytoavailability and recovery of heavy metals, achieving over 50% reduction in metal mobility. Hou et al. [121] demonstrated the synergistic effect of plant-microbe systems in degrading PAHs in petroleum-contaminated soils, with degradation rates exceeding 60%. Similarly, Chen et al. [122] conducted a meta-analysis showing that biochar amendments significantly increased plant uptake efficiency of metals such as Cd, Pb, and Zn by over 30% in many cases.
To improve chelation-assisted uptake, Li et al. [123] used GLDA, a biodegradable chelating agent, with Tagetes patula, reporting high cadmium removal without phytotoxic effects. Chigbo and Batty [124] reported over 70% Cu and pyrene removal using Zea mays with EDDS, confirming the viability of dual-contaminant phytoremediation. Direct phytoextraction was also explored by Shehata et al. [125], showing meaningful reductions in Pb, Cd, and Ni levels in soil.
More advanced strategies involve microbial facilitation. Liu et al. [126] demonstrated enhanced uptake of heavy metals via co-cropping techniques using plant growth-promoting rhizobacteria. Integrated approaches were validated by Lai et al. [127], who achieved over 65% reduction in TPH and heavy metals by combining phytoremediation with microbial inoculants. Moreover, crop rotation strategies were evaluated by Yang et al. [112], revealing significant variation in phytoextraction efficiency based on annual fluxes of Cd. Finally, Bhat et al. (2022) [128] provided a comprehensive review highlighting the scalability, eco-compatibility, and future potential of phytoremediation as a green technology.

Table 5.
Previous studies on biological remediation.
Table 5.
Previous studies on biological remediation.
| Method | Soil Type | Contaminants | Experimental Conditions | Efficiency | Refs. |
|---|---|---|---|---|---|
| Bioremediation | Sandy loam | Automotive residual oil | pH 4.5; 37 °C; enzyme addition + 3–5% vermicompost; 49 days | 99.9% removal efficiency | [113] |
| Clay | Petroleum Hydrocarbons | Temperature-controlled environment with nutrient amendments | 88% | [129] | |
| Contaminated soils | PAHs (Polycyclic Aromatic Hydrocarbons) | Inoculation with PAH-degrading microbes | 99% | [115] | |
| Clay | 2,4-dichlorophe noxyacetic acid | Temperature-controlled environment with applied electric field 1.0 V cm−1 | 75~85% | [117] | |
| Contaminated soils | TPH | open and closed microcosms systems, Humidity-controlled environment with nutrient amendments | 84.1% and 78.7% | [130] | |
| Crude Oil-Contaminated Soils | TPH | microbial inoculum and nutrient addition | 35~66% | [111] | |
| Contaminated Soils | Cr | Humidity-controlled environment with microbial inoculum | 74.94% | [116] | |
| Petroleum-contaminated soil | TPH | Room temperature environment with nutrient addition | 60% and 34% | [114] | |
| Petroleum-contaminated soil | TPH | room temperature of 20–25 °C (autumn) and 10 °C(winter). | 59.89% and 51.38% | [131] | |
| Phytoremediation | Agricultural soil | As, Hg, Cd, and Pb | Field experiment using T. capensis and H. psittacorum with foliar application of fulvic acid, auxin, compost, coconut coir, and sulfur | Enhanced uptake and translocation of heavy metals due to amendments altering soil pH and microbial activity | [120] |
| Petroleum-contaminated soil | Polycyclic aromatic hydrocarbons (PAHs) | Use of microorganism-assisted phytoremediation in heavily polluted soils | Significant degradation of PAHs observed, indicating enhanced remediation efficiency | [121] | |
| Contaminated soil | Cd, Pb, Cu, and Zn | Application of biochar derived from various sources to contaminated soils | Reduction in bioavailable Cd by 52%, Pb by 46%, Cu by 29%, and Zn by 36% | [122] | |
| Agricultural soil | Cd | Application of tetrasodium glutamate diacetate (GLDA) with Tagetes patula L. | Removal of 12.9% of Cd from contaminated agricultural land | [123] | |
| Co-contaminated soil | Heavy metals and PAHs | Application of EDTA and citric acid to enhance phytoextraction | Enhanced plant growth by 41% and increased phytoextraction of Cu; pyrene dissipation improved by up to 90.4% | [124] | |
| Contaminated soil | Cr, Co, Cd, and Mn | Use of kenaf and flax plants with sulfur and gibberellin treatments over two growing seasons | Double-season phytoremediation efficiencies: Cr (66.87%), Mn (65.63%), Co (54.66%), Cd (23.40%) | [125] | |
| Contaminated soil | Heavy metals | Co-cropping of Sedum alfredii with alfalfa, associated with plant growth-promoting rhizobacteria (PGPR) | Enhanced phytoextraction of heavy metals due to synergistic plant-microbe interactions | [126] | |
| Contaminated soil | TPH and heavy metals | Use of Populus bonatii and Crotalaria juncea with earthworms and petroleum-degrading bacteria over 20 months | Higher TPH removal efficacy at 30–60 cm soil depth; more Zn than Cr absorbed and accumulated by plants | [127] | |
| Heavy metal-contaminated soil | Cd | Evaluation of phytoremediation potential based on the balance between annual input and output fluxes of Cd in farmland soil | Demonstrated potential for Cd removal through plant uptake | [112] | |
| Contaminated soil | Heavy metals | Use of hyperaccumulator plant species in various soil types | Effective removal of heavy metals from polluted areas | [128] |
4. Practical Examples of Remediation and Redevelopment in Shipyard Environments
Table 6 summarizes representative examples of shipyard remediation and redevelopment projects from various regions. Although multiple advanced technologies such as soil washing, in situ chemical oxidation, and bioremediation have been introduced in the literature, the actual remediation practices at shipyards have largely relied on conventional physical approaches such as excavation and dredging. This trend can be attributed to multiple practical factors, including the urgency of site reuse, economic constraints, and the complex heterogeneity of contamination at such industrial sites. The following cases highlight specific contamination profiles, applied remediation methods, their performance, and the scale of intervention. At the Hunters Point Naval Shipyard in San Francisco (~3.5 million m2), the site was impacted by petroleum hydrocarbons, VOCs (e.g., benzene, TCE), PCBs (<0.74 mg/kg), heavy metals (e.g., Pb up to 287 mg/kg), and radionuclides (e.g., Cs-137, Sr-90) [132,133,134]. The remediation involved the excavation of approximately 1 million tons of contaminated soil, the installation of landfill capping systems, vapor extraction, and the removal of radiologically contaminated structures. These interventions achieved more than 95% removal of PCB and TPH concentrations, with radionuclide levels reduced to near-background conditions. Advanced in situ chemical or biological treatments were not actively utilized despite the complexity of contamination. While the site presented a complex mixture of pollutants, advanced in situ chemical or biological treatments were not predominantly used.

Table 6.
Projects of shipyard remediation and redevelopment.
Similarly, At the Mare Island Naval Shipyard (~22.7 million m2), the remediation strategy was similarly excavation-focused, involving the removal of over 300 underground storage tanks (USTs), excavation of hotspots contaminated with PCBs (11–14 mg/kg), lead (up to 8300 mg/kg), zinc (11,000 mg/kg), TPH (82 mg/L), and various explosives [134,135,136]. A pilot-scale thermal desorption test was conducted, but large-scale implementation was not pursued. The project achieved over 90% reduction in key contaminants and enabled the site to be certified for commercial reuse, with regulatory monitoring scheduled until 2050.
The Philadelphia Navy Yard (~5.9 million m2) was remediated through a combination of asbestos abatement, soil excavation targeting PCB (>50 mg/kg), lead (>2000 mg/kg), petroleum hydrocarbons, and UST removal [137,138]. Groundwater contaminated with VOCs was treated through pump-and-treat systems. Post-remediation assessments indicated PCB levels below 1 mg/kg, and asbestos was fully removed, enabling large-scale redevelopment that currently hosts over 150 businesses [137].
At the Charlestown Navy Yard in Boston (~526,000 m2), the site contained PCBs (~12 mg/kg), PAHs (benzo[a]pyrene up to 21 mg/kg), dioxins (~0.046 mg/kg), and lead (~7 mg/kg). Remediation activities included excavation of approximately 917 m3 of contaminated soil and sediment, PCB equipment removal, and capping of residual contamination zones [139,140,141]. The site has since been redeveloped as a National Historical Park [139].
In Varvsstaden (Kockums Shipyard), Malmö (~180,000 m2), remediation targeted petroleum hydrocarbons, chlorinated solvents (e.g., TCE, PCE), and heavy metals. The excavation was conducted in grid-based units (~400 m2), with high-contamination soils (~20,000 m3) removed for off-site disposal. Soil washing was used for partial reuse of lower-risk materials, and the site now meets Swedish residential soil standards [142,143].
The Oslo Harbor Cleanup (Norway) primarily addressed sediment contamination with heavy metals (e.g., Hg ~0.8 mg/kg, Pb ~94 mg/kg), PAH16 (~3.6 mg/kg), PCBs (~28 µg/kg), and TBT (~112 µg/kg). Approximately 700,000 m3 of sediments were dredged, confined in a designated CAD system, and capped with 0.5 m of clay. TBT levels were reduced to 2.4 µg/kg, and heavy metals were decreased to near-background levels, allowing the reactivation of fishing and harbor activities [144,145,146,147].
In Howards Bay (Fraser Shipyard), Wisconsin, roughly 64,742 m3 of contaminated sediments were dredged and stabilized using cement-based solidification. These materials were then disposed of in licensed landfills and capped with clean sand and topsoil. TBT was reduced by over 99%, and portions of the treated sediment were reused for landscaping and green infrastructure development [148,149,150,151].
Finally, the Tongyeong (Shina Shipyard) project in South Korea (~180,000 m2) is one of the few examples that actively adopted advanced soil treatment methods. Heavy metals (e.g., Cu, Cd, As, Pb, Zn), TPH, and TBT were detected at concentrations exceeding Korea’s Region 1 residential standards, reaching depths of up to 7 m. A combination of deep grid excavation and high-pressure soil washing was applied. Recovered clean soils were reused, and a slurry wall was installed for hydraulic containment. The remediation achieved over 99% removal efficiency for heavy metals, making the site suitable for future redevelopment into a mixed-use and marina district [152].
Collectively, these case studies demonstrate that physical removal techniques such as excavation and dredging have remained the primary remediation strategies at shipyard sites, not merely because of their operational simplicity, but also because they offered reliable, large-scale contaminant removal under complex and heterogeneous conditions. These methods provided a practical response to urgent redevelopment timelines, regulatory certainty, and site-specific constraints. However, it is also important to situate these choices within their historical and technological context. Most remediation projects reviewed here were implemented between the early 1990s and 2010s, a period when many advanced in situ technologies, such as in situ chemical oxidation, electrokinetic remediation, or large-scale bioremediation, were either still in the pilot phase or lacked the regulatory frameworks necessary for full-scale deployment. At that time, their performance under field conditions remained uncertain, and their operational risks were perceived to be high. In that sense, the dominance of conventional physical methods does not necessarily reflect a disregard for innovation, but rather a rational response to the technological readiness levels, economic constraints, and risk-averse institutional decision-making frameworks of the time. As remediation technologies continue to evolve, addressing issues of cost, efficiency, and regulatory compatibility, there is strong potential for a more integrated and adaptive use of advanced techniques in future shipyard redevelopment projects.
5. Evaluation of Soil Remediation Technologies for Shipyards
The previous section emphasized that remediation projects at shipyard sites have predominantly relied on conventional physical methods, such as excavation and dredging. This tendency stems from site-specific constraints and technological limitations, as well as the operational simplicity and predictability of these approaches. Only a few soil remediation techniques discussed in this review, such as soil washing and soil vapor extraction, have occasionally been applied. However, the increasing complexity of contamination, rising demand for sustainable remediation strategies, stricter environmental regulations, and growing expectations for post-remediation land reuse suggest that broader and more flexible remediation approaches will be required in the future. Accordingly, this section examines the technical advantages and limitations, economic and environmental considerations, and practical applicability of selected alternative remediation methods in shipyard settings. Based on this multifaceted analysis, the study provides a comprehensive evaluation of the potential integration of these technologies into future shipyard soil remediation strategies.
5.1. Technological Advantages and Limitations
A variety of remediation technologies have been developed and applied to address the diverse nature of soil contamination. In shipyard settings, where contamination typically includes heavy metals, petroleum hydrocarbons, and POPs, the selection of an appropriate remediation strategy requires careful consideration of each technology’s technical performance, applicability to site conditions, and potential constraints.
Table 7 summarizes the advantages and challenges of the selected soil remediation technologies [30]. Soil washing separates contaminants from soil particles using water or chemical solutions, often aided by physical agitation. It is effective for soils contaminated with heavy metals and hydrophobic organic pollutants. However, the process generates large volumes of wastewater and is generally more suitable for coarse-grained soils, as fine particles reduce separation efficiency. According to the U.S. EPA [153], the soil washing process typically consumes between approximately 640 L/m3 and 3960 L/m3 of water, depending on the specific process configuration and soil characteristics.

Table 7.
Benefits and challenges of soil remediation technologies [30].
Soil flushing involves injecting chemical solutions into the subsurface to mobilize and extract contaminants in situ. It minimizes surface disruption and is technically applicable to sensitive areas. Its performance, however, is highly dependent on soil permeability and hydraulic control. In poorly permeable soils, flushing efficiency declines, and there is a risk of uncontrolled migration of mobilized contaminants; in a full-scale field application at a Korean military site, modified in situ soil flushing combined with air sparging achieved TPH removal rates of about 53–62% over depths of 6–9 m and increased the soil’s hydraulic conductivity nearly fivefold, demonstrating that flushing efficiency is highly dependent on soil permeability, with lower-permeability layers showing reduced treatment effectiveness [154].
Thermal desorption heats soil to volatilize and remove organic contaminants. It is particularly effective for persistent pollutants such as PAHs, PCBs, and TBT. Technically, it provides high removal rates, but it requires significant energy input and uniform heating, making it less suitable for large or deeply contaminated areas. Previously reported field data indicate thermal desorption systems typically require substantial energy inputs, with electrical resistance heating consuming approximately 200–400 kWh/m3, and gas-fired thermal conduction systems using between 620 and 890 kWh/m3, depending on site-specific conditions and heating configuration [155].
Adsorption-enhanced remediation utilizes materials such as activated carbon or biochar to immobilize pollutants within the soil matrix. This method offers versatility for both organic and inorganic contaminants. Nevertheless, its success depends on the correct selection and dosage of the adsorbent material, and it may produce secondary solid-phase waste that requires further handling.
Soil vapor extraction removes volatile organic compounds from unsaturated zones by applying a vacuum to draw contaminated vapors out of the soil. It is effective in permeable, dry soils and can be combined with other technologies. Limitations include the need for off-gas treatment systems and the inapplicability of saturated or fine-grained soils. Off-gas treatment systems, which are essential to managing extracted vapor and preventing environmental release, can account for up to 50% of total soil vapor extraction remediation project costs [156].
Chemical oxidation introduces oxidants such as hydrogen peroxide, permanganate, or persulfate to break down organic contaminants in place. It provides rapid degradation and can be applied across heterogeneous soils. However, incomplete reactions, secondary by-product formation, and challenges in oxidant distribution reduce its reliability in complex field conditions [157]. For example, field-scale in situ chemical oxidation treatment has limitations in fully mineralizing petroleum hydrocarbons into CO2 and H2O, often resulting in the formation of various low-molecular-weight hydrophilic intermediates. In particular, BTEX and MTBE generate complex mixtures of aromatic and aliphatic intermediates, and the complete removal of these by-products is essential to prevent secondary contamination [158,159].
Chemical reduction targets redox-sensitive contaminants like chlorinated solvents and certain metals by chemically reducing them to less toxic or immobilized forms. It offers long-term stabilization potential but requires precise control of redox conditions and effective reagent delivery, particularly in heterogeneous subsurface environments. Chemical reduction efficiency is limited in low-permeability soils such as clays and tills, primarily due to the restricted injection rate of treatment solutions [160].
Electrokinetic remediation uses a low-voltage electric current to mobilize charged contaminants across electrodes embedded in the soil. It is one of the few technologies suitable for fine-grained, low-permeability soils, allowing for the removal of both organics and metals. However, it is energy-intensive and demands strict control of electrolyte composition and system stability over extended durations. Studies on periodic-power electrokinetic remediation have demonstrated energy consumption ranging from 45 to 3669 kWh/m3, depending on soil chemistry and applied power regimes, underlining the need for careful energy optimization in field applications [161].
Stabilization/solidification involves mixing binding agents with contaminated soil to immobilize pollutants and improve structural integrity. It is technically effective for preventing contaminant leaching. However, it increases the volume of treated material and poses long-term monitoring challenges due to possible matrix degradation [162].
Microbial bioremediation relies on microbial activity to degrade biodegradable organic contaminants, especially petroleum hydrocarbons. It is applicable in situ with minimal disturbance, but it is slow, sensitive to environmental factors (e.g., temperature, pH), and ineffective for recalcitrant compounds [163].
Phytoremediation employs plants to extract, degrade, or stabilize contaminants in shallow soils. It is technically feasible over large surface areas but limited by the depth of root systems, the slow pace of remediation, and seasonal dependencies. Phytoremediation is generally limited to the effective rooting depth of plants, which typically ranges from 0.3 m for grasses to 5–6 m for woody species like poplars under field conditions. Depending on contaminant type and site characteristics, remediation may require multiple growing seasons, with typical treatment durations spanning 2–5 years for hydrocarbons and significantly longer for heavy metals [164].
In summary, while each technology offers distinct advantages under specific conditions, their field-scale applicability in shipyard environments depends heavily on contaminant characteristics, soil properties, and operational constraints. As shipyard sites increasingly face complex and diverse contamination profiles, a nuanced understanding of these technologies’ technical trade-offs is essential to inform appropriate remediation design.
5.2. Economic and Life Cycle Assessment
While the technical efficacy of remediation technologies is a primary consideration in site selection and engineering design, their full-scale implementation depends equally on cost efficiency and environmental sustainability. This is particularly critical for shipyard sites, where large-scale remediation must address diverse contaminants, including heavy metals, petroleum hydrocarbons, PAHs, and persistent organic pollutants, under complex spatial and regulatory constraints. Cost analysis and life cycle assessment (LCA) approaches have quantified these differences, providing an evidence-based foundation for remediation planning [165,166,167,168].
Among the soil remediation methods, soil washing is effective for removing heavy metals and PAHs but entails substantial secondary burdens from sludge and wash water treatment. It also requires high energy input. Typical treatment costs range from approximately $80 to $200 per ton, and its CO2 footprint is lower than thermal and desorption or off-site landfilling [169,170]. Soil flushing, which injects surfactants or chelating agents into the subsurface, has a similar cost profile—generally $75–$210 per ton—though its field applicability may vary based on soil permeability and contaminant distribution [169].
In contrast, thermal desorption allows rapid remediation and is highly effective for VOCs and other volatile contaminants. However, this method demands substantial capital investment and energy input, leading to the highest global warming potential (GWP) among the reviewed technologies—typically 300–400 kg CO2-eq per ton. Costs can also reach up to $300 per ton [171,172]. While some LCA models suggest long-term environmental benefits from thorough source removal and reduced plume migration, these gains may only offset initial impacts over extended timeframes [165].
Adsorption-enhanced remediation represents a more flexible and low-cost alternative. However, its environmental footprint is highly dependent on the production route of the adsorbents. Cradle-to-gate LCA studies show that coal-based GAC produces the highest emissions (5.2–5.5 kg CO2-eq/kg), while biomass-derived GACs—from coconut or wood—have significantly lower GWPs (0.9–1.1 kg CO2-eq/kg). Reactivated GAC, with a GWP of 2.1 kg CO2-eq/kg, also offers a promising, more sustainable option [173].
Soil vapor extraction is simple to operate and highly effective for VOC removal. While its overall cost is moderate, the treatment of off-gases can incur significant additional expenses [174]. Typical operations range around $20–$220 per ton of soil for extraction and treatment of off-gases [174,175]. In contrast, despite its rapid action, chemical oxidation performs poorly in most LCA categories due to the use of strong reagents like Fenton’s reagent or persulfate, which contribute significantly to ecotoxicity and human toxicity [176]. Chemical reduction using zero-valent iron (ZVI) exhibits intermediate GWP, typically ranging from 70 to 180 kg CO2-eq per ton of soil treated, depending on synthesis methods [176]. Among production methods, sodium borohydride-based synthesis shows the lowest environmental burden, whereas hydrogen gas reduction incurs higher impacts due to energy intensity. Key contributors include electricity usage, NaBH4 manufacturing, and reagent emissions. Cleaner energy sources and optimized production routes can mitigate these effects.
Electrokinetic remediation, especially when combined with permeable reactive barriers, demonstrates the potential for both organic and inorganic contaminants. Field-scale costs are approximately $200 per m3 [177]. In terms of carbon footprint, electrokinetic remediation shows 0.36–0.49 kg CO2-eq/kg soil, with energy consumption contributing 88–93% of the overall impact. Atrazine-contaminated soils required the highest energy, while Chlorsulfuron needed the least. Renewable energy integration (e.g., solar) could cut water footprint by up to 90% [178].
Among biological approaches, a recent project in Southeast Asia reported $310/m3 (~$174 material costs) for combined bioaugmentation–biostimulation [179]. A recent LCA-based study on petroleum-contaminated saline soils demonstrated that bioremediation using inorganic nutrients (e.g., urea) can result in significant environmental burdens, including 84.2 kg CO2-eq per ton and high impacts on human health and ecosystem quality [180]. In contrast, organic waste-based bioremediation (e.g., molasses with bacterial inoculants) reduced the overall environmental footprint by up to 90%. These findings highlight that nutrient selection critically affects the sustainability of bioremediation processes. Integrating waste valorization within bioremediation offers both ecological and circular economy benefits.
Lastly, phytoremediation is recognized as a sustainable option for heavy metal-contaminated soils, with LCA studies showing 30–100% lower environmental impacts compared to excavation or soil washing [181]. However, long treatment durations (18 months to 16 years) and biomass disposal remain key challenges. TEA indicates that soil preparation and biomass management drive most of the costs, while chelator-assisted phytoextraction can boost metal uptake by up to 200-fold, enhancing efficiency. Integrating LCA and techno-economic analysis supports informed, site-specific decision-making for sustainable remediation.
5.3. Integrated Assessment and Site-Specific Considerations
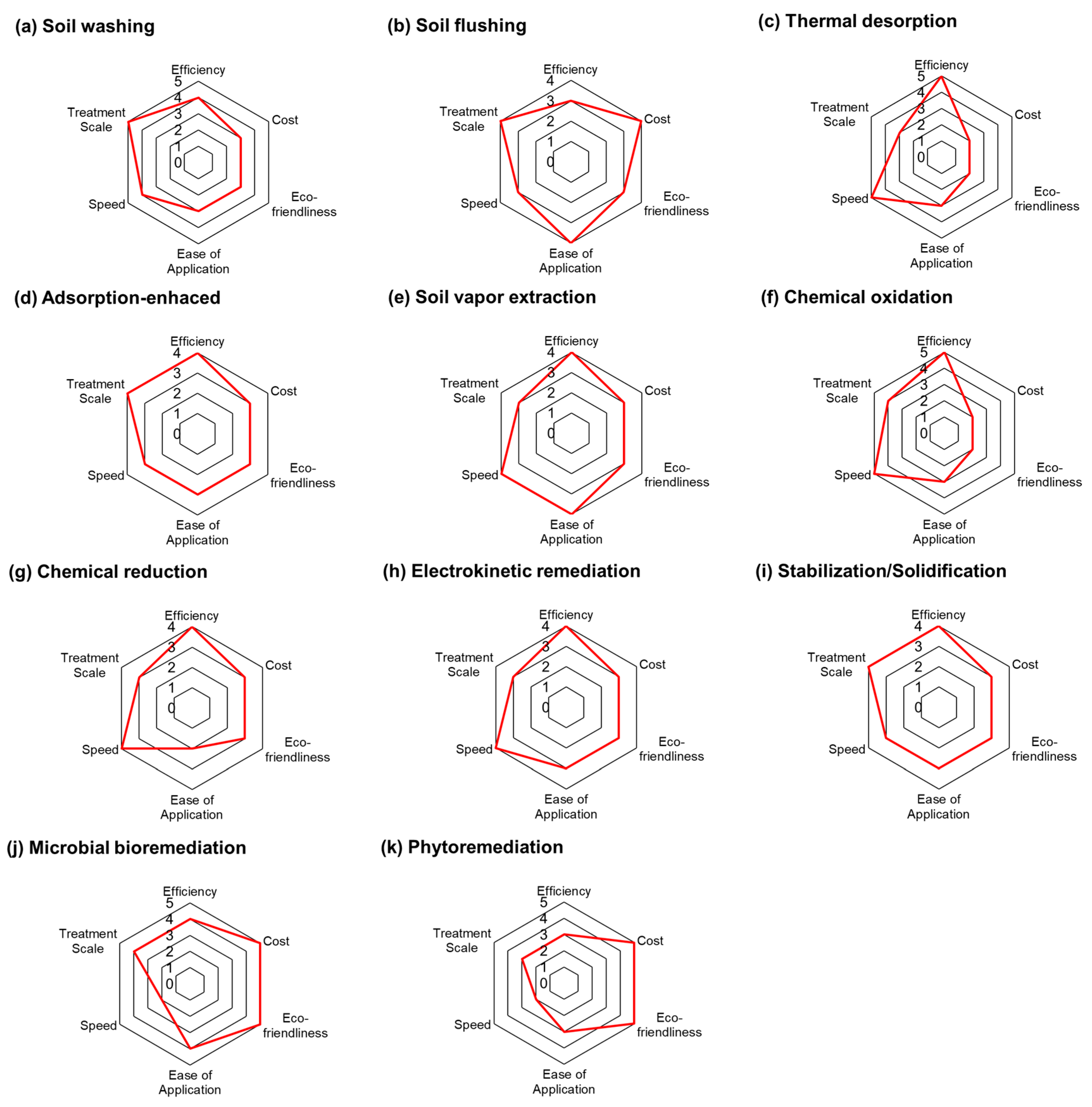
The previous two sections discussed the strengths and limitations of soil remediation technologies, with a particular focus on their economic and environmental aspects. Based on these considerations and previous studies, this section identifies six key evaluation criteria and conducts an integrated assessment of each technology [30]. These criteria—efficiency, cost, eco-friendliness, ease of application, speed, and treatment scale—provide a comprehensive framework for assessing practical performance.
Figure 6 summarizes how each representative technology performs across these dimensions. Thermal desorption and chemical oxidation show strong performance in terms of efficiency and speed, making them highly effective for the rapid removal of persistent contaminants. However, their high energy requirements and associated emissions lead to lower scores in cost and environmental sustainability. These characteristics make them suitable for hotspot or time-sensitive remediation but less viable for long-term, large-scale applications.
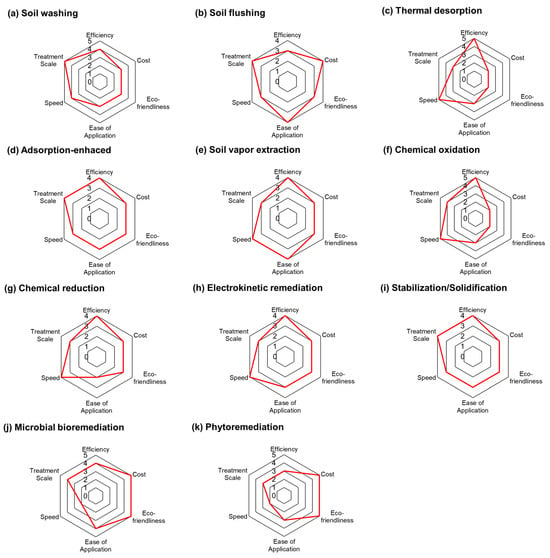
Figure 6.
Performance evaluation of representative soil remediation technologies (rated from 1 = poor to 5 = excellent) [28].
In contrast, phytoremediation and microbial bioremediation stand out for their environmental and economic advantages. They are associated with the lowest GWP emissions and offer additional benefits such as landscape restoration and potential biomass utilization. That said, their slow treatment rates and limited applicability to deep or complex contamination reduce their potential as standalone solutions. Instead, they are better positioned as follow-up or polishing stages within a multi-step remediation process.
Soil washing demonstrates relatively balanced performance across all criteria, particularly excelling in efficiency and treatment capacity. Its flexibility and compatibility with diverse contamination profiles highlight its value in shipyard contexts, especially when integrated into phased or combined strategies. Stabilization and solidification also offer consistent results across various pollutant types, though their long-term effectiveness may be influenced by environmental changes and material durability.
Electrokinetic remediation and soil flushing show variable performance depending heavily on soil characteristics, contaminant type, and energy availability. While they can be highly effective under specific conditions, especially in fine-grained or low-permeability soils, their broader applicability remains limited. Similarly, technologies like adsorption-enhanced remediation and soil vapor extraction offer clear advantages for targeted use (e.g., VOC removal or shallow zone treatment) but fall short in scalability and speed.
Overall, the comparative results reinforce the idea that no single technology satisfies all remediation goals. Optimal outcomes will depend on selecting and combining technologies based on the dominant site constraints, contamination profiles, and long-term land use plans. Hybrid approaches, where high-impact, front-end methods such as thermal treatment or washing are followed by sustainable polishing techniques like phytoremediation or solidification, can offer more balanced solutions. To translate this understanding into practical strategies, it is essential to apply structured decision-making frameworks that integrate environmental, technical, and socio-economic dimensions.
5.4. Toward Practical Applicability of Selected Soil Remediation Technologies for Shipyard Remediation
Table 8 highlights differences in the range of pollutants that each technology can effectively target. This table displays the applicability of each technology to major contaminants commonly found in shipyard environments. Soil washing shows the broadest spectrum of applicability, capable of addressing PAHs, PCBs, heavy metals, and organotin compounds. Its ability to physically separate contaminants from the soil matrix allows it to respond flexibly to diverse contamination profiles, making it a highly promising primary remediation technology for complex shipyard sites. Thermal desorption similarly demonstrated high applicability across almost all major contaminants, particularly excelling in treating persistent pollutants such as high concentrations of PAHs, PCBs, and organotin compounds. However, as discussed earlier, its high energy demands, and operational costs must be considered when applying it at large scales. Chemical oxidation proved effective mainly for organic pollutants such as PAHs, PCBs, TBT, and other organotin compounds, indicating its potential for hotspot remediation in shipyard sites where organic contamination predominates. Chemical reduction exhibited strong treatment capabilities for heavy metals and some organotin compounds, although its overall applicable contaminant range was more limited. Given its selective reaction characteristics, careful site-specific design is required for its effective application.

Table 8.
Summary of pollutant types remediated by each technology (○ = applicable, △ = partially applicable, × = not applicable).
Phytoremediation and bioremediation showed applicability primarily to petroleum hydrocarbons and low concentrations of PAHs but were less effective against PCBs and heavy metals. Therefore, these technologies are more suitable as complementary strategies to stabilize residual contamination or support long-term ecological restoration rather than serving as primary remediation methods for shipyard sites. However, recent LCA-based assessments suggest that phytoremediation, despite its limitations, offers significant advantages in terms of low greenhouse gas emissions, land aesthetics, and cost-effectiveness when coupled with biomass valorization strategies such as anaerobic digestion or energy recovery. In scenarios involving moderate contamination and long-term site redevelopment goals, phytoremediation may serve as a viable secondary or finishing-stage option.
Soil flushing and electrokinetic remediation were primarily applicable to heavy metals (e.g., Pb, Cd, Zn, Cu) and are particularly promising for fine-grained soils or low-permeability formations. However, their applicability to organic contaminants remains limited. Furthermore, electrokinetic remediation’s high electricity consumption and potential for secondary waste streams necessitate consideration of energy sources and long-term environmental trade-offs. Stabilization and solidification techniques show consistent performance in immobilizing a broad range of pollutants but raise concerns regarding future leachability and monitoring requirements under changing environmental conditions, such as those driven by climate change.
Site heterogeneity presents a significant obstacle to the universal deployment of any single technology. The co-occurrence of diverse contaminants at varying depths and concentrations often requires a hierarchical or hybrid strategy. For example, the initial removal of high-concentration contaminants through soil washing or thermal desorption could be followed by bioremediation or phytoremediation to address residual contamination in a sustainable manner. Importantly, site-specific geostatistical characterization and modeling of contaminant mobility should guide remediation design to prevent cross-media transfer and uncontrolled migration, especially in coastal sites vulnerable to sea-level rise or groundwater intrusion.
The practicality of advanced remediation techniques also varies significantly depending on regional socio-economic contexts. In developing countries, barriers such as limited financial resources, weak regulatory enforcement, and lack of technical capacity can hinder adoption. In such cases, cost-effective and ecologically adaptive approaches such as phytoremediation or low-tech bioremediation, supported by local biomass reuse strategies, may offer more feasible options.
Future shipyard site restoration projects must carefully balance economic feasibility, environmental sustainability, and long-term resilience by tailoring remediation strategies to specific site conditions and contamination profiles. The integration of LCA and TEA into remediation planning processes allows stakeholders to evaluate trade-offs more systematically and select combinations of technologies that align with both short-term cleanup goals and long-term land use visions.
6. Conclusions
In this review, soil contamination issues at shipyard sites, applicable soil remediation technologies, practical cases of soil and sediment remediation, and the advantages and limitations of various remediation approaches were comprehensively discussed. Particular attention was given to evaluating future directions for the development and application of soil remediation technologies in shipyard environments. To date, compared to remediation efforts for other contaminated sites, documented remediation cases specifically targeting shipyard soils have been relatively limited. Moreover, even in the available examples, the remediation methods employed were predominantly simple physical techniques such as excavation and dredging, rather than the use of advanced or integrated treatment technologies. Given the unique characteristics of shipyard environments, such as the co-occurrence of petroleum hydrocarbons, heavy metals, PCBs, and organotin compounds, as well as site-specific challenges like coastal proximity and subsurface heterogeneity, future remediation strategies must evolve beyond single-process approaches. A more effective restoration of shipyard soils will require the integration of multiple remediation technologies, carefully selected and combined based on site-specific contamination profiles, environmental conditions, and project goals. Specifically, a hierarchical or hybrid treatment framework should be pursued, wherein high-concentration contaminants are initially removed using high-efficiency methods such as soil washing or thermal desorption, followed by secondary or complementary treatments such as bioremediation or phytoremediation for residual contaminants. Additionally, careful attention must be paid to secondary environmental issues, particularly the generation and management of contaminated wastewater produced during soil remediation processes. Efficient handling, treatment, and disposal of remediation by-products are essential to ensure that secondary pollution does not compromise the overall effectiveness and environmental integrity of remediation efforts. Finally, considerations of economic feasibility, environmental sustainability, and long-term resilience must be balanced when designing shipyard soil remediation projects. Advancing the application of combined and adaptive remediation technologies will be critical to achieving more efficient, sustainable, and cost-effective restoration of complexly contaminated shipyard sites.
Author Contributions
Conceptualization, Methodology, Data curation, validation, D.K., H.J., C.P., Y.J., and S.S.; Writing—review, and editing, Writing—original draft, Visualization, Funding acquisition, Formal analysis, Data curation, Conceptualization, E.Y.; Writing—original draft, Visualization, Methodology, Data curation, Conceptualization, J.H.J.; Writing—original draft, Visualization, Methodology, Formal analysis, Data curation, Conceptualization, M.A.K. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Research Resurgence under the Glocal University 30 Project at Gyeongsang National University in 2024.
Data Availability Statement
All data are presented in the article.
Conflicts of Interest
Jae Ho Jung was employed by the company IDEA Environment Service Co., Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| BOD | Biochemical Oxygen Demand |
| BTX | Benzene, Toluene, and Xylene |
| COD | Chemical Oxygen Demand |
| Cd | Cadmium |
| Cr | Chromium |
| Cu | Copper |
| DDT | Dichlorodiphenyltrichloroethane |
| EDTA | Ethylenediaminetetraacetic Acid |
| EDX | Energy-Dispersive X-Ray Spectroscopy |
| EK-PRB | Electrokinetic-Permeable Reactive Barrier |
| ERL | Effects Range Low |
| ERM | Effects Range Median |
| FAO | Food And Agriculture Organization |
| GLDA | Tetrasodium Glutamate Diacetate |
| IMO | International Maritime Organization |
| LZSS | Lead-Zinc Smelting Slag LZSS |
| MSWI | Municipal Solid Waste Incinerator |
| NZVI | Nanoscale Zero-Valent Iron |
| OCP | Octacalcium Phosphate |
| OCPs | Organochlorine Pesticides |
| PAHs | Polycyclic Aromatic Hydrocarbons |
| PCBs | Polychlorinated Biphenyls |
| PFAS | Per-And Poly-Fluoroalkyl Substances |
| PFOS | Perfluorooctanesulfonic Acid |
| POPs | Persistent Organic Pollutants |
| Pb | Lead |
| SEM | Scanning Electron Microscopy |
| SVE | Soil Vapor Extraction |
| TBT | Tributyltin |
| TBT | Tributyltin |
| TCAS | Tetrasulfonate |
| TCE | Trichloroethylene |
| TGA | Thermogravimetric Analysis |
| TOC | Total Organic Carbon |
| TPH | Total Petroleum Hydrocarbons |
| USEPA | U.S. Environmental Protection Agency |
| UTS | Universal Treatment Standards |
| VOCs | Volatile Organic Compounds |
| WHO | World Health Organization |
| XRD | X-Ray Crystallography |
| Zn | Zinc |
References
- Kwak, S.-J.; Yoo, S.-H.; Chang, J.-I. The role of the maritime industry in the Korean national economy: An input–output analysis. Mar. Policy 2005, 29, 371–383. [Google Scholar] [CrossRef]
- Chiu, S.; Ho, K.; Chan, S.; So, O.; Lai, K. Characterization of contamination in and toxicities of a shipyard area in Hong Kong. Environ. Pollut. 2006, 142, 512–520. [Google Scholar] [CrossRef] [PubMed]
- Makri, P.; Hermides, D.; Kontakiotis, G.; Zarkogiannis, S.D.; Besiou, E.; Janjuhah, H.T.; Antonarakou, A. Integrated Ecological Assessment of Heavily Polluted Sedimentary Basin within the Broader Industrialized Area of Thriassion Plain (Western Attica, Greece). Water 2022, 14, 382. [Google Scholar] [CrossRef]
- Karakas, S.; Acar, A.Z.; Kirmizi, M. Maritime Sustainability: Navigating Complex Challenges and Ecological Footprints. In Sustainable Development Seen Through the Lenses of Ethnoeconomics and the Circular Economy; Springer: Berlin/Heidelberg, Germany, 2024; pp. 19–47. [Google Scholar]
- Eklund, B.; Eklund, D. Pleasure Boatyard Soils are Often Highly Contaminated. Environ. Manag. 2014, 53, 930–946. [Google Scholar] [CrossRef]
- Paipa-Sanabria, E.; González-Montoya, D.; Coronado-Hernández, J.R. Green Technologies for Environmental Air and Water Impact Reduction in Ships: A Systematic Literature Review. J. Mar. Sci. Eng. 2025, 13, 839. [Google Scholar] [CrossRef]
- Hayman, B.; Dogliani, M.; Kvale, I.; Fet, A.M. Technologies for Reduced Environmental Impact from Ships-Ship Building, Maintenance and Dismantling Aspects; ENSUS-2000: Newcastle upon Tyne, UK, 2000. [Google Scholar]
- Li, P.; Dsikowitzky, L.; Diao, X.; Yang, F.; Li, Q.X.; Schwarzbauer, J. Unusual tin organics, DDX and PAHs as specific pollutants from dockyard work in an industrialized port area in China. Chemosphere 2020, 243, 125284. [Google Scholar] [CrossRef]
- Hong, S.H.; Yim, U.H.; Shim, W.J.; Oh, J.R. Congener-Specific Survey for Polychlorinated Biphenlys in Sediments of Industrialized Bays in Korea: Regional Characteristics and Pollution Sources. Environ. Sci. Technol. 2005, 39, 7380–7388. [Google Scholar] [CrossRef]
- Yılmaz, A.; Karacık, B.; Yakan, S.D.; Henkelmann, B.; Schramm, K.-W.; Okay, O.S. Organic and heavy metal pollution in shipbreaking yards. Ocean Eng. 2016, 123, 452–457. [Google Scholar] [CrossRef]
- Guo, G.; Zhang, C.; Wu, G.; Ding, Q.; Wang, S.; Li, F. Health and ecological risk-based characterization of soil and sediment contamination in shipyard with long-term use of DDT-containing antifouling paint. Sci. Total Environ. 2013, 450–451, 223–229. [Google Scholar] [CrossRef]
- Kim, N.S.; Shim, W.J.; Yim, U.H.; Ha, S.Y.; An, J.G.; Shin, K.H. Three decades of TBT contamination in sediments around a large scale shipyard. J. Hazard. Mater. 2011, 192, 634–642. [Google Scholar] [CrossRef]
- Kotrikla, A. Environmental management aspects for TBT antifouling wastes from the shipyards. J. Environ. Manag. 2009, 90, S77–S85. [Google Scholar] [CrossRef] [PubMed]
- Staniszewska, M.; Radke, B.; Namieśnik, J.; Bolałek, J. Analytical methods and problems related to the determination of organotin compounds in marine sediments. Int. J. Environ. Anal. Chem. 2008, 88, 747–774. [Google Scholar] [CrossRef]
- Choi, J.-S.; Jeon, S.-K. A Geo-statistical Assessment of Heavy Metal Pollution in the Soil Around a Ship Building Yard in Busan, Korea. Korean Soc. Mar. Environ. Saf. 2018, 24, 907–915. [Google Scholar] [CrossRef]
- Islam, M.N.; Ganguli, S.; Saha, N.; Mamun Huda, M.; Hoque, M.A.; Peng, C.; Ng, J.C. Uncovering the impact of mega-scale shipbreaking yards on soil and crop quality in Bangladesh: A spatiotemporal dynamics and associated health risks of metal/loid contamination. J. Hazard. Mater. 2024, 464, 132931. [Google Scholar] [CrossRef]
- Hasan, A.B.; Reza, A.H.M.S.; Kabir, S.; Siddique, M.A.B.; Ahsan, M.A.; Akbor, M.A. Accumulation and distribution of heavy metals in soil and food crops around the ship breaking area in southern Bangladesh and associated health risk assessment. SN Appl. Sci. 2020, 2, 155. [Google Scholar] [CrossRef]
- Alam, I.; Barua, S.; Ishii, K.; Mizutani, S.; Hossain, M.M.; Rahman, I.M.M.; Hasegawa, H. Assessment of health risks associated with potentially toxic element contamination of soil by end-of-life ship dismantling in Bangladesh. Environ. Sci. Pollut. Res. 2019, 26, 24162–24175. [Google Scholar] [CrossRef]
- Ali, M.M.; Islam, M.S.; Islam, A.R.M.T.; Bhuyan, M.S.; Ahmed, A.S.S.; Rahman, M.Z.; Rahman, M.M. Toxic metal pollution and ecological risk assessment in water and sediment at ship breaking sites in the Bay of Bengal Coast, Bangladesh. Mar. Pollut. Bull. 2022, 175, 113274. [Google Scholar] [CrossRef]
- Rahman, M.S.; Rahman, M.; Jolly, Y.N.; Hossain, M.K.; Semme, S.A.; Paray, B.A.; Arai, T.; Yu, J.; Hossain, M.B. Heavy metals in afforested mangrove sediment from the world’s largest delta: Distributional mapping, contamination status, risk assessment and source tracing. Mar. Pollut. Bull. 2024, 203, 116429. [Google Scholar] [CrossRef]
- Chowdhury, N.; Rasid, M.M. Heavy metal contamination of soil and vegetation in ambient locality of ship breaking yards in Chittagong, Bangladesh. J. Environ. Sci. Toxicol. Food Technol. 2016, 10, 20–27. [Google Scholar]
- Rakib, M.R.J.; Rahman, M.A.; Onyena, A.P.; Kumar, R.; Sarker, A.; Hossain, M.B.; Islam, A.R.M.T.; Islam, M.S.; Rahman, M.M.; Jolly, Y.N.; et al. A comprehensive review of heavy metal pollution in the coastal areas of Bangladesh: Abundance, bioaccumulation, health implications, and challenges. Environ. Sci. Pollut. Res. 2022, 29, 67532–67558. [Google Scholar] [CrossRef]
- Luo, Y.; Tong, G.; Song, Q.; Tao, P.; Jin, M.; Gu, N.; Zheng, M.; Yu, X.; Yu, X. Impacts of shipyard oil leakage on the PAHs and PCBs occurrence in Xiangshan Bay, China. Mar. Environ. Res. 2023, 190, 106057. [Google Scholar] [CrossRef] [PubMed]
- Biswas, B.; Sarkar, B.; Mandal, A.; Naidu, R. Heavy metal-immobilizing organoclay facilitates polycyclic aromatic hydrocarbon biodegradation in mixed-contaminated soil. J. Hazard. Mater. 2015, 298, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Gauthier, P.T.; Norwood, W.P.; Prepas, E.E.; Pyle, G.G. Metal–PAH mixtures in the aquatic environment: A review of co-toxic mechanisms leading to more-than-additive outcomes. Aquat. Toxicol. 2014, 154, 253–269. [Google Scholar] [CrossRef] [PubMed]
- Dermont, G.; Bergeron, M.; Mercier, G.; Richer-Laflèche, M. Soil washing for metal removal: A review of physical/chemical technologies and field applications. J. Hazard. Mater. 2008, 152, 1–31. [Google Scholar] [CrossRef]
- Hu, G.; Liu, H.; Chen, C.; Hou, H.; Li, J.; Hewage, K.; Sadiq, R. Low-temperature thermal desorption and secure landfill for oil-based drill cuttings management: Pollution control, human health risk, and probabilistic cost assessment. J. Hazard. Mater. 2021, 410, 124570. [Google Scholar] [CrossRef]
- Ambika, S.; Kumar, M.; Pisharody, L.; Malhotra, M.; Kumar, G.; Sreedharan, V.; Singh, L.; Nidheesh, P.; Bhatnagar, A. Modified biochar as a green adsorbent for removal of hexavalent chromium from various environmental matrices: Mechanisms, methods, and prospects. Chem. Eng. J. 2022, 439, 135716. [Google Scholar] [CrossRef]
- Zhang, S.; Zhao, J.; Zhu, L. Predicting removal efficiency of organic pollutants by soil vapor extraction based on an optimized machine learning method. Sci. Total Environ. 2024, 927, 172438. [Google Scholar] [CrossRef]
- Aparicio, J.D.; Raimondo, E.E.; Saez, J.M.; Costa-Gutierrez, S.B.; Álvarez, A.; Benimeli, C.S.; Polti, M.A. The current approach to soil remediation: A review of physicochemical and biological technologies, and the potential of their strategic combination. J. Environ. Chem. Eng. 2022, 10, 107141. [Google Scholar] [CrossRef]
- Liu, J.; Zhao, L.; Liu, Q.; Li, J.; Qiao, Z.; Sun, P.; Yang, Y. A critical review on soil washing during soil remediation for heavy metals and organic pollutants. Int. J. Environ. Sci. Technol. 2021, 19, 601–624. [Google Scholar] [CrossRef]
- Chiu, J.C.; Gani, P. Soil Washing Methods for Effective Removal of Heavy Metal Contaminants. Ind. Domest. Waste Manag. 2024, 4, 56–71. [Google Scholar] [CrossRef]
- Zhang, M.; Chen, W.; Chuan, X.; Guo, X.; Shen, X.; Zhang, H.; Wu, F.; Hu, J.; Wu, Z.; Wang, X. Remediation of heavily PAHs-contaminated soil with high mineral content from a coking plant using surfactant-enhanced soil washing. Sci. Total Environ. 2024, 909, 168499. [Google Scholar] [CrossRef] [PubMed]
- Grimison, C.; Knight, E.R.; Nguyen, T.M.H.; Nagle, N.; Kabiri, S.; Bräunig, J.; Navarro, D.A.; Kookana, R.S.; Higgins, C.P.; McLaughlin, M.J. The efficacy of soil washing for the remediation of per-and poly-fluoroalkyl substances (PFASs) in the field. J. Hazard. Mater. 2023, 445, 130441. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Z.; Peng, A.; Chu, Z.; Zhang, X.; Huang, H.; Mi, Y.; Xia, D.; Wu, X.; Ye, Z.; Tao, Y. Sustainable remediation of Cr (VI)-contaminated soil by soil washing and subsequent recovery of washing agents using biochar supported nanoscale zero-valent iron. Sci. Total Environ. 2024, 921, 171107. [Google Scholar] [CrossRef] [PubMed]
- Rabieian, M.; Taghavijeloudar, M. Simultaneously removal of PAHs from contaminated soil and effluent by integrating soil washing and advanced oxidation processes in a continuous system: Water saving, optimization and scale up modeling. Water Res. 2024, 256, 121563. [Google Scholar] [CrossRef]
- Huang, J.; Xiao, X.; Chen, B. Insight into the electrochemical process of EDTA-assisted soil washing effluent under alternating current. J. Hazard. Mater. 2024, 470, 134115. [Google Scholar] [CrossRef]
- Xing, L.-Y.; Meng, G.-H.; Yang, J.-X.; Xu, M.-H.; Xu, Y.-M.; Xie, H.-X.; Wang, A.-J.; Sun, Y.-L. PAHs removal by soil washing with thiacalix [4] arene tetrasulfonate. Environ. Sci. Ecotechnol. 2024, 21, 100422. [Google Scholar] [CrossRef]
- Corres, X.; Sierra, C.; Diez-Mestas, A.; Gallego, J.; Baragaño, D. A novel heuristic tool for selecting the best upgrading conditions for the removal of potentially toxic elements by soil washing. J. Hazard. Mater. 2024, 466, 133529. [Google Scholar] [CrossRef]
- Keochanh, D.; Phenrat, T. Magnet-assisted nanoscale zerovalent iron soil washing in chlorpyrifos-contaminated soil remediation: Proof of concept. Process Saf. Environ. Prot. 2024, 191, 723–735. [Google Scholar] [CrossRef]
- Sikdar, S.K.; Grosse, D.; Rogut, I. Membrane technologies for remediating contaminated soils: A critical review. J. Membr. Sci. 1998, 151, 75–85. [Google Scholar] [CrossRef]
- Li, Y.; Wei, M.; Yu, B.; Liu, L.; Xue, Q. Thermal desorption optimization for the remediation of hydrocarbon-contaminated soils by a self-built sustainability evaluation tool. J. Hazard. Mater. 2022, 436, 129156. [Google Scholar] [CrossRef]
- Liu, C.; Shi, H.; Wang, C.; Fei, Y.; Han, Z. Thermal remediation of Soil contaminated with polycyclic aromatic hydrocarbons: Pollutant removal process and influence on Soil Functionality. Toxics 2022, 10, 474. [Google Scholar] [CrossRef] [PubMed]
- Sang, Y.; Yu, W.; He, L.; Wang, Z.; Ma, F.; Jiao, W.; Gu, Q. Sustainable remediation of lube oil-contaminated soil by low temperature indirect thermal desorption: Removal behaviors of contaminants, physicochemical properties change and microbial community recolonization in soils. Environ. Pollut. 2021, 287, 117599. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Cheng, F.; Shao, Z.; Wu, B.; Guo, S. Effects of thermal desorption on ecotoxicological characteristics of heavy petroleum-contaminated soil. Sci. Total Environ. 2023, 857, 159405. [Google Scholar] [CrossRef] [PubMed]
- Bykova, M.V.; Alekseenko, A.V.; Pashkevich, M.A.; Drebenstedt, C. Thermal desorption treatment of petroleum hydrocarbon-contaminated soils of tundra, taiga, and forest steppe landscapes. Environ. Geochem. Health 2021, 43, 2331–2346. [Google Scholar] [CrossRef]
- Ilyas, N.; Shoukat, U.; Saeed, M.; Akhtar, N.; Yasmin, H.; Khan, W.; Iqbal, S. Comparison of plant growth and remediation potential of pyrochar and thermal desorption for crude oil-contaminated soils. Sci. Rep. 2021, 11, 2817. [Google Scholar] [CrossRef]
- Zhang, S.; Zhao, J.; Zhu, L. New insights into thermal desorption remediation of pyrene-contaminated soil based on an optimized numerical model. J. Hazard. Mater. 2024, 461, 132687. [Google Scholar] [CrossRef]
- Xue, Y.; Chen, L.; Xiang, L.; Zhou, Y.; Wang, T. Experimental investigation on electromagnetic induction thermal desorption for remediation of petroleum hydrocarbons contaminated soil. J. Environ. Manag. 2023, 328, 117200. [Google Scholar] [CrossRef]
- Yue, R.; Zhang, X.; Zhong, Y.; Chen, Z.; Zhao, Y.; Wang, D.; Wang, Z.; Mao, X. Thermal reduction-desorption of cadmium from contaminated soil by a biomass co-pyrolysis process. J. Hazard. Mater. 2022, 423, 126937. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, A.; Liu, L.; Duan, X.; Ge, W.; Liu, C.; Qiu, G. Enhanced remediation of cadmium-polluted soil and water using facilely prepared MnO2-coated rice husk biomass. Chem. Eng. J. 2023, 457, 141311. [Google Scholar] [CrossRef]
- Chen, Y.; Fan, J.; Ma, R.; Xue, Y.; Ma, Q.; Chen, X.; Yuan, S.; Zheng, H.; Qiu, Y.; Fu, R. Enhanced Remediation of Pb (Ii), Cd (Ii) and As (V) Contaminated Water and Soil by A-Feooh Incorporated Carboxylated Cellulose Nanocrystals: Synergistic Effect and Immobilization Mechanism. SSRN. Available online: https://papers.ssrn.com/sol3/papers.cfm?abstract_id=4091491 (accessed on 15 May 2025). [CrossRef]
- Lin, H.; Yang, Y.; Shang, Z.; Li, Q.; Niu, X.; Ma, Y.; Liu, A. Study on the enhanced remediation of petroleum-contaminated soil by biochar/g-C3N4 composites. Int. J. Environ. Res. Public Health 2022, 19, 8290. [Google Scholar] [CrossRef]
- Qi, X.; Gou, J.; Chen, X.; Xiao, S.; Ali, I.; Shang, R.; Wang, D.; Wu, Y.; Han, M.; Luo, X. Application of mixed bacteria-loaded biochar to enhance uranium and cadmium immobilization in a co-contaminated soil. J. Hazard. Mater. 2021, 401, 123823. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wen, J.; Xue, Z.; Yin, X.; Yuan, L.; Yang, C. Removal of Cr (VI) by polyaniline embedded polyvinyl alcohol/sodium alginate beads− Extension from water treatment to soil remediation. J. Hazard. Mater. 2022, 426, 127809. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Yin, H.; Zhu, M.; Yu, X.; Shao, P.; Dang, Z. MgO-loaded nitrogen and phosphorus self-doped biochar: High-efficient adsorption of aquatic Cu2+, Cd2+, and Pb2+ and its remediation efficiency on heavy metal contaminated soil. Chemosphere 2022, 294, 133733. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Xue, B.; Jiao, L.; Meng, X.; Zhang, L.; Li, B.; Sun, H. Preparation of ball-milled phosphorus-loaded biochar and its highly effective remediation for Cd-and Pb-contaminated alkaline soil. Sci. Total Environ. 2022, 813, 152648. [Google Scholar] [CrossRef]
- Li, Y.; Shao, M.; Huang, M.; Sang, W.; Zheng, S.; Jiang, N.; Gao, Y. Enhanced remediation of heavy metals contaminated soils with EK-PRB using β-CD/hydrothermal biochar by waste cotton as reactive barrier. Chemosphere 2022, 286, 131470. [Google Scholar] [CrossRef]
- Lyu, Z.; Zhou, W.; Gao, X.; Zheng, H.; Jia, J. Remediation of toluene-contaminated soils by sequential treatment: Soil vapor extraction systems and internal combustion engine units. J. Contam. Hydrol. 2025, 271, 104532. [Google Scholar] [CrossRef]
- Cao, W.; Zhang, L.; Miao, Y.; Qiu, L. Research progress in the enhancement technology of soil vapor extraction of volatile petroleum hydrocarbon pollutants. Environ. Sci. Process. Impacts 2021, 23, 1650–1662. [Google Scholar] [CrossRef]
- Liu, Y.; Huang, F.; Zhou, D.; Wang, T.; Wang, G. Study of factors affecting the remediation of contaminated soil through pneumatic fracturing and soil vapor extraction. J. Hazard. Mater. 2024, 467, 133776. [Google Scholar] [CrossRef]
- Xu, L.; Hu, X.; Zha, F.; Kang, H.; Fang, L.; Kang, B.; Chu, C.; Yang, C. Mass transfer enhancement of air sparging on VOCs contaminated low-permeability soil by establishing pressure gradient. Chemosphere 2023, 313, 137416. [Google Scholar] [CrossRef]
- de Melo Henrique, J.M.; Isidro, J.; Sáez, C.; López-Vizcaíno, R.; Yustres, A.; Navarro, V.; Dos Santos, E.V.; Rodrigo, M.A. Enhancing soil vapor extraction with EKSF for the removal of HCHs. Chemosphere 2022, 296, 134052. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, T.; Zhang, M.; Wang, L.; Kang, G.; Wu, S.; She, Y. Ultrasonically activated persulfate process for the degradation of phenanthrene in soil-washing effluent: Experimental, DFT calculation and toxicity evaluation. J. Environ. Chem. Eng. 2024, 12, 113035. [Google Scholar] [CrossRef]
- Jun, B.-M.; Oh, M.; Jung, J.-Y.; Eun, H.-C.; Yoon, Y. A feasibility study on clearance levels for real U (VI)-contaminated soil and the mechanisms involving metal ions after neutralization of effluents generated from the soil-washing process. Nucl. Eng. Technol. 2025, 57, 103518. [Google Scholar] [CrossRef]
- Alazaiza, M.Y.; Albahnasawi, A.; Ali, G.A.; Bashir, M.J.; Copty, N.K.; Amr, S.S.A.; Abushammala, M.F.; Al Maskari, T. Recent advances of nanoremediation technologies for soil and groundwater remediation: A review. Water 2021, 13, 2186. [Google Scholar] [CrossRef]
- Petrillo, A.; Fraternali, F.; Acampora, A.; Di Chiara, G.; Colangelo, F.; Farina, I. Innovative Solidification and Stabilization Techniques Using Industrial By-Products for Soil Remediation. Appl. Sci. 2025, 15, 4002. [Google Scholar] [CrossRef]
- Hu, W.; Cheng, W.-C.; Wen, S. Investigating the effect of degree of compaction, initial water content, and electric field intensity on electrokinetic remediation of an artificially Cu-and Pb-contaminated loess. Acta Geotech. 2023, 18, 937–949. [Google Scholar] [CrossRef]
- Siegrist, R.L.; Crimi, M.; Thomson, N.R.; Clayton, W.S.; Marley, M.C. In situ chemical oxidation. In Chlorinated Solvent Source Zone Remediation; Springer: Berlin/Heidelberg, Germany, 2014; pp. 253–305. [Google Scholar]
- Nie, J.; Wang, Q.-m.; Han, L.-j.; Li, J.-s. Synergistic remediation strategies for soil contaminated with compound heavy metals and organic pollutants. J. Environ. Chem. Eng. 2024, 12, 113145. [Google Scholar] [CrossRef]
- Zeng, Y.; Li, T.; Ding, Y.; Fang, G.; Wang, X.; Ye, B.; Ge, L.; Gao, J.; Wang, Y.; Zhou, D. Biochar-supported nano-scale zerovalent iron activated persulfate for remediation of aromatic hydrocarbon-contaminated soil: An in-situ pilot-scale study. Biochar 2022, 4, 64. [Google Scholar] [CrossRef]
- Song, I.-G.; Kang, Y.-G.; Kim, J.-H.; Yoon, H.; Um, W.Y.; Chang, Y.-S. Assessment of sulfidated nanoscale zerovalent iron for in-situ remediation of cadmium-contaminated acidic groundwater at a zinc smelter. J. Hazard. Mater. 2023, 441, 129915. [Google Scholar] [CrossRef]
- Wang, L.; Ma, J.; Guo, Q.; Liu, L.; Shou, J.; Sun, A.; Zhao, L. In situ generation of hydrogen peroxide using polymetallic-doped g-C3N4 for pollutant removal. Appl. Sci. 2021, 11, 10797. [Google Scholar] [CrossRef]
- Liu, M.-H.; Hsiao, C.-M.; Lin, C.-E.; Leu, J. Application of combined in situ chemical reduction and enhanced bioremediation to accelerate TCE treatment in groundwater. Appl. Sci. 2021, 11, 8374. [Google Scholar] [CrossRef]
- Reginatto, C.; Cecchin, I.; Heineck, K.S.; Thomé, A.; Reddy, K.R. Use of nanoscale zero-valent iron for remediation of clayey soil contaminated with hexavalent chromium: Batch and column tests. Int. J. Environ. Res. Public Health 2020, 17, 1001. [Google Scholar] [CrossRef] [PubMed]
- Geng, W.; Wei, Y.; Ke, Y.; Qin, J.; Yu, X.; Guo, X.; Long, M. Unveiling molecular transformations of soil organic matter after remediation by chemical oxidation based on ESI-FT-ICR-MS analysis. ACS EST Eng. 2023, 3, 831–840. [Google Scholar] [CrossRef]
- Hong, Y.K.; Kim, J.W.; Lee, D.J.; Kim, H.S.; Park, Y.-H.; Kim, S.C. Effect of In-Situ Oxidation Method on Soil Biological Properties in Total Petroleum Hydrocarbon Contaminated Soil. J. Soil Sci. Fertil. 2023, 56, 90–100. [Google Scholar] [CrossRef]
- Rodrigues, R.; Betelu, S.; Colombano, S.; Tzedakis, T.; Masselot, G.; Ignatiadis, I. In situ chemical reduction of chlorinated organic compounds. Environ. Soil Remediat. Rehabil. Exist. Innov. Solut. 2020, 283–398. [Google Scholar] [CrossRef]
- Mouvet, C.; Collet, B.; Gaude, J.-M.; Rangon, L.; Bristeau, S.; Senergues, M.; Lesueur-Jannoyer, M.; Jestin, A.; Hellal, J.; Woignier, T. Physico-chemical and agronomic results of soil remediation by In Situ Chemical Reduction applied to a chlordecone-contaminated nitisol at plot scale in a French Caribbean banana plantation. Environ. Sci. Pollut. Res. 2020, 27, 41063–41092. [Google Scholar] [CrossRef]
- Cameselle, C.; Reddy, K.R. Electrobioremediation: Combined Electrokinetics and Bioremediation Technology for Contaminated Site Remediation. Indian Geotech. J. 2022, 52, 1205–1225. [Google Scholar] [CrossRef]
- Kim, S.-O.; Jeong, J.Y.; Lee, W.-C.; Yun, S.-T.; Jo, H.Y. Electrokinetic remediation of heavy metal-contaminated soils: Performance comparison between one-and two-dimensional electrode configurations. J. Soils Sediments 2021, 21, 2755–2769. [Google Scholar] [CrossRef]
- Shi, C.; Tong, M.; Cai, Q.; Li, Z.; Li, P.; Lu, Y.; Cao, Z.; Liu, H.; Zhao, H.-P.; Yuan, S. Electrokinetic-enhanced bioremediation of trichloroethylene-contaminated low-permeability soils: Mechanistic insight from spatio-temporal variations of indigenous microbial community and biodehalogenation activity. Environ. Sci. Technol. 2023, 57, 5046–5055. [Google Scholar] [CrossRef]
- Narenkumar, J.; Das, B.; Abilaji, S.; Sathishkumar, K.; AlSalhi, M.S.; Devanesan, S.; Rajasekar, A.; Malik, T. Biosurfactant-assisted bio-electrokinetic enhanced remediation of heavy metal-contaminated soil. Front. Microbiol. 2024, 15, 1458369. [Google Scholar] [CrossRef]
- Sun, Z.; Xu, S.; Zhang, J.; Eugene, B.D.; Li, S. Effect of Electrode Positioning on Electrokinetic Remediation of Contaminated Soft Clay with Surface Electrolyte. Toxics 2024, 12, 758. [Google Scholar] [CrossRef]
- Wang, Y.; Han, Z.; Li, A.; Cui, C. Enhanced electrokinetic remediation of heavy metals contaminated soil by biodegradable complexing agents. Environ. Pollut. 2021, 283, 117111. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.; Wang, B.; Ai, H.; Qi, Y.; Liu, Z. A comparative study on solidification/stabilization characteristics of coal fly ash-based geopolymer and Portland cement on heavy metals in MSWI fly ash. J. Clean. Prod. 2021, 319, 128790. [Google Scholar] [CrossRef]
- Wang, L.; Cho, D.-W.; Tsang, D.C.; Cao, X.; Hou, D.; Shen, Z.; Alessi, D.S.; Ok, Y.S.; Poon, C.S. Green remediation of As and Pb contaminated soil using cement-free clay-based stabilization/solidification. Environ. Int. 2019, 126, 336–345. [Google Scholar] [CrossRef]
- Chen, T.; Wang, L.; He, B.; Peng, X.; Nie, X.; Ma, F.; Han, P.; Bai, X. Study on the solidification/stabilization of cadmium-contaminated soil by red mud-assisted blast furnace slag under excitation conditions. J. Clean. Prod. 2024, 435, 140505. [Google Scholar] [CrossRef]
- Xia, M.; Muhammad, F.; Zeng, L.; Li, S.; Huang, X.; Jiao, B.; Shiau, Y.; Li, D. Solidification/stabilization of lead-zinc smelting slag in composite based geopolymer. J. Clean. Prod. 2019, 209, 1206–1215. [Google Scholar] [CrossRef]
- Liu, J.; Zha, F.; Xu, L.; Kang, B.; Yang, C.; Feng, Q.; Zhang, W.; Zhang, J. Strength and microstructure characteristics of cement-soda residue solidified/stabilized zinc contaminated soil subjected to freezing–thawing cycles. Cold Reg. Sci. Technol. 2020, 172, 102992. [Google Scholar] [CrossRef]
- Zhang, Y.; Ong, Y.J.; Yi, Y. Comparison between CaO-and MgO-activated ground granulated blast-furnace slag (GGBS) for stabilization/solidification of Zn-contaminated clay slurry. Chemosphere 2022, 286, 131860. [Google Scholar] [CrossRef]
- Kim, E.-J.; Park, S.; Adil, S.; Lee, S.; Cho, K. Biogeochemical alteration of an aquifer soil during in situ chemical oxidation by hydrogen peroxide and peroxymonosulfate. Environ. Sci. Technol. 2021, 55, 5301–5311. [Google Scholar] [CrossRef]
- Zarei, A.A.; Bazrafshan, E.; Mosafer, J.; Foroughi, M.; Khaksefidi, R.; Boghsani, G.T.; Mohammadi, L.; Dargahi, A. In situ chemical oxidation of tinidazole in aqueous media by heat-activated persulfate: Kinetics, thermodynamic, and mineralization studies. Appl. Water Sci. 2024, 14, 71. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, Y.; Zhou, A. A potential novel approach for in situ chemical oxidation based on the combination of persulfate and dithionite. Sci. Total Environ. 2019, 693, 133635. [Google Scholar] [CrossRef]
- Dangi, M.B.; Urynowicz, M.A.; Schultz, C.L.; Budhathoki, S. A comparison of the soil natural oxidant demand exerted by permanganate, hydrogen peroxide, sodium persulfate, and sodium percarbonate. Environ. Chall. 2022, 7, 100456. [Google Scholar] [CrossRef]
- Fang, Z.; Zhou, Z.; Xue, G.; Yu, Y.; Wang, Q.; Cheng, B.; Ge, Y.; Qian, Y. Application of sludge biochar combined with peroxydisulfate to degrade fluoroquinolones: Efficiency, mechanisms and implication for ISCO. J. Hazard. Mater. 2022, 426, 128081. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.-Y.; Pokhrel, P.; Wang, Y.-S.; Lin, S.-L.; Liu, M.-H. Combined application of ozone and hydrogen peroxide to degrade diesel contaminants in soil and groundwater. Water 2021, 13, 3404. [Google Scholar] [CrossRef]
- Park, S.; Oh, S.; Kim, I. Role of Borate Buffer in Organic Degradation by Peroxymonosulfate in the Presence of Metal Oxides. Water 2021, 13, 2698. [Google Scholar] [CrossRef]
- Lea, K.; Eric, S.; Rula, D. Verification Monitoring for In Situ Chemical Reduction Using Zero-Valent Zinc, A Novel Technology for Remediation of Chlorinated Alkanes. 2020. Available online: https://serdp-estcp.mil/projects/details/785bb4d4-9971-4722-a636-6ae8f895c636 (accessed on 15 May 2025).
- Huang, T.; Li, Y.; Qian, J.; Liu, S.; Wang, J. Remediation of Cr (VI)-contaminated soil by double-modified nanoscale zero-valent iron: Performance and mechanism. J. Soils Sediments 2024, 24, 1724–1738. [Google Scholar] [CrossRef]
- Galvão, Á.G.; Costa, L.G.; Costa, E.C.d.A.; da Silva, D.R.; Martínez-Huitle, C.A.; Vieira dos Santos, E. Application of a Zero-Valent Iron/Cork as Permeable Reactive Barrier for In Situ Remediation of Phenanthrene in Soil. Catalysts 2022, 12, 1591. [Google Scholar] [CrossRef]
- Wang, L.; Wei, J.; Yang, L.; Chen, Y.; Wang, M.; Xiao, L.; Yuan, G. Enhancing soil remediation of copper-contaminated soil through washing with a soluble humic substance and chemical reductant. Agronomy 2023, 13, 1754. [Google Scholar] [CrossRef]
- Singh, P.; Pal, P.; Mondal, P.; Saravanan, G.; Nagababu, P.; Majumdar, S.; Labhsetwar, N.; Bhowmick, S. Kinetics and mechanism of arsenic removal using sulfide-modified nanoscale zerovalent iron. Chem. Eng. J. 2021, 412, 128667. [Google Scholar] [CrossRef]
- Hussain, A.A.; Kamran, K.; Ishaq, M.; Akram, A.; Hina, M. Optimization of electroosmotic flow to enhance the removal of contaminants from low-permeable soils. J. Appl. Electrochem. 2023, 53, 1245–1258. [Google Scholar] [CrossRef]
- Asadollahfardi, G.; Sarmadi, M.S.; Rezaee, M.; Khodadadi-Darban, A.; Yazdani, M.; Paz-Garcia, J.M. Comparison of different extracting agents for the recovery of Pb and Zn through electrokinetic remediation of mine tailings. J. Environ. Manag. 2021, 279, 111728. [Google Scholar] [CrossRef]
- Wang, L.; Yu, K.; Li, J.-S.; Tsang, D.C.; Poon, C.S.; Yoo, J.-C.; Baek, K.; Ding, S.; Hou, D.; Dai, J.-G. Low-carbon and low-alkalinity stabilization/solidification of high-Pb contaminated soil. Chem. Eng. J. 2018, 351, 418–427. [Google Scholar] [CrossRef]
- Jiang, M.; Wang, K.; Xue, R.; Zhao, Q.; Wang, L.; Wang, W.; Ding, J. Remediation of antimony-contaminated soil with composite materials of Fe/Fe2+-fly ash-concrete additive in seasonal freezing regions. Process Saf. Environ. Prot. 2022, 167, 247–256. [Google Scholar] [CrossRef]
- Xia, W.-Y.; Du, Y.-J.; Li, F.-S.; Guo, G.-L.; Yan, X.-L.; Li, C.-P.; Arulrajah, A.; Wang, F.; Wang, S. Field evaluation of a new hydroxyapatite based binder for ex-situ solidification/stabilization of a heavy metal contaminated site soil around a Pb-Zn smelter. Constr. Build. Mater. 2019, 210, 278–288. [Google Scholar] [CrossRef]
- Kan, L.; Shi, R.; Zhao, Y.; Duan, X.; Wu, M. Feasibility study on using incineration fly ash from municipal solid waste to develop high ductile alkali-activated composites. J. Clean. Prod. 2020, 254, 120168. [Google Scholar] [CrossRef]
- Wu, H.-L.; Jin, F.; Bo, Y.-L.; Du, Y.-J.; Zheng, J.-X. Leaching and microstructural properties of lead contaminated kaolin stabilized by GGBS-MgO in semi-dynamic leaching tests. Constr. Build. Mater. 2018, 172, 626–634. [Google Scholar] [CrossRef]
- Yaman, C. Performance and kinetics of bioaugmentation, biostimulation, and natural attenuation processes for bioremediation of crude oil-contaminated soils. Processes 2020, 8, 883. [Google Scholar] [CrossRef]
- Yang, Y.; Xiao, C.; Wang, F.; Peng, L.; Zeng, Q.; Luo, S. Assessment of the potential for phytoremediation of cadmium polluted soils by various crop rotation patterns based on the annual input and output fluxes. J. Hazard. Mater. 2022, 423, 127183. [Google Scholar] [CrossRef]
- Sánchez Mata, O.; Aguilera Flores, M.M.; Ureño García, B.G.; Ávila Vázquez, V.; Cabañas García, E.; Franco Villegas, E.A. Bioremediation of Automotive Residual Oil-Contaminated Soils by Biostimulation with Enzymes, Surfactant, and Vermicompost. Int. J. Environ. Res. Public Health 2023, 20, 6600. [Google Scholar] [CrossRef]
- Wu, M.; Dick, W.A.; Li, W.; Wang, X.; Yang, Q.; Wang, T.; Xu, L.; Zhang, M.; Chen, L. Bioaugmentation and biostimulation of hydrocarbon degradation and the microbial community in a petroleum-contaminated soil. Int. Biodeterior. Biodegrad. 2016, 107, 158–164. [Google Scholar] [CrossRef]
- Haleyur, N.; Shahsavari, E.; Jain, S.S.; Koshlaf, E.; Ravindran, V.B.; Morrison, P.D.; Osborn, A.M.; Ball, A.S. Influence of bioaugmentation and biostimulation on PAH degradation in aged contaminated soils: Response and dynamics of the bacterial community. J. Environ. Manag. 2019, 238, 49–58. [Google Scholar] [CrossRef]
- Wang, X.; Zhao, R.; Wu, H.; Jia, X.; Liu, Y.; Zhou, G.; Chen, S.; Zhao, F.; Li, L.; Hu, S. Enhanced bioremediation of hexavalent chromium via Stenotrophomonas acidaminiphila 4–1 assisted with agricultural wastes-derived biochar. Biochem. Eng. J. 2024, 208, 109355. [Google Scholar] [CrossRef]
- Barba, S.; Villaseñor, J.; Rodrigo, M.A.; Cañizares, P. Biostimulation versus bioaugmentation for the electro-bioremediation of 2,4-dichlorophenoxyacetic acid polluted soils. J. Environ. Manag. 2021, 277, 111424. [Google Scholar] [CrossRef] [PubMed]
- Sharma, J.K.; Kumar, N.; Singh, N.P.; Santal, A.R. Phytoremediation technologies and their mechanism for removal of heavy metal from contaminated soil: An approach for a sustainable environment. Front. Plant Sci. 2023, 14, 1076876. [Google Scholar] [CrossRef] [PubMed]
- Phang, L.-Y.; Mingyuan, L.; Mohammadi, M.; Tee, C.-S.; Yuswan, M.H.; Cheng, W.-H.; Lai, K.-S. Phytoremediation as a viable ecological and socioeconomic management strategy. Environ. Sci. Pollut. Res. 2024, 31, 50126–50141. [Google Scholar] [CrossRef]
- Wiafe, S.; Boahen, C.; Bandoh, T. Enhanced Phytoremediation of Heavy Metals By Soil-applied Organic And Inorganic Amendments: Heavy Metal Phytoavailability, Accumulation, And Metal Recovery. Soil Sediment Contam. Int. J. 2024, 34, 634–658. [Google Scholar] [CrossRef]
- Hou, L.; Liu, R.; Li, N.; Dai, Y.; Yan, J. Study on the efficiency of phytoremediation of soils heavily polluted with PAHs in petroleum-contaminated sites by microorganism. Environ. Sci. Pollut. Res. 2019, 26, 31401–31413. [Google Scholar] [CrossRef]
- Chen, D.; Liu, X.; Bian, R.; Cheng, K.; Zhang, X.; Zheng, J.; Joseph, S.; Crowley, D.; Pan, G.; Li, L. Effects of biochar on availability and plant uptake of heavy metals–A meta-analysis. J. Environ. Manag. 2018, 222, 76–85. [Google Scholar] [CrossRef]
- Li, H.; Jin, R.; Xu, Z.; Hu, H.; Kalkhajeh, Y.K.; Zhao, Y.; Zhan, L. Application of chelate GLDA for remediating Cd-contaminated farmlands using Tagetes patula L. Environ. Sci. Pollut. Res. 2023, 30, 3774–3782. [Google Scholar] [CrossRef]
- Chigbo, C.; Batty, L. Chelate-assisted phytoremediation of Cu-pyrene-contaminated soil using Z. mays. Water Air Soil Pollut. 2015, 226, 74. [Google Scholar] [CrossRef]
- Shehata, S.M.; Badawy, R.K.; Aboulsoud, Y.I. Phytoremediation of some heavy metals in contaminated soil. Bull. Natl. Res. Cent. 2019, 43, 189. [Google Scholar] [CrossRef]
- Liu, Z.-F.; Ge, H.-G.; Li, C.; Zhao, Z.-P.; Song, F.-M.; Hu, S.-B. Enhanced phytoextraction of heavy metals from contaminated soil by plant co-cropping associated with PGPR. Water Air Soil Pollut. 2015, 226, 29. [Google Scholar] [CrossRef]
- Lai, W.-L.; Lee, F.-Y.; Chen, C.S.; Hseu, Z.-Y.; Kuo, Y.-L. The removal efficacy of heavy metals and total petroleum hydrocarbons from contaminated soils by integrated bio-phytoremediation. J. Soil Groundw. Environ. 2014, 19, 35–44. [Google Scholar] [CrossRef]
- Bhat, S.A.; Bashir, O.; Haq, S.A.U.; Amin, T.; Rafiq, A.; Ali, M.; Américo-Pinheiro, J.H.P.; Sher, F. Phytoremediation of heavy metals in soil and water: An eco-friendly, sustainable and multidisciplinary approach. Chemosphere 2022, 303, 134788. [Google Scholar] [CrossRef] [PubMed]
- Kalhor, A.X.; Movafeghi, A.; Mohammadi-Nassab, A.D.; Abedi, E.; Bahrami, A. Potential of the green alga Chlorella vulgaris for biodegradation of crude oil hydrocarbons. Mar. Pollut. Bull. 2017, 123, 286–290. [Google Scholar] [CrossRef]
- Cerqueira, V.S.; Maria do Carmo, R.P.; Camargo, F.A.; Bento, F.M. Comparison of bioremediation strategies for soil impacted with petrochemical oily sludge. Int. Biodeterior. Biodegrad. 2014, 95, 338–345. [Google Scholar] [CrossRef]
- Chen, W.; Wang, F.; Zeng, L.; Li, Q. Bioremediation of petroleum-contaminated soil by semi-aerobic aged refuse biofilter: Optimization and mechanism. J. Clean. Prod. 2021, 294, 125354. [Google Scholar] [CrossRef]
- Dillon, L. Race, waste, and space: Brownfield redevelopment and environmental justice at the Hunters Point Shipyard. Antipode 2014, 46, 1205–1221. [Google Scholar] [CrossRef]
- Navy USDOT. Finding of Suitability to Transfer for Parcel A (Revision 3), Hunters Point Shipyard; Department of the Navy, Base Realignment and Closure: San Francisco, CA, USA, 2004. [Google Scholar]
- U.S. EPA; Department of Defense; Department of Energy; Department of Interior I; National Aeronautics and Space Administration. Tennessee Valley Authority; Coast Guard; Abstracts of Remediation Case Studies; Federal Remediation Technologies Roundtable: Washington, DC, USA, 2006; Volume 10. [Google Scholar]
- White, R. Deconstructing Mare Island: Reconnaissance in the Ruins. Boom A J. Calif. 2012, 2, 55–69. [Google Scholar] [CrossRef][Green Version]
- Battelle. Remedial Investigation and Focused Feasibility Study (RI/FFS) Report for the Defense Reutilization and Marketing Office (DRMO), Former Mare Island Naval Shipyard; Battelle, 505 King Avenue: Columbus, OH, USA, 2014. [Google Scholar]
- Woods, E.E.S. Designing an Integrated Waterfront: Responsive Redevelopment at the Philadelphia Navy Yard; Massachusetts Institute of Technology: Cambridge, MA, USA, 2012. [Google Scholar]
- U.S. EPA. Philadelphia Navy Yard Site Spotlight; U.S. EPA, Region III: Philadelphia, PA, USA, 2025. [Google Scholar]
- Calisti, S.P. The Redevelopment of the Charlestown Navy Yard: A Model for the Military Base Closure Process. Ph.D. Thesis, University Of Florida, Gainesville, FL, USA, 1992. [Google Scholar]
- U.S. EPA. Environmental Summary Report for Charlestown Navy Yard Parcels 6 and 7; U.S. EPA Region 1: Boston, MA, USA, 2010. [Google Scholar]
- Massachusetts Department of Environmental Protection (MassDEP); Phase II Site Assessment Report: Charlestown Navy Yard Building 105 Area. U.S. Army Corps of Engineers (USACE): Boston, MA, USA, 2012.
- Andrén, S. Urban sustainable development from a place-based and a system-based approach: Case study Malmö. In Proceedings of the International Conference City Futures 09, Bangkok, Thailand, 7–9 March 2009. [Google Scholar]
- Varvsstaden, A.B. Varvstaden—An Example of Transformation and Upcycling of Local Resources; Nordic Sustainable Construction: Malmö, Sweden, 2023. [Google Scholar]
- Lehoux, A.P.; Petersen, K.; Leppänen, M.T.; Snowball, I.; Olsen, M. Status of contaminated marine sediments in four Nordic countries: Assessments, regulations, and remediation approaches. J. Soils Sediments 2020, 20, 2619–2629. [Google Scholar] [CrossRef]
- Oslo Port, A. Oslo Harbor Remediation Project Summary; Oslo Havn KF: Oslo, Norway, 2015. [Google Scholar]
- Pettersen, A.; Solerød, A.W.; Norwegian Geotechnical Institute (NGI); Port of Oslo. The Oslo Harbour Remediation Project: Oslo Fjord Clean Up; Oslo Havn KF: Oslo, Norway, 2007. [Google Scholar]
- Ministry of Climate and Environment; Port of Oslo. RenOslofjord: Final Report on Harbor Sediment Remediation; Norwegian Environment Agency: Oslo, Norway, 2018. [Google Scholar]
- U.S. EPA. Howards Bay Remediation, Great Lakes Areas of Concern. Available online: https://www.epa.gov/great-lakes-aocs/howards-bay-remediation (accessed on 19 May 2025).
- U.S. EPA. Howards Bay Area of Concern Cleanup Summary; U.S. EPA Region 5: Chicago, IL, USA, 2021. [Google Scholar]
- Wisconsin Department of Natural Resources. Dredging Complete at Howards Bay; WDNR Remediation and Redevelopment Program: Madison, WI, USA, 2022. [Google Scholar]
- Arcadis; Wisconsin Department of Natural Resources. Howards Bay Cleanup and Restoration Overview; Arcadis and WDNR: Madison, WI USA, 2020. [Google Scholar]
- City of Tongyeong. Project Overview of Soil and Groundwater Remediation at Former Tongyeong Shipyard; Internal Meeting Document: Tongyeong-si, Republic of Korea, 2023. [Google Scholar]
- U.S. EPA. Soil Washing Treatment: Engineering Bulletin; EPA Office of Emergency and Remedial Response: Washington, DC, USA, 1990. [Google Scholar]
- Lee, H.; Lee, Y.; Kim, J.; Kim, C. Field Application of Modified In Situ Soil Flushing in Combination with Air Sparging at a Military Site Polluted by Diesel and Gasoline in Korea. Int. J. Environ. Res. Public Health 2014, 11, 8806–8824. [Google Scholar] [CrossRef]
- Haemers, T. Comparison of Power Consumption for Thermal Conductive Heating vs Electrical Resistance in In Situ Thermal Desorption; Haemers Technologies: Brussels, Belgium, 2018. [Google Scholar]
- California Environmental Protection Agency – Department of Toxic Substances Control (DTSC). Remediation of Chlorinated Volatile Organic Compounds in Vadose Zone Soil (PT&R Guidance); California Environmental Protection Agency, DTSC: Sacramento, CA, USA, 2010. [Google Scholar]
- Wei, K.-H.; Ma, J.; Xi, B.-D.; Yu, M.-D.; Cui, J.; Chen, B.-L.; Li, Y.; Gu, Q.-B.; He, X.-S. Recent progress on in-situ chemical oxidation for the remediation of petroleum contaminated soil and groundwater. J. Hazard. Mater. 2022, 432, 128738. [Google Scholar] [CrossRef] [PubMed]
- Andrade, L.N.; Araujo, S.F.; Matos, A.T.; Henriques, A.B.; Oliveira, L.C.; Souza, P.P.; Chagas, P.; Leão, M.M.D.; Amorim, C.C. Performance of different oxidants in the presence of oxisol: Remediation of groundwater contaminated by gasoline/ethanol blend. Chem. Eng. J. 2017, 308, 428–437. [Google Scholar] [CrossRef]
- Piscitelli, D.; Zingaretti, D.; Verginelli, I.; Gavasci, R.; Baciocchi, R. The fate of MtBE during Fenton-like treatments through laboratory scale column tests. J. Contam. Hydrol. 2015, 183, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Tratnyek, P.G.; Johnson, R.L.; Lowry, G.V.; Brown, R.A. In Situ Chemical Reduction for Source Remediation. In Chlorinated Solvent Source Zone Remediation; Kueper, B.H., Stroo, H.F., Vogel, C.M., Ward, C.H., Eds.; Springer: New York, NY, USA, 2014; pp. 307–351. [Google Scholar]
- Yuan, L.; Xu, X.; Li, H.; Wang, Q.; Wang, N.; Yu, H. The influence of macroelements on energy consumption during periodic power electrokinetic remediation of heavy metals contaminated black soil. Electrochim. Acta 2017, 235, 604–612. [Google Scholar] [CrossRef]
- U.S. EPA. Solidification/Stabilization Resource Guide; EPA Office of Solid Waste and Emergency Response: Washington, DC, USA, 1999. [Google Scholar]
- Wang, L.; Chen, G.; Du, X.; Li, M.; Zhang, Z.; Liang, H.; Gao, D. Bioremediation of PAHs-contaminated site in a full-scale biopiling system with immobilized enzymes: Removal efficiency and microbial communities. Environ. Res. 2024, 262, 119763. [Google Scholar] [CrossRef]
- Pivetz, B.E. Phytoremediation of Contaminated Soil and Ground Water at Hazardous Waste Sites; US Environmental Protection Agency, Office of Research and Development: Washington DC, USA, 2001. [Google Scholar]
- Lemming, G.; Hauschild, M.Z.; Bjerg, P.L. Life cycle assessment of soil and groundwater remediation technologies: Literature review. Int. J. Life Cycle Assess. 2010, 15, 115–127. [Google Scholar] [CrossRef]
- Cadotte, M.; Deschênes, L.; Samson, R. Selection of a remediation scenario for a diesel-contaminated site using LCA. Int. J. Life Cycle Assess. 2007, 12, 239–251. [Google Scholar] [CrossRef]
- Cai, Y.; Chen, H.; Zhao, W.; Sun, Y.; Wang, M.; Chen, Y.; Ma, J. Investigation of greenhouse gas emissions in soil remediation activities based on the integration of machine learning models and life cycle assessment. J. Clean. Prod. 2025, 487, 144630. [Google Scholar] [CrossRef]
- Ashkanani, Z.; Mohtar, R.; Al-Enezi, S.; Smith, P.K.; Calabrese, S.; Ma, X.; Abdullah, M. AI-assisted systematic review on remediation of contaminated soils with PAHs and heavy metals. J. Hazard. Mater. 2024, 468, 133813. [Google Scholar] [CrossRef]
- Khalid, S.; Shahid, M.; Niazi, N.K.; Murtaza, B.; Bibi, I.; Dumat, C. A comparison of technologies for remediation of heavy metal contaminated soils. J. Geochem. Explor. 2017, 182, 247–268. [Google Scholar] [CrossRef]
- Falconi, M. Soil Washing (SW) Report; IMPEL: Brussels, Belgium, 2023; p. 120. ISBN 978-2-931225-30-1. [Google Scholar]
- Song, Y.H.; Kim, G.Y.; Kim, D.Y.; Hwang, Y.W. Life Cycle Assessment of Crude Oil-Contaminated Soil Treated by Low-Temperature Thermal Desorption and Its Beneficial Reuse for Soil Amendment. Sustainability 2024, 16, 10900. [Google Scholar] [CrossRef]
- Geoengineer.Org. Contaminated Soil Remediation through Thermal Desorption: Synthesis of Case Histories and Comparison of In-Situ and Ex-Situ Applications; Geoengineer.Org, 29 April 2021. Available online: https://www.geoengineer.org/education/web-class-projects/ce-176-environmental-geotechnics/assignments/contaminated-soil-remediation-through-thermal-desorption (accessed on 15 May 2025).
- Vilén, A.; Laurell, P.; Vahala, R. Comparative life cycle assessment of activated carbon production from various raw materials. J. Environ. Manag. 2022, 324, 116356. [Google Scholar] [CrossRef] [PubMed]
- U.S. EPA. How to Evaluate Alternative Cleanup Technologies for Underground Storage Tank Sites: A Guide for Corrective Action Plan Reviewers; EPA 510-B-17-003; EPA Office of Underground Storage Tanks: Washington, DC, USA, 2017. [Google Scholar]
- Inogen Alliance. How Much Does It Cost for Site Remediation? Inogen Alliance Website. 23 June 2023. Available online: https://inogenalliance.com/ (accessed on 15 May 2025).
- Deng, B.; Carter, R.A.; Cheng, Y.; Liu, Y.; Eddy, L.; Wyss, K.M.; Ucak-Astarlioglu, M.G.; Luong, D.X.; Gao, X.; JeBailey, K.; et al. High-temperature electrothermal remediation of multi-pollutants in soil. Nat. Commun. 2023, 14, 6371. [Google Scholar] [CrossRef]
- Cameselle, C. Electrokinetic Remediation, Cost Estimation. In Encyclopedia of Applied Electrochemistry; Kreysa, G., Ota, K.-I., Savinell, R.F., Eds.; Springer: New York, NY, USA, 2014; pp. 723–725. [Google Scholar]
- Fernández-Marchante, C.M.; Vieira dos Santos, E.; Souza, F.L.; Martínez-Huitle, C.A.; Rodríguez-Gómez, A.; Lobato, J.; Rodrigo, M.A. Environmental impact assessment of the electrokinetic adsorption barriers to remove different herbicides from agricultural soils. Sci. Total Environ. 2024, 927, 172287. [Google Scholar] [CrossRef]
- Orellana, R.; Cumsille, A.; Piña-Gangas, P.; Rojas, C.; Arancibia, A.; Donghi, S.; Stuardo, C.; Cabrera, P.; Arancibia, G.; Cárdenas, F.; et al. Economic Evaluation of Bioremediation of Hydrocarbon-Contaminated Urban Soils in Chile. Sustainability 2022, 14, 11854. [Google Scholar] [CrossRef]
- Vafa, H.J.; Pourbabaee, A.A.; Alikhani, H.A.; Yazdanfar, N.; Khanali, M. A comparative life cycle analysis of bioremediation approaches for old-aged petroleum pollution in hypersaline soil. Chemosphere 2025, 373, 144150. [Google Scholar] [CrossRef]
- Aghili, S.; Golzary, A. Greening the earth, healing the soil: A comprehensive life cycle assessment of phytoremediation for heavy metal contamination. Environ. Technol. Innov. 2023, 32, 103241. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).