Wet-Process-Modified Blue-Green Algae Biochar by K2FeO4 for the Efficient Adsorption of Cr(VI) from Water
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Biochar Preparation
2.3. Characterization
2.4. Batch Adsorption Experiments
3. Results and Discussion
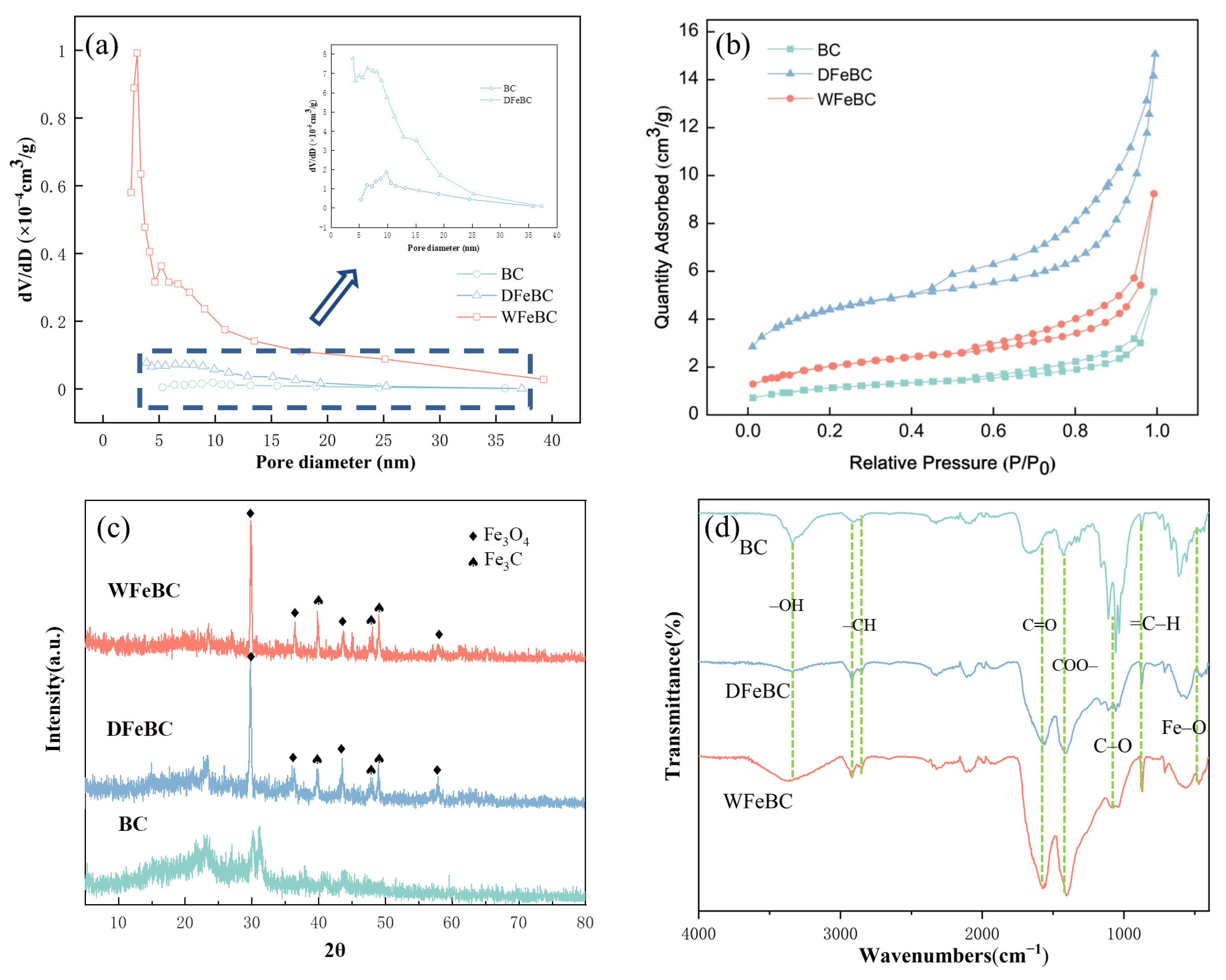
3.1. Characterization
3.2. Adsorption of Hexavalent Chromium
3.2.1. Effect of Preparation Parameters
3.2.2. Effect of Initial pH
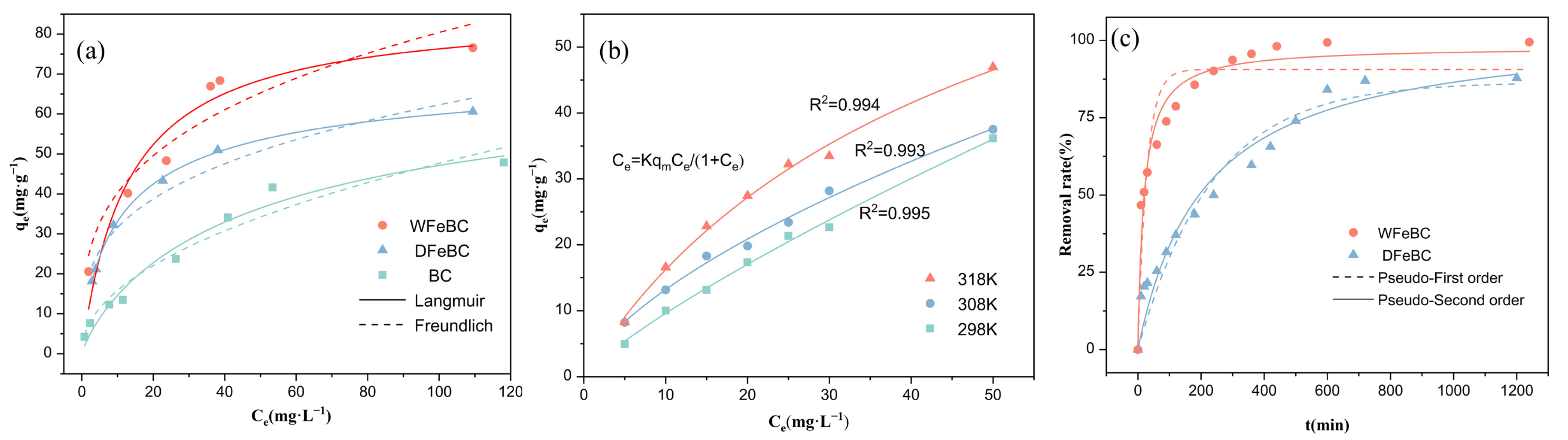
3.3. Kinetics and Isotherms
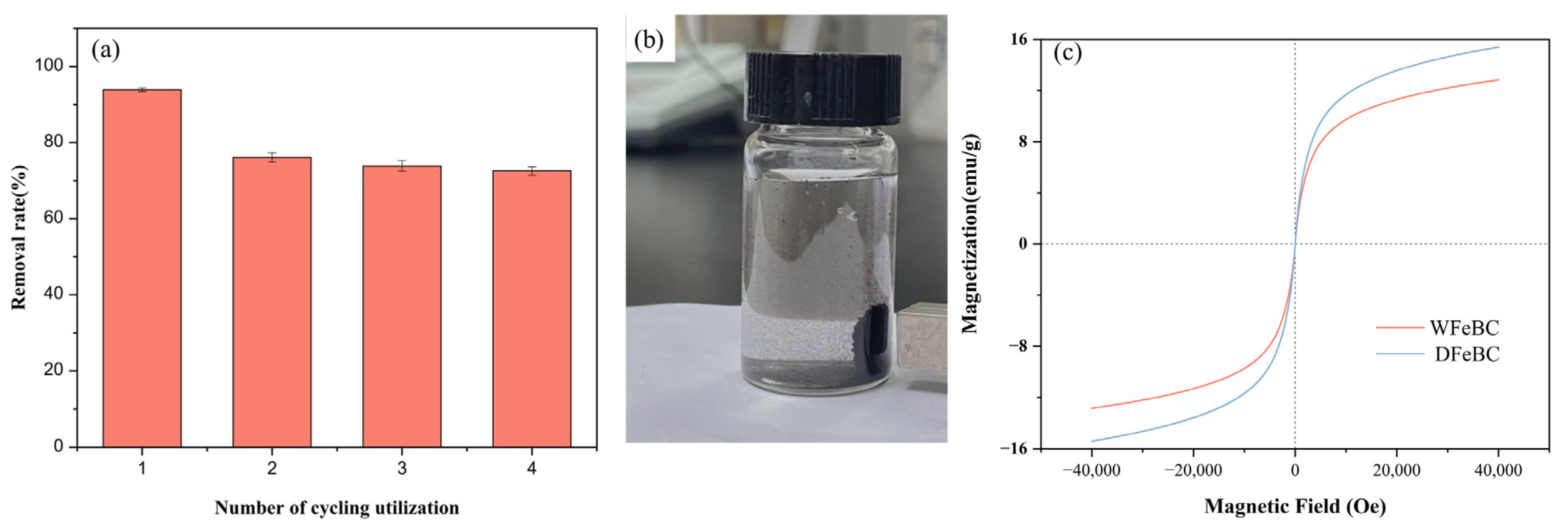
3.4. Regeneration
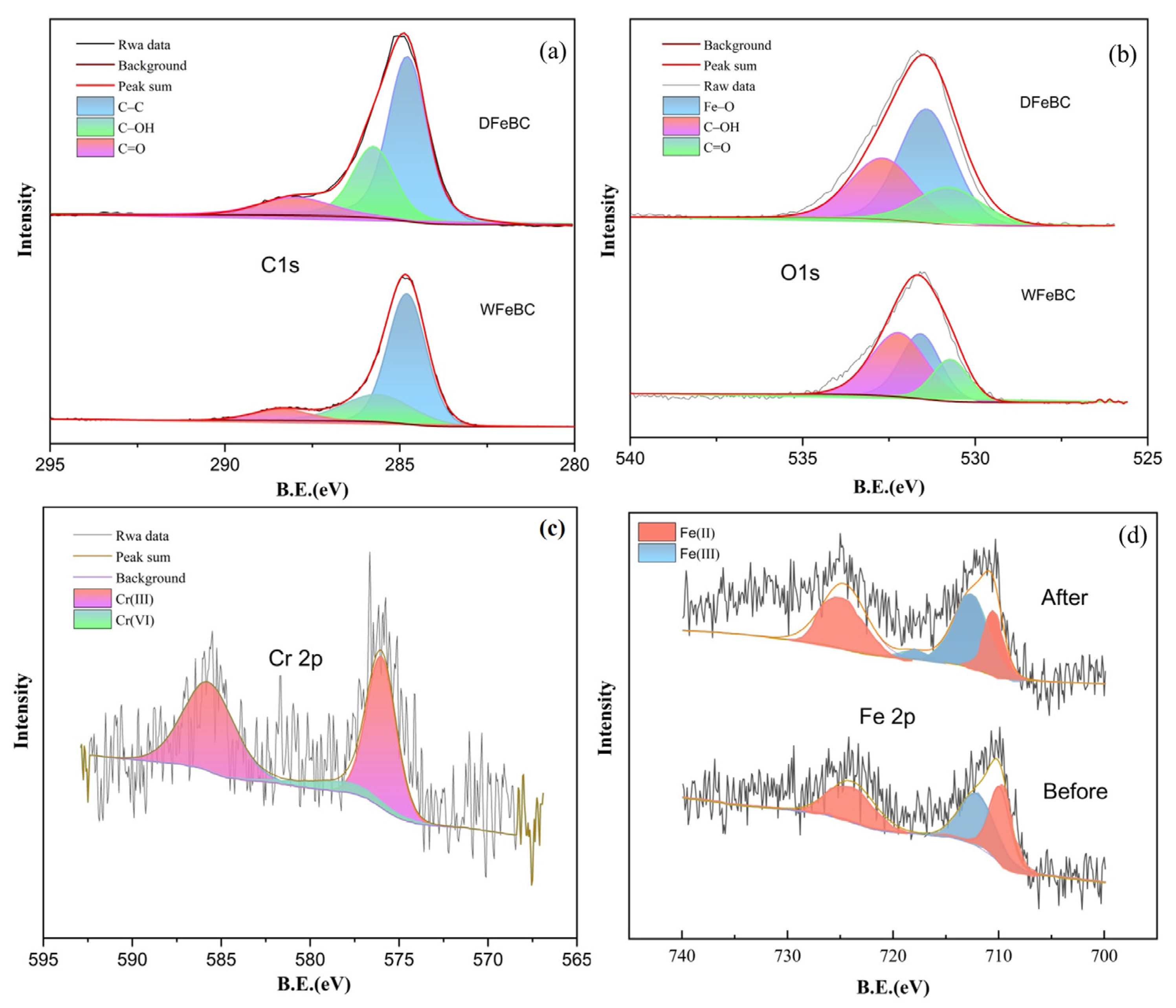
3.5. Adsorption Mechanism
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Fenti, A.; Chianese, S.; Iovino, P.; Musmarra, D.; Salvestrini, S. Cr(VI) Sorption from Aqueous Solution: A Review. Appl. Sci. 2020, 10, 6477. [Google Scholar] [CrossRef]
- Enniya, I.; Rghioui, L.; Jourani, A. Adsorption of hexavalent chromium in aqueous solution on activated carbon prepared from apple peels. Sustain. Chem. Pharm. 2018, 7, 9–16. [Google Scholar] [CrossRef]
- Tran, H.V.; Tran, T.L.; Le, T.D.; Le, T.D.; Nguyen, H.M.T.; Dang, L.T. Graphene oxide enhanced adsorption capacity of chitosan/magnetite nanocomposite for Cr(VI) removal from aqueous solution. Mater. Res. Express 2018, 6, 025018. [Google Scholar] [CrossRef]
- Mikhaylov, V.I.; Maslennikova, T.P.; Krivoshapkina, E.F.; Tropnikov, E.M.; Krivoshapkin, P.V. Express Al/Fe oxide–oxyhydroxide sorbent systems for Cr(VI) removal from aqueous solutions. Chem. Eng. J. 2018, 350, 344–355. [Google Scholar] [CrossRef]
- Kumar, K.Y.; Muralidhara, H.B.; Nayaka, Y.A.; Balasubramanyam, J.; Hanumanthappa, H. Low-cost synthesis of metal oxide nanoparticles and their application in adsorption of commercial dye and heavy metal ion in aqueous solution. Powder Technol. 2013, 246, 125–136. [Google Scholar] [CrossRef]
- Bagbi, Y.; Sarswat, A.; Mohan, D.; Pandey, A.; Solanki, P.R. Lead and Chromium Adsorption from Water using L-Cysteine Functionalized Magnetite (Fe3O4) Nanoparticles. Sci. Rep. 2017, 7, 7672. [Google Scholar] [CrossRef]
- Kerur, S.S.; Bandekar, S.; Hanagadakar, M.S.; Nandi, S.S.; Ratnamala, G.M.; Hegde, P.G. Removal of hexavalent Chromium-Industry treated water and Wastewater: A review. Mater. Today Proc. 2021, 42, 1112–1121. [Google Scholar] [CrossRef]
- Peng, H.; Guo, J. Removal of chromium from wastewater by membrane filtration, chemical precipitation, ion exchange, adsorption electrocoagulation, electrochemical reduction, electrodialysis, electrodeionization, photocatalysis and nanotechnology: A review. Environ. Chem. Lett. 2020, 18, 2055–2068. [Google Scholar] [CrossRef]
- Aryee, A.A.; Dovi, E.; Li, Q.; Han, R.; Li, Z.; Qu, L. Magnetic biocomposite based on peanut husk for adsorption of hexavalent chromium, Congo red and phosphate from solution: Characterization, kinetics, equilibrium, mechanism and antibacterial studies. Chemosphere 2022, 287 Pt 1, 132030. [Google Scholar] [CrossRef]
- Yin, Z.; Xu, S.; Liu, S.; Xu, S.; Li, J.; Zhang, Y. A novel magnetic biochar prepared by K2FeO4-promoted oxidative pyrolysis of pomelo peel for adsorption of hexavalent chromium. Bioresour. Technol. 2020, 300, 122680. [Google Scholar] [CrossRef]
- Zou, H.; Zhao, J.; He, F.; Zhong, Z.; Huang, J.; Zheng, Y.; Zhang, Y.; Yang, Y.; Yu, F.; Bashir, M.A.; et al. Ball milling biochar iron oxide composites for the removal of chromium (Cr(VI)) from water: Performance and mechanisms. J. Hazard. Mater. 2021, 413, 125252. [Google Scholar] [CrossRef]
- Yao, X.; Ji, L.; Guo, J.; Ge, S.; Lu, W.; Cai, L.; Wang, Y.; Song, W.; Zhang, H. Magnetic activated biochar nanocomposites derived from wakame and its application in methylene blue adsorption. Bioresour. Technol. 2020, 302, 122842. [Google Scholar] [CrossRef]
- Yang, C.X.; Zhu, Q.; Dong, W.P.; Fan, Y.Q.; Wang, W.L. Preparation and Characterization of Phosphoric Acid-Modified Biochar Nanomaterials with Highly Efficient Adsorption and Photodegradation Ability. Langmuir 2021, 37, 9253–9263. [Google Scholar] [CrossRef]
- Huang, X.; Niu, X.; Zhang, D.; Li, X.; Li, H.; Wang, Z.; Lin, Z.; Fu, M. Fate and mechanistic insights into nanoscale zerovalent iron (nZVI) activation of sludge derived biochar reacted with Cr(VI). J. Environ. Manag. 2022, 319, 115771. [Google Scholar] [CrossRef]
- Zhang, J.; Yu, H.; Xu, W.; Shi, H.; Hu, X.; Xu, J.; Lou, L. Adsorption-reduction coupling mechanism and reductive species during efficient florfenicol removal by modified biochar supported sulfidized nanoscale zerovalent iron. Environ. Res. 2023, 216 Pt 4, 114782. [Google Scholar] [CrossRef]
- Liu, Y.; Shan, H.; Pang, Y.; Zhan, H.; Zeng, C. Iron modified chitosan/coconut shell activated carbon composite beads for Cr(VI) removal from aqueous solution. Int. J. Biol. Macromol. 2023, 224, 156–169. [Google Scholar] [CrossRef]
- Shan, A.; Idrees, A.; Zaman, W.Q.; Abbas, Z.; Ali, M.; Rehman, M.S.U.; Hussain, S.; Danish, M.; Gu, X.; Lyu, S. Synthesis of nZVI-Ni@BC composite as a stable catalyst to activate persulfate: Trichloroethylene degradation and insight mechanism. J. Environ. Chem. Eng. 2021, 9, 104808. [Google Scholar] [CrossRef]
- Ayyob, M.; Bakry, W.M.; Alamier, R.S.; Salama, M.S.; El-Shall, F.S. Awad.Remediation of water containing phosphate using ceria nanoparticles decorated partially reduced graphene oxide (CeO2-PRGO) composite. Surf. Interfaces 2022, 31, 102006. [Google Scholar]
- Amr, A.; Ibrahim, R.S.; Salama, S.A.; El-Hakam, A.S.; Khder, A.I.A. Synthesis of 12-tungestophosphoric acid supported on Zr/MCM-41 composite with excellent heterogeneous catalyst and promising adsorbent of methylene blue. Colloids Surf. A Physicochem. Eng. Asp. 2021, 631, 127753. [Google Scholar]
- Younes, Y.A.; Kospa, D.A.; Salama, R.S.; Ahmed, A.I.; Ibrahim, A.A. Hydrophilic candle wastes microcapsules as a thermal energy storage material for all-day steam and electricity cogeneration. Desalination 2023, 550, 116377. [Google Scholar] [CrossRef]
- Kim, Y.; Oh, J.-I.; Vithanage, M.; Park, Y.-K.; Lee, J.; Kwon, E.E. Modification of biochar properties using CO2. Chem. Eng. J. 2019, 372, 383–389. [Google Scholar] [CrossRef]
- Wang, Q.; Zhang, Z.; Xu, G.; Li, G. Magnetic porous biochar with nanostructure surface derived from penicillin fermentation dregs pyrolysis with K2FeO4 activation: Characterization and application in penicillin adsorption. Bioresour. Technol. 2021, 327, 124818. [Google Scholar] [CrossRef] [PubMed]
- Xiong, T.; Yuan, X.; Chen, X.; Wu, Z.; Wang, H.; Leng, L.; Wang, H.; Jiang, L.; Zeng, G. Insight into highly efficient removal of cadmium and methylene blue by eco-friendly magnesium silicate-hydrothermal carbon composite. Appl. Surf. Sci. 2018, 427, 1107–1117. [Google Scholar] [CrossRef]
- Leng, L.; Yuan, X.; Zeng, G.; Shao, J.; Chen, X.; Wu, Z.; Wang, H.; Peng, X. Surface characterization of rice husk bio-char produced by liquefaction and application for cationic dye (Malachite green) adsorption. Fuel 2015, 155, 77–85. [Google Scholar] [CrossRef]
- Li, R.; Wang, J.J.; Zhou, B.; Zhang, Z.; Liu, S.; Lei, S.; Xiao, R. Simultaneous capture removal of phosphate, ammonium and organic substances by MgO impregnated biochar and its potential use in swine wastewater treatment. J. Clean. Prod. 2017, 147, 96–107. [Google Scholar] [CrossRef]
- Liu, S.; Li, J.; Xu, S.; Wang, M.; Zhang, Y.; Xue, X. A modified method for enhancing adsorption capability of banana pseudostem biochar towards methylene blue at low temperature. Bioresour. Technol. 2019, 282, 48–55. [Google Scholar] [CrossRef]
- Stoia, M.; Istratie, R.; Păcurariu, C. Investigation of magnetite nanoparticles stability in air by thermal analysis and FTIR spectroscopy. J. Therm. Anal. Calorim. 2016, 125, 1185–1198. [Google Scholar] [CrossRef]
- Zhang, Z.; Huang, G.; Zhang, P.; Shen, J.; Wang, S.; Li, Y. Development of iron-based biochar for enhancing nitrate adsorption: Effects of specific surface area, electrostatic force, and functional groups. Sci. Total Environ. 2023, 856 Pt 1, 159037. [Google Scholar] [CrossRef]
- Yang, Q.; Sun, Y.; Sun, W.; Qin, Z.; Liu, H.; Ma, Y.; Wang, X. Cellulose derived biochar: Preparation, characterization and Benzo[a]pyrene adsorption capacity. Grain Oil Sci. Technol. 2021, 4, 182–190. [Google Scholar] [CrossRef]
- Guo, X.; Liu, A.; Lu, J.; Niu, X.; Jiang, M.; Ma, Y.; Liu, X.; Li, M. Adsorption Mechanism of Hexavalent Chromium on Biochar: Kinetic, Thermodynamic, and Characterization Studies. ACS Omega 2020, 5, 27323–27331. [Google Scholar] [CrossRef]
- Dong, H.; Deng, J.; Xie, Y.; Zhang, C.; Jiang, Z.; Cheng, Y.; Hou, K.; Zeng, G. Stabilization of nanoscale zero-valent iron (nZVI) with modified biochar for Cr(VI) removal from aqueous solution. J. Hazard. Mater. 2017, 332, 79–86. [Google Scholar] [CrossRef]
- He, R.; Yuan, X.; Huang, Z.; Wang, H.; Jiang, L.; Huang, J.; Tan, M.; Li, H. Activated biochar with iron-loading and its application in removing Cr (VI) from aqueous solution. Colloids Surf. A Physicochem. Eng. Asp. 2019, 579, 123642. [Google Scholar] [CrossRef]
- Dobrzyńska, J.; Wysokińska, A.; Olchowski, R. Raspberry stalks-derived biochar, magnetic biochar and urea modified magnetic biochar—Synthesis, characterization and application for As(V) and Cr(VI) removal from river water. J. Environ. Manag. 2022, 316, 115260. [Google Scholar] [CrossRef]
- Huang, D.; Liu, C.; Zhang, C.; Deng, R.; Wang, R.; Xue, W.; Luo, H.; Zeng, G.; Zhang, Q.; Guo, X. Cr(VI) removal from aqueous solution using biochar modified with Mg/Al-layered double hydroxide intercalated with ethylenediaminetetraacetic acid. Bioresour. Technol. 2019, 276, 127–132. [Google Scholar] [CrossRef]
- Wang, F.; Liu, L.-Y.; Liu, F.; Wang, L.-G.; Ouyang, T.; Chang, C.-T. Facile one-step synthesis of magnetically modified biochar with enhanced removal capacity for hexavalent chromium from aqueous solution. J. Taiwan Inst. Chem. Eng. 2017, 81, 414–418. [Google Scholar] [CrossRef]
- Zhang, X.; Lv, L.; Qin, Y.; Xu, M.; Jia, X.; Chen, Z. Removal of aqueous Cr(VI) by a magnetic biochar derived from Melia azedarach wood. Bioresour. Technol. 2018, 256, 1–10. [Google Scholar] [CrossRef]
- Shi, S.; Yang, J.; Liang, S.; Li, M.; Gan, Q.; Xiao, K.; Hu, J. Enhanced Cr (VI) removal from acidic solutions using biochar modified by Fe3O4@ SiO2-NH2 particles. Sci. Total Environ. 2018, 628, 499–508. [Google Scholar] [CrossRef]
- Dong, X.; Ma, L.Q.; Li, Y. Characteristics and mechanisms of hexavalent chromium removal by biochar from sugar beet tailing. J. Hazard. Mater. 2011, 190, 909–915. [Google Scholar] [CrossRef]
- Zhou, Z.; Liu, P.; Wang, S.; Finfrock, Y.Z.; Ye, Z.; Feng, Y.; Li, X. Iron-modified biochar-based bilayer permeable reactive barrier for Cr(VI) removal. J. Hazard. Mater. 2022, 439, 129636. [Google Scholar] [CrossRef]




| Adsorbent | Langmuir Isothermal Adsorption Model | Freundlich Isothermal Adsorption Model | ||||
|---|---|---|---|---|---|---|
| b | R2 | Qm/(mg/g) | Kf | R2 | n | |
| DFeBC | 0.586 | 0.877 | 48.54 | 18.68 | 0.9626 | 3.58 |
| WFeBC | 0.228 | 0.948 | 66.22 | 16.81 | 0.957 | 2.84 |
| Materials | pH | Qe(mg/g) | Reference |
|---|---|---|---|
| BC-Fe-U | 2.3–5.7 | 19.2 | [33] |
| BC@EDTA-LDH | 3 | 38.0 | [34] |
| NMBC | 2 | 48 | [35] |
| Fe/PBC-ND | 2 | 25.68 | [36] |
| Magnetic biochar | 1 | 27.2 | [37] |
| WFeBC | 2 | 66.22 | This work. |
| Adsorbent | Pseudo-First-Order Model | Pseudo-Second-Order Model | ||
|---|---|---|---|---|
| k1/min−1 | R2 | K2/(g/mg·min) | R2 | |
| DFeBC | 0.0023 | 0.9135 | 0.0197 | 0.9567 |
| WFeBC | 0.0028 | 0.6798 | 0.0027 | 0.9993 |
| C0 (mg/L) | 298 K | 308 K | 318 K | ||
|---|---|---|---|---|---|
| ΔH | ΔS | ΔG | ΔG | ΔG | |
| 5 | 32.0164 | 0.2004 | −27.7129 | −29.7173 | −31.7216 |
| 10 | 28.5312 | 0.1849 | −26.5651 | −28.4140 | −30.2628 |
| 15 | 35.2372 | 0.1894 | −21.1994 | −23.0932 | −24.9871 |
| 20 | 38.4897 | 0.1997 | −21.0290 | −23.0263 | −25.0236 |
| 25 | 27.9982 | 0.1581 | −19.1227 | −20.7040 | −22.2852 |
| 30 | 27.0288 | 0.1539 | −18.8211 | −20.3597 | −21.8983 |
| 50 | 34.7758 | 0.1767 | −17.8850 | −19.6521 | −21.4193 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, A.; Wang, X.; Liu, X.; He, C. Wet-Process-Modified Blue-Green Algae Biochar by K2FeO4 for the Efficient Adsorption of Cr(VI) from Water. Processes 2023, 11, 1489. https://doi.org/10.3390/pr11051489
Cheng A, Wang X, Liu X, He C. Wet-Process-Modified Blue-Green Algae Biochar by K2FeO4 for the Efficient Adsorption of Cr(VI) from Water. Processes. 2023; 11(5):1489. https://doi.org/10.3390/pr11051489
Chicago/Turabian StyleCheng, Aihua, Xingwen Wang, Xiaohe Liu, and Chi He. 2023. "Wet-Process-Modified Blue-Green Algae Biochar by K2FeO4 for the Efficient Adsorption of Cr(VI) from Water" Processes 11, no. 5: 1489. https://doi.org/10.3390/pr11051489
APA StyleCheng, A., Wang, X., Liu, X., & He, C. (2023). Wet-Process-Modified Blue-Green Algae Biochar by K2FeO4 for the Efficient Adsorption of Cr(VI) from Water. Processes, 11(5), 1489. https://doi.org/10.3390/pr11051489







