Microwave-Hydrogen Peroxide Assisted Anaerobic Treatment as an Effective Method for Short-Chain Fatty Acids Production from Tannery Sludge
Abstract
1. Introduction
2. Materials and Methods
2.1. Substrate Collection and Characteristics
2.2. Batch Tests’ Rationale
2.3. Batch Tests’ Preparation and Monitoring
2.4. Analytical Methods and Calculations
3. Results
3.1. Effect of the Initial pH on the Tannery Sludge Fermentation
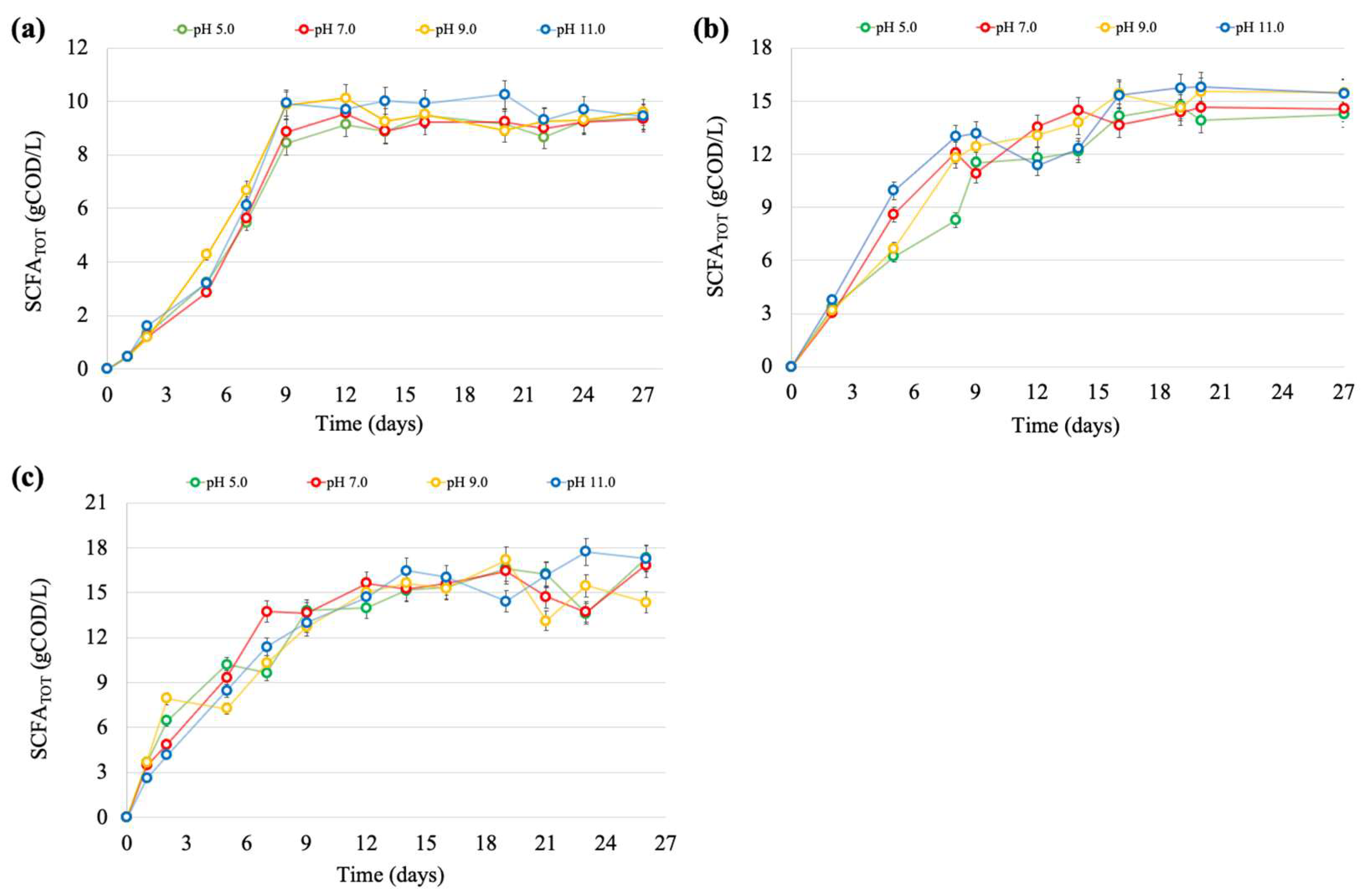
3.2. Production of SCFAs over Time
3.3. SCFAs/CODSOL Ratio
3.4. Fermentation Yield (YF)
3.5. Nutrients’ Release and Quantification
3.6. SCFAs Composition
3.7. Perspective of the Tannery Sludge Utilization in a New Biorefinery Value-Chain
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- 2030 Climate & Energy Framework. Available online: https://ec.europa.eu/clima/policies/strategies/2030_en (accessed on 26 May 2022).
- UNIC Italian Tanneries. Business Overview on Italian Tanning Industry; UNIC Italian Tanneries: San Miniato, Italy, 2020. [Google Scholar]
- Abreu, M.A.; Toffoli, S.M. Characterization of a Chromium-Rich Tannery Waste and Its Potential Use in Ceramics. Ceram. Int. 2009, 35, 2225–2234. [Google Scholar] [CrossRef]
- Alibardi, L.; Cossu, R. Pre-Treatment of Tannery Sludge for Sustainable Landfilling. Waste Manag. 2016, 52, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Basegio, T.; Berutti, F.; Bernardes, A.; Pérez, C.; Bergmann, P. Environmental and Technical Aspects of the Utilisation of Tannery Sludge as a Raw Material for Clay Products. J. Eur. Ceram. Soc. 2002, 22, 2251–2259. [Google Scholar] [CrossRef]
- Liu, H.; Wang, Y.; Zhang, H.; Huang, G.; Yang, Q.; Wang, Y. Synchronous Detoxification and Reduction Treatment of Tannery Sludge Using Cr (VI) Resistant Bacterial Strains. Sci. Total Environ. 2019, 687, 34–40. [Google Scholar] [CrossRef]
- Mpofu, A.B.; Welz, P.J.; Oyekola, O.O. Anaerobic Digestion of Secondary Tannery Sludge: Optimisation of Initial PH and Temperature and Evaluation of Kinetics. Waste Biomass Valorization 2020, 11, 873–885. [Google Scholar] [CrossRef]
- Mpofu, A.B.; Kibangou, V.A.; Kaira, W.M.; Oyekola, O.O.; Welz, P.J. Anaerobic Co-Digestion of Tannery and Slaughterhouse Wastewater for Solids Reduction and Resource Recovery: Effect of Sulfate Concentration and Inoculum to Substrate Ratio. Energies 2021, 14, 2491. [Google Scholar] [CrossRef]
- Silva, J.D.C.; Leal, T.T.B.; Araújo, A.S.F.; Araujo, R.M.; Gomes, R.L.F.; Melo, W.J.; Singh, R.P. Effect of Different Tannery Sludge Compost Amendment Rates on Growth, Biomass Accumulation and Yield Responses of Capsicum Plants. Waste Manag. 2010, 30, 1976–1980. [Google Scholar] [CrossRef]
- Zhai, S.; Li, M.; Xiong, Y.; Wang, D.; Fu, S. Dual Resource Utilization for Tannery Sludge: Effects of Sludge Biochars (BCs) on Volatile Fatty Acids (VFAs) Production from Sludge Anaerobic Digestion. Bioresour. Technol. 2020, 316. [Google Scholar] [CrossRef]
- Ronchi Decree. 1997. Available online: https://www.parlamento.it/parlam/leggi/deleghe/97022dl.htm (accessed on 24 March 2022).
- Pietrelli, L.; Ippolito, N.M.; Reverberi, A.P.; Vocciante, M. Heavy Metals Removal and Recovery from Hazardous Leather Sludge. Chem. Eng. Trans. 2019, 76, 1327–1332. [Google Scholar] [CrossRef]
- Skrzypczak, D.; Szopa, D.; Mikula, K.; Izydorczyk, G.; Baśladyńska, S.; Hoppe, V.; Pstrowska, K.; Wzorek, Z.; Kominko, H.; Kułażyński, M.; et al. Tannery Waste-Derived Biochar as a Carrier of Micronutrients Essential to Plants. Chemosphere 2022, 294, 133720. [Google Scholar] [CrossRef]
- Valentino, F.; Munarin, G.; Biasiolo, M.; Cavinato, C.; Bolzonella, D.; Pavan, P. Enhancing Volatile Fatty Acids (VFA) Production from Food Waste in a Two-Phases Pilot-Scale Anaerobic Digestion Process. J. Environ. Chem. Eng. 2021, 9, 106062. [Google Scholar] [CrossRef]
- Naresh Kumar, A.; Sarkar, O.; Chandrasekhar, K.; Raj, T.; Narisetty, V.; Mohan, S.V.; Pandey, A.; Varjani, S.; Kumar, S.; Sharma, P.; et al. Upgrading the Value of Anaerobic Fermentation via Renewable Chemicals Production: A Sustainable Integration for Circular Bioeconomy. Sci. Total Environ. 2022, 806, 150312. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.S.; Chua, A.S.M.; Yeoh, H.K.; Ngoh, G.C. A Review of the Production and Applications of Waste-Derived Volatile Fatty Acids. Chem. Eng. J. 2014, 235, 83–99. [Google Scholar] [CrossRef]
- Valentino, F.; Moretto, G.; Gottardo, M.; Pavan, P.; Bolzonella, D.; Majone, M. Novel Routes for Urban Bio-Waste Management: A Combined Acidic Fermentation and Anaerobic Digestion Process for Platform Chemicals and Biogas Production. J. Clean. Prod. 2019, 220, 368–375. [Google Scholar] [CrossRef]
- Soomro, A.F.; Abbasi, I.A.; Ni, Z.; Ying, L.; Liu, J. Influence of Temperature on Enhancement of Volatile Fatty Acids Fermentation from Organic Fraction of Municipal Solid Waste: Synergism between Food and Paper Components. Bioresour. Technol. 2020, 304, 122980. [Google Scholar] [CrossRef]
- Battista, F.; Almendros, M.G.; Rousset, R.; Boivineau, S.; Bouillon, P.A. Enzymatic Hydrolysis at High Dry Matter Content: The Influence of the Substrates’ Physical Properties and of Loading Strategies on Mixing and Energetic Consumption. Bioresour. Technol. 2018, 250, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Horn, E.J.; Oyekola, O.O.; Welz, P.J.; van Hille, R.P. Biological Desulfurization of Tannery Effluent Using Hybrid Linear Flow Channel Reactors. Water 2022, 14, 32. [Google Scholar] [CrossRef]
- Woodard and Curran. Chapter 7 Methods for Treating Wastewater from Industry. In Industrial Waste Treatment Handbook; Woodard and Curran: Portland, ME, USA, 2006; ISBN 978-0-7506-7963-3. [Google Scholar] [CrossRef]
- Tyagi, V.K.; Lo, S.L. Application of Physico-Chemical Pretreatment Methods to Enhance the Sludge Disintegration and Subsequent Anaerobic Digestion: An up to Date Review. Rev. Environ. Sci. Biotechnol. 2011, 10, 215–242. [Google Scholar] [CrossRef]
- Ambrose, H.W.; Chin, C.T.L.; Hong, E.; Philip, L.; Suraishkumar, G.K.; Sen, T.K.; Khiadani, M. Effect of Hybrid (Microwave-H2O2) Feed Sludge Pretreatment on Single and Two-Stage Anaerobic Digestion Efficiency of Real Mixed Sewage Sludge. Process Saf. Environ. Prot. 2020, 136, 194–202. [Google Scholar] [CrossRef]
- Liu, J.; Jia, R.; Wang, Y.; Wei, Y.; Zhang, J.; Wang, R.; Cai, X. Does Residual H2O2 Result in Inhibitory Effect on Enhanced Anaerobic Digestion of Sludge Pretreated by Microwave-H2O2 Pretreatment Process? Environ. Sci. Pollut. Res. 2017, 24, 9016–9025. [Google Scholar] [CrossRef]
- Özön, E.; Erdinçler, A. Effects of Microwave, H2O2/MW and H2O2/Heat Pre-Treatments on the Methane Production from Wastewater Sludges: Experimental and Modeling Approach. Environ. Sci. Pollut. Res. 2019, 26, 35411–35421. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Yang, M.; Zhang, J.; Zheng, J.; Xu, H.; Wang, Y.; Wei, Y. A Comprehensive Insight into the Effects of Microwave-H2O2 Pretreatment on Concentrated Sewage Sludge Anaerobic Digestion Based on Semi-Continuous Operation. Bioresour. Technol. 2018, 256, 118–127. [Google Scholar] [CrossRef] [PubMed]
- Apha Awwa, W.E.F. Standard Methods for the Examination of Water and Wastewater; American Public Health Association, American Water Works Association, and Water Environment Federation: Washington, DC, USA, 2005. [Google Scholar]
- Franke-Whittle, I.H.; Walter, A.; Ebner, C.; Insam, H. Investigation into the Effect of High Concentrations of Volatile Fatty Acids in Anaerobic Digestion on Methanogenic Communities. Waste Manag. 2014, 34, 2080–2089. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Tsui, T.H.; Loh, K.C.; Dai, Y.; Tong, Y.W. Acidogenic Fermentation of Organic Wastes for Production of Volatile Fatty Acids. In Biomass, Biofuels, Biochemicals: Microbial Fermentation of Biowastes; Elsevier: Amsterdam, The Netherlands, 2022; pp. 343–366. [Google Scholar] [CrossRef]
- Vidal-Antich, C.; Perez-Esteban, N.; Astals, S.; Peces, M.; Mata-Alvarez, J.; Dosta, J. Assessing the Potential of Waste Activated Sludge and Food Waste Co-Fermentation for Carboxylic Acids Production. Sci. Total Environ. 2021, 757, 143763. [Google Scholar] [CrossRef] [PubMed]
- Niero, L.; Morgan-Sagastume, F.; Lagerkvist, A. Accelerating Acidogenic Fermentation of Sewage Sludge with Ash Addition. J. Environ. Chem. Eng. 2021, 9, 106564. [Google Scholar] [CrossRef]
- Zhang, D.; Jiang, H.; Chang, J.; Sun, J.; Tu, W.; Wang, H. Effect of Thermal Hydrolysis Pretreatment on Volatile Fatty Acids Production in Sludge Acidification and Subsequent Polyhydroxyalkanoates Production. Bioresour. Technol. 2019, 279, 92–100. [Google Scholar] [CrossRef]
- Presti, D.; Cosenza, A.; Capri, F.C.; Gallo, G.; Alduina, R.; Mannina, G. Influence of Volatile Solids and PH for the Production of Volatile Fatty Acids: Batch Fermentation Tests Using Sewage Sludge. Bioresour. Technol. 2021, 342, 125853. [Google Scholar] [CrossRef]
- Morgan-Sagastume, F.; Hjort, M.; Cirne, D.; Gérardin, F.; Lacroix, S.; Gaval, G.; Karabegovic, L.; Alexandersson, T.; Johansson, P.; Karlsson, A.; et al. Integrated Production of Polyhydroxyalkanoates (PHAs) with Municipal Wastewater and Sludge Treatment at Pilot Scale. Bioresour. Technol. 2015, 181, 78–89. [Google Scholar] [CrossRef]
- Gottardo, M.; Bolzonella, D.; Adele Tuci, G.; Valentino, F.; Majone, M.; Pavan, P.; Battista, F. Producing Volatile Fatty Acids and Polyhydroxyalkanoates from Foods By-Products and Waste: A Review. Bioresour. Technol. 2022, 361, 127716. [Google Scholar] [CrossRef]
- Liu, H.; Han, P.; Liu, H.; Zhou, G.; Fu, B.; Zheng, Z. Full-Scale Production of VFAs from Sewage Sludge by Anaerobic Alkaline Fermentation to Improve Biological Nutrients Removal in Domestic Wastewater. Bioresour. Technol. 2018, 260, 105–114. [Google Scholar] [CrossRef]
- Morgan-Sagastume, F.; Valentino, F.; Hjort, M.; Cirne, D.; Karabegovic, L.; Gerardin, F.; Johansson, P.; Karlsson, A.; Magnusson, P.; Alexandersson, T.; et al. Polyhydroxyalkanoate (PHA) Production from Sludge and Municipal Wastewater Treatment. Water Sci. Technol. 2014, 69, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Jin, M.; Lian, F.; Xia, R.; Wang, Z. Formulation and Durability of a Geopolymer Based on Metakaolin/Tannery Sludge. Waste Manag. 2018, 79, 717–728. [Google Scholar] [CrossRef] [PubMed]
- Righetti, E.; Nortilli, S.; Fatone, F.; Frison, N.; Bolzonella, D. A Multiproduct Biorefinery Approach for the Production of Hydrogen, Methane and Volatile Fatty Acids from Agricultural Waste. Waste Biomass Valorization 2020, 11, 5239–5246. [Google Scholar] [CrossRef]
- Moretto, G.; Lorini, L.; Pavan, P.; Crognale, S.; Tonanzi, B.; Rossetti, S.; Majone, M.; Valentino, F. Biopolymers from Urban Organic Waste: Influence of the Solid Retention Time to Cycle Length Ratio in the Enrichment of a Mixed Microbial Culture (MMC). ACS Sustain. Chem. Eng. 2020, 8, 14531–14539. [Google Scholar] [CrossRef]
- Uçkun Kiran, E.; Trzcinski, A.P.; Liu, Y. Platform Chemical Production from Food Wastes Using a Biorefinery Concept. J. Chem. Technol. Biotechnol. 2015, 90, 1364–1379. [Google Scholar] [CrossRef]





| Operating Conditions | Acidogenic Fermentation Batch Tests (Series) | |||||
|---|---|---|---|---|---|---|
| M8 | M12 | M8-P | T8 | T12 | T8-P | |
| Temperature (°C) | 40 | 40 | 40 | 55 | 55 | 55 |
| Solids’ content (gTSs/L) | 80 | 120 | 80 | 80 | 120 | 80 |
| Range of the initial pH | 5–11 | 5–11 | 5–11 | 5–11 | 5–11 | 5–11 |
| MW-H2O2 pretreatment 1 | no | no | yes | no | no | yes |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tuci, G.A.; Valentino, F.; Bonato, E.; Pavan, P.; Gottardo, M. Microwave-Hydrogen Peroxide Assisted Anaerobic Treatment as an Effective Method for Short-Chain Fatty Acids Production from Tannery Sludge. Processes 2022, 10, 2167. https://doi.org/10.3390/pr10112167
Tuci GA, Valentino F, Bonato E, Pavan P, Gottardo M. Microwave-Hydrogen Peroxide Assisted Anaerobic Treatment as an Effective Method for Short-Chain Fatty Acids Production from Tannery Sludge. Processes. 2022; 10(11):2167. https://doi.org/10.3390/pr10112167
Chicago/Turabian StyleTuci, Giulia Adele, Francesco Valentino, Edoardo Bonato, Paolo Pavan, and Marco Gottardo. 2022. "Microwave-Hydrogen Peroxide Assisted Anaerobic Treatment as an Effective Method for Short-Chain Fatty Acids Production from Tannery Sludge" Processes 10, no. 11: 2167. https://doi.org/10.3390/pr10112167
APA StyleTuci, G. A., Valentino, F., Bonato, E., Pavan, P., & Gottardo, M. (2022). Microwave-Hydrogen Peroxide Assisted Anaerobic Treatment as an Effective Method for Short-Chain Fatty Acids Production from Tannery Sludge. Processes, 10(11), 2167. https://doi.org/10.3390/pr10112167








