The Fitter the Better? Cardiopulmonary Exercise Testing Can Predict Pulmonary Exacerbations in Cystic Fibrosis
Abstract
:1. Introduction
2. Methods
2.1. Study Design and Subjects
2.2. Spirometry
2.3. Cardiopulmonary Exercise Testing
2.4. Statistical Methods
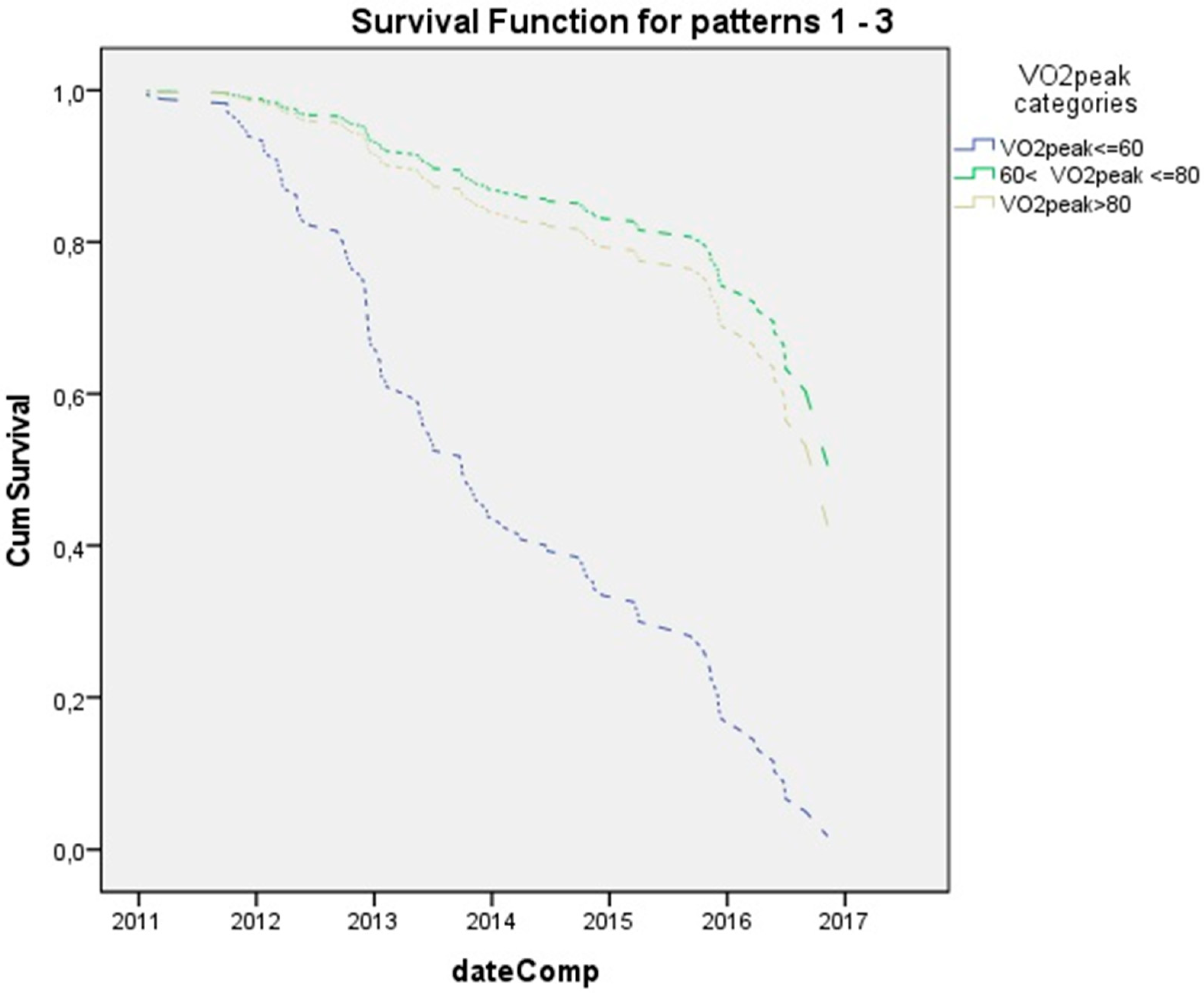
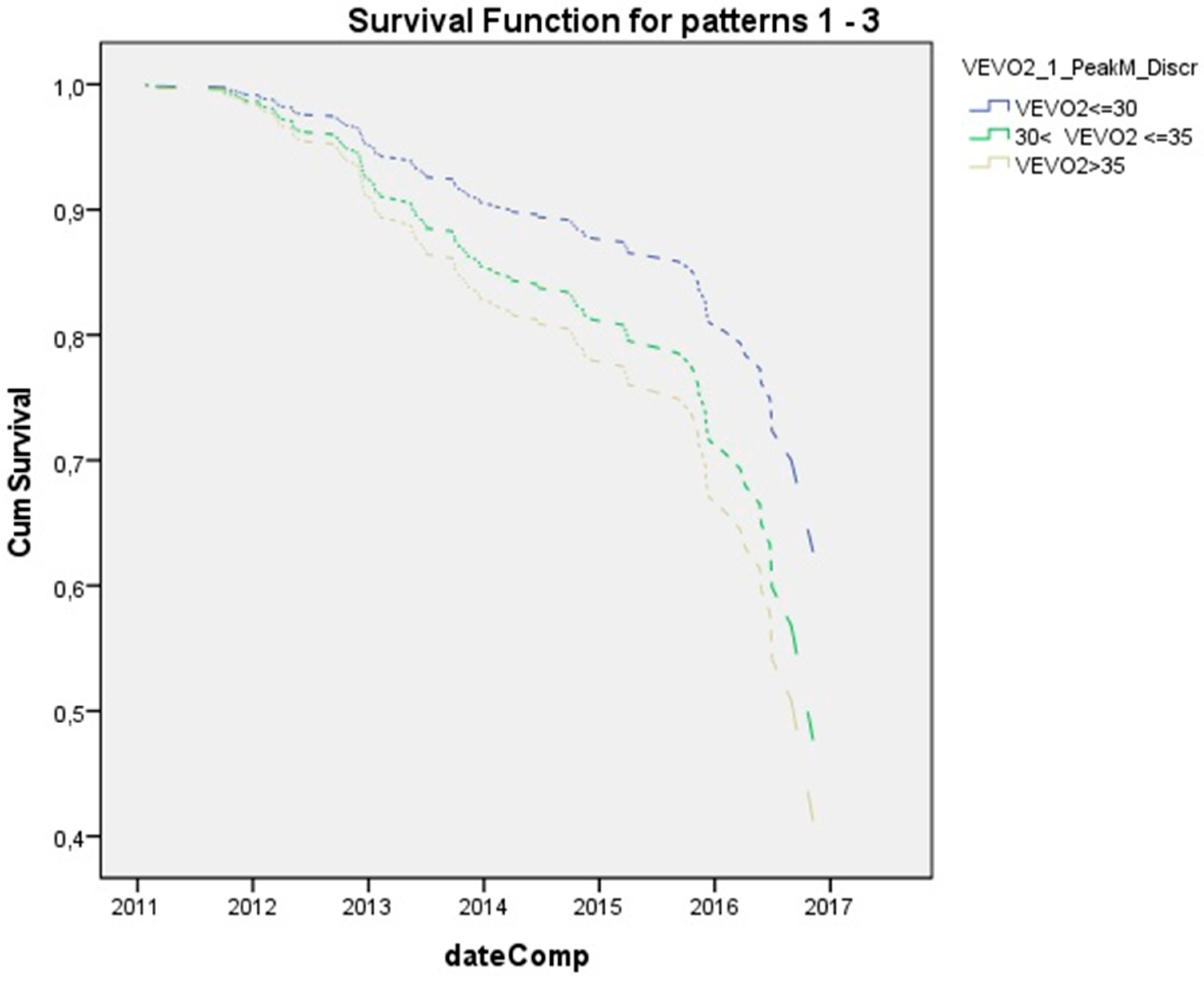
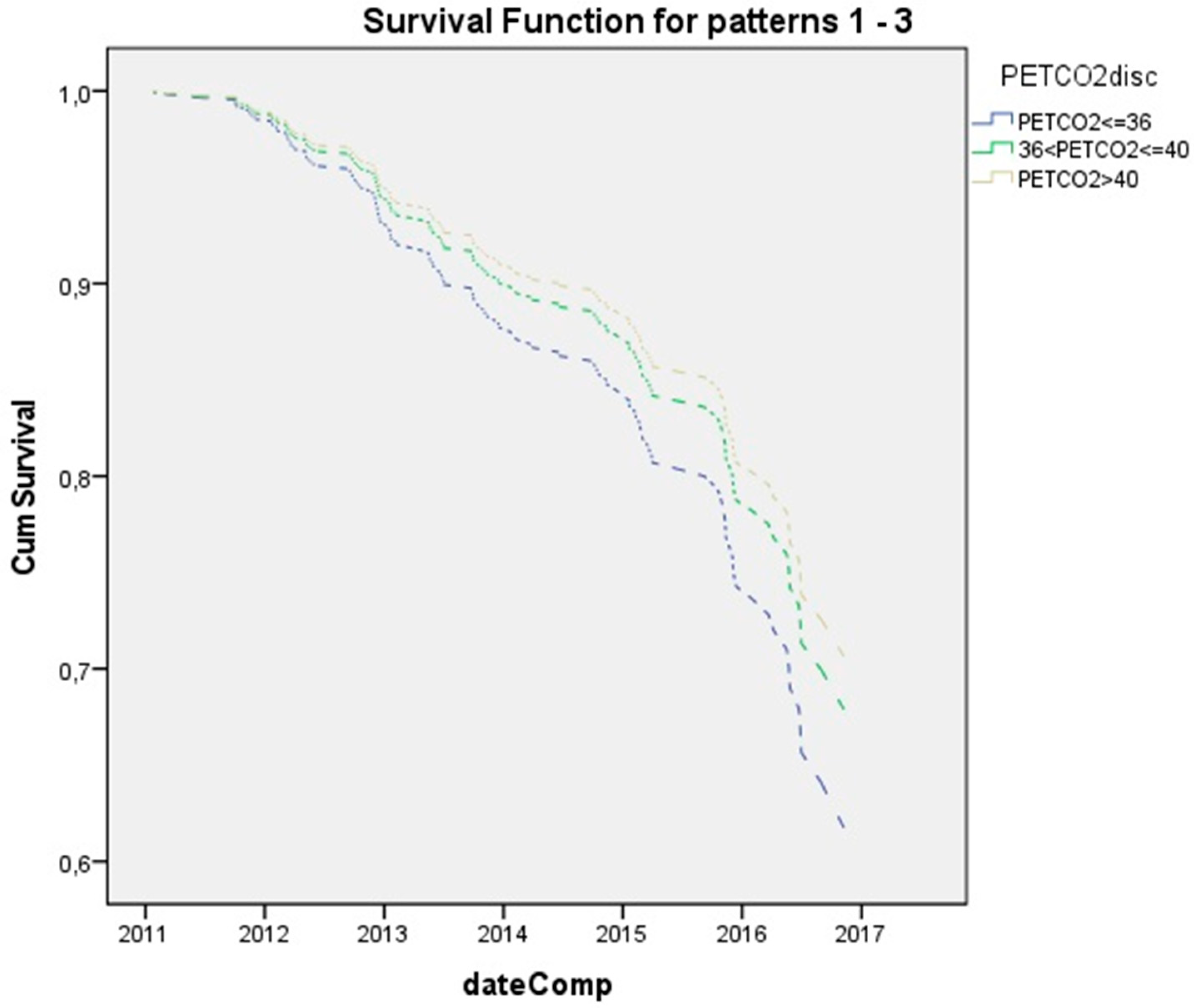
3. Results
Cox Proportional Hazards Models
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Radtke, T.; Crook, S.; Kaltsakas, G.; Louvaris, Z.; Berton, D.; Urquhart, D.S.; Kampouras, A.; Rabinovich, R.A.; Verges, S.; Kontopidis, D.; et al. ERS statement on standardisation of cardiopulmonary exercise testing in chronic lung diseases. Eur. Respir. Rev. 2019, 28, 180101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nixon, P.A.; Orenstein, D.M.; Kelsey, S.F.; Doershuk, C.F. The prognostic value of exercise testing in patients with cystic fibrosis. N. Engl. J. Med. 1992, 327, 1785–1788. [Google Scholar] [CrossRef] [PubMed]
- Moorcroft, A.J.; Dodd, M.E.; Webb, A.K. Exercise testing and prognosis in adult cystic fibrosis. Thorax 1997, 52, 291–293. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pianosi, P.; Leblanc, J.; Almudevar, A. Peak oxygen uptake and mortality in children with cystic fibrosis. Thorax 2005, 60, 50–54. [Google Scholar] [CrossRef] [Green Version]
- Hulzebos, E.H.; Bomhof-Roordink, H.; van de Weert-van Leeuwen, P.B.; Twisk, J.W.; Arets, H.G.; van der Ent, C.K.; Takken, T. Prediction of mortality in adolescents with cystic fibrosis. Med. Sci. Sports Exerc. 2014, 46, 2047–2052. [Google Scholar] [CrossRef] [PubMed]
- Hebestreit, H.; Hulzebos, E.H.; Schneiderman, J.E.; Karila, C.; Boas, S.R.; Kriemler, S.; Dwyer, T.; Sahlberg, M.; Urquhart, D.S.; Lands, L.C.; et al. Cardiopulmonary Exercise Testing Provides Additional Prognostic Information in Cystic Fibrosis. Am. J. Respir. Crit. Care Med. 2019, 199, 987–995. [Google Scholar]
- Urquhart, D.S. Exercise testing in cystic fibrosis: Why (and how)? J. R. Soc. Med. 2011, 104 (Suppl. 1), S6–S14. [Google Scholar] [CrossRef] [Green Version]
- Villaverde-Hueso, A.; Sanchez-Diaz, G.; Molina-Cabrero, F.J.; Gallego, E.; Posada de la Paz, M.; Alonso-Ferreira, V. Mortality Due to Cystic Fibrosis over a 36-Year Period in Spain: Time Trends and Geographic Variations. Int. J. Environ. Res. Public Health 2019, 16, 119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sanders, D.B.; Ostrenga, J.S.; Rosenfeld, M.; Fink, A.K.; Schechter, M.S.; Sawicki, G.S.; Flume, P.A.; Morgan, W.J. Predictors of pulmonary exacerbation treatment in cystic fibrosis. J. Cyst. Fibros. 2020, 19, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Sanders, D.B.; Solomon, G.M.; Beckett, V.V.; West, N.E.; Daines, C.L.; Heltshe, S.L.; Dasenbrook, E.C.D.; van Decanter, D.R.; Solomon, G.M.; Goss, C.H.; et al. Standardized Treatment of Pulmonary Exacerbations (STOP) study: Observations at the initiation of intravenous antibiotics for cystic fibrosis pulmonary exacerbations. J. Cyst. Fibros. 2017, 16, 592–599. [Google Scholar] [CrossRef] [Green Version]
- Vermeulen, F.; Proesmans, M.; Boon, M.; Havermans, T.; De Boeck, K. Lung clearance index predicts pulmonary exacerbations in young patients with cystic fibrosis. Thorax 2014, 69, 39–45. [Google Scholar] [CrossRef] [Green Version]
- Bilton, D.; Canny, G.; Conway, S.; Dumcius, S.; Hjelte, L.; Proesmans, M.; Tümmler, B.; Vavrova, V.; De Boeck, K. Pulmonary exacerbation: Towards a definition for use in clinical trials. Report from the EuroCareCF Working Group on outcome parameters in clinical trials. J. Cyst. Fibros. 2011, 10 (Suppl. 2), S79–S81. [Google Scholar] [CrossRef] [Green Version]
- Beydon, N.; Davis, S.D.; Lombardi, E.; Allen, J.L.; Arets, H.G.M.; Aurora, P.; Bisgaard, H.; Davis, G.M.; Ducharme, F.M.; Eigen, H.; et al. An official American Thoracic Society/European Respiratory Society statement: Pulmonary function testing in preschool children. Am. J. Respir. Crit. Care Med. 2007, 175, 1304–1345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Godfrey, S.; Davies, C.T.; Wozniak, E.; Barnes, C.A. Cardio-respiratory response to exercise in normal children. Clin. Sci. 1971, 40, 419–431. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Orenstein, D. Assessment of Exercise Pulmonary Function; Human Kinetics: Champaign, IL, USA, 1993. [Google Scholar]
- Hebestreit, H.; Kieser, S.; Junge, S.; Ballmann, M.; Hebestreit, A.; Schindler, C.; Shcenk, T.; Posselt, H.-G.; Kriemler, S. Long-term effects of a partially supervised conditioning programme in cystic fibrosis. Eur. Respir. J. 2010, 35, 578–583. [Google Scholar] [CrossRef] [PubMed]
- Hatziagorou, E.; Kampouras, A.; Avramidou, V.; Georgopoulou, V.; Kirvasilis, F.; Kontouli, K.; Hebestreit, H.; Tsanakas, J. Exercise responses are related to structural lung damage in CF pulmonary disease. Pediatr. Pulmonol. 2016, 51, 914–920. [Google Scholar] [CrossRef] [PubMed]
- Kampouras, A.; Hatziagorou, E.; Avramidou, V.; Georgopoulou, V.; Kirvassilis, F.; Hebestreit, H.; Tsanakas, J. Ventilation efficiency to exercise in patients with cystic fibrosis. Pediatr. Pulmonol. 2019, 54, 1584–1590. [Google Scholar] [CrossRef] [PubMed]
- De Bentzmann, S.; Roger, P.; Puchelle, E. Pseudomonas aeruginosa adherence to remodelling respiratory epithelium. Eur. Respir. J. 1996, 9, 2145–2150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Paolo, M.; Teopompi, E.; Savi, D.; Crisafulli, E.; Longo, C.; Tzani, P.; Longo, F.; Ielpo, A.; Pisi, G.; Cimino, G.; et al. Reduced exercise ventilatory efficiency in adults with cystic fibrosis and normal to moderately impaired lung function. J. Appl. Physiol. 2019, 127, 501–512. [Google Scholar] [CrossRef]
- Argo, C.K.; Stine, J.G.; Henry, Z.H.; Lackner, C.; Patrie, J.T.; Weltman, A.L.; Caldwell, S.H. Physical deconditioning is the common denominator in both obese and overweight subjects with nonalcoholic steatohepatitis. Aliment. Pharmacol. Ther. 2018, 48, 290–299. [Google Scholar] [CrossRef]
- Barker, M.; Hebestreit, A.; Gruber, W.; Hebestreit, H. Exercise testing and training in German CF centers. Pediatr. Pulmonol. 2004, 37, 351–355. [Google Scholar] [CrossRef] [PubMed]
- Werkman, M.S.; Hulzebos, E.H.; Helders, P.J.; Arets, B.G.; Takken, T. Estimating peak oxygen uptake in adolescents with cystic fibrosis. Arch. Dis. Child. 2014, 99, 21–25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radtke, T.; Puhan, M.A.; Hebestreit, H.; Kriemler, S. The 1-min sit-to-stand test—A simple functional capacity test in cystic fibrosis? J. Cyst. Fibros. 2016, 15, 223–226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lang, R.L.; Stockton, K.; Wilson, C.; Russell, T.G.; Johnston, L.M. Exercise testing for children with cystic fibrosis: A systematic review. Pediatr. Pulmonol. 2020, 55, 1996–2010. [Google Scholar] [CrossRef] [PubMed]
- Andrade Lima, C.; Dornelas de Andrade, A.; Campos, S.L.; Brandão, D.C.; Mourato, I.P.; Britto, M.C.A. Six-minute walk test as a determinant of the functional capacity of children and adolescents with cystic fibrosis: A systematic review. Respir. Med. 2018, 137, 83–88. [Google Scholar] [CrossRef] [Green Version]
- Selvadurai, H.C.; Blimkie, C.J.; Meyers, N.; Mellis, C.M.; Cooper, P.J.; Van Asperen, P.P. Randomized controlled study of in-hospital exercise training programs in children with cystic fibrosis. Pediatr. Pulmonol. 2002, 33, 194–200. [Google Scholar] [CrossRef]
- Kriemler, S.; Kieser, S.; Junge, S.; Ballmann, M.; Hebestreit, A.; Schindler, C.; Stüssi, C.; Hebestreit, H. Effect of supervised training on FEV1 in cystic fibrosis: A randomised controlled trial. J. Cyst. Fibros. 2013, 12, 714–720. [Google Scholar] [CrossRef] [Green Version]
- Kriemler, S.; Radtke, T.; Christen, G.; Kerstan-Huber, M.; Hebestreit, H. Short-Term Effect of Different Physical Exercises and Physiotherapy Combinations on Sputum Expectoration, Oxygen Saturation, and Lung Function in Young Patients with Cystic Fibrosis. Lung 2016, 194, 659–664. [Google Scholar] [CrossRef]
- Bhatt, J.M. Treatment of pulmonary exacerbations in cystic fibrosis. Eur. Respir. Rev. 2013, 22, 205–216. [Google Scholar] [CrossRef] [Green Version]
- Hebestreit, H.; the ACTIVATE-CF Study Working Group; Lands, L.C.; Alarie, N.; Schaeff, J.; Karila, C.; Orenstein, D.M.; Urquhart, D.S.; Hulzebos, E.H.J.; Stein, L.; et al. Effects of a partially supervised conditioning programme in cystic fibrosis: An international multi-centre randomised controlled trial (ACTIVATE-CF): Study protocol. BMC Pulm Med. 2018, 18, 31. [Google Scholar] [CrossRef] [Green Version]
| Variable | Mean | SD |
|---|---|---|
| Age, years | 14, 9 | 4, 7 |
| Βody Mass Index (BMI), kg/m2 | 19, 6 | 3, 3 |
| Height cm | 153, 7 | 14, 4 |
| Weight kg | 47, 2 | 13, 4 |
| Variable | Relative Hazard (95% CI) | p-Value |
|---|---|---|
| Age, years | 0.878, (0.851, 0.906) | <0.001 |
| ΒΜΙ, kg/m2 | 0.872 (0.842, 0.903) | <0.001 |
| Gender | 1.171 (0.88, 1.56) | 0.279 |
| Sputum culture | 0.314 (0.227, 0.433) | <0.001 |
| FEV1% (%predicted) | 0.99 (0.984, 0.996) | 0.002 |
| FVC% (%predicted) | 0.98 (0.972, 0.988) | <0.001 |
| VO2peak % (%predicted) | 1.003 (0.994, 1.012) | 0.536 |
| VO2max % (%predicted) | 0.993 (0.984, 1.02) | 0.147 |
| VE/VO2peak (Peak Ex) | 1.037 (1.004, 1.071) | 0.029 |
| VE/VCO2peak (Peak Ex) | 1.027 (1.001, 1.055) | 0.045 |
| PetCO2 | 0.96 (0.923. 0.999) | 0.045 |
| CPET Variable | |||||
|---|---|---|---|---|---|
| CPET variables | VO2peak | VE/VO2 Peak | VE/VCO2 Peak | PetCO2 | VO2max |
| 0.988 (0.978, 0.998) p = 0.019 | 1.033 (1.002, 1.065) p = 0.038 | 1.015 (0.992, 1.039) p = 0.213 | 0.954 (0.917, 0.992) p = 0.017 | 0.988 (0.979, 0.997) p = 0.007 | |
| Age | 0.918 (0.886, 0.951) p < 0.001 | 0.929 (0.897, 0.961) p < 0.001 | 0.931 (0.900, 0.964) p < 0.001 | 0.929 (0.897, 0.917) p < 0.001 | 0.926 (0.895, 0.958) p < 0.001 |
| Culture | 0.451 (0.303, 0.672) p < 0.001 | 0.492 (0.328, 7.39) p = 0.001 | 0.479 (0.320, 0.717) p < 0.001 | 0.468 (0.313, 0.701) p < 0.001 | 0.461 (0.310, 0.686) p < 0.001 |
| CPET Variable | |||||
|---|---|---|---|---|---|
| CPET variables | VO2peak | VE/VO2 peak | VE/VCO2 peak | PetCO2 | VO2max |
| 0.988 (0.976, 0.999) p = 0.042 | 1.002 (0.967, 1.038) p = 0.919 | 0.980 (0.949, 1.011) p = 0.2 | 0.964 (0.927, 1.003) p = 0.067 | 0.994 (0.985, 1.004) p = 0.244 | |
| Age | 0.846 (0.810, 0.885) p < 0.001 | 0.860 (0.824, 0. 897) p < 0.001 | 0.853 (0.816, 0.891) p < 0.001 | 0.862 (0.826, 0.899) p < 0.001 | 0.861 (0.826, 0.962) p < 0.001 |
| Culture | 0.597 (0.390, 0.912) p = 0.017 | 0.492 (0.328, 7.39) p = 0.001 | 0.648 (0.423, 0.991) p = 0.045 | 0.633 (0.413, 0.970) p = 0.036 | 0.632 (0.415, 0.962) p = 0.032 |
| FEV1 | 0.980 (0.977, 0.990) p < 0.001 | 0.492 (0.328, 7.39) p = 0.001 | 0.977 (0.967, 0.987) p < 0.001 | 0.980 (0.970, 0.989) p < 0.001 | 0.981 (0.972, 0.990) p < 0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kampouras, A.; Hatziagorou, E.; Kalantzis, T.; Avramidou, V.; Kontouli, K.; Kirvassilis, F.; Tsanakas, J. The Fitter the Better? Cardiopulmonary Exercise Testing Can Predict Pulmonary Exacerbations in Cystic Fibrosis. Children 2021, 8, 527. https://doi.org/10.3390/children8060527
Kampouras A, Hatziagorou E, Kalantzis T, Avramidou V, Kontouli K, Kirvassilis F, Tsanakas J. The Fitter the Better? Cardiopulmonary Exercise Testing Can Predict Pulmonary Exacerbations in Cystic Fibrosis. Children. 2021; 8(6):527. https://doi.org/10.3390/children8060527
Chicago/Turabian StyleKampouras, Asterios, Elpis Hatziagorou, Thomas Kalantzis, Vasiliki Avramidou, Kalliopi Kontouli, Fotios Kirvassilis, and John Tsanakas. 2021. "The Fitter the Better? Cardiopulmonary Exercise Testing Can Predict Pulmonary Exacerbations in Cystic Fibrosis" Children 8, no. 6: 527. https://doi.org/10.3390/children8060527
APA StyleKampouras, A., Hatziagorou, E., Kalantzis, T., Avramidou, V., Kontouli, K., Kirvassilis, F., & Tsanakas, J. (2021). The Fitter the Better? Cardiopulmonary Exercise Testing Can Predict Pulmonary Exacerbations in Cystic Fibrosis. Children, 8(6), 527. https://doi.org/10.3390/children8060527







