Abstract
Utilizing bio-based nano-fuel additives presents a promising path towards improved engine performance and reduced emissions. The response surface method is used in this investigation to predict and optimize the performance parameters and exhaust emissions of a single-cylinder diesel engine. The engine operates with B20 at 2900, 3100, and 3300 rpm and nanobiochar concentrations of 30, 60, and 90 ppm. The results showed a declining trend in all of the engine-out emissions when the nanobiochar additive was used. Based on the optimization results, an engine speed of 3108 rpm and a nanobiochar ratio of 90 ppm were found to be the optimal conditions within the defined range of the input parameters. At this point, the engine power, torque, BSFC, and emissions of NOx, CO, and UHC were measured at 5.78 kW, 17.96 Nm, and 309 g/kW·h, 104.9 ppm, 1.25 (%Vol.), and 104.9 ppm, respectively. These values represent significant improvements compared to the baseline B20 fuel. The modeling results showed that RSM could effectively predict engine performance and emissions when running on a green-based fuel like B20, with a 90 ppm nanobiochar additive.
1. Introduction
Since 1950, the global population has more than tripled, reaching 7.6 billion people, and is expected to grow to 9.6 billion by 2050 [1]. This rapid population growth has intensified the demand for energy, where the world’s primary energy supply is still derived from non-renewable resources, such as oil, gas, and coal [2]. However, the depletion of these resources, coupled with environmental concerns, has driven global efforts to pursue of sustainable energy solutions, with biofuels emerging as a significant alternative. Among these, renewable and clean biofuels derived from plant and animal sources have attracted considerable attention. These cleaner-burning fuels are a crucial part of the energy blend in many countries due to their reduced emissions [3]. Compared to other biofuels, biodiesel has made the most progress. Vegetable oils, waste cooking oils, animal fats, and algae are all sources of the long-chain fatty acids that produce biodiesel’s monoalkyl esters. The continuous replenishment of biofuels eliminates concerns about the depletion of traditional fuels [4]. Despite the promising environmental benefits of biodiesel, several limitations hinder its widespread adoption as a mainstream fuel alternative. Biodiesel is known to have high viscosity, which can lead to poor fuel atomization and incomplete combustion in engines. Moreover, the use of biodiesel can increase of nitrogen oxide (NOx) emissions, posing a challenge for meeting stringent emission standards. Other concerns include storage instability, as biodiesel can degrade over time, leading sedimentation and the formation of acids that affect fuel quality. These challenges highlight the need for performance-enhancement strategies, such as the use of nano-additives, to improve the combustion efficiency, reduce emissions, and enhance the overall sustainability of biodiesel as an alternative fuel [5]. Building on this need, Consequently, significant efforts have been made to improve biodiesel properties, including the use of fuel additives to enhance combustion of both diesel and biodiesel. The concept of utilizing nanoparticles as fuel additives aligns with the promise of driving technological innovation and significant advancements, as evidenced by recent research exploring the effects of nanoparticle-based additives on engine performance and emissions [5,6]. Recent research highlights the use of a variety of nanocatalysts, including aluminum oxide (Al2O3), copper oxide (CuO), cerium oxide (CeO2), iron oxide (Fe2O3), and titanium dioxide (TiO2), in diesel and biodiesel fuel mixtures to improve combustion efficiency and lower emissions [7,8]. The stability of nanoparticles in fuel blends, particularly their impact on lubrication and fuel properties, is a primary concern when using them as fuel additives [9].
To address these concerns and promote sustainability, researchers have increasingly turned to biological and bio-based nanoparticles as alternatives. Metallic nanoparticles are made of metals (e.g., gold, silver), non-metallic nanoparticles are composed of non-metals (e.g., carbon, polymers), catalytic nanoparticles use their surface to speed up chemical reactions, and bio-based nanoparticles are derived from or synthesized using biological matter. The classifications are not mutually exclusive, as a nanoparticle can be both metallic and catalytic or both bio-based and non-metallic [10]. Biological nanoparticles are naturally occurring or biogenic nanoparticles with sizes between 1 and 100 nm, including viruses, vesicles, and protein-based structures. They are diverse, biocompatible, and have myriad applications in medicine for diagnosis, drug delivery, and as vaccine platforms, as well as being synthesized through “green” methods using plants and microorganisms [11]. Adding biological nanoparticles to diesel fuel can improve the combustion process [12]. Thermochemical conversion of biomass produces biochar, a carbon-based material. This biochar, when processed into nanobiochar, offers a renewable and eco-friendly option for fuel enhancement.
In recent years, there has been growing interest in using biochar derived from agricultural residues as an additive to fuel blends. Among various biomass materials, walnut shells have emerged as a significant source due to their abundance, cost-effectiveness, and distinctive characteristics. Walnut shell biochar is particularly beneficial due to its large surface area and porous structure, which makes it effective in improving engine performance and reducing pollutant emissions when incorporated into biodiesel mixtures. Moreover, the use of walnut shell biochar as a renewable fuel additive offers an eco-friendly solution to decrease engine emissions while simultaneously improving overall engine performance. Its unique catalytic and adsorptive properties make walnut shell biochar a superior alternative to conventional metallic nanoparticles [13].
Adding biocatalysts to biodiesel enhances specific physicochemical properties of the fuel blend, resulting in improved engine performance and reduced diesel engine exhaust emissions [14]. Recently, developed countries have used nanobiochar as an additive in internal combustion engines to evaluate performance and environmental emissions. Nanobiochar shows excellent promise as a substitute for conventional metal fuel additives, as the addition of nanoparticles to biodiesel reduces greenhouse gas emissions and increases combustion efficiency in engines [12]. Biochar is mainly produced from inexpensive sources like plant waste, animal dung, and sewage sludge [15]. Iran’s energy production does not typically use these residues [16]. Due to its carbon content and low cost, nanobiochar is a promising, cost-effective organic nano-additive for diesel engines.
Additionally, converting plant residues into nanobiochar offers an environmentally friendly method for managing plant-derived biomass [17]. Adding biochar to diesel and biodiesel blends as a renewable fuel additive is beneficial because it enables the use of new, cost-effective, and renewable diesel engines [18]. Biochar is widely used in various environmental applications, including solid waste management, soil improvement, water purification, and carbon capture [19].
Researchers have conducted studies on nanobiochar as an additive for internal combustion engines. The performance and emissions of a single-cylinder diesel engine were studied using a fuel blend comprising diesel, biodiesel, ethanol, and a nanobiochar additive. The results showed that incorporating nanobiochar into the diesel–biodiesel-ethanol blend improved engine performance and reduced exhaust emissions. The optimal parameters obtained were 7.7 Nm for engine torque, 7.1 kW for power, 219 g/kWh for brake-specific fuel consumption (BSFC), 184 ppm for NOx, and 0.011% for CO [20]. Research has shown that walnut shell biochar can enhance the physical and chemical properties of fuel blends, thereby increasing combustion efficiency and reducing emissions of pollutants such as carbon monoxide (CO) and nitrogen oxides (NOx). For instance, a study conducted in Iran demonstrated that biochar derived from walnut shells significantly reduces CO and NOx emissions when used as a fuel additive [21]. This makes walnut shell biochar a promising candidate for enhancing the sustainability of biofuel applications [22]. In a study by Mofijur et al. (2024) [5], the effect of nanoparticle-based fuel additives, including nanobiochar, on the performance of biodiesel engines was investigated. The results showed a 7.1% increase in engine power and a 6.4% reduction in brake-specific fuel consumption (BSFC) when nanobiochar was added to biodiesel. Furthermore, nanobiochar reduced NOx emissions by 12% and CO emissions by 10%, demonstrating its potential to enhance engine efficiency and reduce harmful emissions [5]. In the study conducted by Le et al. (2024) [23], the role of nanobiochar in biodiesel production and its effects on diesel engine performance were explored. The findings revealed that adding nanobiochar increased fuel efficiency by 8%, reduced NOx emissions by 14%, and reduced CO emissions by 9%. The study concluded that nanobiochar can significantly improve both fuel properties and engine performance while minimizing environmental impacts [23]. In another study, Ardabili et al. (2020) [24], investigated the impact of nanobiochar derived from sugarcane particles as an additive to a diesel fuel blend on engine performance and greenhouse gas emissions. Engine performance variables (power, torque, and brake-specific fuel consumption) and exhaust emissions (NOx, UHC, and CO) showed statistically significant differences. Best performance was achieved with 8.1 kW of power, 7.7 N.m of torque, and 227 g/kWh of brake-specific fuel consumption. NOx, CO, and UHC exhaust emissions were optimally measured at 217 ppm, 0.01%, and 30 ppm. Adding nanobiochar resulted in a 20.51% decrease in NOx emissions, a 25% reduction in UHC emissions, and a 33% increase in CO emissions [24]. The impact of Aegle marmelos (bael tree) nanobiochar-enhanced diesel fuel on a single-cylinder diesel engine’s combustion, performance, and emissions was examined by researchers. Adding nanobiochar to the diesel fuel blend reduced CO emissions by 27.57%, HC emissions by 19.11%, and increased CO2 emissions by 18.46% [25]. Ramachandran et al. (2021) [26] used nanobiochar at concentrations of 25 and 50 ppm as an additive in biodiesel derived from grape seeds. The results showed reductions of 2.2%, 2.8%, and 3.5% in hydrocarbon, carbon monoxide, and soot emissions, respectively, while NO emissions increased [26].
Despite these advancements, a key gap remains in exploring modified forms of nanobiochar to further optimize biodiesel blends. This study aims to investigate the use of amine-modified nanobiochar as an additive in diesel and biodiesel blends to enhance engine performance and reduce harmful emissions. The primary objective is to evaluate the impact of amine-modified nanobiochar on key engine parameters—power output, torque, and emissions—and to identify the optimal nanobiochar concentration for improved fuel efficiency and environmental performance. The novelty of this research lies in its unique integration of amine modification with nanobiochar derived specifically from walnut shells, which has not been extensively studied in diesel–biodiesel blends. This approach provides a sustainable, low-cost alternative to metallic nanoparticles by leveraging amine groups to enhance dispersion stability and catalytic activity, potentially addressing limitations in previous nanobiochar applications such as inconsistent emission reductions. The contribution of this study to the existing literature is significant. While previous studies have examined the use of biochar and nanoparticles in fuel additives, this research uniquely combines amine-modified nanobiochar derived from walnut shells, offering a sustainable and cost-effective alternative to traditional metallic nanoparticles. Additionally, this study explores the application of amine-modified nanobiochar in a diesel–biodiesel blend, a topic that has not been thoroughly investigated in earlier research. The findings will provide valuable insights into the potential of amine-modified nanobiochar as a multifunctional additive for improving both engine efficiency and environmental sustainability.
2. Materials and Methods
Iranian gas station diesel and Tarbiat Modares University’s (Tehran) Renewable Energy Research Institute’s waste cooking oil-derived biodiesel were used in this research. Waste cooking oil biodiesel was produced via transesterification process, in which waste cooking oil reacts with methanol in the presence of potassium hydroxide on a batch scale. The production was performed at 60 °C and a 6:1 molar ratio under air pressure. The resulting biodiesel was tested to meet international fuel standards and the purity was reported as 98%. For biochar preparation, walnut shells were selected as the raw material. The shells were washed with deionized water to remove impurities and dried in the sun at 25 °C for three days. After drying, the shells were gently crushed and sieved through a 1 mm mesh to ensure uniform particle size. The biochar was then produced from the dried walnut shells using a slow pyrolysis process in the absence of air (oxygen) in a furnace (2 h at 600 °C with a heating rate of 5 °C per min).
2.1. Functionalization of BIOCHAR
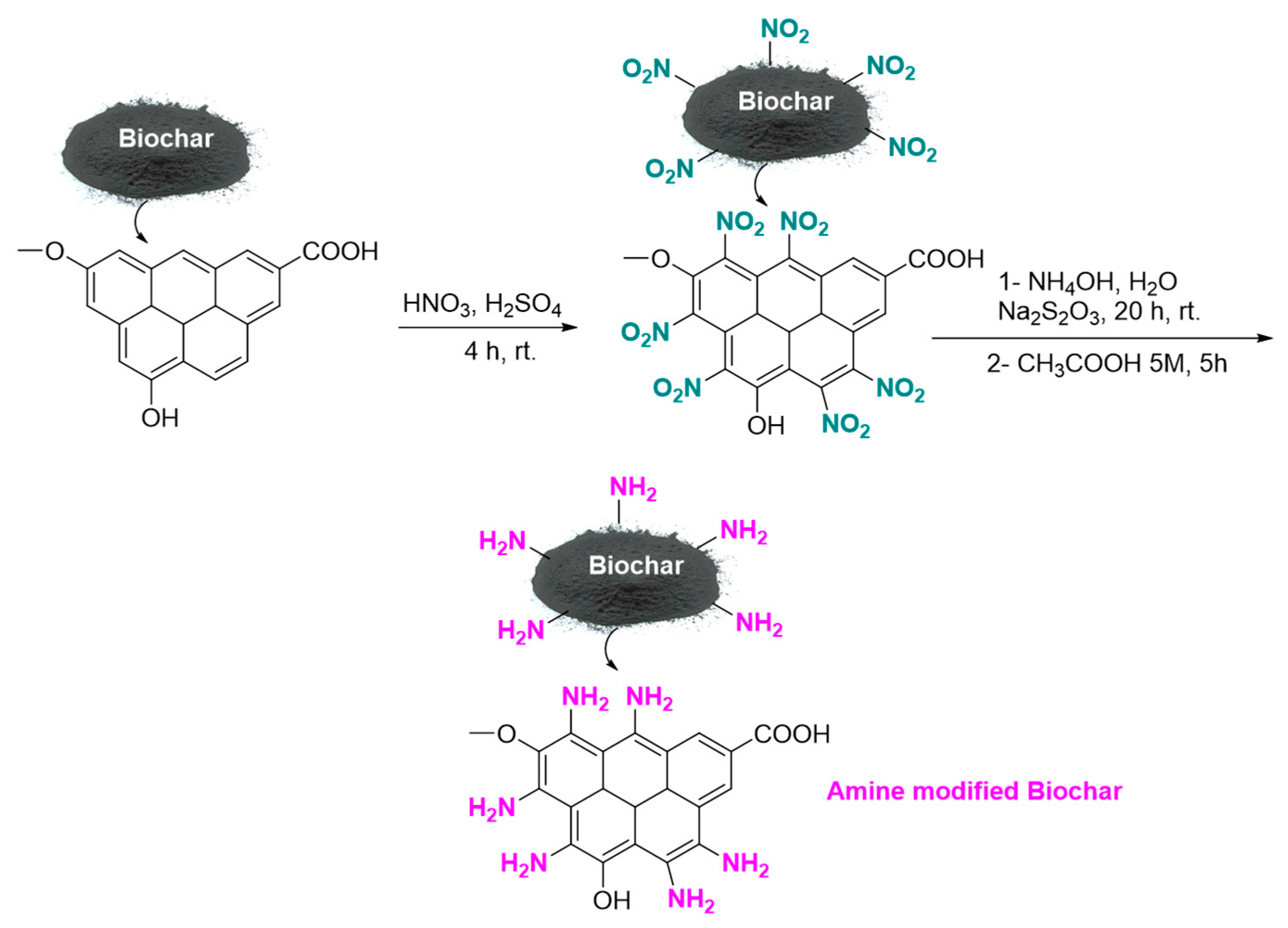
To prepare basic functionalized biochar, 1 g of walnut shell biochar was mixed with 30 mL of 60% nitric acid and 20 mL of concentrated sulfuric acid for 4 h at room temperature in a sealed glass vial on a stirrer. Filtration separated the biochar-NO2, followed by multiple distilled-water washes and a 24 h drying period at room temperature. Next, 10 mL of water and 5 mL of ammonia were added to 1 g of biochar-NO2 in a sealed glass vial; this mixture was stirred for 10 min at room temperature. Then, 8.5 g of sodium thiosulfate were added and the mixture was stirred for 20 h at room temperature in a sealed glass vial. Finally, 20 mL of 5 M acetic acid was added to the reaction mixture, which was stirred under reflux for 5 h. Room-temperature drying for 24 h, followed by filtration and several distilled-water washes of the resulting basic biochar and amine-modified nanobiochar. This experiment utilized amine-modified Nanobiochar, as shown in Figure 1.
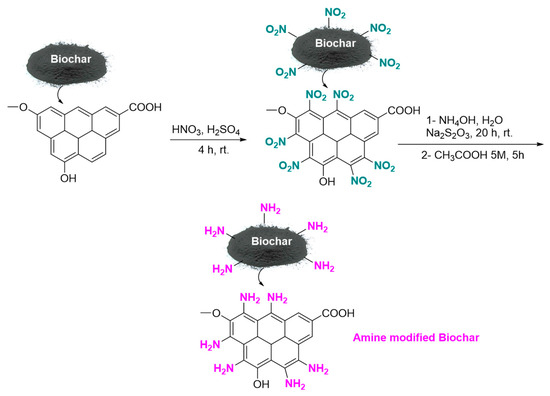
Figure 1.
Synthesis of Amine modified Nanobiochar.
2.2. Preparation of Fuel Blends
After preparing the diesel, biodiesel, and amine-modified biochar, the fuel blends were prepared according to the specified volume percentages and ratios. To ensure long-term stability, nano-fuels often require surfactants to reduce surface tension and prevent agglomeration. Typical surfactants include oleic acid, Span 80, and Triton X-100, typically used at concentrations of 1–5 wt%. The blending procedure usually involves high-shear mixing or ultrasonication. For instance, a nano-fuel might be prepared by dispersing nanoparticles in a base fuel with 2 wt% oleic acid under ultrasonication for 30 min at room temperature. Stability is then assessed via zeta potential measurements, where values above +30 mV or below −30 mV generally indicate good stability due to electrostatic repulsion. Alternatively, sedimentation tests can be performed by observing the fuel over time (e.g., 24 h, 1 week, 1 month) and noting any settling of nanoparticles. Reproducibility is verified by repeating the blending procedure multiple times and confirming consistent zeta potential values and sedimentation behavior.
In this study, the density and viscosity of the fuel blends were measured. The density was measured using a Density-Meter (Model CY-001, Code 87643), manufactured by Chenaryaz Ebtekar Industry, Tabriz, Iran, for both homogeneous and heterogeneous liquids. Viscosity was measured using an optical viscometer (Model OVM-10, Code 100200), designed by Nano Engineering Zhikan Company, Tehran, Iran. Table 1 lists the composition and selected physicochemical properties of the fuel samples used in this study. Density and viscosity are essential for understanding the overall behavior of fuel in the engine, and their optimization can enhance both fuel efficiency and emissions control. The correct balance of density and viscosity in the fuel blend is critical for achieving high combustion efficiency while minimizing environmental impact [27,28].

Table 1.
Fuel composition and main physicochemical properties.
In Table 1, the letter “B” represents biodiesel; the first number indicates the percentage of biodiesel, and the second number indicates the amount of Amine-modified Biochar added to fuel blends. For instance, “B20-30” represents a fuel blend containing 20% biodiesel, 80% diesel, and 30 ppm nano-amine-modified biochar as an additive.
2.3. Engine Setup and Testing Procedure
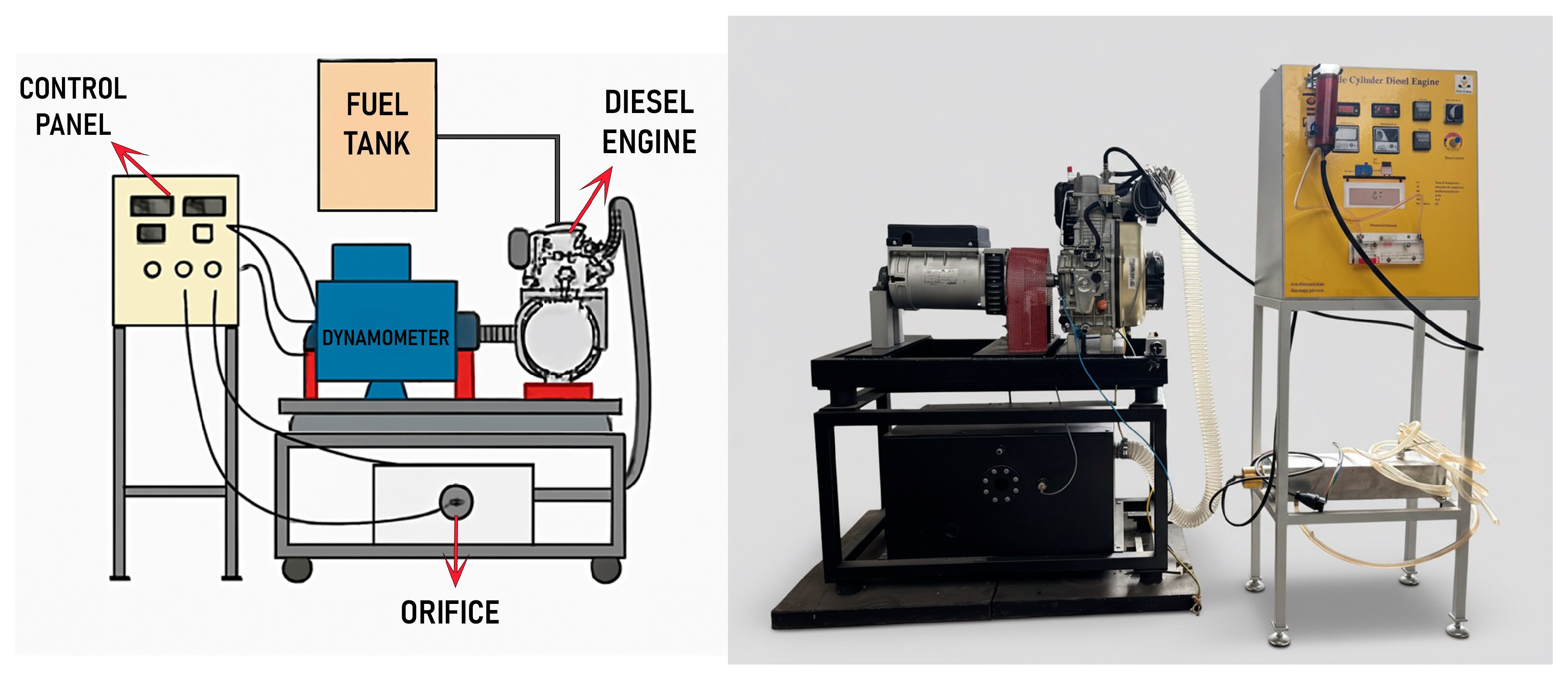
In this research, a single-cylinder air-cooled engine available in the Renewable Energy Laboratory of the Faculty of Agriculture at Bu-Ali Sina University was used for the experiments. In this experiment, a dynamometer is used to measure the diesel engine’s power and torque output. The system is equipped with a load cell that measures force and calculates torque. The dynamometer is coupled to the engine via a standard mechanical coupling process, with a torque of 21 ± 7% Nm applied to the fasteners. The dynamometer itself meets industry standards, with a protection grade of IP23, ensuring reliability across various testing environments. The generator used in the system is a brushless, single-phase alternator from the S16F series, which is known for its high reliability and low maintenance requirements. It operates at 3600 RPM for 60 Hz or 3000 RPM for 50 Hz and has a nominal power output range of 5.5 to 6.5 kVA, with efficiency reaching up to 79.7%. The alternator features H-class insulation and is designed for operation in environments with a maximum temperature of 40 °C and altitudes below 1000 m. This dynamometer setup allows accurate measurements of engine power, torque, and airflow under different load conditions. The system provides critical data for evaluating engine performance, ensuring the testing process meets the required accuracy and safety standards.
The engine tests were conducted at full load and different engine speeds (2900, 3100, and 3300 rpm). The engine setup is shown in Figure 2, while Table 2 details its specifications.
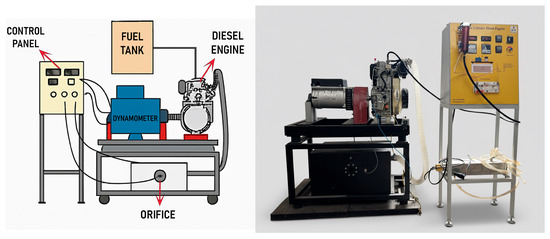
Figure 2.
Engine setup.

Table 2.
Engine Specifications.
To measure the exhaust emissions, an AIRREX HG-550 gas analyzer, HEPHZIBAH Co., Ltd., Incheon, South Korea, was used (Figure 3). The measurement range, units, and accuracy of the device are provided in Table 3.

Figure 3.
Gas analyzer.

Table 3.
Specifications of the gas analyzer.
2.4. Field Emission Scanning Electron Microscopy (FESEM)
The physical characteristics and morphological structure of the amine-modified biochar were studied using field emission scanning electron microscopy (FESEM). FESEM images are displayed in Figure 4. The amine-modified biochar exhibited a rough surface structure, good shape homogeneity, and an average size of less than 100 nm, with various macro- and micropores. These features offer abundant adsorption cavities and spaces.

Figure 4.
FESEM images of Amine modified Nanobiochar as a basic additive.
2.5. Energy-Dispersive X-Ray Spectroscopy Analysis
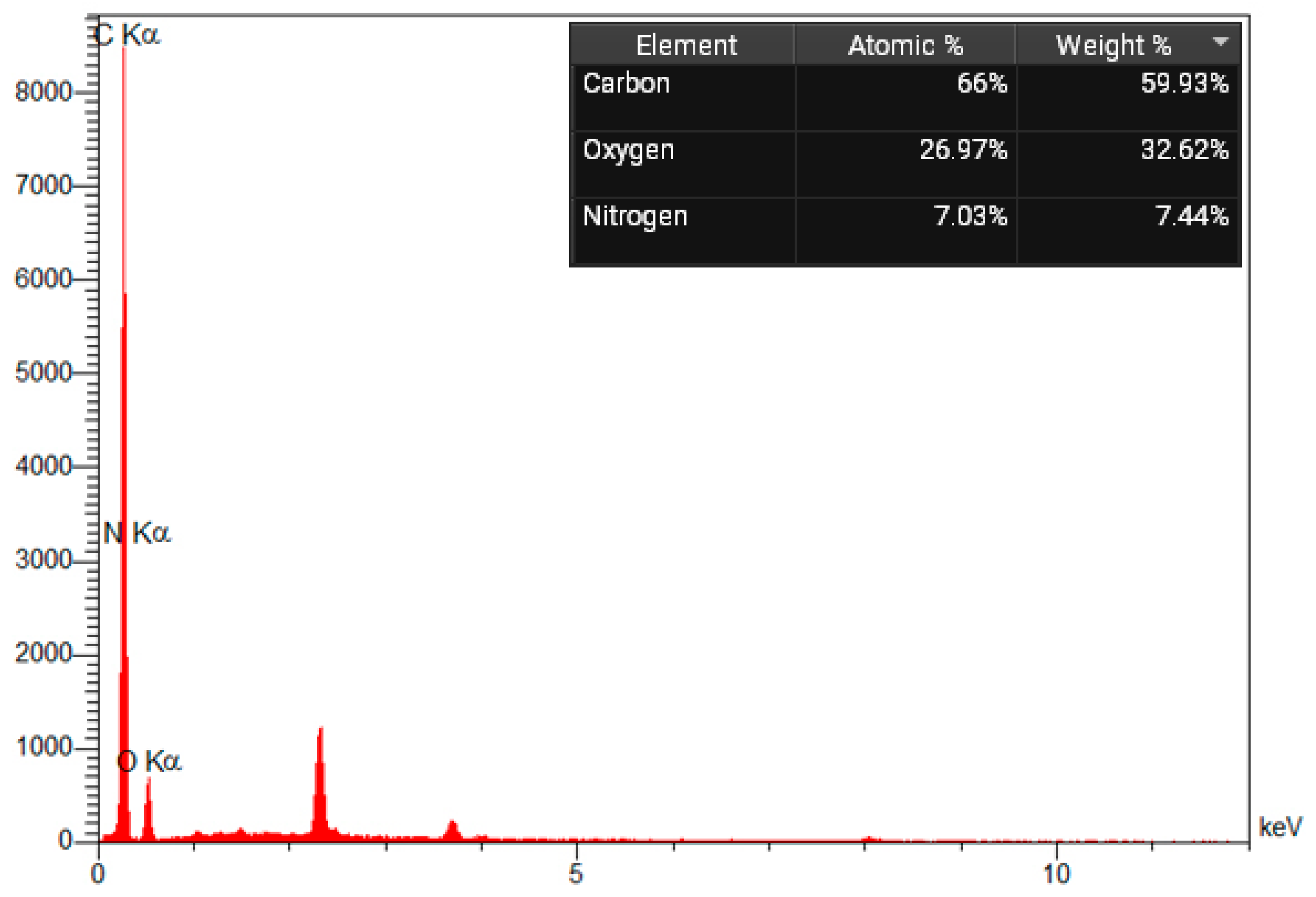
As indicated by energy-dispersive X-ray spectroscopy (EDS) analysis, the element content of amine-modified biochar is composed of oxygen, nitrogen, and carbon. The EDS result shows good agreement with this structure. Additionally, the mass percentages of C, N, and O are 59.93%, 7.44%, and 32.62%, respectively (Figure 5). A nitrogen (N) peak in an Energy Dispersive X-ray Spectroscopy (EDS) spectrum corresponds to the emission of characteristic X-rays from nitrogen atoms, specifically the N K-alpha line at approximately 392 eV. However, because nitrogen is a light element, its peak is often weak, found in the low-energy region, and can be challenging to detect due to overlap with the carbon (C) peak (around 277 eV) and absorption by the detector window and sample. Gold exhibits peaks at approximately 2.2 and 3.7 keV.
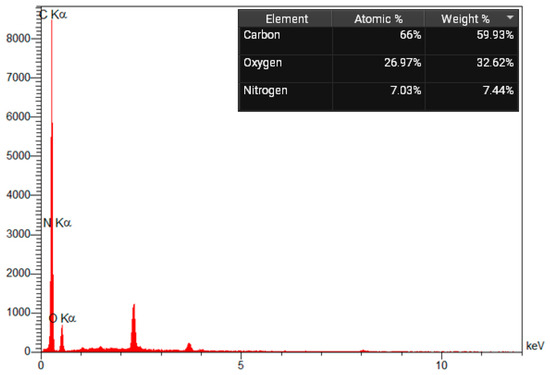
Figure 5.
EDX spectrum of Amine modified Biochar additive.
2.6. BET Analysis (Surface Area and Porosity Analysis)
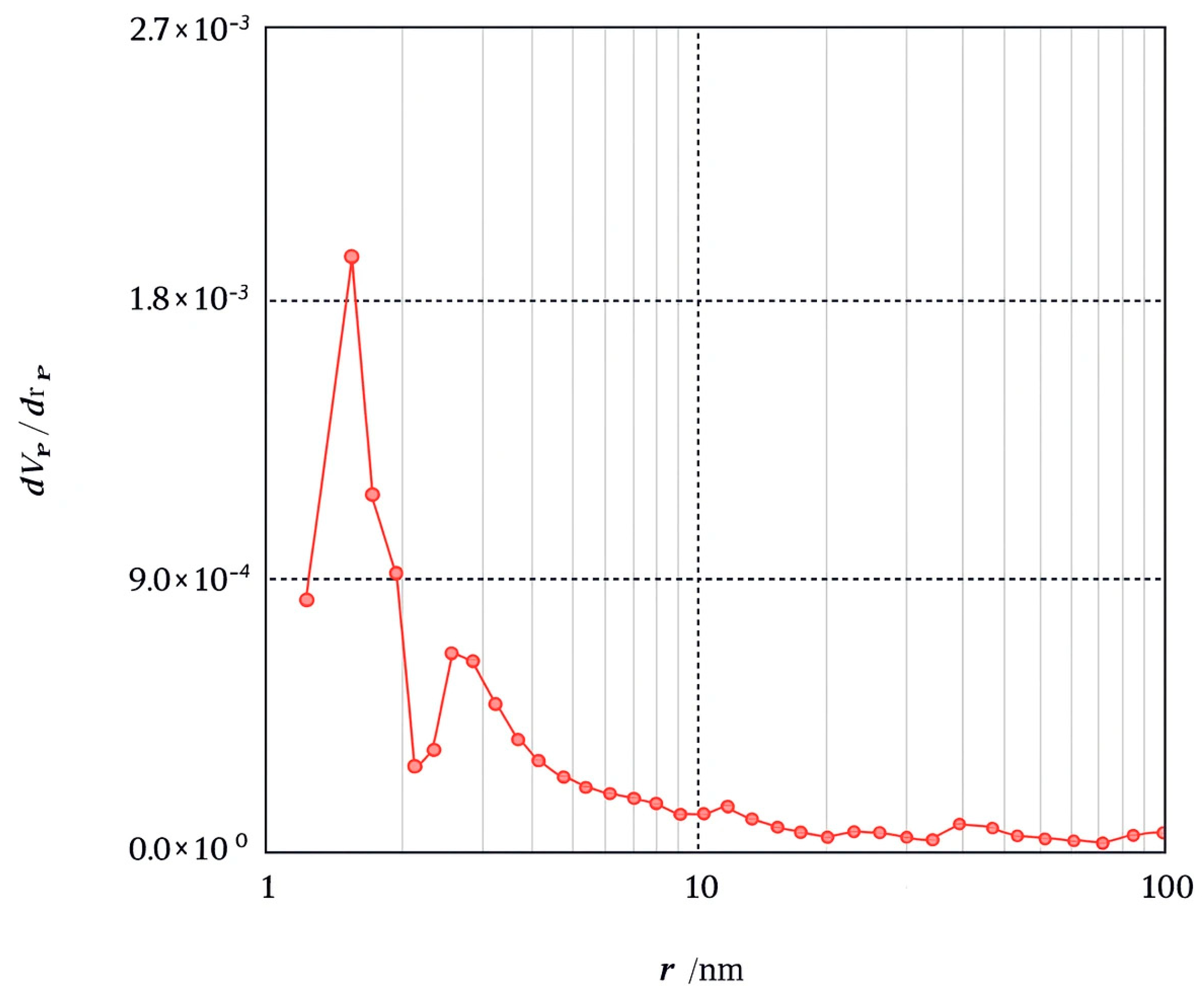
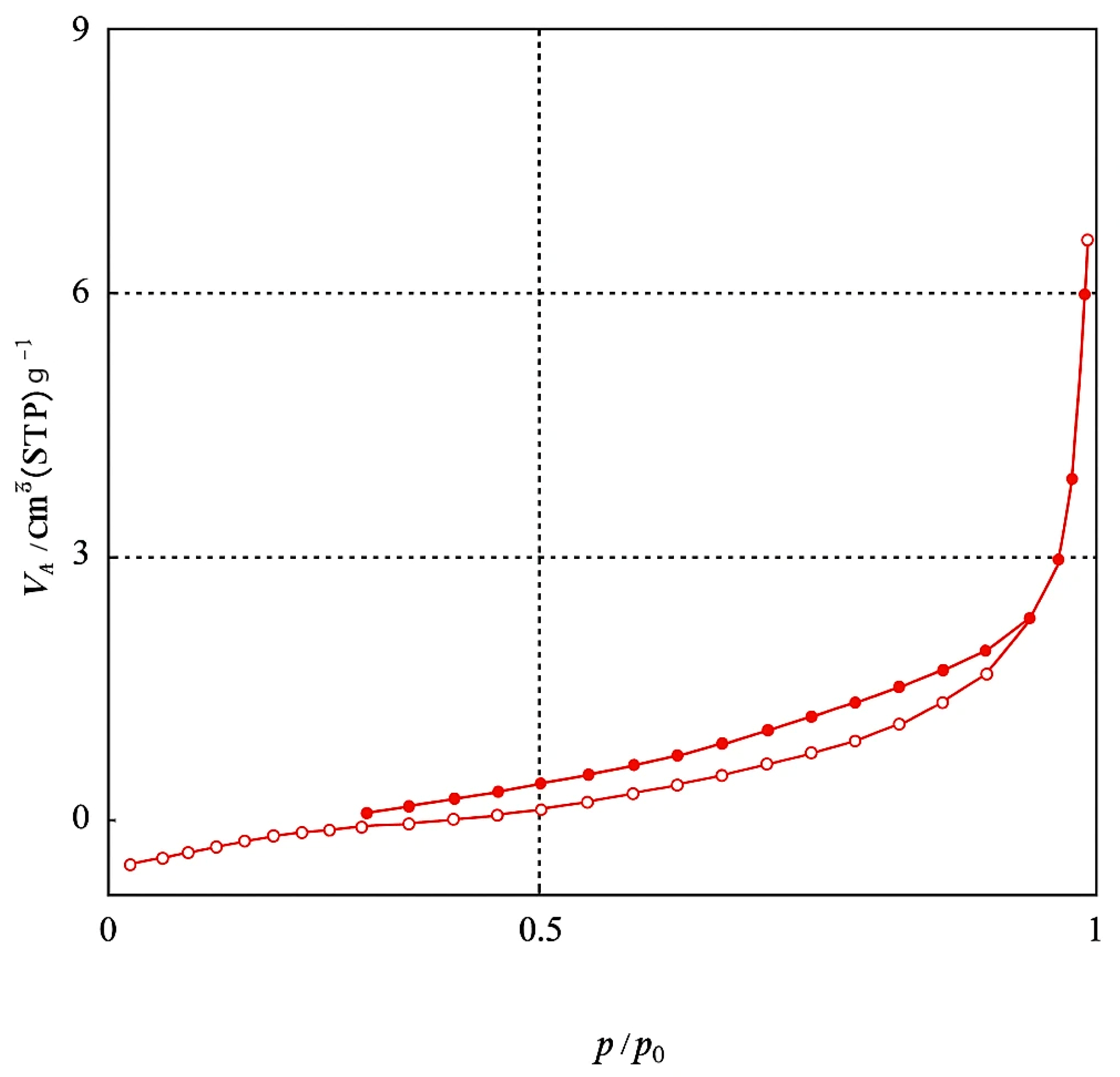
The BET method is commonly used to measure the surface area of an adsorbent by utilizing N2 gas as the adsorbate on the material’s surface. Adsorption characterization of Amine-modified Biochar nanoparticles derived from walnut shells at a pyrolysis temperature of 600 °C was analyzed through N2 adsorption at 77.35 K over a relative pressure (P/P0) range of 0.0–1. Modified biochar sample pyrolyzed at 600 °C for 2 h showed a specific surface area of 2.77 m2/g, pore volumes of 0.0095 cm3/g, and an average pore diameter of 13.68 nm, according to Brunauer–Emmett–Teller (BET) analysis. The BJH method was used to determine the pore-size distribution of the adsorbent. Figure 6 illustrates the pore size distribution curve for the Amine modified Biochar nanoparticles sample. The curve depicts pore size distribution in the micropore (<2 nm), mesopore (2–50 nm), and macropore regime (>50 nm). The analyzed sample displays sizes ranging from 1.2 to 100 nm, distributed mainly in the mesoporous regime. The nitrogen adsorption–desorption isotherm of Amine-modified Biochar nanoparticles is depicted in Figure 7, illustrating that the adsorbent exhibits a type IV isotherm with an H3 hysteresis loop across a broad P/P0 range of 0.02 to 0.99, indicative of a mesoporous structure [29].
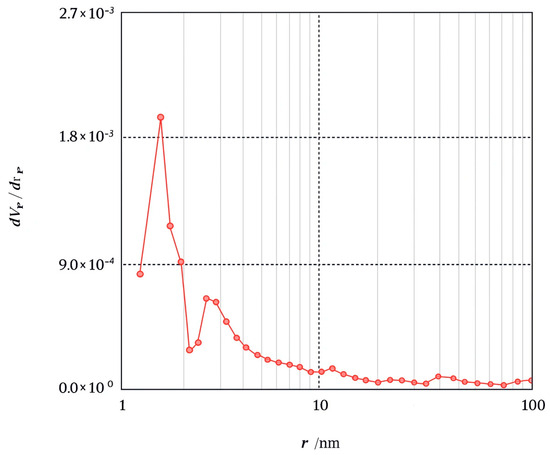
Figure 6.
BJH adsorption dV/dr Pore volume distribution for Amine modified Nanobiochar.
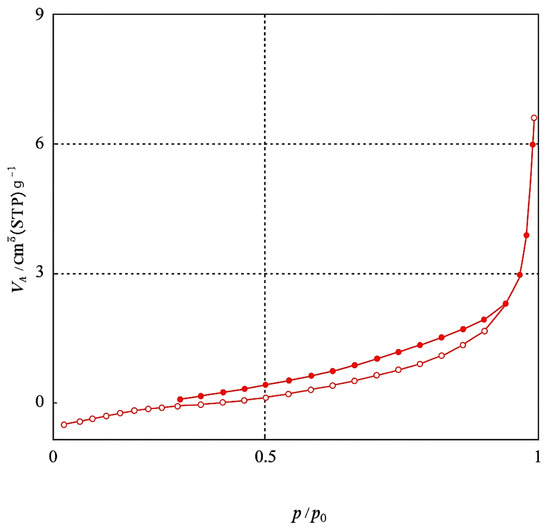
Figure 7.
Nitrogen adsorption isotherms for Amine modified Nanobiochar.
2.7. FTIR Spectra
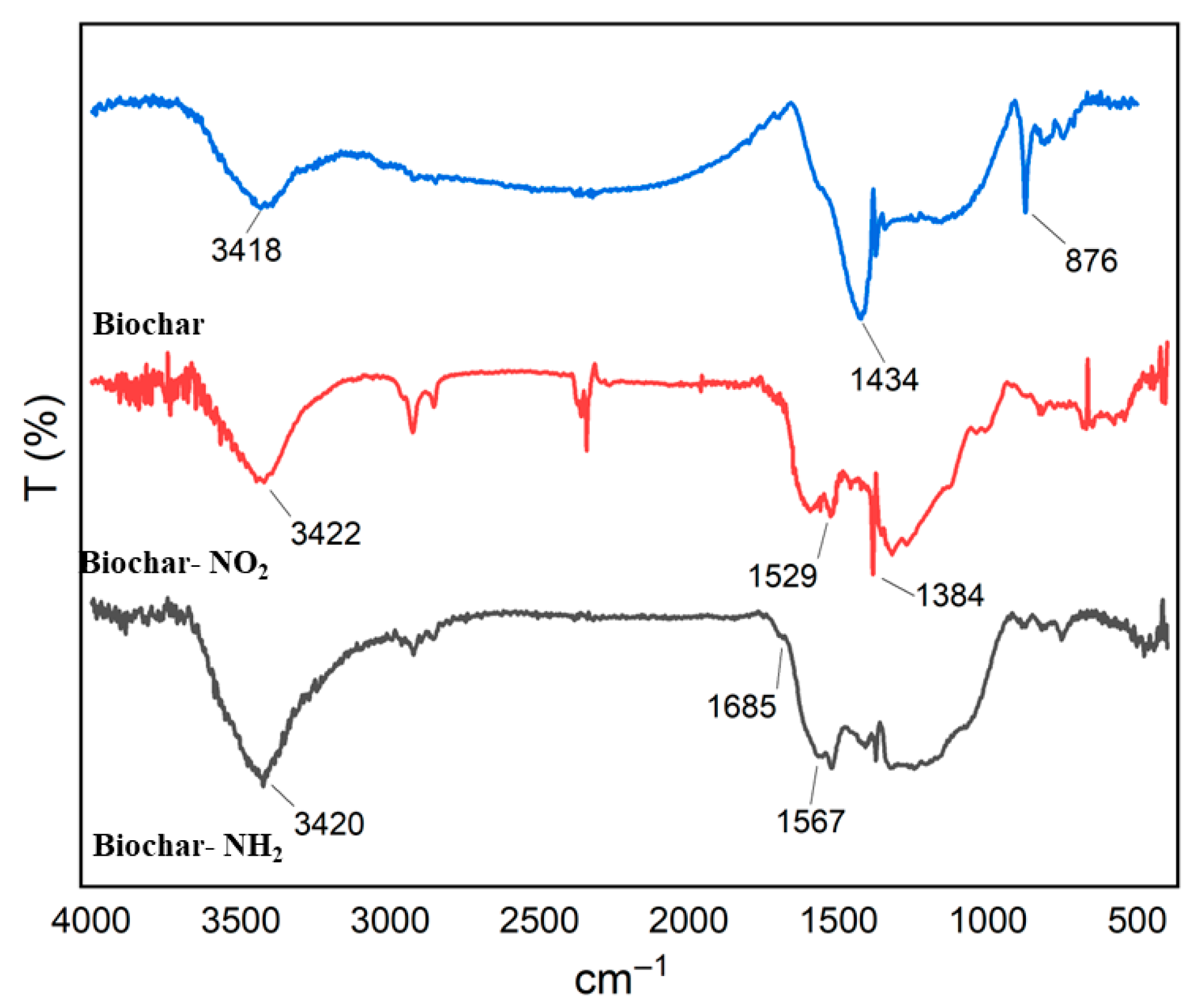
Biochar functional groups influence its chemical properties. FTIR analysis revealed that modified biochar exhibits a great-er abundance of functional groups, including amino moieties, compared to pristine biochar (Figure 8). Figure 8 shows the FT-IR spectrum of biochar, biochar-NO2, biochar-NH2 in the 400–4000 cm−1 range on the absorbent surface for identifying functional groups. From FT-IR data of the biochar Figure 8, the broad absorption peaks around 3418 cm−1 could be assigned to the stretching vibration of –OH. Peak at about 1434 cm−1 correspond to C=C stretching, while weak peak at 876 cm−1 is related to C–H stretching vibrations. Figure 8 (FT-IR data of the biochar-NO2) relates to biochar nitrification. Peaks at about 1529 cm−1 and 1384 cm−1 correspond to N–O stretching. The peak at 1529 cm−1 is likely associated with the asymmetric stretching of the N–O bond in nitro compounds or adsorbed NO2, while the peak at 1384 cm−1 can be attributed to the symmetric stretching of the N–O bond in nitrate ions (NO3−). The presence of these peaks suggests that NO2 is being oxidized to nitrate on the surface of the bio-char. Figure 8 (FT-IR data of the biochar-NH2) relates to Amine modified Biochar, the peak around 3650 cm−1 corresponds to N-H stretching, and the band near 1567 cm−1 indicates N-H bending vibration [30]. The peak at 1394 cm−1 is mainly assigned to C-N stretching, whereas the peaks at 2976, 2898, and 1066 cm−1 are attributed to alkyl CH2 and C-O stretching, respectively.
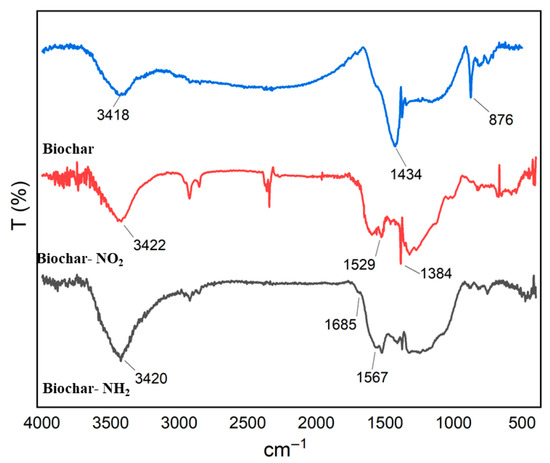
Figure 8.
FT-IR spectrum of biochar (Up), biochar-NO2 (Middle) and biochar-NH2.
2.8. Response Surface Model
Diesel engine performance and emissions were optimized using a response surface method with a blend of biodiesel, diesel, and biochar additives. Design Expert version 10 was used for statistical and optimization calculations. In this study, a historical-based RSM design was used. The p-value and F-value of the modified models were used to evaluate the engine-out parameters. The engine worked for 5 min to warm up. The experiments were conducted at full load. All the central points in the design are repeated 5 times. The Design Expert software suggested the design table. The uncertainty analysis was performed using actual and predicted data.
3. Results
This investigation comprehensively examines the effects of blending biodiesel-based waste cooking oil with conventional diesel fuel and nanobiochar. The studied parameters were engine speed (2900, 3100, and 3300 rpm) and biochar ratio (30, 60, and 90 ppm). The impact of biodiesel, diesel, and biochar additives on engine-out emissions (including UHC, CO, and NOx emissions) was analyzed. Additionally, the diesel engine performance parameters, including engine power and torque, were evaluated using the fuels mentioned above.
3.1. RSM Modeling
The optimal range of each parameters is also suggested by the RSM models. An ANOVA-based RSM technique was used to assess the models in this study. RSM is known as a powerful statistical approach that helps modify mathematical models to understand how different variables interact with a response. It is also used to identify complex relationships between engineering factors by combining statistical and mathematical techniques. The p-value and F-value of the model are the main parameters to evaluate the defined models. Statistical significance of the model was indicated by a p-value below 0.05. The suggested models showed statistical significance (p-values < 0.05), as illustrated in Table 4. The degree of freedom was considered as 5, and the mean square for engine power and torque was obtained as 0.13 and 1.16, respectively. Additionally, the mean squares for CO, NOx, and UHC emissions are reported as 0.93, 6323.06, and 122,000, respectively.

Table 4.
Statistical findings for the studied engine-out parameters.
According to Table 4, the engine speed variable has a greater effect than the nanobiochar additive, as indicated by higher F-values for all engine-out parameters. Furthermore, the F-value coefficient for NOX emissions was larger than that for the other studied variables.
3.2. Engine Performance
3.2.1. Engine Power
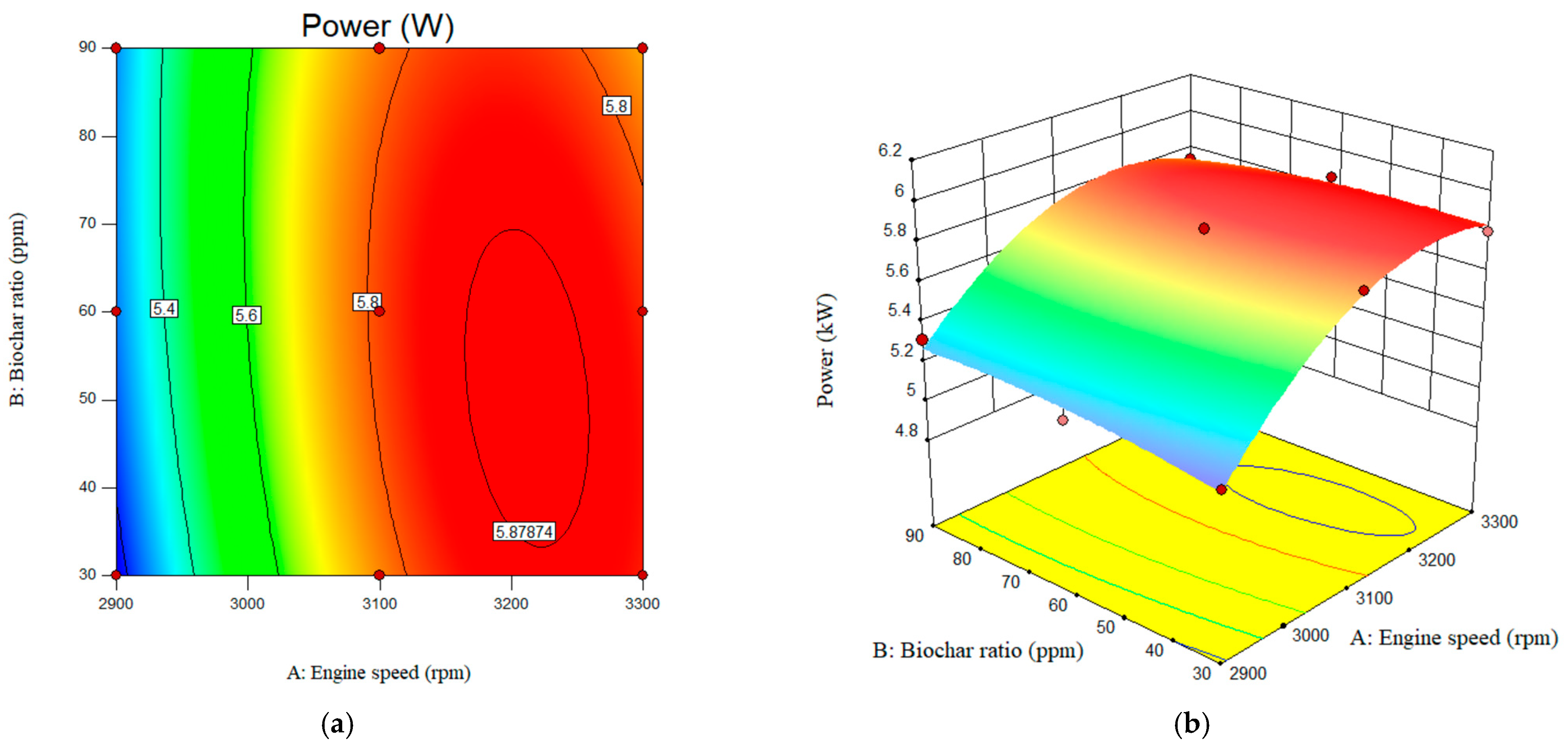
The interactive effects of engine-out performance parameters, such as engine power, are depicted in Figure 9. As can be seen, with an increase in engine speed from 2900 rpm to 3300 rpm, engine power increased from 5.27 to 5.8 W. A slight increase in engine torque was also observed as biochar increased from 30 to 90 ppm. The adjusted R2 value, which characterizes the impact of the studied parameters on the response factors, was achieved as 0.98. The predicted R-square was also achieved as 0.80. The results showed that engine power increased from 5.76 to 5.81 W. The maximum engine power was achieved at 3100 rpm with a B20-nanobiochar blended fuel. The value of the engine-out power at the engine speed of the 2900 rpm and nano-biochar of 30 ppm was achieved 5.17 W. The value of the engine power was 3300 rpm and nano biochar of 90 ppm was also reported as 5.74 W. The value for the engine power for pure B20 at engine speed of 2900, 3100, and 3300 rpm was obtained as 5.04, 5.47, and 5.53 W, respectively.
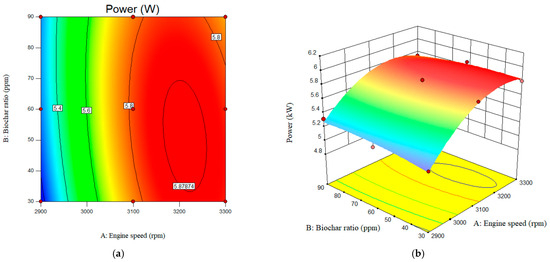
Figure 9.
Interactive effects of engine speed and nanobiochar ratio on the engine power. (a) 2D; (b) 3D.
According to the results, compared to engine speed, adding nanobiochar at the specified ratio had a less significant effect on both engine power and torque. However, the impact of the nanobiochar concentration on the diesel/biodiesel fuel was considerably less than of engine speed for both engine-out performance factors.
Various studies investigating the addition of B20 and nano additives to diesel fuel have proposed that higher combustion temperatures and the structural oxygen content of biodiesel are the most widely accepted explanations for the increased engine performance, which could result in enhanced combustion efficiency [31,32].
The variables of the modified equation are obtained for engine power as below:
3.2.2. Engine Torque
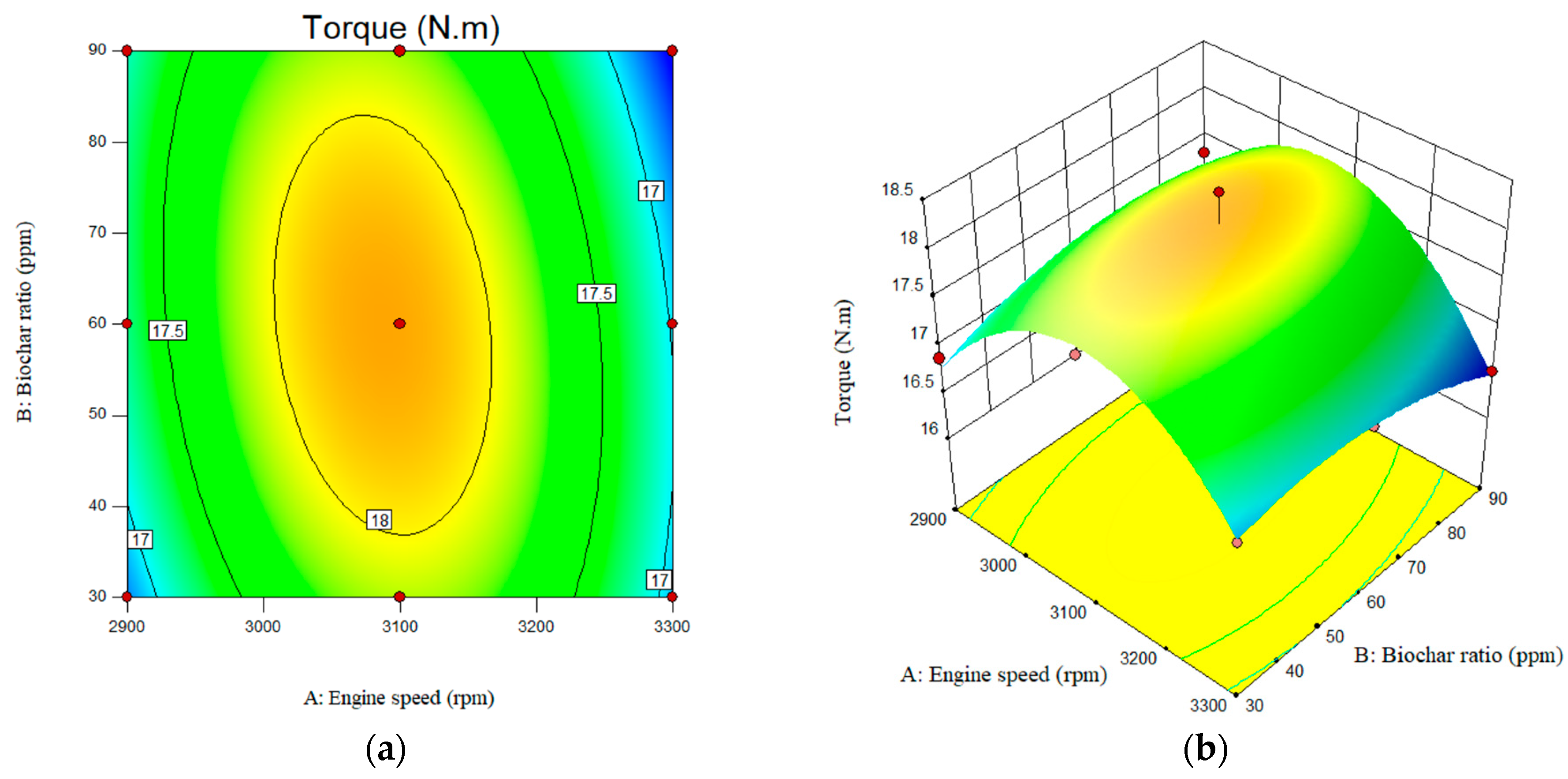
In this study, the adjusted R2 value, which characterizes the impact of the studied parameters on the response factors, was achieved as ~0.8 for engine torque. The predicted R-squares were also 0.80 and 0.52 for engine power and engine torque, respectively. The value of the engine torque for B20 at engine speed of the 2900 and nanobiochar of 30, 60, and 90 ppm was obtained as 16.88, 16.93, and 17.31 N.m, respectively. The engine torque for engine speed of the 3300 rpm at nanobiochar of 30, 60, and 90 ppm was achieved as 5.8, 5.85, and 5.74 N.m, respectively. The value of the engine torque for B20 was reported as 16.61, 16.88, and 15.99 N.m, respectively.
The regression coefficients confirm the reliability and accuracy of the models in this investigation, demonstrating a robust fit to the experimentally observed diesel engine data. Based on the modified data, it can be concluded that the optimal range for the variable, within the defined range of variables, lies between an engine speed of 3100 rpm and a nanobiochar concentration of 60 ppm. It is also worth noting that adding nanobiochar to B20 resulted in a slight increase in the engine torque. Additionally, as shown in Figure 10, engine torque increases as engine speed rises from approximately 2900 rpm to about 3100 rpm. However, a further increase in engine speed reduced torque. When the engine speed was increased from 3100 to 3300 rpm, the engine torque decreased sharply from 17.9 N.m to 16.89 N.m.
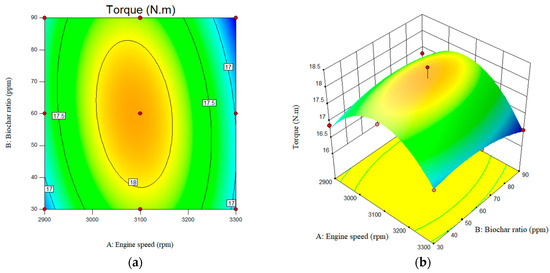
Figure 10.
Interactive effects of engine speed and nanobiochar ratio on the Torque. (a) 2D; (b) 3D.
Such a slight increase in engine torque could be attributed to potentially complete combustion and also energy content of the fuel blend studied [33,34].
The variables in the modified equation are obtained for engine torque and can be explained by Equation (2).
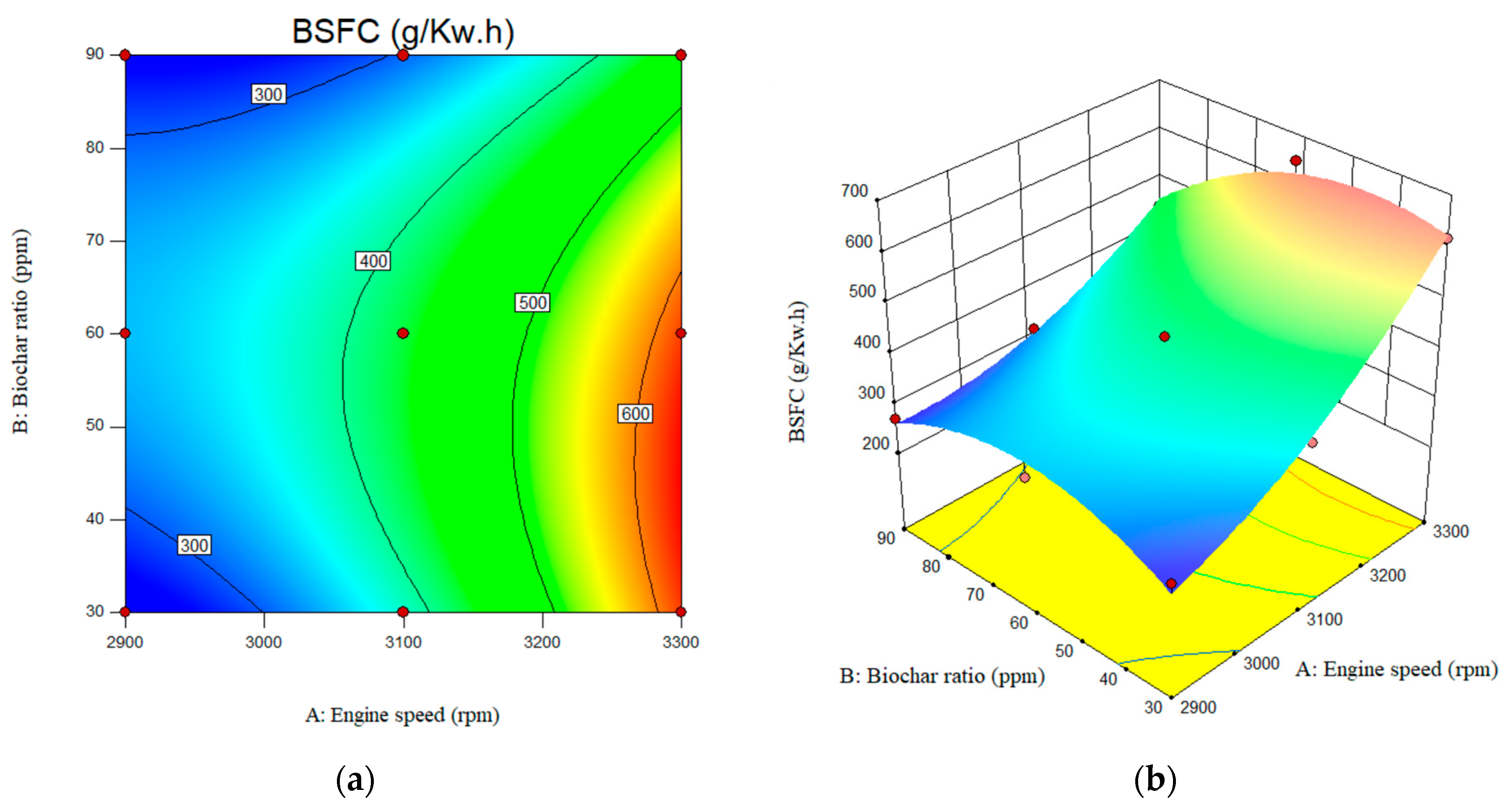
3.2.3. Brake-Specific Fuel Consumption (BSFC)
As shown in Figure 11, the surface plots depict the predicted BSFC over the input parameter ranges. As engine speed increases from 2900 rpm to 3100 rpm, brake-specific fuel consumption (BSFC) increases significantly. However, BSFC reached at a maximum of 600 g/kW·h at an engine speed of 3300 rpm. The value of the BSFC for the engine speed of the 2900 rpm and nano biochar of 30, 60, and 90 ppm was reported as 16.88, 16,933, and 17.32 g/kW·h, respectively. The BSFC for the B20 at engine speed of the 3300 rpm and nano biochar of 30, 60, and 90 ppm was calculated as 617.243, 647.59, and 440.01 g/kW·h, respectively.
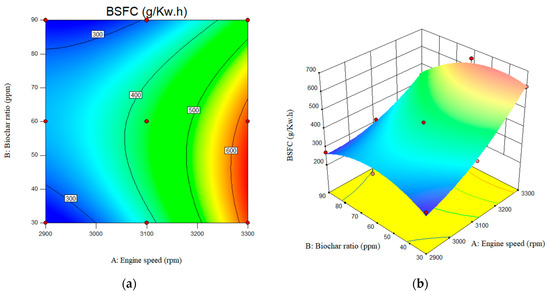
Figure 11.
Interactive effects of engine speed and biochar ratio on the BSFC. (a) 2D; (b) 3D.
Additionally, it was observed that increasing the nanobiochar concentration from 30 ppm to 60 ppm resulted in a slight increase in BSFC, from 386 g/kW·h to 429 g/kW·h. It was also observed that higher nanobiochar concentrations led to a decrease in BSFC levels up to 305 g/kW·h level. When compared to B20, the amount of brake-specific fuel consumption (BSFC) was reduced with the inclusion of biochar. The BSFC for B20 was observed to be 375 g/kW·h, 368 g/kW·h, and 684 g/kW·h at engine speeds of 2900, 3100, and 3300 rpm, respectively.
3.3. Engine Emissions
3.3.1. NOX Emission
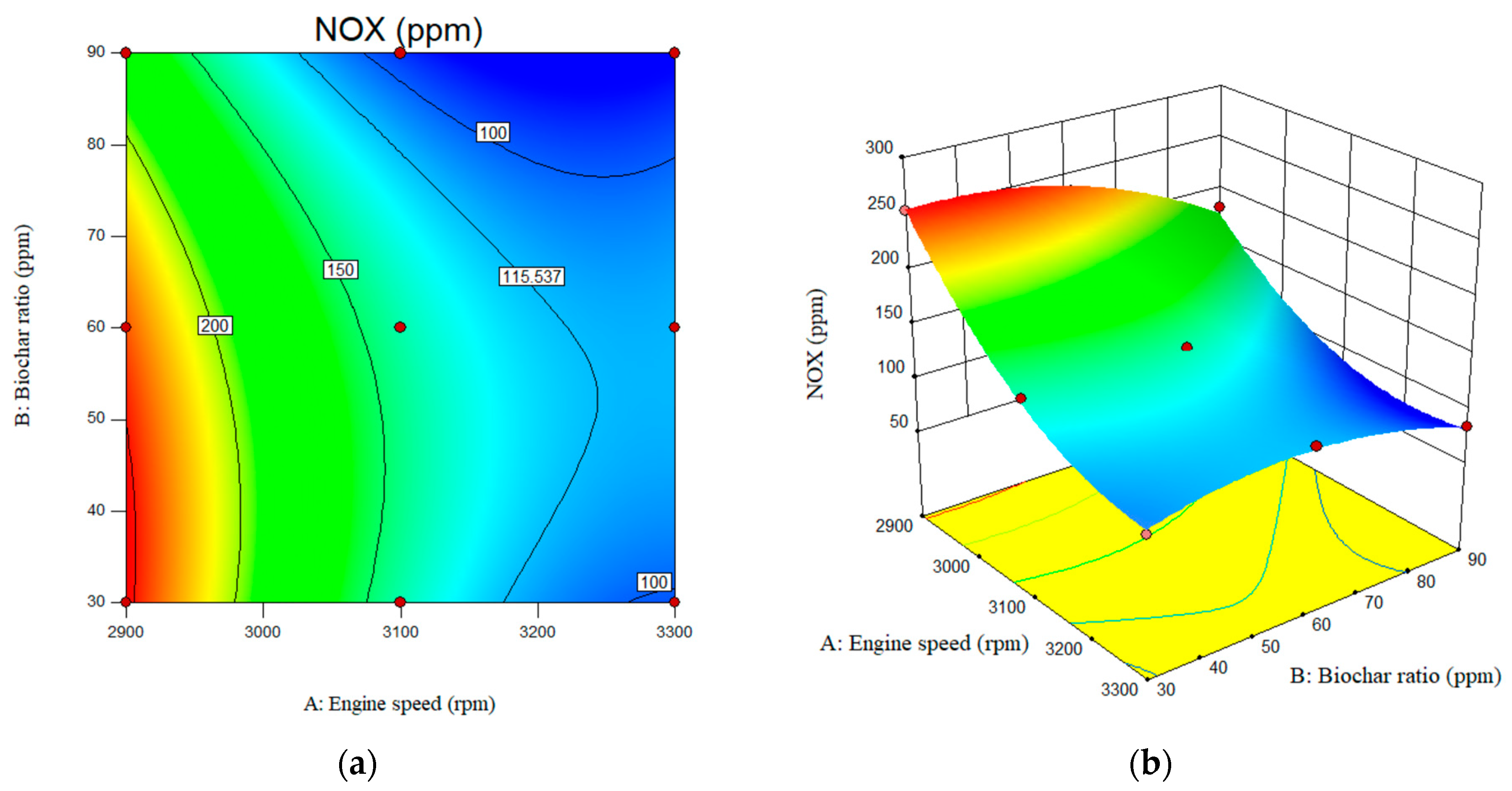
The results of this experimental investigation showed that the NOx emissions values of nanobiochar blended with B20 fuel were significantly lower compared to B20. According to the results, increasing nanobiochar from 30 ppm to 90 ppm resulted in lower NOx emissions for B20/nanobiochar emulsion, from 139 ppm to 94 ppm, compared to B20 fuel. NOx emissions are further reduced at higher nanobiochar concentrations. As shown in Figure 12, the NOx emission decreases sharply from 240 ppm to 114 ppm with increasing engine speed from 2900 rpm to 3300 rpm. The value of the NOx for B20 at engine speed of 2900 rpm and biochar ratio of the 30, 60, and 90 ppm was reported as 254, 234.5, and 182 ppm, respectively. This value for engine speed of the 3300 rpm and biochar ratio of 30, 60, and 90 ppm was obtained as 94, 116.5, and 84 ppm, respectively. The value for the NOx emissions for the engine speed of the 2900, 3100, and 3300 rpm for B20 was reported as 314, 220, and 170 ppm, respectively.
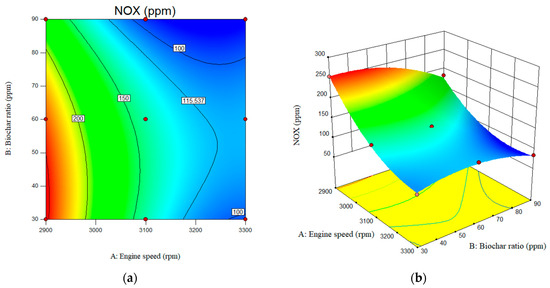
Figure 12.
Interactive effects of engine speed and nanobiochar ratio on the NOX emissions. (a) 2D; (b) 3D.
As shown in Figure 12b, the reduction in NOx emissions became increasingly pronounced with higher ratios of nanobiochar in the fuel mixture. As is clear, the amine-modified Biochar plays a crucial role as a catalyst in reducing NOx emissions. The Biochar itself, due to its porous structure, acts as an adsorbent. When enriched with amines, the catalytic role becomes more pronounced, and the amino group, as a base, enhances the reducing role. This mechanism becomes stronger as the ratio increases.
In addition, according to the F-value of the predicted models, the effect of engine speed on the emitted NOx was much higher than the nanobiochar concentration. The F-values for engine speed and nanobiochar were 470 and 65, respectively. According to the predicted models, the lowest NOX emission concentration is typically observed at 3000 rpm and a nanobiochar concentration of 90 ppm. This can be attributed to the formation of oxygen at a maximum rate of nanobiochar during combustion [35].
Numerous studies have shown that NOx emissions in diesel engines are driven by oxygen availability, residence time, and high combustion temperatures [36].
The derived model, based on the input parameters, including engine speed (A) and nanobiochar concentrations (B), is presented in Equation (3).
According to the results, both engine speed and nanobiochar concentration were statistically significant at the 1% level. Additionally, the interaction effect of engine speed and nanobiochar ratio on NOx formation was found to be substantial at the 5% significance level. The model F-value for NOx emissions was 126.37, indicating that the extracted model is significant and valid. Furthermore, the value of R-square (0.9953) corresponded well with the amount of adjusted R-square (0.9874). The predicted R-square for the emitted NOx was also reported as 0.94.
3.3.2. UHC Emission
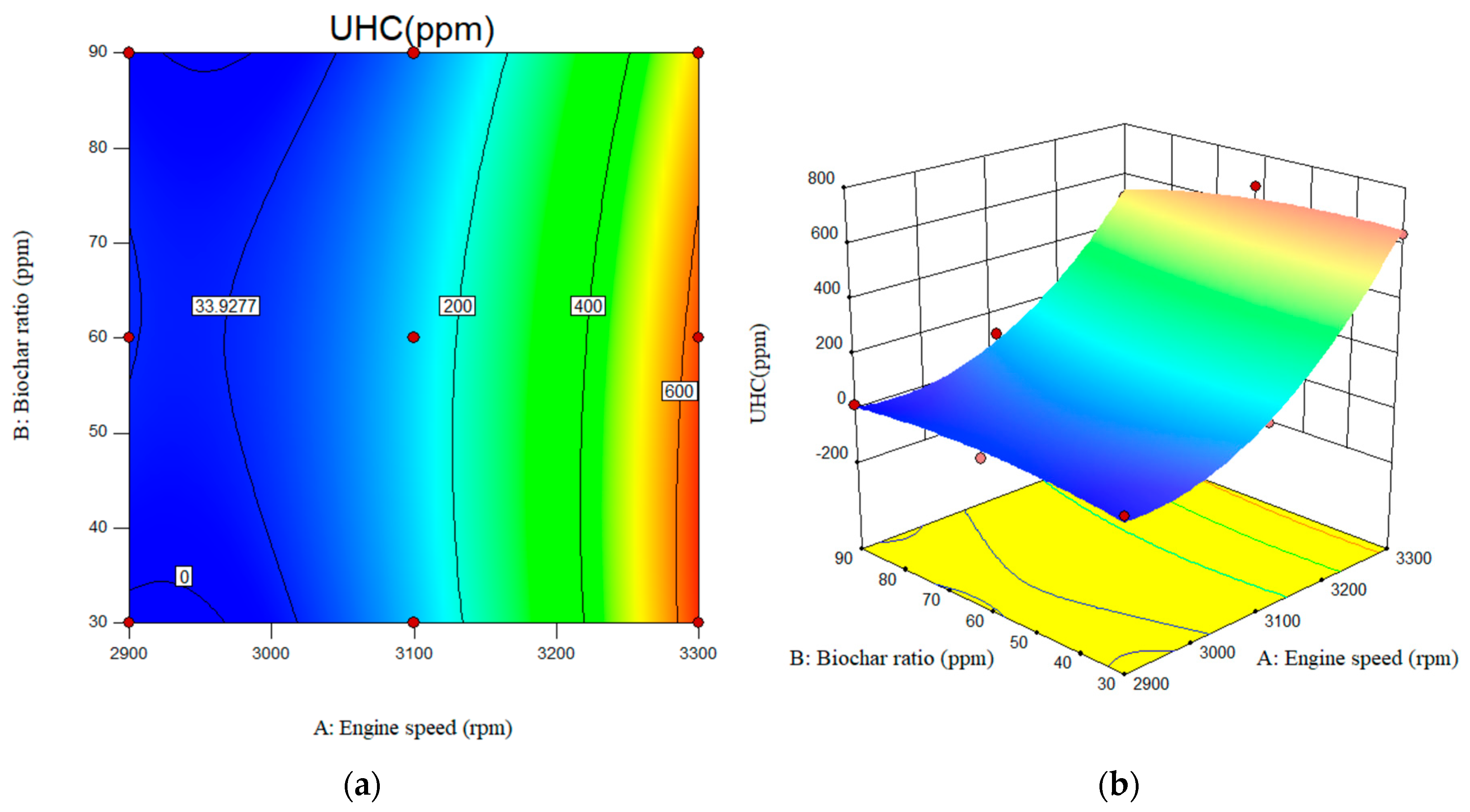
As can be seen in Table 4, the p-value for the UHC emissions indicates the relevance of the suggested model. The model’s p-value was 65, indicating that it effectively predicted the engine’s experimental data. The effect of engine speed on UHC emissions was statistically significant. However, the impact of nanobiochar on the studied levels was not statistically significant. Similarly, according to the results, the interaction influence of engine speed and nanobiochar concentration was not significant. According to the RSM results, the calculated R-square (0.9909) was in strong agreement with the adjusted R-square (0.9758). The predicted R-square for UHC emissions was also 0.90.
In Figure 13, variations in UHC emissions levels with engine speed and nano-biochar concentrations are illustrated. As shown in Figure 13a, UHC emissions increase with engine speed. As engine speed increased from 2900 rpm to 3300 rpm, UHC emissions increased from 30 ppm to 630 ppm. The maximum UHC emissions were 654 ppm for a B20/nanobiochar mixture at 3300 rpm.
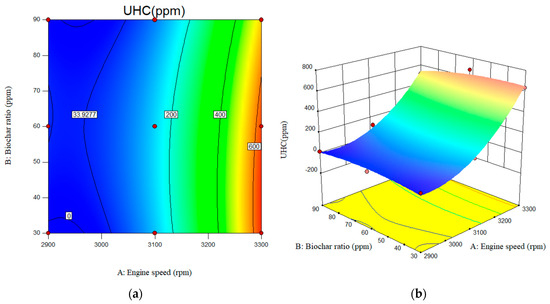
Figure 13.
Interactive effects of engine speed and biochar ratio on the UHC emissions. (a) 2D; (b) 3D.
The UHC emissions for B20 have been measured at 23, 148, and 726 at engine speeds of 2900, 3100, and 3300 rpm, respectively. Incorporating 90 ppm of nanobiochar resulted in a ~29% reduction in UHC emissions compared to B20.
3.3.3. CO Emission
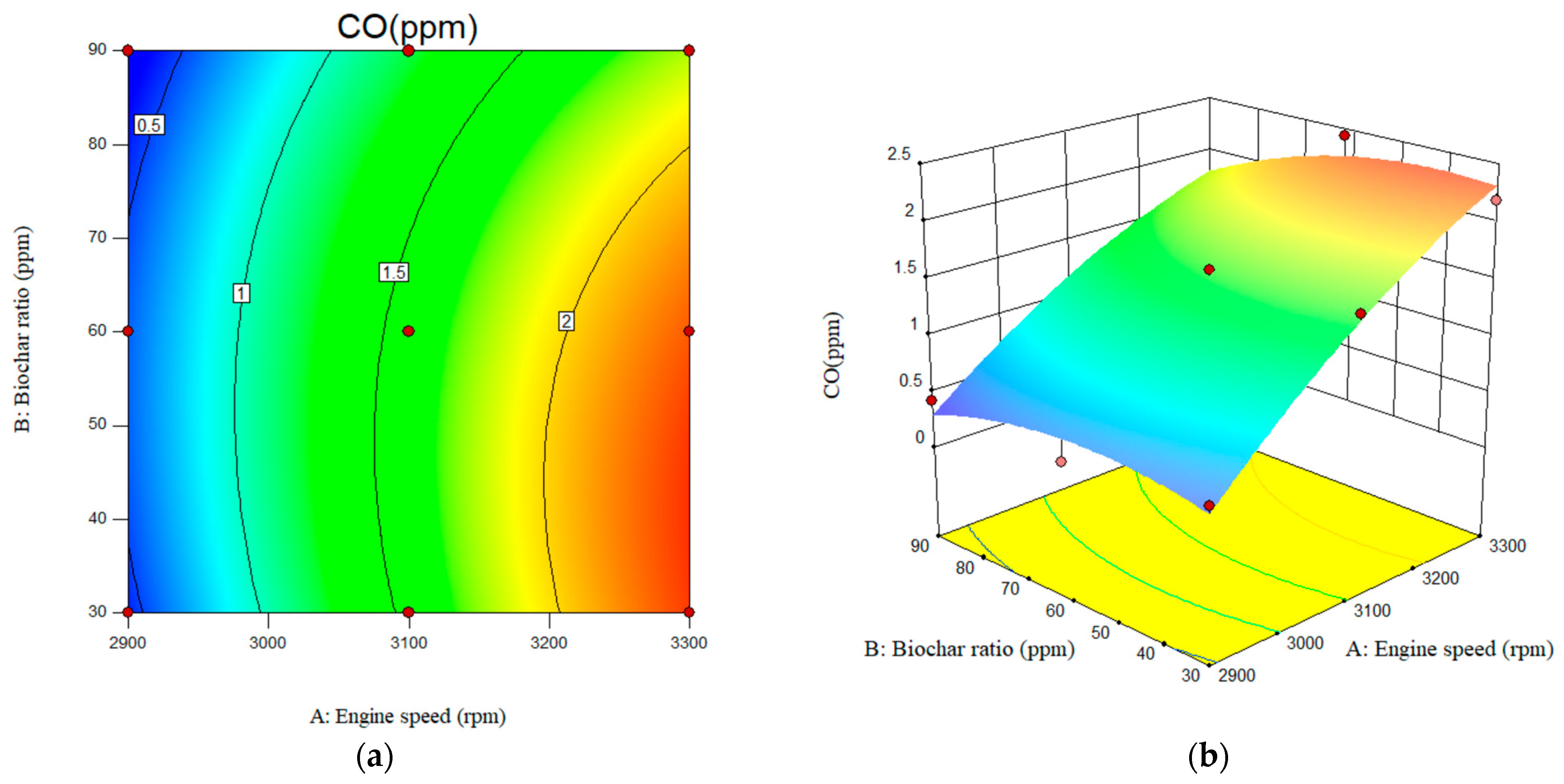
Figure 14 shows the variation in CO emissions levels with respect to engine speed and nanobiochar ratio for the studied fuel mixtures. As shown in Figure 14a, when the engine speed increases from 2900 rpm to 3300 rpm, there is a corresponding rise in the engine-out CO emissions. CO emissions were observed at the maximum engine speed and a minimum nanobiochar ratio of 2.25%. In terms of the effect of nanobiochar, a significant reduction in engine-out CO emissions was accomplished across the board for all studied nanobiochar ratios. When compared to B20 at an engine speed of 2900 rpm, the value of engine-out CO emissions that are reduced by B20 blended with 60 ppm of nanobiochar is ~35%. A quadratic equation was obtained based on the RSM results, as shown in Equation 5. Generally, due to biodiesel’s oxygen content, it burns completely, resulting in lower CO emissions with B20 compared to neat diesel. The value of the CO emissions for the B20 fuel was recorded as 0.55, 1.81, and 2.76 ppm at the engine speed of the 2900, 3100, and 3300 rpm, respectively. In this study, the addition of nanobiochar to B20 resulted in greater CO emissions reductions, attributed to its higher surface area and chemical reactivity. Reviewing the literature, Other published papers in this area have shown a similar tendency to use nanoparticles as a fuel additive [37,38,39].
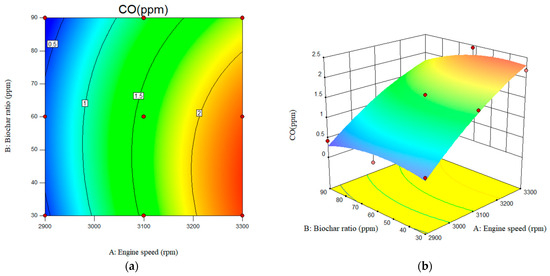
Figure 14.
Interactive effects of engine speed and nanobiochar ratio on the CO emissions. (a) 2D; (b) 3D.
The RSM-based analysis indicated that the model was highly accurate in predicting CO emissions, as evidenced by an R-square value of 0.9738. The F-value of 22.3 further supports the model’s reliability in estimating diesel-engine-out CO emissions. Additionally, according to the RSM-based results obtained, among the studied variables, engine speed plays the most significant role in the formation of CO emissions, as indicated by its high F-value of 103.67, compared to the nanobiochar ratio, which has a much lower F-value of 3.78. The predicted R-square for CO emissions was also reported as 0.68.
3.4. Multi-Objective Optimization
In the present experimental work, we optimized the engine speed range and the nanobiochar additive mixed with B20 to achieve maximum engine performance and minimum emissions. The results confirmed that engine speed had the most significant influence on both engine performance and engine-out emissions, followed by nanobiochar ratio. The criteria and limits used to optimize the performance and emissions of a diesel engine running on B20 at different engine speeds and nanobiochar ratios are presented in Table 5. The optimization process was performed using Design-Expert software Version 10, with the goal of maximizing engine performance (i.e., torque and power) and minimizing engine emissions, including CO, NOx, and UHC. The optimization criteria are listed in Table 5. The parameters, including importance and parameter weights, were selected based on experience and other published papers in this area [40]. The maximum and minimum values of the variables selected from the experimental data were imported into the software.

Table 5.
The criteria of the multi-objective optimization process.
According to the results of the RSM-based desirability function, the optimal conditions for engine performance and emissions within the studied ranges were determined to be an engine speed of 3108 rpm and a nanobiochar concentration of 90 ppm.
The corresponding values of engine performance and emissions at the optimal point are 5.78 W for engine power, 17.96 N.m for engine torque, and 309 g/kW·h for BSFC.h, NOX emissions of 90.92 ppm, UHC emissions of 104.9 ppm, and CO emissions of 1.25. For this point, a high value of the desirability function was achieved, at 0.76, indicating the model’s accuracy in predicting the curves of the experimental data for each engine parameter. The overall uncertainly was calculated for engine power and engine torque as 0.035 and0.26, respectively. In addition, the overall uncertainly for NOx, UHC, and CO emissions also was obtained as 4.08, 24.99, and 0.11, respectively.
4. Discussion
The experimental results demonstrated that incorporating amine-functionalized nanobiochar into diesel–biodiesel blends significantly improved engine performance and reduced exhaust emissions.
4.1. Engine Performance Improvement
Compared to the base B20 fuel, the addition of 90 ppm of the modified biochar resulted in higher torque and power output, along with a marked reduction in brake-specific fuel consumption (BSFC). According to the results obtained, with increasing biochar ratio from 30 ppm to 90 ppm, the engine power increased slightly from 5.7 to 5.81 W. In addition, with increasing engine speed the engine power increased from 5.2 W to 5.8 W. The effect of the engine speed on the engine performance was statistically significant at level of 5%, but the effect of the biochar ratio was reported not significant. This improvement can be attributed to the porous nanostructure and the biochar’s amine groups, which provide both catalytic and adsorptive effects, thereby enhancing combustion.
4.2. Emission Reduction
In terms of emissions, the most notable effect was observed for nitrogen oxides (NOx). The results showed that NOx emissions decreased sharply as the concentration of amine-modified biochar increased from 30 ppm to 90 ppm, with a reduction of nearly 40% compared to B20 fuel at the optimal operating condition. A Similar trend was also observed when the engine speed increased from 2900 rpm to 3300 rpm and NOx emission reduced from ~234 ppm to ~117 ppm. This trend is consistent with previous studies on nano-additives in biodiesel blends, where the presence of oxygenated nanostructures facilitated more complete combustion and altered the thermal pathways of NOx formation.
Similarly, carbon monoxide (CO) and unburned hydrocarbons (UHC) showed substantial reductions when nano-biochar was blended with B20. The high surface area and reactivity of the nanoparticles enhanced oxidation of incomplete combustion products, which explains the improved CO and UHC levels compared to neat biodiesel–diesel blends. These findings align with the literature on nanocatalysts such as cerium oxide and graphene-based additives, but highlight the novelty of biochar as a sustainable, low-cost alternative.
In this study, the observed reductions in engine-out emissions can be attributed to several combustion-related factors. The oxygen content in the B20 blend, which is inherently higher than that of conventional diesel, facilitates more fuel oxidation, resulting in lower levels of carbon monoxide (CO) and unburned hydrocarbons (UHCs). Furthermore, the inclusion of amine-modified nanobiochar in the fuel blend enhances the combustion process in multiple ways. The nanobiochar’s porous structure and high surface area act as a catalyst, improving fuel atomization and promoting better mixing of the fuel and air. This leads to a more efficient combustion process, optimizing engine performance and reducing NOx emissions. The amine groups in the nanobiochar can further reduce NOx emissions by facilitating nitrogen oxide reduction via chemical reactions during combustion.
4.3. Multi-Objective Optimization
The multi-objective optimization analysis further supported these experimental findings. Using response surface methodology (RSM), the optimal condition was determined to be an engine speed of ~3100 rpm and 90 ppm biochar concentration, which simultaneously maximized engine power and torque while minimizing NOx, CO, and UHC emissions. The high desirability index (0.76) obtained in the optimization confirms the robustness of the models and the practical applicability of the proposed additive.
4.4. Practical Implications of Biochar
Overall, the use of amine-functionalized nanobiochar as a green additive offers dual benefits: improving combustion efficiency and reducing harmful emissions. Unlike conventional metallic nanoparticles, which may pose environmental and cost concerns, biochar provides a renewable, economical, and environmentally benign solution. This highlights the potential of functionalized biochar not only in diesel engines but also in broader energy and environmental applications. Adding biochar to a diesel–biodiesel blend can improve engine performance, boost efficiency, and reduce harmful emissions like nitrogen oxides (NO) and carbon monoxide (CO). These additives serve as eco-friendly alternatives to traditional metal-based additives, offering benefits such as better combustion and lower pollution without the toxic side effects of some conventional additives [23]. Moreover, the use of biochar in diesel–biodiesel blends presents a pathway to utilize waste biomass, converting it into a valuable resource that simultaneously addresses fuel enhancement and waste management challenges. Different types of biochar, derived from various feedstocks and production methods, exhibit varying degrees of effectiveness, influencing the blend’s properties and its subsequent impact on engine performance and emissions. Factors such as biochar particle size, surface area, and chemical composition play critical roles in determining its efficacy as a fuel additive, necessitating careful optimization to achieve desired combustion efficiency and emission reduction.
4.5. Comparison with Metal and Carbon-Based Additives
When comparing metallic (CeO2, Fe3O4) and carbon-based (graphene, GQDs) additives for a diesel engine using biodiesel, several factors must be considered. Metallic additives like CeO2 and Fe3O4 can act as oxidation catalysts, potentially improving combustion efficiency and reducing particulate matter (PM) emissions. Cerium oxide, in particular, is known for its oxygen storage capacity, which can promote soot oxidation. Iron oxide can also help reduce emissions, though its catalytic activity may differ from that of CeO2.
4.6. Future Research on GQDs and Biochar
Graphene and Graphene Quantum Dots (GQDs) can be used as additives for biodiesel to improve engine performance and reduce emissions. Studies show that adding GQDs to biodiesel blends can increase torque and power, decrease specific fuel consumption, and significantly reduce emissions of carbon monoxide (CO) and unburned hydrocarbons (UHC). The exact benefits depend on the blend concentration and engine conditions, but GQDs show promise as an environmentally friendly additive compared to metal-based nanoparticles. Further research is needed to optimize the use of these additives and fully understand their long-term impact on engine performance and durability [12].
5. Conclusions
The performance of a single-cylinder diesel engine was analyzed for the biodiesel blends of B20 blended with nanobiochar as a bio-based fuel additive. The nano-sized and porous morphology of amines-modified biochar, as observed using FESEM techniques, indicated a nanocatalyst. Additionally, the EDS analysis showed the element content of the amine-modified biochar. This additive, along with its higher proportion, has shown a positive effect on reducing gas emissions. These results are attributed to its structure and catalytic and reducing roles of both amine and biochar surfaces. The results showed that input parameters, such as nanobiochar additive concentration and engine speed, had a significantly influenced on engine performance and engine-out emissions. According to the results, higher engine load levels appeared to have an adverse influence on diesel engine-out emissions, including NOx, CO, and UHC. Unlike engine speed, a favorable declining trend in the formation of all the above-mentioned emissions was observed when varying the nanobiochar additive concentration.
For all derived models using the RSM technique, the statistical significance of the predicted models was observed, as indicated by a p-value less than 0.05, demonstrating the high accuracy of the models. In this study, the optimized diesel engine working conditions, fueled with B20 with nanobiochar, for improved engine performance and minimal engine-out emissions, including CO, NOx, and UHC emissions, were found to be 3108 engine speed and a nanobiochar concentration of 90 ppm, resulting in 5.78 kW power, 17.96 N.m torque, and 309 g/kW·h BSFC, 90.92 ppm NOx, 104.9 ppm of UHC, and 1.25% CO emission.
Author Contributions
Conceptualization, B.S. and M.H.; methodology, B.S., M.Y., S.M.S.A. and M.H.; software, B.S., M.Y. and S.M.S.A.; validation, B.S., H.H.A.A. and S.M.S.A.; formal analysis, B.S., S.M.S.A. and M.H.; investigation B.S., M.Y. and M.H.; resources, B.S., H.H.A.A. and M.Y.; data curation, B.S. and M.Y.; writing—original draft preparation, B.S., M.Y., S.M.S.A. and M.H.; writing—review and editing, S.M.S.A. and M.H.; visualization, B.S. and M.H.; supervision, B.S.; project administration, B.S.; funding acquisition, B.S. and H.H.A.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. Please contact to b.shadidi@basu.ac.ir.
Acknowledgments
This research was conducted in collaboration with the Faculty of Agriculture, Bu-Ali Sina University.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Gierer, F.; Vaughan, S.; Slater, M.; Thompson, H.M.; Elmore, J.S.; Girling, R.D. A review of the factors that influence pesticide residues in pollen and nectar: Future research requirements for optimising the estimation of pollinator exposure. Environ. Pollut. 2019, 249, 236–247. [Google Scholar] [CrossRef]
- Ahmed, M.; Shan, S.; Ismail, F.; Ahmad, M. The effect of energy consumption, income, and population growth on CO2 emissions in China, India and the USA. Sustainability 2023, 15, 11956. [Google Scholar] [CrossRef]
- Najafi, G.; Shadidi, B.; Mamat, R.; Safieddin Ardebili, S.M.; Hazrati, S.; Ebadi, M.T.; Ghazali, M.F. Multi-objective optimization of the performance and emissions characteristics of a CI engine powered by Pyrus glabra biodiesel as a novel feedstock using response surface method. Biofuels 2024, 15, 1099–1108. [Google Scholar] [CrossRef]
- Mhetras, N.; Gokhale, D. Sustainable biodiesel production: Importance of feedstock resources and production methods. RSC Adv. 2025, 15, 26739–26754. [Google Scholar] [CrossRef] [PubMed]
- Mofijur, M.; Ahmed, S.F.; Ahmed, B.; Mehnaz, T.; Mehejabin, F.; Shome, S.; Almomani, F.; Chowdhury, A.A.; Kalam, M.A.; Badruddin, I.A.; et al. Impact of nanoparticle-based fuel additives on biodiesel combustion: An analysis of fuel properties, engine performance, emissions, and combustion characteristics. Energy Convers. Manag. X 2024, 21, 100515. [Google Scholar] [CrossRef]
- Ghanati, S.G.; Doğan, B.; Yeşilyurt, M.K. The effects of the usage of silicon dioxide (SiO2) and titanium dioxide (TiO2) as nano-sized fuel additives on the engine characteristics in diesel engines: A review. Biofuels 2023, 15, 229–243. [Google Scholar] [CrossRef]
- Elkelawy, M.; Kabeel, A.E.; El Shenawy, H.; Panchal, H.; Elbanna, A.; Bastawissi, H.A.-E.; Sadasivuni, K.K. Experimental investigation on the influences of acetone organic compound additives into the diesel/biodiesel mixture in CI engine. Sustain. Energy Technol. Assess. 2023, 50, 101951. [Google Scholar] [CrossRef]
- Bitire, S.O.; Akinmoladun, O.O.; Oladimeji, M.O.; Olatunji, S.O.; Oladipo, A.A.; Olayanju, T.M.A. The impact of CuO nanoparticles as fuel additives in diesel engines: A review. Energy Environ. 2023, 237, 333–350. [Google Scholar] [CrossRef]
- Jokubynienė, V.; Slavinskas, S.; Kreivaitis, R. The Effect of Nanoparticle Additives on the Lubricity of Diesel and Biodiesel Fuels. Lubricants 2023, 11, 290. [Google Scholar] [CrossRef]
- Ying, S.; Guan, Z.; Ofoegbu, P.C.; Clubb, P.; Rico, C.; He, F.; Hong, J. Green synthesis of nanoparticles: Current developments and limitations. Environ. Technol. Innov. 2022, 26, 102336. [Google Scholar] [CrossRef]
- Stanley, S. Biological nanoparticles and their influence on organisms. Curr. Opin. Biotechnol. 2014, 28, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Heidari-Maleni, A.; Gundoshmian, T.M.; Jahanbakhshi, A.; Ghobadian, B. Performance improvement and exhaust emissions reduction in diesel engine through the use of graphene quantum dot (GQD) nanoparticles and ethanol-biodiesel blends. Fuel 2020, 267, 117116. [Google Scholar] [CrossRef]
- Uslu, O.S.; Babur, E.; Alma, M.H.; Solaiman, Z.M. Walnut Shell Biochar Increases Seed Germination and Early Growth of Seedlings of Fodder Crops. Agriculture 2020, 10, 427. [Google Scholar] [CrossRef]
- Farrokh, M.; Hajjami, M.; Zolfigol, M.A.; Jalali-Mola, S. Catalytic application of biochar functionalized copper L-histidine for the chemo and homoselective conversion of cyanides to amides and reduction of nitroarenes to anilines. ACS Omega 2024, 9, 47811–47821. [Google Scholar] [CrossRef]
- Yang, Z.; Xing, R.; Zhou, W. Adsorption of ciprofloxacin and Cu2+ onto biochars in the presence of dissolved organic matter derived from animal manure. Environ. Sci. Pollut. Res. 2019, 26, 14382–14392. [Google Scholar] [CrossRef]
- Taghizadeh-Alisaraei, A.; Motevali, A.; Ghobadian, B. Ethanol production from date wastes: Adapted technologies, challenges, and global potential. Renew. Energy 2019, 143, 1094–1110. [Google Scholar] [CrossRef]
- Jeong, C.Y.; Dodla, S.K.; Wang, J.J. Fundamental and molecular composition characteristics of biochars produced from sugarcane and rice crop residues and by-products. Chemosphere 2016, 142, 4–13. [Google Scholar] [CrossRef]
- Yusri, I.M.; Majeed, A.A.; Mamat, R.; Ghazali, M.F.; Awad, O.I.; Azmi, W.H. A review on the application of response surface method and artificial neural network in engine performance and exhaust emissions characteristics in alternative fuel. Renew. Sustain. Energy Rev. 2018, 90, 665–686. [Google Scholar] [CrossRef]
- Afshar, M.; Mofatteh, S. Biochar for a sustainable future: Environmentally friendly applications and challenges. Sci. Total Environ. 2024, 860, 159466. [Google Scholar] [CrossRef]
- Mirbagheri, S.A.; Safieddin Ardebili, S.M.; Kiani, M.K.D. Modeling of the engine performance and exhaust emissions characteristics of a single-cylinder diesel using nano-biochar added into ethanol-biodiesel-diesel blends. Fuel 2020, 278, 118238. [Google Scholar] [CrossRef]
- Matin, N.H.; Sadeghi, H.; Baghery, R.; Bahrami, S.; Vali, L. Almond and Walnut Shell-Derived Biochars Affect Sorption and Degradation of Pesticides in Soil. Sci. Total Environ. 2020, 744, 140941. [Google Scholar] [CrossRef]
- Behbahan, A.K.; Khodadadi, M.; Ghorbani, M.; Ahmadpour, A. Application of MOF Modified Walnut Biochars for the Extraction of Pesticides from Wheat Samples. J. Hazard. Mater. 2023, 442, 130050. [Google Scholar] [CrossRef]
- Le, T.; Nguyen, T.; Phan, C.; Huynh, T. The role of nanobiochar in biodiesel production and its effects on diesel engine performance. Energy Rep. 2024, 10, 1123–1134. [Google Scholar]
- Safieddin Ardebili, S.M.; Taghipoor, A.; Solmaz, H.; Mostafaei, M. The effect of nano-biochar on the performance and emissions of a diesel engine fueled with fusel oil-diesel fuel. Fuel 2020, 268, 117356. [Google Scholar] [CrossRef]
- Paramasivam, B. Investigation on emission, performance, and combustion distinctiveness of bael seed cake pyrolysis oil/nano-biochar/diesel blend fuelled in a compression ignition engine. Environ. Prog. Sustain. Energy 2022, 41, e13871. [Google Scholar] [CrossRef]
- Ramachandran, S.; Thangavelu, M.; Kamaraj, L.; Sorakka Ponnappan, V.; Arumugam, R. Ignition analysis of diesel engine propelled with neat biodiesel containing nanoparticles. Energy Sources Part A Recovery Util. Environ. Eff. 2021, 47, 7225–7237. [Google Scholar] [CrossRef]
- Sánchez-Rodríguez, G.; Martínez, L.; González, M.; López, P.; Hernández, A. Density and Viscosity in Biodiesel + Diesel Mixtures from Waste Oils. Appl. Sci. 2025, 15, 3812. [Google Scholar] [CrossRef]
- Tüccar, G.; Aydın, A.; Yılmaz, A.; Sevinç, V.; Koç, M. Investigations of effects of density and viscosity of diesel & biodiesel fuels on engine exhaust emissions. Energy Rep. 2018, 4, 621–630. [Google Scholar] [CrossRef]
- Green, R.A.; Hartwig, J.F. Palladium-catalyzed amination of aryl chlorides and bromides with ammonium salts. Org. Lett. 2014, 16, 4388–4391. [Google Scholar] [CrossRef]
- Chen, Y.L.; Pan, B.C.; Li, H.Y.; Zhang, W.M.; Lv, L.; Wu, J. Selective removal of Cu(II) ions by using cation-exchange resin-supported polyethyleneimine (PEI) nanoclusters. Environ. Sci. Technol. 2010, 44, 3508–3513. [Google Scholar] [CrossRef]
- Prabu, A. Exploring the impact of aluminum oxide nanoparticles on waste transformer biodiesel blend under variable injection timing. Sustain. Energy Res. 2024, 11, 19. [Google Scholar] [CrossRef]
- Hameed, A.Z.; Muralidharan, K. Mahua oil transesterified biofuel (B20) + Al2O3 and CeO2 nano-additives: Performance, emission, and catalytic activity analysis. ACS Omega 2023, 8, 5692–5701. [Google Scholar] [CrossRef]
- Dhande, D.Y.; Choudhari, C.S.; Gaikwad, D.P.; Sinaga, N.; Dahe, K.B. Prediction of spark ignition engine performance with bioethanol–gasoline mixes using a multilayer perception model. Pet. Sci. Technol. 2022, 40, 1437–1461. [Google Scholar] [CrossRef]
- Zhang, M.; Cao, J. Effects of lean burn on combustion and emissions of a DISI engine fueled with methanol–gasoline blends. Energies 2024, 17, 4023. [Google Scholar] [CrossRef]
- Sujin, P.; Roji, S.S.S.; Kings, A.J.; Miriam, L.M. Optimization of engine performance, emission and combustion parameters by using mixed nonedible oil biodiesel with nano-additives using hybrid techniques. Energy 2024, 305, 132413. [Google Scholar] [CrossRef]
- Rohani, A.; Zareei, J.; Ghadamkheir, K.; Farkhondeh, S.A. Optimization of engine parameters and emission profiles through bio-additives: Insights from ANFIS Modeling of Diesel Combustion. Cleaner Eng. Technol. 2025, 27, 100994. [Google Scholar] [CrossRef]
- Soudagar, M.E.M.; Shelare, S.; Marghade, D.; Belkhode, P.; Nur-E-Alam, M.; Kiong, T.S.; Fattah, I.M.R. Optimizing IC engine efficiency: A comprehensive review on biodiesel, nanofluid, and the role of artificial intelligence and machine learning. Energy Convers. Manag. 2024, 307, 118337. [Google Scholar] [CrossRef]
- Gad, M.S.; Kamel, B.M.; Badruddin, I.A. Improving the diesel engine performance, emissions and combustion characteristics using biodiesel with carbon nanomaterials. Fuel 2021, 288, 119665. [Google Scholar] [CrossRef]
- Sharma, V.; Hossain, A.K.; Ahmed, A.; Rezk, A. Study on using graphene and graphite nanoparticles as fuel additives in waste cooking oil biodiesel. Fuel 2022, 328, 125270. [Google Scholar] [CrossRef]
- Solmaz, H.; Ardebili, S.M.S.; Aksoy, F.; Calam, A.; Yılmaz, E.; Arslan, M. Optimization of the operating conditions of a beta-type rhombic drive Stirling engine by using response surface method. Energy 2020, 198, 117377. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).