Exopolysaccharides of Fungal Origin: Properties and Pharmaceutical Applications
Abstract
1. Introduction
2. Methodology
3. Results on Pharmaceutical Properties and Applications of Fungal Polysaccharides
3.1. Pullulan
3.2. Schizophyllan
3.3. Scleroglucan
3.4. Botryosphaeran
3.5. Lentinan
3.6. Grifolan
3.7. Lasiodiplodan
4. Extraction and Analytical Methods of Fungal Exopolysaccharides
5. Discussions
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- van Dam, J.E.G.; van den Broek, L.A.M.; Boeriu, C.G. Polysaccharides in human health care. Nat. Prod. Commun. 2017, 12, 821–830. [Google Scholar] [CrossRef]
- Naranjo-Ortiz, M.A.; Gabaldon, T. Fungal evolution: Major ecological adaptations and evolutionary transitions. Biol. Rev. 2019, 94, 1443–1476. [Google Scholar] [CrossRef] [PubMed]
- Giavasis, I. Bioactive fungal polysaccharides as potential functional ingredients in food and nutraceuticals. Curr. Opin. Biotechnol. 2014, 26, 162–173. [Google Scholar] [CrossRef]
- Jaroszuk, M.O.; Wilkolazka, A.J.; Ścisel, J.J.; Szalapata, K.; Nowak, A.; Jaszek, M.; Ozimek, E.; Majewska, M. Extracellular polysaccharides from Ascomycota and Basidiomycota: Production conditions, biochemical characteristics, and biological properties. World J. Microbiol. Biotechnol. 2015, 31, 1823–1844. [Google Scholar] [CrossRef]
- Mahapatra, S.; Banerjee, D. Fungal exopolysaccharide: Production, composition and applications. Microbiol. Insights 2013, 6, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Herrera, J.R.; Ortiz-Castellanos, L. Cell wall glucans of fungi. A review. Cell Surf. 2019, 5, 100022. [Google Scholar] [CrossRef]
- Złotko, K.; Wiater, A.; Waśko, A.; Pleszczyńska, M.; Paduch, R.; Jaroszuk-Ściseł, J.; Bieganowski, A. A report on fungal (1→3)-α-d-glucans: Properties, functions and application. Molecules 2019, 24, 3972. [Google Scholar] [CrossRef]
- Chimilovski, J.S.; Habu, S.; Teixeira, R.F.B.; Thomaz-Soccol, V.; Noseda, M.D.; Medeiros, A.B.P.; Pandey, A.; Soccol, C.R. Antitumour activity of Grifola frondosa exopolysaccharides produced by submerged fermentation using sugar cane and soy molasses as carbon sources. Food Technol. Biotechnol. 2011, 49, 359–363. [Google Scholar]
- Özcan, E.; Öner, E.T. Microbial of extracellular polysaccharide production from biomass sources. In Polysaccharides; Ramawat, K., Mérillon, J.M., Eds.; Springer: Berlin/Heidelberg, Germany, 2018; pp. 1–21. [Google Scholar] [CrossRef]
- Venkatachalam, G.; Arumugam, S.; Doble, M. Industrial production and applications of α/β linear and branched glucans. Indian Chem. Eng. 2020, 63, 533–547. [Google Scholar] [CrossRef]
- Barbosa, J.R.; de Carvalho, R.N., Jr. Occurrence and possible roles of polysaccharides in fungi and their influence on the development of new technologies. Carbohydr. Polym. 2020, 246, 116613. [Google Scholar] [CrossRef]
- Ganie, S.A.; Rather, L.J.; Li, Q. A review on anticancer applications of pullulan and pullulan derivative nanoparticles. Carbohydr. Polym. Technol. Appl. 2021, 2, 116613. [Google Scholar] [CrossRef]
- Sugumaran, K.R.; Ponnusami, V. Review on production, downstream processing and characterization of microbial pullulan. Carbohydr. Polym. 2017, 173, 573–591. [Google Scholar] [CrossRef]
- Singh, R.S.; Saini, G.K.; Kennedy, J.F. Pullulan: Microbial sources, production and applications. Carbohydr. Polym. 2008, 73, 515–531. [Google Scholar] [CrossRef] [PubMed]
- Key, S.W.C.; Dailin, D.J.; Selvamani, S.; Malek, R.A.; Sukmawati, D.; El Enshasy, H. Pullulan production in submerged cultivation: A review. J. Crit. Rev. 2020, 7, 3220–3231. [Google Scholar]
- Dailin, D.J.; Low, L.Z.M.I.; Kumar, K.; Malek, R.A.; Natasya, K.H.; Keat, H.C.; Sukmawati, D.; El Enshasy, H. Agro-industrial waste: A potential feedstock for pullulan production. Biosci. Biotechnol. Res. Asia. 2019, 16, 229–250. [Google Scholar] [CrossRef]
- Akdeniz Oktay, B.; Bozdemir, M.T.; Özbaş, Z.Y. Evaluation of some agro-industrial wastes as fermentation medium for pullulan production by Aureobasidium pullulans AZ-6. Curr. Microbiol. 2022, 79, 93. [Google Scholar] [CrossRef]
- Thirumavalavan, K.; Manikkadan, T.R.; Dhanasekar, R. Pullulan production from coconut by-products by Aureobasidium pullulans. Afr. J. Biotechnol. 2009, 8, 254–258. [Google Scholar]
- Kumar, A.S.; Mody, K.; Jha, B. Bacterial exopolysaccharides-a perception. J. Basic Microbiol. 2007, 47, 103–117. [Google Scholar] [CrossRef]
- West, T.P. Production of the polysaccharide pullulan by Aureobasidium pullulans cell immobilization. Polysaccharides 2022, 3, 544–555. [Google Scholar] [CrossRef]
- Kumar, D.; Saini, N.; Pandit, V.; Ali, S. An insight to pullulan: A biopolymer in pharmaceutical approaches. Int. J. Basic Appl. Sci. 2012, 1, 202–219. [Google Scholar] [CrossRef]
- Thangavelu, M.; Kulandhaivelu, S.V. Development and Characterization of Pullulan-Carboxymethyl cellulose blend film for packaging applications. Int. J. Polym. Sci. 2022, 2022, 9649726. [Google Scholar] [CrossRef]
- Singh, R.S.; Kaur, N.; Hassan, M.; Kennedy, J.F. Pullulan in biomedical research and development—A review. Int. J. Biol. Macromol. 2021, 166, 694–706. [Google Scholar] [CrossRef] [PubMed]
- Raychaudhuri, R.; Naik, S.; Shreya, A.B.; Kandpal, N.; Pandey, A.; Kalthur, G.; Mutalik, S. Pullulan based stimuli responsive and sub cellular targeted nanoplatforms for biomedical application: Synthesis, nanoformulations and toxicological perspective. Int. J. Biol. Macromol. 2020, 161, 1189–1205. [Google Scholar] [CrossRef] [PubMed]
- Rekha, M.R.; Chandra, P.S. Pullulan as a promising biomaterial for biomedical applications: A perspective. Trends Biomater. Artif. Organs 2007, 20, 111–116. [Google Scholar]
- Coltelli, M.B.; Danti, S.; de Clerck, K.; Lazzeri, A.; Morganti, P. Pullulan for advanced sustainable body- and skin-contact applications. J. Funct. Biomater. 2020, 11, 20. [Google Scholar] [CrossRef]
- Moscovici, M. Present and future medical applications of microbial exopolysaccharides. Front. Microbiol. 2015, 6, 1012. [Google Scholar] [CrossRef]
- Ferreira, L.M.; Velasquez, A.A.; Schaffazick, S.R.; Cruz, L. Pullulan: An advantageous natural polysaccharide excipient to formulate tablets of alendronate-loaded microparticles. Braz. J. Pharm. Sci. 2015, 51, 28–33. [Google Scholar] [CrossRef]
- Cheng, K.C.; Demirci, A.; Catchmark, J.M. Pullulan: Biosynthesis, production, and applications. Appl. Microbiol. Biotechnol. 2011, 92, 29–44. [Google Scholar] [CrossRef]
- Singh, R.S.; Kaur, N.; Kennedy, J.F. Pullulan and pullulan derivatives as promising biomolecules for drug and gene targeting. Carbohydr. Polym. 2015, 123, 190–207. [Google Scholar] [CrossRef]
- Li, H.; Xue, Y.; Jia, B.; Bai, Y.; Zuo, Y.; Wang, S.; Zhao, Y.; Yang, W.; Tang, H. The preparation of hyaluronic acid grafted pullulan polymers and their use in the formation of novel biocompatible wound healing film. Carbohydr. Polym. 2018, 188, 92–100. [Google Scholar] [CrossRef]
- Singh, S.; Kaur, N.; Rana, V.; Kennedy, J.F. Recent insights on applications of pullulan in tissue engineering. Carbohydr. Polym. 2016, 153, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.S.; Kaur, N.; Rana, V.; Kennedy, J.F. Pullulan: A novel molecule for biomedical applications. Carbohydr. Polym. 2017, 171, 102–121. [Google Scholar] [CrossRef]
- Mishra, B.; Vuppu, S.; Rath, K. The role of microbial pullulan, a biopolymer in pharmaceutical approaches: A review. J. Appl. Pharm. Sci. 2011, 1, 45–50. [Google Scholar]
- Wu, D.; Chen, Y.; Wen, S.; Wen, Y.; Wang, R.; Zhang, Q.; Qin, G.; Yi, H.; Wu, M.; Lu, L.; et al. Synergistically enhanced inhibitory effects of pullulan nanoparticle-mediated co-delivery of lovastatin and doxorubicin totriple-negative breast cancer cells. Nanoscale Res. Lett. 2019, 14, 1–12. [Google Scholar] [CrossRef]
- Wu, S.; Yang, X.; Yang, X. Methotrexate and 10-hydroxycamptothecine loaded pullulan nanoparticles with the targeting property for efficient cancer therapy. Mater. Technol. 2022, 37, 2777–2784. [Google Scholar] [CrossRef]
- Rekha, M.R.; Pal, K.; Bala, P.; Shetty, M.; Mittra, I.; Bhuvaneshwar, G.S.; Sharma, C.P. Pullulan-histone antibody nanoconjugates for the removal of chromatin fragments from systemic circulation. Biomaterials 2013, 34, 6328–6338. [Google Scholar] [CrossRef]
- Coseri, S.; Spatareanu, A.; Sacarescu, L.; Rimbu, C.; Suteu, D.; Spik, S.; Harabagiu, V. Green synthesis of the silver nanoparticles mediated by pullulan and 6-carboxypullulan. Carbohydr. Polym. 2015, 116, 9–17. [Google Scholar] [CrossRef]
- Boridy, S.; Takahashi, H.; Akiyoshi, K.; Maysinger, D. The binding of pullulan modified cholesteryl nanogels to Aβ oligomers and their suppression of cytotoxicity. Biomaterials 2009, 30, 5583–5591. [Google Scholar] [CrossRef]
- Kobayashi, H.; Katakura, O.; Morimoto, N.; Akiyoshi, K.; Kasugai, S. Effects of colesterol-bearing pullulan (CHP)-nanogels in combination with prostaglandin E1 wound healing. J. Biomed. Mater. Res. B Appl. Biomater. 2009, 91, 55–60. [Google Scholar] [CrossRef]
- Tabernero, A.; Cardea, S. Microbial exopolysaccharides as drug carriers. Polymers 2020, 12, 2142. [Google Scholar] [CrossRef]
- Iswariya, S.; Bhanukeerthi, A.V.; Velswamy, P.; Uma, T.S.; Perumal, P.T. Design and development of a piscine collagen blended pullulan hydrogel for skin tissue engineering. RSC Adv. 2016, 6, 57863–57871. [Google Scholar] [CrossRef]
- Mahmoud, Y.A.G.; El-Naggar, M.E.; Abdel-Megeed, A.; El-Newehy, M. Recent advancements in microbial polysaccharides: Synthesis and applications. Polymers 2021, 13, 4136. [Google Scholar] [CrossRef]
- Kumar, A.; Bharti, A.K.; Bezie, Y. Schizophyllum commune: A fungal cell-factory for production of valuable metabolites and enzymes. BioResources 2022, 17, 5420–5436. [Google Scholar] [CrossRef]
- Zhong, K.; Liu, L.; Tong, L.; Zhong, X.; Wang, Q.; Zhou, S. Rheological properties and antitumor produced activity of schizophyllan with solid-state fermentation. Int. J. Biol. Macromol. 2013, 62, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Kumari, M.; Survase, S.A.; Singhal, R.S. Production of schizophyllan using Schizophyllum commune NRCM. Bioresour Technol. 2008, 99, 1036–1043. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, A.; Shojaosadati, S.A.; Tehrani, H.J.; Mousavi, S.M.; Saleh, T.; Khorasani, A.K. Schizophyllan production by newly isolated fungus Schizophyllum commune IBRC-M 30213: Optimization of culture medium using response surface methodology. Ann. Microbiol. 2018, 68, 47–62. [Google Scholar] [CrossRef]
- Teoh, Y.P.; Don, M.M. Optimization of parameters for mycelia growth by Schizophyllum commune and a kinetic model study of its growth morphology. J. Appl. Sci. 2012, 12, 1100–1105. [Google Scholar] [CrossRef]
- Mironczuk-Chodakowska, I.; Kujawowicz, K.; Witkowska, A.M. Beta-glucans from fungi: Biological and health-promoting potential in the COVID-19 Pandemic Era. Nutrients 2021, 13, 3960. [Google Scholar] [CrossRef]
- Abdel-Mohsen, A.M.; Abdel-Rahman, R.M.; Fouda, M.M.G.; Vojtova, L.; Uhrova, L.; Hassan, A.F.; Al-Deyab, S.S.; El-Shamy, I.E.; Jancar, J. Preparation, characterization and cytotoxicity of schizophyllan/silver nanoparticle composite. Carbohydr. Polym. 2014, 102, 238–245. [Google Scholar] [CrossRef]
- Saetang, N.; Ramaraj, R.; Unpaprom, Y. Optimization of ethanol precipitation of schizophyllan from Schizophyllum commune by applied statistical modelling. Biomass Convers. Biorefin. 2022, 1–13. [Google Scholar] [CrossRef]
- Zhang, Y.; Kong, H.; Fang, Y.; Nishinari, K.; Phillips, G.O. Schizophyllan: A review of its structure, properties, bioactivity and recent developments. Bioact. Carbohydr. Diet. Fibre 2013, 1, 53–71. [Google Scholar] [CrossRef]
- Zhao, S.; Gao, Q.; Rong, C.; Wang, S.; Zhao, Z.; Liu, Y. Immunomodulatory effects of edible and medicinal mushrooms and their bioactive immunoregulatory products. J. Fungi. 2020, 6, 269. [Google Scholar] [CrossRef]
- Mansour, A.; Daba, A.; Baddour, N.; El-Saadani, M.; Aleem, E. Schizophyllan inhibits the development of mammary and hepatic carcinomas induced by 7,12-dimethylbenz(α)anthracene and decreases cell proliferation: Comparison with tamoxifen. J. Cancer Res. Clin. Oncol. 2012, 138, 1579–1596. [Google Scholar] [CrossRef]
- Zhou, B.; Fu, Q.; Song, S.S.; Zheng, H.L.; Wei, Y.Z. Inhibitory effect of schizophyllan on rat glioma cells. Bangladesh J. Pharmacol. 2015, 10, 759–764. [Google Scholar] [CrossRef]
- Thongsiri, C.; Nagai-Yoshioka, Y.; Yamasaki, R.; Adachi, Y.; Usui, M.; Nakashima, K.; Nishihara, T.; Ariyoshi, W. Schizophyllum commune β-glucan: Effect on interleukin-10 expression induced by lipopolysaccharide from periodontopathic bacteria. Carbohydr. Polym. 2021, 253, 117285. [Google Scholar] [CrossRef]
- Lee, S.; Ki, C.S. Inflammatory responses of macrophage-like RAW264. 7 cells in a 3D hydrogel matrix to ultrasonicated schizophyllan. Carbohydr. Polym. 2020, 229, 115555. [Google Scholar] [CrossRef]
- Atiq, A.; Parhar, I. Anti-neoplastic potential of flavonoids and polysaccharide phytochemicals in glioblastoma. Molecules 2020, 25, 4895. [Google Scholar] [CrossRef]
- Matsumoto, T.; Numata, M.; Anada, T.; Mizu, M.; Koumoto, K.; Sakurai, K.; Nagasaki, T.; Shinkai, S. Chemically modified polysaccharide schizophyllan for antisense oligonucleotides delivery to enhance the cellular uptake efficiency. Biochim. Biophys. Acta 2004, 1670, 91–104. [Google Scholar] [CrossRef]
- Takedatsu, H.; Mitsuyama, K.; Mochizuki, S.; Kobayashi, T.; Sakurai, K.; Takeda, H.; Fujiyama, Y.; Koyama, Y.; Nishihira, J.; Sata, M. A new therapeutic approach using a schizophyllan-based drug delivery system for inflammatory bowel disease. Mol. Ther. 2012, 20, 1234–1241. [Google Scholar] [CrossRef]
- Bai, T.; Wang, T.; Li, Y.; Gao, N.L.; Zhang, L.; Chen, W.H.; Yin, X. Optimization of scleroglucan production by Sclerotium rolfsii by lowering pH during fermentation via oxalate metabolic pathway manipulation using CRISPR/Cas9. Fungal Biol. Biotechnol. 2021, 8, 1–9. [Google Scholar] [CrossRef]
- Survase, S.A.; Saudagar, S.; Bajaj, I.B.; Singhal, R.S. Scleroglucan: Fermentative production, downstream processing and applications. Food Technol. Biotechnol. 2007, 45, 107–118. [Google Scholar]
- Fosmer, A.; Gibbons, W.R.; Heisel, N.J. Reducing the cost of scleroglucan production by use of a condensed corn solubles medium. J. Biotech. Res. 2010, 2, 131. [Google Scholar]
- Zeng, W.; Wang, J.; Shan, X.; Yu, S.; Zhou, J. Efficient production of scleroglucan by Sclerotium rolfsii and insights into molecular weight modification by high-pressure homogenization. Front. Bioeng. Biotechnol. 2021, 9, 799. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Lu, Y.; Adams, G.G.; Zobel, H.; Ballance, S.; Wolf, B.; Harding, S.E. Characterisation of the molecular properties of scleroglucan as an alternative rigid rod molecule to xanthan gum for oropharyngeal dysphagia. Food Hydrocoll. 2020, 101, 105446. [Google Scholar] [CrossRef]
- Marchetti, M.; Pisani, S.; Petropaolo, V.; Seganti, L.; Nicoletti, R.; Degener, A.; Orsi, N. Antiviral effect of polysaccha-ride from Sclerotium glucanicum towards herpes simplex virus type I infection. Planta Med. 1996, 62, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Mastromarino, P.; Petruzziello, R.; Macchia, S.; Rieti, S.; Nicoletti, R.; Orsi, N. Antiviral activity of natural and semi-synthetic polysaccharides on early steps of rubella virus infection. J. Antimicrob. Chemother. 1997, 39, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Manjanna, K.M.; Shivakumar, B.; Pramodkumar, T.M. Natural exopolysaccharides as novel excipients in drug delivery: A review. Arch. Appl. Sci. Res. 2009, 1, 230–253. [Google Scholar]
- Coviello, T.; Palleschi, A.; Matricardi, P.; Bocchinfuso, G.; Grassi, M.; Alhaique, F. Scleroglucan: A versatile polysaccharide for modified drug delivery. Molecules 2005, 10, 6–33. [Google Scholar] [CrossRef]
- Rizk, S.; Duru, C.; Gaudy, D.; Jacob, M.; Ferrari, F.; Bertoni, M.; Caramella, C. Physico-chemical characterization andtabletting properties of scleroglucan. Int. J. Pharm. 1994, 112, 125–130. [Google Scholar] [CrossRef]
- Vinarta, S.C.; Francois, N.J.; Daraio, M.E.; Figueroa, L.I.C.; Farina, J.I. Sclerotium rolfsii scleroglucan: The promising behavior of a natural polysaccharide as a drug delivery vehicle, suspension stabilizer and emulsifier. Int. J. Biol. Macromol. 2007, 41, 314–323. [Google Scholar] [CrossRef]
- Geraldelli, D.; Ribeiro, M.C.; Madeiros, T.C.; Comiran, P.K.; Martins, K.O.; Oliveira, M.F.; Oliveira, G.A.; Dekker, R.F.H.; Barbosa-Dekker, A.M.; Alegranci, P.; et al. Botryosphaeran, a (1→3)(1→6)-β-D-glucan, reduces tumor development and cachexia syndrome in obese male rats by increasing insulin sensitivity and FOXO3a activity. Int. J. Biol. Macromol. 2020, 165, 985–994. [Google Scholar] [CrossRef] [PubMed]
- Giese, E.C.; Dekker, R.F.H.; Barbosa, A.M.; Silva, R. Triple helix conformation of botryosphaeran, a (1→3; 1→6)-β-D-glucan produced by Botryosphaeria rhodina MAMB-05. Carbohydr. Polym. 2008, 74, 953–956. [Google Scholar] [CrossRef]
- Giese, E.C.; Sumiya, A.F.G.; Borsato, D.; Dekker, R.F.H.; Barbosa, A.M. Evaluation of fermentative parameters for the production of botryosphaeran (a (1→3,1→6)- β-D-glucan) and mycelial biomass by Botryosphaeria rhodina MAMB-05. Orbital Electron. J. Chem. 2015, 7, 36–43. [Google Scholar] [CrossRef]
- Fonseca, P.R.M.S.; Dekker, R.F.H.; Barbosa, A.M.; Silveira, J.L.M.; Vasconcelos, A.F.D.; Monteiro, N.K.; Aranda-Silverio, G.; Corradi da Silva, M.L. Thermal and rheological properties of a family of botryosphaerans produced by Botryosphaeria rhodina MAMB-05. Molecules 2011, 16, 7488–7501. [Google Scholar] [CrossRef] [PubMed]
- Dekker, R.F.H.; Queiroz, E.A.I.F.; Cunha, M.A.A.; Barbosa-Dekker, A.M. Botryosphaeran - A fungal exopolysaccharide of the (1→3)(1→6)-β-d-Glucan kind: Structure and biological functions. In Extracellular Sugar-Based Biopolymers Matrices. Biologically-Inspired Systems; Cohen, E., Merzendorfer, H., Eds.; Springer: Berlin/Heidelberg, Germany, 2019; Volume 12; pp. 433–484. [Google Scholar]
- Dekker, R.F.H.; Barbosa-Dekker, A.M. Botryosphaeran. An unusual exocellular fungal (1→3)(1→6)-β-D-Glucan with notable biomedical applications. In Polysaccharides of Microbial Origin; Oliveira, J., Radhouani, H., Reis, R.L., Eds.; Springer International Publishing: Berlin/Heidelberg, Germany, 2021; pp. 1–17. [Google Scholar]
- Miranda, C.C.B.O.; Dekker, R.F.H.; Serpeloni, J.M.; Fonseca, E.A.L.; Colus, I.M.S.; Barbosa, A.M. Anticlastogenic activity exhibited by botryosphaeran, a new exopolysaccharide produced by Botryosphaeria rhodina MAMB-05. Int. J. Biol. Macromol. 2008, 42, 172–177. [Google Scholar] [CrossRef]
- Weng, B.B.C.; Lin, Y.C.; Hu, C.W.; Kao, M.Y.; Wang, S.H.; Lo, D.Y.; Lai, T.; Kan, L.S.; Chiou, R.Y.Y. Toxicological and immunomodulatory assessments of botryosphaeran (β-glucan) produced by Botryosphaeria rhodina RCYU 30101. Food Chem. Toxicol. 2011, 49, 910–916. [Google Scholar] [CrossRef] [PubMed]
- Miranda-Nantes, C.C.B.O.; Fonseca, E.A.I.; Zaia, C.T.B.V.; Dekker, R.F.H.; Khaper, N.; Castro, I.A.; Barbosa, A.M. Hypoglycemic and hypocholesterolemic effects of botryosphaeran from Botryosphaeria rhodina MAMB-05 in diabetes induced and hyperlipidemia conditions in rats. Mycobiology 2011, 39, 187–193. [Google Scholar] [CrossRef]
- Silva-Sena, G.G.; Malini, M.; Delarmelina, J.M.; Dutra, J.C.V.; Gervasio, S.V.; Leal, M.A.S.; Pereira, T.M.C.; Barbosa-Dekker, A.M.; Dekker, R.F.H.; de Paula, F.; et al. In vivo antimutagenic and antiatherogenic effects of the (1→3)(1→6)-β-D-glucan botryosphaeran. Mutat. Res. /Genet. Toxicol. Environ. 2018, 826, 6–14. [Google Scholar] [CrossRef]
- Brandi, J. Chemical modification of botryosphaeran: Structural characterization and anticoagulant activity of a water-soluble sulfonated (1→3)(1→6)-β-D-glucan. J. Microbiol. Biotechnol. 2011, 21, 1036–1042. [Google Scholar] [CrossRef]
- Sacchelli, B.A.L.; Faccin-Galhardi, L.C.; Ito, V.Y.; Lopes, J.L.; Dekker, R.F.H.; Barbosa-Dekker, A.M.; Orsato, A. Botryosphaeran and sulfonated derivatives as novel antiviral agents for herpes simplex and dengue fever. Int. J. Biol. Macromol. 2019, 138, 334–339. [Google Scholar] [CrossRef]
- Queiroz, A.I.F.; Fortes, Z.B.; Cunha, M.A.A.; Barbosa, A.M.; Khaper, N.; Dekker, R.F.H. Antiproliferative and pro-apoptotic effects of three fungal exocellular-glucans in MCF-7 breast cancer cells is mediated by oxidative stress, AMP-activated protein kinase (AMPK) and the Forkhead transcription factor, FOXO3a. Int. J. Biochem. Cell Biol. 2015, 67, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Malini, M.; Souza, M.F.; Oliveira, M.T.; Antunes, L.M.G.; Figueiredo, S.G.; Barbosa, A.M.; Dekker, R.F.H.; Cólus, I.M.S. Modulation of gene expression and cell cycle by botryosphaeran, a (1→3) (1→6)-D-glucan in human lymphocytes. Int. J. Biol. Macromol. 2015, 77, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Kerche-Silva, L.E.; Colus, I.M.S.; Malini, M.; Mori, M.P.; Dekker, R.F.H.; Barbosa-Dekker, A.M.B. In vitro protective effects of botryosphaeran, a (1→3;1→6)-d-glucan, against mutagens in normal and tumor rodent cells. Mutat. Res. 2017, 814, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Fujiike, A.Y.; Lee, C.Y.A.L.; Rodrigues, F.S.T.; Oliveira, L.C.B.; Barbosa-Dekker, A.M.; Dekker, R.F.H.; Colus, I.M.S.; Serpeloni, J.M. Anticancer effects of carboxymethylated (1→3)(1→6)-β-D-glucan (botryosphaeran) on multicellular tumor spheroids of MCF-7 cells as a model of breast cancer. J. Toxicol. Environ. Health 2022, 85, 521–537. [Google Scholar] [CrossRef]
- Silva, N.A.; Pereira, B.G.; Santos, J.A.; Guarnier, F.A.; Barbosa-Dekker, A.M.; Dekker, R.F.H.; Kassuya, C.A.L.; Sara, S.B. Oral administration of botryosphaeran [(1→3) (1→6)-β-D-glucan] reduces inflammation through modulation of leukocytes and has limited effect on inflammatory nociception. Cell Biochem. Funct. 2022, 40, 578–588. [Google Scholar] [CrossRef]
- Jaros, D.; Kobsch, J.; Rohm, H. Exopolysaccharides from Basidiomycota: Formation, isolation and techno-functional properties. Eng. Life Sci. 2018, 18, 743–752. [Google Scholar] [CrossRef]
- Feng, Y.L.; Li, W.Q.; Wu, X.Q.; Cheng, J.W.; Ma, S.Y. Statistical optimization of media for mycelial growth and exo-polysaccharide production by Lentinus edodes and a kinetic model study of two growth morphologies. Biochem. Eng. J. 2010, 49, 104–112. [Google Scholar] [CrossRef]
- Qiuyang, L.; Yuguo, L.; Guoyuan, H.; Yuanzheng, L.; Dongmei, D. Effects of Tween 80 on the liquid fermentation of Lentinus Edodes. Food Sci. Biotechnol. 2018, 27, 1103–1109. [Google Scholar] [CrossRef]
- Bisko, N.; Lomberg, M.; Mustafin, K.; Al-Maali, G.; Suleimenova, Z.; Narmuratova, Z.; Mykchaylova, O.; Mytropolska, N.; Zhakipbekova, A. Effects of cultivation parameters on intracellular polysaccharide production in submerged culture of the edible medicinal mushroom Lentinula edodes. Czech Mycol. 2020, 72, 1–17. [Google Scholar] [CrossRef]
- Sobieralski, K.; Siwulski, M.; Lisiecka, J.; Jedryczka, M.; Golak, I.S.; Jozwiak, D.F. Fungi-derived β-glucans as a component of functional food. Acta Sci. Pol. Hortorum Cultus 2012, 11, 111–128. [Google Scholar]
- Ooi, V.E.C.; Liu, F. Immunomodulation and anti-cancer activity of polysaccharide-protein complexes. Curr. Med. Chem. 2000, 7, 715–729. [Google Scholar] [CrossRef] [PubMed]
- Laroche, C.; Michaud, P. New developments and prospective for (1,3) glucans. Recent Pat. Biotechnol. 2007, 1, 59–73. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, S.; Wang, X.; Zhang, L.; Cheung, P.C.K. Advances in lentinan, isolation, structure, chain conformation and bioactives. Food Hydrocoll. 2011, 25, 196–206. [Google Scholar] [CrossRef]
- Jampilek, J.; Kralova, K. Advances in drug delivery nanosystems using graphene-based materials and carbon nanotubes. Materials 2021, 14, 1059. [Google Scholar] [CrossRef]
- Yagi, M.; Watanabe, S.; Yoshino, S.; Hazama, S.; Suga, T.; Nakazama, S. Provision for adverse effect of S-1 containing chemotherapy in patients with advanced digestive cancer--combination with superfine dispersed lentinan. Gan Kagaku Ryoho. 2010, 37, 457–462. [Google Scholar] [PubMed]
- Liu, W.; Gu, J.; Qi, J.; Zeng, X.N.; Ji, J.; Chen, Z.Z.; Sun, X.L. Lentinan exerts synergistic apoptotic effects with paclitaxel in A549 cells via activating ROS-TXNIP- NLRP3 inflammasome. J. Cell Mol. Med. 2015, 19, 1949–1955. [Google Scholar] [CrossRef]
- Oba, K.; Kobayashi, M.; Matsui, T.; Kodera, Y.; Sakamoto, J. Individual patient based meta-analysis of lentinan for unresectable/recurrent gastric cancer. Anticancer Res. 2009, 29, 2739–2745. [Google Scholar]
- Hori, T.; Ikehara, T.; Takatsuka, S.; Fukuoka, T.; Tendo, M.; Tezuka, K.; Dan, N.; Nishino, H.; Hirakawa, K. Combination chemotherapy of S-1/low-dose CDDP/lentinan for advanced gastric cancer. Gan Kagaku Ryoho. 2011, 38, 293–295. [Google Scholar] [PubMed]
- Ina, K.; Furuta, R.; Kataoka, T.; Kayukawa, S.; Yoshida, T.; Miwa, T.; Yamamura, Y.; Takeuchi, Y. Lentinan prolonged survival in patients with gastric cancer receiving S-1-based chemotherapy. World J. Clin. Oncol. 2011, 2, 339–343. [Google Scholar] [CrossRef]
- Vannucci, L.; Sima, P.; Vetvicka, V.; Křižan, J. Lentinan properties in anticancer therapy: A review on the last 12-year literature. Am. J. Immunol. 2017, 13, 50–61. [Google Scholar] [CrossRef]
- Seo, Y.R.; Patel, D.K.; Shin, W.C.; Sim, W.S.; Lee, O.H.; Lim, K.T. Structural elucidation and immune-enhancing effects of novel polysaccharide from Grifola frondosa. Biomed. Res. Int. 2019, 2019, 7528609. [Google Scholar] [CrossRef]
- Wu, J.Y.; Siu, K.C.; Geng, P. Bioactive ingredients and medicinal values of Grifola frondosa (Maitake). Foods 2021, 10, 95. [Google Scholar] [CrossRef] [PubMed]
- Shih, I.L.; Chou, B.W.; Chen, C.C.; Wu, J.Y.; Hsieh, C. Study of mycelial growth and bioactive polysaccharide production in batch and fed-batch culture of Grifola frondosa. Bioresour. Technol. 2008, 99, 785–793. [Google Scholar] [CrossRef]
- He, Y.; Li, X.; Hao, C.; Zeng, P.; Zhang, M.; Liu, Y.; Chang, Y.; Zhang, L. Grifola frondosa polysaccharide: A review of antitumor and other biological activity studies in China. Discov. Med. 2018, 25, 159–176. [Google Scholar] [PubMed]
- He, Y.; Zhang, L.; Wang, H. The biological activities of the antitumor drug Grifola frondosa polysaccharide. Prog. Mol. Biol. Transl. Sci. 2019, 163, 221–261. [Google Scholar] [CrossRef] [PubMed]
- Masuda, Y.; Inoue, H.; Ohta, H.; Miyake, A.; Konishi, M.; Nanba, H. Oral administration of soluble β-glucans extracted from Grifola frondosa induces systemic antitumor immune response and decreases immunosuppression in tumor-bearing mice. Int. J. Cancer 2013, 133, 108–119. [Google Scholar] [CrossRef] [PubMed]
- Kodama, N.; Murata, Y.; Asakawa, A.; Inui, A.; Hayashi, M.; Sakai, N.; Nanba, H. Maitake D-fraction enhances antitumor effects and reduces immunosuppression by mitomycin-C in tumor-bearing mice. Nutrition 2005, 21, 624–629. [Google Scholar] [CrossRef]
- Nie, X.; Shi, B.; Ding, Y.; Tao, W. Preparation of a chemically sulfated polysaccharide derived from Grifola frondosa and its potential biological activities. Int. J. Biol. Macromol. 2006, 39, 228–233. [Google Scholar] [CrossRef]
- Lema, D.R.; Iglesias, O.M.; Portela, C.F.A.; Blanco, A.R.; Ayerbes, M.V.; Díaz, A.D.; Pais, A.C.; Prego, C.; Figueroa, A. In vitro anti-proliferative and anti-invasive effect of polysaccharide-rich extracts from Trametes Versicolor and Grifola Frondosa in colon cancer cells. Int. J. Med. Sci. 2019, 16, 231–240. [Google Scholar] [CrossRef]
- Shomori, K.; Yamamoto, M.; Arifuku, I.; Teramachi, K.; Ito, H. Antitumor effects of a water-soluble extract from Maitake (Grifola frondosa) on human gastric cancer cell lines. Oncol. Rep. 2009, 22, 615–620. [Google Scholar] [CrossRef]
- Kou, L.; Du, M.; Liu, P.; Zhang, B.; Zhang, Y.; Yang, P.; Shang, M.; Wang, X. Anti-diabetic and anti-nephritic activities of Grifola frondosa mycelium polysaccharides in diet-streptozotocin-induced diabetic rats via modulation on oxidative stress. Appl. Biochem. Biotechnol. 2019, 187, 310–322. [Google Scholar] [CrossRef] [PubMed]
- Wasser, S.P. Medicinal mushrooms as a source of antitumor and immunomodulating polysaccharides. Appl. Microbiol. Biotechnol. 2002, 60, 258–274. [Google Scholar] [CrossRef] [PubMed]
- Mayell, M. Maitake extracts and their therapeutic potential—A Review. Altern. Med. Rev. 2001, 6, 48–60. [Google Scholar] [PubMed]
- Abdeshahian, P.; Ascencio, J.J.; Philippini, R.R.; Antunes, F.A.F.; Abdeshahian, M.; dos Santos, C.J.; da Silva, S.S. Fermentative production of lasiodiplodan by Lasiodiplodia theobromae CCT3966 from pretreated sugarcane straw. Sustainability 2021, 13, 9697. [Google Scholar] [CrossRef]
- Acosta, S.B.P.; Marchioro, M.L.K.; Santos, V.A.Q.; Calegari, G.C.; Lafay, C.B.B.; Barbosa-Dekker, A.M.; Dekker, R.F.H.; Da Cunha, M.A.A. Valorization of soybean molasses as fermentation substrate for the production of microbial exocellular β-Glucan. J. Polym. Environ. 2020, 28, 2149–2160. [Google Scholar] [CrossRef]
- Ascensio, J.J.; Philippini, R.R.; Gomes, F.M.; Pereira, F.M.; da Silva, S.S.; Kumar, V.; Chandel, A.K. Comparative highly efficient production of beta-glucan by Lasiodiplodia theobromae CCT 3966 and its multiscale characterization. Fermentation 2021, 7, 108. [Google Scholar] [CrossRef]
- Renganathan, P.; Karan, R.; Dhaarani, S.; Saravanan, K.R.; Premkumar, R. Effect of different media, temperature, pH, carbon source and nitrogen source on mycelial growth of Lasiodiplodia theobromae causing crown rot of banana. Ann. Rom. Soc. Cell Biol. 2020, 24, 1494–1506. [Google Scholar]
- Mello, M.B.; Machado, C.S.; Ribeiro, D.L.; Aissa, A.F.; Burim, R.V.; Alves da Cunha, M.A.; Barcelos, G.R.M.; Antunes, L.M.G.; Bianchi, M.L.P. Protective effects of the exopolysaccharide lasiodiplodan against DNA damage and inflammation induced by doxorubicin in rats: Cytogenetic and gene expression assays. Toxicology 2017, 376, 66–74. [Google Scholar] [CrossRef]
- Vasconcelos, A.F.D.; Dekker, R.F.H.; Barbosa, A.M.; Carbonero, E.R.; Silveira, J.L.M.; Glauser, B.; Pereira, M.S.; da Silva, C.M.L. Sulfonation and anticoagulant activity of fungal exocellular ß-(1→6)-D-glucan (lasiodiplodan). Carbohydr. Polym. 2013, 92, 1908–1914. [Google Scholar] [CrossRef]
- Turmina, J.A.; Carraro, E.; Cunha, M.A.A.; Dekker, R.F.; Barbosa, A.M.; Santos, F.S.; Silva, L.A.; Malfatti, C.R. Toxicological assessment of β-(1→6)-glucan (lasiodiplodan) in mice during a 28-day feeding study by gavage. Molecules 2012, 17, 14298–14309. [Google Scholar] [CrossRef]
- Alves da Cunha, M.A.; Turmina, J.A.; Ivanov, R.C.; Barroso, R.R.; Marques, P.T.; Fonseca, E.A.; Fortes, Z.B.; Dekker, R.F.H.; Khaper, N.; Barbosa, A.M. Lasiodiplodan, an exocellular (1→6)-D-glucan from Lasiodiplodia theobromae MMPI: Production on glucose, fermentation kinetics, rheology and anti-proliferative activity. J. Ind. Microbiol. Biotechnol. 2012, 39, 1179–1188. [Google Scholar] [CrossRef] [PubMed]
- Kagimura, F.Y.; da Cunha, M.A.A.; Theis, T.V.; Malfatti, C.R.M.; Dekker, R.F.H.; Barbosa, A.M.; Teixeira, S.D.; Salome, K. Carboxymethylation of (1→6)-β-glucan (lasiodiplodan): Preparation, characterization and antioxidant evaluation. Carbohydr. Polym. 2015, 127, 390–399. [Google Scholar] [CrossRef] [PubMed]
- Calegari, G.C.; Santos, V.A.Q.; Teixeira, S.; Barbosa, A.; Dekker, R.F.H.; Alves da Cunha, M.A. Sulfonation of (1→6)-β-D-Glucan (Lasiodiplodan) and its antioxidant and antimicrobial potential. J. Pharm. Pharmacol. 2017, 5, 850–863. [Google Scholar] [CrossRef]
- Luna, W.N.S.; Santos, V.A.Q.; Teixeira, S.D.; Barbosa-Dekker, A.M.; Dekker, R.F.H.; Alves da Cunha, M.A. O-Acetylated (1→6)-β-D-Glucan (Lasiodiplodan): Chemical derivatization, characterization and antioxidant activity. J. Pharm. Pharmacol. 2018, 6, 320–332. [Google Scholar] [CrossRef]
- Malfatti, C.R.M.; dos Santos, F.S.; Wouk, J.; da Silva, L.A.; Michel, R.G.; Snak, A.L.; Czervinski, T.; Alves da Cunha, M.A.; Barbosa, A.M.; Dekker, F.H. Intracerebroventricular administration of the (1-6)-β-d-glucan (lasiodiplodan) in male rats prevents d-penicillamine-induced behavioral alterations and lipoperoxidation in the cortex. Pharm. Biol. 2017, 55, 1289–1294. [Google Scholar] [CrossRef]
- Nissola, C.; Marchioro, M.L.K.; de Souza Leito Mello, E.V.; Guidi, A.C.; de Medeiros, D.C.; da Silva, C.G.; de Mello, J.C.P.; Pereira, E.A.; Barbosa-Dekker, A.M.; Cunha, M.A.A. Hydrogel containing (1→6)-β-D-glucan (lasiodiplodan) effectively promotes dermal wound healing. Int. J. Biol. Macromol. 2021, 183, 316–330. [Google Scholar] [CrossRef]
- Elisashvili, V.I.; Kachlishvili, E.T.; Wasser, S.P. Carbon and nitrogen source effects on basidiomycetes exopolysaccharide production. Appl. Biochem. Microbiol. 2009, 45, 531–535. [Google Scholar] [CrossRef]
- Maziero, R.; Cavazzoni, V.; Bononi, V.L.R. Screening of basidiomycetes for the production of exopolysaccharide and biomass in submerged culture. Rev. Microbiol. 1999, 30, 77–84. [Google Scholar] [CrossRef]
- Kagimura, F.Y.; da Cunha, M.A.A.; Barbosa, A.M.; Dekker, R.F.; Malfatti, C.R.M. Biological activities of derivatized d-glucans: A review. Int. J. Biol. Macromol. 2015, 72, 588–598. [Google Scholar] [CrossRef]
- Osińska-Jaroszuk, M.; Sulej, J.; Jaszek, M.; Jaroszuk-Ściseł, J. Applications of Fungal Polysaccharides. Encycl. Micol. 2021, 2, 613–628. [Google Scholar] [CrossRef]
- United States Pharmacopeia (2022). NF Monographs, Pullulan. Available online: https://www.uspnf.com/sites/default/files/usp_pdf/EN/USPNF/pf-48-1-toc-archive.pdf (accessed on 7 July 2022).
- European Directorate for the Quality of Medicines and Healthcare of the Council of Europe, European Pharmacopoeia (Ph. Eur.). Available online: https://www.edqm.eu/en/european-pharmacopoeia (accessed on 26 July 2022).
- The Japanese Pharmacopoeia Eighteenth Edition (JP18), October 2021, p. 1607, JP18: Japanese Pharmacopoeia Seventeenth Edition (nihs.go.jp). Available online: https://jpdb.nihs.go.jp/kyokuhou/files/000904449.pdf (accessed on 12 August 2022).
- British Pharmacopoeia Commission. Available online: https://www.pharmacopoeia.com/file/NOM---February-2015.pdf (accessed on 22 August 2022).

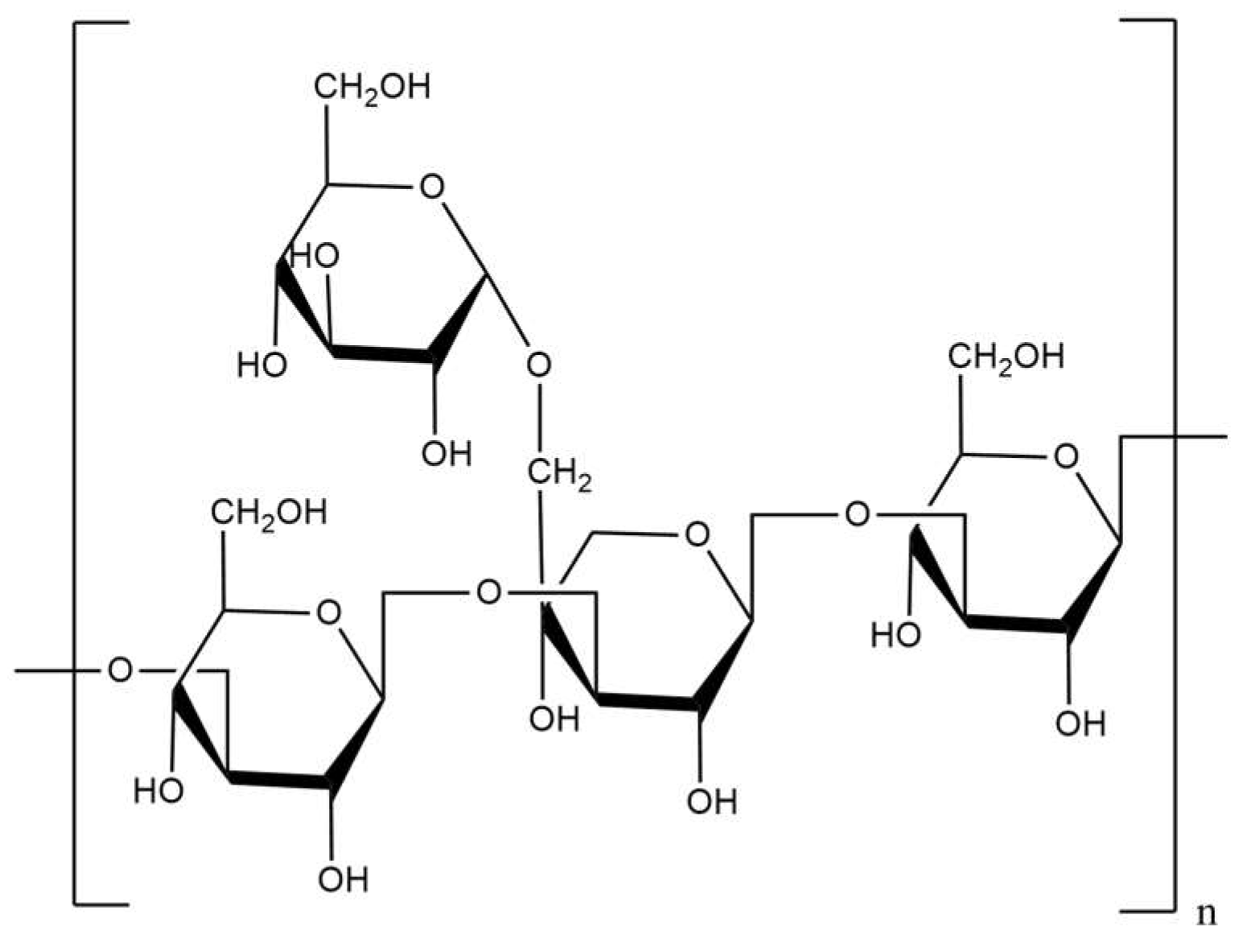
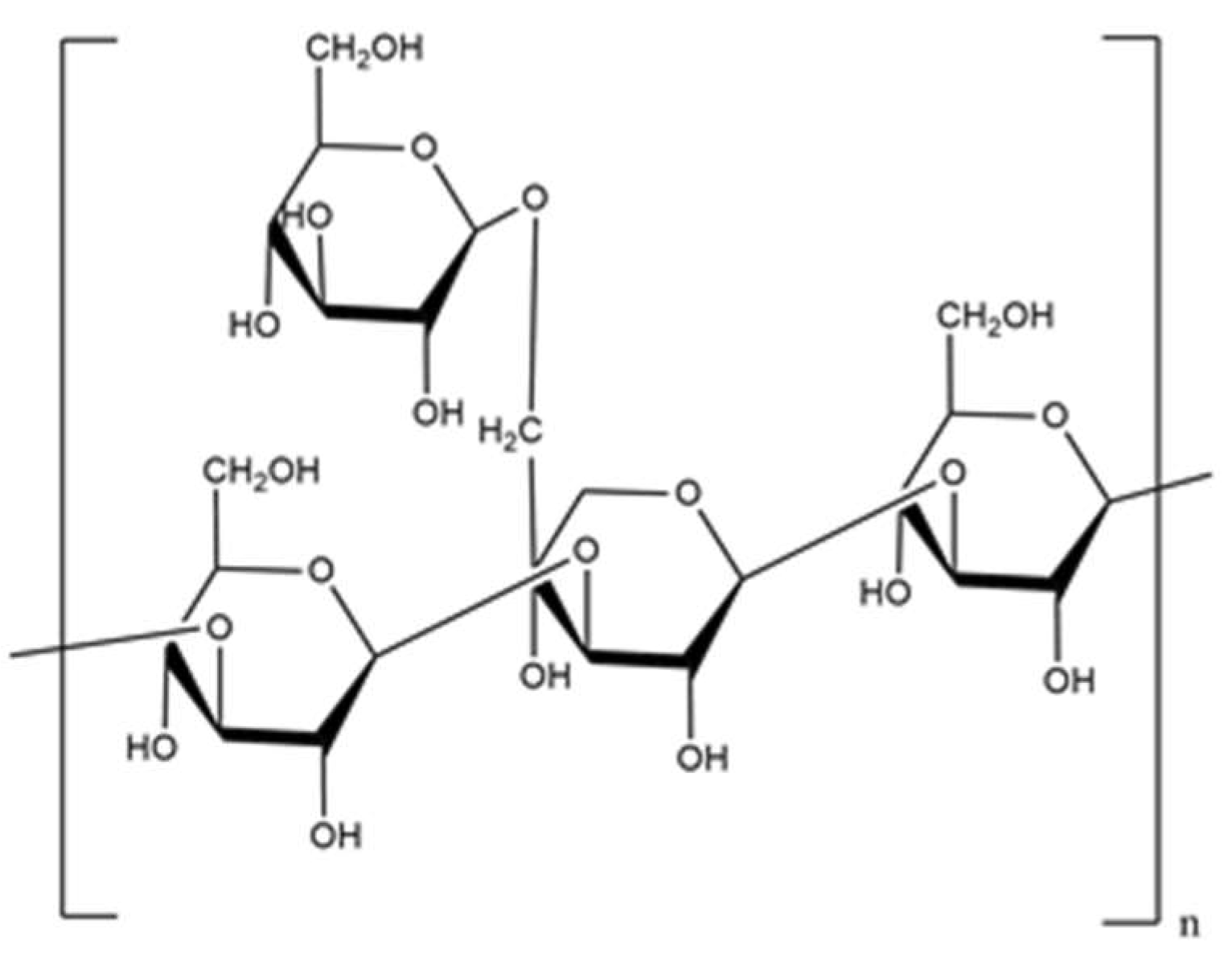
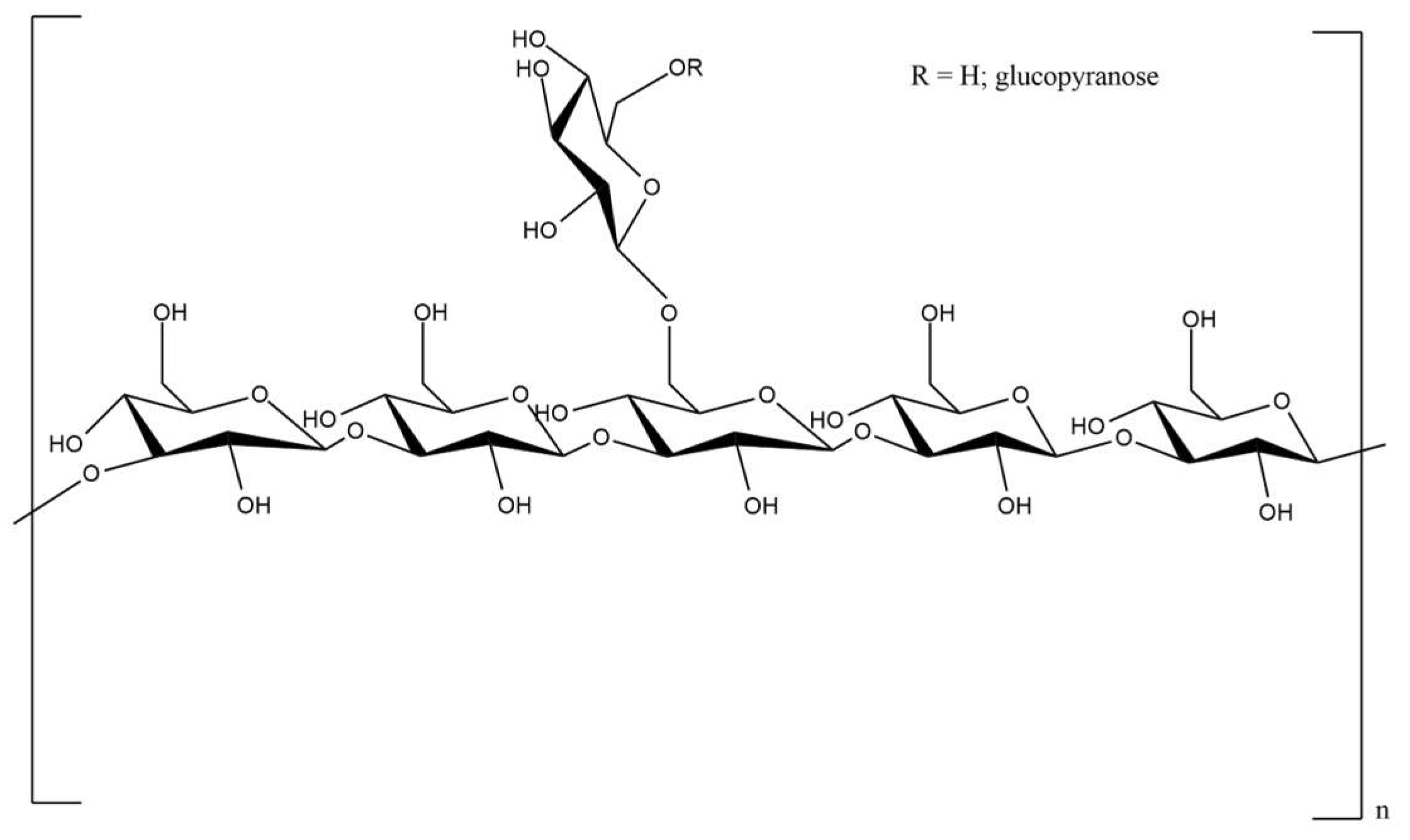

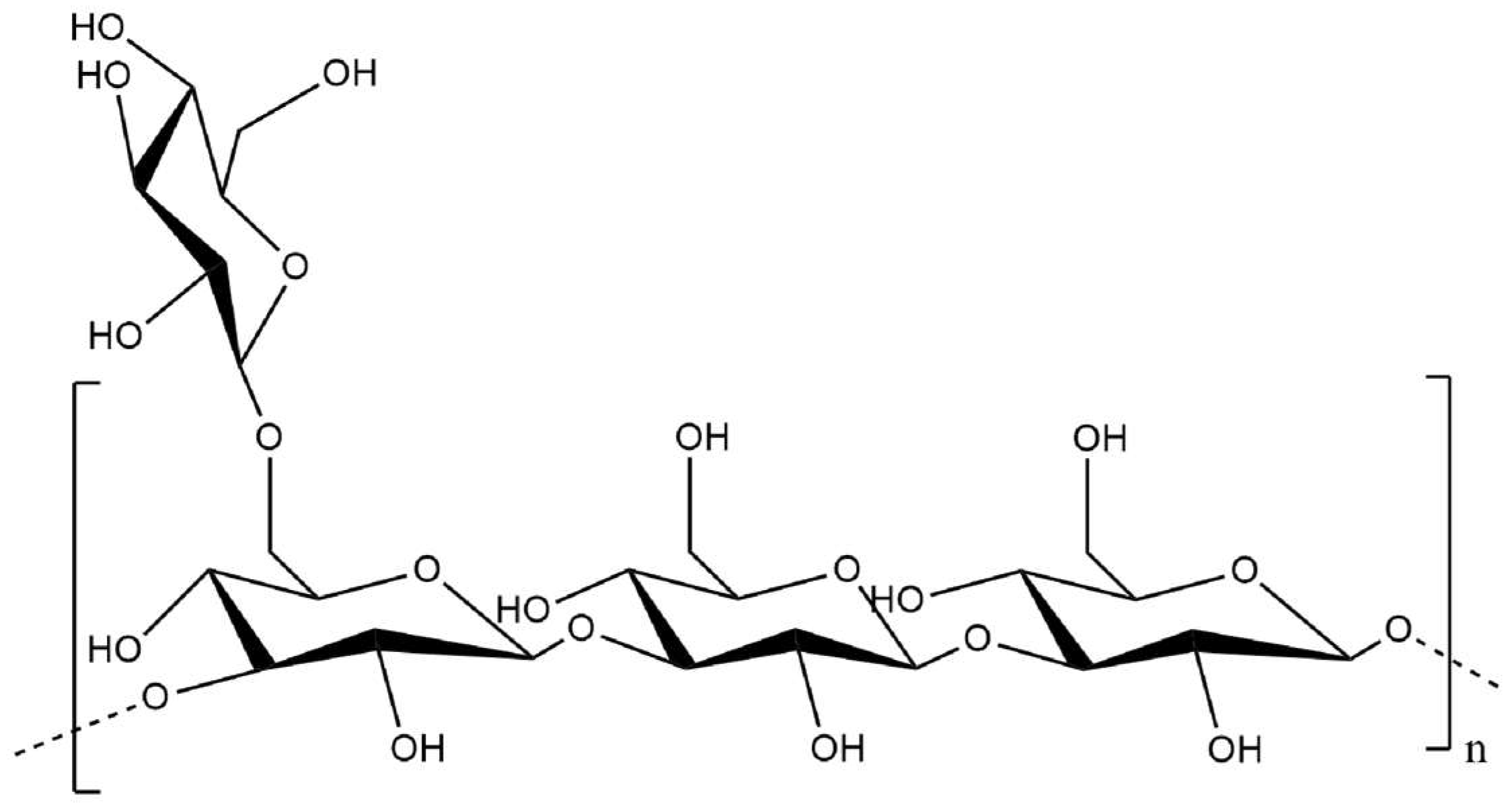

| Sources | Polymer | Monosaccharide Constituents | Types of Glycosidic Linkages | Pharmaceutical Applications | References |
|---|---|---|---|---|---|
| Aureobasidium pullulans | pullulan | d-Glucose | α (1,6), α (1,4) | targeted drug delivery, tissue engineering, wound healing, anticancer activity | [30,31,32,33,34,35] |
| Schizophyllum commune | schizophyllan | d-Glucose | β (1,3), β (1,6) | anti-head, neck and mammary cancers, antibacterial, antiparasitic, hypoglycemic properties, immunobiological activities | [51,52,53,54,55,56,57] |
| Sclerotium rolfsii | scleroglucan | d-Glucose | β (1,3), β (1,6) | antitumor, antiviral, antimicrobial activities, drug delivery systems, immunomodulatory effects | [65,66,67,68,69,70,71] |
| Botryosphaeria rhodina | botryosphaeran | d-Glucose | β (1,3), β (1,6) | antiproliferative and immunomodulatory activities, anti-diabetic and hypocholesterolemic properties | [78,79,80,81,84] [85,86,87] |
| Lentinus edodes | lentinan | d-Glucose | β (1,3), β (1,6) | immunomodulatory and anti-cancer properties, such as sarcoma, lung, colon, and gastric cancers, K36 murine lymphoma | [98,99,100,101,102,103] |
| Grifola frondosa | grifolan | d-Glucose | β (1,3), β (1,6) | breast, stomach, and colon cancer activity, immunomodulatory effect, anti-diabetic, antilipidemic, antiviral and anti-hypertensive properties | [109,110,111,112,113,114,115,116] |
| Lasiodiplodia theobromae | lasiodiplodan | d-Glucose | β (1,6) | antiproliferative activity in breast cancer MCF-7 cells, anticoagulant, hypoglycemic, antimicrobial, antioxidant, and wound healing effects | [122,124,125,126,127,129] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stoica, R.M.; Moscovici, M.; Lakatos, E.S.; Cioca, L.I. Exopolysaccharides of Fungal Origin: Properties and Pharmaceutical Applications. Processes 2023, 11, 335. https://doi.org/10.3390/pr11020335
Stoica RM, Moscovici M, Lakatos ES, Cioca LI. Exopolysaccharides of Fungal Origin: Properties and Pharmaceutical Applications. Processes. 2023; 11(2):335. https://doi.org/10.3390/pr11020335
Chicago/Turabian StyleStoica, Roxana Mădălina, Misu Moscovici, Elena Simina Lakatos, and Lucian Ionel Cioca. 2023. "Exopolysaccharides of Fungal Origin: Properties and Pharmaceutical Applications" Processes 11, no. 2: 335. https://doi.org/10.3390/pr11020335
APA StyleStoica, R. M., Moscovici, M., Lakatos, E. S., & Cioca, L. I. (2023). Exopolysaccharides of Fungal Origin: Properties and Pharmaceutical Applications. Processes, 11(2), 335. https://doi.org/10.3390/pr11020335









