Effect of Action Observation Training on Spasticity, Gross Motor Function, and Balance in Children with Diplegia Cerebral Palsy
Abstract
1. Introduction
2. Materials and Methods
2.1. Action Observation Training
2.2. General Physical Therapy
2.3. Outcome Measurements
2.3.1. Spasticity of Ankle Joint
2.3.2. Gross Motor Function
2.3.3. Balance Function Measurement
2.3.4. Data Analyses
3. Results
3.1. Spasticity of Ankle Joint
3.2. Gross Motor Function
3.3. Balance Function Measurement
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Bax, M.; Goldstein, M.; Rosenbaum, P.; Leviton, A.; Paneth, N.; Dan, B.; Jacobsson, B.; Damiano, D. Executive Committee for the Definition of Cerebral Palsy. Proposed definition and classification of cerebral palsy, April 2005. Dev. Med. Child Neurol. 2005, 47, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, M.H.; Viehweger, E.; Stout, J.; Novacheck, T.F.; Gage, J.R. Comprehensive treatment of ambulatory children with cerebral palsy: An outcome assessment. J. Pediatr. Orthop. 2004, 24, 45–53. [Google Scholar] [CrossRef]
- Yokochi, K. Motor functions in non-ambulatory children with spastic diplegia and periventricular leukomalacia. Brain Dev. 2001, 23, 327–332. [Google Scholar] [CrossRef]
- Woollacott, M.H.; Shumway-Cook, A. Postural dysfunction during standing and walking in children with cerebral palsy: What are the underlying problems and what new therapies might improve balance? Neural Plast. 2005, 12, 211–219; discussion 63–72. [Google Scholar] [CrossRef] [PubMed]
- Case-Smith, J. Parenting a child with a chronic medical condition. Am. J. Occup. Ther. 2004, 58, 551–560. [Google Scholar] [CrossRef]
- Yonetsu, R.; Iwata, A.; Surya, J.; Unase, K.; Shimizu, J. Sit-to-stand movement changes in preschool-aged children with spastic diplegia following one neurodevelopmental treatment session--a pilot study. Disabil. Rehabil. 2015, 37, 1643–1650. [Google Scholar] [CrossRef]
- Trompetto, C.; Marinelli, L.; Mori, L.; Pelosin, E.; Curra, A.; Molfetta, L.; Abbruzzese, G. Pathophysiology of spasticity: Implications for neurorehabilitation. Biomed. Res. Int. 2014, 2014, 354906. [Google Scholar] [CrossRef]
- Bani-Ahmed, A. The evidence for prolonged muscle stretching in ankle joint management in upper motor neuron lesions: Considerations for rehabilitation - a systematic review. Top Stroke Rehabil. 2019, 26, 153–161. [Google Scholar] [CrossRef]
- Multani, I.; Manji, J.; Hastings-Ison, T.; Khot, A.; Graham, K. Botulinum Toxin in the Management of Children with Cerebral Palsy. Paediatr. Drugs 2019, 21, 261–281. [Google Scholar] [CrossRef] [PubMed]
- Ross Raftemo, A.E.; Mahendran, A.; Hollung, S.J.; Jahnsen, R.B.; Lydersen, S.; Vik, T.; Andersen, G.L. Use of botulinum toxin A in children with cerebral palsy. Tidsskr. Nor. Laegeforen. 2019, 139, 8. [Google Scholar]
- Shiratori, T.; Girolami, G.L.; Aruin, A.S. Anticipatory postural adjustments associated with a loading perturbation in children with hemiplegic and diplegic cerebral palsy. Exp. Brain Res. 2016, 234, 2967–2978. [Google Scholar] [CrossRef] [PubMed]
- Lintanf, M.; Bourseul, J.S.; Houx, L.; Lempereur, M.; Brochard, S.; Pons, C. Effect of ankle-foot orthoses on gait, balance and gross motor function in children with cerebral palsy: A systematic review and meta-analysis. Clin. Rehabil. 2018, 32, 1175–1188. [Google Scholar] [CrossRef]
- Pin, T.W.; Butler, P.B.; Purves, S. Use of whole body vibration therapy in individuals with moderate severity of cerebral palsy- a feasibility study. BMC Neurol. 2019, 19, 80. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Wang, G.; Wang, B. Rehabilitation treatment of spastic cerebral palsy with radial extracorporeal shock wave therapy and rehabilitation therapy. Med. (Baltim.) 2018, 97, e13828. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Kan, L.; Dong, A.; Zhang, J.; Bai, Z.; Xie, Y.; Liu, Q.; Peng, Y. The effects of action observation training on improving upper limb motor functions in people with stroke: A systematic review and meta-analysis. PLoS ONE 2019, 14, e0221166. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Lee, B.H. Action observation training for functional activities after stroke: A pilot randomized controlled trial. Neuro Rehabil. 2013, 33, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Simon-Martinez, C.; Mailleux, L.; Ortibus, E.; Fehrenbach, A.; Sgandurra, G.; Cioni, G.; Desloovere, K.; Wenderoth, N.; Demaerel, P.; Sunaert, S.; et al. Combining constraint-induced movement therapy and action-observation training in children with unilateral cerebral palsy: A randomized controlled trial. BMC Pediatr. 2018, 18, 250. [Google Scholar] [CrossRef]
- Buccino, G.; Molinaro, A.; Ambrosi, C.; Arisi, D.; Mascaro, L.; Pinardi, C.; Rossi, A.; Gasparotti, R.; Fazzi, E.; Galli, J. Action Observation Treatment Improves Upper Limb Motor Functions in Children with Cerebral Palsy: A Combined Clinical and Brain Imaging Study. Neural. Plast. 2018, 2018, 4843985. [Google Scholar] [CrossRef]
- Sgandurra, G.; Cecchi, F.; Beani, E.; Mannari, I.; Maselli, M.; Falotico, F.P.; Inguaggiato, E.; Perazza, S.; Sicola, E.; Feys, H.; et al. Tele-UPCAT: Study protocol of a randomised controlled trial of a home-based Tele-monitored UPper limb Children Action observation Training for participants with unilateral cerebral palsy. BMJ Open 2018, 8, e017819. [Google Scholar]
- Leonard, G.; Tremblay, F. Corticomotor facilitation associated with observation, imagery and imitation of hand actions: A comparative study in young and old adults. Exp. Brain Res. 2007, 177, 167–175. [Google Scholar] [CrossRef]
- Numanoglu, A.; Gunel, M.K. Intraobserver reliability of modified Ashworth scale and modified Tardieu scale in the assessment of spasticity in children with cerebral palsy. Acta Orthop. Traumatol. Turc. 2012, 46, 196–200. [Google Scholar] [CrossRef]
- Ko, J.; Kim, M. Reliability and responsiveness of the gross motor function measure-88 in children with cerebral palsy. Phys. Ther. 2013, 93, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, D.; Birmingham, T. Validity and reliability of a pediatric reach test. Pediatr. Phys. Ther. 2003, 15, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Surveillance of cerebral palsy in Europe. A collaboration of cerebral palsy surveys and registers. Surveillance of Cerebral Palsy in Europe (SCPE). Dev. Med. Child Neurol. 2000, 42, 816–824. [Google Scholar] [CrossRef] [PubMed]
- Lemmens, R.J.; Janssen-Potten, Y.J.; Timmermans, A.A.; Defesche, A.; Smeets, R.J.; Seelen, H.A. Arm hand skilled performance in cerebral palsy: Activity preferences and their movement components. BMC Neurol. 2014, 14, 52. [Google Scholar] [CrossRef]
- Curtis, D.J.; Bencke, J.; Mygind, B. The effect of training in an interactive dynamic stander on ankle dorsiflexion and gross motor function in children with cerebral palsy. Dev. Neurorehabil. 2014, 17, 393–397. [Google Scholar] [CrossRef]
- Tieman, B.L.; Palisano, R.J.; Gracely, E.J.; Rosenbaum, P.L. Gross motor capability and performance of mobility in children with cerebral palsy: A comparison across home, school, and outdoors/community settings. Phys. Ther. 2004, 84, 419–429. [Google Scholar] [CrossRef]
- Damiano, D.L.; Abel, M.F. Relation of gait analysis to gross motor function in cerebral palsy. Dev. Med. Child Neurol. 1996, 38, 389–396. [Google Scholar] [CrossRef]
- Park, E.S.; Rha, D.W.; Shin, J.S.; Kim, S.; Jung, S. Effects of hippotherapy on gross motor function and functional performance of children with cerebral palsy. Yonsei Med. J. 2014, 55, 1736–1742. [Google Scholar] [CrossRef]
- Mahasup, N.; Sritipsukho, P.; Lekskulchai, R.; Hansakunachai, T. Effects of mirror neurons stimulation on motor skill rehabilitation in children with cerebral palsy: A clinical trial. J. Med. Assoc. Thai. 2012, 95, S166–S172. [Google Scholar]
- Chan, G.; Miller, F. Assessment and treatment of children with cerebral palsy. Orthop. Clin. N. Am. 2014, 45, 313–325. [Google Scholar] [CrossRef] [PubMed]
| The first stage is to improve balance in the sitting position. | |||||
|---|---|---|---|---|---|
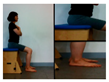 |  |  |  |  | 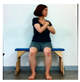 |
| Upright the pelvis | Move weight forward | Move weight to the left and right | Rotate right and left | ||
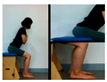
| The second stage is the sit-to-stand movement. | |||||
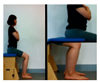 |  |  | |||
| Upright the pelvis in a sitting position | Move weight forward from a sitting position | Stand up from a sitting position | |||
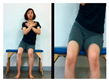
| The third stage is standing movements to improve balance. | |||||
 |  |  | 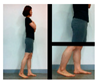 | 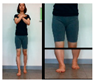 | 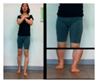 |
| Move Weight Right and left | Forward weight shift with right foot and left foot (lateral view) | Forward weight shift with right foot and left foot (front) | |||
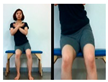
| The fourth stage is walking sideways. | |||||
 |  |  |  | ||
| Walking sideway to the left | Walking sideway to the right | ||||
| Parameters | AOT (n = 9) | Control (n = 9) | t (p) |
|---|---|---|---|
| Gender, M/F (n, %) | 3 (33.3)/6 (66.7) | 5 (55.0)/4 (45.0) | 1.141 (0.270) |
| Age (years) | 7.44 ± 1.88 a | 6.90 ± 1.79 | 0.646 (0.527) |
| Height (cm) | 122.60 ± 13.86 | 123.24 ± 14.18 | −0.099 (0.922) |
| Weight (kg) | 23.01 ± 6.71 | 27.73 ± 10.19 | −1.176 (0.256) |
| GMFCS (I/II/III) | 4/2/3 | 4/3/2 | 0.210 (0.837) |
| Parameters | AOT (n = 9) | Control (n = 9) | t (p) | |
|---|---|---|---|---|
| Ankle stiffness-right side (°) | Before | 4.00 ± 4.72 a | 3.77 ± 4.87 | −1.651 (0.118) |
| After | 10.58 ± 3.36 | 8.16 ± 3.58 | ||
| Before-after | −6.58 ± 2.46 | −4.39 ± 3.12 | ||
| t(p) | −8.018 (0.000) | −4.223 (0.003) | ||
| Ankle stiffness-left side (°) | Before | 3.40 ± 4.92 | 3.34 ± 4.13 | −1.411 (0.177) |
| After | 9.50 ± 3.45 | 7.71 ± 2.98 | ||
| Before-after | −6.10 ± 2.76 | −4.38 ± 2.41 | ||
| t(p) | −6.632 (0.000) | −5.439 (0.001) | ||
| MTS-right (kg) | Before | 4.78 ± 3.26 | 3.57 ± 3.35 | 1.325 (0.204) |
| After | 2.91 ± 2.53 | 2.47 ± 2.99 | ||
| Before-after | 1.87 ± 1.46 | 1.09 ± 0.98 | ||
| t(p) | −4.987 (0.001) | 3.335 (0.010) | ||
| MTS-left (kg) | Before | 3.77 ± 1.46 | 4.29 ± 3.19 | 2.236 (0.040) |
| After | 1.91 ± 1.20 | 3.94 ± 3.28 | ||
| Before-after | 1.86 ± 1.80 | 0.34 ± 0.93 | ||
| t(p) | 3.093 (0.015) | 1.106 (0.301) | ||
| Parameters | AOT (n = 9) | Control (n = 9) | t (p) | |
|---|---|---|---|---|
| GMFM-B (%) | Before | 93.33 ± 6.07 a | 93.89 ± 5.40 | −1.991 (0.064) |
| After | 98.45 ± 2.34 | 95.74 ± 4.72 | ||
| Before-after | −5.12 ± 4.60 | −1.85 ± 1.76 | ||
| t(p) | −3.339 (0.010) | −3.162 (0.013) | ||
| GMFM-C (%) | Before | 88.36 ± 10.68 | 89.94 ± 9.88 | −1.737 (0.102) |
| After | 94.92 ± 5.89 | 91.83 ± 8.49 | ||
| Before-after | −6.56 ± 5.28 | −2.89 ± 3.53 | ||
| t(p) | −3.731 (0.006) | −2.449 (0.040) | ||
| GMFM-D (%) | Before | 60.68 ± 29.79 | 68.94 ± 23.02 | −1.928 (0.072) |
| After | 77.48 ± 20.61 | 75.78 ± 21.79 | ||
| Before-after | −16.80 ± 12.76 | −6.84 ± 8.79 | ||
| t(p) | −3.949 (0.004) | −2.334 (0.048) | ||
| GMFM-E (%) | Before | 46.26 ± 38.00 | 50.30 ± 32.82 | −3.583 (0.002) |
| After | 52.77 ± 37.93 | 51.54 ± 32.26 | ||
| Before-after | −6.51 ± 4.11 | −1.23 ± 1.62 | ||
| t(p) | −4.752 (0.001) | −2.285 (0.052) | ||
| Parameters | AOT (n = 9) | Control (n = 9) | t (p) | |
|---|---|---|---|---|
| PRT lateral-right (cm) | Before | 15.52 ± 6.12 a | 14.94 ± 6.21 | −2.327 (0.033) |
| After | 18.80 ± 6.42 | 16.33 ± 6.87 | ||
| Before-after | −3.28 ± 1.71 | −1.40 ± 1.72 | ||
| t(p) | −5.751 (0.000) | −2.433 (0.041) | ||
| PRT lateral-left (cm) | Before | 14.34 ± 5.17 | 14.96 ± 6.68 | −3.551 (0.003) |
| After | 18.42 ± 5.27 | 16.37 ± 6.75 | ||
| Before-after | −4.08 ± 1.52 | −1.42 ± 1.65 | ||
| t(p) | −8.022 (0.000) | −2.578 (0.033) | ||
| PRT frontal-right (cm) | Before | 22.78 ± 7.16 | 20.35 ± 7.64 | −2.154 (0.047) |
| After | 27.38 ± 7.81 | 22.37 ± 6.79 | ||
| Before-after | −4.60 ± 2.87 | −2.02 ± 2.16 | ||
| t(p) | −4.811 (0.001) | −2.803 (0.023) | ||
| PRT frontal-left (cm) | Before | 22.60 ± 7.58 | 19.86 ± 7.54 | −2.339 (0.033) |
| After | 26.85 ± 7.19 | 21.90 ± 7.41 | ||
| Before-after | −4.25 ± 2.05 | −2.04 ± 1.96 | ||
| t(p) | −6.213 (0.000) | −3.116 (0.014) | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jeong, Y.-a.; Lee, B.-H. Effect of Action Observation Training on Spasticity, Gross Motor Function, and Balance in Children with Diplegia Cerebral Palsy. Children 2020, 7, 64. https://doi.org/10.3390/children7060064
Jeong Y-a, Lee B-H. Effect of Action Observation Training on Spasticity, Gross Motor Function, and Balance in Children with Diplegia Cerebral Palsy. Children. 2020; 7(6):64. https://doi.org/10.3390/children7060064
Chicago/Turabian StyleJeong, Young-a, and Byoung-Hee Lee. 2020. "Effect of Action Observation Training on Spasticity, Gross Motor Function, and Balance in Children with Diplegia Cerebral Palsy" Children 7, no. 6: 64. https://doi.org/10.3390/children7060064
APA StyleJeong, Y.-a., & Lee, B.-H. (2020). Effect of Action Observation Training on Spasticity, Gross Motor Function, and Balance in Children with Diplegia Cerebral Palsy. Children, 7(6), 64. https://doi.org/10.3390/children7060064






