Thermal Behavior of Estonian Graptolite–Argillite from Different Deposits
Abstract
1. Introduction
2. Materials and Methods
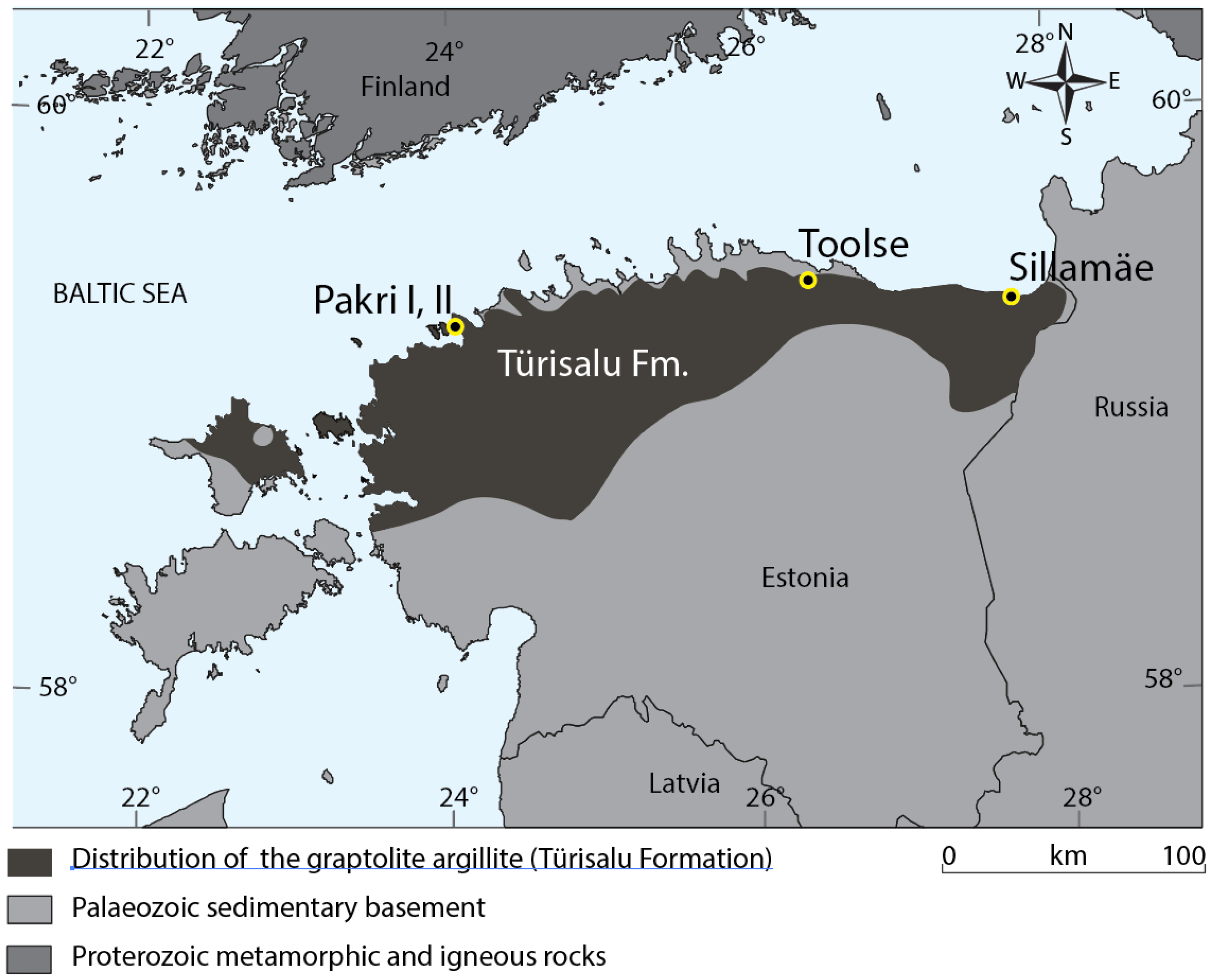
2.1. Materials
2.2. Methods
2.2.1. Material Characterization
2.2.2. Thermal and Kinetic Analysis
3. Results and Discussion
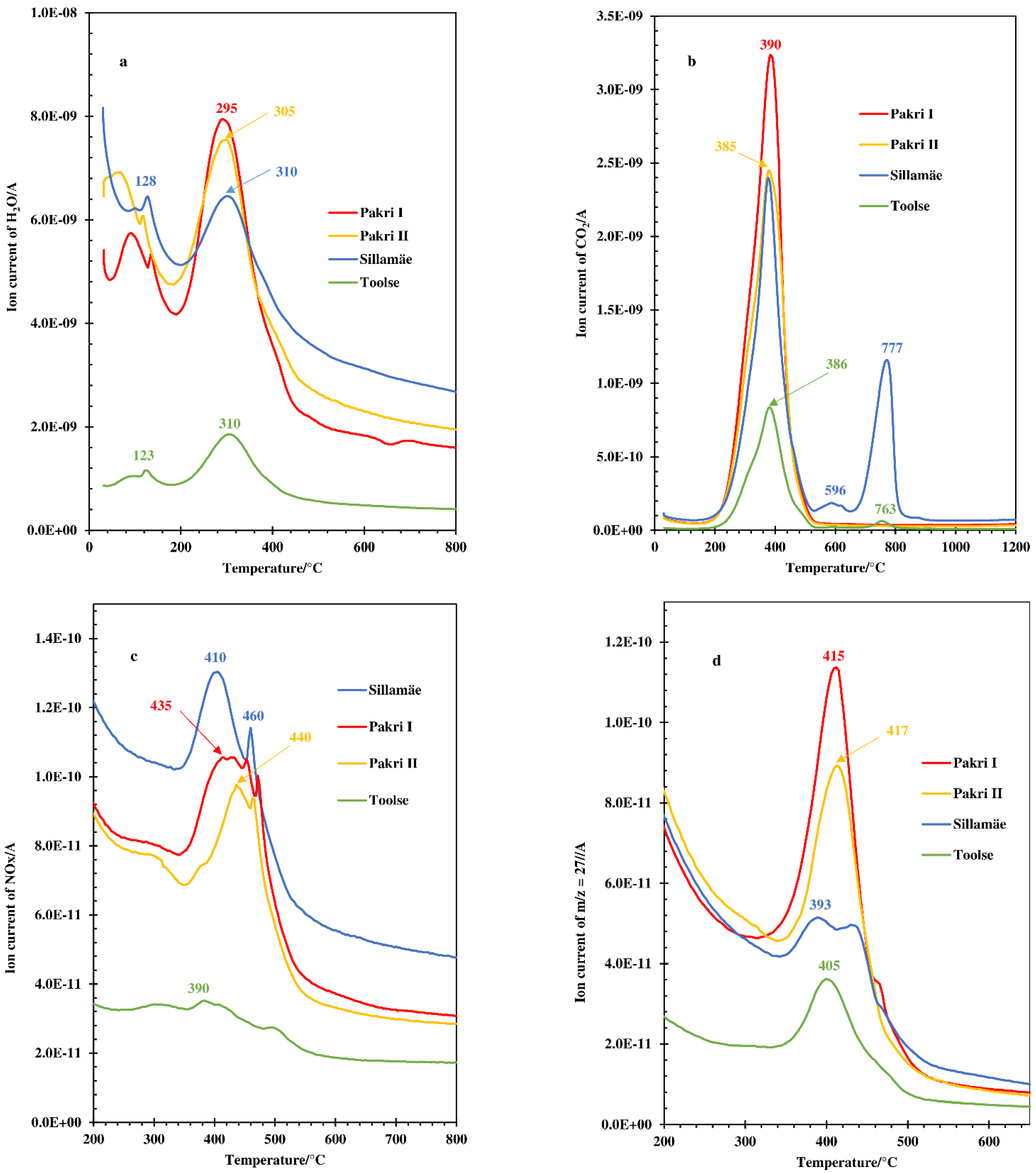
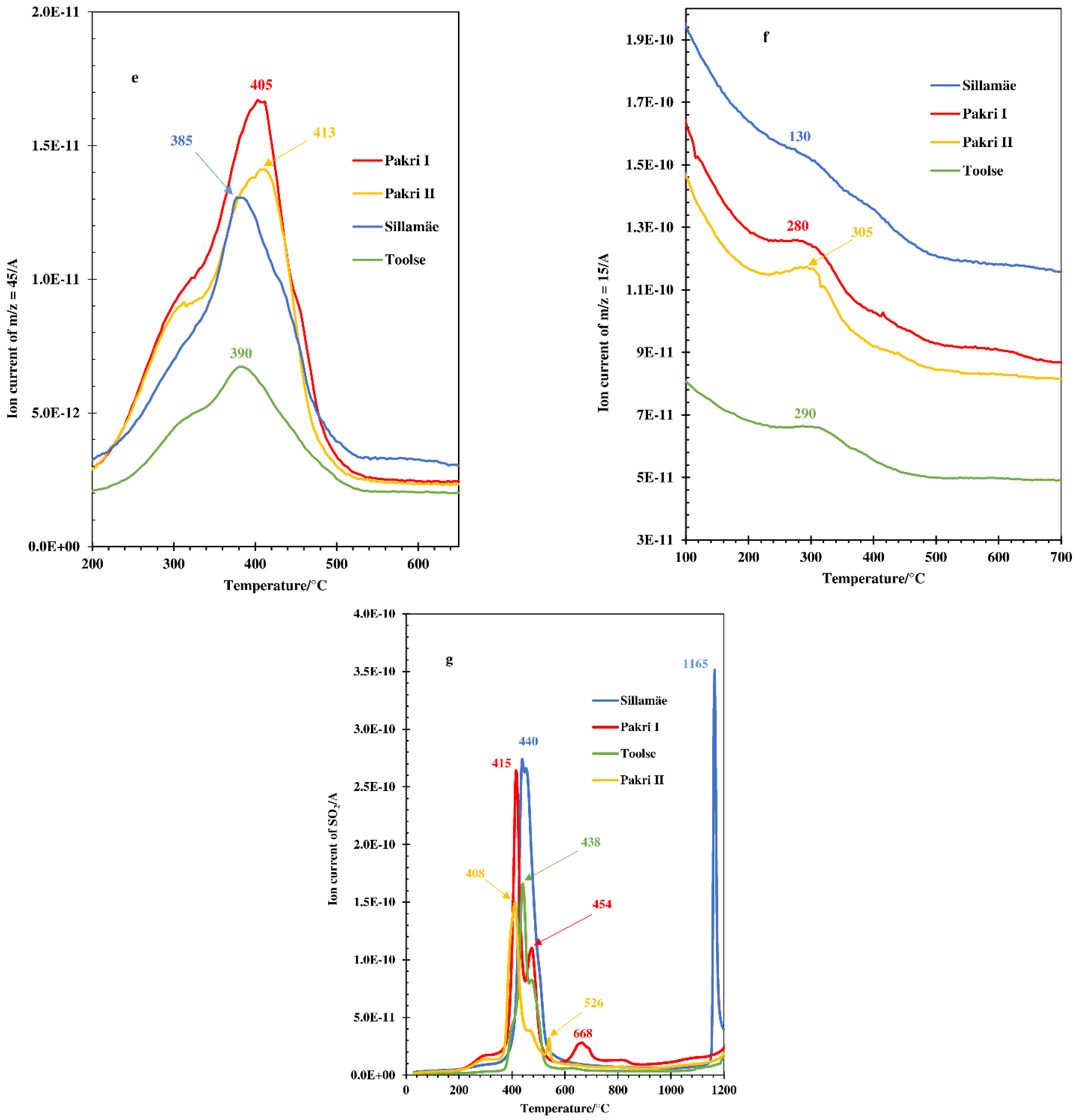
3.1. Thermal and MS Analysis
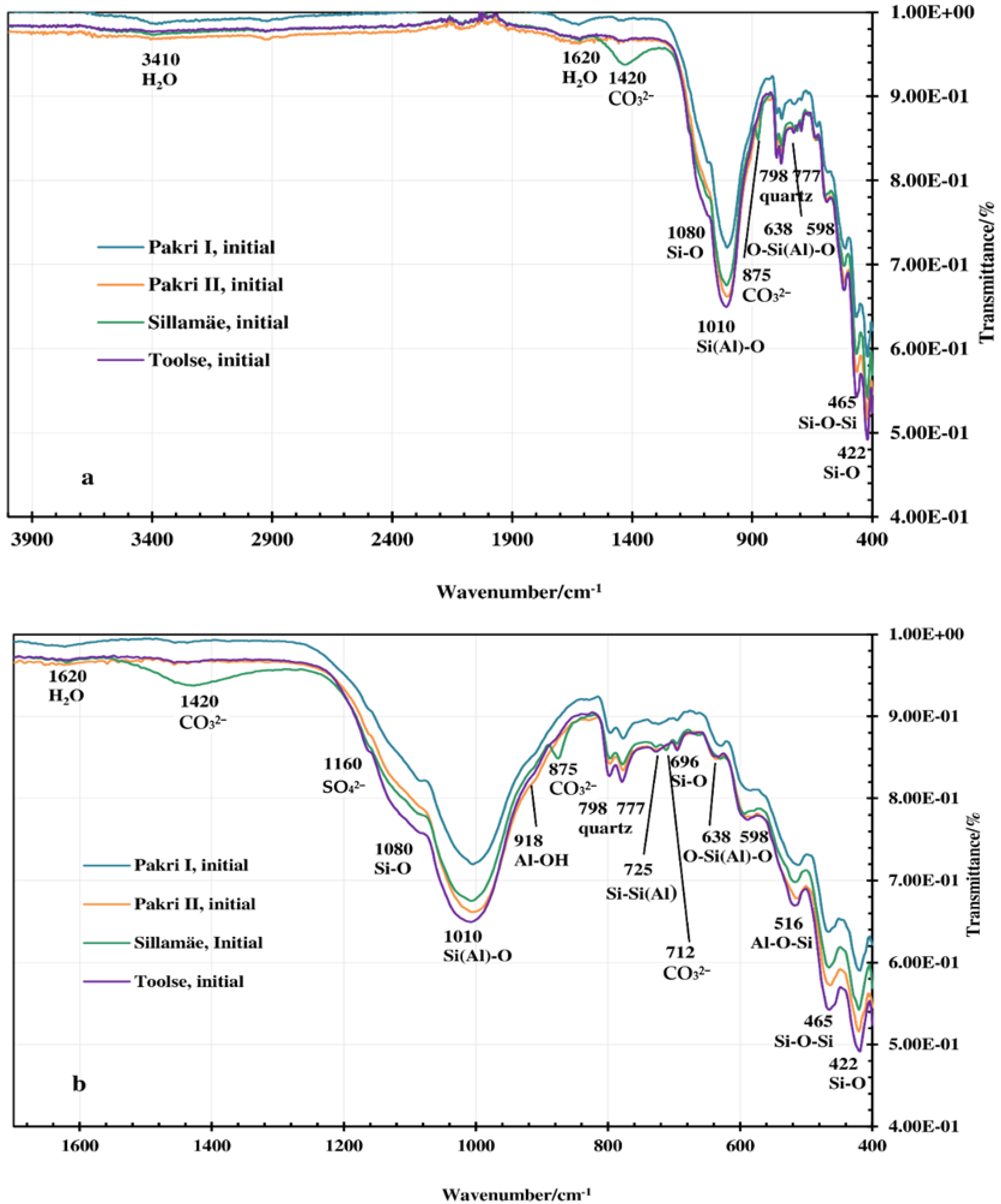
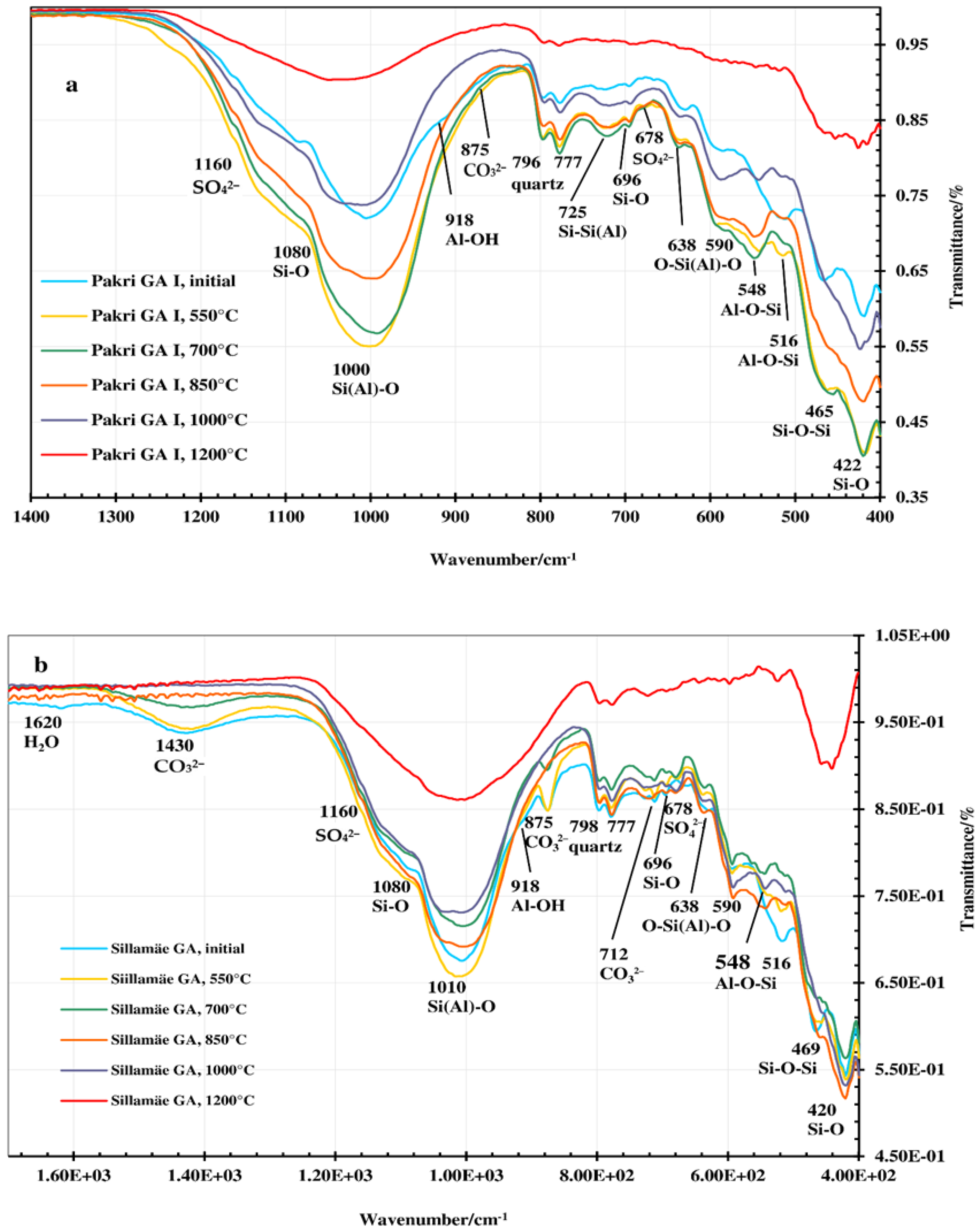
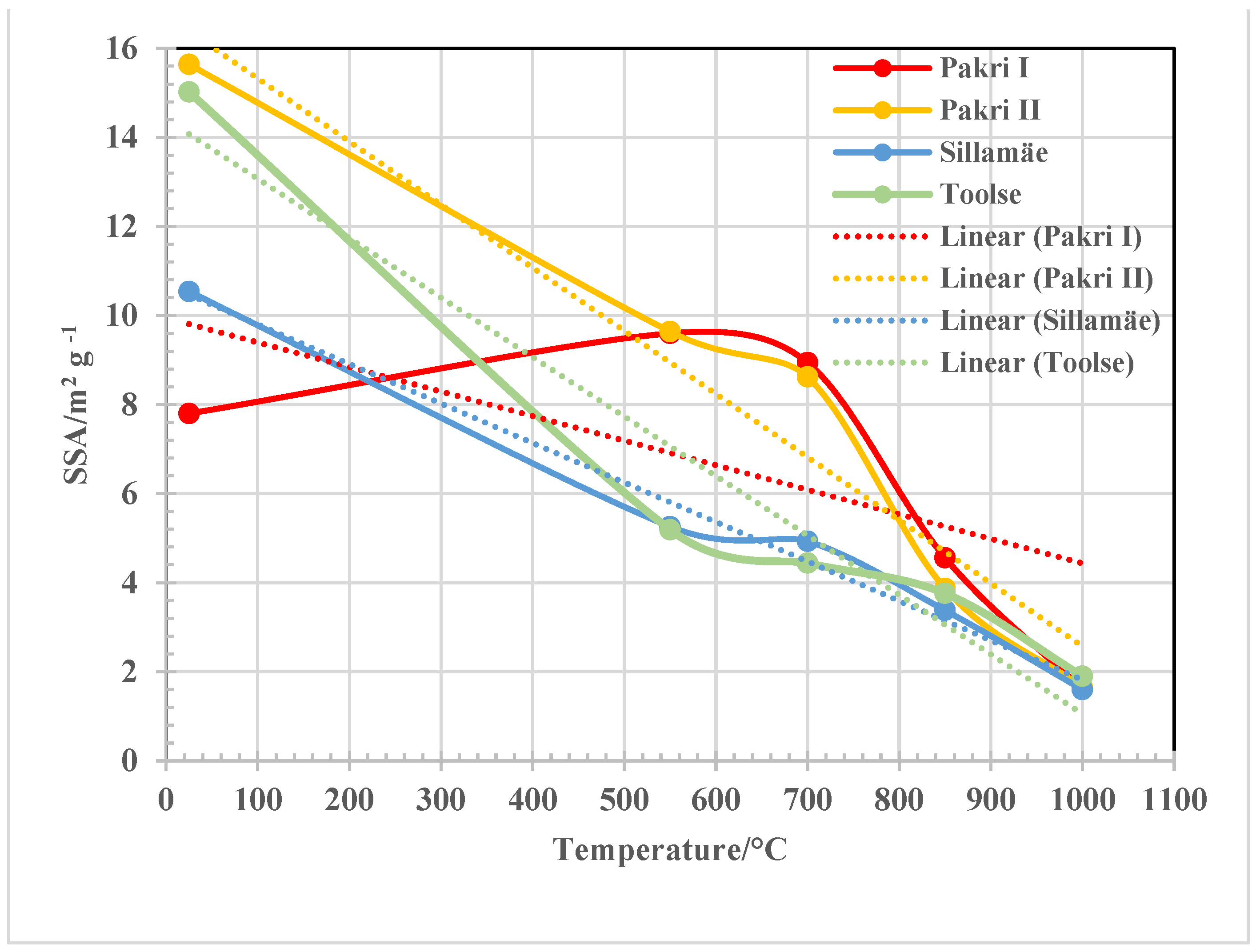
3.2. FT-IR, XRD and Morphology Analysis
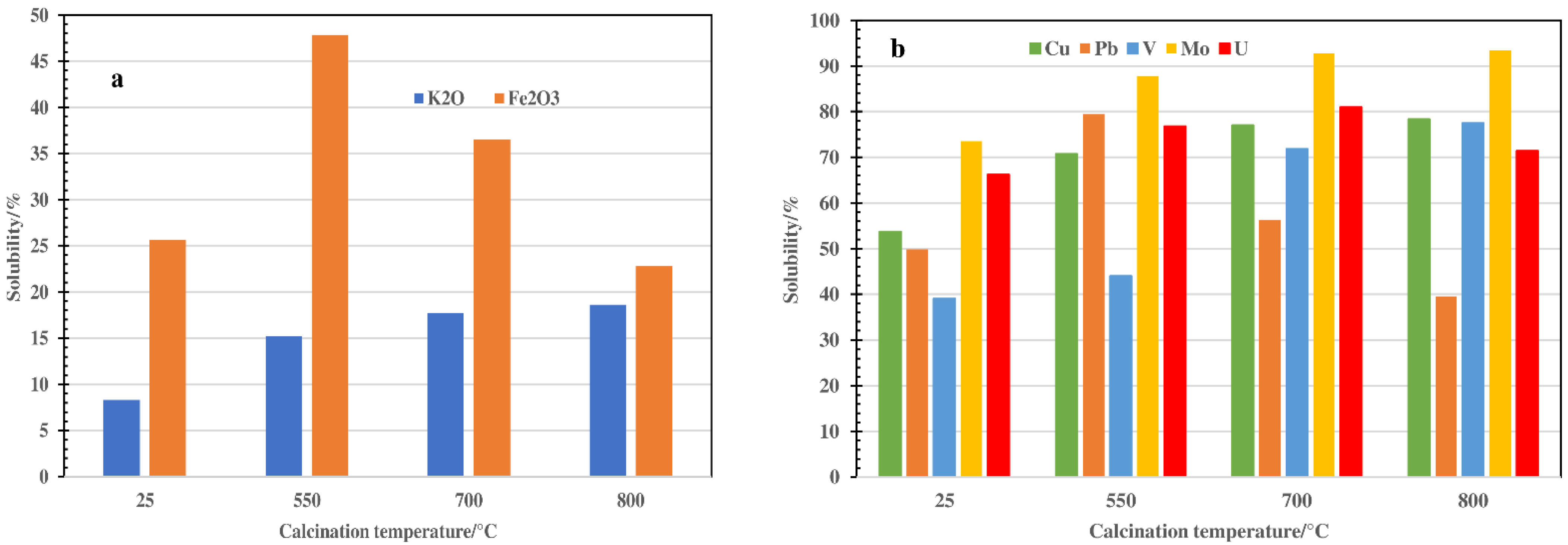
3.3. Solubility Test in Sulpuric Acid
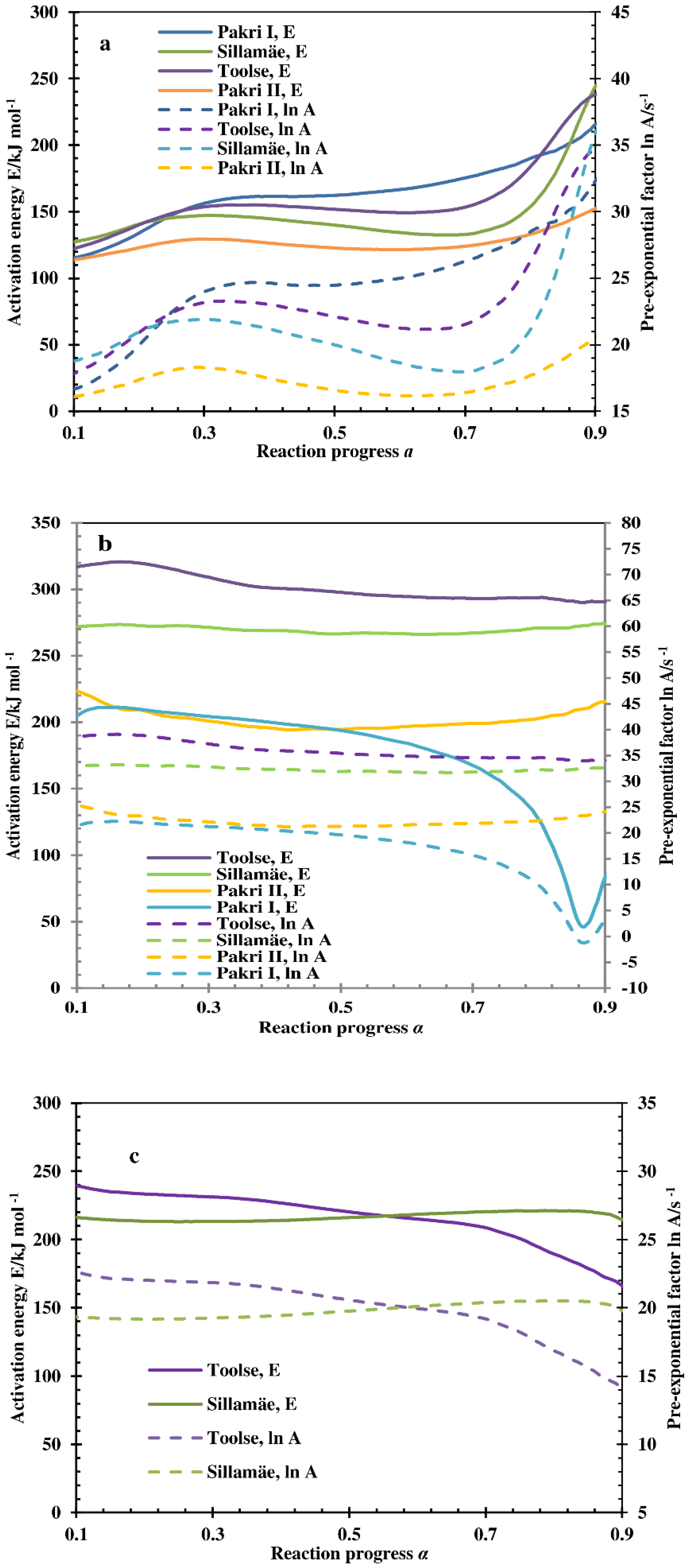
3.4. Kinetic Calculations
4. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| DTA | Differential thermal analysis |
| DTG | Differential TG |
| EGA | Evolved gas analysis |
| FTIR | Fourier transform infrared |
| GA | Graptolite–argillite |
| MP-AES | Microwave plasma atomic emission spectroscopy |
| MS | Mass spectroscopy |
| SEM | Scanning electron spectroscopy |
| BET SSA | Brunauer–Emmett–Teller specific surface area |
| TG | Thermogravimetry |
| XRD | X-ray diffraction |
References
- Brumsack, H.-J. The trace metal content of recent organic carbon-rich sediments: Implications for Cretaceous black shale formation. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2006, 232, 344–361. [Google Scholar] [CrossRef]
- Johnson, S.C.; Large, R.R.; Coveney, R.M.; Kelley, K.D.; Slack, J.F.; Steadman, J.A.; Gregory, D.D.; Sack, P.J.; Meffre, S. Secular distribution of highly metalliferous black shales corresponds with peaks in past atmosphere oxigenation. Min. Deposita. 2017, 52, 791–798. [Google Scholar] [CrossRef]
- Lavergren, U.; Åström, M.E.; Bergback, B.; Holmström, H. Mobility of trace elements in black shale assessed by leaching tests and sequential chemical reaction. Geochem. Explor. Environ. Analysis. 2009, 9, 71–79. [Google Scholar] [CrossRef]
- Canfield, D.E. A new model for Proterozoic ocean chemistry. Nature 1998, 396, 450–453. [Google Scholar] [CrossRef]
- Meyer, K.; Kump, L. Oceanic Euxinia in Earth History: Causes and Consequences. Annu. Rev. Earth Planet. Sci. 2008, 36, 251–288. [Google Scholar] [CrossRef]
- Lyons, T.W.; Anbar, A.D.; Severmann, S.; Scott, C.; Gill, B.C. Tracking Euxinia in the Ancient Ocean: A Multiproxy Perspective and Proterozoic Case Study. Annu. Rev. Earth Planet. Sci. 2009, 37, 507–534. [Google Scholar] [CrossRef]
- Blumenberg, M.; Wiese, F. Imbalanced nutrients as triggers for black shale formation in a shallow shelf setting during the OAE 2 (Wunstorf, Germany). Biogeosciences 2012, 9, 4139–4153. [Google Scholar] [CrossRef]
- Ozaki, K.; Tajima, S.; Tajika, E. Conditions required for oceanic anoxia/euxinia: Constraints from a one-dimensional ocean biochemical cycle model. Earth Planet. Sci. Lett. 2011, 304, 270–279. [Google Scholar] [CrossRef]
- Turgeon, S.C.; Creaser, R.A. Cretaceous oceanic anoxic event 2 triggered by a massive magmatic episode. Nature 2008, 454, 323–326. [Google Scholar] [CrossRef]
- Jenkyns, H.C. Geochemistry of oceanic anoxic events. Geochem. Geophys. Geosyst. 2010, 11, Q03004. [Google Scholar] [CrossRef]
- Voigt, S.; Gale, A.S.; Voigt, T. Sea-level change, carbon cycling and paleoclimate during the Late Cenomanian of northwest Europe; an integrated paleoenvironmental analysis. Creteaceous Res. 2006, 27, 836–858. [Google Scholar] [CrossRef]
- Weissert, H.; Erba, E. Volcanism, CO2 and palaeoclimate: A Late Jurassic—Early Cretaceous carbon and oxygen isotope record. J. Geol. Soc. 2004, 161, 695–702. [Google Scholar] [CrossRef]
- Mattioli, E.; Pittet, B.; Palliani, R.B.; Röhl, H.J.; Schmid-Röhl, A.; Morettini, E. Phytoplankton evidence for the timing and correlation of palaeoceanographical changes during the early Toarcian oceanic anoxic event (Early Jurassic). J. Geol. Soc. 2004, 161, 685–693. [Google Scholar] [CrossRef]
- Nederbragt, A.J.; Thurow, J.; Vonhof, H.; Brumsack, H.-J. Modelling oceanic carbon and phosphorus fluxes: Implications for the cause of the late Cenomanian oceanic Anoxic Event (OAE2). J. Geol. Soc. 2004, 161, 721–728. [Google Scholar] [CrossRef]
- Algeo, T.J.; Ingall, E. Sedimentary Corg:P ratios, paleocean ventilation, and Phanerozoic atmospheric pO2. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2007, 256, 130–155. [Google Scholar] [CrossRef]
- Mort, H.P.; Adatte, T.; Keller, G.; Bartels, D.; Föllmi, K.B.; Steinmann, P.; Berner, Z.; Chellai, E.H. Organic carbon deposition and phosphorus accumulation during Oceanic Anoxic Event 2 in Tarfaya, Morocco. Creteaceous Res. 2008, 29, 1008–1023. [Google Scholar] [CrossRef]
- Hade, S.; Soesoo, A. Estonian graptolite argillites revisited: A future resource? Oil Shale. 2014, 31, 4–18. [Google Scholar] [CrossRef]
- Voolma, M.; Soesoo, A.; Hade, S.; Hints, R.; Kallaste, T. Geochemical heterogeneity of Estonian graptolite argillite. Oil Shale. 2013, 30, 377–401. [Google Scholar] [CrossRef]
- Pagés, A.; Barnes, S.; Schmid, S.; Coveney, R.M.J.; Schwark, L.; Liu, W.; Grice, K.; Fan, H.; Wen, H. Geochemical investigation of the lower Cambrian mineralised black shales of South China and the late Devonian nick deposit, Canada. Ore Geol. Rev. 2018, 94, 396–413. [Google Scholar] [CrossRef]
- Dai, S.; Zheng, X.; Wang, X.; Finkelman, R.B.; Jiang, Y.; Ren, D.; Yan, X.; Zhou, Y. Stone coal in China: A review. Int. Geol. Review. 2018, 60, 736–753. [Google Scholar] [CrossRef]
- Marynowski, L.; Zatoń, M.; Rakociński, M.; Filipiak, P.; Kurkiewicz, S.; Pearce, T.J. Deciphering the upper Famennian Hangenberg Black Shale depositional environments based on multi-proxy record. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2012, 346–347, 66–86. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, W.; Zhang, Y.; Song, S.; Bao, S. In -situ investigation on mineral phase transition during roasting of vanadium-bearing stone coal. Adv. Powder Techn. 2017, 28, 1103–1107. [Google Scholar] [CrossRef]
- Li, W.; Ma, C.; Gong, W.; Zhu, X. Clean production technology for effective recovery of vanadium from shale: Interaction between activators and vanadium-loaded minerals. J. Clean. Prod. 2021, 315, 128170. [Google Scholar] [CrossRef]
- Lippmaa, E.; Maremäe, E.; Pihlak, A.T.; Aguraiuja, R. Estonian graptolitic argillites—ancient ores or future fuels? Oil Shale. 2009, 26, 530–539. [Google Scholar] [CrossRef]
- Palvadre, R. Possibilities of utilization organic-poor metalliferous black shales (argillite). Oil Shale. 2020, 37, 242–267. [Google Scholar] [CrossRef]
- Hints, R.; Hade, S.; Soesoo, A.; Voolma, M. Depositional framework of the East Baltic Tremadocian black shale revisited. GFF 2014, 136, 464–482. [Google Scholar] [CrossRef]
- Hints, R.; Pajusaar, S.; Urtson, K.; Liiv, M.; Kallaste, T. Metal enrichment in lithologically complex black shales: A case study from the Tremadocian of NE Estonia. Est. J. Earth Sci. 2021, 70, 36–50. [Google Scholar] [CrossRef]
- Lippmaa, E.; Maremäe, E.; Pihlak, A.T. Resources, production and processing of Baltoscandian multimetal black shales. Oil Shale. 2011, 28, 68–77. [Google Scholar] [CrossRef]
- Tribovillard, N.; Algeo, J.T.; Lyons, T.; Riboulleau, A. Trace metals as paleoredox and paleoproductivity proxies: An update. Chem. Geol. 2006, 232, 12–32. [Google Scholar] [CrossRef]
- Soesoo, A.; Vind, J.; Hade, S. Uranium and Thorium resources of Estonia. Minerals 2020, 10, 798. [Google Scholar] [CrossRef]
- Vind, J.; Bauert, H. Geochemical Characterization of the Tremadocian Black Shale in North-Western Estonia; Research Report No. EGF9330; Geological Survey of Estonia: Rakvere, Estonia, 2020; Volume 91. [Google Scholar]
- Schmidt, C.M.; Heide, K. Thermal analysis of hydrocarbons in Paleozoic black shales. J. Therm. Anal. Calorim. 2001, 64, 1297–1302. [Google Scholar] [CrossRef]
- Labus, M. Thermal methods implementation in analysis of fine-grained rocks containing organic matter. J. Therm. Anal. Calorim. 2017, 129, 965–973. [Google Scholar] [CrossRef]
- Labus, M.; Lempart, M. Studies of Polish Paleozoic shale rocks using FTIR and TG/DSC methods. J. Petrol. Sci. Eng. 2018, 161, 311–318. [Google Scholar] [CrossRef]
- Taylor, J.C. Computer programs for standardless quantitative analysis of minerals using the full powder diffraction profile. Powder Diffr. 1991, 6, 2–9. [Google Scholar] [CrossRef]
- Ward, C.R.; Taylor, J.C.; Cohen, D.R. Quantitative mineralogy of sandstones by X-ray diffractrometry and normative analysis. J. Sed. Geo. 1999, 69, 1050–1062. [Google Scholar] [CrossRef]
- Friedman, H.L. Kinetics of thermal degradation of char-forming plastics from thermogravimetry. Application to phenolic plastic. J. Polym. Sci. 1965, 6, 183–195. [Google Scholar] [CrossRef]
- AKTS Softwear and Setaram Instruments: A Global Solution for Kinetic Analysis and Determination of the Thermal Stability of Materials; AKTS AG: Sider, Switzerland, 2006; p. 88.
- Gaines, G.L.; Vedder, W. Dehydroxylation of Muscovite. Nature 1964, 4918, 495. [Google Scholar] [CrossRef]
- Kodama, H.; Brydon, J.E. Dehydroxylation of Microcrystalline Muscovite. Trans. Faraday Soc. 1968, 64, 3112–3119. [Google Scholar] [CrossRef]
- Lempart, M.; Derkowski, A.; Luberda-Durnaś, K.; Skiba, M.; Błachowski, A. Dehydrogenation and dihydroxylation as drivers of the thermal decomposition of Fe-chlorites. Am. Mineral. 2018, 103, 1837–1850. [Google Scholar] [CrossRef]
- Frost, R.L.; Weier, M.L.; Martens, W. Thermal decomposition of jarosite of potassium, sodium and lead. J. Therm. Anal. Calorim. 2005, 82, 115–118. [Google Scholar] [CrossRef]
- Paulik, F.; Paulik, J.; Arnold, M. Kinetics and mechanism of decomposition of pyrite under conventional and quasi-isothermal –quasi-isobaric thermoanalytical conditions. J. Therm. Anal. 1982, 25, 313–325. [Google Scholar] [CrossRef]
- Hu, G.; Dam-Johansen, K.; Wedel, S.; Hansen, J.P. Decomposition and oxidation of pyrite. Prog. Energy Combust. Sci. 2006, 32, 295–314. [Google Scholar] [CrossRef]
- Diko, M.; Ekosse, G.; Ogola, J. Fourier Transform Infrared Spectroscopy and thermal analysis of kaolinite clays from South Africa and Cameroon. Acta Geodyn. Geomater. 2016, 13, 149–158. [Google Scholar]
- Zhirong, L.; Uddin, M.A.; Zhanxue, S. FT-IR and XRD analysis of Natural Na-bentonite and Cu (II)-loaded Na-bentonite. Spectrochim. Acta Part A 2011, 79, 1013–1016. [Google Scholar] [CrossRef] [PubMed]
- Eisazadeh, A.; Kassim, K.A.; Nur, H. Solid-state NMR and FTIR studies of lime stabilized montmorillonitic and lateritic clays. Appl. Clay Sci. 2012, 67–68, 5–10. [Google Scholar] [CrossRef]
- Cizer, Ö.; Rodrigues-Navarro, C.; Ruiz-Agudo, E.; Elsen, J.; Van Gemert, D.; Van Balen, K. Phase and morphology evolution of carbonate precipitated by carbonation of hydrated lime. J. Mat. Sci. 2012, 47, 6151–6165. [Google Scholar] [CrossRef]
- Müller, C.M.; Pejcic, B.; Esteban, L.; Piane, C.D.; Raven, M.; Mizaikoff, B. Infrared Attenuated Total Reflectance Spectroscopy: An Innovative Strategy for Analyzing Mineral components in Energy Relevant Systems. Sci. Rep. 2014, 4, 6764. [Google Scholar] [CrossRef] [PubMed]
- Madejová, J. FTIR techniques in clay mineral studies. Vib. Spectrosc. 2012, 31, 1–10. [Google Scholar] [CrossRef]
- Davarcioğlu, B.; Ҫiftҁi, E. Investigation of Central Anatolian Clays by FTIR Spectroscopy (Arapli-Yesilhisar-Kayseri, Turkey). Int. J. Nat. Engin. Sci. 2009, 3, 167–174. [Google Scholar]
- Theodosoglou, E.; Koroneos, A.; Soldatos, T.; Zorba, T.; Paraskevopoulos, K.M. Comparative Fourier transform infrared and X-ray powder diffraction analysis of naturally occurred K-feldspars. Bull. Geol. Soc. Greece Jan. 2010, XLIII, 2752–2761. [Google Scholar] [CrossRef]
- Mozgawa, W.; Król, M.; Dyczek, J.; Deja, J. Investigation of the coal fly ashes using IR spectroscopy. Spectrochim. Acta Part A 2000, 56, 1819–1823. [Google Scholar] [CrossRef] [PubMed]
- Matteson, A.; Herron, M.M. End-member feldspar concentrations determined by FTIR spectral analysis. J. Sediment. Petrol. 1993, 63, 1144–1148. [Google Scholar]
- Alver, B.E.; Dikmen, G.; Alver, Ö. Investigation of the influence of heat treatment on the structural properties of illite-rich clay mineral using FT-IR, 29Si MAS NMR, TG and DTA methods. Anadolu. Univ. J. Sci. Technol. A Appl. Sci. Engin. 2016, 17, 823–829. [Google Scholar]
- Smith, D.H.; Seshadri, K.S. Infrared spectra of Mg2Ca(SO4)3, MgSO4, hexagonal CaSO4 and orthorhombic CaSO4. Spectrochim. Acta Part A 1999, 55, 795–805. [Google Scholar] [CrossRef]
- Bishop, J.L.; Murad, E. The visible and infrared properties of jarosite and alunite. Am. Mineral. 2005, 90, 1100–1107. [Google Scholar] [CrossRef]
- Barlow, S.G.; Manning, D.A.C. Influence of time and temperature on reactions and transformations of muscovite mica. Br. Ceram. Trans. 1999, 98, 122–126. [Google Scholar] [CrossRef]



| Compound/Formula | Pakri I | Pakri II | Sillamäe | Toolse |
|---|---|---|---|---|
| Quartz, SiO2 | 24.2 | 24.7 | 23.8 | 35.5 |
| Orthoclase, KAlSi3O8 | 54.7 | 57.3 | 38.0 | 41.8 |
| Muscovite, KAl2(AlSi3O10)(F,OH)2 | 7.2 | 7.8 | 3.8 | 9.1 |
| Jarosite, KFe33+(OH)6(SO4)2 | 4.5 | 1.0 | - | 0.4 |
| Chlorite, (Mg,Fe)3(SiAl)4O10(OH)2(Mg,Fe)3(OH)6 | 0.5 | 1.7 | 0.4 | - |
| Pyrite, FeS2 | 8.4 | 5.7 | 7.2 | 9.9 |
| Anatase, TiO2 | 0.5 | 0.7 | 0.3 | 0.4 |
| Hematite, Fe2O3 | - | 1.1 | - | 1.1 |
| Calcite, CaCO3 | - | - | 19.1 | - |
| Dolomite, CaMg(CO3)2 | - | - | 6.9 | 1.8 |
| Sphalerite, (Zn,Fe)S | - | - | 0.5 | - |
| Item | Sample/Content | |||
|---|---|---|---|---|
| Pakri I | Pakri II | Sillamäe | Toolse | |
| SiO2, % | 45.3 | 45.2 | 41.2 | 54.0 |
| Al2O3, % | 11.4 | 11.3 | 9.1 | 9.6 |
| K2O, % | 6.8 | 8.4 | 5.5 | 5.9 |
| Fe2O3, % | 7.9 | 5.4 | 8.1 | 9.4 |
| CaO, % | 0.3 | 0.2 | 11.8 | 1.6 |
| MgO, % | 0.8 | 1.0 | 1.4 | 1.8 |
| SO3total, % | 12.7 | 8.1 | 10.3 | 13.2 |
| CTC, % | 12.97 | 14.42 | 9.51 | 9.59 |
| CTIC, % | 0.40 | 0.24 | 2.23 | 0.32 |
| Corg *, % | 12.57 | 14.18 | 7.28 | 9.27 |
| V, ppm | 890 | 958 | 868 | 916 |
| Mo, ppm | 336 | 137 | 956 | 414 |
| U, ppm | 90 | 120 | 254 | 146 |
| Cu, ppm | 110 | 97 | 89 | 87 |
| Pb, ppm | 135 | 104 | 194 | 158 |
| BET SSA, m2 g−1 | 7.81 | 15.64 | 10.54 | 15.02 |
| Goss calorific value, MJ kg−1 | 5.27 | 6.13 | 3.25 | 4.37 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaljuvee, T.; Tõnsuaadu, K.; Einard, M.; Mikli, V.; Kivimäe, E.-K.; Kallaste, T.; Trikkel, A. Thermal Behavior of Estonian Graptolite–Argillite from Different Deposits. Processes 2022, 10, 1986. https://doi.org/10.3390/pr10101986
Kaljuvee T, Tõnsuaadu K, Einard M, Mikli V, Kivimäe E-K, Kallaste T, Trikkel A. Thermal Behavior of Estonian Graptolite–Argillite from Different Deposits. Processes. 2022; 10(10):1986. https://doi.org/10.3390/pr10101986
Chicago/Turabian StyleKaljuvee, Tiit, Kaia Tõnsuaadu, Marve Einard, Valdek Mikli, Eliise-Koidula Kivimäe, Toivo Kallaste, and Andres Trikkel. 2022. "Thermal Behavior of Estonian Graptolite–Argillite from Different Deposits" Processes 10, no. 10: 1986. https://doi.org/10.3390/pr10101986
APA StyleKaljuvee, T., Tõnsuaadu, K., Einard, M., Mikli, V., Kivimäe, E.-K., Kallaste, T., & Trikkel, A. (2022). Thermal Behavior of Estonian Graptolite–Argillite from Different Deposits. Processes, 10(10), 1986. https://doi.org/10.3390/pr10101986







