Diagnostic Accuracy of Acid-Base Status in Infants with Hypertrophic Pyloric Stenosis
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Study Design
2.3. Diagnostic Procedures
2.4. Outcome Measures
2.5. Statistical Analysis
3. Results
3.1. Pylorostenosis Group (Q40.0)
3.2. Control Group (R11.0)
3.3. Differences between Groups
3.4. Differences in Acid-Base Status between Groups
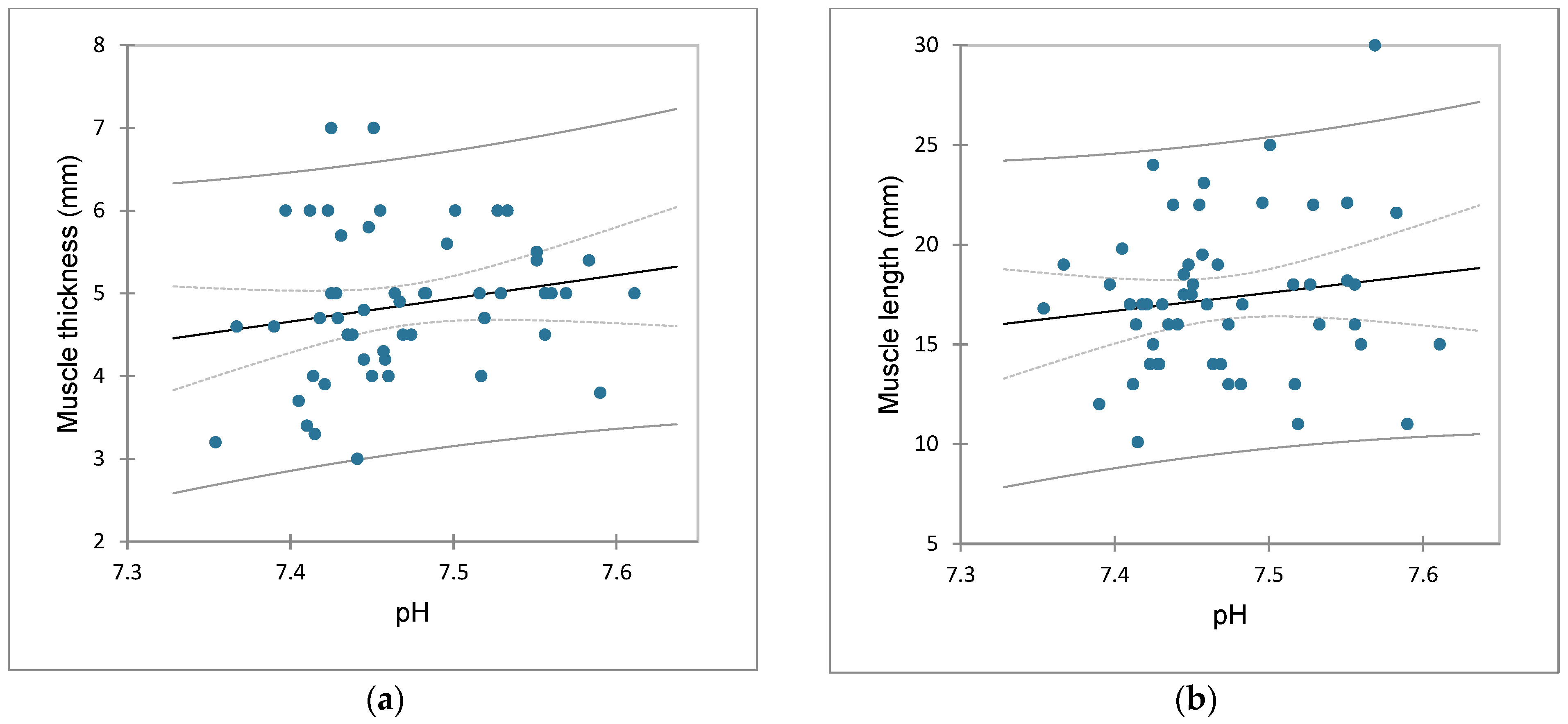
3.5. Correlation between Thickness and Length of Pyloric Muscle and Values of Acid-Base Status
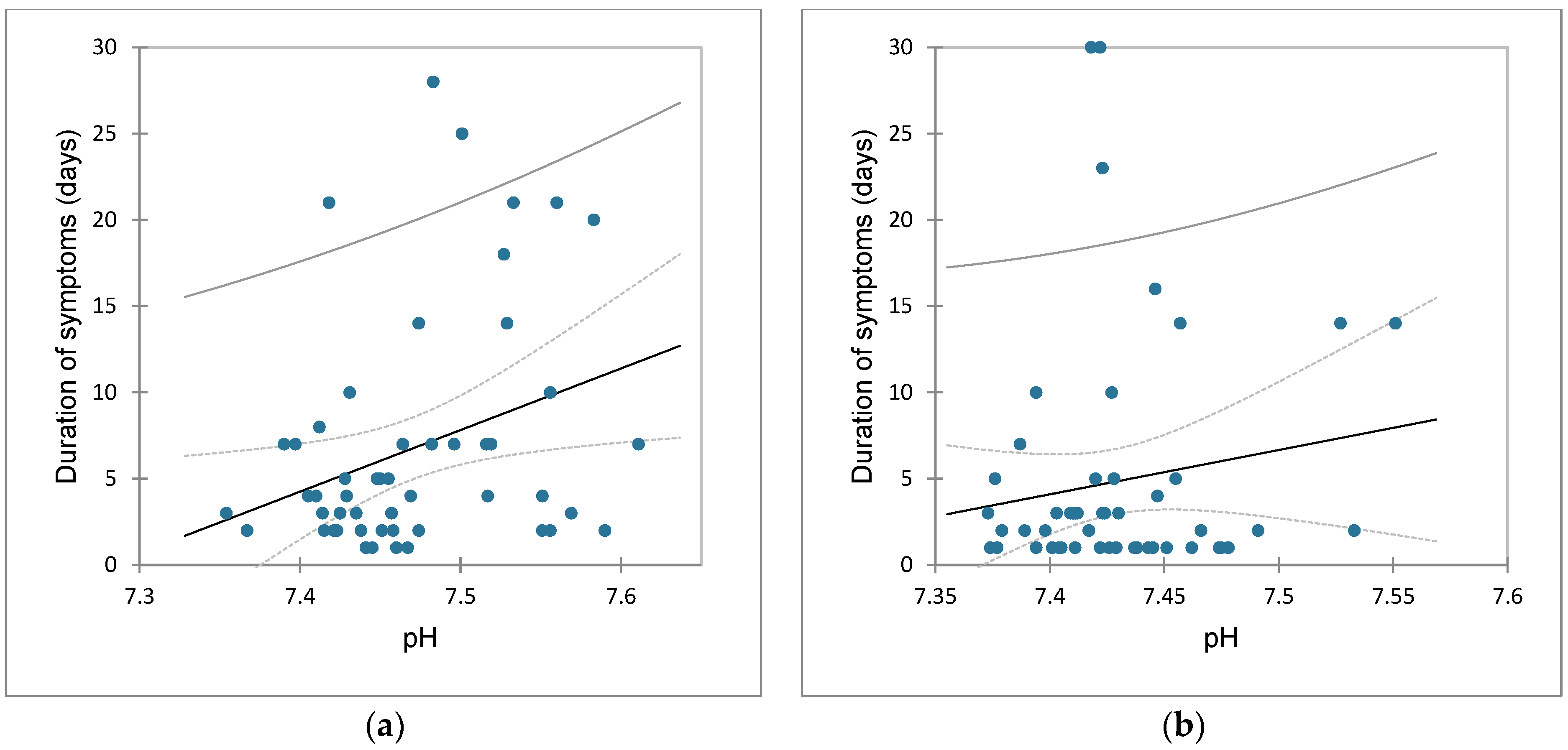
3.6. Correlation between Duration of Vomiting and Acid-Base Status
3.7. Influence of Duration of Vomiting Longer and Shorter than Three Days on Acid-Base Status
4. Discussion
Limitations of Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kopač, M. Evaluation and Treatment of Alkalosis in Children. J. Pediatr. Intensive Care 2019, 8, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Shaw, A.M. Bicarbonate and chloride equilibrium and acid-base balance in the neonate. Neonatal Netw. 2008, 27, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.E. Guyton and Hall Textbook of Medical Physiology, 13th ed.; W B Saunders: Philadelphia, PA, USA, 2015. [Google Scholar]
- Bašković, M.; Župančić, B.; Lesjak, N.; Vukasović, I. Hypertrophic pyloric stenosis—Five-year retrospective analysis. Acta Med. Croat. 2016, 70, 103–106. [Google Scholar]
- Pogorelić, Z.; Čagalj, I.Č.; Žitko, V.; Nevešćanin, A.; Krželj, V. Late-Onset Hypertrophic Pyloric Stenosis in a 14-Weeks-Old Full Term Male Infant. Acta Med. 2019, 62, 82–84. [Google Scholar] [CrossRef] [PubMed][Green Version]
- El-Gohary, Y.; Yeap, B.H.; Hempel, G.; Gillick, J. A 9-year single-center experience with circumumbilical Ramstedt’s pyloromyotomy. Eur. J. Pediatr. Surg. 2010, 20, 387–390. [Google Scholar] [CrossRef] [PubMed]
- To, T.; Wajja, A.; Wales, P.W.; Langer, J.C. Population demographic indicators associated with incidence of pyloric stenosis. Arch. Pediatr. Adolesc. Med. 2005, 159, 520–525. [Google Scholar] [CrossRef] [PubMed]
- Ames, M.D. Gastric acidity in the first ten days of life of the prematurely born baby. Am. J. Dis. Child. 1960, 100, 252–256. [Google Scholar] [CrossRef]
- Krogh, C.; Biggar, R.J.; Fischer, T.K.; Lindholm, M.; Wohlfahrt, J.; Melbye, M. Bottle-feeding and the Risk of Pyloric Stenosis. Pediatrics 2012, 130, e943–e949. [Google Scholar] [CrossRef]
- Zhu, J.; Zhu, T.; Lin, Z.; Qu, Y.; Mu, D. Perinatal risk factors for infantile hypertrophic pyloric stenosis: A meta-analysis. J. Pediatr. Surg. 2017, 52, 1389–1397. [Google Scholar] [CrossRef] [PubMed]
- Piotrowska, A.P.; Solari, V.; Puri, P. Distribution of heme oxygenase-2 in nerves and interstitial cells of Cajal in the normal pylorus and in infantile hypertrophic pyloric stenosis. Arch. Pathol. Lab. Med. 2003, 127, 1182–1186. [Google Scholar] [CrossRef]
- Oue, T.; Puri, P. Smooth muscle cell hypertrophy versus hyperplasia in infantile hypertrophic pyloric stenosis. Pediatr. Res. 1999, 45, 853–857. [Google Scholar] [CrossRef] [PubMed]
- Aspelund, G.; Langer, J.C. Current management of hypertrophic pyloric stenosis. Semin. Pediatr. Surg. 2007, 16, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Gale, H.I.; Gee, M.S.; Westra, S.J.; Nimkin, K. Abdominal ultrasonography of the pediatric gastrointestinal tract. World J. Radiol. 2016, 8, 656–667. [Google Scholar] [CrossRef] [PubMed]
- Kamata, M.; Cartabuke, R.S.; Tobias, J.D. Perioperative care of infants with pyloric stenosis. Paediatr. Anaesth. 2015, 25, 1193–1206. [Google Scholar] [CrossRef]
- St. Peter, S.D.; Tsao, K.; Sharp, S.W.; Holcomb, G.W. 3rd; Ostlie, D.J. Predictors of emesis and time to goal intake after pyloromyotomy: Analysis from a prospective trial. J. Pediatr. Surg. 2008, 43, 2038–2041. [CrossRef] [PubMed][Green Version]
- Jobson, M.; Hall, N.J. Contemporary management of pyloric stenosis. Semin. Pediatr. Surg. 2016, 25, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Alberti, D.; Cheli, M.; Locatelli, G. A new technical variant for extramucosal pyloromyotomy: The Tan-Bianchi operation moves to the right. J. Pediatr. Surg. 2004, 39, 53–56. [Google Scholar] [CrossRef]
- Alain, J.L.; Grousseau, D.; Terrier, G. Extramucosal pylorotomy by laparoscopy. J. Pediatr. Surg. 1991, 26, 1191–1192. [Google Scholar] [CrossRef]
- Oomen, M.W.; Hoekstra, L.T.; Bakx, R.; Ubbink, D.T.; Heij, H.A. Open versus laparoscopic pyloromyotomy for hypertrophic pyloric stenosis: A systematic review and meta-analysis focusing on major complications. Surg. Endosc. 2012, 26, 2104–2110. [Google Scholar] [CrossRef]
- Pogorelić, Z.; Zelić, A.; Jukić, M.; Muñoz, C.M.L. The Safety and Effectiveness of Laparoscopic Pyloromyotomy Using 3-mm Electrocautery Hook versus Open Surgery for Treatment of Hypertrophic Pyloric Stenosis in Infants. Children 2021, 8, 701. [Google Scholar] [CrossRef]
- Mahida, J.B.; Asti, L.; Deans, K.J.; Minneci, P.C.; Groner, J.I. Laparoscopic pyloromyotomy decreases postoperative length of stay in children with hypertrophic pyloric stenosis. J. Pediatr. Surg. 2016, 51, 1436–1439. [Google Scholar] [CrossRef] [PubMed]
- White, J.S.; Clements, W.D.; Heggarty, P.; Sidhu, S.; Mackle, E.; Stirling, I. Treatment of infantile hypertrophic pyloric stenosis in a district general hospital: A review of 160 cases. J. Pediatr. Surg. 2003, 38, 1333–1336. [Google Scholar] [CrossRef]
- Al-Jazaeri, A.; Al-Shehri, A.; Zamakhshary, M.; Al-Zahem, A. Can the duration of vomiting predict postoperative outcomes in hypertrophic pyloric stenosis? Ann. Saudi Med. 2011, 31, 609–612. [Google Scholar] [CrossRef] [PubMed]
- Shanbhogue, L.K.; Sikdar, T.; Jackson, M.; Lloyd, D.A. Serum electrolytes and capillary blood gases in the management of hypertrophic pyloric stenosis. Br. J. Surg. 1992, 79, 251–253. [Google Scholar] [CrossRef]
- Beasley, S.W.; Hudson, I.; Yuen, H.P.; Jones, P.G. Influence of age, sex, duration of symptoms and dehydration of serum electrolytes in hypertrophic pyloric stenosis. Aust. Paediatr. J. 1986, 22, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Goh, D.W.; Hall, S.K.; Gornall, P.; Buick, R.G.; Green, A.; Corkery, J.J. Plasma chloride and alkalaemia in pyloric stenosis. Br. J. Surg. 1990, 77, 922–923. [Google Scholar] [CrossRef] [PubMed]
- Tutay, G.J.; Capraro, G.; Spirko, B.; Garb, J.; Smithline, H. Electrolyte profile of pediatric patients with hypertrophic pyloric stenosis. Pediatr. Emerg. Care 2013, 29, 465–468. [Google Scholar] [CrossRef]
- Wilkinson, D.J.; Chapman, R.A.; Owen, A.; Olpin, S.; Marven, S.S. Hypertrophic pyloric stenosis: Predicting the resolution of biochemical abnormalities. Pediatr. Surg. Int. 2011, 27, 695–698. [Google Scholar] [CrossRef]
- Oakley, E.A.; Barnett, P.L. Is acid base determination an accurate predictor of pyloric stenosis? J. Paediatr. Child Health 2000, 36, 587–589. [Google Scholar] [CrossRef]
- Johnson, P.R.V. Infantile hypertrophic pyloric stenosis. Surgery 2004, 22, 212–215. [Google Scholar] [CrossRef]
- Niedzielski, J.; Kobielski, A.; Sokal, J.; Krakós, M. Accuracy of sonographic criteria in the decision for surgical treatment in infantile hypertrophic pyloric stenosis. Arch. Med. Sci. 2011, 7, 508–511. [Google Scholar] [CrossRef] [PubMed]
- Godbole, P.; Sprigg, A.; Dickson, J.A.; Lin, P.C. Ultrasound compared with clinical examination in infantile hypertrophic pyloric stenosis. Arch. Dis. Child. 1996, 75, 335–337. [Google Scholar] [CrossRef] [PubMed]
- Conn, A.W. Anaesthesia for pyloromyotomy in infancy. Can. Anaesth. Soc. J. 1963, 10, 18–29. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Steven, I.M.; Allen, T.H.; Sweeney, D.B. Congenital hypertrophic pyloric stenosis: The anaesthetist’s view. Anaesth. Intensive Care 1973, 1, 544–546. [Google Scholar] [CrossRef] [PubMed]
- Van der Schouw, Y.T.; van der Velden, M.T.; Hitge-Boetes, C.; Verbeek, A.L.; Ruijs, S.H. Diagnosis of hypertrophic pyloric stenosis: Value of sonography when used in conjunction with clinical findings and laboratory data. Am. J. Roentgenol. 1994, 163, 905–909. [Google Scholar] [CrossRef]
- Janik, J.S.; Wayne, E.R.; Janik, J.P. Pyloric stenosis in premature infants. Arch. Pediatr. Adolesc. Med. 1996, 150, 223–224. [Google Scholar] [CrossRef]
| Variable | Pylorostenosis Group (Q40.0) | Control Group (R11.0) | p-Value |
|---|---|---|---|
| Sex (male, female) | 41, 12 | 41, 12 | 1.000 |
| Age (days) | 31 (24–44) | 35 (21–50) | 0.674 |
| Duration of symptoms (days) | 4 (2–7) | 2 (1–5) | 0.002 * |
| Family history | 1 | 0 | <0.0001 * |
| Birth (preterm, term, post-term) | 5, 48, 0 | 6, 47, 0 | 0.563 |
| Birth weight (grams) | 3506 (±558) | 3428 (±525) | 0.464 |
| Weight during examination (grams) | 3880 (±601) | 4439 (±1025) | 0.001 * |
| Difference between birth weight and weight during examination (grams) | 374 (±568) | 1010 (±858) | <0.0001 * |
| Type of vomiting (explosive, regurgitation) | 45, 8 | 22, 31 | 0.023 * |
| Variable | Pylorostenosis Group (Q40.0) | Control Group (R11.0) | p-Value | Reference Interval |
|---|---|---|---|---|
| pH | 7.457 (7.425–7.517) | 7.422 (7.403–7.446) | <0.0001 * | 7.350–7.450 |
| pCO2 (kPa) | 4.49 (4.16–5.10) | 4.70 (4.22–5.10) | 0.604 | 3.60–5.50 |
| pO2 (kPa) | 8 (7.2–8.7) | 8.1 (7.1–8.8) | 0.867 | 4.8–10.6 |
| HCO3 (mmol/L) | 25 (23–27) | 23 (22–24) | 0.000 * | 19–24 |
| Total CO2 (mmol/L) | 25 (23–28) | 24 (22–25) | 0.011 * | 22–29 |
| BE (mmol/L) | 0.8 (−1.2–2.9) | −1.3 (−2.7–−0.2) | 0.000 * | −7–−1 |
| sO2 (%) | 95.1 (93.3–96.0) | 94.1 (91.3–95.7) | 0.162 | 94–98 |
| Potassium (mmol/L) | 5 (4.2–5.6) | 5.3 (5–5.9) | 0.006 * | 3–7 |
| Sodium (mmol/L) | 137 (136–138) | 137 (136–138) | 0.278 | 134–142 |
| Chloride (mmol/L) | 103 (100–105) | 104 (103–106) | 0.071 | 96–111 |
| Ionized Calcium (mmol/L) | 1.28 (1.25–1.31) | 1.31 (1.27–1.34) | 0.011 * | 0.95–1.50 |
| Glucose (mmol/L) | 4.5 (4.1–4.8) | 4.9 (4.5–5.5) | 0.007 * | 2.8–5.5 |
| Lactate (mmol/L) | 1.98 (1.39–2.57) | 1.75 (1.52–2.21) | 0.509 | 0–1.8 |
| Total bilirubin (µmol/L) | <34 (<34–87) | <34 (<34–54) | 0.143 | <34 |
| Oxyhemoglobin (%) | 93.1 (91.5–94.4) | 91.9 (89.9–94.3) | 0.138 | 94–100 |
| Carboxyhemoglobin (%) | 0.7 (0.5–1) | 0.8 (0.4–1) | 0.682 | <1.5 |
| Methemoglobin (%) | 1 (0.9–1.1) | 1.1 (0.9–1.2) | 0.132 | <1.5 |
| Deoxyhemoglobin (%) | 4.8 (4–6.6) | 5.9 (4.2–8.5) | 0.159 | <5 |
| Variable | <3 Days (n = 23) | >3 Days (n = 30) | p-Value |
|---|---|---|---|
| pH | 7.456 (±0.059) | 7.481 (±0.059) | 0.123 |
| pCO2 (kPa) | 4.502 (±0.658) | 4.630 (±0.770) | 0.525 |
| HCO3 (mmol/L) | 24.261 (±1.839) | 26.700 (±4.419) | 0.016 * |
| Total CO2 (mmol/L) | 24.174 (±2.249) | 26.533 (±5.204) | 0.048 * |
| BE (mmol/L) | −0.161 (±2.174) | 2.307 (±4.923) | 0.030 * |
| Potassium (mmol/L) | 5.234 (±0.671) | 4.633 (±0.987) | 0.015 * |
| Sodium (mmol/L) | 137.913 (±2.466) | 137.400 (±2.027) | 0.410 |
| Chloride (mmol/L) | 103.565 (±4.620) | 101.633 (±6.636) | 0.239 |
| Variable | <3 Days (n = 38) | >3 Days (n = 15) | p-Value |
|---|---|---|---|
| pH | 7.424 (±0.036) | 7.439 (±0.047) | 0.227 |
| pCO2 (kPa) | 4.601 (±0.637) | 4.826 (±0.519) | 0.229 |
| HCO3 (mmol/L) | 23.105 (±1.737) | 24.733 (±3.432) | 0.026 * |
| Total CO2 (mmol/L) | 23.132 (±2.580) | 25.133 (±3.889) | 0.033 * |
| BE (mmol/L) | −1.668 (±2.124) | 0.267 (±3.694) | 0.020 * |
| Potassium (mmol/L) | 5.374 (±0.588) | 5.487 (±0.689) | 0.551 |
| Sodium (mmol/L) | 136.947 (±1.610) | 137.200 (±1.568) | 0.606 |
| Chloride (mmol/L) | 104.184 (±2.276) | 103.400 (±5.422) | 0.458 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bašković, M.; Sinjeri, D. Diagnostic Accuracy of Acid-Base Status in Infants with Hypertrophic Pyloric Stenosis. Children 2022, 9, 1815. https://doi.org/10.3390/children9121815
Bašković M, Sinjeri D. Diagnostic Accuracy of Acid-Base Status in Infants with Hypertrophic Pyloric Stenosis. Children. 2022; 9(12):1815. https://doi.org/10.3390/children9121815
Chicago/Turabian StyleBašković, Marko, and Dorotea Sinjeri. 2022. "Diagnostic Accuracy of Acid-Base Status in Infants with Hypertrophic Pyloric Stenosis" Children 9, no. 12: 1815. https://doi.org/10.3390/children9121815
APA StyleBašković, M., & Sinjeri, D. (2022). Diagnostic Accuracy of Acid-Base Status in Infants with Hypertrophic Pyloric Stenosis. Children, 9(12), 1815. https://doi.org/10.3390/children9121815






