Laryngeal Masks in Neonatal Resuscitation—A Narrative Review of Updates 2022
Abstract
1. Introduction
2. Methods
3. Discussion
3.1. Historical Perspectives
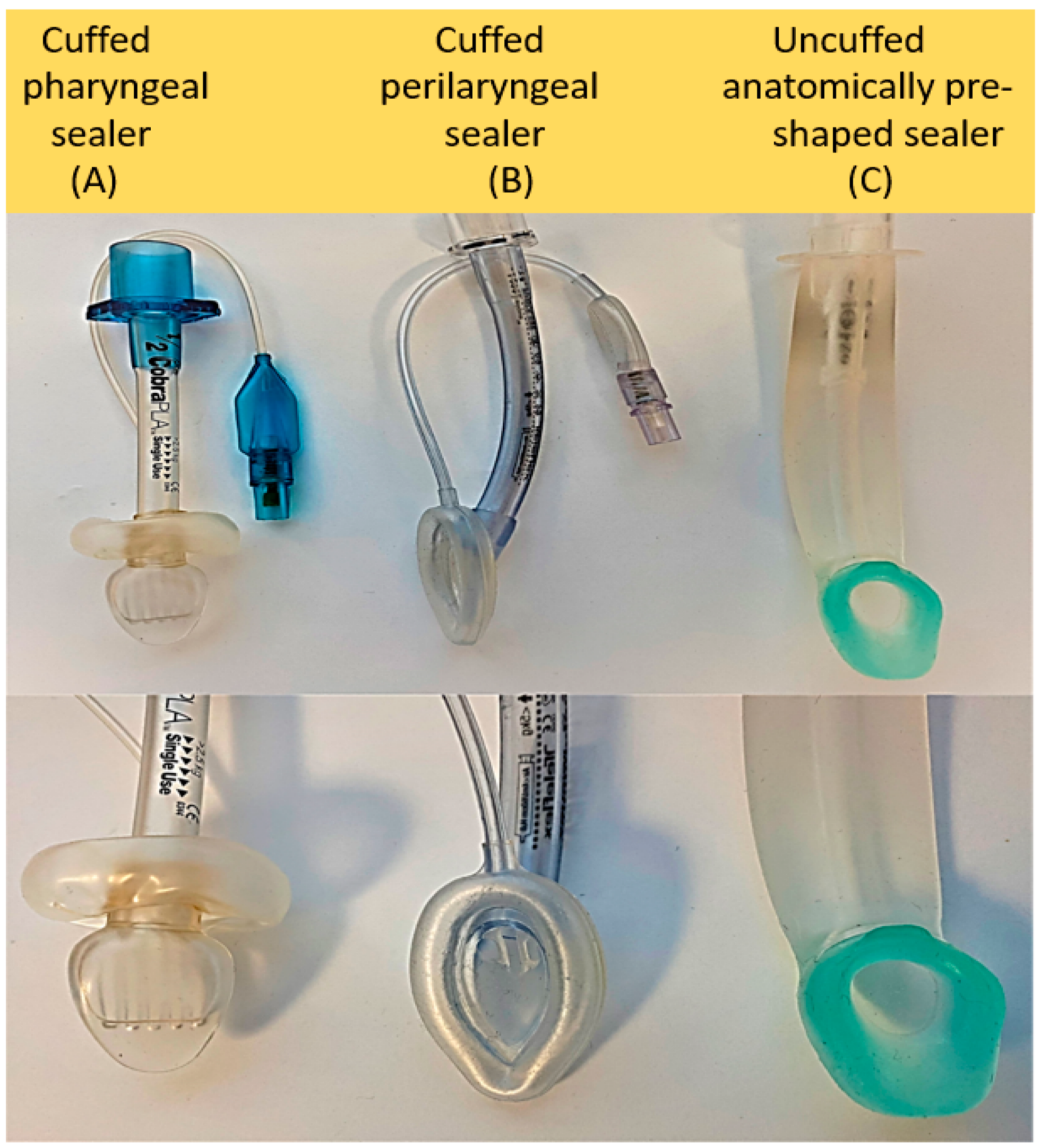
3.2. Classification of Supraglottic Airway Devices
3.3. LM and Its Effect on Respiratory Mechanics
3.4. LM—A Peri-Laryngeal Sealing Device
3.5. A Second-Generation LM
3.6. LM Use in Current Neonatal Practice and Perceptions
3.7. Insertion Technique and Training
3.8. LM during Initial Stabilization
3.8.1. LM as Primary PPV Interface
3.8.2. LM as Secondary PPV Interface
3.9. Advantages of LM as a PPV Interface
3.10. Disadvantages of LM as a PPV Interface
3.11. LM during Chest Compressions
3.12. LM for Medications during Resuscitation
3.13. LM Use in Preterm Infants
3.14. LM for Difficult Airway
3.15. LM Use as an Element of Palliative Care
3.16. LM Use in Prehospital Settings
3.17. LM Use in Low Resource Settings
3.18. LM Use—NRP 2020
3.19. Future Directions
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Wyllie, J. Resuscitation of the depressed newborn. Semin. Fetal Neonatal Med. 2006, 11, 158–165. [Google Scholar] [CrossRef]
- Ersdal, H.L.; Mduma, E.; Svensen, E.; Perlman, J.M. Early initiation of basic resuscitation interventions including face mask ventilation may reduce birth asphyxia related mortality in low-income countries: A prospective descriptive observational study. Resuscitation 2012, 83, 869–873. [Google Scholar] [CrossRef] [PubMed]
- Aziz, K.; Lee, H.C.; Escobedo, M.B.; Hoover, A.V.; Kamath-Rayne, B.D.; Kapadia, V.S.; Magid, D.J.; Niermeyer, S.; Schmölzer, G.M.; Szyld, E.; et al. Part 5: Neonatal Resuscitation: 2020 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2020, 142 (Suppl. 2), S524–S550. [Google Scholar] [CrossRef]
- Kuypers, K.L.; Lamberska, T.; Martherus, T.; Dekker, J.; Böhringer, S.; Hooper, S.B.; Plavka, R.; Pas, A.B.T. The effect of a face mask for respiratory support on breathing in preterm infants at birth. Resuscitation 2019, 144, 178–184. [Google Scholar] [CrossRef]
- Martherus, T.; Oberthuer, A.; Dekker, J.; Hooper, S.B.; McGillick, E.; Kribs, A.; Pas, A.T. Supporting breathing of preterm infants at birth: A narrative review. Arch. Dis. Child. Fetal Neonatal Ed. 2019, 104, F102–F107. [Google Scholar] [CrossRef]
- Singh, G.P.; Chowdhury, T.; Bindu, B.; Schaller, B. Sudden Infant Death Syndrome—Role of Trigeminocardiac Reflex: A Review. Front. Neurol. 2016, 7, 221. [Google Scholar] [CrossRef] [PubMed]
- Gaertner, V.D.; Rüegger, C.M.; O’Currain, E.; Kamlin, C.O.F.; Hooper, S.B.; Davis, P.G.; Springer, L. Physiological responses to facemask application in newborns immediately after birth. Arch. Dis. Child. Fetal Neonatal Ed. 2021, 106, 381–385. [Google Scholar] [CrossRef] [PubMed]
- Schmölzer, G.M.; Dawson, J.A.; Kamlin, C.O.; O’Donnell, C.P.; Morley, C.J.; Davis, P.G. Airway obstruction and gas leak during mask ventilation of preterm infants in the delivery room. Arch. Dis. Child. Fetal Neonatal Ed. 2011, 96, F254–F257. [Google Scholar] [CrossRef]
- Perlman, J.M.; Risser, R. Cardiopulmonary resuscitation in the delivery room. Associated clinical events. Arch. Pediatr. Adolesc. Med. 1995, 149, 20–25. [Google Scholar] [CrossRef]
- Kuypers, K.L.; Lamberska, T.; Martherus, T.; Dekker, J.; Böhringer, S.; Hooper, S.B.; Plavka, R.; Pas, A.B.T. Comparing the effect of two different interfaces on breathing of preterm infants at birth: A matched-pairs analysis. Resuscitation 2020, 157, 60–66. [Google Scholar] [CrossRef]
- Mangat, A.K.; Bruckner, M.; Schmölzer, G.M. Face mask versus nasal prong or nasopharyngeal tube for neonatal resuscitation in the delivery room: A systematic review and meta-analysis. Arch. Dis. Child. 2021, 106, 561–567. [Google Scholar] [CrossRef] [PubMed]
- Bansal, S.C.; Caoci, S.; Dempsey, E.; Trevisanuto, D.; Roehr, C.C. The Laryngeal Mask Airway and Its Use in Neonatal Resuscitation: A Critical Review of Where We Are in 2017/2018. Neonatology 2018, 113, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, M.J.; Kumar, M. Laryngeal mask airway versus bag-mask ventilation or endotracheal intubation for neonatal resuscitation. Cochrane Database Syst. Rev. 2018, 3, CD003314. [Google Scholar] [CrossRef] [PubMed]
- Brain, A.I. The laryngeal mask—A new concept in airway management. Br. J. Anaesth. 1983, 55, 801–805. [Google Scholar] [CrossRef] [PubMed]
- Mizushima, A.; Wardall, G.J.; Simpson, D.L. The laryngeal mask airway in infants. Anaesthesia 1992, 47, 849–851. [Google Scholar] [CrossRef]
- Paterson, S.J.; Byrne, P.J.; Molesky, M.G.; Seal, R.F.; Finucane, B.T. Neonatal resuscitation using the laryngeal mask airway. Anesthesiology 1994, 80, 1248–1253; discussion 27A. [Google Scholar] [CrossRef]
- Singh, R. Controlled trial to evaluate the use of LMA for neonatal resuscitation. J. Anaesth. Clin. Pharmacol. 2005, 21, 303–306. [Google Scholar]
- Feroze, F.; Masood, N.; Khuwaja, A.; Malik, F.I. Neonatal Resuscitation. Prof. Med. J. 2008, 15, 148–152. [Google Scholar] [CrossRef]
- Trevisanuto, D.; Cavallin, F.; Nguyen, L.N.; Nguyen, T.V.; Tran, L.D.; Tran, C.D.; Doglioni, N.; Micaglio, M.; Moccia, L. Supreme Laryngeal Mask Airway versus Face Mask during Neonatal Resuscitation: A Randomized Controlled Trial. J. Pediatr. 2015, 167, 286–291.e1. [Google Scholar] [CrossRef]
- American Academy of Pediatrics; American Heart Association. Textbook of Neonatal Resuscitation; Weiner, G.M., Zaichkin, J., Kattwinkel, J., Eds.; American Academy of Pediatrics: Elk Grove Village, IL, USA, 2016. [Google Scholar]
- Ramaiah, R.; Das, D.; Bhananker, S.M.; Joffe, A.M. Extraglottic airway devices: A review. Int. J. Crit. Illn. Inj. Sci. 2014, 4, 77–87. [Google Scholar] [CrossRef]
- Miller, D.M. A proposed classification and scoring system for supraglottic sealing airways: A brief review. Anesth. Analg. 2004, 99, 1553–1559. [Google Scholar] [CrossRef] [PubMed]
- Wahlen, B.M.; Heinrichs, W.; Latorre, F. Gastric insufflation pressure, air leakage and respiratory mechanics in the use of the laryngeal mask airway (LMATM)in children. Pediatr. Anesth. 2004, 14, 313–317. [Google Scholar] [CrossRef]
- Ozden, E.S.; Meco, B.C.; Alanoglu, Z.; Alkıs, N. Comparison of ProSeal laryngeal mask airway (PLMA) with cuffed and uncuffed endotracheal tubes in infants. Bosn. J. Basic Med. Sci. 2016, 16, 286–291. [Google Scholar] [CrossRef]
- Pejovic, N.J.; Cavallin, F.; Mpamize, A.; Lubulwa, C.; Höök, S.M.; Byamugisha, J.; Nankunda, J.; Tylleskär, T.; Trevisanuto, D. Respiratory monitoring during neonatal resuscitation using a supraglottic airway device vs. a face mask. Resuscitation 2022, 171, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Mahdavi, A.; Razavi, S.S.; Malekianzadeh, B.; Sadeghi, A. Comparison of the Peak Inspiratory Pressure and Lung Dynamic Compliance between a Classic Laryngeal Mask Airway and an Endotracheal Tube in Children Under Mechanical Ventilation. Tanaffos 2017, 16, 289–294. [Google Scholar] [PubMed]
- Reissmann, H.; Pothmann, W.; Füllekrug, B.; Dietz, R.; Schulte am Esch, J. Resistance of laryngeal mask airway and tracheal tube in mechanically ventilated patients. Br. J. Anaesth. 2000, 85, 410–416. [Google Scholar] [CrossRef]
- Natalini, G.; Rosano, A.; Lanza, G.; Martinelli, E.; Pletti, C.; Bernardini, A. Resistive load of laryngeal mask airway and ProSeal laryngeal mask airway in mechanically ventilated patients. Acta Anaesthesiol. Scand. 2003, 47, 761–764. [Google Scholar] [CrossRef] [PubMed]
- Keller, C.; Brimacombe, J.R.; Keller, K.; Morris, R. Comparison of four methods for assessing airway sealing pressure with the laryngeal mask airway in adult patients. Br. J. Anaesth. 1999, 82, 286–287. [Google Scholar] [CrossRef]
- Patel, B.; Bingham, R. Laryngeal mask airway and other supraglottic airway devices in paediatric practice. Contin. Educ. Anaesth. Crit. Care Pain. 2009, 9, 6–9. [Google Scholar] [CrossRef]
- Van Zundert, A.; Brimacombe, J. The LMA SupremeTM—A pilot study. Anaesthesia 2008, 63, 209–210. [Google Scholar] [CrossRef]
- Pejovic, N.J.; Höök, S.M.; Byamugisha, J.; Alfvén, T.; Lubulwa, C.; Cavallin, F.; Nankunda, J.; Ersdal, H.; Blennow, M.; Trevisanuto, D.; et al. A Randomized Trial of Laryngeal Mask Airway in Neonatal Resuscitation. N. Engl. J. Med. 2020, 383, 2138–2147. [Google Scholar] [CrossRef] [PubMed]
- Ankay-Yılbaş, A.; Başaran, B.; Üzümcügil, F.; Akça, B.; İzgi, M.; Canbay, Ö. Comparison of i-gel, LMA-supreme, LMA-classic and LMAproseal as conduits of endotracheal intubation in newborns and infants: A manikin study. Turk J. Pediatr. 2019, 61, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Goel, D.; Shah, D.; Hinder, M.; Tracy, M. Laryngeal mask airway use during neonatal resuscitation: A survey of practice across newborn intensive care units and neonatal retrieval services in Australian New Zealand Neonatal Network. J. Paediatr. Child. Health 2020, 56, 1346–1350. [Google Scholar] [CrossRef]
- Mani, S.; Rawat, M. Proficiency of Laryngeal Mask Airway Insertion Skill in NRP Certified Providers. Am. J. Perinatol. Available online: https://www.thieme-connect.de/products/ejournals/abstract/10.1055/s-0040-1721379 (accessed on 29 November 2020). [CrossRef] [PubMed]
- Shah, B.A.; Foulks, A.; Lapadula, M.C.; McCoy, M.; Hallford, G.; Bedwell, S.; DeShea, L.; Szyld, E. Laryngeal Mask Use in the Neonatal Population: A Survey of Practice Providers at a Regional Tertiary Care Center in the United States. Am. J. Perinatol. Available online: https://www.thieme-connect.de/products/ejournals/abstract/10.1055/s-0041-1736662 (accessed on 2 November 2020). [CrossRef]
- Pinheiro, J.M.; Santana-Rivas, Q.; Pezzano, C. Randomized trial of laryngeal mask airway versus endotracheal intubation for surfactant delivery. J. Perinatol. 2016, 36, 196–201. [Google Scholar] [CrossRef]
- American Academy of Pediatrics; American Heart Association. Textbook of Neonatal Resuscitation; Weiner, G.M., Zaichkin, J., Eds.; American Academy of Pediatrics: Itasca, IL, USA, 2021. [Google Scholar]
- Brimacombe, J.; Berry, A. Insertion of the laryngeal mask airway—A prospective study of four techniques. Anaesth. Intensiv. Care 1993, 21, 89–92. [Google Scholar] [CrossRef]
- Hwang, J.-W.; Park, H.-P.; Lim, Y.-J.; Do, S.-H.; Lee, S.-C.; Jeon, Y.-T. Comparison of Two Insertion Techniques of ProSeal™ Laryngeal Mask Airway: Standard versus90-degree Rotation. Anesthesiology 2009, 110, 905–907. [Google Scholar] [CrossRef]
- Koo, B.-W.; Oh, A.-Y.; Hwang, J.-W.; Na, H.-S.; Min, S.-W. Comparison of standard versus 90° rotation technique for LMA Flexible™ insertion: A randomized controlled trial. BMC Anesthesiol. 2019, 19, 95. [Google Scholar] [CrossRef]
- Yun, M.-J.; Hwang, J.-W.; Park, S.-H.; Han, S.-H.; Park, H.-P.; Kim, J.-H.; Jeon, Y.-T.; Lee, S.-C. The 90° rotation technique improves the ease of insertion of the ProSeal™ laryngeal mask airway in children. Can. J. Anesth. 2011, 58, 379–383. [Google Scholar] [CrossRef]
- Shyam, T.; Selvaraj, V. Airway management using LMA-evaluation of three insertional techniques-a prospective randomised study. J. Anaesthesiol. Clin. Pharmacol. 2021, 37, 108–113. [Google Scholar] [PubMed]
- Gandini, D.; Brimacombe, J. Manikin training for neonatal resuscitation with the laryngeal mask airway. Paediatr. Anaesth. 2004, 14, 493–494. [Google Scholar] [CrossRef]
- Belkhatir, K.; Scrivens, A.; O’Shea, J.E.; Roehr, C.C. Experience and training in endotracheal intubation and laryngeal mask airway use in neonates: Results of a national survey. Arch. Dis. Child. Fetal Neonatal Ed. 2021, 106, 223–224. [Google Scholar] [CrossRef] [PubMed]
- Konrad, C.; Schüpfer, G.; Wietlisbach, M.; Gerber, H. Learning manual skills in anesthesiology: Is there a recommended number of cases for anesthetic procedures? Anesth. Analg. 1998, 86, 635–639. [Google Scholar] [CrossRef] [PubMed]
- Wood, F.; Morley, C.J.; Dawson, J.A.; Kamlin, C.O.F.; Owen, L.S.; Donath, S.; Davis, P.G. Assessing the effectiveness of two round neonatal resuscitation masks: Study 1. Arch. Dis. Child. Fetal Neonatal Ed. 2008, 93, F235–F237. [Google Scholar] [CrossRef]
- O’Currain, E.; Davis, P.G.; Thio, M. Educational Perspectives: Toward More Effective Neonatal Resuscitation: Assessing and Improving Clinical Skills. Neoreviews 2019, 20, e248–e257. [Google Scholar] [CrossRef]
- Wood, F.E.; Morley, C.J.; Dawson, J.A.; Davis, P.G. A respiratory function monitor improves mask ventilation. Arch. Dis. Child. Fetal Neonatal Ed. 2008, 93, F380–F381. [Google Scholar] [CrossRef]
- Trevisanuto, D.; Parotto, M.; Doglioni, N.; Ori, C.; Zanardo, V.; Micaglio, M. The Supreme Laryngeal Mask Airway (LMA): A new neonatal supraglottic device: Comparison with Classic and ProSeal LMA in a manikin. Resuscitation 2012, 83, 97–100. [Google Scholar] [CrossRef]
- Micaglio, M.; Doglioni, N.; Parotto, M.; Zanardo, V.; Ori, C.; Trevisanuto, D. Training for neonatal resuscitation with the laryngeal mask airway: A comparison of the LMA-ProSeal and the LMA-Classic in an airway management manikin. Paediatr. Anaesth. 2006, 16, 1028–1031. [Google Scholar] [CrossRef]
- Zhu, X.Y.; Lin, B.C.; Zhang, Q.S.; Ye, H.M.; Yu, R.J. A prospective evaluation of the efficacy of the laryngeal mask airway during neonatal resuscitation. Resuscitation 2011, 82, 1405–1409. [Google Scholar] [CrossRef]
- Pejovic, N.J.; Trevisanuto, D.; Lubulwa, C.; Höök, S.M.; Cavallin, F.; Byamugisha, J.; Nankunda, J.; Tylleskär, T. Neonatal resuscitation using a laryngeal mask airway: A randomised trial in Uganda. Arch. Dis. Child. 2018, 103, 255–260. [Google Scholar] [CrossRef]
- Mathai, S.S.; Adhikari, K.; Joy, A. Laryngeal Mask Airway as Primary Mode in Neonatal Resuscitation–Does it Reduce Need of Positive Pressure Ventilation? Pediatr. Res. Int. J. 2014, 2014, 216089. [Google Scholar] [CrossRef]
- Trevisanuto, D.; Micaglio, M.; Pitton, M.; Magarotto, M.; Piva, D.; Zanardo, V. Laryngeal mask airway: Is the management of neonates requiring positive pressure ventilation at birth changing? Resuscitation 2004, 62, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Zanardo, V.; Weiner, G.; Micaglio, M.; Doglioni, N.; Buzzacchero, R.; Trevisanuto, D. Delivery room resuscitation of near-term infants: Role of the laryngeal mask airway. Resuscitation 2010, 81, 327–330. [Google Scholar] [CrossRef] [PubMed]
- Brimacombe, J.; Gandini, D. Airway rescue and drug delivery in an 800 g neonate with the laryngeal mask airway. Paediatr. Anaesth. 1999, 9, 178. [Google Scholar] [CrossRef] [PubMed]
- Esmail, N.; Saleh, M.; Ali, A. Laryngeal mask airway versus endotracheal intubation for Apgar score improvement in neonatal resuscitation. Egypt J. Anesthesiol. 2002, 18, 115–121. [Google Scholar]
- Yang, C.; Zhu, X.; Lin, W.; Zhang, Q.; Su, J.; Lin, B.; Ye, H.; Xiaoyu, Z. Randomized, controlled trial comparing laryngeal mask versus endotracheal intubation during neonatal resuscitation—A secondary publication. BMC Pediatr. 2016, 16, 17. [Google Scholar] [CrossRef]
- el Shimi, M.S.; Abusaif, I.; Khafagy, S. Efficacy of Laryngeal Mask Airway in Neonatal Resuscitation. Egypt J. Hosp. Med. 2018, 70, 1767–1772. [Google Scholar] [CrossRef]
- Zanardo, V.; Simbi, A.K.; Savio, V.; Micaglio, M.; Trevisanuto, D. Neonatal resuscitation by laryngeal mask airway after elective cesarean section. Fetal Diagn. Ther. 2004, 19, 228–231. [Google Scholar] [CrossRef] [PubMed]
- Foglia, E.E.; Ades, A.; Napolitano, N.; Leffelman, J.; Nadkarni, V.; Nishisaki, A. Factors Associated with Adverse Events during Tracheal Intubation in the NICU. Neonatology 2015, 108, 23–29. [Google Scholar] [CrossRef]
- Drake-Brockman, T.F.; Ramgolam, A.; Zhang, G.; Hall, G.L.; von Ungern-Sternberg, B.S. The effect of endotracheal tubes versus laryngeal mask airways on perioperative respiratory adverse events in infants: A randomised controlled trial. Lancet 2017, 389, 701–708. [Google Scholar] [CrossRef]
- Mizumoto, H.; Motokura, K.; Kurosaki, A.; Hata, D. Introduction of laryngeal mask airway in Japan, and its rescue use for newborns. Pediatr. Int. 2018, 60, 954–956. [Google Scholar] [CrossRef] [PubMed]
- Reinhart, D.J.; Simmons, G. Comparison of Placement of the Laryngeal Mask Airway With Endotracheal Tube by Paramedics and Respiratory Therapists. Ann. Emerg. Med. 1994, 24, 260–263. [Google Scholar] [CrossRef]
- Ye, Q.; Wu, D.; Fang, W.; Wong, G.T.C.; Lu, Y. Comparison of gastric insufflation using LMA-supreme and I-gel versus tracheal intubation in laparoscopic gynecological surgery by ultrasound: A randomized observational trial. BMC Anesthesiol. 2020, 20, 136. [Google Scholar] [CrossRef] [PubMed]
- Dodd, K.W.; Strobel, A.M.; Driver, B.E.; Reardon, R.F. Use of a Supraglottic Airway to Relieve Ventilation-Impeding Gastric Insufflation During Emergency Airway Management in an Infant. Ann. Emerg. Med. 2016, 68, 452–455. [Google Scholar] [CrossRef]
- Schwindt, J.; Schwindt, E.; Grass, B.; Schäfer, S.; Kreth, U.; Hoffmann, F. Intubation in neonatal resuscitation—Compelling necessity or incalculable risk? Resuscitation 2021, 165, 188–189. [Google Scholar] [CrossRef]
- Madar, J.; Roehr, C.C.; Ainsworth, S.; Ersdal, H.; Morley, C.; Rüdiger, M.; Skåre, C.; Szczapa, T.; Pas, A.T.; Trevisanuto, D.; et al. European Resuscitation Council Guidelines 2021: Newborn resuscitation and support of transition of infants at birth. Resuscitation 2021, 161, 291–326. [Google Scholar] [CrossRef]
- Roehr, C.C.; Madar, J.; Morley, C.J.; Ainsworth, S.; Ersdal, H.; Rüdiger, M.; Skåre, C.; Szczapa, T.; Pas, A.T.; Trevisanuto, D.; et al. Reply letter to: Intubation in neonatal resuscitation—Compelling necessity or incalculable risk? Resuscitation 2021, 165, 190–191. [Google Scholar] [CrossRef]
- Wang, H.E.; Schmicker, R.H.; Daya, M.R.; Stephens, S.W.; Idris, A.H.; Carlson, J.N.; Colella, M.R.; Herren, H.; Hansen, M.; Richmond, N.J.; et al. Effect of a Strategy of Initial Laryngeal Tube Insertion vs. Endotracheal Intubation on 72-Hour Survival in Adults with Out-of-Hospital Cardiac Arrest: A Randomized Clinical Trial. JAMA 2018, 320, 769–778. [Google Scholar] [CrossRef]
- Benger, J.R.; Kirby, K.; Black, S.; Brett, S.J.; Clout, M.; Lazaroo, M.J.; Nolan, J.P.; Reeves, B.C.; Robinson, M.; Scott, L.J.; et al. Effect of a Strategy of a Supraglottic Airway Device vs. Tracheal Intubation During Out-of-Hospital Cardiac Arrest on Functional Outcome: The AIRWAYS-2 Randomized Clinical Trial. JAMA 2018, 320, 779–791. [Google Scholar] [CrossRef]
- Wang, H.E.; Benger, J.R. Endotracheal intubation during out-of-hospital cardiac arrest: New insights from recent clinical trials. J. Am. Coll. Emerg. Physicians Open 2020, 1, 24–29. [Google Scholar] [CrossRef]
- Mani, S.; Gugino, S.; Helman, J.; Bawa, M.; Nair, J.; Chandrasekharan, P.; Rawat, M. Laryngeal mask ventilation with chest compression during neonatal resuscitation: Randomized, non-inferiority trial in lambs. Pediatr. Res. Available online: https://www.nature.com/articles/s41390-021-01820-z (accessed on 3 November 2021). [CrossRef]
- Halling, C.; Raymond, T.; Brown, L.S.; Ades, A.; Foglia, E.E.; Allen, E.; Wyckoff, M.H.; Guerguerian, A.-M.; Atkins, D.; Fink, E.; et al. Neonatal delivery room CPR: An analysis of the Get with the Guidelines®-Resuscitation Registry. Resuscitation 2021, 158, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Vali, P.; Laskminrusimha, S. Laryngeal mask airway: An alternate option for all phases of neonatal resuscitation. Pediatr. Res. Available online: https://www.nature.com/articles/s41390-021-01917-5 (accessed on 20 December 2021). [CrossRef]
- McKinsey, S.; Perlman, J.M. Resuscitative interventions during simulated asystole deviate from the recommended timeline. Arch. Dis. Child. Fetal Neonatal Ed. 2016, 101, F244–F247. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.T.; Lin, H.J.; Jeng, H.W.; Lin, C.C.; Guo, H.R. The pharmacological effect of epinephrine administration via laryngeal mask airway in a porcine model of asphyxial cardiac arrest. Emerg. Med. J. 2008, 25, 722–724. [Google Scholar] [CrossRef]
- Liao, C.-K.; Lin, H.-J.; Foo, N.-P.; Lin, C.-C.; Guo, H.-R.; Chen, K.-T. Epinephrine administration via a laryngeal mask airway: What is the optimal dose? Signa Vitae 2010, 5, 25–28. [Google Scholar]
- Attridge, J.T.; Stewart, C.; Stukenborg, G.J.; Kattwinkel, J. Administration of rescue surfactant by laryngeal mask airway: Lessons from a pilot trial. Am. J. Perinatol. 2013, 30, 201–206. [Google Scholar]
- Sadeghnia, A.; Tanhaei, M.; Mohammadizadeh, M.; Nemati, M. A comparison of surfactant administration through i-gel and ET-tube in the treatment of respiratory distress syndrome in newborns weighing more than 2000 grams. Adv. Biomed Res. 2014, 3, 160. [Google Scholar] [CrossRef]
- Barbosa, R.F.; Simões, E.S.A.C.; Silva, Y.P. A randomized controlled trial of the laryngeal mask airway for surfactant administration in neonates. J. Pediatr. 2017, 93, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Roberts, K.D.; Brown, R.; Lampland, A.L.; Leone, T.A.; Rudser, K.D.; Finer, N.N.; Rich, W.D.; Merritt, T.A.; Czynski, A.J.; Kessel, J.M.; et al. Laryngeal Mask Airway for Surfactant Administration in Neonates: A Randomized, Controlled Trial. J. Pediatr. 2018, 193, 40–46.e1. [Google Scholar] [CrossRef] [PubMed]
- Amini, E.; Sheikh, M.; Shariat, M.; Dalili, H.; Azadi, N.; Nourollahi, S. Surfactant Administration in Preterm Neonates Using Laryngeal Mask Airway: A Randomized Clinical Trial. Acta Med. Iran. 2019, 57, 348–354. [Google Scholar] [CrossRef]
- Gharehbaghi, M.; Moghaddam, Y.J.; Radfar, R. Comparing the Efficacy of Surfactant Administration by Laryngeal Mask Airway and Endotracheal Intubation in Neonatal Respiratory Distress Syndrome. Crescent J. Med. Biol. Sci. 2018, 5, 222–227. [Google Scholar]
- Gallup, J.A.; MBPinheiro, J.; Ndakor, S.M.; Pezzano, C. Randomized Trial of Surfactant Therapy via Laryngeal Mask Airway vs. Brief Tracheal Intubation. Pediatrics 2021, 147, 755–756. [Google Scholar]
- Roberts, C.T.; Manley, B.J.; O’Shea, J.E.; Stark, M.; Andersen, C.; Davis, P.G.; Buckmaster, A. Supraglottic airway devices for administration of surfactant to newborn infants with respiratory distress syndrome: A narrative review. Arch. Dis. Child.-Fetal Neonatal Ed. 2021, 106, 336–341. [Google Scholar] [CrossRef]
- Trevisanuto, D.; Grazzina, N.; Ferrarese, P.; Micaglio, M.; Verghese, C.; Zanardo, V. Laryngeal mask airway used as a delivery conduit for the administration of surfactant to preterm infants with respiratory distress syndrome. Biol. Neonate 2005, 87, 217–220. [Google Scholar] [CrossRef]
- Trevisanuto, D.; Parotto, M.; Doglioni, N.; Zanardo, V.; Micaglio, M. Upper esophageal lesion following laryngeal mask airway resuscitation in a very low birth weight infant. Resuscitation 2011, 82, 1251–1252. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.T.; Wang, J.N.; Tai, Y.T.; Tsai, T.Y.; Wu, J.M. Successful management of a neonate with Pierre-Robin syndrome and severe upper airway obstruction by long term placement of a laryngeal mask airway. Resuscitation 2004, 61, 97–99. [Google Scholar] [CrossRef]
- Baraka, A. Laryngeal Mask Airway for Resuscitation of a Newborn with Pierre-Robin Syndrome. Anesthesiology 1995, 83, 646–647. [Google Scholar] [CrossRef]
- Leal-Pavey, Y.R. Use of the LMA classic to secure the airway of a premature neonate with Smith-Lemli-Opitz syndrome: A case report. AANA J. 2004, 72, 427–430. [Google Scholar]
- Bucx, M.J.; Grolman, W.; Kruisinga, F.H.; Lindeboom, J.A.; Van Kempen, A.A. The prolonged use of the laryngeal mask airway in a neonate with airway obstruction and Treacher Collins syndrome. Paediatr. Anaesth. 2003, 13, 530–533. [Google Scholar] [CrossRef] [PubMed]
- Galderisi, A.; De Bernardo, G.; Lorenzon, E.; Trevisanuto, D. i-gel: A new supraglottic device for effective resuscitation of a very low birthweight infant with Cornelia de Lange syndrome. BMJ Case Rep. 2015, 2015, bcr2014209124. [Google Scholar] [CrossRef]
- Jöhr, M.; Berger, T.M.; Ruppen, W.; Schlegel, C. Congenital laryngotracheo-oesophageal cleft: Successful ventilation with the Laryngeal Mask Airway. Paediatr. Anaesth. 2003, 13, 68–71. [Google Scholar] [CrossRef]
- Carenzi, B.; Corso, R.; Stellino, V.; Carlino, G.; Tonini, C.; Rossini, L.; Gentili, G. Airway management in an infant with congenital centrofacial dysgenesia. Br. J. Anaesth. 2002, 88, 726–728. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gunnarsson, B.; Smárason, A.K.; Skogvoll, E.; Fasting, S. Characteristics and outcome of unplanned out-of-institution births in Norway from 1999 to 2013: A cross-sectional study. Acta Obstet. Gynecol. Scand. 2014, 93, 1003–1010. [Google Scholar] [CrossRef]
- Javaudin, F.; Hamel, V.; Legrand, A.; Goddet, S.; Templier, F.; Potiron, C.; Pes, P.; Bagou, G.; Montassier, E. Unplanned out-of-hospital birth and risk factors of adverse perinatal outcome: Findings from a prospective cohort. Scand. J. Trauma Resusc. Emerg. Med. 2019, 27, 26. [Google Scholar] [CrossRef] [PubMed]
- Huynh, T.; Bahr, N.; Harrod, T.; Guise, J.-M. When Seconds Matter: Neonatal Resuscitation in the Prehospital Setting. Pediatrics 2020, 146, 372. [Google Scholar]
- Berry, A.M.; Brimacombe, J.R.; Verghese, C. The laryngeal mask airway in emergency medicine, neonatal resuscitation, and intensive care medicine. Int. Anesthesiol. Clin. 1998, 36, 91–109. [Google Scholar] [CrossRef]
- Garces, A.; McClure, E.; Espinoza, L.; Saleem, S.; Figueroa, L.; Bucher, S.; Goldenberg, R.L. Traditional birth attendants and birth outcomes in low-middle income countries: A review. Semin. Perinatol. 2019, 43, 247–251. [Google Scholar] [CrossRef]
- Reisman, J.; Arlington, L.; Jensen, L.; Louis, H.; Suarez-Rebling, D.; Nelson, B.D. Newborn Resuscitation Training in Resource-Limited Settings: A Systematic Literature Review. Pediatrics 2016, 138, e20154490. [Google Scholar] [CrossRef]
- Niermeyer, S.; Robertson, N.J.; Ersdal, H.L. Beyond basic resuscitation: What are the next steps to improve the outcomes of resuscitation at birth when resources are limited? Semin. Fetal Neonatal Med. 2018, 23, 361–368. [Google Scholar] [CrossRef] [PubMed]

| Study | Population | Sample Size (n) | Intervention | Comparator | Primary Outcome (Definition) |
|---|---|---|---|---|---|
| Randomized controlled trials | |||||
| Singh et al. (2005) [17] | GA > 34 w BW > 1500 g | 25 V 25 | LMA Classic | FM | Success of ventilation (Chest expansion and bilateral breath sounds) (96% vs. 88%) |
| Feroze et al. (2008) [18] | BW > 1500 g | 25 V 25 | LMA Classic | FM | Success of resuscitation (Not clearly defined) (96% vs. 80%) |
| Zhu et al. (2011) [52] | GA ≥ 34 w BW ≥ 2 kg | 205 V 164 | LMA Classic | FM | Success of resuscitation (Prevention of need for tracheal intubation) (99% vs. 84.1%) |
| Mathai et al. (2014) [54] | GA > 36 w BW > 2 kg | 32 V 35 | LMA Classic | FM | Duration of PPV until spontaneous breathing 95.31 s (23.22 s) vs. 180.86 s (37.83 s) (p = 0.024) |
| Trevisanuto et al. (2015) [19] | GA ≥ 34 w BW ≥ 1500 g | 71 V 71 | LMA Supreme | FM | Success of resuscitation (Prevention of need for endotracheal intubation) (91.5% vs. 78.9%) |
| Pejovic et al. (2018) [53] | GA > 34 w BW > 2 kg | 25 V 25 | i-gel | FM | Time to spontaneous breathing (153 s (59) vs. 216 s (92)) |
| Pejovic et al. (2020) [32] | GA > 34 w BW > 2 kg | 563 V 591 | i-gel | FM | Death or moderate- severe HIE (27.4% vs. 24.4%) |
| Observational studies | |||||
| Cohort studies | |||||
| Trevisanuto et al. (2004) [55] | GA ≥ 34 w BW ≥ 2 kg | 74 V 74 | LMA Classic | FM | Need for tracheal intubation—no difference |
| Zanardo et al. (2010) [56] | 34w–36 w 7 d | 36 V 34 | LMA Classic | FM | Admission to NICU [OR—0.30 (0.10–0.89)] and length of hospitalization decreased with LM |
| Case series | |||||
| Paterson et al. (1994) [16] | GA 35–41 w | 20 | LMA Classic | - | Success of resuscitation—100% (Improvement in APGAR score) |
| Gandini et al. (1999) [57] | GA 28–42 w BW 1–4.7 Kg | 104 | LMA Classic | - | Success of resuscitation—99% (Improvement in APGAR score) |
| Study | Population | Sample Size (n) | Intervention | Comparator | Primary Outcome (Definition) |
|---|---|---|---|---|---|
| Randomized controlled trials | |||||
| Esmail et al. (2002) [58] | GA ≥ 35 w BW ≥ 2.5 kg | 20 V 20 | LMA Classic | ETT | Success of resuscitation (Improvement in APGAR score) (100% in both groups) |
| Feroze et al. (2008) [18] | BW >1500 g | 25 V 25 | LMA Classic | ETT | Success of resuscitation (Improvement in APGAR score) (96% vs. 90%) |
| Yang et al. (2016) [59] | GA ≥ 34 w BW ≥ 2 kg | 36 V 32 | LMA Classic | ETT | Success of resuscitation (Establishment of spontaneous breathing, HR > 100, good muscle tone) (86% vs. 97%) p = 0.20 |
| El-Shimi et al. (2018) [60] | GA ≥ 34 w BW ≥ 2 kg | 40 V 40 | LMA Classic | ETT | Need for ETT insertion in LMA group (0%) Success of resuscitation (Improvement in APGAR score at 5 min) (100% vs. 100%) |
| Observational studies | |||||
| Cohort study | |||||
| Zanardo et al. (2010) [56] | 34 w–36 w 7 d | 36 V 16 | LMA Classic | ETT | Admission to NICU [OR—0.08 (0.02–0.33)] and length of hospitalization decreased with LM |
| Case-Control study | |||||
| Zanardo et al. (2004) [61] (Secondary comparison) | GA > 37 w | 43 V 18 | LM (Mode of delivery – cesarean section + vaginal) | ETT (Mode of delivery – cesarean section + vaginal) | Success of resuscitation with LM—97.6% Need for NICU admission and mechanical ventilation—decreased in LM group |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mani, S.; Pinheiro, J.M.B.; Rawat, M. Laryngeal Masks in Neonatal Resuscitation—A Narrative Review of Updates 2022. Children 2022, 9, 733. https://doi.org/10.3390/children9050733
Mani S, Pinheiro JMB, Rawat M. Laryngeal Masks in Neonatal Resuscitation—A Narrative Review of Updates 2022. Children. 2022; 9(5):733. https://doi.org/10.3390/children9050733
Chicago/Turabian StyleMani, Srinivasan, Joaquim M. B. Pinheiro, and Munmun Rawat. 2022. "Laryngeal Masks in Neonatal Resuscitation—A Narrative Review of Updates 2022" Children 9, no. 5: 733. https://doi.org/10.3390/children9050733
APA StyleMani, S., Pinheiro, J. M. B., & Rawat, M. (2022). Laryngeal Masks in Neonatal Resuscitation—A Narrative Review of Updates 2022. Children, 9(5), 733. https://doi.org/10.3390/children9050733







