Post-Polymerization Modifications of Polymeric Monolithic Columns: A Review
Abstract
:1. Introduction
2. Photo-Grafting Reactions
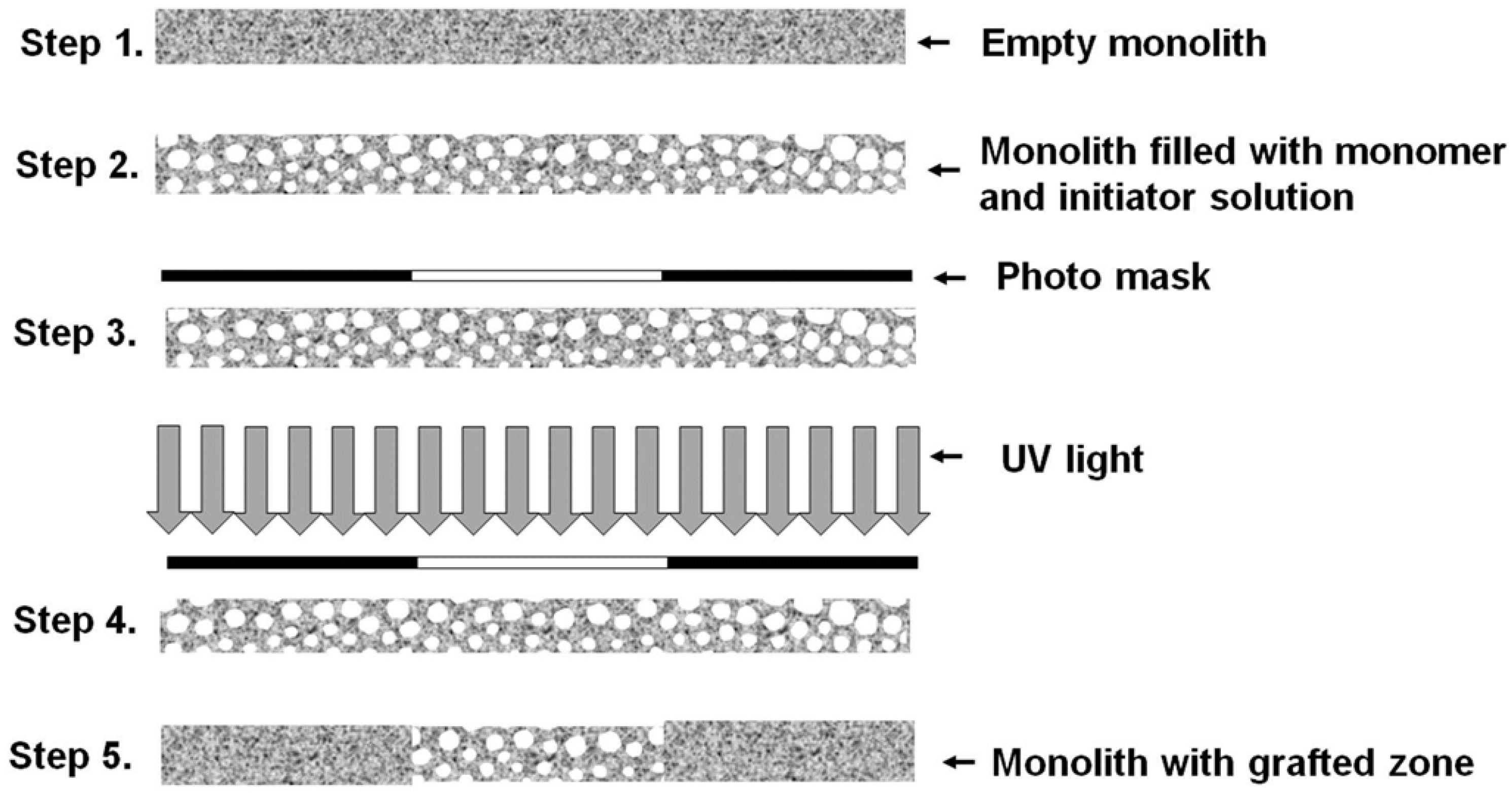

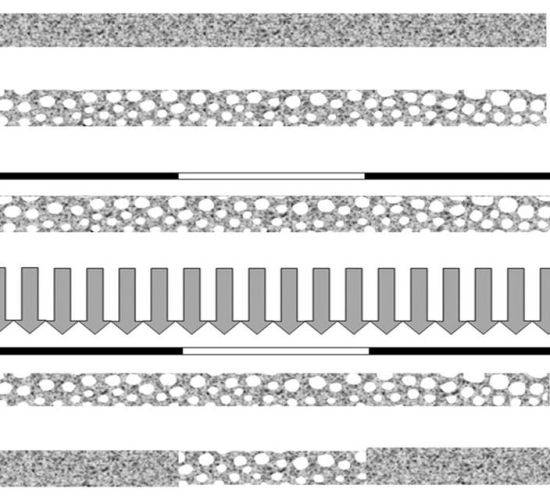
2.1. Monolithic Columns Grafted with Ionizable Exchange Groups
2.2. Grafted Monolithic Columns for Biomolecule Immobilization
2.3. Monolithic Columns with Bonded Ligands
| Monolith | Reaction | Graft energy | Time | Reference |
|---|---|---|---|---|
| Poly(BuMA-co-EDMA) | Single and sequential two step grafting of monomers. | 500 W | 60 * min | [7] |
| Poly(BuMA-co-EDMA) | Sequential grafting of PEGMA and VAL. | 500 W | 60 * min | [8] |
| Poly(BuMA-co-EDMA) | Single step grafting of gradients of AMPS. | 500 W | 1 to 10 min | [10] |
| Poly(BuMA-co-EDMA) | Single step grafting of gradients of AMPS. | 3 J/cm2 | N/A | [11] |
| Poly(BuMA-co-EDMA) | Sequential grafting of PEGMA, VAL, and SPM. | 1 J/cm2 each step | N/A | [13] |
| Poly(BuMA-co-EDMA) | Single step grafting of AMPS. | 0.25 to 7 J/cm2 | N/A | [29] |
| Poly(BuMA-co-EDMA) | Single step grafting of AMPS and butyl acrylate. | 500 W | 0.5 to 1.5 min | [36] |
| Poly(BuMA-co-EDMA) | Single step grafting of AMPS and VAL. | 500 W | 30 min | [37] |
| Poly(BuMA-co-EDMA) | Single step grafting of META and butyl acrylate. | 1.4 mW/cm2 | 3 min | [67] |
| Poly(BuMA-co-EDMA) | Single step grafting of VAL for dual function monolith. | 15 mW/cm2 | 3 min | [68] |
| Poly(GMA-co-EDMA) | Single step grafting of META. | 0.25 J/cm2 | N/A | [69] |
| Poly(BuMA-co-EDMA) | Single step grafting of AMPS and VAL. | N/A | 60, 30 min respectively | [70] |
| Poly(GMA-co-EDMA) | Sequential grafting of PEGMA and VAL. | 12 mW/cm2 | 4 * 1 to 6 min (VAL) | [71] |
| Poly(BuMA-co-EDMA) and Poly(GMA-co-EDMA) | Sequential grafting of PEGMA and VAL. | N/A | 4 *, 2 min | [72] |
| Poly(BuMA-co-EDMA) | Sequential grafting of PEGMA and VAL. | 3 * J/cm2 | N/A | [74] |
| Poly(BuMA-co-EDMA) | Single step grafting of VAL. | 3 J/cm2 | N/A | [75] |
| Poly(BuMA-co-EDMA) | Single step grafting of spiropyran monomer using VIS LED at 660 nm. | 0.5 cd | 2 min | [77] |
| Poly(LMA-co-EDMA) | Multi step grafting of VAL. | 1 * J/cm2 | N/A | [78] |
| Poly(LMA-co-EDMA) | Multi step grafting of GMA. | 1 * J/cm2 | N/A | [79] |
2.4. Monolithic Columns Grafted with Heterogeneous Grafting Energies
3. “Click” Chemistry
| Monolith | Reaction | λ (nm) | Temperature (C°) | Time | Reference |
|---|---|---|---|---|---|
| Poly(NAS-co-EDMA) | Propargylamine modification to produce surface alkyne. Click grafting of 1-adamantanethiol using UV irradiation at 313 nm, using darocure as initiator. | 313 | N/A | 30, 60, 120, and 180 min | [43] |
| Poly(GMA-co-EDMA), Poly(GMA-co-MMA-co-EDMA), Poly(GMA-co-HEMA-co-EDMA) | Surface sulfhydryl groups clicked with (S)-N-(4-allyloxy-3,5-dichlorobenzoyl)-2-amino-3,3-dimethylbutanephosphonic acid in the presence of AIBN initiator. | N/A | 60 | 24 h | [50] |
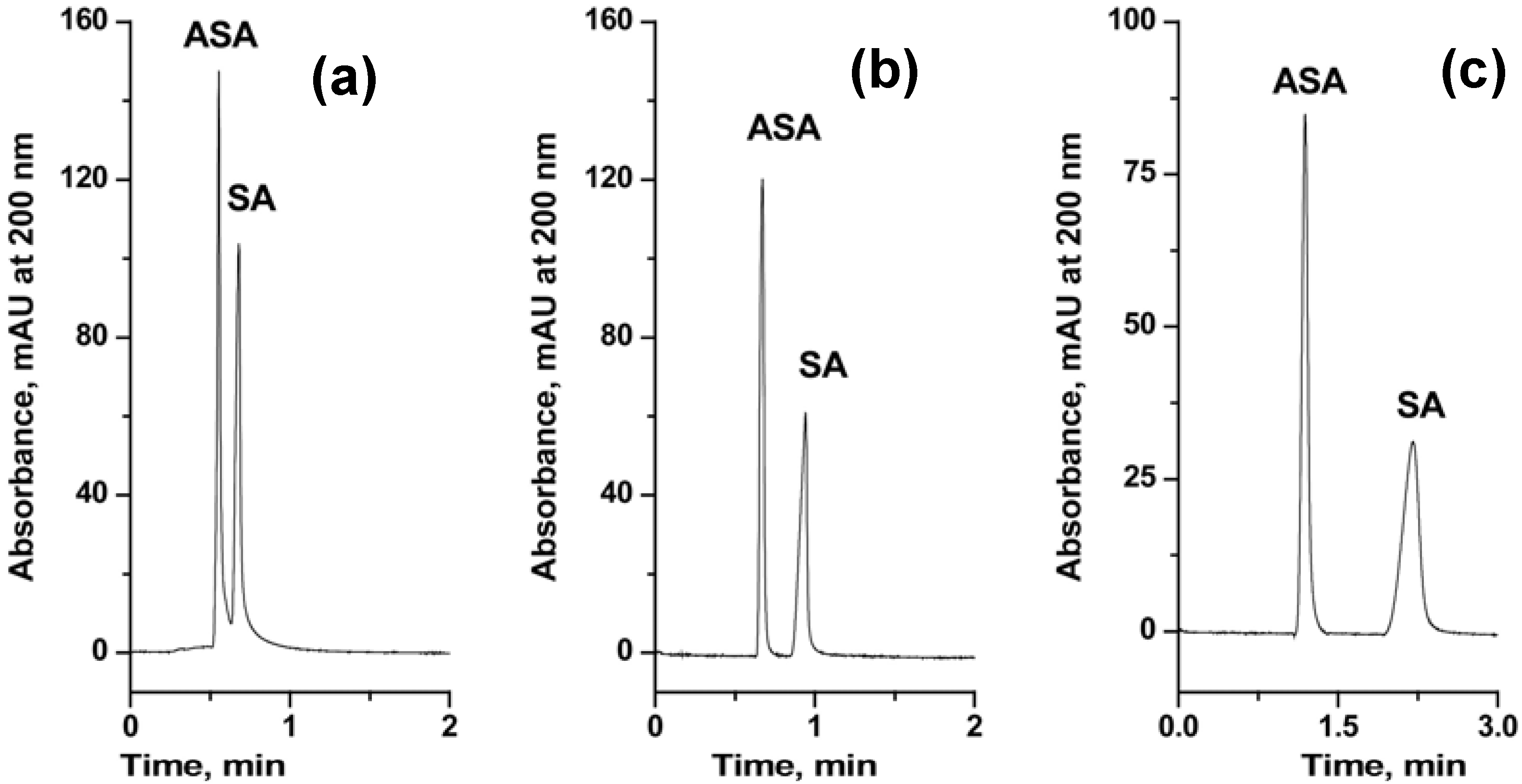
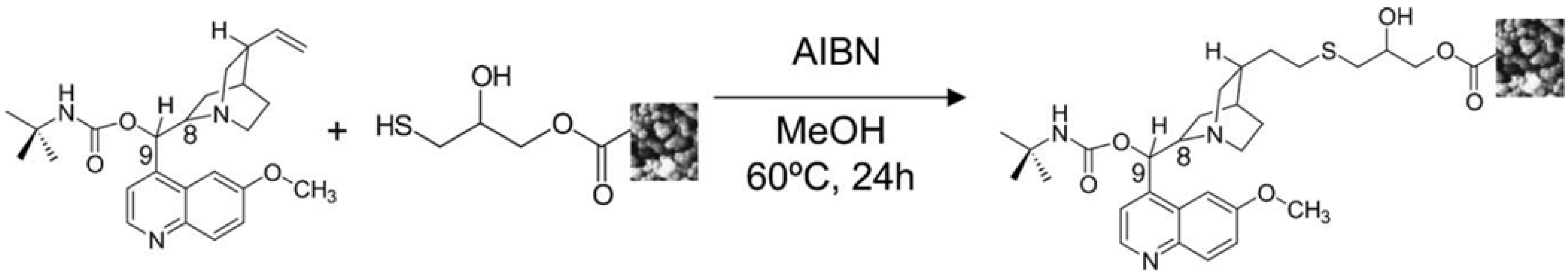
| Poly(GMA-co-EDMA) | Surface thiol groups clicked with O-9-tert-butylcarbamoylquinine in the presence of AIBN. | N/A | 60 | 24 h | [87] |
| Poly(IEM-co-MMA-co-EGDMA) | Monoliths modified with 1-octanol, 1-decanol, 1-dodecanol, 1-octadecanol, n-decylamine and 1-decanethiol. | N/A | 60 | N/A | [88] |
| Poly(GMA-co-EDMA) | Monolith thiolated with cysteamine, with cleavage of the disulphide thus exposing thiols. MEDSA or LMA clicked using either heat or UV initiation. | 360 | 80 | N/A | [89] |
| Poly(NAS-co-EDMA) | Allylamine modified monolith surface reacted with 1-octadecanethiol in the presence of AIBN and UV initiation. | 365 | N/A | 4 h | [90] |
| Poly(propargyl methacrylate-co-EDMA) | Click addition of 1-azidooctane and 1-azidooctadecane using a Cu(I) catalyst. | N/A | 30 | 120 h | [91] |
| Poly(GMA-co-EDMA) and poly(VBC-co-DVB) | Azide modified surfaces clicked with 1-decyne using a Cu(I) catalyst. | N/A | 30 to 60 | 48 h | [92] |
| Poly(3-(Trimethoxysilyl)propyl acrylate-co-propargylacrylate-co-AMPS-co-TRIM-co-PETRA) | Active surface modified with cinnamidyl azide or 6-azido-6-deoxy-beta-cyclodextrin. | N/A | 84 | 16 h | [93] |
3.1. Thiol-Ene Click Reactions in Monolithic Column Functionalization
3.2. Thiol-Yne Click Reactions in Monolithic Column Functionalization
4. Thermally Initiated Graft Chain Growth
5. Hypercross-Linking
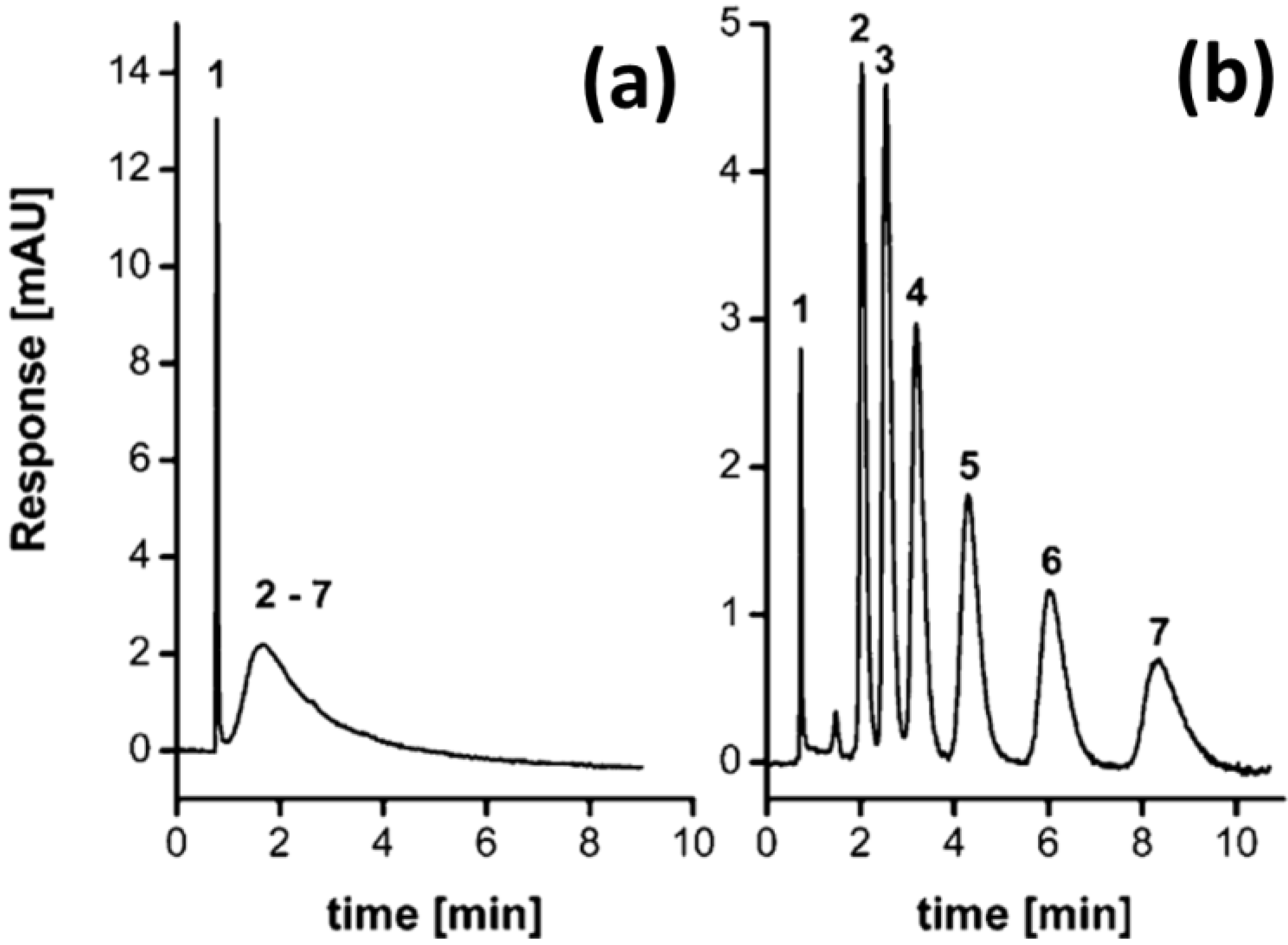
| Monolith | Reaction | Temperature (°C) | Time (h) | Reference |
|---|---|---|---|---|
| Poly(styrene-co-VBC-co-DVB) | Solution of FeCl3 in DCE flushed across column (held in ice for 2 h). | 80 | 24 | [112] |
| Poly(styrene-co-VBC-co-DVB) | Solution of FeCl3 in DCE flushed across column (held in ice for 1 h). | 80 | 24 | [114] |
| Poly(styrene-co-VBC-co-DVB) and poly(4-methyl styrene -co-VBC-co-DVB) | Solution of FeCl3 in DCE flushed across column (held in ice for 2 h). | 90 | 2 | [115] |
| Poly(styrene-co-VBC-co-DVB) | Solution of FeCl3 in DCE flushed across column. | 90 | 2 | [116] |
| Poly(styrene-co-VBC-co-DVB) | Solution of FeCl3 in DCE flushed across column. Thermal grafting of MEDSA using 4,4’-azobis(4-cyanovaleric acid) (8 h at 70 °C). | 90 | 2 | [117] |
| Poly(4-methyl styrene -co-VBC-co-DVB) | Hypercross-linked monolith brominated to support cysteamine modification (microwave assisted, 30 min). Disulphide bonds cleaved. Pendent thiols modified with AuNPs. | 90 | 4 | [118] |
6. Nano-Particles and Nano-Structures
| Monolith | Reaction | Temperature (C°) | Time | Nano-Particle Type | Reference |
|---|---|---|---|---|---|
| Poly(BuMA-co-EDMA) and poly(LMA-co-EDMA) | Introduction of photo-grafted zones of monomers suitable for amination, supporting AuNP immobilization. Dual function monolith. | Room Temperature (RT) | N/A | 20 nm Au | [9] |
| Poly(GMA-co-EDMA) and poly(styrene-co-DVB) | For poly(STY-co-DVB) chlorosulfonic acid in dry dichloromethane was used for sulfonation. For poly(GMA-co-EDMA), sulfonation was performed in three techniques: (i) 4-hydroxybenezenesulfonic acid and triethylamine, (ii) thiobenzoic acid and triethylamine (thiol groups were oxidized by pumping a solution of tert-butylhydroperoxide). (iii) Sodium sulfite and tetra-n-butylammonium hydroxide. | 60, 60, 70, respectively | 20 h each | 65 nm Latex | [28] |
| Poly(BuMA-co-AMPS-co-EDMA) | Co-polymerization of sulfonic acid group suitable for latex nano-particle agglomeration. | RT | 2 h | 60 nm Latex | [46] |
| Poly(BuMA-co-AMPS-co-EDMA) | Co-polymerization of sulfonic acid group suitable for latex nano-particle agglomeration. | RT | 2 h | 65 nm Latex | [47] |
| Poly(BuMA-co-AMPS-co-EDMA) | Co-polymerization of sulfonic acid group suitable for latex nano-particle agglomeration. | RT | 2 h | 65 nm Latex | [48] |
| Poly(GMA-co-EDMA) | Introduction of surface immobilized AuNPs via cysteamine and subsequent cleavage of the disulfide bond, to reveal surface thiols. AuNP modification. | RT | N/A | 10 nm Au | [118] |
| Poly(GMA-co-EDMA) | Generation of quaternary ammonium for IONP immobilization. | RT | N/A | 19 nm iron oxide | [119] |
| Poly(EDMA) | Preparation of AuNP modified extraction pipette tip, for glycoprotein selectivity. | RT | N/A | 20 nm Au | [129] |
| Poly(GMA-co-EDMA) | Coupling of α-glucosidase to AuNP modified monolith for PMME. | RT | 3 h | 15 nm Au | [130] |
| Poly(GMA-co-EDMA) | For AuNP immobilization, and functionalizing groups such as 3-mercapto propionic acid (CEC), 1-octadecanethiol (RPLC), and sodium 2-mercaptoethane sulfonate (SCX). | RT | N/A | 15 nm Au | [131] |
| Poly(NAS-co-EDMA) | Surface generation of alkyne group, followed by thiol-yne photo-addition of cysteamine for AuNP modification. | RT | N/A | 20 nm Au | [132] |
| Poly(GMA-co-EDMA) | Introduction of surface AuNPs by generation of surface thiols from surface epoxide groups. Two methods were used: (i) Hydrogen sulfide, and (ii) Cysteamine. | 100RTRT | 30 min2 h20 min | 40–50 nm Au | [133] |
| Poly(BuMA-co-EDMA) | Preparation of thiolated and aminated surfaces and comparison of immobilization pathways for AuNPs. | RT | N/A | 20 nm Au | [134] |
| Poly(GMA-co-EDMA) | Immobilization of SNPs upon a polymer monolith. | RT | 60 min | 50 nm Ag | [127] |
| Poly(HEMA-co-EDMA) | Generation of polymer monolith extraction device within a pipette tip, immobilized with IONPs via surface grafted functional groups (META). | RT | N/A | 20 nm iron oxide | [135] |
| Poly(NIPAAm-co-GMA-co-EDMA) | Generation of γ-alumina nano-particle monolith for PMME. | 60 | 14 h | 10–20 nm Al2O3 | [136] |
| Poly(GMA-co-EDMA), poly(BuMA-co-EDMA), poly(GMA-co-BuMA-co-EDMA) | Immobilization of Pd/Pt NFs via aminated surface groups. | RT | 72 h | Pd/Pt NFs | [137] |
| Poly(GMA-co-EDMA) | Coating of monolith surface with graphene and graphene oxide nano-sheets for PMME. | RT | N/A | Graphene nano-sheets | [138] |
| Poly(GMA-co-EDMA) | Encapsulation of MWCNTs and surface attachment of MWCNTs via surface aminated groups. | RT | N/A | MWCNTs | [139] |
6.1. Polymeric Nano-Particles
6.2. Gold Nano-Particles

6.3. Other Metallic Nano-Particles
6.4. Carbon Nano-Structures
7. Concluding Remarks
List of Abbreviations:
| AMPS | 2-acrylamido-2-methyl-1-propanesulphonic acid |
| AIBN | α, α’-azoisobutyronitrile |
| ATP | adenosine triphosphate |
| ATRP | atom transfer radical polymerization |
| BPO | benzoyl peroxide |
| BSA | bovine serum albumin |
| BuMA | butyl methacrylate |
| CEC | capillary electrochromatography |
| CE | capillary electrophoresis |
| COC | cyclic olefin copolymer |
| DAP | 2,2-dimethoxy-2-phenylacetophenone |
| DCE | dichlorethane |
| DMF | dimethyl formamide |
| DVB | divinyl benzene |
| EDMA | ethyleneglycol dimethacrylate |
| EOF | electroosmostic flow |
| GFP | green fluorescent protein |
| GMA | glycidyl methacrylate |
| AuNP(s) | gold nano-particle(s) |
| HEMA | hydroxyethyl methacrylate |
| HILIC | hydrophilic interaction liquid chromatography |
| IDA | iminodiacetic acid |
| IEM | isocyanoethyl methacrylate |
| IONP(s) | iron oxide nano-particle(s) |
| LMA | lauryl methacrylate |
| MEDSA | N,N-dimethyl-N-methacryloxyethyl-N-(3-sulfopropyl) ammonium betaine |
| META | methacryloyloxyethyl trimethylammonium chloride |
| MMA | methyl methacrylate |
| MST | 4-methyl styrene |
| MWCNT(s) | multi-walled carbon nano-tube(s) |
| NAS | N-acryloxysuccinimide |
| NF(s) | nano-flower(s) |
| OEG | oligoethylene glycol |
| PEGMA | polyethyleneglycol methacrylate |
| PEI | polyethylimine |
| PMME | polymer monolith micro-extraction |
| PTFE | Teflon |
| RAFT | reversible addition-fragmentation chain transfer |
| RP | reversed-phase |
| ROMP | ring opening metathesis polymerization |
| sC4D | scanning capacitively coupled contactless conductivity detection |
| SEM | scanning electron microscope |
| SNP(s) | silver nano-particle(s) |
| SPE | solid phase extraction |
| SPM | sulphopropyl methacrylate |
| STY | styrene |
| TEMPO | 2,2,6,6-tetramethylpiperidyl-1-oxy |
| TRIM | trimethylolpropane trimethacrylate |
| VAL | vinyl azlactone |
| VBC | vinylbenzyl chloride |
Acknowledgments
Conflicts of Interest
References
- Hjertén, S.; Liao, J.; Zhang, R. High-performance liquid chromatography on continuous polymer beds. J. Chromatogr. 1989, 473, 273–275. [Google Scholar] [CrossRef]
- Svec, F.; Fréchet, J. Continuous rods of macroporous polymer as high-performance liquid chromatography separation media. Anal. Chem. 1992, 64, 820–822. [Google Scholar] [CrossRef]
- Svec, F.; Fréchet, J. Modified poly(glycidyl methacrylate-co-ethylene dimethacrylate) continuous rod columns for preparative-scale ion-exchange chromatography of proteins. J. Chromatogr. A 1995, 702, 89–95. [Google Scholar] [CrossRef]
- Peters, E.; Petro, M.; Svec, F.; Fréchet, J. Molded rigid polymer monoliths as separation media for capillary electrochromatography. Anal. Chem. 1997, 69, 3646–3649. [Google Scholar] [CrossRef]
- Xie, S.; Svec, F.; Fréchet, J. Design of reactive porous polymer supports for high throughput bioreactors: Poly(2-vinyl-4,4-dimethylazlactone-co-acrylamide-co-ethylene dimethacrylate) monoliths. Biotechnol. Bioeng. 1999, 62, 30–35. [Google Scholar] [CrossRef]
- Peterson, D.; Rohr, T.; Svec, F.; Fréchet, J. Enzymatic microreactor-on-a-chip: Protein mapping using trypsin immobilized on porous polymer monoliths molded in channels of microfluidic devices. Anal. Chem. 2002, 74, 4081–4088. [Google Scholar] [CrossRef]
- Stachowiak, T.; Svec, F.; Fréchet, J. Patternable protein resistant surfaces for multifunctional microfluidic devices via surface hydrophilization of porous polymer monoliths using photografting. Chem. Mater. 2006, 18, 5950–5957. [Google Scholar] [CrossRef]
- Logan, T.; Clarke, D.; Stachowiak, T.; Svec, F.; Fréchet, J. Photopatterning enzymes on polymer monoliths in microfluidic devices for steady-state kinetic analysis and spatially separated multi-enzyme reactions. Anal. Chem. 2007, 79, 6592–6598. [Google Scholar] [CrossRef]
- Currivan, S.; Connolly, D.; Paull, B. Production of polymer monolithic capillary columns with integrated gold nano-particle modified segments for on-capillary extraction. Microchem. J. 2013, 111, 32–39. [Google Scholar] [CrossRef]
- Pucci, V.; Raggi, M.; Svec, F.; Fréchet, J. Monolithic columns with a gradient of functionalities prepared via photoinitiated grafting for separations using capillary electrochromatography. J. Sep. Sci. 2004, 27, 779–788. [Google Scholar] [CrossRef]
- Currivan, S.; Connolly, D.; Gillespie, E.; Paull, B. Fabrication and characterisation of capillary polymeric monoliths incorporating continuous stationary phase gradients. J. Sep. Sci. 2010, 33, 484–492. [Google Scholar] [CrossRef]
- Collins, D.; Nesterenko, E.; Connolly, D.; Vasquez, M.; Macka, M.; Brabazon, D.; Paull, B. Versatile capillary column temperature control using a thermoelectric array based platform. Anal. Chem. 2011, 83, 4307–4313. [Google Scholar] [CrossRef]
- Currivan, S.; Connolly, D.; Paull, B. Production of novel polymer monolithic columns, with stationary phase gradients, using cyclic olefin co-polymer (COC) optical filters. Analyst 2012, 137, 2559–2566. [Google Scholar] [CrossRef]
- Peters, E.; Svec, F.; Fréchet, J. Rigid macroporous polymer monoliths. Adv. Mater. 1999, 11, 1169–1181. [Google Scholar] [CrossRef]
- Svec, F.; Fréchet, J. Molded rigid monolithic porous polymers: An inexpensive, efficient, and versatile alternative to beads for the design of materials for numerous applications. Ind. Eng. Chem. Res. 1999, 38, 34–48. [Google Scholar] [CrossRef]
- Svec, F. Preparation and HPLC applications of rigid macroporous organic polymer monoliths. J. Sep. Sci. 2004, 27, 747–766. [Google Scholar] [CrossRef]
- Svec, F. Porous polymer monoliths: Amazingly wide variety of techniques enabling their preparation. J. Chromatogr. A 2010, 1217, 902–924. [Google Scholar] [CrossRef]
- Svec, F. Quest for organic polymer-based monolithic columns affording enhanced efficiency in high performance liquid chromatography separations of small molecules in isocratic mode. J. Chromatogr. A 2012, 1228, 250–262. [Google Scholar]
- Schaller, D.; Hilder, E.; Haddad, P. Monolithic stationary phases for fast ion chromatography and capillary electrochromatography of inorganic ions. J. Sep. Sci. 2006, 29, 1705–1719. [Google Scholar] [CrossRef]
- Nordborg, A.; Hilder, E. Recent advances in polymer monoliths for ion-exchange chromatography. Anal. Bioanal. Chem. 2009, 394, 71–84. [Google Scholar] [CrossRef]
- Svec, F. Less common applications of monoliths: I. Microscale protein mapping with proteolytic enzymes immobilized on monolithic supports. Electrophoresis 2006, 27, 947–961. [Google Scholar] [CrossRef]
- Svec, F. Less common applications of monoliths: Preconcentration and solid-phase extraction. J. Chromatogr. B 2006, 841, 52–64. [Google Scholar] [CrossRef]
- Potter, O.; Hilder, E. Porous polymer monoliths for extraction: Diverse applications and platforms. J. Sep. Sci. 2008, 31, 1881–1906. [Google Scholar] [CrossRef]
- Mallik, R.; Hage, D. Affinity monolith chromatography. J. Sep. Sci. 2006, 29, 1686–1704. [Google Scholar] [CrossRef]
- Arrua, R.; Alvarez-Igarzabal, C. Macroporous monolithic supports for affinity chromatography. J. Sep. Sci. 2011, 34, 1974–1987. [Google Scholar]
- Calleri, E.; Ambrosini, S.; Temporini, C.; Massolini, G. New monolithic chromatographic supports for macromolecules immobilization: Challenges and opportunities. J. Pharm. Biomed. Anal. 2012, 69, 64–76. [Google Scholar] [CrossRef]
- Connolly, D.; Currivan, S.; Paull, B. Polymeric monolithic materials modified with nanoparticles for separation and detection of biomolecules: A review. Proteomics 2012, 12, 2904–2917. [Google Scholar] [CrossRef]
- Hutchinson, J.; Hilder, E.; Shellie, R.; Smith, J.; Haddad, P. Towards high capacity latex-coated porous polymer monoliths as ion-exchange stationary phases. Analyst 2006, 131, 215–221. [Google Scholar] [CrossRef]
- Gillespie, E.; Connolly, D.; Paull, B. Using scanning contactless conductivity to optimise photografting procedures and capacity in the production of polymer ion-exchange monoliths. Analyst 2009, 134, 1314–1321. [Google Scholar] [CrossRef]
- Jiang, Z.; Smith, N.; Ferguson, P.; Taylor, M. Hydrophilic interaction chromatography using methacrylate-based monolithic capillary column for the separation of polar analytes. Anal. Chem. 2007, 79, 1243–1250. [Google Scholar] [CrossRef]
- Jiang, Z.; Reilly, J.; Everatt, B.; Smith, N. Novel zwitterionic polyphosphorylcholine monolithic column for hydrophilic interaction chromatography. J. Chromatogr. A 2009, 1216, 2439–2448. [Google Scholar] [CrossRef]
- Jiang, Z.; Smith, N.; Ferguson, P.; Taylor, M. Novel highly hydrophilic zwitterionic monolithic column for hydrophilic interaction chromatography. J. Sep. Sci. 2009, 32, 2544–2555. [Google Scholar] [CrossRef]
- Jandera, P.; Staňková, M.; Škeříková, V.; Urban, J. Cross-linker effects on the separation efficiency on (poly)methacrylate capillary monolithic columns. Part I. Reversed-phase liquid chromatography. J. Chromatogr. A 2013, 1274, 97–106. [Google Scholar] [CrossRef]
- Staňková, M.; Jandera, P.; Škeříková, V.; Urban, J. Cross-linker effects on the separation efficiency on (poly)methacrylate capillary monolithic columns. Part II. Aqueous normal-phase liquid chromatography. J. Chromatogr. A 2013, 1289, 47–57. [Google Scholar] [CrossRef]
- Liu, Z.; Peng, Y.; Wang, T.; Yuan, G.; Zhang, Q.; Guo, J.; Jiang, Z. Preparation and application of novel zwitterionic monolithic column for hydrophilic interaction chromatography. J. Sep. Sci. 2013, 36, 262–269. [Google Scholar] [CrossRef]
- Hilder, E.; Svec, F.; Fréchet, J. Shielded stationary phases based on porous polymer monoliths for the capillary electrochromatography of highly basic biomolecules. Anal. Chem. 2004, 76, 3887–3892. [Google Scholar] [CrossRef]
- Rohr, T.; Hilder, E.; Donovan, J.; Svec, F.; Fréchet, J. Photografting and the control of surface chemistry in three-dimensional porous polymer monoliths. Macromolecules 2003, 36, 1677–1684. [Google Scholar] [CrossRef]
- Peters, E.; Svec, F.; Fréchet, J. Thermally responsive rigid polymer monoliths. Adv. Mater. 1997, 9, 630–633. [Google Scholar] [CrossRef]
- Chen, H.; Huang, T.; Zhang, X. Immunoaffinity extraction of testosterone by antibody immobilized monolithic capillary with on-line laser-induced fluorescence detection. Talanta 2009, 78, 259–264. [Google Scholar] [CrossRef]
- Guerrouache, M.; Millot, M.; Carbonnier, B. Functionalization of macroporous organic polymer monolith based on succinimide ester reactivity for chiral capillary chromatography: A cyclodextrin click approach. Macromol. Rapid Commun. 2009, 30, 109–113. [Google Scholar] [CrossRef]
- Palm, A.; Novotny, M. A monolithic PNGase F enzyme microreactor enabling glycan mass mapping of glycoproteins by mass spectrometry. Rapid Commun. Mass Spectrom. 2005, 19, 1730–1738. [Google Scholar] [CrossRef]
- Guerrouche, M.; Millot, M.; Carbonnier, B. Capillary columns for reversed-phase CEC prepared via surface functionalization of polymer monolith with aromatic selectors. J. Sep. Sci. 2011, 34, 2271–2278. [Google Scholar]
- Dao, T.; Guerrouche, M.; Carbonnier, B. Thiol-yne click adamantane monolithic stationary phase for capillary electrochromatography. Chin. J. Chem. 2012, 30, 2281–2284. [Google Scholar] [CrossRef]
- Sýkora, D.; Svec, F.; Fréchet, J. Separation of oligonucleotides on novel monolithic columns with ion-exchange functional surfaces. J. Chromatogr. A 1999, 852, 297–304. [Google Scholar] [CrossRef]
- Ueki, Y.; Umemura, T.; Li, J.; Odake, T.; Tsunoda, K. Preparation and application of methacrylate-based cation-exchange monolithic columns for capillary ion chromatography. Anal. Chem. 2004, 76, 7007–7012. [Google Scholar] [CrossRef]
- Hilder, E.; Svec, F.; Fréchet, J. Latex-functionalized monolithic columns for the separation of carbohydrates by micro anion-exchange chromatography. J. Chromatogr. A 2004, 1053, 101–106. [Google Scholar] [CrossRef]
- Hutchinson, J.; Zakaria, P.; Bowie, A.; Macka, M.; Avdalovic, N.; Haddad, P. Latex-coated polymeric monolithic ion-exchange stationary phases. 1. Anion-exchange capillary electrochromatography and in-line sample preconcentration in capillary electrophoresis. Anal. Chem. 2005, 77, 407–416. [Google Scholar] [CrossRef]
- Zakaria, P.; Hutchinson, J.; Avdalovic, N.; Liu, Y.; Haddad, P. Latex-coated polymeric monolithic ion-exchange stationary phases. Micro-ion chromatography. Anal. Chem. 2005, 77, 417–423. [Google Scholar] [CrossRef]
- Bisjak, C.; Bakry, R.; Huck, C.; Bonn, G. Amino-functionalized monolithic poly(glycidyl methacrylate-co-divinylbenzene) ion-exchange stationary phases for the separation of oligonucleotides. Chromatographia 2005, 62, S31–S36. [Google Scholar] [CrossRef]
- Preinerstorfer, B.; Linder, W.; Lämmerhofer, M. Polymethacrylate-type monoliths functionalized with chiral amino phosphonic acid-derived strong cation exchange moieties for enantioselective nonaqueous capillary electrochromatography and investigation of the chemical composition of the monolithic polymer. Electrophoresis 2005, 26, 2005–2018. [Google Scholar] [CrossRef]
- Weider, W.; Bisjak, C.; Huck, C.; Bakry, R.; Bonn, G. Monolithic poly(glycidyl methacrylate-co-divinylbenzene) capillary columns functionalized to strong anion exchangers for nucleotide and oligonucleotide separation. J. Sep. Sci. 2006, 29, 2478–2484. [Google Scholar] [CrossRef]
- Lou, Q.; Zou, H.; Xiao, X.; Gou, Z.; Kong, L.; Mao, X. Chromatographic separation of proteins on metal immobilized iminodiacetic acid-bound molded monolithic rods of macroporous poly(glycidyl methacrylate-co-ethylene dimethacrylate). J. Chromatogr. A 2001, 926, 255–264. [Google Scholar] [CrossRef]
- Pan, Z.; Zou, H.; Mo, W.; Huang, X.; Wu, R. Protein A immobilized monolithic capillary column for affinity chromatography. Anal. Chim. Acta 2002, 466, 141–150. [Google Scholar] [CrossRef]
- Bedair, M.; El Rassi, Z. Affinity chromatography with monolithic capillary columns I. Polymethacrylate monoliths with immobilized mannan for the separation of mannose-binding proteins by capillary electrochromatography and nano-scale liquid chromatography. J. Chromatogr. A 2004, 1044, 177–186. [Google Scholar] [CrossRef]
- Bedair, M.; El Rassi, Z. Affinity chromatography with monolithic capillary columns II. Polymethacrylate monoliths with immobilized lectins for the separation of glycoconjugates by nano-liquid affinity chromatography. J. Chromatogr. A 2005, 1079, 236–245. [Google Scholar] [CrossRef]
- Bedair, M.; Oleschuk, R. Lectin affinity chromatography using porous polymer monolith assisted nanoelectrospray MS/MS. Analyst 2006, 131, 1316–1321. [Google Scholar] [CrossRef]
- Yang, C.; Zhu, G.; Zhang, L.; Zhang, W.; Zhang, Y. Repeatedly usable immobilized pH gradient in a monolithic capillary column. Electrophoresis 2004, 25, 1729–1734. [Google Scholar] [CrossRef]
- Zhu, G.; Yuan, H.; Zhao, P.; Zhang, L.; Liang, Z.; Zhang, W.; Zhang, Y. Macroporous polyacrylamide-based monolithic column with immobilized pH gradient for protein analysis. Electrophoresis 2006, 27, 3578–3583. [Google Scholar] [CrossRef]
- Liang, Y.; Zhu, G.; Wang, T.; Zhang, X.; Liang, Z.; Zhang, L.; Zhang, Y. Fast preparation of monolithic immobilized pH gradient column by photopolymerization and photografting techniques for isoelectric focusing separation of proteins. Electrophoresis 2011, 32, 2911–2914. [Google Scholar] [CrossRef]
- Rånby, B. Photochemical modification of polymers-photocrosslinking, surface photografting, and lamination. Polym. Eng. Sci. 1998, 38, 1229–1243. [Google Scholar] [CrossRef]
- Rånby, B.; Yang, W.; Tretinnikov, O. Surface photografting of polymer fibers, films and sheets. Nucl. Instrum. Methods Phys. Res. B 1999, 151, 301–305. [Google Scholar] [CrossRef]
- Rånby, B. Surface modification and lamination of polymers by photografting. Int. J. Adhes. Adhes. 1999, 19, 337–343. [Google Scholar] [CrossRef]
- Rånby, B. Photoinitiated modifications of polymers: Photocrosslinking, surface photografting and photolamination. Mater. Res. Innov. 1998, 2, 64–71. [Google Scholar] [CrossRef]
- Yang, W.; Rånby, B. Bulk surface photografting process and its applications. I. Reactions and kinetics. J. App. Polym. Sci. 1996, 62, 533–543. [Google Scholar] [CrossRef]
- Yang, W.; Rånby, B. Photoinitiation performance of some ketones in the LDPE-acrylic acid surface photografting system. Eur. Polym. J. 1999, 35, 1557–1568. [Google Scholar] [CrossRef]
- Viklund, C.; Ponten, E.; Glad, B.; Irgum, K.; Horstedt, P.; Svec, F. “Molded” macroporous poly(glycidyl methacrylate-co-trimethylolpropane trimethacrylate) materials with fine controlled porous properties: Preparation of monoliths using photoinitiated polymerization. Chem. Mater. 1997, 9, 463–471. [Google Scholar] [CrossRef]
- Eeltink, S.; Hilder, E.; Geiser, L.; Svec, F.; Fréchet, J.; Rozing, G.; Shoenmakers, P.; Kok, W. Controlling the surface chemistry and chromatographic properties of methacrylate-ester based monolithic capillary columns via photografting. J. Sep. Sci. 2007, 30, 407–413. [Google Scholar] [CrossRef]
- Peterson, D.; Rohr, T.; Svec, F.; Fréchet, J. Dual-function microanalytical device by in situ photolithographic grafting of porous polymer monolith: Integrating solid-phase extraction and enzymatic digestion for peptide mass mapping. Anal. Chem. 2003, 75, 5328–5335. [Google Scholar] [CrossRef]
- Connolly, D.; Paull, B. High-performance separation of small inorganic anions on a methacrylate-based polymer monolith grafted with (2(methacryloyloxy)ethyl) trimethylammonium chloride. J. Sep. Sci. 2009, 32, 2653–2658. [Google Scholar] [CrossRef]
- Connolly, D.; O’ Shea, V.; Clark, P.; O’Connor, B.; Paull, B. Evaluation of photografted charged sites within polymer monoliths in capillary columns using contactless conductivity detection. J. Sep. Sci. 2007, 30, 3060–3068. [Google Scholar] [CrossRef]
- Křenková, J.; Lacher, N.; Svec, F. Highly efficient enzyme reactors containing trypsin and endoproteinase lysc immobilized on porous polymer monolith coupled to MS suitable for analysis of antibodies. Anal. Chem. 2009, 81, 2004–2012. [Google Scholar] [CrossRef]
- Křenková, J.; Lacher, N.; Svec, F. Multidimensional system enabling deglycosylation of proteins using a capillary reactor with peptide-N-glycosidase F immobilized on a porous polymer monolith and hydrophilic interaction liquid chromatography–mass spectrometry of glycans. J. Chromatogr. A 2009, 1216, 3252–3259. [Google Scholar] [CrossRef]
- Potter, O.; Breadmore, M.; Hilder, E. Boronate functionalised polymer monoliths for microscale affinity chromatography. Analyst 2006, 131, 1094–1096. [Google Scholar] [CrossRef]
- Gillespie, E.; Connolly, D.; Nesterenko, P.; Paull, B. Accurate non-invasive determination of pKa of surface functionalised ion exchange monoliths using capacitively coupled contactless conductivity detection. Analyst 2008, 133, 874–876. [Google Scholar] [CrossRef]
- Gillespie, E.; Connolly, D.; Nesterenko, P.; Paull, B. On-column titration and investigation of metal complex formation for aminopolycarboxylate functionalised monoliths using scanning contactless conductivity detection. J. Sep. Sci. 2009, 32, 2659–2667. [Google Scholar] [CrossRef]
- Walsh, Z.; Levkin, P.; Jain, V.; Paull, B.; Svec, F.; Macka, M. Visible light initiated polymerization of styrenic monolithic stationary phases using 470 nm light emitting diode arrays. J. Sep. Sci. 2010, 33, 61–66. [Google Scholar] [CrossRef]
- Walsh, Z.; Levkin, P.; Abele, S.; Scarmagnani, S.; Heger, D.; Klán, P.; Diamond, D.; Paull, B.; Svec, F.; Macka, M. Polymerisation and surface modification of methacrylate monoliths in polyimide channels and polyimide coated capillaries using 660 nm light emitting diodes. J. Chromatogr. A 2011, 1218, 2954–2962. [Google Scholar] [CrossRef]
- Moyna, A.; Connolly, D.; Nesterenko, E.; Nesterenko, P.; Paull, B. Separation of selected transition metals by capillary chelation ion chromatography using acetyl-iminodiacetic acid modified capillary polymer monoliths. J. Chromatogr. A 2012, 1249, 155–163. [Google Scholar] [CrossRef]
- Moyna, A.; Connolly, D.; Nesterenko, E.; Nesterenko, P.; Paull, B. Iminodiacetic acid functionalised organopolymer monoliths: Application to the separation of metal cations by capillary high-performance chelation ion chromatography. Anal. Bioanal. Chem. 2013, 405, 2207–2217. [Google Scholar] [CrossRef]
- Rohr, T.; Ogletree, D.; Svec, F.; Fréchet, J. Surface functionalization of thermoplastic polymers for the fabrication of microfluidic devices by photoinitiated grafting. Adv. Funct. Mater. 2003, 13, 264–270. [Google Scholar] [CrossRef]
- Keppler, M.; Hüsing, N. Space-confined click reactions in hierarchically organized silica monoliths. New J. Chem. 2011, 35, 681–690. [Google Scholar] [CrossRef]
- Slater, M.; Fréchet, J.; Svec, F. In-column preparation of a brush-type chiral stationary phase using click chemistry and a silica monolith. J. Sep. Sci. 2009, 32, 21–28. [Google Scholar] [CrossRef]
- Keppler, M.; Holzbock, J.; Akbarzadeh, J.; Peterlik, H.; Hüsing, N. Inorganic–organic hybrid materials through postsynthesis modification: Impact of the treatment with azides on the mesopore structure. Beilstein J. Nanotechnol. 2011, 2, 486–498. [Google Scholar] [CrossRef]
- Chen, Y.; Wu, M.; Wang, K.; Chen, B.; Yao, S.; Zou, H.; Nie, L. Vinyl functionalized silica hybrid monolith-based trypsin microreactor for on line digestion and separation via thiol-ene “click” strategy. J. Chromatogr. A 2011, 1218, 7982–7988. [Google Scholar] [CrossRef]
- Wang, K.; Chen, Y.; Yang, H.; Li, Y.; Nie, L.; Yao, S. Modification of VTMS hybrid monolith via thiol-ene click chemistry for capillary electrochromatography. Talanta 2012, 91, 52–59. [Google Scholar] [CrossRef]
- Yang, H.; Chen, Y.; Liu, Y.; Nie, L.; Yao, S. One-pot synthesis of (3-sulfopropyl methacrylate potassium)-silica hybrid monolith via thiol-ene click chemistry for CEC. Electrophoresis 2013, 34, 510–517. [Google Scholar] [CrossRef]
- Preinerstorfer, B.; Bicker, W.; Linder, W.; Lämmerhofer, M. Development of reactive thiol-modified monolithic capillaries and in-column surface functionalization by radical addition of a chromatographic ligand for capillary electrochromatography. J. Chromatogr. A 2004, 1044, 187–199. [Google Scholar] [CrossRef]
- Lv, Y.; Hughes, T.; Hao, X.; Hart, N.; Littler, S.; Zhang, X.; Tan, T. A novel route to prepare highly reactive and versatile chromatographic monoliths. Macromol. Rapid Commun. 2010, 31, 1785–1790. [Google Scholar] [CrossRef]
- Lv, Y.; Lin, Z.; Svec, F. “Thiol–ene’’ click chemistry: A facile and versatile route for the functionalization of porous polymer monoliths. Analyst 2012, 137, 4114–4118. [Google Scholar] [CrossRef]
- Tijunelyte, I.; Babinot, J.; Guerrouache, M.; Valincius, G.; Carbonnier, B. Hydrophilic monolith with ethylene glycol-based grafts prepared via surface confined thiol-ene click photoaddition. Polymer 2012, 53, 29–36. [Google Scholar] [CrossRef]
- Sun, X.; Lin, D.; He, X.; Chen, L.; Zhang, Y. A facile and efficient strategy for one-step in situ preparation of hydrophobic organic monolithic stationary phases by click chemistry and its application on protein separation. Talanta 2010, 82, 404–408. [Google Scholar] [CrossRef]
- Sun, X.; He, X.; Chen, L.; Zhang, Y. In-column “click” preparation of hydrophobic organic monolithic stationary phases for protein separation. Anal. Bioanal. Chem. 2011, 399, 3407–3413. [Google Scholar] [CrossRef]
- Salwinski, A.; Roy, V.; Agrofoglio, L.; Delépée, R. In situ one-step method for synthesis of “click”-functionalized monolithic stationary phase for capillary electrochromatography. Macromol. Chem. Phys. 2011, 212, 2700–2707. [Google Scholar] [CrossRef]
- Viklund, C.; Svec, F.; Fréchet, J.; Irgum, K. Fast ion-exchange HPLC of proteins using porous poly(glycidyl methacrylate-co-ethylene dimethacrylate) monoliths grafted with poly(2-acrylamido-2-methyl-1-propanesulfonic acid). Biotechnol. Prog. 1997, 13, 597–600. [Google Scholar] [CrossRef]
- Viklund, C.; Irgum, K. Synthesis of porous zwitterionic sulfobetaine monoliths and characterization of their interaction with proteins. Macromolecules. 2000, 33, 2539–2544. [Google Scholar] [CrossRef]
- Meyer, U.; Svec, F.; Fréchet, J.; Hawker, C.; Irgum, K. Use of stable free radicals for the sequential preparation and surface grafting of functionalized macroporous monoliths. Macromolecules 2000, 33, 7769–7775. [Google Scholar] [CrossRef]
- Georges, M.; Veregin, R.; Kazmaier, P.; Hamer, G. Narrow molecular weight resins by a free-radical polymerisation process. Macromolecules 1993, 26, 2987–2988. [Google Scholar] [CrossRef]
- Buchmeiser, M.; Sinner, F.; Mupa, M.; Wurst, K. Ring-opening metathesis polymerization for the preparation of surface-grafted polymer supports. Macromolecules 2000, 33, 32–39. [Google Scholar] [CrossRef]
- Sinner, F.; Buchmeiser, M. A new class of continuous polymer supports prepared by ring-opening metathesis polymerization: A straightforward route to functionalized monoliths. Macromolecules 2000, 33, 5777–5786. [Google Scholar] [CrossRef]
- Kanamori, K.; Hasegawa, J.; Nakanishi, K.; Hanada, T. Facile synthesis of macroporous cross-linked methacrylate gels by atom transfer radical polymerization. Macromolecules 2008, 41, 7186–7193. [Google Scholar] [CrossRef]
- Limé, F.; Irgum, K. Hydrobromination of residual vinyl groups on divinylbenzene polymer particles followed by atom transfer radical surface graft polymerization. J. Polym. Sci.: Part A: Polym. Chem. 2009, 47, 1259–1265. [Google Scholar] [CrossRef]
- Liu, H.; Zhuang, X.; Turson, M.; Zhang, M.; Dong, X. Enrofloxacin-imprinted monolithic columns synthesized using reversible addition-fragmentation chain transfer polymerization. J. Sep. Sci. 2008, 31, 1694–1701. [Google Scholar] [CrossRef]
- Barlow, K.; Hao, X.; Hughes, T.; Hutt, O.; Polyzos, A.; Turner, K.; Moad, G. Porous, functional poly(styrene-co-divinylbenzene) monoliths by RAFT polymerization. Polym. Chem. 2014, 5, 722–732. [Google Scholar] [CrossRef]
- Viklund, C.; Nordstrӧm, A.; Irgum, K.; Svec, F.; Fréchet, J. Preparation of porous poly(styrene-co-divinylbenzene) monoliths with controlled pore size distributions initiated by stable free radicals and their pore surface functionalization by grafting. Macromolecules 2001, 34, 4361–4369. [Google Scholar] [CrossRef]
- Peters, E.; Svec, F.; Fréchet, J.; Viklund, C.; Irgum, K. Control of porous properties and surface chemistry in “molded” porous polymer monoliths prepared by polymerization in the presence of TEMPO. Macromolecules. 1999, 32, 6377–6379. [Google Scholar] [CrossRef]
- Germain, J.; Fréchet, J.; Svec, F. Nanoporous, hypercrosslinked polypyrroles: Effect of crosslinking moiety on pore size and selective gas adsorption. Chem. Commun. 2009. [Google Scholar] [CrossRef]
- Davankov, V.; Rogozhin, S.; Tsyurupa, M. Macronet Polystyrene Structures for Ionites and Method of Producing Same. U.S. Patent 3,729,457, 24 April 1973. [Google Scholar]
- Pastukhov, A.; Tsyurupa, M.; Davankov, V. Hypercrosslinked polystyrene: A polymer in a non-classical physical state. J. Polym. Sci. Polym. Phys. 1999, 37, 2324–2333. [Google Scholar] [CrossRef]
- Davankov, V.; Tsyurupa, M. Structure and properties of hypercrosslinked polystyrene—The first representative of a new class of polymer networks. React. Polym. 1990, 13, 27–42. [Google Scholar] [CrossRef]
- Davankov, V.; Tsyurupa, M.; Ilyin, M.; Pavlova, L. Hypercross-linked polystyrene and its potentials for liquid chromatography: A mini-review. J. Chromatogr. A 2002, 965, 65–73. [Google Scholar] [CrossRef]
- Tsyurupa, M.; Davankov, V. Porous structure of hypercrosslinked polystyrene: State-of-the-art mini-review. React. Funct. Polym. 2006, 66, 768–779. [Google Scholar] [CrossRef]
- Urban, J.; Svec, F.; Fréchet, J. Efficient separation of small molecules using a large surface area hypercrosslinked monolithic polymer capillary column. Anal. Chem. 2010, 82, 1621–1623. [Google Scholar] [CrossRef]
- Minakuchi, H.; Nakanishi, K.; Soga, N.; Ishizuka, N.; Tanaka, N. Octadecylsilylated porous silica rods as separation media for reversed-phase liquid chromatography. Anal. Chem. 1996, 68, 3498–3501. [Google Scholar] [CrossRef]
- Urban, J.; Svec, F.; Fréchet, J. Hypercrosslinking: New approach to porous polymer monolithic capillary columns with large surface area for the highly efficient separation of small molecules. J. Chromatogr. A 2010, 1217, 8212–8221. [Google Scholar] [CrossRef]
- Chen, X.; Dinh, N.; Zhao, J.; Wang, Y.; Li, S.; Svec, F. Effect of ion adsorption on CEC separation of small molecules using hypercrosslinked porous polymer monolithic capillary columns. J. Sep. Sci. 2012, 35, 1502–1505. [Google Scholar] [CrossRef]
- Teisseyre, T.; Urban, J.; Halpern-Manners, N.; Chambers, S.; Bajaj, V.; Svec, F.; Pines, A. Remotely detected NMR for the characterization of flow and fast chromatographic separations using organic polymer monoliths. Anal. Chem. 2011, 83, 6004–6010. [Google Scholar] [CrossRef]
- Škeříková, V.; Urban, J. Highly stable surface modification of hypercrosslinked monolithic capillary columns and their application in hydrophilic interaction chromatography. J. Sep. Sci. 2013, 36, 2806–2812. [Google Scholar] [CrossRef]
- Lv, Y.; Lin, Z.; Svec, F. Hypercrosslinked large surface area porous polymer monoliths for hydrophilic interaction liquid chromatography of small molecules featuring zwitterionic functionalities attached to gold nanoparticles held in layered structure. Anal. Chem. 2012, 84, 8457–8460. [Google Scholar] [CrossRef]
- Křenková, J.; Foret, F. Iron oxide nanoparticle coating of organic polymer-based monolithic columns for phosphopeptide enrichment. J. Sep. Sci. 2011, 34, 2106–2112. [Google Scholar]
- Li, Y.; Chen, Y.; Xiang, R.; Ciuparu, D.; Pfefferle, L.; Horvath, C.; Wilkins, J. Incorporation of single-wall carbon nanotubes into an organic polymer monolithic stationary phase for µ-HPLC and capillary electrochromatography. Anal. Chem. 2005, 77, 1398–1406. [Google Scholar] [CrossRef]
- Křenková, J.; Lacher, N.; Svec, F. Control of selectivity via nanochemistry: Monolithic capillary column containing hydroxyapatite nanoparticles for separation of proteins and enrichment of phosphopeptides. Anal. Chem. 2010, 82, 8335–8341. [Google Scholar] [CrossRef]
- Chambers, S.; Holcombe, T.; Svec, F.; Fréchet, J. Porous polymer monoliths functionalized through copolymerization of a C60 fullerene-containing methacrylate monomer for highly efficient separations of small molecules. Anal. Chem. 2011, 83, 9478–9484. [Google Scholar] [CrossRef]
- Tong, S.; Liu, Q.; Li, Y.; Zhou, W.; Jia, Q.; Duan, T. Preparation of porous polymer monolithic column incorporated with graphene nanosheets for solid phase microextraction and enrichment of glucocorticoids. J. Chromatogr. A 2012, 1253, 22–31. [Google Scholar] [CrossRef]
- Tobal, K.; Guerre, O.; Rolando, C.; Le Gac, S. Metal nanoparticle-based polymer monolithic columns dedicated to the specific trapping of phosphopeptides. Mol. Cell. Proteomics 2006, 5, S278. [Google Scholar]
- Hsieh, H.; Sheu, C.; Shi, F.; Li, D. Development of a titanium dioxide nanoparticle pipette-tip for the selective enrichment of phosphorylated peptides. J. Chromatogr. A 2007, 1165, 128–135. [Google Scholar] [CrossRef]
- Rainer, M.; Sonderegger, H.; Bakry, R.; Huck, C.; Morandell, S.; Huber, L.; Gjerde, D.; Bonn, G. Analysis of protein phosphorylation by monolithic extraction columns based on poly(divinylbenzene) containing embedded titanium dioxide and zirconium dioxide nano-powders. Proteomics 2008, 8, 4593–4602. [Google Scholar] [CrossRef]
- Liu, J.; White, I.; DeVoe, D. Nanoparticle-functionalized porous polymer monolith detection elements for surface-enhanced raman scattering. Anal. Chem. 2011, 83, 2119–2124. [Google Scholar] [CrossRef]
- Lei, W.; Zhang, L.; Wan, L.; Shi, B.; Wang, Y.; Zhang, W. Hybrid monolithic columns with nanoparticles incorporated for capillary electrochromatography. J. Chromatogr. A 2012, 1239, 64–71. [Google Scholar] [CrossRef]
- Alwael, H.; Connolly, D.; Clarke, P.; Thompson, R.; Twamley, B.; O’Connor, B.; Paull, B. Pipette-tip selective extraction of glycoproteins with lectin modified gold nano-particles on a polymer monolithic phase. Analyst 2011, 136, 2619–2628. [Google Scholar] [CrossRef]
- Zhang, A.; Ye, F.; Lu, J.; Zhao, S. Screening α-glucosidase inhibitor from natural products by capillary electrophoresis with immobilized enzyme onto polymer monolith modified by gold nanoparticles. Food Chem. 2013, 141, 1854–1859. [Google Scholar] [CrossRef]
- Cao, Q.; Xu, Y.; Liu, F.; Svec, F.; Fréchet, J. Polymer monoliths with exchangeable chemistries: Use of gold nanoparticles as intermediate ligands for capillary columns with varying surface functionalities. Anal. Chem. 2010, 82, 7416–7421. [Google Scholar] [CrossRef]
- Guerrouache, M.; Mahouche-Chergui, S.; Chehimi, M.; Carbonnier, B. Site-specific immobilisation of gold nanoparticles on a porous monolith surface by using a thiol–yne click photopatterning approach. Chem. Commun. 2012, 48, 7486–7488. [Google Scholar] [CrossRef]
- Xu, Y.; Cao, Q.; Svec, F.; Fréchet, J. Porous polymer monolithic column with surface-bound gold nanoparticles for the capture and separation of cysteine-containing peptides. Anal. Chem. 2010, 82, 3352–3358. [Google Scholar] [CrossRef]
- Connolly, D.; Twamley, B.; Paull, B. High-capacity gold nanoparticle functionalised polymer monoliths. Chem. Commun. 2010, 46, 2109–2111. [Google Scholar] [CrossRef]
- Křenková, J.; Foret, F. Nanoparticle-modified monolithic pipette tips for phosphopeptide enrichment. Anal. Bioanal. Chem. 2013, 405, 2175–2183. [Google Scholar] [CrossRef]
- Li, W.; Zhou, X.; Ye, J.; Jia, Q. Develompent of a γ-alumina-nanoparticle functionalized porous polymer monolith for the enrichment of sudan dyes in red wine samples. J. Sep. Sci. 2013, 36, 3330–3337. [Google Scholar]
- Floris, P.; Twamley, B.; Nesterenko, P.; Paull, B.; Connolly, D. Agglomerated polymer monoliths with bimetallic nano-particles as flow-through micro-reactors. Microchim. Acta 2012, 179, 149–156. [Google Scholar] [CrossRef]
- Tong, S.; Zhou, X.; Zhou, C.; Li, Y.; Li, W.; Zhou, W.; Jia, Q. A strategy to decorate porous polymer monoliths with graphene oxide and graphene nanosheets. Analyst 2013, 138, 1549–1557. [Google Scholar] [CrossRef]
- Chambers, S.; Svec, F.; Fréchet, J. Incorporation of carbon nanotubes in porous polymer monolithic capillary columns to enhance the chromatographic separation of small molecules. J. Chromatogr. A 2011, 1218, 2546–2552. [Google Scholar] [CrossRef]
- Křenková, J.; Foret, F.; Svec, F. Less common applications of monoliths: V. Monolithic scaffolds modified with nanostructures for chromatographic separations and tissue engineering. J. Sep. Sci. 2012, 35, 1266–1283. [Google Scholar] [CrossRef]
- Nesterenko, E.; Nesterenko, P.; Connolly, D.; He, X.; Floris, P.; Duffy, E.; Paull, B. Nano-particle modified stationary phases for high-performance liquid chromatography. Analyst 2013, 138, 4229–4254. [Google Scholar] [CrossRef]
- Frens, G. Controlled nucleation for regulation of particle-size in monodisperse gold suspensions. Nat. Phys. Sci. 1973, 241, 20–22. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Currivan, S.; Jandera, P. Post-Polymerization Modifications of Polymeric Monolithic Columns: A Review. Chromatography 2014, 1, 24-53. https://doi.org/10.3390/chromatography1010024
Currivan S, Jandera P. Post-Polymerization Modifications of Polymeric Monolithic Columns: A Review. Chromatography. 2014; 1(1):24-53. https://doi.org/10.3390/chromatography1010024
Chicago/Turabian StyleCurrivan, Sinéad, and Pavel Jandera. 2014. "Post-Polymerization Modifications of Polymeric Monolithic Columns: A Review" Chromatography 1, no. 1: 24-53. https://doi.org/10.3390/chromatography1010024
APA StyleCurrivan, S., & Jandera, P. (2014). Post-Polymerization Modifications of Polymeric Monolithic Columns: A Review. Chromatography, 1(1), 24-53. https://doi.org/10.3390/chromatography1010024