Synthesis, Applications, and Inhibition Mechanisms of Carbon Dots as Corrosion Inhibitors: A Review
Abstract
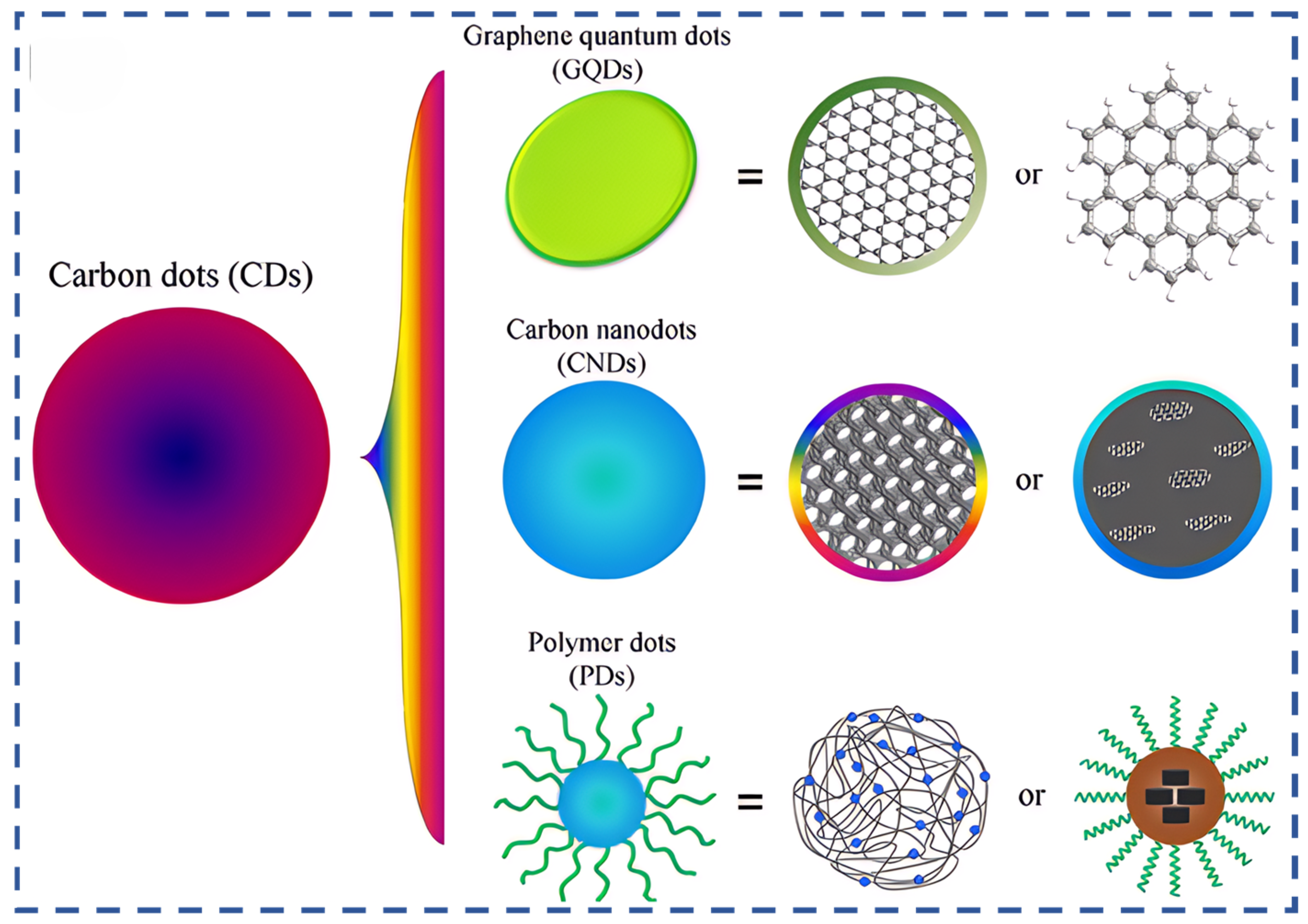
1. Introduction
2. Synthesis and Applications of CDs as Corrosion Inhibitors
2.1. Acid Solutions
2.2. Alkaline Environments
2.3. Neutral Solutions
2.4. CO2-Saturated Solutions
2.5. Microbiologically Influenced Corrosion
3. Mechanisms of Corrosion Inhibition by CDs
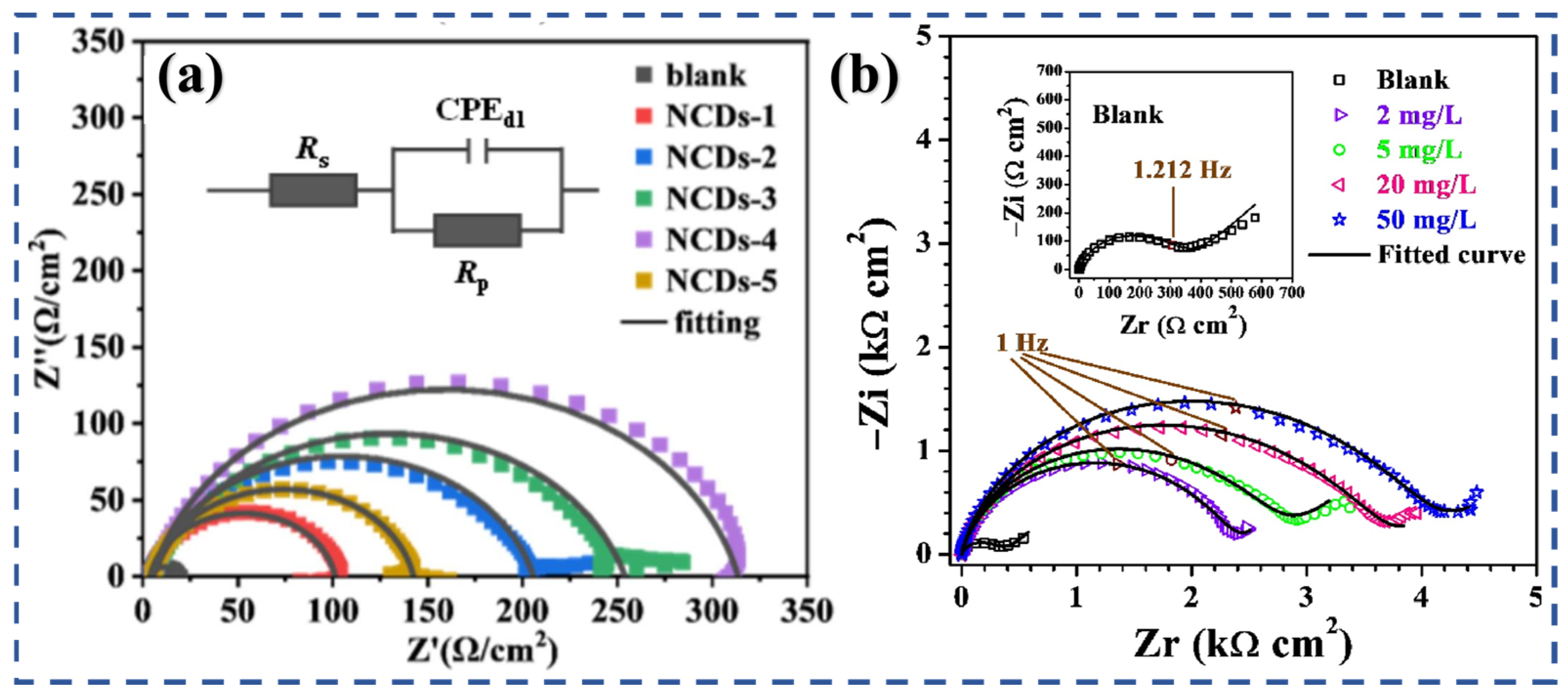
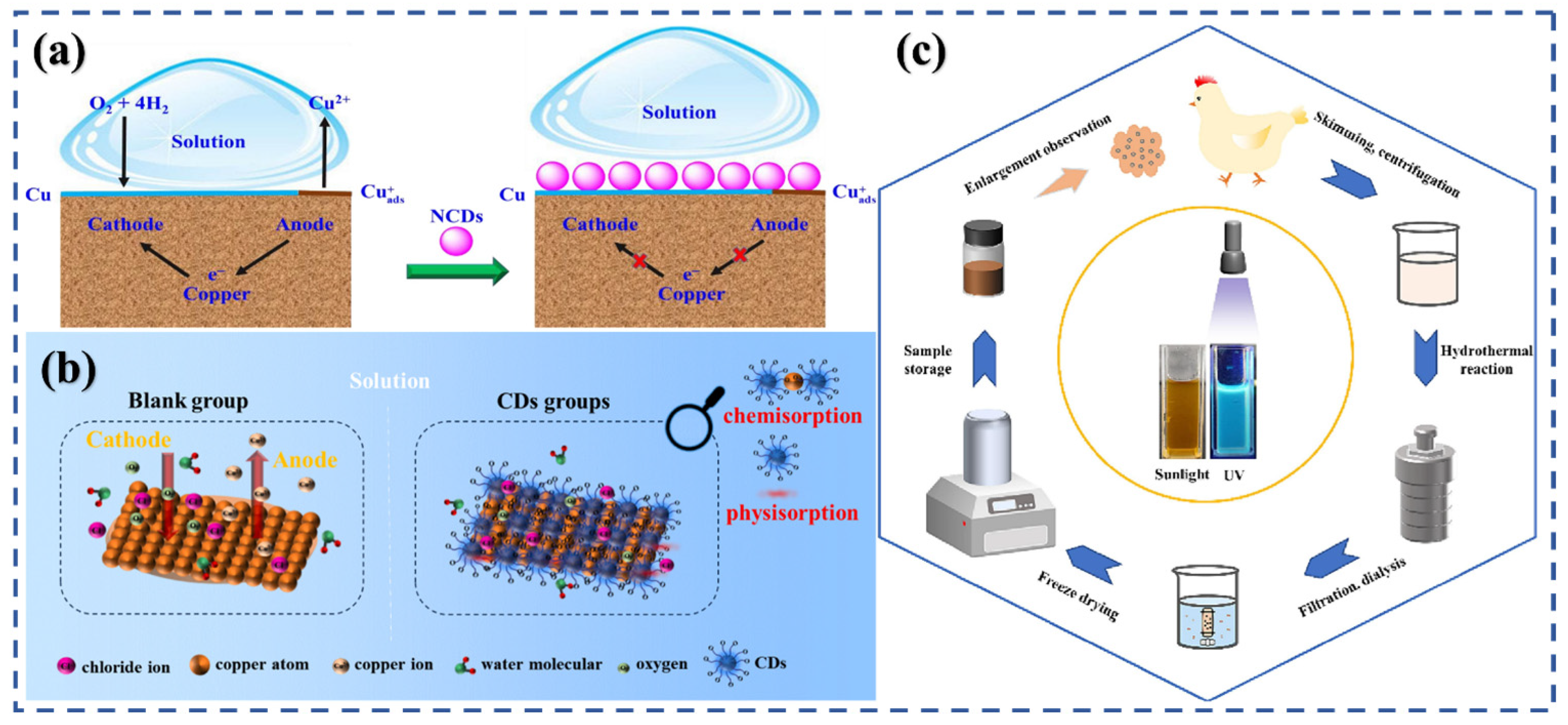
3.1. Adsorption and Film Formation
3.2. Influence of Heteroatom Doping and Electronic Structure
3.3. Electrochemical Inhibition Pathways
3.4. Synergistic Effects of Size, Structure, and Sustainability
4. Conclusions and Future Prospects
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CDs | Carbon dots |
| GQDs | Graphene quantum dots |
| CNDs | Carbon nanodots |
| PDs | Polymer dots |
| MIC | Microbiologically influenced corrosion |
| N-CDs | N-doped CDs |
References
- Hou, B.; Li, X.; Ma, X.; Du, C.; Zhang, D.; Zheng, M.; Xu, W.; Lu, D.; Ma, F. The cost of corrosion in China. npj Mater. Degrad. 2017, 1, 4. [Google Scholar] [CrossRef]
- Verma, C.; Ebenso, E.E.; Bahadur, I.; Quraishi, M.A. An overview on plant extracts as environmental sustainable and green corrosion inhibitors for metals and alloys in aggressive corrosive media. J. Mol. Liq. 2018, 266, 577–590. [Google Scholar] [CrossRef]
- Verma, C.; Olasunkanmi, L.O.; Ebenso, E.E.; Quraishi, M.A. Substituents effect on corrosion inhibition performance of organic compounds in aggressive ionic solutions: A review. J. Mol. Liq. 2018, 251, 100–118. [Google Scholar] [CrossRef]
- Gupta, R.K.; Malviya, M.; Verma, C.; Quraishi, M.A. Aminoazobenzene and diaminoazobenzene functionalized graphene oxides as novel class of corrosion inhibitors for mild steel: Experimental and DFT studies. Mater. Chem. Phys. 2017, 198, 360–373. [Google Scholar] [CrossRef]
- Gupta, R.K.; Malviya, M.; Verma, C.; Gupta, N.K.; Quraishi, M.A. Pyridine-based functionalized graphene oxides as a new class of corrosion inhibitors for mild steel: An experimental and DFT approach. RSC Adv. 2017, 7, 39063–39074. [Google Scholar] [CrossRef]
- Pourhashem, S.; Rashidi, A.; Vaezi, M.R.; Bagherzadeh, M.R. Excellent corrosion protection performance of epoxy composite coatings filled with amino-silane functionalized graphene oxide. Surf. Coat. Technol. 2017, 317, 1–9. [Google Scholar] [CrossRef]
- Chang, C.-H.; Huang, T.-C.; Peng, C.-W.; Yeh, T.-C.; Lu, H.-I.; Hung, W.-I.; Weng, C.-J.; Yang, T.-I.; Yeh, J.-M. Novel anticorrosion coatings prepared from polyaniline/graphene composites. Carbon 2012, 50, 5044–5051. [Google Scholar] [CrossRef]
- Iannuzzi, M.; Frankel, G.S. The carbon footprint of steel corrosion. npj Mater. Degrad. 2022, 6, 101. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.-G.; Zhang, Y.; Wang, H.; Wan, S.; Song, L.-F.; Liao, B.-K.; Guo, X.-P. Modified nano-lignin as a novel biomass-derived corrosion inhibitor for enhanced corrosion resistance of carbon steel. Corros. Sci. 2024, 227, 111705. [Google Scholar] [CrossRef]
- Rahimi, A.; Farhadian, A.; Berisha, A.; Shaabani, A.; Varfolomeev, M.A.; Mehmeti, V.; Zhong, X.; Yousefzadeh, S.; Djimasbe, R. Novel sucrose derivative as a thermally stable inhibitor for mild steel corrosion in 15% HCl medium: An experimental and computational study. Chem. Eng. J. 2022, 446, 136938. [Google Scholar] [CrossRef]
- Ma, L.; Qiang, Y.; Zhao, W. Designing novel organic inhibitor loaded MgAl-LDHs nanocontainer for enhanced corrosion resistance. Chem. Eng. J. 2021, 408, 127367. [Google Scholar] [CrossRef]
- Mioč, E.K.; Gretić, Z.H.; Ćurković, H.O. Modification of cupronickel alloy surface with octadecylphosphonic acid self–assembled films for improved corrosion resistance. Corros. Sci. 2018, 134, 189–198. [Google Scholar] [CrossRef]
- Qian, K.; Wang, C.; Yang, M.; Zou, X.; Zhang, Y.; Dong, Q.; Wang, X.; Shao, Y.; Cheng, Z.; Ju, J.; et al. Self-assembly Zn-containing layer on PEO-coated Mg with enhanced corrosion resistance, antibacterial activity, and osteogenic property. Corros. Sci. 2023, 226, 111674. [Google Scholar] [CrossRef]
- Daroonparvar, M.; Khan, M.U.F.; Saadeh, Y.; Kay, C.M.; Kasar, A.K.; Kumar, P.; Esteves, L.; Misra, M.; Menezes, P.; Kalvala, P.R.; et al. Modification of surface hardness, wear resistance and corrosion resistance of cold spray Al coated AZ31B Mg alloy using cold spray double layered Ta/Ti coating in 3.5 wt % NaCl solution. Corros. Sci. 2020, 176, 109029. [Google Scholar] [CrossRef]
- Ren, S.; Cui, M.; Liu, C.; Wang, L. A comprehensive review on ultrathin, multi-functionalized, and smart graphene and graphene-based composite protective coatings. Corros. Sci. 2023, 212, 110939. [Google Scholar] [CrossRef]
- Cui, M.; Chen, X.; Mei, S.; Ren, S. Bioinspired polydopamine nanosheets for the enhancement in anti-corrosion performance of water-borne epoxy coatings. Chem. Eng. J. 2023, 471, 144760. [Google Scholar] [CrossRef]
- Wei, L.; Li, W.; Wang, Z.-J.; Liu, W.; Zhu, C.; Liu, F.; Li, W. Confinement assembly of a novel Nb2O5&ZnIn2S4 photoanode and its highly efficient and sensitive photoelectrochemical cathodic protection performance. Chem. Eng. J. 2023, 463, 142233. [Google Scholar] [CrossRef]
- Ji, X.; Zhang, D.; Zhou, L.; Xu, Z.; Wu, Y.; Yang, C. Efficient self-powered cathodic corrosion protection system based on multi-layer grid synergistic triboelectric nanogenerator and power management circuits. Chem. Eng. J. 2024, 485, 149753. [Google Scholar] [CrossRef]
- Dehghani, A.; Bahlakeh, G.; Ramezanzadeh, B.; Ramezanzadeh, M. Potential role of a novel green eco-friendly inhibitor in corrosion inhibition of mild steel in HCl solution: Detailed macro/micro-scale experimental and computational explorations. Constr. Build. Mater. 2020, 245, 118464. [Google Scholar] [CrossRef]
- Zhang, W.; Zhang, D.; Li, X.; Li, C.; Gao, L. Excellent performance of dodecyl dimethyl betaine and calcium gluconate as hybrid corrosion inhibitors for Al alloy in alkaline solution. Corros. Sci. 2022, 207, 110556. [Google Scholar] [CrossRef]
- Zheng, Z.; Hu, J.; Eliaz, N.; Zhou, L.; Yuan, X.; Zhong, X. Mercaptopropionic acid-modified oleic imidazoline as a highly efficient corrosion inhibitor for carbon steel in CO2-saturated formation water. Corros. Sci. 2022, 194, 109930. [Google Scholar] [CrossRef]
- Wang, J.; An, L.; Wang, J.; Gu, J.; Sun, J.; Wang, X. Frontiers and advances in N-heterocycle compounds as corrosion inhibitors in acid medium: Recent advances. Adv. Colloid Interface Sci. 2023, 321, 103031. [Google Scholar] [CrossRef]
- Koch, I.; Moriarty, M.; House, K.; Sui, J.; Cullen, W.R.; Saper, R.B.; Reimer, K.J. Bioaccessibility of lead and arsenic in traditional Indian medicines. Sci. Total Environ. 2011, 409, 4545–4552. [Google Scholar] [CrossRef]
- Schem, M.; Schmidt, T.; Gerwann, J.; Wittmar, M.; Veith, M.; Thompson, G.E.; Molchan, I.S.; Hashimoto, T.; Skeldon, P.; Phani, A.R.; et al. CeO2-filled sol–gel coatings for corrosion protection of AA2024-T3 aluminium alloy. Corros. Sci. 2009, 51, 2304–2315. [Google Scholar] [CrossRef]
- Wang, X.; Liu, J.; Jin, M.; Yan, Y.; Tang, J.; Jin, Z. A review of organic corrosion inhibitors for resistance under chloride attacks in reinforced concrete: Background, Mechanisms and Evaluation methods. Constr. Build. Mater. 2024, 433, 136583. [Google Scholar] [CrossRef]
- Ehsani, A.; Mahjani, M.G.; Hosseini, M.; Safari, R.; Moshrefi, R.; Shiri, H.M. Evaluation of Thymus vulgaris plant extract as an eco-friendly corrosion inhibitor for stainless steel 304 in acidic solution by means of electrochemical impedance spectroscopy, electrochemical noise analysis and density functional theory. J. Colloid Interface Sci. 2017, 490, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Wang, M.; Fadhil, A.A.; Fu, C.; Chen, T.; Chen, M.; Khadom, A.A.; Mahood, H.B. Preparation characterization, and corrosion inhibition performance of graphene oxide quantum dots for Q235 steel in 1 M hydrochloric acid solution. Colloids Surfaces A Physicochem. Eng. Asp. 2021, 627, 127209. [Google Scholar] [CrossRef]
- Guo, L.; Zhang, R.; Tan, B.; Li, W.; Liu, H.; Wu, S. Locust Bean Gum as a green and novel corrosion inhibitor for Q235 steel in 0.5 M H2SO4 medium. J. Mol. Liq. 2020, 310, 113239. [Google Scholar] [CrossRef]
- Faustin, M.; Maciuk, A.; Salvin, P.; Roos, C.; Lebrini, M. Corrosion inhibition of C38 steel by alkaloids extract of Geissospermum laeve in 1M hydrochloric acid: Electrochemical and phytochemical studies. Corros. Sci. 2015, 92, 287–300. [Google Scholar] [CrossRef]
- Mourya, P.; Banerjee, S.; Singh, M.M. Corrosion inhibition of mild steel in acidic solution by Tagetes erecta (Marigold flower) extract as a green inhibitor. Corros. Sci. 2014, 85, 352–363. [Google Scholar] [CrossRef]
- Tan, B.; Xiang, B.; Zhang, S.; Qiang, Y.; Xu, L.; Chen, S.; He, J. Papaya leaves extract as a novel eco-friendly corrosion inhibitor for Cu in H2SO4 medium. J. Colloid Interface Sci. 2021, 582, 918–931. [Google Scholar] [CrossRef]
- Wu, Y.; Guo, L.; She, Y. Insight on the corrosion inhibition performance of Psidium guajava linn leaves extract. J. Mol. Liq. 2022, 346, 117858. [Google Scholar] [CrossRef]
- Huang, W.; Tang, Z.; Liu, X.; Liu, L.; Zhong, H.; Yu, Y.; Chen, H.; Wang, C.; Jiang, Q.; Ye, Y.; et al. A green and effective anti-corrosion and anti-microbial inhibitor of citric acid-based carbon dots: Experiment and mechanism analysis. J. Mater. Res. Technol. 2024, 32, 2149–2159. [Google Scholar] [CrossRef]
- Li, J.; Zhao, X.; Gong, X. The Emerging Star of Carbon Luminescent Materials: Exploring the Mysteries of the Nanolight of Carbon Dots for Optoelectronic Applications. Small 2024, 20, e2400107. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Wang, J.H.; Mao, Q.; Chen, X. Tunable Organelle Imaging by Rational Design of Carbon Dots and Utilization of Uptake Pathways. ACS Nano 2021, 15, 14465–14474. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Sun, T.; Wang, X.; He, H.; E, S. Development of high-dispersion CLDH/carbon dot composites to boost chloride binding of cement. Cem. Concr. Compos. 2024, 152, 105669. [Google Scholar] [CrossRef]
- He, H.; Shuang, E.; Lu, D.; Hu, Y.; Yan, C.; Shan, H.; He, C. Deciphering size-induced influence of carbon dots on mechanical performance of cement composites. Constr. Build. Mater. 2024, 425, 136030. [Google Scholar] [CrossRef]
- Yan, F.; Bai, R.; Huang, J.; Bian, X.; Fu, Y. Machine learning-assisted carbon dots synthesis and analysis: State of the art and future directions. TrAC Trends Anal. Chem. 2025, 184, 118141. [Google Scholar] [CrossRef]
- E, S.; Xing, Y.Z.; Du, S.; Liu, A.M.; Gao, Z.; Zhou, Q.; Xuan, Y.; Zhao, Y.N.; Chen, X.W.; Zhang, S.B. Shape Control of Carbon Nanoparticles via a Simple Anion-Directed Strategy for Precise Endoplasmic Reticulum-Targeted Imaging. Angew. Chem. Int. Ed. 2023, 62, e202311008. [Google Scholar] [CrossRef]
- He, P.; Bai, J.; Yang, G.; Qin, F.; Wang, X.; Yu, X.; Yao, Y.; Tang, X.; Ren, L. Regulation of the unconventional luminescence behaviors of phenylenediamine-based carbon dots with high PLQY values. Chem. Eng. J. 2025, 506, 160342. [Google Scholar] [CrossRef]
- Guo, H.; Raj, J.; Wang, Z.; Zhang, T.; Wang, K.; Lin, L.; Hou, W.; Zhang, J.; Wu, M.; Wu, J.; et al. Synergistic Effects of Amine Functional Groups and Enriched-Atomic-Iron Sites in Carbon Dots for Industrial-Current–Density CO2 Electroreduction. Small 2024, 20, 2311132. [Google Scholar] [CrossRef]
- Lei, Q.; Liu, C.; Nan, X.; Zhu, Y.; Fu, L.; Lin, X.; Zhang, H.; Yang, M.; Fang, X.; Luo, Y.; et al. Carbon dots-based electrochemical and fluorescent biosensors for the detection of foodborne pathogens: Current advance and challenge. Coord. Chem. Rev. 2025, 529, 216457. [Google Scholar] [CrossRef]
- He, C.; E, S.; Yan, H.; Li, X. Structural engineering design of carbon dots for lubrication. Chin. Chem. Lett. 2021, 32, 2693–2714. [Google Scholar] [CrossRef]
- Gao, Y.; Qiu, Z.; Zhao, J.; He, H.; Wang, B. A comprehensive review on the applications of carbon dots in civil engineering materials. J. Mater. Sci. 2025, 60, 3227–3252. [Google Scholar] [CrossRef]
- Zhu, S.; Song, Y.; Zhao, X.; Shao, J.; Zhang, J.; Yang, B. The photoluminescence mechanism in carbon dots (graphene quantum dots, carbon nanodots, and polymer dots): Current state and future perspective. Nano Res. 2015, 8, 355–381. [Google Scholar] [CrossRef]
- Cui, M.; Ren, S.; Xue, Q.; Zhao, H.; Wang, L. Carbon dots as new eco-friendly and effective corrosion inhibitor. J. Alloys Compd. 2017, 726, 680–692. [Google Scholar] [CrossRef]
- Ye, Y.; Jiang, Z.; Zou, Y.; Chen, H.; Guo, S.; Yang, Q.; Chen, L. Evaluation of the inhibition behavior of carbon dots on carbon steel in HCl and NaCl solutions. J. Mater. Sci. Technol. 2020, 43, 144–153. [Google Scholar] [CrossRef]
- Cen, H.; Chen, Z.; Guo, X. N, S co-doped carbon dots as effective corrosion inhibitor for carbon steel in CO2-saturated 3.5% NaCl solution. J. Taiwan Inst. Chem. Eng. 2019, 99, 224–238. [Google Scholar] [CrossRef]
- Kalajahi, S.T.; Rasekh, B.; Yazdian, F.; Neshati, J.; Taghavi, L. Green mitigation of microbial corrosion by copper nanoparticles doped carbon quantum dots nanohybrid. Environ. Sci. Pollut. Res. 2020, 27, 40537–40551. [Google Scholar] [CrossRef]
- Singh, A.; Ansari, K.R.; Chauhan, D.S.; Quraishi, M.A.; Lgaz, H.; Chung, I.-M. Comprehensive investigation of steel corrosion inhibition at macro/micro level by ecofriendly green corrosion inhibitor in 15% HCl medium. J. Colloid Interface Sci. 2020, 560, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Berdimurodov, E.; Verma, D.K.; Kholikov, A.; Akbarov, K.; Guo, L. The recent development of carbon dots as powerful green corrosion inhibitors: A prospective review. J. Mol. Liq. 2022, 349, 118124. [Google Scholar] [CrossRef]
- Li, J.; Lv, J.; Fu, L.; Tang, M.; Wu, X. New Ecofriendly Nitrogen-Doped Carbon Quantum Dots as Effective Corrosion Inhibitor for Saturated CO2 3% NaCl Solution. Russ. J. Appl. Chem. 2020, 93, 380–392. [Google Scholar] [CrossRef]
- Long, W.-J.; Li, X.-Q.; Zheng, S.-Y.; He, C. A novel effective carbon dots-based inhibitor for carbon steel against chloride corrosion: From inhibition behavior to mechanism. Carbon 2023, 218, 118708. [Google Scholar] [CrossRef]
- He, H.; Zhang, Z.; Jiang, C.; He, C. Research progress of carbon dots as novel corrosion inhibitors. J. Mol. Struct. 2025, 1334, 141894. [Google Scholar] [CrossRef]
- Al Jahdaly, B.A.; Elsadek, M.F.; Ahmed, B.M.; Farahat, M.F.; Taher, M.M.; Khalil, A.M. Outstanding Graphene Quantum Dots from Carbon Source for Biomedical and Corrosion Inhibition Applications: A Review. Sustainability 2021, 13, 2127. [Google Scholar] [CrossRef]
- Verma, C.; Alfantazi, A.; Quraishi, M.A. Quantum dots as ecofriendly and aqueous phase substitutes of carbon family for traditional corrosion inhibitors: A perspective. J. Mol. Liq. 2021, 343, 117648. [Google Scholar] [CrossRef]
- Wang, S.W.J.; Chen, Z.; Wang, Y.; Ma, L.; Zhang, D. Research progress of carbon dots in the field of metal corrosion and protection. Surf. Technol. 2024, 53, 75–78. [Google Scholar]
- Zhang, Q.; Zhang, R.; Wu, R.; Luo, Y.; Guo, L.; He, Z. Green and high-efficiency corrosion inhibitors for metals: A review. J. Adhes. Sci. Technol. 2022, 37, 1501–1524. [Google Scholar] [CrossRef]
- Panossian, Z.; Almeida, N.L.D.; Sousa, R.M.F.D.; Pimenta, G.D.S.; Marques, L.B.S. Corrosion of carbon steel pipes and tanks by concentrated sulfuric acid: A review. Corros. Sci. 2012, 58, 1–11. [Google Scholar] [CrossRef]
- Qing, L.; Zhou, T.-Y. Inhibition Behavior of Graphene Quantum Dots for Carbon Steel in HCl Solution. Corros. Prot. 2015, 36, 152–156. [Google Scholar]
- S, E.; Gao, Z.; Cong, X.; He, C.; Gao, L.; Yin, Z. Novel green corrosion inhibitor for carbon steel: A comprehensive analysis of the effect of nitrogen content on corrosion inhibition performance. Chem. Eng. Sci. 2025, 320, 122394. [Google Scholar] [CrossRef]
- Cui, M.; Ren, S.; Zhao, H.; Wang, L.; Xue, Q. Novel nitrogen doped carbon dots for corrosion inhibition of carbon steel in 1 M HCl solution. Appl. Surf. Sci. 2018, 443, 145–156. [Google Scholar] [CrossRef]
- Qiang, Y.; Zhang, S.; Zhao, H.; Tan, B.; Wang, L. Enhanced anticorrosion performance of copper by novel N-doped carbon dots. Corros. Sci. 2019, 161, 108193. [Google Scholar] [CrossRef]
- Ye, Y.; Yang, D.; Chen, H. A green and effective corrosion inhibitor of functionalized carbon dots. J. Mater. Sci. Technol. 2019, 35, 2243–2253. [Google Scholar] [CrossRef]
- Long, W.-J.; Li, X.-Q.; Yu, Y.; He, C. Green synthesis of biomass-derived carbon dots as an efficient corrosion inhibitor. J. Mol. Liq. 2022, 360, 119522. [Google Scholar] [CrossRef]
- Wang, Q.; Wang, R.; Sun, X.; Aslam, R.; Zhou, X.; Zhang, Q.; Zhao, C.; Sun, Y.; Yan, Z.; Li, X. Protein-derived carbon dots as green corrosion inhibitors for carbon steel in sulfuric acid solution. Diam. Relat. Mater. 2024, 145, 111135. [Google Scholar] [CrossRef]
- He, C.; Li, X.-Q.; Feng, G.-L.; Long, W.-J. A universal strategy for green and in situ synthesis of carbon dot-based pickling solution. Green Chem. 2022, 24, 5842–5855. [Google Scholar] [CrossRef]
- Tang, Z.; Huang, W.; Liu, L.; Li, H.; Meng, H.; Zeng, T.; Ye, X.; Jiang, Q.; Ye, Y.W.; Liu, Y. Study on structure and molecular scale protection mechanism of green Ce,N-CDs anti-bacterial and anti-corrosive inhibitor. J. Mater. Res. Technol. 2024, 28, 3865–3881. [Google Scholar] [CrossRef]
- Wan, S.; Chen, H.; Liao, B.; Guo, X. Adsorption and anticorrosion mechanism of glucose-based functionalized carbon dots for copper in neutral solution. J. Taiwan Inst. Chem. Eng. 2021, 129, 289–298. [Google Scholar] [CrossRef]
- Cen, H.; Zhang, X.; Zhao, L.; Chen, Z.; Guo, X. Carbon dots as effective corrosion inhibitor for 5052 aluminium alloy in 0.1 M HCl solution. Corros. Sci. 2019, 161, 108197. [Google Scholar] [CrossRef]
- Bao, H.; Lian, Y.; Qiao, Z.; Zhang, S.; Meng, X.; Ma, K.; Zhu, D.; Zhao, J.; Zhang, H. N-doped carbon quantum dots as corrosion inhibitor for ultra-high voltage etched Al foil. Surf. Coat. Technol. 2023, 476, 130240. [Google Scholar] [CrossRef]
- Zheng, S.; Feng, L.; Hu, Z.; Li, J.; Zhu, H.; Ma, X. Study on the corrosion inhibition of biomass carbon quantum dot self-aggregation on Q235 steel in hydrochloric acid. Arab. J. Chem. 2023, 16, 104605. [Google Scholar] [CrossRef]
- Xu, Z.; Gan, Y.; Zeng, J.; Chen, J.; Fu, A.; Zheng, X.; Li, W. Green synthesis of functionalized fluorescent carbon dots from biomass and their corrosion inhibition mechanism for copper in sulfuric acid environment. Chem. Eng. J. 2023, 470, 144425. [Google Scholar] [CrossRef]
- Liu, Y.; Ren, H.; Gong, Z.; Tan, B.; Lan, W.; Dai, Q.; Zheng, X.; Guo, L.; Alobaid, A.A.; Warad, I. Melon seed shell synthesis N, S-carbon quantum dots as ultra-high performance corrosion inhibitors for copper in 0.5 M H2SO4. J. Ind. Eng. Chem. 2024, 137, 593–605. [Google Scholar] [CrossRef]
- Wang, S.; Wang, J.; Wang, Z.; Zhang, L.; Xu, H. The effect of pyrrolic nitrogen on corrosion inhibition performance of N-doped carbon dots. Surf. Interfaces 2023, 44, 103740. [Google Scholar] [CrossRef]
- Wang, T.; Cao, S.; Sun, Y.; Liu, D.; Pan, S.; Ma, A.; Kuvarega, A.T.; Mamba, B.B.; Gui, J. Ionic liquid-assisted preparation of N, S-rich carbon dots as efficient corrosion inhibitors. J. Mol. Liq. 2022, 356, 118943. [Google Scholar] [CrossRef]
- He, H.; Shi, J.; Yu, S.; Yang, J.; Xu, K.; He, C.; Li, X. Exploring green and efficient zero-dimensional carbon-based inhibitors for carbon steel: From performance to mechanism. Constr. Build. Mater. 2023, 411, 134334. [Google Scholar] [CrossRef]
- Padhan, S.; Rout, T.K.; Nair, U.G. N-doped carbon dots as corrosion inhibitor for mild steel corrosion in acid medium. Colloids Surf. A Physicochem. Eng. Asp. 2022, 653, 129905. [Google Scholar] [CrossRef]
- Peng, Y.; Tang, M.; Qiang, Y.; Deng, S.; Li, X. High-value utilization of renewable biofuel tree species waste: Inhibition mechanism of carbon steel in trichloroacetic acid by Jatropha curcas L. cake meal carbon dots. Colloids Surf. A Physicochem. Eng. Asp. 2025, 729, 138847. [Google Scholar] [CrossRef]
- Tang, Y.; He, H.; Qiao, H.; Wang, S.; He, C.; Xiang, T. Carbon dot aggregates: A new strategy to promote corrosion inhibition performance of carbon dots. J. Mater. Res. Technol. 2024, 33, 1479–1493. [Google Scholar] [CrossRef]
- Shi, C.; Wei, G.; Deng, S.; Li, X. Valorization strategy of invasive weed: Interfacial corrosion inhibition of steel in 1.0 M HCl media by Erigeron canadensis extract. J. Mater. Res. Technol. 2025, 39, 6363–6380. [Google Scholar] [CrossRef]
- Kokalj, A.; Lozinšek, M.; Kapun, B.; Taheri, P.; Neupane, S.; Losada-Pérez, P.; Xie, C.; Stavber, S.; Crespo, D.; Renner, F.U.; et al. Simplistic correlations between molecular electronic properties and inhibition efficiencies: Do they really exist? Corros. Sci. 2021, 179, 108856. [Google Scholar] [CrossRef]
- Li, X.; He, C.; Yu, K.; Luo, Q.; Long, W. Facile Preparation and Characterization of Carbon Dots with Schiff Base Structures Toward an Efficient Corrosion Inhibitor. Surf. Technol. 2023, 52, 229. [Google Scholar]
- Jin, G.; Xue, S.; Zhao, B.; Wang, Y.; Liu, S.; Ye, Q.; Zhou, F. N-doped carbon dots based nano-composite coatings with ultra-low coefficient of friction and superior corrosion resistance. Friction 2025. [Google Scholar] [CrossRef]
- Pooja, K.; Kumari, K.; Kumar, M.R.; Mahendra, Y. Synthesis of novel carbon dots as efficient green corrosion inhibitor for mild steel in an acidic environment: Electrochemical, gravimetric, and XPS analysis. Prog. Org. Coat. 2025, 209, 109561. [Google Scholar]
- Tan, Y.; Zhao, P.; Guo, L.; Zhu, L.; Lgaz, H.; Adam, A.M.M. Spent coffee grounds-derived N-doped carbon dots as efficient green corrosion inhibitors for Q235 steel in acidic environment. Prog. Org. Coat. 2026, 210, 109700. [Google Scholar] [CrossRef]
- Wu, X.; Li, J.; Lv, J.; Deng, C.; Yang, L. Novel Carbon Dots for Corrosion Inhibition of N80 Carbon Steel in 3% Saturated CO2 Saline Solution. Russ. J. Appl. Chem. 2021, 94, 1111–1121. [Google Scholar] [CrossRef]
- Wu, X.; Li, J.; Deng, C.; Yang, L.; Lv, J.; Fu, L. Novel carbon dots as effective corrosion inhibitor for N80 steel in 1 M HCl and CO2-saturated 3.5 wt% NaCl solutions. J. Mol. Struct. 2022, 1250, 131897. [Google Scholar] [CrossRef]
- Shao, H.; Li, D.; Chen, Z.; Yin, X.; Chen, Y.; Liu, Y.; Yang, W. Sulfur dots corrosion inhibitors with superior antibacterial and fluorescent properties. J. Colloid Interface Sci. 2023, 654, 878–894. [Google Scholar] [CrossRef]
- Miao, C.; Wang, Q.; Yang, S.; Tang, Y.; Liu, X.; Lu, S. Hydrothermal route upcycling surgical masks into dual-emitting carbon dots as ratiometric fluorescent probe for Cr (VI) and corrosion inhibitor in saline solution. Talanta 2024, 275, 126070. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Zhang, D.; Wu, P.; Gao, L. Corrosion inhibition of high-nitrogen-doped CDs for copper in 3wt% NaCl solution. J. Taiwan Inst. Chem. Eng. 2022, 138, 104462. [Google Scholar] [CrossRef]
- Luo, J.; Cheng, X.; Chen, X.; Zhong, C.F.; Xie, H.; Ye, Y.W.; Zhao, H.C.; Li, Y.; Chen, H. The effect of N and S ratios in N, S co-doped carbon dot inhibitor on metal protection in 1 M HCl solution. J. Taiwan Inst. Chem. Eng. 2021, 127, 387–398. [Google Scholar] [CrossRef]
- Li, F.; Zhou, Y.; Wang, Z.; Qu, W.; Su, H.; Li, M.; Cui, N.; Sun, S.; Fu, Y.; Hu, S. Ce-doped carbon dots enhanced by ionic liquids: A potent corrosion shield for N80 steel in aggressive 1M HCl environment. Colloids Surf. A Physicochem. Eng. Asp. 2025, 709, 136132. [Google Scholar]
- Aslam, R.; Wang, Q.; Sun, Y.; Yan, Z. Fabrication, characterization and corrosion inhibition performance of sustainable metal-organic framework nanocomposite. Colloids Surf. A Physicochem. Eng. Asp. 2025, 720, 137095. [Google Scholar]
- Wang, Z.; Jin, Z.; Liu, H.; Liu, R.; Zhang, Y.; Yin, Y.; Liu, H.; Yuan, X.; Fan, S.; Liu, H. Insights into the hydrophobic coating with integrated high-efficiency anti-corrosion, anti-biofouling and self-healing properties based on anti-bacterial nano LDH materials. Corros. Sci. 2024, 231, 111995. [Google Scholar] [CrossRef]
- Yang, D.; Ye, Y.; Su, Y.; Liu, S.; Gong, D.; Zhao, H. Functionalization of citric acid-based carbon dots by imidazole toward novel green corrosion inhibitor for carbon steel. J. Clean. Prod. 2019, 229, 180–192. [Google Scholar] [CrossRef]
- Saraswat, V.; Yadav, M. Carbon Dots as Green Corrosion Inhibitor for Mild Steel in HCl Solution. ChemistrySelect 2020, 5, 7347–7357. [Google Scholar] [CrossRef]
- Guan, Y.; Li, X.; Tan, B.; Yang, S.; Deng, S. Hydrothermal synthesis of peanut shell-derived carbon dots as the novel efficient inhibitor for the corrosion of steel. Colloids Surf. A Physicochem. Eng. Asp. 2026, 728, 138517. [Google Scholar] [CrossRef]
- Yang, S.; Deng, S.; Qiang, Y.; Li, X. Nature’s nano-shield: Jatropha curcas fruit seeds meal derived carbon dots as potent green inhibitor. J. Mater. Sci. Technol. 2025, 262, 140–156. [Google Scholar] [CrossRef]







Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, Y.; Hong, T.; Zhou, S.; Wang, Y.; Sheng, S.; Hong, J.; Wang, S.; Liu, C.; He, C.; He, H.; et al. Synthesis, Applications, and Inhibition Mechanisms of Carbon Dots as Corrosion Inhibitors: A Review. Processes 2025, 13, 4002. https://doi.org/10.3390/pr13124002
Hu Y, Hong T, Zhou S, Wang Y, Sheng S, Hong J, Wang S, Liu C, He C, He H, et al. Synthesis, Applications, and Inhibition Mechanisms of Carbon Dots as Corrosion Inhibitors: A Review. Processes. 2025; 13(12):4002. https://doi.org/10.3390/pr13124002
Chicago/Turabian StyleHu, Yin, Tianyao Hong, Sheng Zhou, Yangrui Wang, Shiyu Sheng, Jie Hong, Shifang Wang, Chang Liu, Chuang He, Haijie He, and et al. 2025. "Synthesis, Applications, and Inhibition Mechanisms of Carbon Dots as Corrosion Inhibitors: A Review" Processes 13, no. 12: 4002. https://doi.org/10.3390/pr13124002
APA StyleHu, Y., Hong, T., Zhou, S., Wang, Y., Sheng, S., Hong, J., Wang, S., Liu, C., He, C., He, H., & Xu, M. (2025). Synthesis, Applications, and Inhibition Mechanisms of Carbon Dots as Corrosion Inhibitors: A Review. Processes, 13(12), 4002. https://doi.org/10.3390/pr13124002





