New Polyfunctional Nanocatalysts for the Hydrogen-Free Processing of N-Alkanes and Gasoline Fractions
Abstract
1. Introduction
- -
- elemental sulfur (S) and hydrogen sulfide (H2S), which can be dissolved in the fuel;
- -
- mercaptans (thiols)—organic compounds containing a thiol functional group (–SH);
- -
- sulfides (thioethers)—compounds of the general formula R–S–R′, where R and R′—organic radicals;
- -
- disulfides—compounds containing a disulfide linkage (–S–S–);
- -
- thiophenes and their derivatives, such as thiophane, thiophene, benzothiophene, and dibenzothiophene.
2. Results and Discussion
3. Materials and Methods
3.1. Experimental Procedure and Preparation of Catalyst
3.2. Study of Samples
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| GHSV | Gas Hourly Space Velocity |
| BET | Brunauer–Emmett–Teller Method |
| MM | Motor Method |
| RM | Research Method |
| P | Pressure |
| SEM | Scanning Electron Microscopy |
| TEM | Transmission Electron Microscopy |
| TPD-NH3 | Temperature-Programmed Desorption of Ammonia |
| Tmax | Maximum Temperature |
| XRF | X-ray Fluorescence Spectrum |
References
- Wang, Z.; Ke, M.; Song, Z.; Li, J.; Sun, J. Benzene reduction process simulation and optimization in catalytic cracking gasoline distillation. Processes 2023, 11, 151. [Google Scholar] [CrossRef]
- Chen, F.; Hao, J.; Yu, Y.; Cheng, D.; Zhan, X. The influence of external acid strength of hierarchical ZSM-5 zeolites on n-heptane catalytic cracking. Microporous Mesoporous Mater. 2022, 330, 575–582. [Google Scholar] [CrossRef]
- Tanimu, A.; Tanimu, G.; Alasiri, H.; Aitani, A. Catalytic cracking of crude oil: Mini review of catalyst formulations for enhanced selectivity to light olefins. Energy Fuels 2022, 36, 5152–5166. [Google Scholar] [CrossRef]
- Wang, J.; Mu, H.T.; Dai, T.L. Operatio industrialis FUDS-6 catalyst in fractione pellentesque hydrotreating plantae. Oleum Expolitio Petrochem. 2017, 43, 49–53. [Google Scholar]
- Verma, D.; Rana, B.S.; Kumar, R.; Sibi, M.G.; Sinha, A.K. Diesel and aviation kerosene with desired aromatics from hydroprocessing of jatropha oil over hydroprocessing over hydrogenation catalysts supported on hierarchical mesoporous SAPO-11. Appl. Catal. A Gen. 2015, 490, 108–116. [Google Scholar] [CrossRef]
- Chiosso, M.E.; Crespo, I.; Merlo, A.B.; Valle, B. Metal-doped HZSM-5 zeolite catalysts for catalytic cracking of raw bio-oil: Exploring activity toward value-added products. Catalysts 2023, 13, 1198. [Google Scholar] [CrossRef]
- Wei, Y.J.; Zhang, Y.J.; Zhu, X.D.; Gu, H.M.; Zhu, Z.Q.; Liu, S.H.; Sun, X.Y.; Jiang, X.L. Effects of diesel hydrocarbon components on cetane number and engine combustion and emission characteristics. Appl. Sci. 2022, 12, 3549. [Google Scholar] [CrossRef]
- Gao, L.; Shi, Z.; Etim, U.J.; Wu, P.; Han, D.; Xing, W.; Mintova, S.; Bai, P.; Yan, Z. Beta-MCM-41 micro-mesoporous catalysts in the hydroisomerization of n-heptane: Definition of an indexed isomerization factor as a performance descriptor. Microporous Mesoporous Mater. 2019, 277, 17–28. [Google Scholar] [CrossRef]
- Chebotova, V.I.; Ulanov, V.V. Depth of oil refining in Russia. Business J. Neftegaz Ru. 2021, 109, 14–18. [Google Scholar]
- Milina, M.; Mitchell, S.; Michels, N.L.; Kenvin, J.; Pérez-Ramírez, J. Interdependence between porosity, acidity, and catalytic performance in hierarchical ZSM-5 zeolites prepared by post-synthetic modification. J. Catal. 2013, 308, 398–407. [Google Scholar] [CrossRef]
- Alabdullah, M.; Rodriguez-Gomez, A.; Vittenet, J.; Sedjerari, A.B.; Xu, W.; Abba, I.; Gascon, J. Viewpoint on the refinery of the future: Catalyst and process challenges. ASC Catal. 2020, 1, 8131–8140. [Google Scholar] [CrossRef]
- Ji, Y.; Yang, H.; Yan, W. Strategies to enhance the catalytic performance of ZSM-5 zeolite in hydrocarbon cracking: A review. Catalysts 2017, 7, 367. [Google Scholar] [CrossRef]
- Bogdanov, I.; Altynov, A.; Kirgina, M. Hydrogen-free upgradingon ZSM-5 type zeolite catalyst—Efficient way to obtain low-freezing diesel fuel. S. Afr. J. Chem. Eng. 2022, 41, 1–8. [Google Scholar] [CrossRef]
- Solmanov, P.S.; Maximov, N.M.; Tomina, N.N.; Zanozina, I.I.; Pimerzin, A.A.; Verevkin, S.P. NiMoW/P-Al2O3 four-component catalysts with different Mo:W molar ratios and P2O5 contents: The effect of the composition and active phase morphology on the catalytic activity. React. Kinet. Mech. Cata. 2020, 129, 253–264. [Google Scholar] [CrossRef]
- Solntanali, S.; Mohaddesy, R.S.; Mashayekhi, M.; Rashidzadeh, M. Catalytic upgrading of heavy naphtha to gasoline: Simultaneous operation of reforming and desulfurization in the absence of hydrogen. J. Environ. Chem. Eng. 2020, 8, 104548. [Google Scholar] [CrossRef]
- Salim, M.A.-R.; Saed, U.A. Production of high-octane number gasoline from Basra low octane-number gas condensate and ethanol over modified Zn/ZSM-5 zeolite catalyst. Bull. Chem. React. Eng. Catal. 2023, 18, 59–70. [Google Scholar] [CrossRef]
- Kosinov, N.; Coumans, F.J.A.G.; Li, G.; Uslamin, E.; Mezari, B.; Wijpkema, A.S.G.; Pidko, E.A.; Hensen, E.J.M. Stable Mo/HZSM-5 methane dehydroaromatization catalysts optimized for high-temperature calcination-regeneration. J. Catal. 2017, 346, 125–133. [Google Scholar] [CrossRef]
- Gounder, R.; Iglesia, E. Catalytic hydrogenation of alkenes on acidic zeolites: Mechanistic connections to monomolecular alkane dehydrogenation reactions. J. Catal. 2011, 277, 36–45. [Google Scholar] [CrossRef]
- Standl, S.; Hinrichsen, O. Kinetic modeling of catalytic olefin cracking and methanol-to-olefins over zeolites: A review. Catalysts 2018, 8, 626. [Google Scholar] [CrossRef]
- Al Wasif-Ruiz, T.; Alvarez-Mateos, P.; Sanchez-Martín, J.A.; Guirado, M.; Barrios-Sánchez, C.C. Influence of fuel formulation on exhaust emissions from gasoline direct injection vehicle. Fuel Process. Technol. 2025, 272, 108215. [Google Scholar] [CrossRef]
- Akopyan, A.V.; Fedorov, R.A.; Andreev, B.V.; Tarakanova, A.V.; Anisimov, A.V.; Karakhanov, E.A. Oxidative desulfurization of hydrocarbon feedstock. Russ. J. Appl. Chem. 2018, 91, 457–471. [Google Scholar] [CrossRef]
- Kim, S.W.; Yeo, C.E.; Lee, D.Y. Effect of fines content on fluidity of FCC catalysts for stable operation of fluid catalytic cracking unit. Energies 2019, 12, 293. [Google Scholar] [CrossRef]
- Kong, J.; Dong, M.; Zhang, Z.; Yan, J.; Li, J.; Sun, L. Design and control of fluid catalytic cracking gasoline fractionator. Chem. Eng. Res. Des. 2024, 203, 663–687. [Google Scholar] [CrossRef]
- Li, J.; Xiong, L.; Ding, F.; Zhang, H.; Chen, X.F. Non-hydrogen catalytic dewaxing for oil production. Adv. Mater. Res. 2011, 1677, 418–420. [Google Scholar] [CrossRef]
- Otalvaro, N.D.; Sogne, G.; Delgado, K.H.; Wild, S.; Pitter, S.; Sauer, J. Kinetics of the direct DME synthesis from CO2 rich syngas under variation of the CZA-to-γ-Al2O3 ratio of a mixed catalyst bed. RSC Adv. 2021, 11, 24556–24569. [Google Scholar] [CrossRef]
- Tuktin, B.; Omarova, A.; Saidilda, G.; Nurzhanova, S.; Tungatarova, S.; Ongarbayev, Y. Modified polymetallic zeolite-based catalysts for hydroprocessing diesel oil fraction and tetradecane. Int. J. Technol. 2024, 15, 812–823. [Google Scholar] [CrossRef]
- Tuktin, B.; Saidilda, G.; Nurzhanova, S.; Ongarbayev, Y. Hydroprocessing of gasoline on modified alumina catalysts. Catalysts 2024, 14, 404. [Google Scholar] [CrossRef]
 , iso-alkanes—
, iso-alkanes— , olefins—
, olefins— , aromatic hydrocarbons—
, aromatic hydrocarbons— , naphthenic hydrocarbons—
, naphthenic hydrocarbons— , gas-phase yield—
, gas-phase yield— , liquid-phase yield—
, liquid-phase yield— .
.
 , iso-alkanes—
, iso-alkanes— , olefins—
, olefins— , aromatic hydrocarbons—
, aromatic hydrocarbons— , naphthenic hydrocarbons—
, naphthenic hydrocarbons— , gas-phase yield—
, gas-phase yield— , liquid-phase yield—
, liquid-phase yield— .
.
 , iso-alkanes—
, iso-alkanes— , olefins—
, olefins— , aromatic hydrocarbons—
, aromatic hydrocarbons— , naphthenic hydrocarbons—
, naphthenic hydrocarbons— , gas-phase yield—
, gas-phase yield— , liquid-phase yield—
, liquid-phase yield— .
.
 , iso-alkanes—
, iso-alkanes— , olefins—
, olefins— , aromatic hydrocarbons—
, aromatic hydrocarbons— , naphthenic hydrocarbons—
, naphthenic hydrocarbons— , gas-phase yield—
, gas-phase yield— , liquid-phase yield—
, liquid-phase yield— .
.
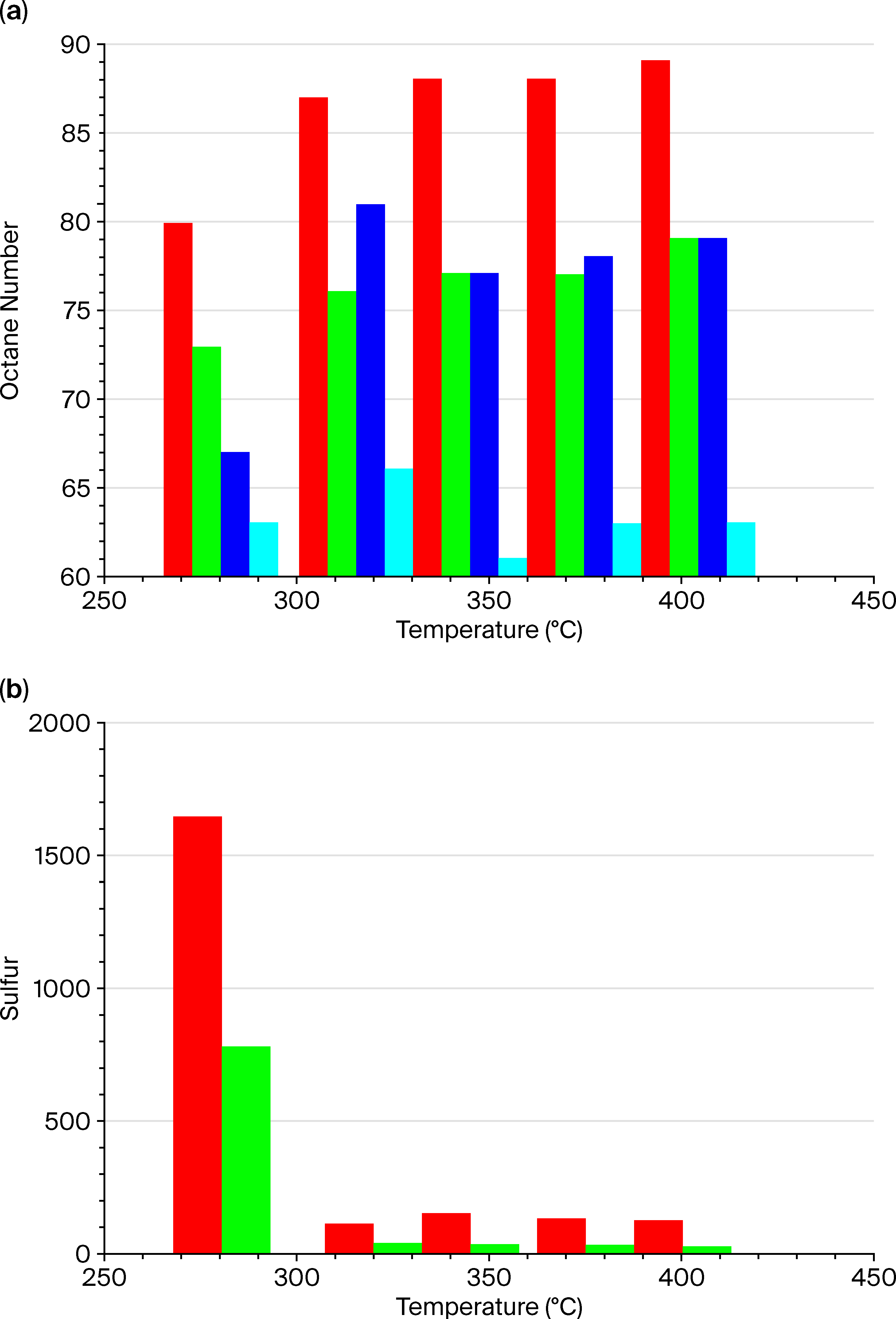
 , iso-alkanes—
, iso-alkanes— , olefins—
, olefins— , aromatic hydrocarbons—
, aromatic hydrocarbons— , naphthenic hydrocarbons—
, naphthenic hydrocarbons— , gas-phase yield—
, gas-phase yield— , liquid-phase yield—
, liquid-phase yield— .
.
 , iso-alkanes—
, iso-alkanes— , olefins—
, olefins— , aromatic hydrocarbons—
, aromatic hydrocarbons— , naphthenic hydrocarbons—
, naphthenic hydrocarbons— , gas-phase yield—
, gas-phase yield— , liquid-phase yield—
, liquid-phase yield— .
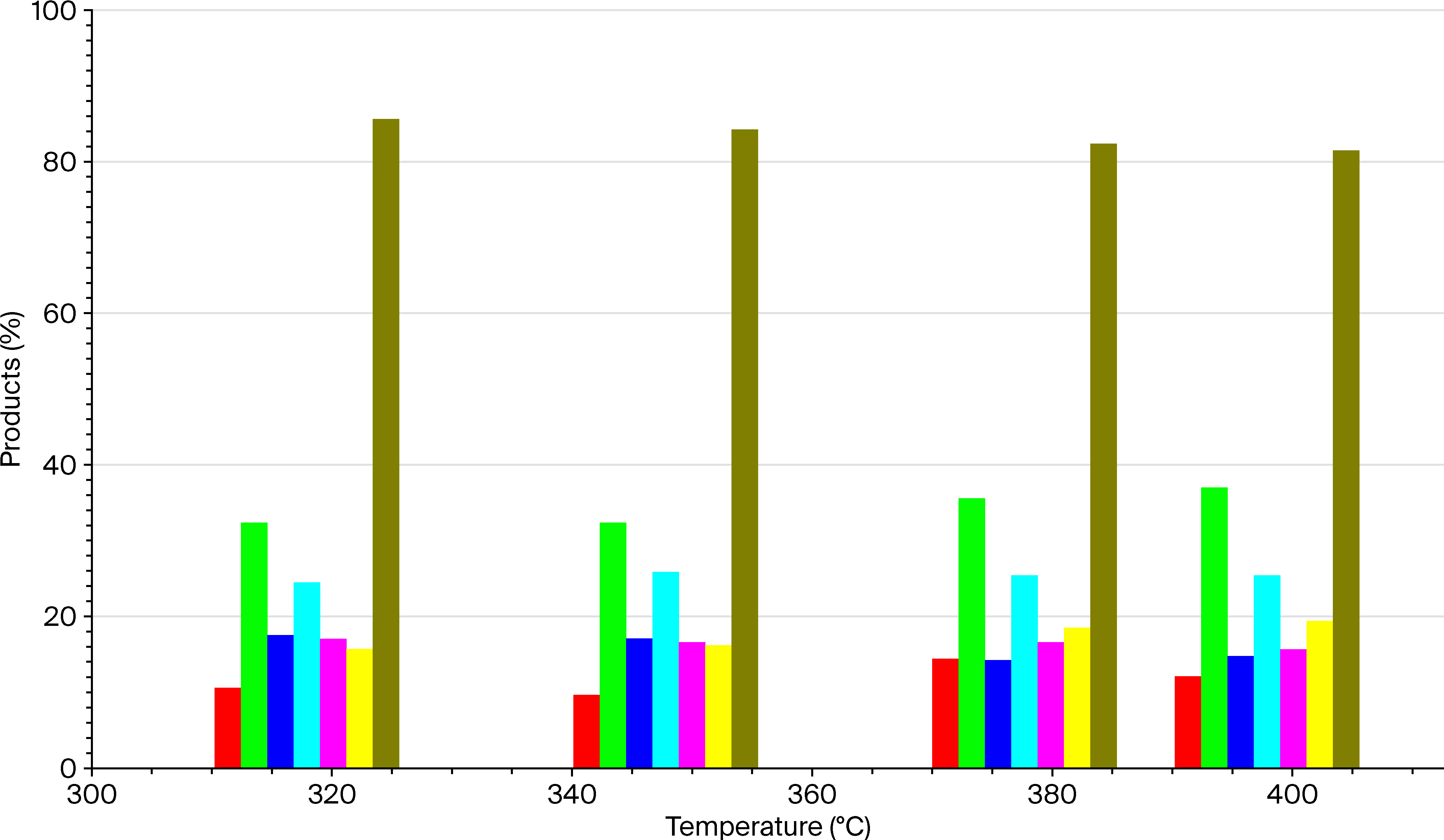
.




| Products (%) | Temperature (°C) | |||
|---|---|---|---|---|
| 320 | 350 | 380 | 400 | |
| Hexane | ||||
| Paraffins | 22.9 | 22.5 | 25.9 | 23.3 |
| Iso-alkanes | 24.2 | 19.1 | 20.7 | 16.6 |
| Olefins | 36.9 | 39.6 | 34.7 | 38.4 |
| Aromatic hydrocarbons | 6.7 | 6.1 | 5.3 | 6.4 |
| Naphthenic hydrocarbons | 9.3 | 12.7 | 13.4 | 15.3 |
| Gas-phase yield | 7.0 | 11.0 | 13.0 | 16.0 |
| Liquid-phase yield | 93.0 | 89.0 | 87.0 | 84.0 |
| Octane number according to the RM | 93.1 | 92.6 | 95.9 | 92.2 |
| Octane number according to the MM | 79.4 | 95.3 | 97.3 | 98.2 |
| Decane | ||||
| Paraffins | 98.1 | 97.5 | 95.1 | 93.6 |
| Iso-alkanes | 0.4 | 0.7 | 2.4 | 4.5 |
| Olefins | 0.6 | 0.7 | 1.1 | 1.0 |
| Aromatic hydrocarbons | 0.1 | 0.2 | 0.7 | 0.5 |
| Naphthenic hydrocarbons | 0.2 | 0.5 | 0.1 | 0.4 |
| Gas-phase yield | 12.0 | 16.0 | 15.0 | 17.0 |
| Liquid-phase yield | 88.0 | 84.0 | 85.0 | 83.0 |
| Octane number according to the RM | 76.6 | 89.4 | 90.1 | 89.9 |
| Octane number according to the MM | 51.8 | 95.3 | 97.3 | 98.2 |
| Products (%) | Temperature (°C) | ||||
|---|---|---|---|---|---|
| Original Gasoline | 320 | 350 | 380 | 400 | |
| Paraffins | 28.4 | 33.9 | 24.8 | 18.3 | 15.2 |
| Iso-alkanes | 35.0 | 33.5 | 31.4 | 34.8 | 35.2 |
| Olefins | 4.7 | 6.5 | 5.3 | 6.4 | 7.9 |
| Aromatic hydrocarbons | 5.7 | 6.1 | 7.8 | 8.3 | 9.8 |
| Naphthenic hydrocarbons | 26.2 | 19.9 | 30.7 | 32.2 | 34.4 |
| Gas-phase yield | 11.0 | 14.0 | 15.0 | 18.0 | |
| Liquid-phase yield | 89.0 | 86.0 | 85.0 | 82.0 | |
| Octane number according to the RM | 66.8 | 80.9 | 77.3 | 78.5 | 79.1 |
| Octane number according to the MM | 62.6 | 65.8 | 61.3 | 62.7 | 63.0 |
| Mass fraction of sulfur (ppm) | 776 | 354 | 283 | 248 | 253 |
| Products (%) | Temperature (°C) | ||||
|---|---|---|---|---|---|
| Original Gasoline | 320 | 350 | 380 | 400 | |
| Paraffins | 6.2 | 10.2 | 9.5 | 9.4 | 8.9 |
| Iso-alkanes | 53.7 | 31.9 | 32.0 | 35.2 | 36.3 |
| Olefins | 10.6 | 17.1 | 16.9 | 14.0 | 14.6 |
| Aromatic hydrocarbons | 7.2 | 24.3 | 25.5 | 25.1 | 25.2 |
| Naphthenic hydrocarbons | 22.6 | 16.5 | 16.1 | 16.3 | 15.0 |
| Gas-phase yield | 15.0 | 16.0 | 18.0 | 19.0 | |
| Liquid-phase yield | 85.0 | 84.0 | 82.0 | 81.0 | |
| Octane number according to the RM | 80.4 | 87.3 | 88.5 | 87.8 | 88.9 |
| Octane number according to the MM | 73.5 | 75.6 | 76.7 | 77.1 | 78.9 |
| Mass fraction of sulfur (ppm) | 1650.0 | 1178 | 1540 | 1280 | 1230 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nurzhanova, S.B.; Saidilda, G.T.; Nurlan, A.; Abilmagzhanov, A.Z.; Nagashybayeva, A.S.; Tungatarova, S.A. New Polyfunctional Nanocatalysts for the Hydrogen-Free Processing of N-Alkanes and Gasoline Fractions. Processes 2025, 13, 3841. https://doi.org/10.3390/pr13123841
Nurzhanova SB, Saidilda GT, Nurlan A, Abilmagzhanov AZ, Nagashybayeva AS, Tungatarova SA. New Polyfunctional Nanocatalysts for the Hydrogen-Free Processing of N-Alkanes and Gasoline Fractions. Processes. 2025; 13(12):3841. https://doi.org/10.3390/pr13123841
Chicago/Turabian StyleNurzhanova, Saule B., Galymzhan T. Saidilda, Annas Nurlan, Arlan Z. Abilmagzhanov, Aizada S. Nagashybayeva, and Svetlana A. Tungatarova. 2025. "New Polyfunctional Nanocatalysts for the Hydrogen-Free Processing of N-Alkanes and Gasoline Fractions" Processes 13, no. 12: 3841. https://doi.org/10.3390/pr13123841
APA StyleNurzhanova, S. B., Saidilda, G. T., Nurlan, A., Abilmagzhanov, A. Z., Nagashybayeva, A. S., & Tungatarova, S. A. (2025). New Polyfunctional Nanocatalysts for the Hydrogen-Free Processing of N-Alkanes and Gasoline Fractions. Processes, 13(12), 3841. https://doi.org/10.3390/pr13123841







