The Influence of Binding of Selected Mycotoxin Deactivators and Aflatoxin M1 on the Content of Selected Micronutrients in Milk
Abstract
1. Introduction
2. Materials and Methods
2.1. Aim and Hypothesis
2.2. Laboratory Methodology
2.3. Statistical Analysis
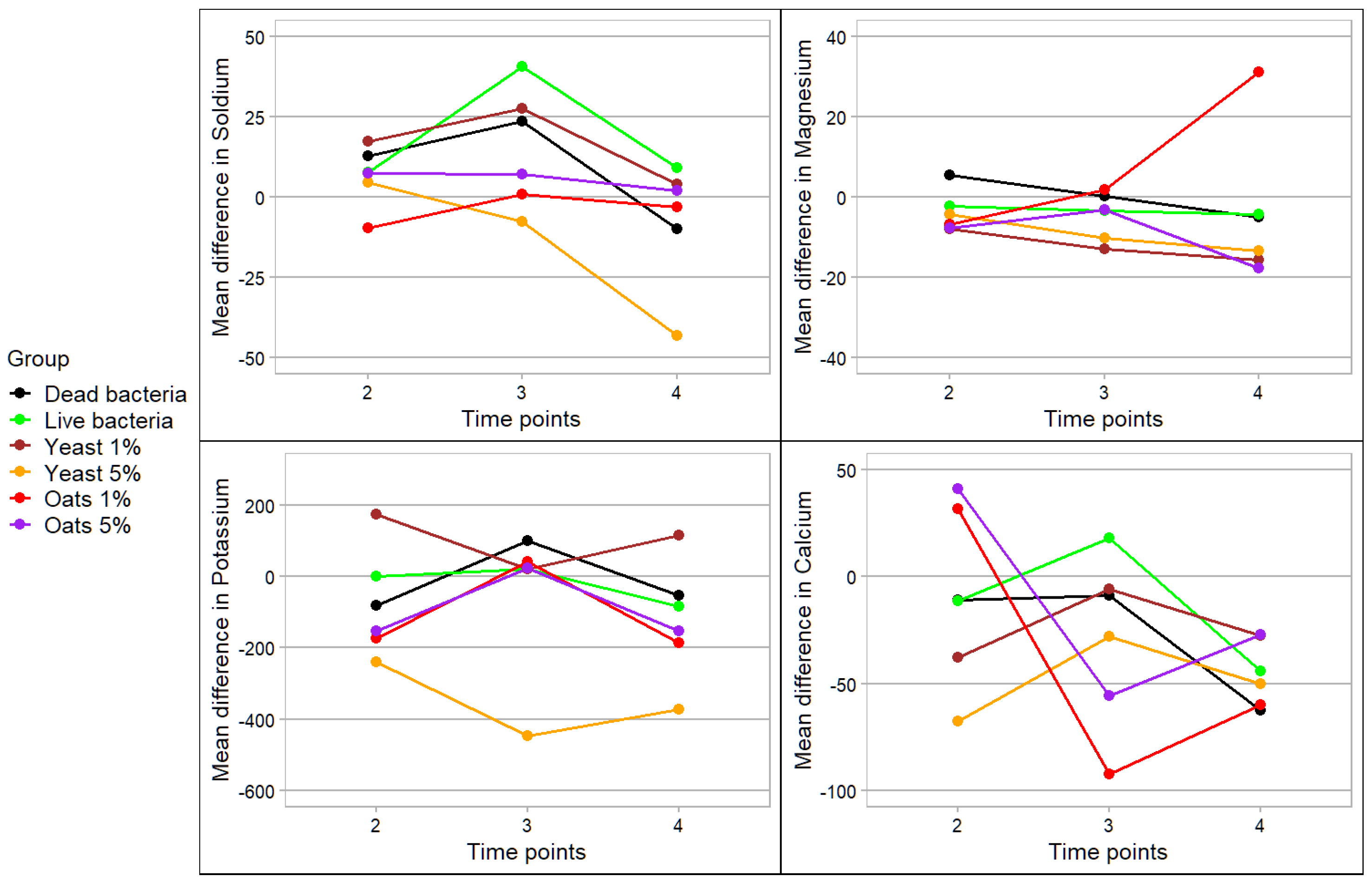
3. Results
4. Discussion
5. Conclusions
6. Limitations of the Study
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ATM1 | aflatoxin M1 |
| AFB1 | aflatoxin B1 |
| EFSA | European Food Safety Authority |
References
- Republic of Croatia. Regulation on Milk and Dairy Products. In Official Gazette; 2007; 133, p. 3798. Available online: https://narodne-novine.nn.hr/clanci/sluzbeni/2007_12_133_3798.html (accessed on 16 September 2022).
- Pike, R.L.; Brown, M.L. Nutrition: An Integrated Approach; John, Wiley and Sons: New York, NY, USA, 1984. [Google Scholar]
- Vahčić, N.; Hruškar, M.; Marković, K.; Banović, M.; Colić Barić, I. Essential minerals in milk and their daily intake through milk consumption. Mljekarstvo 2010, 60, 77–85. [Google Scholar]
- Zamberlin, Š.; Antunac, N.; Havranek, J.; Samaržija, D. Mineral elements in milk and dairy products. Mljekarstvo 2012, 62, 111–125. [Google Scholar]
- Bezie, A. The effect of different heat treatment on the nutritional value of milk and milk products and shelf-life of milk products. A review. J. Dairy Vet. Sci. 2019, 11, 555822. [Google Scholar] [CrossRef]
- De Jong, P. Thermal processing of milk. In Advanced Dairy Science and Technology; The Blackwell Publishing Ltd: Oxford, UK, 2008; pp. 1–34. [Google Scholar]
- Malbe, M.; Otstavel, T.; Kodis, I.; Viitak, A. Content of selected micro and macro elements in dairy cows’ milk in Estonia. Agron. Res. 2010, 8, 323–326. [Google Scholar]
- Cashman, K.D. Macroelements, Nutritional Significance. In Encyclopedia of Dairy Sciences, No 3; Roginski, H., Fuquay, J.W., Fox, P.F., Eds.; Academic Press: London, UK, 2002; pp. 2051–2058. [Google Scholar]
- Moreno-Rojas, R.; Zurera-Cosano, G.; Amaro-Lopez, M.A. Concentration and seasonal variation of calcium, magnesium, sodium and potassium in raw cow, ewe and goat milk. Int. J. Food Sci. Nutr. 1994, 45, 99–105. [Google Scholar] [CrossRef]
- Fuente de La Olano, A.; Juárez, M. Distribution of calcium, magnesium, phosphorus, zinc, manganese, copper and iron between the soluble and colloidal phases. Lait 1997, 77, 515–520. [Google Scholar] [CrossRef]
- Morris, R.C.; Sebastian, A.; Forman, A.; Tanaka, M.; Schmidlin, O. Normotensive salt-sensitivity: Effects of race and dietary potassium. Hypertension 1999, 33, 18–23. [Google Scholar] [CrossRef]
- Canabady-Rochellea, L.S.; Mellemab, M. Physical-chemical comparison of cow’s milk proteins versus soy proteins in their calcium-binding capacities. Colloids Surf. A Physicochem. Eng. Asp. 2010, 366, 110–112. [Google Scholar] [CrossRef]
- Dorić, I.; Lisak Jakopović, K.; Barukčić, I.; Božanić, R. Influence of milk on human health. Croat. J. Food Technol. Biotechnol. Nutr. 2019, 14, 24–32. [Google Scholar]
- Theobald, H.E. Dietary calcium and health. Br. Nutr. Found. Nutr. Bull. 2005, 30, 237–277. [Google Scholar] [CrossRef]
- Buzinaro, E.F.; Almeida, R.N.; Mazeto, G.M. Bioavailability of dietary calcium. Arq. Bras. Endocrinol. Metabol. 2006, 50, 852–861. [Google Scholar] [CrossRef] [PubMed]
- Soetan, K.O.; Olaiya, C.O.; Oyewole, O.E. The importance of mineral elements for humans, domestic animals and plants—A review. Afr. J. Food Sci. 2021, 4, 200–222. [Google Scholar]
- Muneer, K. Concentrations of Major and Minor Elements in Cow’s Milk at Najaf Province. Kufa J. Vet. Med. Sci. 2016, 7, 9–14. [Google Scholar]
- Korley Kortei, N.; Barnabas Teye Djaba, B.; Okraku Tettey, C.; Ofori Agyemang, A.; Aninagyei, E.; Essuman, E.K.; Agyemang Boakye, A.; Annan, T. Toxicogenic Fungi, Aflatoxins, and Antimicrobial Activities Associated with Some Spices and Herbs from Three Selected Markets in Ho Municipality, Ghana. Int. J. Food Sci. 2022, 2022, 7195890. [Google Scholar] [CrossRef] [PubMed]
- James, M.; Owino, W.; Imathiu, S. Microbial Contamination and Occurrence of Aflatoxins in Processed Baobab Products in Kenya. Int. J. Food Sci. 2022, 2022, 2577222. [Google Scholar] [CrossRef] [PubMed]
- Marchese, S.; Polo, A.; Ariano, A.; Velotto, S.; Costantini, S.; Severino, L. Aflatoxin B1 and M1, Biological Properties and Their Involvement in Cancer Development. Toxins 2018, 10, 214. [Google Scholar] [CrossRef]
- EFSA Panel on Contaminants in the Food Chain (CONTAM). Risk assessment of aflatoxins in food. EFSA J. 2020, 18, 6040. [Google Scholar]
- Sklan, D.; Klipper, E.; Friedman, A.; Shelly, M.; Makovsky, B. The Effect of Chronic Feeding of Diacetoxyscirpenol, T-2 Toxin, and Aflatoxin on Performance, Health, and Antibody Production in Chicks. J. Appl. Poult. Res. 2001, 10, 79–85. [Google Scholar] [CrossRef]
- Ayres, J.L.; Lee, D.J.; Wales, J.H.; Sinnhuber, R.O. Aflatoxin structure and hepatocarcinogenicity in rainbow trout (Salmo gairdneri). J. Natl. Cancer Inst. 1971, 46, 561–564. [Google Scholar]
- Bodine, A.B.; Fisher, S.F.; Gangjee, S. Effect of Aflatoxin B1 and Major Metabolites on Phytohemeagglutinin-Stimulated Lymphoblastogenesis of Bovine Lymphocytes. J. Dairy Sci. 1984, 67, 110–114. [Google Scholar] [CrossRef]
- Wogan, G.N.; Paglialunga, S.; Newberne, P.M. Carcinogenic effects of low dietary levels of aflatoxin B1 in rats. Food Cosmet. Toxicol. 1974, 12, 681–685. [Google Scholar] [CrossRef]
- Cullen, J.M.; Ruebner, B.H.; Hsieh, L.S.; Hyde, D.M.; Hsieh, D.P. Carcinogenicity of Dietary Aflatoxin M1 in Male Fischer Rats Compared to Aflatoxin B1. Cancer Res. 1987, 47, 1913–1917. [Google Scholar] [PubMed]
- World Health Organization; International Agency for Research on Cancer. In IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Aflatoxins; IARC Press: Lyon, France, 1993; Volume 56, pp. 245–395.
- Neal, G.E.; Eaton, D.L.; Judah, D.J.; Verma, A. Metabolism and toxicity of aflatoxins M1 and B1 in human-derived in vitro systems. Toxicol. Appl. Pharmacol. 1998, 151, 152–158. [Google Scholar] [CrossRef]
- Commission Regulation (EC). No 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs. Off. J. Eur. Union 2020, 027.001, 1–31. [Google Scholar]
- García-Pérez, O.D.; Tapia-Salazar, M.; Nieto-López, M.G.; Cavazos, D.V.; Cruz-Suárez, L.E.; Ricque-Marie, D. Effectiveness of aluminosilicate-based products for detoxification of aflatoxin-contaminated diets for juvenile Pacific white shrimp, Litopenaeus vannamei. Cienc. Mar. 2013, 39, 1–13. [Google Scholar] [CrossRef][Green Version]
- Doll, D.; Danicke, S.; Valenta, V.; Flachowsky, G. In vitro studies on the evaluation of mycotoxin detoxifying agents for their efficacy on deoxynivalenol and zearalenone. Arch. Anim. Nutr. 2004, 58, 311–324. [Google Scholar] [CrossRef]
- Chefchaou, H.; Mzabi, A.; Tanghort, M.; Moussa, H.; Chami, N.; Chami, F.; Remmal, A. A comparative study of different mycotoxin adsorbents against DON, T2 Toxin, Aflatoxins and Fumonisins production in maize flour. Livest. Res. Rural. Dev. 2019, 31, 35. [Google Scholar]
- Zouagui, Z.; Asrar, M.; Lakhdissi, H.; Abdennebi, E.H. Prevention of mycotoxin effects in dairy cows by adding an anti-mycotoxin product in feed. J. Mater. Environ. Sci. 2017, 8, 3766–3770. [Google Scholar]
- Yiannikouris, A.; André-Leroux, G.; Poughon, L.; François, J.; Dussap, C.G.; Jeminet, G.; Bertin, G.; Jouany, J.P. Chemical and conformational study of the interactions involved in mycotoxin complexation with beta-D-glucans. Biomacromol. Am. Chem. Soc. 2006, 7, 1147–1155. [Google Scholar] [CrossRef]
- Campagnollo, F.B.; Franco, L.T.; Rottinghaus, G.E.; Kobashigawa, E.; Ledoux, D.R.; Daković, A.; Oliveira, C.A.F. In vitro evaluation of the ability of beer fermentation residue containing Saccharomyces cerevisiae to bind mycotoxins. Food Res. Int. 2015, 77, 643–648. [Google Scholar] [CrossRef]
- Shetty, P.H.; Jespersen, L. Saccharomyces cerevisiae and lactic acid bacteria as potential mycotoxin decontaminating agents. Trends Food Sci. Technol. 2006, 17, 48–55. [Google Scholar] [CrossRef]
- Aazami, M.H.; Nasria, M.H.F.; Mojtahedia, M.; Battaconeb, G. Effect of the yeast cell wall and (1→3)-β-d-glucan on transfer of aflatoxin from feed to milk in Saanen dairy goats. Anim. Feed. Sci. Technol. 2019, 254, 114191. [Google Scholar] [CrossRef]
- Hanif, N.; Muhammad, G.; Siddique, M.; Khanum, A. Clinico-pathomorphological, serum biochemical and histological studies in broilers fed ochratoxin A and a toxin deactivator (Mycofix® Plus). Br. Poult. Sci. 2008, 49, 632–642. [Google Scholar] [CrossRef] [PubMed]
- Hamza, Z.; El-Hashashb, M.; Aly, S.; Hathouta, A.; Sotoc, E.; Sabrya, B.; Ostroffc, G. Preparation and characterization of yeast cell wall beta-glucan encapsulated humic acid nanoparticles as an enhanced aflatoxin B1 binder. Carbohydr. Polym. 2019, 203, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Kolosova, A.; Stroka, J. Substances for reduction of the contamination of feed by mycotoxins: A review. World Mycotoxin J. 2011, 4, 225–256. [Google Scholar] [CrossRef]
- Faucet-Marquis, V.; Joannis-Cassan, C.; Hadjeba-Medjdoub, K.; Ballet, N.; Pfohl-Leszkowicz, A. Development of an in vitro method for the prediction of mycotoxin binding on yeast-based products: Case of aflatoxin B1, zearalenone and ochratoxin A. Appl. Microbiol. Biotechnol. 2014, 98, 7583–7596. [Google Scholar] [CrossRef]
- Ariton, A.M.; Chelmu, S.S.; Neculai–Naleanu ASPostolache, A.N.; Ungureanu, E.; Creangă, S. Innovative approaches for the analysis and decontamination of mycotoxins from feed and milk. Rom. Biotechnol. Lett. 2020, 25, 1304–1310. [Google Scholar] [CrossRef]
- Taheura, F.B.; Fedhila, K.; Chaieb, K.; Kouidhi, B.; Bakhrouf, A.; Abrunhosa, L. Adsorption of aflatoxin B1, zearalenone and ochratoxin A by microorganisms isolated from Kefir grains. Int. J. Food Microbiol. 2017, 251, 1–7. [Google Scholar] [CrossRef]
- Frece, J.; Cvrtila, J.; Topić, I.; Delaš, F.; Markov, K. Lactococcus lactis ssp. lactis as Potential Functional Starter Culture. Food Technol. Biotechnol. 2014, 52, 489–494. [Google Scholar] [CrossRef]
- Noguchi, K.; Gel, Y.R.; Brunner, E.; Konietschke, F. nparLD: An R Software Package for the Nonparametric Analysis of Longitudinal Data in Factorial Experiments. Archives 2012, 50, 1–23. [Google Scholar] [CrossRef]
- Korthauer, K.; Kimes, P.K.; Duvallet, C.; Reyes, A.; Subramanian, A.; Teng, M.; Shukla, C.; Alm, E.J.; Hicks, S.C. A practical guide to methods controlling false discoveries in computational biology. Genome Biol. 2019, 20, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Mgbeahuruike, A.C.; Ejioffor, T.E.; Christian, O.C.; Shoyinka, V.S.; Magnus Karlsson, M.; Nordkvist, E. Detoxification of Aflatoxin-Contaminated Poultry Feeds by 3 Adsorbents, Bentonite, Activated Charcoal, and Fuller’s Earth. J. Appl. Poult. Res. 2018, 27, 461–471. [Google Scholar] [CrossRef]
- Asghar, M.A.; Iqbal, J.; Ahmed, A.; Inam, M.; Khan, M.A.; Jameel, K. In vitro Adsorption of Aflatoxins by Silicon Dioxide in Naturally Contaminated Maize (Zea mays L.) and Compared with Activated Charcoal. J. Res. J. Agric. Biol. Sci. 2015, 12, 9–16. [Google Scholar]
- Hathout, A.S.; Aly, S.E. Biological detoxification of mycotoxins: A review. Ann. Microbiol. 2014, 64, 905–919. [Google Scholar] [CrossRef]
- Yunus, A.W.; Ullah, A.; Lindahl, J.F.; Anwar, Z.; Ullah, A.; Saif, S.; Ali, M.; Zahur, A.B.; Irshad, H.; Javaid, S.; et al. Aflatoxin Contamination of Milk Produced in Peri-urban Farms of Pakistan: Prevalence and Contributory Factors. Front. Microbiol. 2020, 11, 159. [Google Scholar] [CrossRef] [PubMed]
- Perczak, A.; Goliński, P.; Bryła, M.; Waśkiewicz, A. The efficiency of lactic acid bacteria against pathogenic fungi and mycotoxins. Arh. Hig. Rada. Toksikol. 2018, 69, 32–45. [Google Scholar] [CrossRef] [PubMed]
- Mrvčić, J.; Stanzer, D.; Šolić, E.; Stehlik-Toma, V. Interaction of lactic acid bacteria with metal ions: Opportunities for improving food safety and quality. World J. Microbiol. Biotechnol. 2012, 28, 2771–2782. [Google Scholar] [CrossRef]
- Jadan-Piedra, C.; Alcantara, C.; Monedero, V.; Zuniga, M.; Velez, D.; Devesa, V. The use of lactic acid bacteria to reduce mercury bioaccessibility. Food Chem. 2017, 228, 158–166. [Google Scholar] [CrossRef]
- Lin, D.; Cao, H.; Zhong, Y.; Huang, Y.; Zou, J.; He, Q.; Ji, R.; Qin, T.; Chen, Y.; Wang, D.; et al. Screening and identification of Lactic acid bacteria from Ya’an pickle water to effectively remove Pb2+. AMB Express 2019, 9, 101–111. [Google Scholar] [CrossRef]
- Hassan, S.H.A.; Kim, S.J.; Jung, A.Y.; Joo, J.H.; Oh, S.E.; Yang, J.E. Biosorptive capacity of Cd(II) and Cu(II) by lyophilized cells of Pseudomonas stutzeri. J. Gen. Appl. Microbiol. 2009, 55, 27–34. [Google Scholar] [CrossRef]
- Tian, F.; Xiao, Y.; Li, X.; Zhai, Q.; Wang, G.; Zhang, Q.; Zhang, H.; Chen, W. Protective Effects of Lactobacillus plantarum CCFM8246 against Copper Toxicity in Mice. PLoS ONE 2015, 10, 1–14. [Google Scholar] [CrossRef]
- Halttunen, T.; Kankaanpaa, P.; Tahvonen, R.; Salminen, S.; Ouwehand, A.C. Cadmium removal by specific lactic acid bacteria. Biosci. Microflora 2003, 22, 93–97. [Google Scholar] [CrossRef]
- Halttunen, T.; Salminen, S.; Tahvonen, R. Rapid removal of lead and cadmium from water by specific lactic acid bacteria. Int. J. Food Microbiol. 2007, 114, 30–35. [Google Scholar] [CrossRef]
- Wang, J.; Chen, C. Biosorbents for heavy metals removal and their future. Biotechnol. Adv. 2009, 27, 195–226. [Google Scholar] [CrossRef] [PubMed]
- Hodnik, V.; Anderluh, G. Toxin Detection by Surface Plasmon Resonance. Sensors 2009, 9, 1339–1354. [Google Scholar] [CrossRef] [PubMed]
- Zoghi, A.; Khosravi-Darani, K.; Sohrabvandi, S. Surface Binding of Toxins and Heavy Metals by Probiotics. Med. Chem. 2014, 1484–1498. [Google Scholar] [CrossRef] [PubMed]
- Corassin, C.H.; Bovo, F.; Rosim, R.E.; Oliveira, C.A.F. Efficiency of Saccharomyces cerevisiae and lactic acid bacteria strains to bind aflatoxin M1 in UHT skim milk. Food Control. 2013, 31, 80–83. [Google Scholar] [CrossRef]
- Nowak, K.; Wiater, A.; Choma, A.; Wiącek, D.; Bieganowski, A.; Siwulski, M.; Waśko, A. Fungal (1 → 3)-α-D-glucans as a new kind of biosorbent for heavy metals. Int. J. Biol. Macromol. 2019, 137, 960–965. [Google Scholar] [CrossRef]
- Yiannikouris, A.; Francois, J.; Poughon, L.; Dussap, C.G.; Jeminet, G.; Bertin, G.; Jouany, J.P. Influence of pH on Complexing of Model β-D-Glucans with Zearalenone. J. Food Prot. 2004, 67, 2745. [Google Scholar] [CrossRef]
- Modi, B.; Timilsina, H.; Bhandari, S.; Achhami, A.; Pakka, S.; Shrestha, P.; Kandel, D.; Gc, D.B.; Khatri, S.; Chhetri, P.M.; et al. Current trends of food analysis, safety, and packaging. Int. J. Food Sci. 2021, 2021, 9924667. [Google Scholar] [CrossRef]
| ON ICP-MS | The Conditions of Recording |
|---|---|
| Atomizer | MicroMist |
| Injector | quartz |
| Cones | nickel |
| RF power | 1180 W |
| Plasma gas flow | 15.0 L/min |
| Atomizer gas flow | 1.07 L/min |
| Auxiliary gas flow | 0.90 L/min |
| Integration time | 1000 ms |
| Peak points | 100 |
| Number of replicas | 5 |
| Delay time | 30 s |
| Rinse time | 70 s |
| Time 1 | Time 2 | Time 3 | Time 4 | |
|---|---|---|---|---|
| Sodium | ||||
| Yeast 5 | 258 (22.8) | 263 (23.2) | 251 (19.1) | 215 (31.8) |
| Yeast 1 | 222 (20.3) | 239 (24.4) | 249 (26.5) | 226 (24.7) |
| Oats 5 | 228 (24.8) | 235 (23.2) | 222 (25.6) | 230 (24.8) |
| Oats 1 | 205 (38.6) | 195 (38.0) | 206 (34.2) | 202 (26.2) |
| Live bac | 288 (36.8) | 295 (40.6) | 328 (66.2) | 297 (47.5) |
| Dead bac | 240 (11.1) | 253 (15.7) | 264 (26.6) | 230 (17.4) |
| Magnesium | ||||
| Yeast 5 | 78.0 (3.35) | 73.6 (3.74) | 67.8 (4.32) | 64.6 (4.84) |
| Yeast 1 | 78.3 (7.13) | 70.3 (5.40) | 65.2 (4.08) | 62.6 (4.23) |
| Oats 5 | 84.6 (3.98) | 76.9 (5.97) | 81.3 (4.19) | 66.9 (3.51) |
| Oats 1 | 79.0 (4.84) | 72.2 (3.87) | 80.9 (4.10) | 110 (10.6) |
| Live bac | 75.6 (12.3) | 73.3 (12.4) | 72.2 (8.85) | 71.2 (6.59) |
| Dead bac | 71.9 (1.27) | 77.3 (1.88) | 72.1 (2.36) | 66.9 (3.60) |
| Potassium | ||||
| Yeast 5 | 1460 (171) | 1220 (163) | 1010 (91.4) | 1090 (146) |
| Yeast 1 | 931 (73.1) | 1100 (108) | 952 (108) | 1050 (112) |
| Oats 5 | 1240 (57.3) | 1080 (63.9) | 1260 (39.8) | 1080 (52.6) |
| Oats 1 | 1090 (82.8) | 913 (70.3) | 1130 (88.3) | 901 (67.9) |
| Live bac | 1190 (228) | 1190 (193) | 1210 (120) | 1100 (97.4) |
| Dead bac | 1180 (46.3) | 1100 (61.7) | 1280 (75.2) | 1120 (109) |
| Calcium | ||||
| Yeast 5 | 811 (37.8) | 744 (51.0) | 783 (56.8) | 761 (53.6) |
| Yeast 1 | 766 (42.9) | 728 (46.4) | 760 (59.3) | 739 (79.6) |
| Oats 5 | 846 (35.5) | 887 (34.4) | 790 (59.6) | 818 (51.1) |
| Oats 1 | 807 (73.9) | 839 (76.7) | 715 (79.8) | 747 (66.6) |
| Live bac | 288 (36.8) | 295 (40.6) | 328 (66.2) | 297 (47.5) |
| Dead bac | 240 (11.1) | 253 (15.7) | 264 (26.6) | 230 (17.4) |
| F | dfN/dfD | p-Value | GES | |
|---|---|---|---|---|
| Sodium | ||||
| Dead bac | 18.91 | 2/28 | <0.001 * | 0.325 |
| Live bac | 4.016 | 2/28 | 0.029 * | 0.083 |
| Yeast 1 | 15.738 | 2/28 | <0.001 * | 0.152 |
| Yeast 5 | 15.483 | 1.37/19.14 | <0.001 * | 0.393 |
| Oats 1 | 0.775 | 2/28 | 0.471 | 0.032 |
| Oats 5 | 10.614 | 1.06/14.88 | 0.005 * | 0.152 |
| Magnesium | ||||
| Dead bac | 66.624 | 1.42/19.85 | <0.001 * | 0.672 |
| Live bac | 0.223 | 2/28 | 0.802 | 0.005 |
| Yeast 1 | 46.704 | 1.12/15.67 | <0.001 * | 0.188 |
| Yeast 5 | 48.499 | 1.38/19.25 | <0.001 * | 0.51 |
| Oats 1 | 165.803 | 1.15/16.04 | <0.001 * | 0.85 |
| Oats 5 | 131.176 | 2/28 | <0.001 * | 0.755 |
| Potassium | ||||
| Dead bac | 36.545 | 1.4/19.67 | <0.001 * | 0.526 |
| Live bac | 2.228 | 1.32/18.44 | 0.148 | 0.035 |
| Yeast 1 | 16.576 | 1.29/18.07 | <0.001 * | 0.192 |
| Yeast 5 | 17.916 | 2/28 | <0.001 * | 0.204 |
| Oats 1 | 55.917 | 2/28 | <0.001 * | 0.688 |
| Oats 5 | 105.557 | 2/28 | <0.001 * | 0.783 |
| Calcium | ||||
| Dead bac | 2.087 | 1.02/14.23 | 0.17 | 0.072 |
| Live bac | 0.724 | 2/28 | 0.494 | 0.024 |
| Yeast 1 | 2.345 | 1.23/17.29 | 0.14 | 0.046 |
| Yeast 5 | 3.971 | 1.18/16.53 | 0.058 | 0.091 |
| Oats 1 | 41.92 | 2/28 | <0.001 * | 0.471 |
| Oats 5 | 62.647 | 1.16/16.28 | <0.001 * | 0.537 |
| Type of Mycotoxin Deactivators | β-Glucan from Oats | β-Glucan from Yeast | Dead Bacteria | Live Bacteria | ||
|---|---|---|---|---|---|---|
| The concentration of mycotoxin deactivators | 0.01% | 0.005% | 0.01% | 0.005% | 106 BMK | 1 mg biomass/mL |
| Time of binding (hours) | 0.169 | 0.301 | 0.473 | 0.178 | 0.145 | 0.119 |
| 0 | ||||||
| 2 | 0.223 | 0.342 | 0.362 | 0.315 | 0.092 | 0.101 |
| 4 | 0.287 | 0.297 | 0.494 | 0.437 | 0.012 | 0.100 |
| 24 | 0.300 | 0.226 | 0.255 | 0.269 | 0.022 | 0.087 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pavlek, Z.; Bosnir, J.; Kuharic, Z.; Racz, A.; Jurak, I.; Lasic, D.; Markov, K.; Jakopovic, Z.; Frece, J. The Influence of Binding of Selected Mycotoxin Deactivators and Aflatoxin M1 on the Content of Selected Micronutrients in Milk. Processes 2022, 10, 2431. https://doi.org/10.3390/pr10112431
Pavlek Z, Bosnir J, Kuharic Z, Racz A, Jurak I, Lasic D, Markov K, Jakopovic Z, Frece J. The Influence of Binding of Selected Mycotoxin Deactivators and Aflatoxin M1 on the Content of Selected Micronutrients in Milk. Processes. 2022; 10(11):2431. https://doi.org/10.3390/pr10112431
Chicago/Turabian StylePavlek, Zeljka, Jasna Bosnir, Zeljka Kuharic, Aleksandar Racz, Ivan Jurak, Dario Lasic, Ksenija Markov, Zeljko Jakopovic, and Jadranka Frece. 2022. "The Influence of Binding of Selected Mycotoxin Deactivators and Aflatoxin M1 on the Content of Selected Micronutrients in Milk" Processes 10, no. 11: 2431. https://doi.org/10.3390/pr10112431
APA StylePavlek, Z., Bosnir, J., Kuharic, Z., Racz, A., Jurak, I., Lasic, D., Markov, K., Jakopovic, Z., & Frece, J. (2022). The Influence of Binding of Selected Mycotoxin Deactivators and Aflatoxin M1 on the Content of Selected Micronutrients in Milk. Processes, 10(11), 2431. https://doi.org/10.3390/pr10112431







