Clinical Implementation of Predictive Models Embedded within Electronic Health Record Systems: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Information Sources and Searches
2.3. Study Selection
2.4. Data Collection and Quality Assessment
2.5. Synthesis and Analysis of Results
3. Results
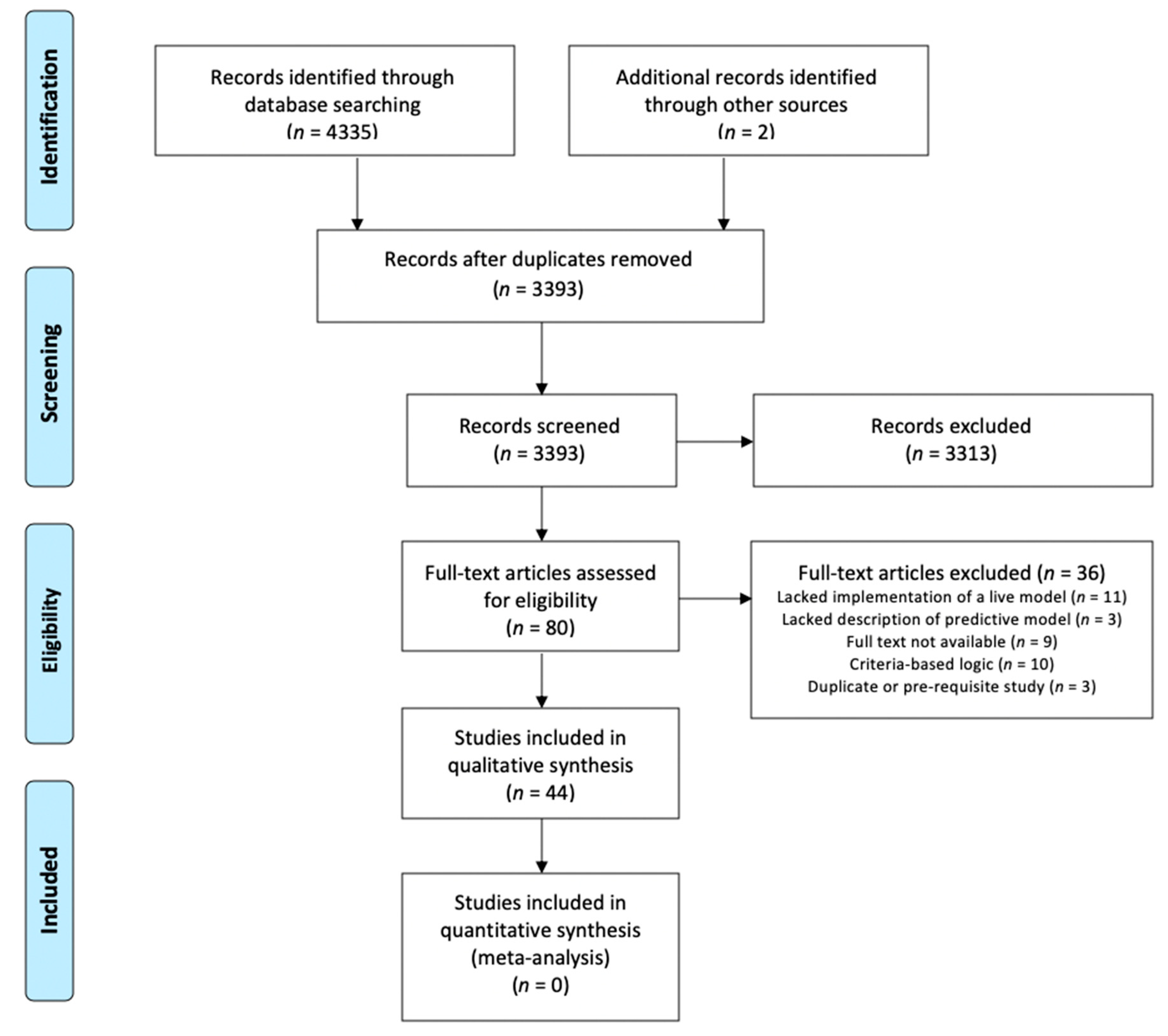
3.1. Study Selection
3.2. Study Characteristics: Study Settings, Study Design, and Clinical Domains
3.3. Predictive Models
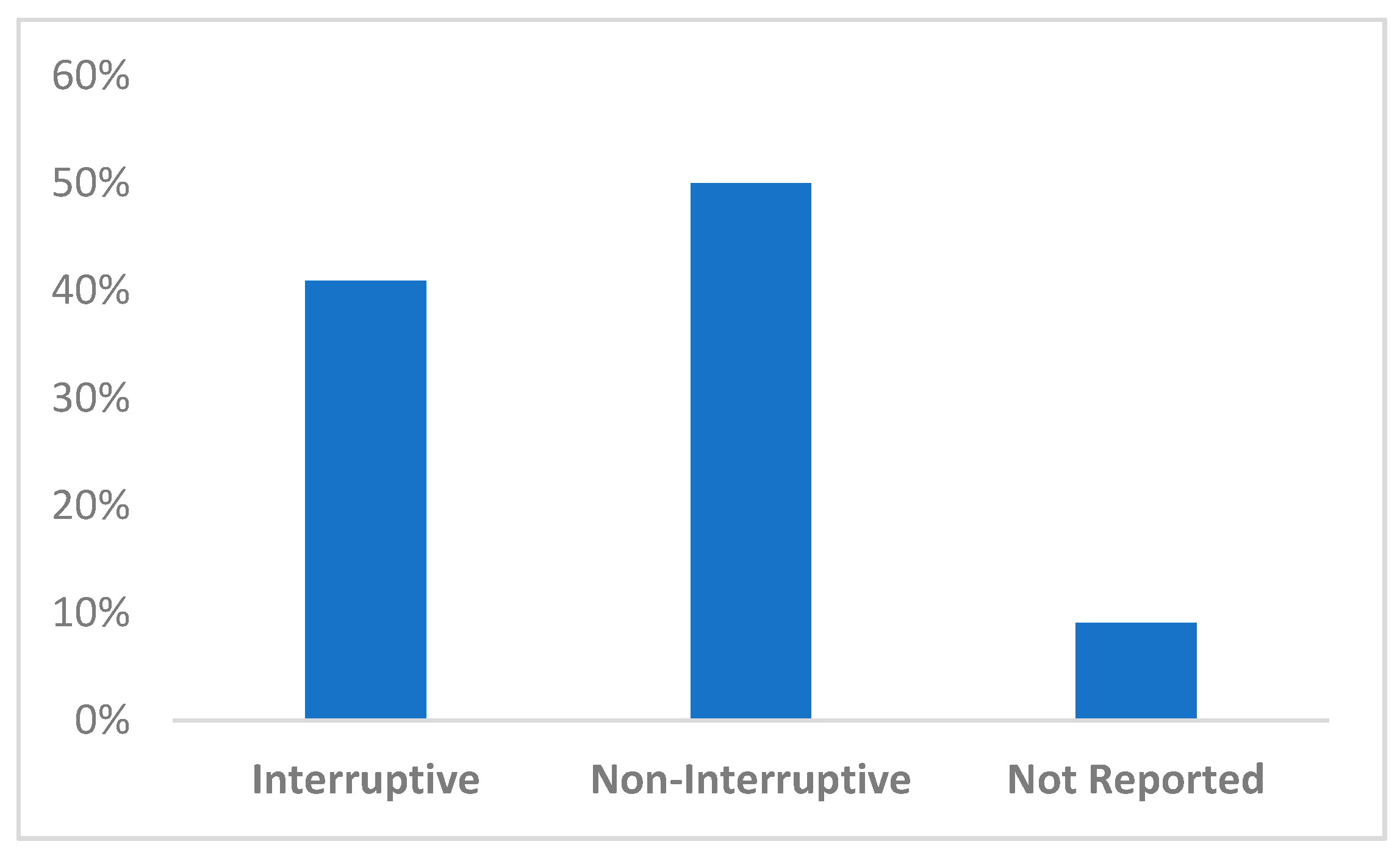
3.4. Integration into EHR Clinical Decision Support Tools and Implementation Challenges
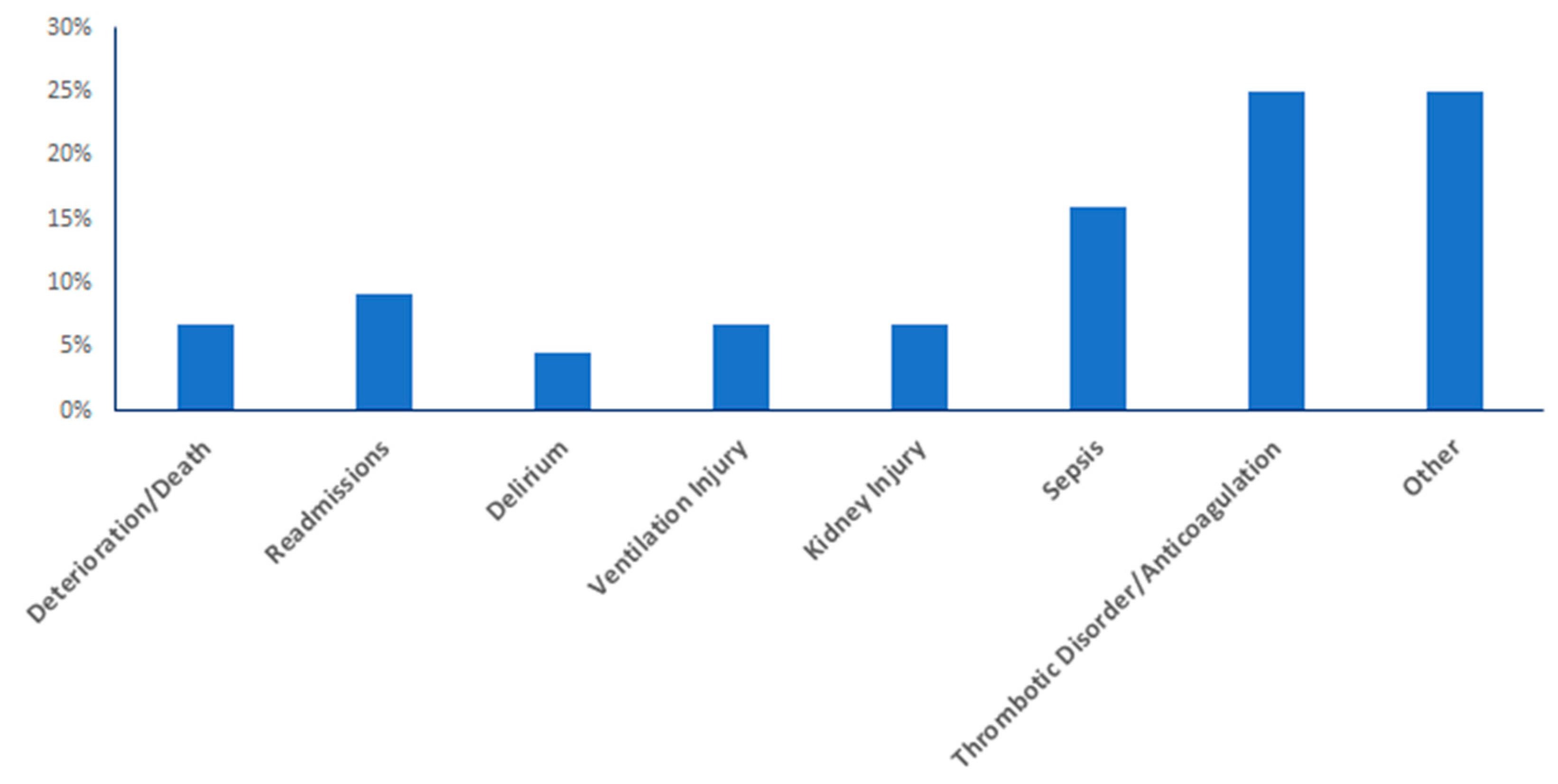
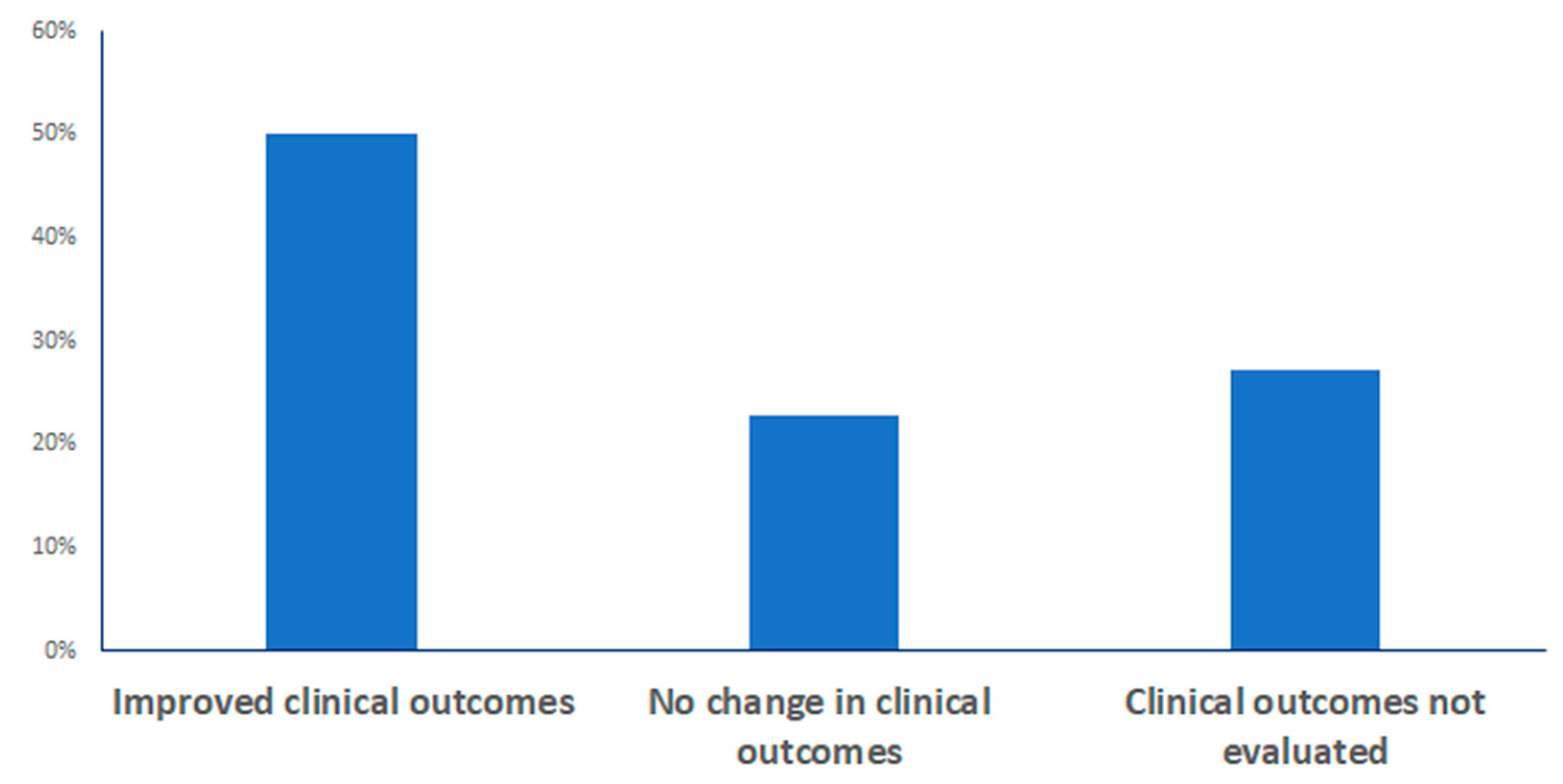
3.5. Impacts on Clinical Outcomes
3.6. Quality Assessment
4. Discussion
4.1. Summary of Evidence and Key Findings
4.2. Gaps in the Literature and Opportunities for Future Investigation
4.3. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. Detailed PubMed Search Strategy
References
- Parikh, R.B.; Kakad, M.; Bates, D.W. Integrating Predictive Analytics into High-Value Care: The Dawn of Precision Delivery. JAMA 2016, 315, 651–652. [Google Scholar] [CrossRef] [PubMed]
- Henry, J.; Pylypchuk, Y.; Searcy, T.; Patel, V. Adoption of Electronic Health Record Systems among U.S. Non-Federal Acute Care Hospitals: 2008–2015. Available online: /evaluations/data-briefs/non-federal-acute-care-hospital-ehr-adoption-2008-2015.php (accessed on 18 September 2019).
- Cohen, M.F. Impact of the HITECH financial incentives on EHR adoption in small, physician-owned practices. Int. J. Med. Inform. 2016, 94, 143–154. [Google Scholar] [CrossRef] [PubMed]
- Mennemeyer, S.T.; Menachemi, N.; Rahurkar, S.; Ford, E.W. Impact of the HITECH Act on physicians’ adoption of electronic health records. J. Am. Med. Inform. Assoc. 2016, 23, 375–379. [Google Scholar] [CrossRef] [PubMed]
- Bihorac, A.; Ozrazgat-Baslanti, T.; Ebadi, A.; Motaei, A.; Madkour, M.; Pardalos, P.M.; Lipori, G.; Hogan, W.R.; Efron, P.A.; Moore, F.; et al. MySurgeryRisk: Development and Validation of a Machine-learning Risk Algorithm for Major Complications and Death After Surgery. Ann. Surg. 2018, 269, 652. [Google Scholar] [CrossRef] [PubMed]
- Weller, G.B.; Lovely, J.; Larson, D.W.; Earnshaw, B.A.; Huebner, M. Leveraging electronic health records for predictive modeling of post-surgical complications. Stat. Methods Med. Res. 2017, 27, 3271–3285. [Google Scholar] [CrossRef]
- Corey, K.M.; Kashyap, S.; Lorenzi, E.; Lagoo-Deenadayalan, S.A.; Heller, K.; Whalen, K.; Balu, S.; Heflin, M.T.; McDonald, S.R.; Swaminathan, M.; et al. Development and validation of machine learning models to identify high-risk surgical patients using automatically curated electronic health record data (Pythia): A retrospective, single-site study. PLoS Med. 2018, 15, e1002701. [Google Scholar] [CrossRef]
- Nemati, S.; Holder, A.; Razmi, F.; Stanley, M.D.; Clifford, G.D.; Buchman, T.G. An Interpretable Machine Learning Model for Accurate Prediction of Sepsis in the ICU. Crit. Care Med. 2018, 46, 547–553. [Google Scholar] [CrossRef]
- Rajkomar, A.; Oren, E.; Chen, K.; Dai, A.M.; Hajaj, N.; Hardt, M.; Liu, P.J.; Liu, X.; Marcus, J.; Sun, M.; et al. Scalable and accurate deep learning with electronic health records. NPJ Digit. Med. 2018, 1, 18. [Google Scholar] [CrossRef]
- Xue, Y.; Liang, H.; Norbury, J.; Gillis, R.; Killingworth, B. Predicting the risk of acute care readmissions among rehabilitation inpatients: A machine learning approach. J. Biomed. Inform. 2018, 86, 143–148. [Google Scholar] [CrossRef]
- Artetxe, A.; Beristain, A.; Graña, M. Predictive models for hospital readmission risk: A systematic review of methods. Comput. Methods Programs Biomed. 2018, 164, 49–64. [Google Scholar] [CrossRef]
- Byrd, R.J.; Steinhubl, S.R.; Sun, J.; Ebadollahi, S.; Stewart, W.F. Automatic identification of heart failure diagnostic criteria, using text analysis of clinical notes from electronic health records. Int. J. Med. Inform. 2014, 83, 983–992. [Google Scholar] [CrossRef] [PubMed]
- Alemi, F.; Avramovic, S.; Schwartz, M.D. Electronic Health Record-Based Screening for Substance Abuse. Big Data 2018, 6, 214–224. [Google Scholar] [CrossRef]
- Taylor, R.A.; Pare, J.R.; Venkatesh, A.K.; Mowafi, H.; Melnick, E.R.; Fleischman, W.; Hall, M.K. Prediction of In-hospital Mortality in Emergency Department Patients with Sepsis: A Local Big Data-Driven, Machine Learning Approach. Acad. Emerg. Med. 2016, 23, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Hoogendoorn, M.; El Hassouni, A.; Mok, K.; Ghassemi, M.; Szolovits, P. Prediction using patient comparison vs. modeling: A case study for mortality prediction. In Proceedings of the 2016 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Orlando, FL, USA, 16–20 August 2016; pp. 2464–2467. [Google Scholar]
- Shickel, B.; Tighe, P.J.; Bihorac, A.; Rashidi, P. Deep EHR: A Survey of Recent Advances in Deep Learning Techniques for Electronic Health Record (EHR) Analysis. IEEE J. Biomed. Health Inform. 2018, 22, 1589–1604. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, B.A.; Navar, A.M.; Pencina, M.J.; Ioannidis, J.P.A. Opportunities and challenges in developing risk prediction models with electronic health records data: A systematic review. J. Am. Med. Inform. Assoc. 2017, 24, 198–208. [Google Scholar] [CrossRef] [PubMed]
- Kruse, C.S.; Goswamy, R.; Raval, Y.; Marawi, S. Challenges and Opportunities of Big Data in Health Care: A Systematic Review. JMIR Med. Inform. 2016, 4, e38. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, S.; Vaidya, V.; Chandrashekaraiah, P.; Kulkarni, H.; Chen, L.; Rudeen, K.; Narayanan, B.; Inbar, O.; Hyde, B. Development of an artificial intelligence model to predict survival at specific time intervals for lung cancer patients. J. Clin. Oncol. 2019, 37, 6556. [Google Scholar] [CrossRef]
- Shaw, J.; Rudzicz, F.; Jamieson, T.; Goldfarb, A. Artificial Intelligence and the Implementation Challenge. J. Med. Internet Res. 2019, 21, e13659. [Google Scholar] [CrossRef]
- He, J.; Baxter, S.L.; Xu, J.; Xu, J.; Zhou, X.; Zhang, K. The practical implementation of artificial intelligence technologies in medicine. Nat. Med. 2019, 25, 30–36. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Health Care Interventions: Explanation and Elaboration. PLoS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. The PRISMA Group Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Rights (OCR), O. for C. HITECH Act Enforcement Interim Final Rule. Available online: https://www.hhs.gov/hipaa/for-professionals/special-topics/HITECH-act-enforcement-interim-final-rule/index.html (accessed on 29 June 2020).
- Downs, S.H.; Black, N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J. Epidemiol. Community Health 1998, 52, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Maynard, G.A.; Morris, T.A.; Jenkins, I.H.; Stone, S.; Lee, J.; Renvall, M.; Fink, E.; Schoenhaus, R. Optimizing Prevention of Hospital-acquired Venous Thromboembolism (VTE): Prospective Validation of a VTE Risk Assessment Model. J. Hosp. Med. 2010, 5, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Novis, S.J.; Havelka, G.E.; Ostrowski, D.; Levin, B.; Blum-Eisa, L.; Prystowsky, J.B.; Kibbe, M.R. Prevention of thromboembolic events in surgical patients through the creation and implementation of a computerized risk assessment program. J. Vasc. Surg. 2010, 51, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Fossum, M.; Alexander, G.L.; Ehnfors, M.; Ehrenberg, A. Effects of a computerized decision support system on pressure ulcers and malnutrition in nursing homes for the elderly. Int. J. Med. Inform. 2011, 80, 607–617. [Google Scholar] [CrossRef] [PubMed]
- Herasevich, V.; Tsapenko, M.; Kojicic, M.; Ahmed, A.; Kashyap, R.; Venkata, C.; Shahjehan, K.; Thakur, S.J.; Pickering, B.W.; Zhang, J.; et al. Limiting ventilator-induced lung injury through individual electronic medical record surveillance. Crit. Care Med. 2011, 39, 34–39. [Google Scholar] [CrossRef]
- Nelson, J.L.; Smith, B.L.; Jared, J.D.; Younger, J.G. Prospective trial of real-time electronic surveillance to expedite early care of severe sepsis. Ann. Emerg. Med. 2011, 57, 500–504. [Google Scholar] [CrossRef]
- Umscheid, C.A.; Hanish, A.; Chittams, J.; Weiner, M.G.; Hecht, T.E.H. Effectiveness of a novel and scalable clinical decision support intervention to improve venous thromboembolism prophylaxis: A quasi-experimental study. BMC Med. Inform. Decis. Mak. 2012, 12, 92. [Google Scholar] [CrossRef]
- Baillie, C.A.; Vanzandbergen, C.; Tait, G.; Hanish, A.; Leas, B.; French, B.; William Hanson, C.; Behta, M.; Umscheid, C.A. The readmission risk flag: Using the electronic health record to automatically identify patients at risk for 30-day readmission. J. Hosp. Med. 2013, 8, 689–695. [Google Scholar] [CrossRef]
- Amarasingham, R.; Patel, P.C.; Toto, K.; Nelson, L.L.; Swanson, T.S.; Moore, B.J.; Xie, B.; Zhang, S.; Alvarez, K.S.; Ma, Y.; et al. Allocating scarce resources in real-time to reduce heart failure readmissions: A prospective, controlled study. BMJ Qual. Saf. 2013, 22, 998–1005. [Google Scholar] [CrossRef]
- Litvin, C.; Ornstein, S.M. Use of clinical decision support tools to improve primary care chronic kidney disease management. J. Gen. Intern. Med. 2013, 28, S448. [Google Scholar]
- Oh, S.-H.; Park, E.-J.; Jin, Y.; Piao, J.; Lee, S.-M. Automatic delirium prediction system in a Korean surgical intensive care unit. Nurs. Crit. Care 2014, 19, 281–291. [Google Scholar] [CrossRef] [PubMed]
- Resetar, E.; McMullen, K.M.; Russo, A.J.; Doherty, J.A.; Gase, K.A.; Woeltje, K.F. Development, Implementation and Use of Electronic Surveillance for Ventilator-Associated Events (VAE) in Adults. AMIA Annu. Symp. Proc. 2014, 2014, 1010–1017. [Google Scholar] [PubMed]
- Amland, R.C.; Dean, B.B.; Yu, H.; Ryan, H.; Orsund, T.; Hackman, J.L.; Roberts, S.R. Computerized Clinical Decision Support to Prevent Venous Thromboembolism Among Hospitalized Patients: Proximal Outcomes from a Multiyear Quality Improvement Project. J. Healthc Qual. 2015, 37, 221–231. [Google Scholar] [CrossRef]
- Faerber, A.E.; Horvath, R.; Stillman, C.; O’Connell, M.L.; Hamilton, A.L.; Newhall, K.A.; Likosky, D.S.; Goodney, P.P. Development and pilot feasibility study of a health information technology tool to calculate mortality risk for patients with asymptomatic carotid stenosis: The Carotid Risk Assessment Tool (CARAT). BMC Med. Inform. Decis. Mak. 2015, 15, 20. [Google Scholar] [CrossRef] [PubMed]
- Hao, S.; Wang, Y.; Jin, B.; Shin, A.Y.; Zhu, C.; Huang, M.; Zheng, L.; Luo, J.; Hu, Z.; Fu, C.; et al. Development, Validation and Deployment of a Real Time 30 Day Hospital Readmission Risk Assessment Tool in the Maine Healthcare Information Exchange. PLoS ONE 2015, 10, e0140271. [Google Scholar] [CrossRef] [PubMed]
- Kharbanda, E.O.; Nordin, J.D.; Sinaiko, A.R.; Ekstrom, H.L.; Stultz, J.M.; Sherwood, N.E.; Fontaine, P.L.; Asche, S.E.; Dehmer, S.P.; Amundson, J.H.; et al. TeenBP: Development and Piloting of an EHR-Linked Clinical Decision Support System to Improve Recognition of Hypertension in Adolescents. EGEMS 2015, 3, 1142. [Google Scholar] [CrossRef] [PubMed]
- Lustig, D.B.; Rodriguez, R.; Wells, P.S. Implementation and validation of a risk stratification method at The Ottawa Hospital to guide thromboprophylaxis in ambulatory cancer patients at intermediate-high risk for venous thrombosis. Thromb. Res. 2015, 136, 1099–1102. [Google Scholar] [CrossRef]
- Umscheid, C.A.; Betesh, J.; VanZandbergen, C.; Hanish, A.; Tait, G.; Mikkelsen, M.E.; French, B.; Fuchs, B.D. Development, Implementation, and Impact of an Automated Early Warning and Response System for Sepsis. J. Hosp. Med. 2015, 10, 26–31. [Google Scholar] [CrossRef]
- Depinet, H.; von Allmen, D.; Towbin, A.; Hornung, R.; Ho, M.; Alessandrini, E. Risk Stratification to Decrease Unnecessary Diagnostic Imaging for Acute Appendicitis. Pediatrics 2016, 138, e20154031. [Google Scholar] [CrossRef]
- Narayanan, N.; Gross, A.K.; Pintens, M.; Fee, C.; MacDougall, C. Effect of an electronic medical record alert for severe sepsis among ED patients. Am. J. Emerg. Med. 2016, 34, 185–188. [Google Scholar] [CrossRef] [PubMed]
- Vinson, D.R.; Ballard, D.W.; Mark, D.G.; Yealy, D.M.; Reed, M.E.; Huang, J.; Rauchwerger, A.S.; Lin, J.S.; Kene, M.V.; Chettipally, U.K. Impact of electronic clinical decision support on initial site of care for emergency department patients with acute pulmonary embolism. Ann. Emerg. Med. 2016, 68, S54. [Google Scholar] [CrossRef]
- Aakre, C.; Franco, P.M.; Ferreyra, M.; Kitson, J.; Li, M.; Herasevich, V. Prospective validation of a near real-time EHR-integrated automated SOFA score calculator. Int. J. Med. Inform. 2017, 103, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Arts, D.L.; Abu-Hanna, A.; Medlock, S.K.; van Weert, H.C.P.M. Effectiveness and usage of a decision support system to improve stroke prevention in general practice: A cluster randomized controlled trial. PLoS ONE 2017, 12, e0170974. [Google Scholar] [CrossRef]
- Bookman, K.; West, D.; Ginde, A.; Wiler, J.; McIntyre, R.; Hammes, A.; Carlson, N.; Steinbruner, D.; Solley, M.; Zane, R. Embedded Clinical Decision Support in Electronic Health Record Decreases Use of High-cost Imaging in the Emergency Department: EmbED study. Acad. Emerg. Med. 2017, 24, 839–845. [Google Scholar] [CrossRef]
- Jin, Y.; Jin, T.; Lee, S.-M. Automated Pressure Injury Risk Assessment System Incorporated Into an Electronic Health Record System. Nurs. Res. 2017, 66, 462–472. [Google Scholar] [CrossRef]
- Samal, L.; D’Amore, J.D.; Bates, D.W.; Wright, A. Implementation of a scalable, web-based, automated clinical decision support risk-prediction tool for chronic kidney disease using C-CDA and application programming interfaces. J. Am. Med. Inform. Assoc. 2017, 24, 1111–1115. [Google Scholar] [CrossRef]
- Shimabukuro, D.W.; Barton, C.W.; Feldman, M.D.; Mataraso, S.J.; Das, R. Effect of a machine learning-based severe sepsis prediction algorithm on patient survival and hospital length of stay: A randomised clinical trial. BMJ Open Respir. Res. 2017, 4, e000234. [Google Scholar] [CrossRef]
- Chaturvedi, S.; Kelly, A.; Prabhakaran, S.; Saposnik, G.; Lee, L.; Malik, A.; Boerman, C.; Serlin, G. Electronic decision support for improvement of contemporary therapy for stroke prevention (EDICTS). Neurology 2018, 90, 569–573. [Google Scholar]
- Cherkin, D.; Balderson, B.; Wellman, R.; Hsu, C.; Sherman, K.J.; Evers, S.C.; Hawkes, R.; Cook, A.; Levine, M.D.; Piekara, D.; et al. Effect of Low Back Pain Risk-Stratification Strategy on Patient Outcomes and Care Processes: The MATCH Randomized Trial in Primary Care. J. Gen. Intern. Med. 2018, 33, 1324–1336. [Google Scholar] [CrossRef]
- Ebinger, J.E.; Strauss, C.E.; Garberich, R.R.; Bradley, S.M.; Rush, P.; Chavez, I.J.; Poulose, A.K.; Porten, B.R.; Henry, T.D. Value-Based ST-Segment-Elevation Myocardial Infarction Care Using Risk-Guided Triage and Early Discharge. Circ. Cardiovasc. Qual. Outcomes 2018, 11, e004553. [Google Scholar] [CrossRef] [PubMed]
- Hebert, C.; Flaherty, J.; Smyer, J.; Ding, J.; Mangino, J.E. Development and validation of an automated ventilator-associated event electronic surveillance system: A report of a successful implementation. Am. J. Infect. Control. 2018, 46, 316–321. [Google Scholar] [CrossRef] [PubMed]
- Jung, A.D.; Baker, J.; Droege, C.A.; Nomellini, V.; Johannigman, J.; Holcomb, J.B.; Goodman, M.D.; Pritts, T.A. Sooner is better: Use of a real-time automated bedside dashboard improves sepsis care. J. Surg. Res. 2018, 231, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.-J.; Jin, Y.; Jin, T.; Lee, S.-M. Automated Medication Error Risk Assessment System (Auto-MERAS). J. Nurs. Care Qual. 2018, 33, 86–93. [Google Scholar] [CrossRef]
- Karlsson, L.; Nilsson, S.; Bång, M.; Nilsson, L.; Charitakis, E.; Janzon, M. A clinical decision support tool for improving adherence to guidelines on anticoagulant therapy in patients with atrial fibrillation at risk of stroke: A cluster-randomized trial in a Swedish primary care setting (the CDS-AF study). PLoS Med. 2018, 15, e1002528. [Google Scholar] [CrossRef]
- Moon, K.-J.; Jin, Y.; Jin, T.; Lee, S.-M. Development and validation of an automated delirium risk assessment system (Auto-DelRAS) implemented in the electronic health record system. Int. J. Nurs. Stud. 2018, 77, 46–53. [Google Scholar] [CrossRef]
- Ridgway, J.P.; Almirol, E.A.; Bender, A.; Richardson, A.; Schmitt, J.; Friedman, E.; Lancki, N.; Leroux, I.; Pieroni, N.; Dehlin, J.; et al. Which Patients in the Emergency Department Should Receive Preexposure Prophylaxis? Implementation of a Predictive Analytics Approach. AIDS Patient Care STDS 2018, 32, 202–207. [Google Scholar] [CrossRef]
- Turrentine, F.E.; Sohn, M.-W.; Wilson, S.L.; Stanley, C.; Novicoff, W.; Sawyer, R.G.; Williams, M.D. Fewer thromboembolic events after implementation of a venous thromboembolism risk stratification tool. J. Surg. Res. 2018, 225, 148–156. [Google Scholar] [CrossRef]
- Villa, S.; Weber, E.J.; Polevoi, S.; Fee, C.; Maruoka, A.; Quon, T. Decreasing triage time: Effects of implementing a step-wise ESI algorithm in an EHR. Int. J. Qual. Health Care 2018, 30, 375–381. [Google Scholar] [CrossRef]
- Vinson, D.R.; Mark, D.G.; Chettipally, U.K.; Huang, J.; Rauchwerger, A.S.; Reed, M.E.; Lin, J.S.; Kene, M.V.; Wang, D.H.; Sax, D.R.; et al. Increasing Safe Outpatient Management of Emergency Department Patients with Pulmonary Embolism A Controlled Pragmatic Trial. Ann. Intern. Med. 2018, 169, 855–865. [Google Scholar] [CrossRef] [PubMed]
- Bedoya, A.D.; Clement, M.E.; Phelan, M.; Steorts, R.C.; O’Brien, C.; Goldstein, B.A. Minimal Impact of Implemented Early Warning Score and Best Practice Alert for Patient Deterioration. Crit. Care Med. 2019, 47, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Brennan, M.; Puri, S.; Ozrazgat-Baslanti, T.; Feng, Z.; Ruppert, M.; Hashemighouchani, H.; Momcilovic, P.; Li, X.; Wang, D.Z.; Bihorac, A. Comparing clinical judgment with the MySurgeryRisk algorithm for preoperative risk assessment: A pilot usability study. Surgery 2019, 165, 1035–1045. [Google Scholar] [CrossRef] [PubMed]
- Ekstrom, H.L.; Kharbanda, E.O.; Ballard, D.W.; Vinson, D.R.; Vazquez-Benitez, G.; Chettipally, U.K.; Dehmer, S.P.; Kunisetty, G.; Sharma, R.; Rauchwerger, A.S.; et al. Development of a Clinical Decision Support System for Pediatric Abdominal Pain in Emergency Department Settings Across Two Health Systems Within the HCSRN. EGEMS 2019, 7, 15. [Google Scholar] [CrossRef] [PubMed]
- Giannini, H.M.; Ginestra, J.C.; Chivers, C.; Draugelis, M.; Hanish, A.; Schweickert, W.D.; Fuchs, B.D.; Meadows, L.; Lynch, M.; Donnelly, P.J.; et al. A Machine Learning Algorithm to Predict Severe Sepsis and Septic Shock: Development, Implementation, and Impact on Clinical Practice. Crit. Care Med. 2019, 47, 1485–1492. [Google Scholar] [CrossRef] [PubMed]
- Khoong, E.C.; Karliner, L.; Lo, L.; Stebbins, M.; Robinson, A.; Pathak, S.; Santoyo-Olsson, J.; Scherzer, R.; Peralta, C.A. A Pragmatic Cluster Randomized Trial of an Electronic Clinical Decision Support System to Improve Chronic Kidney Disease Management in Primary Care: Design, Rationale, and Implementation Experience. JMIR Res. Protoc. 2019, 8, e14022. [Google Scholar] [CrossRef] [PubMed]
- Ogunwole, S.M.; Phillips, J.; Gossett, A.; Downs, J.R. Putting veterans with heart failure FIRST improves follow-up and reduces readmissions. BMJ Open Qual. 2019, 8, e000386. [Google Scholar] [CrossRef]
- Embi, P.J.; Leonard, A.C. Evaluating alert fatigue over time to EHR-based clinical trial alerts: Findings from a randomized controlled study. J. Am. Med. Inform. Assoc. 2012, 19, e145–e148. [Google Scholar] [CrossRef]
- Harris, A.H. Path from Predictive Analytics to Improved Patient Outcomes: A Framework to Guide Use, Implementation, and Evaluation of Accurate Surgical Predictive Models. Ann. Surg. 2017, 265, 461–463. [Google Scholar] [CrossRef]
- Benda, N.C.; Das, L.T.; Abramson, E.L.; Blackburn, K.; Thoman, A.; Kaushal, R.; Zhang, Y.; Ancker, J.S. “How did you get to this number?” Stakeholder needs for implementing predictive analytics: A pre-implementation qualitative study. J. Am. Med. Inform. Assoc. 2020, 27, 709–716. [Google Scholar] [CrossRef]
- Fountaine, T.; McCarthy, B.; Saleh, T. Building the AI-Powered Organization. Available online: https://hbr.org/2019/07/building-the-ai-powered-organization (accessed on 1 July 2019).
- Greenhalgh, T.; Wherton, J.; Papoutsi, C.; Lynch, J.; Hughes, G.; A’Court, C.; Hinder, S.; Fahy, N.; Procter, R.; Shaw, S. Beyond Adoption: A New Framework for Theorizing and Evaluating Nonadoption, Abandonment, and Challenges to the Scale-Up, Spread, and Sustainability of Health and Care Technologies. J. Med. Internet Res. 2017, 19, e367. [Google Scholar] [CrossRef]
- Longhurst, C.A.; Davis, T.; Maneker, A.; Eschenroeder, H.C., Jr.; Dunscombe, R.; Reynolds, G.; Clay, B.; Moran, T.; Graham, D.B.; Dean, S.M.; et al. Local Investment in Training Drives Electronic Health Record User Satisfaction. Appl. Clin. Inform. 2019, 10, 331–335. [Google Scholar] [CrossRef] [PubMed]
| Author | Year | Location | Study Design | Sample Size | Clinical Outcome(s) |
|---|---|---|---|---|---|
| Maynard et al. [26] | 2010 | California, USA | Retrospective cohort | 748 | Venous thromboembolism |
| Novis et al. [27] | 2010 | Illinois, USA | Pre–post | 400 | Deep vein thrombosis |
| Fossum et al. [28] | 2011 | Norway | Quasi-experimental * | 971 | Pressure ulcers, malnutrition |
| Herasevich et al. [29] | 2011 | Minnesota, USA | Pre–post | 1159 | Ventilator-induced lung injury |
| Nelson et al. [30] | 2011 | Michigan, USA | Pre–post | 33,460 | Sepsis |
| Umscheid et al. [31] | 2012 | Pennsylvania, USA | Pre–post | 223,062 | Venous thromboembolism |
| Baillie et al. [32] | 2013 | Pennsylvania, USA | Pre–post | 120,396 | Readmission |
| Amarasingham et al. [33] | 2013 | Texas, USA | Pre–post | 1726 | Readmission |
| Litvin et al. [34] | 2013 | South Carolina, USA | Prospective cohort | 38,983 | Chronic kidney disease |
| Oh et al. [35] | 2014 | South Korea | Pre–post | 1111 | Delirium |
| Resetar et al. [36] | 2014 | Missouri, USA | Prospective cohort | 3691 | Ventilator-associated events |
| Amland et al. [37] | 2015 | Missouri, USA | Pre–post | 45,046 | Venous thromboembolism |
| Faerber et al. [38] | 2015 | New Hampshire, USA | Pre–post | 297 | Mortality |
| Hao et al. [39] | 2015 | Maine, USA | Prospective cohort | 118,951 | Readmission |
| Kharbanda et al. [40] | 2015 | Minnesota, USA | Prospective cohort | 735 | Hypertension |
| Lustig et al. [41] | 2015 | Canada | Prospective cohort | 580 | Venous thromboembolism |
| Umscheid et al. [42] | 2015 | Pennsylvania, USA | Pre–post | 15,526 | Sepsis, deterioration |
| Depinet et al. [43] | 2016 | Ohio, USA | Pre–post | 1886 | Appendicitis |
| Narayanan et al. [44] | 2016 | California, USA | Pre–post | 103 | Sepsis |
| Vinson et al. [45] | 2016 | California, USA | Pre–post | 893 | Pulmonary embolism |
| Aakre et al. [46] | 2017 | Minnesota and Florida, USA | Prospective cohort | 242 | Sepsis |
| Arts et al. [47] | 2017 | Netherlands | Randomized controlled trial | 781 | Stroke |
| Bookman et al. [48] | 2017 | Colorado, USA | Pre–post | 120 | Use of imaging |
| Jin et al. [49] | 2017 | South Korea | Case-control | 1231 | Pressure injury |
| Samal et al. [50] | 2017 | Massachusetts, USA | Prospective cohort | 569,533 | Kidney failure |
| Shimabukuro et al. [51] | 2017 | California, USA | Case-control | 67 | Sepsis |
| Chaturvedi et al. [52] | 2018 | Florida, USA | Prospective cohort | 309 | Anticoagulant therapy |
| Cherkin et al. [53] | 2018 | Washington, USA | Randomized controlled trial | 4709 | Physical function and pain |
| Ebinger et al. [54] | 2018 | Minnesota, USA | Prospective cohort | 549 | Complications, mortality, length of stay, and cost |
| Hebert et al. [55] | 2018 | Ohio, USA | Prospective cohort | 129 | Ventilator-associated events |
| Jung et al. [56] | 2018 | Ohio, USA | Pre–post | 232 | Sepsis, mortality |
| Kang et al. [57] | 2018 | South Korea | Case-control | 8621 | Medical errors |
| Karlsson et al. [58] | 2018 | Sweden | Randomized controlled trial | 444,347 | Anticoagulant therapy |
| Moon et al. [59] | 2018 | South Korea | Retrospective cohort | 4303 | Delirium |
| Ridgway et al. [60] | 2018 | Illinois, USA | Prospective cohort | 180 | HIV |
| Turrentine et al. [61] | 2018 | Virginia, USA | Pre–post | 1864 | Venous thromboembolism |
| Villa et al. [62] | 2018 | California, USA | Pre–post | 33,032 | Triage time |
| Vinson et al. [63] | 2018 | California, USA | Pre–post | 881 | Pulmonary embolism |
| Bedoya et al. [64] | 2019 | North Carolina, USA | Retrospective cohort | 85,322 | Deterioration |
| Brennan et al. [65] | 2019 | Florida, USA | Quasi-experimental * | 20 | Preoperative risk assessment |
| Ekstrom et al. [66] | 2019 | California and Upper Midwest, USA | Prospective cohort | Not stated | Appendicitis |
| Giannini et al. [67] | 2019 | Pennsylvania, USA | Randomized controlled trial | 54,464 | Sepsis |
| Khoong et al. [68] | 2019 | California, USA | Randomized controlled trial | 524 | Chronic kidney disease |
| Ogunwole et al. [69] | 2019 | Texas, USA | Pre–post | 204 | Readmission, Heart failure |
| Author | Interruptive vs. Non-Interruptive | Description of Risk Score Presentation | Quotation Regarding Alert Fatigue |
|---|---|---|---|
| Arts et al. [47] | Non-Interruptive | Floating notification window | “Too many alerts will tend to result in all alerts being ignored, a phenomenon known as ‘alert fatigue.’ Given the possible adverse effects of ‘alert fatigue’ and interruption, we considered the optimal interface to be one which minimized these effects.” |
| Bedoya et al. [64] | Interruptive | Best practice advisory (BPA) triggered requiring response from care nurse | “The majority of BPAs were ignored by care nurses. Furthermore, because nurses were ignoring the BPA, the logic in the background would cause the BPA to repeatedly fire on the same patient. This in turn created a large quantity of alerts that required no intervention by clinicians and led to alert fatigue in frontline nursing staff. Anecdotal feedback from nurses confirmed the constant burden of alerts repeatedly firing on individual patients. Furthermore, alert fatigue begets more alert fatigue and the downstream consequences of alert fatigue can include missed alerts, delay in treatment or diagnosis, or impaired decision-making when responding to future alerts.” |
| Depinet et al. [43] | Interruptive | Alert, data collection screen and feedback interface | “The firing of the CDS tool each time there was a chief complaint related to appendicitis may have led to alert fatigue. Overall, more work is needed to introduce a culture of standardized care in which such a decision support tool could work optimally.” |
| Herasevich et al. [29] | Interruptive | Bedside alert via text paging | “Because the majority of patients are treated with appropriate ventilator settings, unnecessary interruptions with new alert paradigms could have a detrimental effect on performance. It is therefore critical to incorporate contextual stop rules within decision support systems to prevent false positive alerts. Interruptions are often seen as distracting or sometimes devastating elements that need to be minimized or eliminated.” |
| Jin et al. [49] | Non-Interruptive | Display on nursing record screen | “Most computerized risk assessment tools require that nurses measure each score for each item in the scale. Thus, risk assessment scores are obtained only if all item scores are entered into the EHR system. Hence, as reported in a previous study, nurses have experienced work overload and fatigue and expressed their preference to use the paper charts. In addition, nurses felt a lot of time pressure.” |
| Kharbanda et al. [40] | Interruptive | Alert and dashboard display | “Four of eight (50 percent) rooming staff respondents reported that alerts to remeasure a BP [blood pressure] ‘sometimes’ interfered with their workflow, and the remaining responded that the alerts ‘rarely interfered.’” |
| Oh et al. [35] | Non-Interruptive | Pop-up window displayed on primary electronic medical record screen | “Most of the nurses did not recognize the urgent need for delirium care and did not consider it part of their regular routine. Therefore, nurses considered the additional care indicated by the system as extra work.” |
| Shimabukuro et al. [51] | Interruptive | Alert via phone call to charge nurse | “Systems that use these scores deliver many false alarms, which could impact a clinician’s willingness to use the sepsis classification tool.” |
| Custom Model (n = 19) | “Off-the-Shelf” Model (n = 13) | |
|---|---|---|
| Improved clinical outcomes | 16 (84.2%) | 6 (46.2%) |
| No improvements in outcomes | 3 (15.8%) | 7 (53.8%) |
| Physicians as Primary Intended Users (n = 22) | Nurses as Primary Intended Users (n = 8) | Other Intended Users 1 (n = 2) | |
|---|---|---|---|
| Improved clinical outcomes | 15 (68.2%) | 5 (62.5%) | 2 (100%) |
| No improvements in outcomes | 7 (31.8%) | 3 (37.5%) | 0 (0%) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, T.C.; Shah, N.U.; Haack, A.; Baxter, S.L. Clinical Implementation of Predictive Models Embedded within Electronic Health Record Systems: A Systematic Review. Informatics 2020, 7, 25. https://doi.org/10.3390/informatics7030025
Lee TC, Shah NU, Haack A, Baxter SL. Clinical Implementation of Predictive Models Embedded within Electronic Health Record Systems: A Systematic Review. Informatics. 2020; 7(3):25. https://doi.org/10.3390/informatics7030025
Chicago/Turabian StyleLee, Terrence C., Neil U. Shah, Alyssa Haack, and Sally L. Baxter. 2020. "Clinical Implementation of Predictive Models Embedded within Electronic Health Record Systems: A Systematic Review" Informatics 7, no. 3: 25. https://doi.org/10.3390/informatics7030025
APA StyleLee, T. C., Shah, N. U., Haack, A., & Baxter, S. L. (2020). Clinical Implementation of Predictive Models Embedded within Electronic Health Record Systems: A Systematic Review. Informatics, 7(3), 25. https://doi.org/10.3390/informatics7030025







