Studying Plant–Insect Interactions with Solid Phase Microextraction: Screening for Airborne Volatile Emissions Response of Soybeans to the Soybean Aphid, Aphis glycines Matsumura (Hemiptera: Aphididae)
Abstract
:1. Introduction
| Ref. | Sampling | Sample Preparation | Analyses | Matrix and # identified compounds |
|---|---|---|---|---|
| This work | SPME (85 µm Carboxen / PDMS); Extraction conditions: 25 °C, 24 h | SPME | GC-MS | Soybean leaves (A. glycines-infested and non-infested), 62 compounds |
| Zhu and Park [11] | Glass tube containing 100 mg Super Q (80/100 mesh) | Solvent extraction. Direct injection | GC-EAD (electro-antenno-graphic detection) GC-MS | Soybean leaves (A. glycines infested and non-infested), 25 compounds |
| Boue et al. [10] | SPME (100 µm PDMS, 50/30 µm DVB/Carboxen/PDMS); Extraction conditions: 60 °C, 1 h | SPME | GC-MS | Soybeans, 49 compounds |
| Rosario et al. [12] | Dynamic headspace sampling; Tenax-GC adsorbent | Thermal desorption | GC-MS | Winged beans and soybeans, 116 compounds |
| Damiani et al. [13] | 1. Tenax-GC adsorbent, 2. Purge and trap, 3. SPME 100 µm PDMS; Extraction @ 45 °C, 20 min | 1.Thermal desorption 2.SPME | GC-MS GC-FTIR | Soybean leaves, 9 compounds |
2. Experimental Section
2.1. Aphis glycines Colony
2.2. Volatiles from Soybeans with and without A. glycines: Vegetative-Stage, Greenhouse Grown Soybean
2.3. Reproductive-Stage, Field Grown Soybean
2.4. Volatile Emissions Screening with SPME and GC-MS
2.5. Chemical Analysis
2.6. Statistical Analyses
3. Results and Discussion
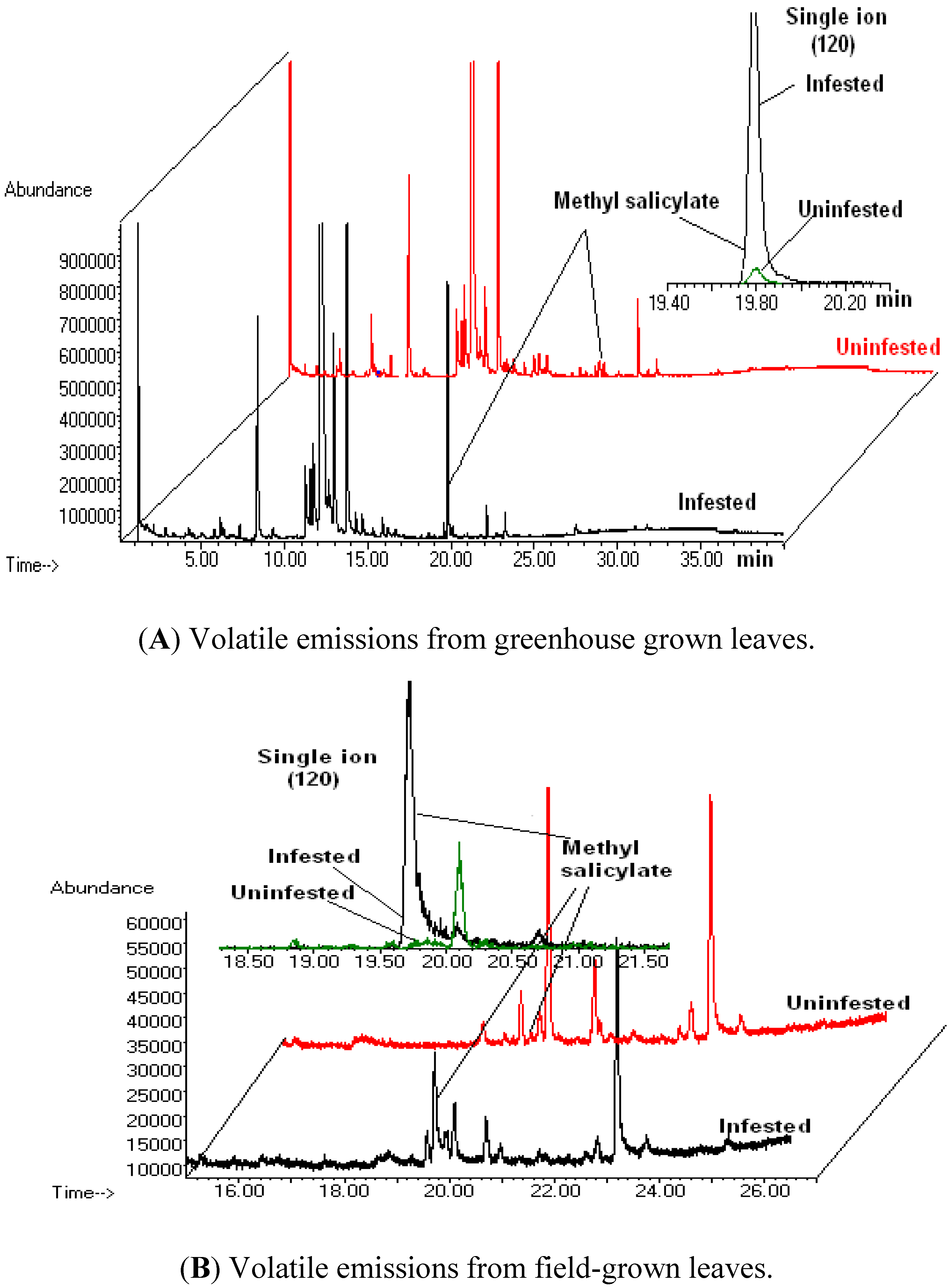
3.1. Volatile Emissions from Vegetative-Stage, Greenhouse Grown Soybean
| GC column RT (min) | Compound | CAS | MS spectral match (%) | Boue et al. [10] beans | Rosario et al. [12] beans | Damiani et al. [13] - beans | Zhu and Park [11] leaves | |
|---|---|---|---|---|---|---|---|---|
| 1 | 2.06 | Acetic acid, methyl ester | 79-20-9 | 35 | y | y | ||
| 2 | 3.25 | Butanal, 2-methyl- | 96-17-3 | 74 | y | |||
| 3 | 3.96 | 2,4-Hexadienal | 142-83-6 | 63 | y | |||
| 4 | 4.16 | Propanenitrile, 2-methyl- | 78-82-0 | 83 | ||||
| 5 | 4.36 | Trimethylsilanol | 1066-40-6 | 69 | ||||
| 6 | 4.81 | 1-Penten-3-one | 1629-58-9 | 38 | y | |||
| 7 | 5.45 | Butanoic acid, 2-methyl-, methyl ester | 868-57-5 | 88 | ||||
| 8 | 5.70 | Propane, 1-chloro-2-methyl- | 513-36-0 | 28 | ||||
| 9 | 6.01 | Butanedinitrile, 2,3-dimethyl- | 16411-13-5 | 82 | ||||
| 10 | 6.23 | 2-Propanol, 2-methyl- | 75-65-0 | 35 | y | |||
| 11 | 6.65 | Hexanal * | 66-25-1 | 88 | y | y | y | |
| 12 | 7.05 | 3,4-Dihydropyran | 110-87-2 | 39 | ||||
| 13 | 7.23 | 1-Penten-3-ol | 616-25-1 | 79 | y | |||
| 14 | 8.13 | 1-Butanol-, acetate | 123-92-2 | 85 | y | |||
| 15 | 8.26 | 1-Butanol, 3-methyl- * | 123-51-3 | 76 | y | |||
| 16 | 9.21 | cis-3-Hexenal | 6789-80-6 | 68 | ||||
| 17 | 9.00 | 1-Butanol, 2-methyl- | 137-32-6 | 31 | y | |||
| 18 | 9.00 | Cyclobutanone, 2, 2-dimethyl- | 1192-14-9 | 68 | ||||
| 19 | 10.58 | trans-2-Methyl-1, 3-pentadiene | n/a | 79 | ||||
| 20 | 11.16 | 3-Octanone * | 106-68-3 | 93 | y | y | ||
| 21 | 11.48 | cycloproane, propyl- | 2415-72-7 | 74 | ||||
| 22 | 11.65 | 3-Hexen-1-ol | 928-97-2 | 69 | y | y | y | |
| 23 | 11.98 | cis-3-Hexen-1-ol | 928-96-1 | 95 | y | |||
| 24 | 12.15 | cis-3-Hexen-1-ol, acetate | 3681-71-8 | 76 | y | y | ||
| 25 | 12.58 | 1,3-Pentadiene, 2-methyl- | 926-54-5 | 83 | ||||
| 26 | 12.88 | 3-Octanol * | 589-98-0 | 83 | y | y | ||
| 27 | 13.30 | Acetic acid * | 64-19-7 | 74 | y | |||
| 28 | 13.61 | 1-Octen-3-ol | 3391-86-4 | 83 | y | y | y | |
| 29 | 14.21 | Oct-1-en-3-one | 50306-18-8 | 72 | ||||
| 30 | 14.58 | Cyclopropene, 3,3-diethyl- | 78578-86-6 | 33 | ||||
| 31 | 14.95 | Benzaldehyde* | 100-52-7 | 93 | y | y | y | |
| 32 | 15.23 | gamma, Heptalactone | 105-21-5 | 24 | ||||
| 33 | 15.80 | Linalool | 78-70-6 | 31 | y | |||
| 34 | 16.13 | 2 [5H]-Furanone, 5-ethyl- | 2407-43-4 | 50 | ||||
| 35 | 16.41 | Benzene, isocyano- | 931-54-4 | 68 | ||||
| 36 | 16.40 | Malonic acid bis-( 2-trimethylsilanyl-ethyl ester) | 90744-45-9 | 31 | ||||
| 37 | 16.80 | Thiophene, 2-pentyl- | 4861-58-9 | 24 | ||||
| 38 | 17.08 | Benzoic, methyl ester | 93-58-3 | 75 | ||||
| 39 | 18.13 | gamma, Hexalactone | 695-06-7 | 88 | ||||
| 40 | 18.6 | 2-Cyclohexene-1,4-dione | 4505-38-8 | 63 | ||||
| 41 | 18.81 | alpha -Copaene | 3856-25-5 | 89 | ||||
| 42 | 18.96 | 2 [5H]-Furanone, 5-ethyl-, [R] | 76291-90-2 | 72 | ||||
| 43 | 19.26 | E-2-Methyl-2-butenoate | 67883-79-8 | 72 | ||||
| 44 | 19.51 | beta-Elemene * | 515-13-9 | 98 | ||||
| 45 | 19.71 | Methyl salicylate * | 119-36-8 | 95 | y | |||
| 46 | 20.03 | trans-Caryophyllene * | 87-44-5 | 93 | ||||
| 47 | 20.21 | 2-Clohexene-1, 4-dione | 4505-38-8 | 72 | ||||
| 48 | 20.73 | Benzenemethanol | 100-51-6 | 95 | ||||
| 49 | 20.93 | cis-Ocimene | 3338-55-4 | 61 | ||||
| 50 | 21.36 | Benzene ethanol * | 60-12-8 | 85 | y | |||
| 51 | 21.80 | alpha, Farnesene | 502-61-4 | 94 | y | |||
| 52 | 22.06 | Methyl ester of 2-oxo-hexanoic acid | 6395-83-1 | 18 | ||||
| 53 | 22.68 | Phenol * | 108-95-2 | 91 | ||||
| 54 | 22.85 | 2-Methyl-3-phenythiopropene | 702-00-1 | 24 | ||||
| 55 | 22.96 | (E)-4,8-Dimethyl-1,3,7-nonatriene | 54 | |||||
| 56 | 23.76 | Ionol | 4130-42-1 | 76 | ||||
| 57 | 24.01 | Methyl anisate | 606-45-1 | 93 | ||||
| 58 | 27.4 | 1-Dodecanamine, n,n-dimethyl-, | 112-18-5 | 72 | ||||
| 59 | 28.25 | 4-Morpholineethanamine | 2038-03-1 | 63 | ||||
| 60 | 28.83 | Indole * | 120-72-9 | 90 | ||||
| 61 | 30.98 | 5-Ethyl-1, 3-dihydro-1, 3-diiminoisoindole | 134940-70-8 | 69 | ||||
| 62 | 31.66 | Morpholine, 4-octadecyl- | 16528-77-1 | 72 |
3.2. Volatile Emissions from Reproductive-Stage, Field Grown Soybean
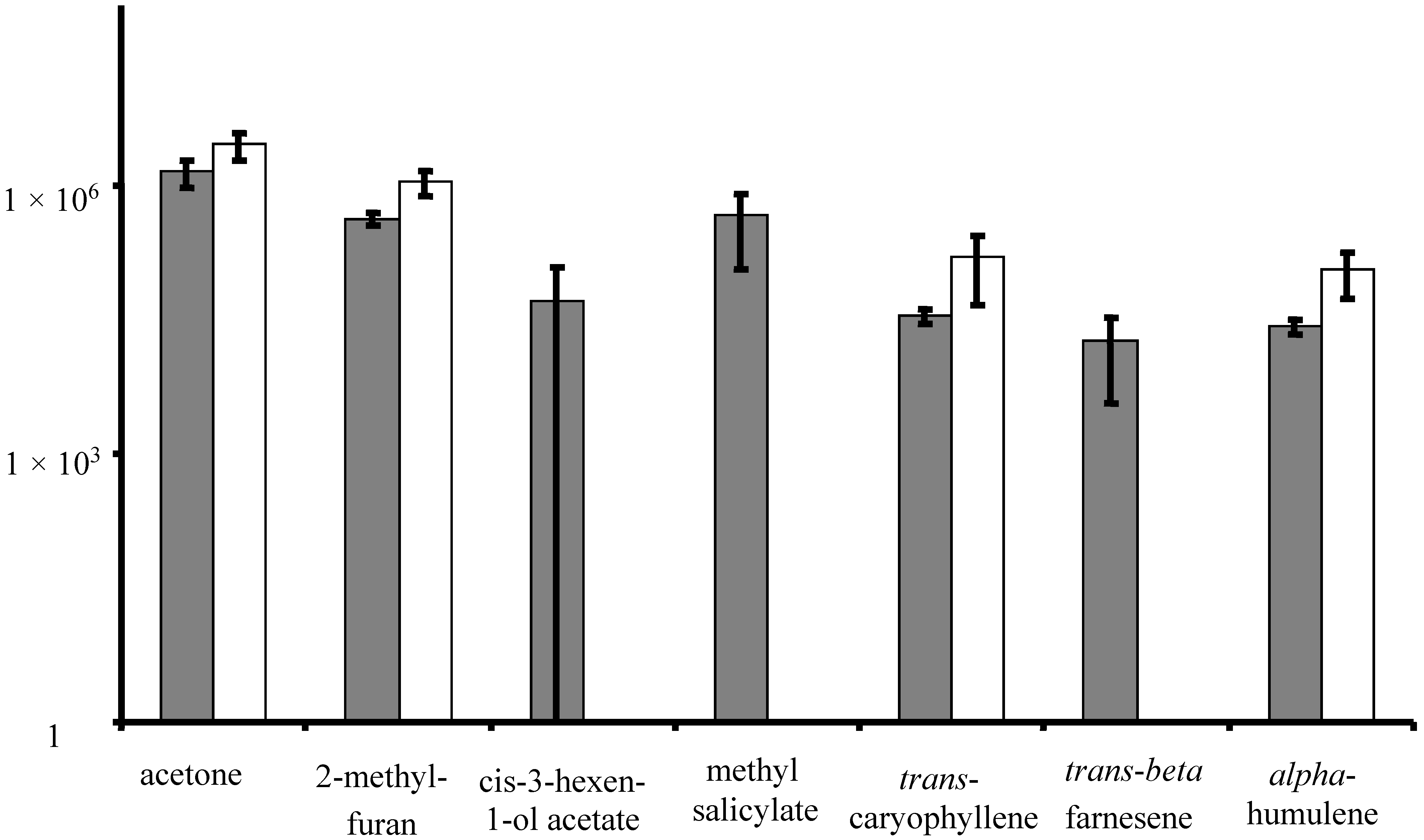
3.3. Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cardoza, Y.J.; Alborn, H.T.; Tumlinson, J.H. In vivo volatile emissions from peanut plants induced by simultaneous fungal infection and insect damage. J. Chem. Ecol. 2002, 28, 161–174. [Google Scholar] [CrossRef] [PubMed]
- deBoer, J.G.; Posthumus, M.A.; Dicke, M. Identification of volatiles that are used in discrimination between plants infested with prey or nonprey herbivores by a predatory mite. J. Chem. Ecol. 2004, 30, 2215–2230. [Google Scholar] [CrossRef] [PubMed]
- De Moraes, C.M.; Lewis, W.J.; Pare, P.W.; Alborn, H.T.; Tumlinson, J.H. Herbivore-infested plants selectively attract parasitoids. Nature 1998, 393, 570–573. [Google Scholar] [CrossRef]
- Van Den Boom, C.E.; Van Beek, T.A.; Posthumus, M.A.; De Groot, A.; Dicke, M. Qualitative and quantitative variation among volatile profiles induced by Tetranychus urticae feeding on plants from various families. J. Chem. Ecol. 2004, 30, 69–89. [Google Scholar] [CrossRef] [PubMed]
- Fox, T.B.; Landis, D.A.; Cardoso, F.F.; DiFonzo, C. Predators suppress Aphis glycines Matsumura population growth in soybean. Environ. Entomol. 2004, 33, 608–618. [Google Scholar] [CrossRef]
- Ragsdale, D.W.; Landis, D.A.; Brodeur, J.; Heimpel, G.E.; Desneux, N. Ecology and management of the soybean aphid in North America. Ann. Rev. Entomol. 2011, 56, 375–399. [Google Scholar] [CrossRef]
- Fox, T.B.; Landis, D.A.; Cardoso, F.F.; DiFonzo, C. Predators suppress Aphis glycines Matsumura population growth in soybean. Environ. Entomol. 2004, 33, 608–618. [Google Scholar] [CrossRef]
- Mallinger, R.E.; Hogg, D.B.; Gratton, C. Methyl salicylate attracts natural enemies and reduces populations of soybean aphids (Hemiptera: Aphididae) in soybean agroecosystems. J. Econ. Entomol. 2011, 104, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Mumm, R.; Dicke, M. Variation in natural plant products and the attraction of bodyguards involved in indirect plant defense. Can. J. Zool. 2010, 88, 628–667. [Google Scholar] [CrossRef]
- Boue, S.M.; Shih, B.Y.; Carter-Wientjes, C.H.; Cleveland, T.E. Identification of volatile compounds in soybean at various developmental stages using solid phase microextraction. J. Agric. Food Chem. 2003, 51, 4873–4876. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.W.; Park, K.C. Methyl salicylate, a soybean aphid-induced plant volatile attractive to the predator Coccinella septempunctata. J. Chem. Ecol. 2005, 31, 1733–1746. [Google Scholar] [CrossRef] [PubMed]
- del Rosario, R.; de Lumen, B.O.; Habu, T.; Flath, R.A.; Richard, M.T.; Teranishi, R. Comparison of headspace volatiles from winged beans and soybeans. J. Agric. Food Chem. 1984, 32, 1011–1015. [Google Scholar] [CrossRef]
- Damiani, P.; Cossignani, L.; Castellini, M.; Bin, F. Clean recovery and HRGC-MS/HRGC-FTIR identification of volatiles from soybean (Glycine max). Ital. J. Food Sci. 2000, 2, 175–182. [Google Scholar]
- Augusto, F.; Lopes, A.L.E.; Zini, C.A. Sampling and sample preparation for analysis of aromas and fragrances. TrAC Trends Anal. Chem. 2003, 22, 160–169. [Google Scholar] [CrossRef]
- Rasmann, S.; Kollner, T.G.; Degenhardt, J.; Hiltpold, I.; Toepfer, S.; Kuhlmann, U.; Gershenzon, J.; Turlings, T.C.J. Recruitment of entomopathogenic nematodes by insect-damaged maize roots. Nature 2005, 434, 732–737. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cai, L.; Koziel, J.A.; O'Neal, M.E. Studying Plant–Insect Interactions with Solid Phase Microextraction: Screening for Airborne Volatile Emissions Response of Soybeans to the Soybean Aphid, Aphis glycines Matsumura (Hemiptera: Aphididae). Chromatography 2015, 2, 265-276. https://doi.org/10.3390/chromatography2020265
Cai L, Koziel JA, O'Neal ME. Studying Plant–Insect Interactions with Solid Phase Microextraction: Screening for Airborne Volatile Emissions Response of Soybeans to the Soybean Aphid, Aphis glycines Matsumura (Hemiptera: Aphididae). Chromatography. 2015; 2(2):265-276. https://doi.org/10.3390/chromatography2020265
Chicago/Turabian StyleCai, Lingshuang, Jacek A. Koziel, and Matthew E. O'Neal. 2015. "Studying Plant–Insect Interactions with Solid Phase Microextraction: Screening for Airborne Volatile Emissions Response of Soybeans to the Soybean Aphid, Aphis glycines Matsumura (Hemiptera: Aphididae)" Chromatography 2, no. 2: 265-276. https://doi.org/10.3390/chromatography2020265
APA StyleCai, L., Koziel, J. A., & O'Neal, M. E. (2015). Studying Plant–Insect Interactions with Solid Phase Microextraction: Screening for Airborne Volatile Emissions Response of Soybeans to the Soybean Aphid, Aphis glycines Matsumura (Hemiptera: Aphididae). Chromatography, 2(2), 265-276. https://doi.org/10.3390/chromatography2020265







