Reference Ranges for Hemoglobin and Hematocrit Levels in Neonates as a Function of Gestational Age (22–42 Weeks) and Postnatal Age (0–29 Days): Mathematical Modeling
Abstract
1. Introduction
2. Materials and Methods
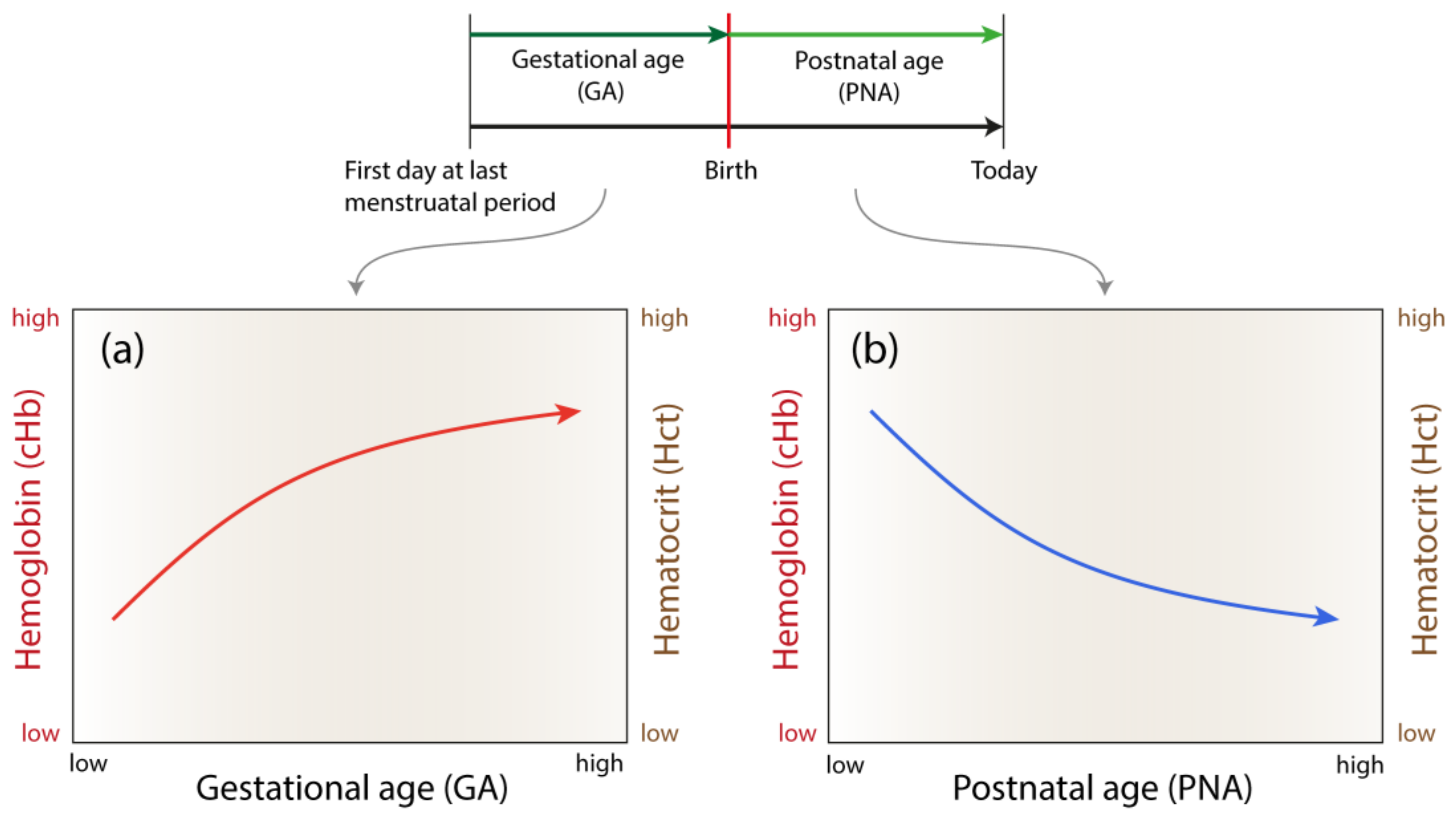
2.1. Hemoglobin and Hematocrit Data
2.2. Derivation and Validation of the General Models
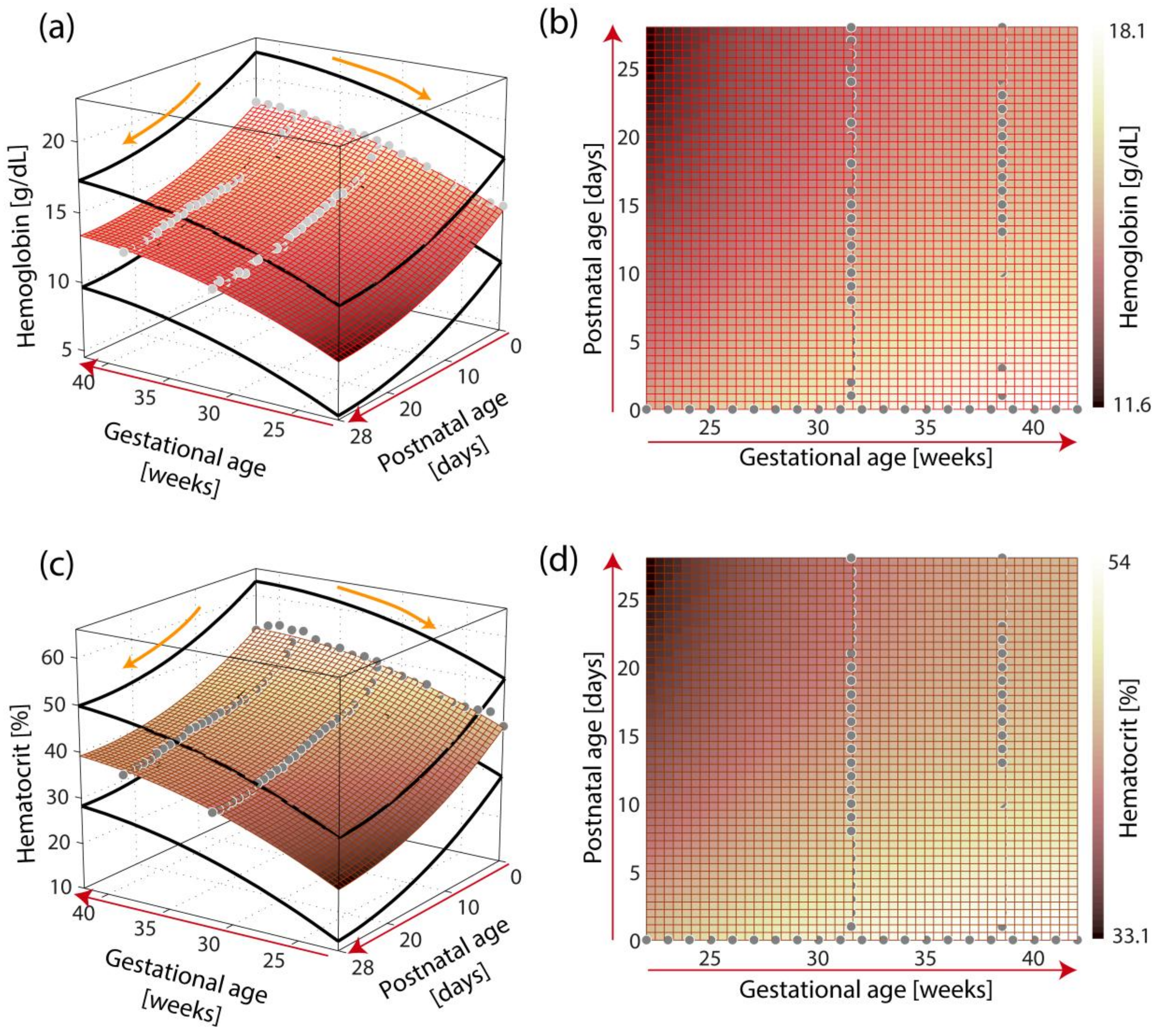
3. Results
3.1. The General Models
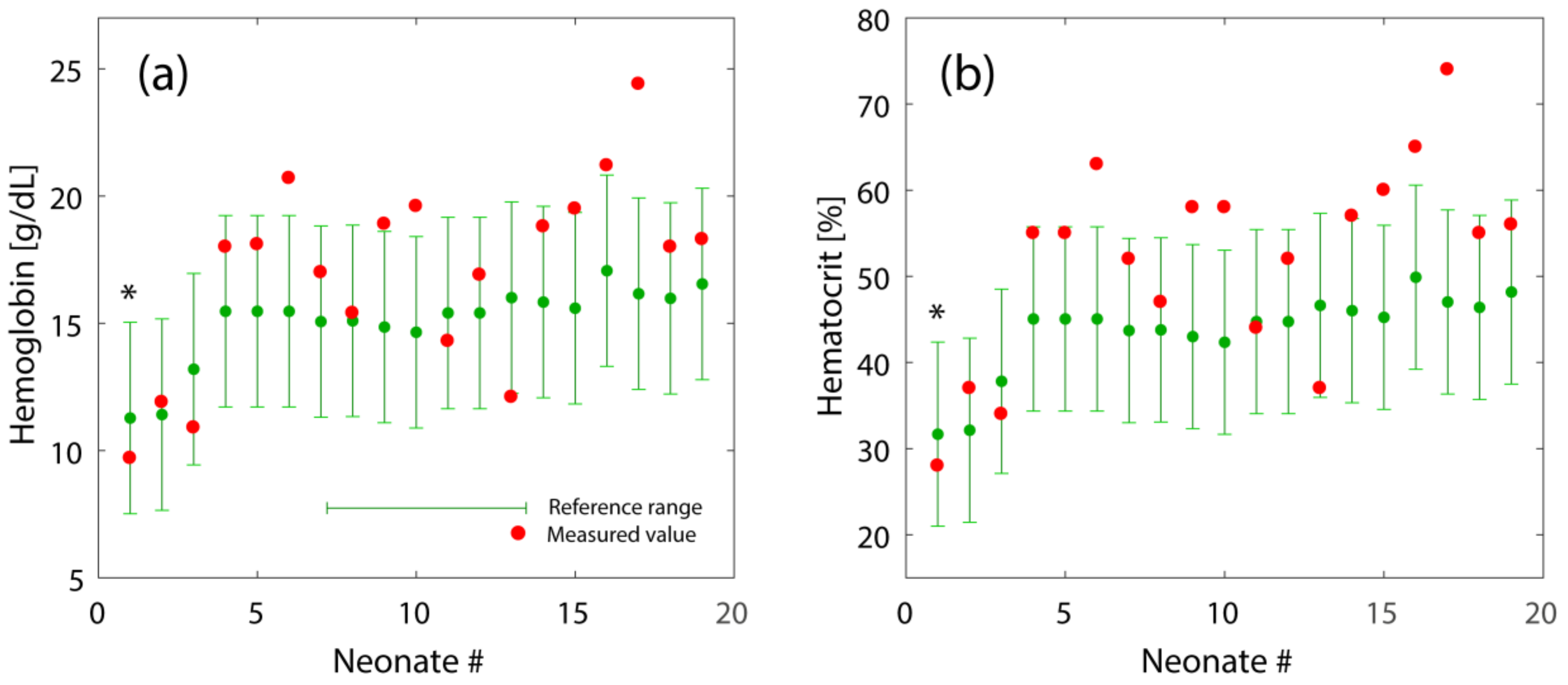
3.2. Validation of the General Models
4. Discussion, Conclusion and Outlook
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Schechter, A.N. Hemoglobin research and the origins of molecular medicine. Blood 2008, 112, 3927–3938. [Google Scholar] [CrossRef] [PubMed]
- Mugrage, E.R. Values for red blood cells of average infants and children. Arch. Pediatr. Adolesc. Med. 1936, 51, 775. [Google Scholar] [CrossRef]
- DeMarsh, Q.B.; Alt, H.L.; Windle, W.F.; Hillis, D.S. The effect of depriving the infant of its placental blood. JAMA 1941, 116, 2568. [Google Scholar] [CrossRef]
- Guest, G.M. Erythrocytes and hemoglobin of the blood in infancy and childhood. AMA Am. J. Dis. Child. 1957, 93, 486. [Google Scholar] [CrossRef]
- Jopling, J.; Henry, E.; Wiedmeier, S.E.; Christensen, R.D. Reference ranges for hematocrit and blood hemoglobin concentration during the neonatal period: Data from a multihospital health care system. Pediatrics 2009. [Google Scholar] [CrossRef] [PubMed]
- Henry, E.; Christensen, R.D. Reference intervals in neonatal hematology. Clin. Perinatol. 2015, 42, 483–497. [Google Scholar] [CrossRef] [PubMed]
- Christensen, R.D.; Henry, E.; Jopling, J.; Wiedmeier, S.E. The cbc: Reference ranges for neonates. Semin. Perinatol. 2009, 33, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Eslami, Z.; Ghilian, R.; Abbasi, F. Evaluation of hemoglobin concentration of cord, capillary and venous sampling in neonates. Iran. J. Ped. Hematol. Oncol. 2012, 2, 159–163. [Google Scholar] [PubMed]
- Crowley, M.; Kirpalani, H. A rational approach to red blood cell transfusion in the neonatal ICU. Curr. Opin. Pediatr. 2010, 22, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Strauss, R.G. Transfusion therapy in neonates. Arch. Pediatr. Adolesc. Med. 1991, 145, 904. [Google Scholar] [CrossRef]
- Schmidt, M.; Lipson, H. Distilling free-form natural laws from experimental data. Science 2009, 324, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Dubčáková, R. Eureqa: Software review. Genet. Program. Evolvable Mach. 2010, 12, 173–178. [Google Scholar] [CrossRef]
- Nasseri, N.; Ostojic, D.; Kleiser, S. In vivo precision assessment of a near-infrared spectroscopy-based tissue oximeter (oxyprem v1.3) in neonates considering systemic hemodynamic fluctuations. J. Biomed. Opt. 2018, 23, 1. [Google Scholar]
- Daae, L.N.W.; Hallerud, M.; Halvorsen, S. A comparison between haematological parameters in ‘capillary’ and venous blood samples from hospitalized children aged 3 months to 14 years. Scand. J. Clin. Lab. Invest. 2009, 51, 651–654. [Google Scholar] [CrossRef]
- Yang, Z.W.; Yang, S.H.; Chen, L.; Qu, J.; Zhu, J.; Tang, Z. Comparison of blood counts in venous, fingertip and arterial blood and their measurement variation. Clin. Lab. Haematol. 2001, 23, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Bosshart, M.; Stover, J.F.; Stocker, R.; Asmis, L.M.; Feige, J.; Neff, T.A.; Schuepbach, R.A.; Cottini, S.R.; Béchir, M. Two different hematocrit detection methods: Different methods, different results? BMC Res. Notes 2010, 3, 65. [Google Scholar] [CrossRef] [PubMed]
- Papa, F.; Rongioletti, M.; Ventura, M.D.; Di Turi, F.; Cortesi, M.; Pasqualetti, P.; Majolini, M.B.; Collegiani, V.; Cicchese, M.; Notarmuzi, M.L.; et al. Blood cell counting in neonates: A comparison between a low volume micromethod and the standard laboratory method. Blood transfus. 2011, 9, 400–406. [Google Scholar] [PubMed]
- Jacob, E.A. Hematological differences in newborn and aging: A review study. HTIJ 2016, 3, 178–190. [Google Scholar] [CrossRef]
- Obladen, M.; Diepold, K.; Maier, R.F. Venous and arterial hematologic profiles of very low birth weight infants. European multicenter rhepo study group. Pediatrics 2000, 106, 707–711. [Google Scholar] [CrossRef] [PubMed]
- Katheria, A.C.; Truong, G.; Cousins, L.; Oshiro, B.; Finer, N.N. Umbilical cord milking versus delayed cord clamping in preterm infants. Pediatrics 2015, 136, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.; Upadhyay, A.; Gothwal, S.; Jaiswal, V.; Joshi, P.; Dubey, K. Umbilical cord milking and hematological parameters in moderate to late preterm neonates: A randomized controlled trial. Indian Pediatr. 2015, 52, 753–757. [Google Scholar] [CrossRef] [PubMed]
- Mercer, J.S.; Erickson-Owens, D.A.; Collins, J.; Barcelos, M.O.; Parker, A.B.; Padbury, J.F. Effects of delayed cord clamping on residual placental blood volume, hemoglobin and bilirubin levels in term infants: A randomized controlled trial. J. Perinatol. 2016, 37, 260–264. [Google Scholar] [CrossRef] [PubMed]
- Bard, H.; Widness, J.A. The life span of erythrocytes transfused to preterm infants. Pediatr. Res. 1997, 42, 9–11. [Google Scholar] [CrossRef] [PubMed]
- ETTNO-Investigators. The ‘effects of transfusion thresholds on neurocognitive outcome of extremely low birth-weight infants (ettno)’ study: Background, aims, and study protocol. Neonatology 2012, 101, 301–305. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-H. Maternal anemia and vitamin d deficiency: Associations with neonatal hemoglobin levels and the vitamin D status. Perinatology 2017, 28, 4. [Google Scholar] [CrossRef]
- MacQueen, B.C.; Henry, E.; Sola-Visner, M.C.; Bennett, S.T.; Christensen, R.D. Hematology, Immunology and Genetics: Neonatology Questions and Controversies, 3rd edition; Ohlis, R.K., Maheshwari, A., Christensen, R.D., Eds.; Elsevier: Oxford, UK, 2018. [Google Scholar]
| Variable | Eq. No. | Parameters | ||||
|---|---|---|---|---|---|---|
| 1 | α1 = −0.3409 | α2 = 0.8300 | α3 = −0.0093 | α4 = 0.0003 | α5 = 0.0058 | |
| 2 | β1 = −1.2956 | β2 = 2.5369 | β3 = −0.0300 | β4 = 0.0069 | β5 = 0.0190 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scholkmann, F.; Ostojic, D.; Isler, H.; Bassler, D.; Wolf, M.; Karen, T. Reference Ranges for Hemoglobin and Hematocrit Levels in Neonates as a Function of Gestational Age (22–42 Weeks) and Postnatal Age (0–29 Days): Mathematical Modeling. Children 2019, 6, 38. https://doi.org/10.3390/children6030038
Scholkmann F, Ostojic D, Isler H, Bassler D, Wolf M, Karen T. Reference Ranges for Hemoglobin and Hematocrit Levels in Neonates as a Function of Gestational Age (22–42 Weeks) and Postnatal Age (0–29 Days): Mathematical Modeling. Children. 2019; 6(3):38. https://doi.org/10.3390/children6030038
Chicago/Turabian StyleScholkmann, Felix, Daniel Ostojic, Helene Isler, Dirk Bassler, Martin Wolf, and Tanja Karen. 2019. "Reference Ranges for Hemoglobin and Hematocrit Levels in Neonates as a Function of Gestational Age (22–42 Weeks) and Postnatal Age (0–29 Days): Mathematical Modeling" Children 6, no. 3: 38. https://doi.org/10.3390/children6030038
APA StyleScholkmann, F., Ostojic, D., Isler, H., Bassler, D., Wolf, M., & Karen, T. (2019). Reference Ranges for Hemoglobin and Hematocrit Levels in Neonates as a Function of Gestational Age (22–42 Weeks) and Postnatal Age (0–29 Days): Mathematical Modeling. Children, 6(3), 38. https://doi.org/10.3390/children6030038






