Evaluation of the Kinetic and Thermodynamic Behavior of Tracers for Their Applicability in SWCTT
Abstract
1. Introduction
2. Experimental Section
2.1. Materials
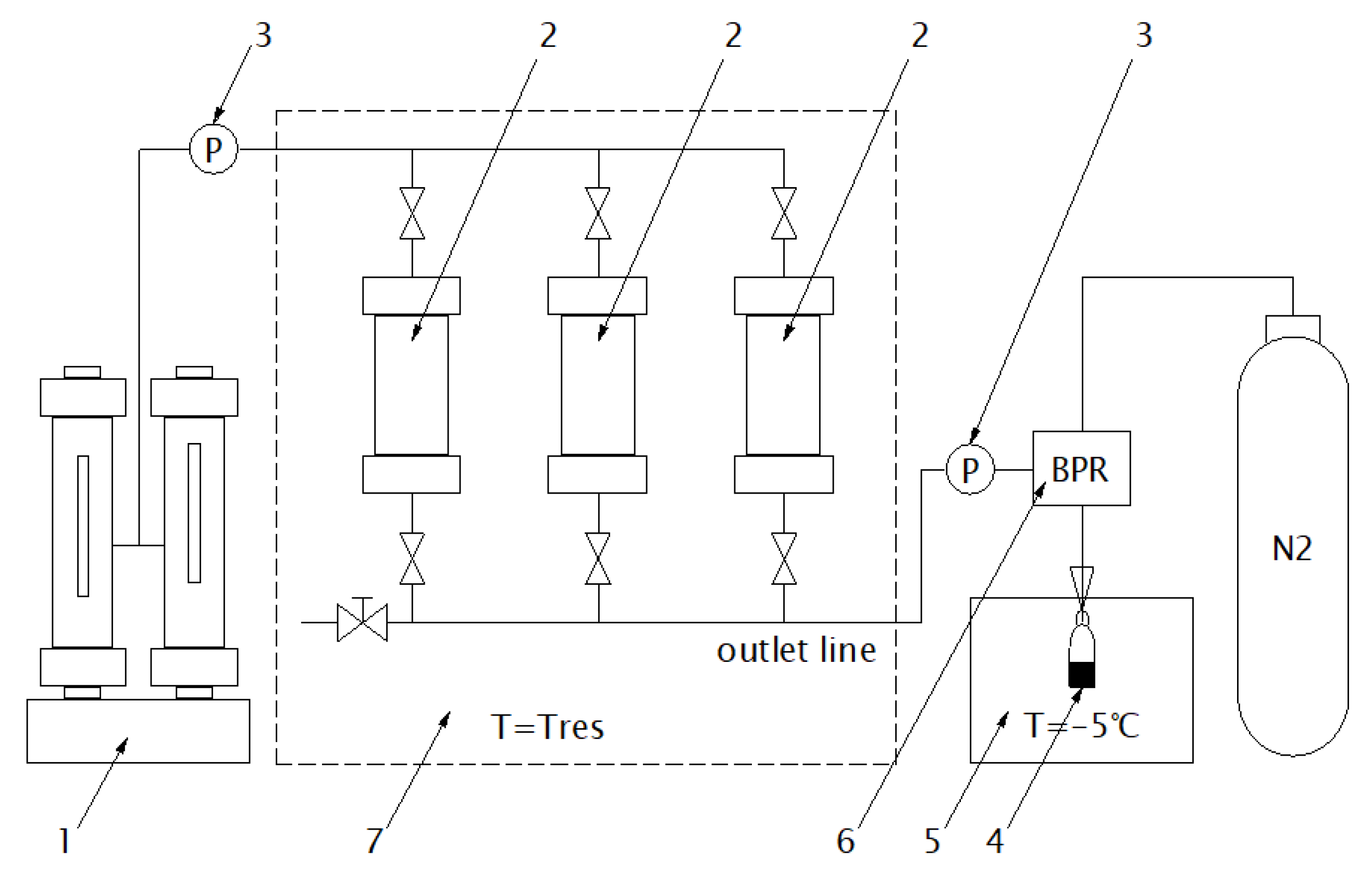
2.2. Experimental Apparatus
2.3. K-Value and Degree of Hydrolysis Calculation
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| kN | Neutral hydrolysis rate constant |
| k(1phase) | Single-phase hydrolysis rate constant |
| k(2phase) | Two-phase hydrolysis rate constant |
| K | Partitioning coefficient |
| Vo | Oil volume |
| Vw | Water volume |
| τ1/2 | Reaction half-life (1/2 hydrolysis) |
| ts | Shut-in time |
| logKow | Octanol/water partition coefficient |
| S | Salinity |
| EtOF | Ethyl formate |
| EtOAc | Ethyl acetate |
| EtOPr | Ethyl propionate |
| MeOAc | Methyl acetate |
| LSW | Low salinity water |
| HSW | High salinity water |
| LT | Low temperature |
| HT | High temperature |
References
- Babadagli, T. Development of mature oil fields—A review. J. Pet. Sci. Eng. 2007, 57, 221–246. [Google Scholar] [CrossRef]
- Chang, M.M.; Maerefat, N.L.; Tomutsa, L.; Honarpour, M.M. Evaluation and Comparison of Residual Oil Saturation Determination Techniques. SPE Form. Eval. 1988, 3, 251–262. [Google Scholar] [CrossRef]
- Teklu, T.W.; Brown, J.S.; Kazemi, H.; Graves, R.M.; Al Sumaiti, A.M. A Critical Literature Review of Laboratory and Field Scale Determination of Residual Oil Saturation. SPE-164483-MS. In Proceedings of the SPE Production and Operations Symposium, Oklahoma City, OK, USA, 23–26 March 2013. [Google Scholar] [CrossRef]
- Deans, H.A. The Single-Well Chemical Tracer Method for Measuring Residual Oil Saturation; Final Report, Contract No. US DOE/BC/20006-18; U.S. Department of Energy Office of Scientific and Technical Information: Washington, DC, USA, 1980. [Google Scholar]
- Doorwar, S.; Tagavifar, M.; Dwarakanath, V. A 1D Analytical Solution to Determine Residual Oil Saturations from Single-well Chemical Tracer Test Copyright. Chevron ETC. In Proceedings of the SPE Improved Oil Recovery Conference, Virtual, 31 August–4 September 2020. [Google Scholar] [CrossRef]
- Mechergui, A.; Agenet, N.; Romero, C.; Nguyen, M.; Batias, J. Design, Operation, and Laboratory Work for Single-Well Tracer Test Campaign in Handil Field Indonesia. In Proceedings of the SPE Enhanced Oil Recovery Conference, Kuala Lumpur, Malaysia, 2–4 July 2013. [Google Scholar] [CrossRef]
- Al Abbad, M.; Balasubramanian, S.; Sanni, M.; Kokal, S.; Zefzafy, I.; Adam, F.; Al Hajji, A. Single-Well Chemical Tracer Test for Residual Oil Measurement: Field Trial and Case Study. In Proceedings of the SPE Kingdom of Saudi Arabia Annual Technical Symposium and Exhibition, Dammam, Saudi Arabia, 25–28 April 2016. [Google Scholar] [CrossRef]
- Al Murayri, M.T.; Hassan, A.A.; Rahim, A.A.; Decroux, B.; Negre, A.; Salaun, M. Surfactant-Polymer Flooding: Single Well Chemical Tracer Test Design and Implementation in a Major Sandstone Kuwaiti Reservoir. In Proceedings of the SPE Kuwait Oil & Gas Show and Conference, Mishref, Kuwait, 13–16 October 2019. [Google Scholar] [CrossRef]
- Zoshchenko, O.; Aleshchenko, A.; Trushin, Y. Assessment of the Potential of Low-Salinity Water Injection Technology to Increase the Oil Recovery of the Carbonate Reservoir of the Kharyaga Field. In Proceedings of the SPE Russian Petroleum Technology Conference, Moscow, Russia, 22–24 October 2019. [Google Scholar] [CrossRef]
- Zecheru, M.; Goran, N. The use of chemical tracers to water injection processes applied on Romanian reservoirs. EPJ Web Conf. 2013, 50, 2005. [Google Scholar] [CrossRef][Green Version]
- Karimi, M. Single Well Tracer Test for Residual Oil Estimation; The Technical University of Crete, School of Mineral Resources Engineering, Petroleum Engineering, MSc Course: Chania, Greece, 2018. [Google Scholar]
- Hernández, C.; Chacón, L.; Anselmi, L.; Angulo, R.; Manrique, E.; Romero, E.; de Audemard, N.; Carlisle, C. Single Well Chemical Tracer Test to Determine ASP Injection Efficiency at Lagomar VLA-6/9/21 Area, C4 Member, Lake Maracaibo, Venezuela. In Proceedings of the SPE/DOE Improved Oil Recovery Symposium, Tulsa, OK, USA, 13–17 April 2002. [Google Scholar] [CrossRef]
- Oyemade, S.N.; Al Harthy, S.A.; Jaspers, H.F.; Van Wunnik, J.; de Kruijf, A.; Stoll, M. Alkaline-Surfactant-Polymer Flood (ASP): Single Well Chemical Tracer Tests-Design, Implementation and Performance. In Proceedings of the SPE EOR Conference at Oil & Gas West Asia, Muscat, Oman, 11–13 April 2010. [Google Scholar] [CrossRef]
- Callegaro, C.; Masserano, F.; Bartosek, M.; Buscaglia, R.; Visintin, R.; Hartvig, S.K.; Huseby, O. Single Well Chemical Tracer Tests to Assess Low Salinity Water and Surfactant EOR Processes in West Africa. In Proceedings of the International Petroleum Technology Conference, Kuala Lumpur, Malaysia, 10–12 December 2014. [Google Scholar] [CrossRef]
- Huseby, O.; Galdiga1, C.; Hartvig, S.; Zarruk, G.; Dugstad, O. A New Generation of Single Well Chemical Tracer Tests—Tracers and Methodologies. In Proceedings of the IOR—20th European Symposium on Improved Oil Recovery, Pau, France, 8–11 April 2019. [Google Scholar] [CrossRef]
- Bursaux, R.; Peltier, S.; Nguyen, M.; Romero, C.; Morel, D.D. Single Well Tracer Test Results in a High Temperature, High Salinity Offshore Carbonate Reservoir for Chemical EOR Pilot Evaluation. In Proceedings of the SPE Improved Oil Recovery Conference, Tulsa, OK, USA, 11–13 April 2016. [Google Scholar] [CrossRef]
- Deans, H.A.; Ghosh, R. pH and Reaction Rate Changes During Single-Well Chemical Tracer Tests. In Proceedings of the SPE/DOE Improved Oil Recovery Symposium, Tulsa, OK, USA, 17–20 April 1994; pp. 203–212. [Google Scholar] [CrossRef]
- Vogel, A.I. 130. Physical properties and chemical constitution. Part XIII. Aliphatic carboxylic esters. J. Chem. Soc. 1948, 2, 624–644. [Google Scholar] [CrossRef] [PubMed]
- Wilhoit, R.C.; Zwolinski, B.J. Physical and Thermodynamic Properties of Aliphatic Alcohols. J. Phys. Chem. Ref. Data 1973, 2, 420. [Google Scholar]
- Barton, A.F.M. Alcohols with Water Solubility Data Series; Pergamon Press: New York, NY, USA, 2013; p. 458. [Google Scholar]
- Getzen, F.; Maczynski, A.; Hefter, G.T. Esters with Water. Part I: Esters 2-C to 6-C; Pergamon Press: New York, NY, USA, 1992; Volume 48, p. 372. [Google Scholar] [CrossRef]
- Cumming, H.; Rücker, C. Octanol–Water Partition Coefficient Measurement by a Simple 1H NMR Method. ACS Omega 2017, 2, 6244–6249. [Google Scholar] [CrossRef] [PubMed]
- Souza, E.S.; Zaramello, L.; Kuhnen, C.A.; Junkes, B.D.S.; Yunes, R.A.; Heinzen, V.E.F. Estimating the Octanol/Water Partition Coefficient for Aliphatic Organic Compounds Using Semi-Empirical Electrotopological Index. Int. J. Mol. Sci. 2011, 12, 7250–7264. [Google Scholar] [CrossRef] [PubMed]
- Galeev, R.I.; Bolotov, A.V.; Varfolomeev, M.A.; Mukhutdinova, A.R.; Smirnov, A.E.; Kornilov, A.V.; Kruglov, D.S.; Zhirov, A.V.; Sansiev, G.V.; Fedorchenko, G.D. New and simple methods of determination partition coefficient and degree hydrolysis of tracer for estimating residual oil saturation by SWCTT technologies. Pet. Sci. Technol. 2021, 39, 1043–1059. [Google Scholar] [CrossRef]
- Mechergui, A.; Romero, C.; Ganzo, A. Design of SWTT at High Salinity and High Temperature: Where are the Limits? In Proceedings of the SPE EOR Conference at Oil and Gas West Asia, Muscat, Oman, 16–18 April 2012. [Google Scholar] [CrossRef]
- Pedersen, T. A Single Well Chemical Tracer model that accounts for temperature gradients, pH changes and buffering. J. Pet. Sci. Eng. 2021, 201, 108500. [Google Scholar] [CrossRef]
- Tang, J.; Zhang, P.-X. Determination of Residual Oil Saturation in A Carbonate Reservoir. SPE-72111-MS. In Proceedings of the SPE Asia Pacific Improved Oil Recovery Conference, Kuala Lumpur, Malaysia, 8–9 October 2001. [Google Scholar] [CrossRef]
- Tomich, J.F.; Dalton, R.L.; Deans, H.A.; Shallenberger, L.K. Single-Well Tracer Method to Measure Residual Oil Saturation. J. Pet. Technol. 1973, 25, 211–218. [Google Scholar] [CrossRef]
- Wang, S.; Shiau, B.; Harwell, J.H. Effect of Reservoirs Conditions on Designing Single-Well Chemical Tracer Tests Under Extreme Brine Conditions. Transp. Porous Media 2018, 121, 1–13. [Google Scholar] [CrossRef]
- Segreda, J.F.M. Spontaneous hydrolysis of ethyl formate: Isobaric activation parameters. Int. J. Chem. Kinet. 2000, 32, 67–71. [Google Scholar] [CrossRef]
- Hsieh, Y.-H.; Weinberg, N.; Wolfe, S. The neutral hydrolysis of methyl acetate—Part 1. Kinetic experiments. Can. J. Chem. 2009, 87, 539–543. [Google Scholar] [CrossRef]
- Comisar, C.M.; Hunter, S.E.; Walton, A.A.; Savage, P.E. Effect of pH on Ether, Ester, and Carbonate Hydrolysis in High-Temperature Water. Ind. Eng. Chem. Res. 2007, 47, 577–584. [Google Scholar] [CrossRef]
- Cooper, G.D.; Williams, B. Hydrolysis of Simple Aromatic Esters and Carbonates. J. Org. Chem. 1962, 27, 3717–3720. [Google Scholar] [CrossRef]
- Tang, J.S.; Harker, B. Mass Balance Method to Determine Residual Oil Saturation from Single Well Tracer Test Data. J. Can. Pet. Technol. 1990, 29, 115–124. [Google Scholar] [CrossRef]
- Bu, P.X.; Al Sofi, A.M.; Liu, J.; Benedek, L.; Han, M. Simulation of single well tracer tests for surfactant–polymer flooding. J. Pet. Explor. Prod. Technol. 2014, 5, 339–351. [Google Scholar] [CrossRef]

| No. | Compound Name | # CAS | Boiling Point, °C | Solubility in Water, g/100 g (20 °C) | log Kow |
|---|---|---|---|---|---|
| Primary (partitioning) tracers | |||||
| 1 | Methyl formate | 107,313 | 31.8 | 30.3 | −0.21 |
| 2 | Ethyl formate | 109,944 | 54.3 | 9.4 | 0.23 |
| 3 | Propyl formate | 110,747 | 81 | 2.8 | 0.83 |
| 4 | Methyl acetate | 79,209 | 57.1 | 31.9 | 0.18 |
| 5 | Ethyl acetate | 141,786 | 77.5 | 8.6 | 0.73 |
| 6 | Propyl acetate | 109,604 | 102 | 2.4 | 1.24 |
| 7 | Isopropyl acetate | 108,214 | 89 | 4,3 | 1.02 |
| 8 | Methyl propionate | 554,121 | 79.8 | 6.5 | 0.82 |
| 9 | Ethyl propionate | 105,373 | 99 | 2.4 | 1.43 |
| Secondary and cover tracers | |||||
| 10 | Methanol | 67,561 | 64.7 | Total | −0.77 |
| 11 | Ethanol | 64,175 | 78.5 | Total | −0.31 |
| 12 | n-Propanol | 71,238 | 97 | Total | 0.34 |
| 13 | i-Propanol | 67,630 | 82.5 | Total | 0.05 |
| 14 | n-Butanol | 71,363 | 117.7 | 7.7 | 0.84 |
| 15 | i-Butanol | 78,831 | 108 | 8.7 | 0.76 |
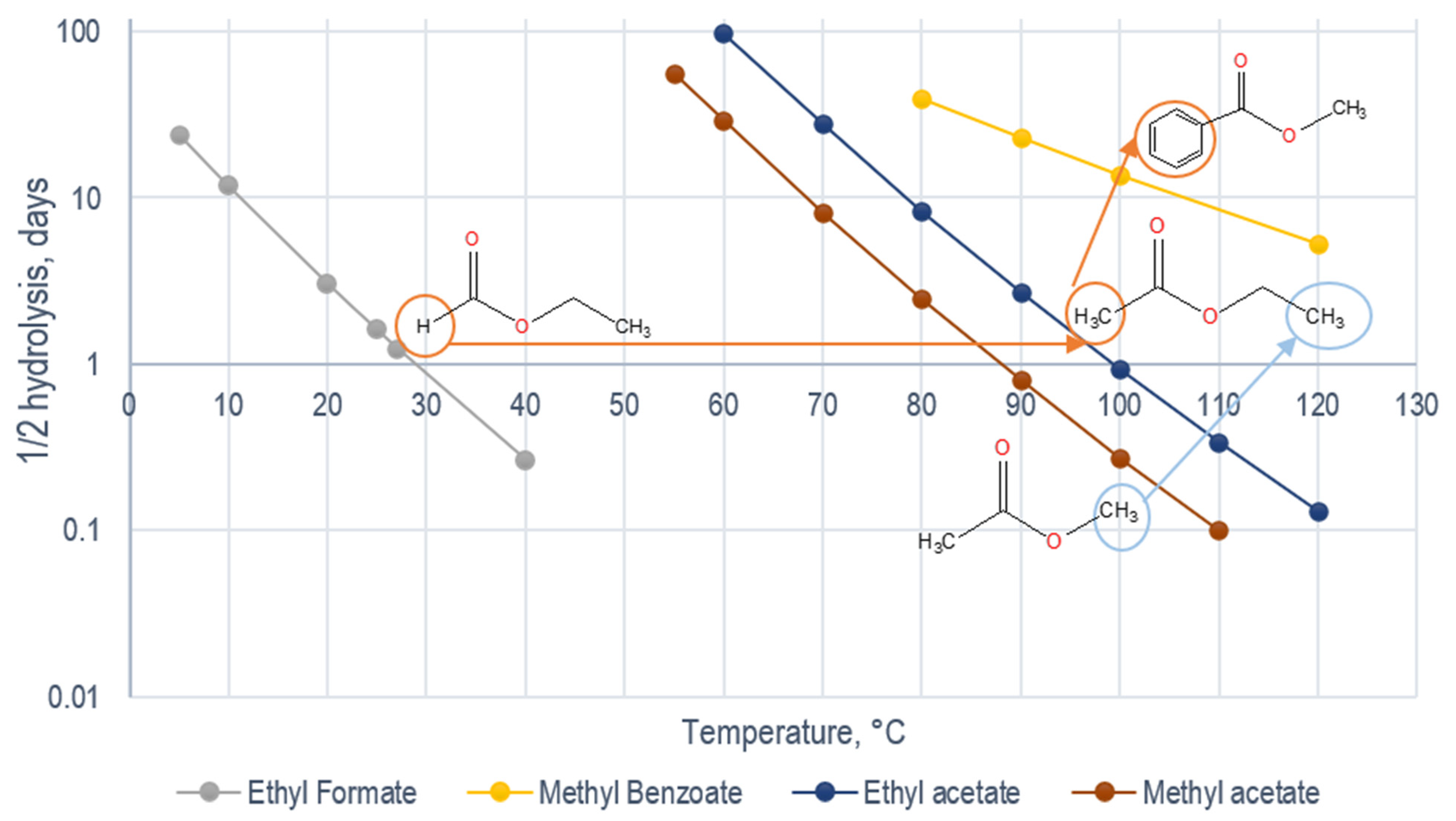
| Ester | T, °C | kN | τ1/2 | EA | Reference |
|---|---|---|---|---|---|
| Ethyl formate | 25 °C | 0.4 | 1.6 | 93 | [30] |
| Methyl acetate | 25 °C | 1.5 × 10−4 | 4600 | - | [31] |
| Ethyl acetate | 25 °C | 5.4 × 10−5 | 12,777 | 120 | [24] |
| Ethyl propionate | 96 °C | - | 4.9 | - | [6] |
| Methyl benzoate | 175 °C | 0.31 | 2.2 | 59 | [32] |
| Ethyl benzoate | 100 °C | 8.6 × 10−3 | 80 | - | [33] |
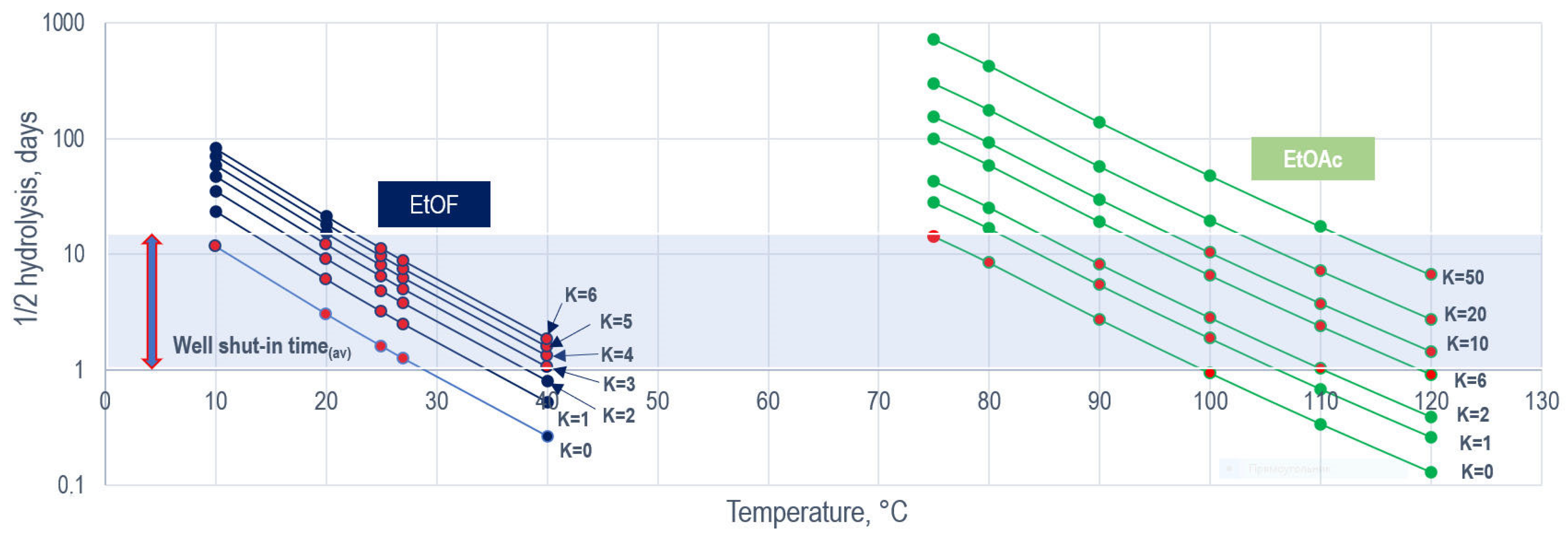
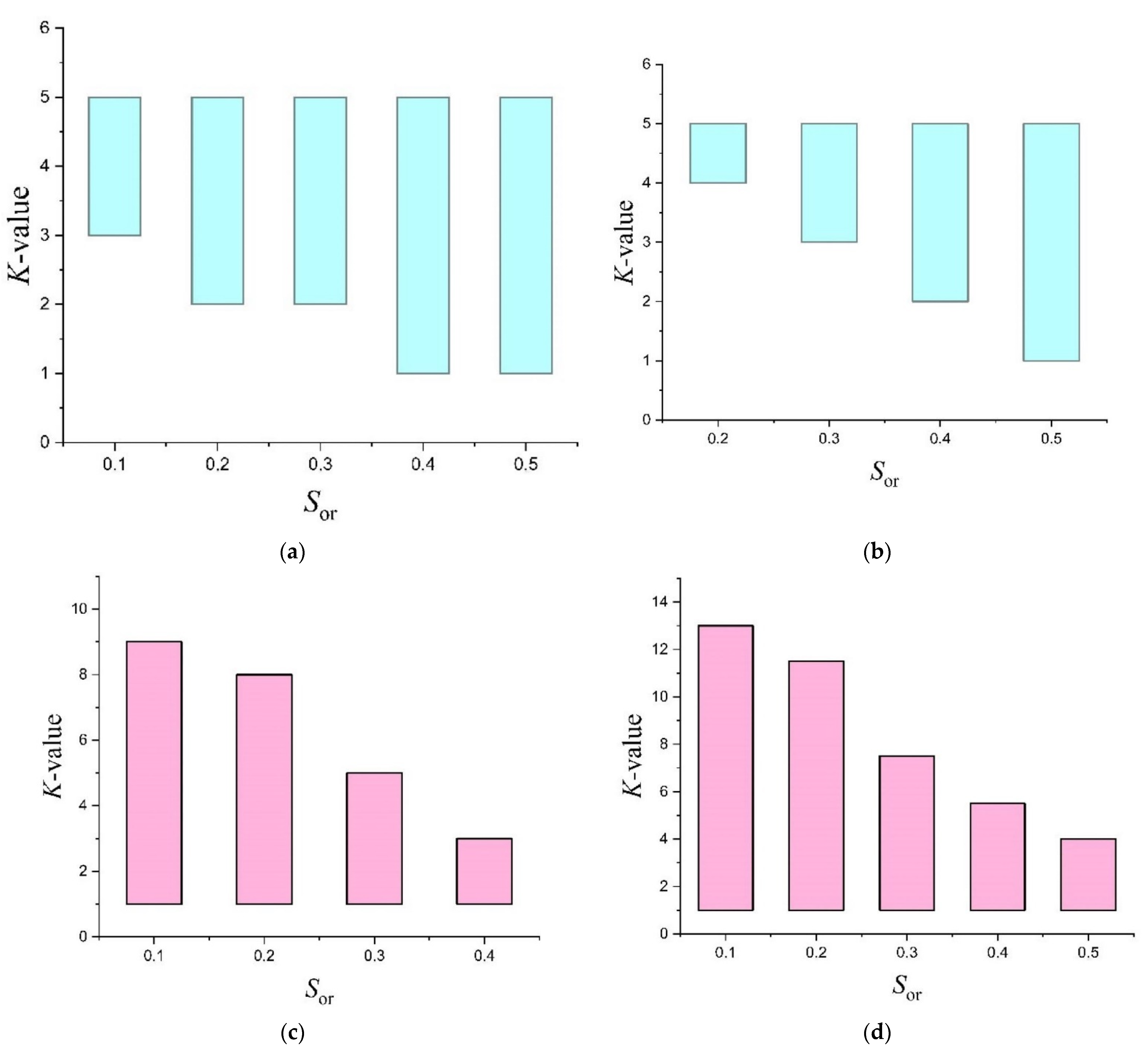
| Tracer | T | K-Value | |||
|---|---|---|---|---|---|
| S = 0 | S = 100 | S = 150 | S = 200 | ||
| Ethyl Formate | 25 | 1 | 1.9 | 2.6 | 3.4 |
| 40 | 1.3 | 2.2 | 3 | 3.6 | |
| 45 | 2.2 | 3 | 3.7 | 4.5 | |
| Ethyl Acetate | 70 | 4 | 6.5 | 9.1 | 10.4 |
| 90 | 4.2 | 8 | 11 | 13.8 | |
| 110 | 4.5 | 10.2 | 14.8 | 17 | |
| Ethyl Propionate | 60 | 16.5 | 27 | 34 | - |
| 90 | 20 | 29 | 37 | - | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anikin, O.V.; Bolotov, A.V.; Mukhutdinova, A.R.; Varfolomeev, M.A. Evaluation of the Kinetic and Thermodynamic Behavior of Tracers for Their Applicability in SWCTT. Processes 2022, 10, 2395. https://doi.org/10.3390/pr10112395
Anikin OV, Bolotov AV, Mukhutdinova AR, Varfolomeev MA. Evaluation of the Kinetic and Thermodynamic Behavior of Tracers for Their Applicability in SWCTT. Processes. 2022; 10(11):2395. https://doi.org/10.3390/pr10112395
Chicago/Turabian StyleAnikin, Oleg V., Alexander V. Bolotov, Alfiya R. Mukhutdinova, and Mikhail A. Varfolomeev. 2022. "Evaluation of the Kinetic and Thermodynamic Behavior of Tracers for Their Applicability in SWCTT" Processes 10, no. 11: 2395. https://doi.org/10.3390/pr10112395
APA StyleAnikin, O. V., Bolotov, A. V., Mukhutdinova, A. R., & Varfolomeev, M. A. (2022). Evaluation of the Kinetic and Thermodynamic Behavior of Tracers for Their Applicability in SWCTT. Processes, 10(11), 2395. https://doi.org/10.3390/pr10112395






