Conducting Psychosocial Intervention Research among Adolescents and Young Adults with Cancer: Lessons from the PRISM Randomized Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
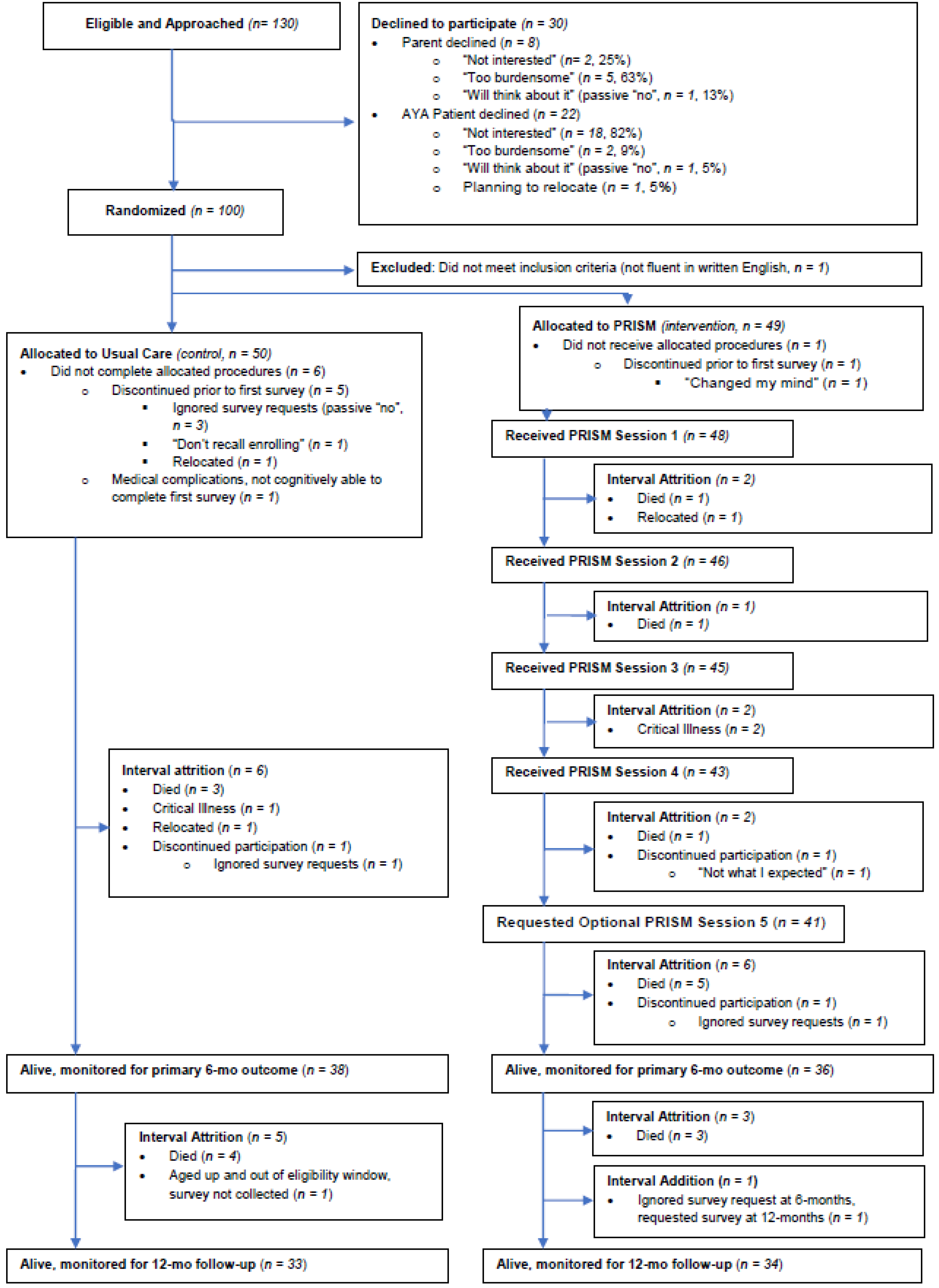
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bleyer, A. Young adult oncology: The patients and their survival challenges. CA Cancer J. Clin. 2007, 57, 242–255. [Google Scholar] [CrossRef] [PubMed]
- Bleyer, A. The adolescent and young adult gap in cancer care and outcome. Curr. Probl. Pediatr. Adolesc. Health Care 2005, 35, 182–217. [Google Scholar] [CrossRef] [PubMed]
- Bleyer, W.A. Cancer in older adolescents and young adults: Epidemiology, diagnosis, treatment, survival, and importance of clinical trials. Med. Pediatr. Oncol. 2002, 38, 1–10. [Google Scholar] [CrossRef]
- Shaw, P.H.; Hayes-Lattin, B.; Johnson, R.; Bleyer, A. Improving enrollment in clinical trials for adolescents with cancer. Pediatrics 2014, 133, S109–S113. [Google Scholar] [CrossRef]
- Ferrari, A.; Bleyer, A. Participation of adolescents with cancer in clinical trials. Cancer Treat. Rev. 2007, 33, 603–608. [Google Scholar] [CrossRef]
- Burke, M.E.; Albritton, K.; Marina, N. Challenges in the recruitment of adolescents and young adults to cancer clinical trials. Cancer 2007, 110, 2385–2393. [Google Scholar] [CrossRef]
- Read, K.; Fernandez, C.V.; Gao, J.; Strahlendorf, C.; Moghrabi, A.; Pentz, R.D.; Barfield, R.C.; Baker, J.N.; Santor, D.; Weijer, C.; et al. Decision-making by adolescents and parents of children with cancer regarding health research participation. Pediatrics 2009, 124, 959–965. [Google Scholar] [CrossRef]
- Varma, S.; Jenkins, T.; Wendler, D. How do children and parents make decisions about pediatric clinical research? J. Pediatr. Hematol. Oncol. 2008, 30, 823–828. [Google Scholar] [CrossRef]
- Barakat, L.P.; Schwartz, L.A.; Reilly, A.; Deatrick, J.A.; Balis, F. A Qualitative Study of Phase III Cancer Clinical Trial Enrollment Decision-Making: Perspectives from Adolescents, Young Adults, Caregivers, and Providers. J. Adolesc. Young Adult Oncol. 2014, 3, 3–11. [Google Scholar] [CrossRef]
- Collins, C.L.; Malvar, J.; Hamilton, A.S.; Deapen, D.M.; Freyer, D.R. Case-linked analysis of clinical trial enrollment among adolescents and young adults at a National Cancer Institute-designated comprehensive cancer center. Cancer 2015, 121, 4398–4406. [Google Scholar] [CrossRef]
- Sanford, S.D.; Beaumont, J.L.; Snyder, M.A.; Reichek, J.; Salsman, J.M. Clinical research participation among adolescent and young adults at an NCI-designated Comprehensive Cancer Center and affiliated pediatric hospital. Support. Care Cancer 2017, 25, 1579–1586. [Google Scholar] [CrossRef] [PubMed]
- Fern, L.A.; Taylor, R.M. Enhancing accrual to clinical trials of adolescents and young adults with cancer. Pediatr. Blood Cancer 2018, 65, e27233. [Google Scholar] [CrossRef]
- Barakat, L.P.; Stevens, E.; Li, Y.; Reilly, A.; Deatrick, J.A.; Goldstein, N.E.; Schwartz, L.A. Evaluation of the Pediatric Research Participation Questionnaire for Measuring Attitudes Toward Cancer Clinical Trials Among Adolescents and Young Adults. J. Adolesc. Young Adult Oncol. 2019, 8, 423–433. [Google Scholar] [CrossRef] [PubMed]
- Zebrack, B.; Mathews-Bradshaw, B.; Siegel, S. Quality cancer care for adolescents and young adults: A position statement. J. Clin. Oncol. 2010, 28, 4862–4867. [Google Scholar] [CrossRef]
- Kazak, A.E.; Abrams, A.N.; Banks, J.; Christofferson, J.; DiDonato, S.; Grootenhuis, M.A.; Kabour, M.; Madan-Swain, A.; Patel, S.K.; Zadeh, S.; et al. Psychosocial Assessment as a Standard of Care in Pediatric Cancer. Pediatr. Blood Cancer 2015, 62, S426–S459. [Google Scholar] [CrossRef] [PubMed]
- Yeager, S. Interdisciplinary collaboration: The heart and soul of health care. Crit. Care Nurs Clin. N. Am. 2005, 17, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Hoge, M.A.; Roth, A.J. Psycho-Oncology, 3rd ed.; Oxford University Press: Oxford, UK, 2015. [Google Scholar]
- Steele, A.C.; Mullins, L.L.; Mullins, A.J.; Muriel, A.C.; Ba, A.J.M. Psychosocial Interventions and Therapeutic Support as a Standard of Care in Pediatric Oncology. Pediatr. Blood Cancer 2015, 62, S585–S618. [Google Scholar] [CrossRef] [PubMed]
- Patenaude, A.F.; Pelletier, W.; Bingen, K. Communication, Documentation, and Training Standards in Pediatric Psychosocial Oncology. Pediatr. Blood Cancer 2015, 62, S870–S895. [Google Scholar] [CrossRef]
- Karlson, C.W.; Rapoff, M.A. Attrition in randomized controlled trials for pediatric chronic conditions. J. Pediatr. Psychol. 2009, 34, 782–793. [Google Scholar] [CrossRef]
- Kwak, M.; Zebrack, B.J.; Meeske, K.A.; Embry, L.; Aguilar, C.; Block, R.; Hayes-Lattin, B.; Li, Y.; Butler, M.; Cole, S. Trajectories of psychological distress in adolescent and young adult patients with cancer: A 1-year longitudinal study. J. Clin. Oncol. 2013, 31, 2160–2166. [Google Scholar] [CrossRef]
- Landolt, M.A.; Vollrath, M.; Niggli, F.K.; Gnehm, H.E.; Sennhauser, F.H. Health-related quality of life in children with newly diagnosed cancer: A one year follow-up study. Health Qual. Life Outcomes 2006, 4, 63. [Google Scholar] [CrossRef] [PubMed]
- Harlan, L.C.; Lynch, C.F.; Keegan, T.H.M.; Hamilton, A.S.; Wu, X.-C.; Kato, I.; West, M.M.; Cress, R.D.; Schwartz, S.M.; Smith, A.W.; et al. Recruitment and follow-up of adolescent and young adult cancer survivors: The AYA HOPE Study. J. Cancer Surviv. 2011, 5, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.W.; Bellizzi, K.M.; Keegan, T.H.; Zebrack, B.; Chen, V.W.; Neale, A.V.; Hamilton, A.S.; Shnorhavorian, M.; Lynch, C.F. Health-related quality of life of adolescent and young adult patients with cancer in the United States: The Adolescent and Young Adult Health Outcomes and Patient Experience study. J. Clin. Oncol. 2013, 31, 2136–2145. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, A.R.; Bona, K.; Wharton, C.M.; Bradford, M.; Shaffer, M.L.; Wolfe, J.; Baker, K.S. Adolescent and Young Adult Patient Engagement and Participation in Survey-Based Research: A Report From the “Resilience in Adolescents and Young Adults With Cancer” Study. Pediatr. Blood Cancer 2016, 63, 734–736. [Google Scholar] [CrossRef]
- Rosenberg, A.R.; Bradford, M.C.; McCauley, E.; Curtis, J.R.; Wolfe, J.; Baker, K.S.; Yi-Frazier, J.P. Promoting resilience in adolescents and young adults with cancer: Results from the PRISM randomized controlled trial. Cancer 2018, 124, 3909–3917. [Google Scholar] [CrossRef]
- Rosenberg, A.R.; Bradford, M.C.; Barton, K.S.; Etsekson, N.; McCauley, E.; Curtis, J.R.; Wolfe, J.; Baker, K.S.; Yi-Frazier, J.P. Hope and benefit finding: Results from the PRISM randomized controlled trial. Pediatr. Blood Cancer 2019, 66, e27485. [Google Scholar] [CrossRef]
- Lau, N.; Bradford, M.C.; Steineck, A.; Junkins, C.C.; Yi-Frazier, J.P.; McCauley, E.; Rosenberg, A.R. Exploratory analysis of treatment response trajectories in the Promoting Resilience in Stress Management (PRISM) Trial: Models of psychosocial care. Psychooncology 2019, 28, 1470–1476. [Google Scholar] [CrossRef]
- Rosenberg, A.R.; Yi-Frazier, J.P.; Eaton, L.; Wharton, C.; Cochrane, K.; Pihoker, C.; Baker, K.S.; McCauley, E. Promoting Resilience in Stress Management: A Pilot Study of a Novel Resilience-Promoting Intervention for Adolescents and Young Adults With Serious Illness. J. Pediatr. Psychol. 2015, 40, 992–999. [Google Scholar] [CrossRef]
- Rosenberg, A.R.; Yi-Frazier, J.P. Commentary: Resilience Defined: An Alternative Perspective. J. Pediatr. Psychol. 2016, 41, 506–509. [Google Scholar] [CrossRef]
- O’Brien, B.C.; Harris, I.B.; Beckman, T.J.; Reed, D.A.; Cook, D.A. Standards for reporting qualitative research: A synthesis of recommendations. Acad. Med. 2014, 89, 1245–1251. [Google Scholar] [CrossRef]
- Vaismoradi, M.; Turunen, H.; Bondas, T. Content analysis and thematic analysis: Implications for conducting a qualitative descriptive study. Nurs. Health Sci. 2013, 15, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Carter, N.; Bryant-Lukosius, D.; DiCenso, A.; Blythe, J.; Neville, A.J. The use of triangulation in qualitative research. Oncol. Nurs. Forum 2014, 41, 545–547. [Google Scholar] [CrossRef] [PubMed]
- Parsons, H.M.; Harlan, L.C.; Seibel, N.L.; Stevens, J.L.; Keegan, T.H. Clinical trial participation and time to treatment among adolescents and young adults with cancer: Does age at diagnosis or insurance make a difference? J. Clin. Oncol. 2011, 29, 4045–4053. [Google Scholar] [CrossRef] [PubMed]
- Parsons, H.M.; Harlan, L.C.; Seibel, N.L.; Stevens, J.L.; Keegan, T.H. Ethics, Emotions, and the Skills of Talking About Progressing Disease With Terminally Ill Adolescents: A Review. JAMA Pediatr. 2016, 170, 1216–1223. [Google Scholar] [CrossRef]
- Rosenberg, A.R.; Wolfe, J. Palliative Care for Adolescents and Young Adults with Cancer. Clin. Oncol. Adolesc. Young Adults 2013, 3, 41–48. [Google Scholar]
- Wiener, L.; Zadeh, S.; Battles, H.; Baird, K.; Ballard, E.; Osherow, J.; Pao, M. Allowing adolescents and young adults to plan their end-of-life care. Pediatrics 2012, 130, 897–905. [Google Scholar] [CrossRef]
- Lyon, M.E.; McCabe, M.A.; Patel, K.M.; D’Angelo, L.J. What do adolescents want? An exploratory study regarding end-of-life decision-making. J. Adolesc. Health 2004, 35, 529. [Google Scholar] [CrossRef]
- Lyon, M.E.; Jacobs, S.; Briggs, L.; Cheng, Y.I.; Wang, J. Family-centered advance care planning for teens with cancer. JAMA Pediatr. 2013, 167, 460–467. [Google Scholar] [CrossRef]
- Hinds, P.S.; Drew, D.; Oakes, L.L.; Fouladi, M.; Spunt, S.L.; Church, C.; Furman, W.L. End-of-life care preferences of pediatric patients with cancer. J. Clin. Oncol. 2005, 23, 9146–9154. [Google Scholar] [CrossRef]
- Lannen, P.K.; Wolfe, J.; Prigerson, H.G.; Onelöv, E.; Kreicbergs, U.C. Unresolved grief in a national sample of bereaved parents: Impaired mental and physical health 4 to 9 years later. J. Clin. Oncol. 2008, 26, 5870–5876. [Google Scholar] [CrossRef]
- Kreicbergs, U.; Valdimarsdóttir, U.; Onelöv, E.; Björk, O.; Steineck, G.; Henter, J.-I. Care-related distress: A nationwide study of parents who lost their child to cancer. J. Clin. Oncol. 2005, 23, 9162–9171. [Google Scholar] [CrossRef] [PubMed]
- Kreicbergs, U.; Valdimarsdóttir, U.; Onelöv, E.; Henter, J.-I.; Steineck, G. Talking about death with children who have severe malignant disease. N. Engl. J. Med. 2004, 351, 1175–1186. [Google Scholar] [CrossRef] [PubMed]
- Bluebond-Langner, M. The Private Worlds of Dying Children; Princeton University Press: Princeton, NJ, USA, 1980. [Google Scholar]
- Coccia, P.F.; Pappo, A.S.; Beaupin, L.; Borges, V.F.; Borinstein, S.C.; Chuch, R.; Dinner, S.; Folbrecht, J.; Frazier, A.L.; Goldsy, R.; et al. Adolescent and Young Adult Oncology Version 2.2018, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Canc. Netw. 2018, 16, 66–97. [Google Scholar] [CrossRef] [PubMed]
| Population | Sample Explanations and Preferences |
|---|---|
| Parents who declined participation (n = 8) | “This would be too much for him to handle right now.” “The questions in this survey would upset him.” |
| Parents whose Adolescents and Young Adults (AYAs) declined participation (n = 22) | “I think this might help him, but he gets to decide.” “Is there a similar study for parents?” |
| AYAs who declined participation (n = 22) | “I don’t believe in this psych crap.” “I am not interested in doing that.” |
| Enrolled AYAs regarding choice of digital or paper-pencil survey | Baseline (n = 92): 23 (25%) prefer digital; 69 (75%) prefer paper-pencil 6-month (n = 74): 50 (68%) prefer digital; 24 (32%) prefer paper-pencil 12-month (n = 67): 58 (87%) prefer digital; 9 (13%) prefer paper-pencil |
| A. AYAs Who Enrolled and Completed Surveys (n = 92) | B. AYAs Who Enrolled and Did Not Complete Surveys (n = 7) | C. AYAs Who Were Approached and Declined Enrollment (n = 30) | |||||
|---|---|---|---|---|---|---|---|
| Characteristic | Usual Care (n = 44) | PRISM (n = 48) | All [n = 92] | Usual Care [n = 6] | PRISM [n = 1] | All [n = 7] | All [n = 30] |
| (n, %) | (n, %) | (n, %) | (n, %) | (n, %) | (n, %) | (n, %) | |
| Female | 24 (55) | 16 (33) | 40 (43) | 2 (33) | 0 (0) | 2 (29) | 6 (20) |
| 12-17 years-old at enrollment | 32 (73) | 35 (73) | 67 (73) | 6 (100) | 1 (100) | 7 (100) | 22 (73) |
| 18-25 years-old at enrollment | 12 (27) | 13 (27) | 25 (27) | 0 (0) | 0 (0) | 0 (0) | 8 (27) |
| Non-White Race | 19 (43) | 15 (31) | 33 (36) | 4 (67) | 0 (0) | 4 (57) | 6 (20) |
| Leukemia/Lymphoma | 27 (61) | 30 (63) | 57 (62) | 4 (67) | 1 (100) | 5 (71) | 17 (57) |
| Central Nervous System (CNS) | 3 (7) | 3 (7) | 6 (7) | 1 (17) | 0 (0) | 1 (14) | 3 (10) |
| Non-CNS Solid Tumor | 14 (32) | 15 (31) | 29 (32) | 1 (17) | 0 (0) | 1 (14) | 10 (33) |
| Advanced Cancer at Enrollment | 14 (32) | 10 (21) | 24 (26) | 3 (50) | 1 (100) | 3 (43) | 11 (37) |
| 1–5 Weeks after Diagnostic News | 6–10 Weeks after Diagnostic News | |
|---|---|---|
| New Diagnosis of Cancer | ||
| Number enrolled | 53 | 20 |
| Number approached | 67 | 25 |
| Enrollment Rate | 79 | 80 |
| New Diagnosis of Recurrent, Progressive, or Refractory Cancer (“Advanced” Cancer) | ||
| Number enrolled | 10 | 17 |
| Number approached | 18 | 20 |
| Enrollment Rate | 56 | 85 |
| Total | ||
| Number enrolled | 63 | 37 |
| Number approached | 85 | 45 |
| Enrollment Rate | 74 | 82 |
| Study Stage | Observations (Source) | Lessons Learned for Future Research |
|---|---|---|
| Screening and Approach | AYAs could not be approached if their parents were not at the bedside. (CRC field notes) | Consider alternative approaches for parents/guardians who are unable to be at the bedside. These may include phone-based consent conferences and/or after-hours staffing. |
| “Flexibility was key. We were most successful when we met families where they were at–both physically, like where they were in the hospital (clinic or infusion or the patient room) and also where they were emotionally. We learned how to read the room to know what to say and when.” (CRC focus group) “It took time and patience and presence. Time and patience to wait for patients to be available, and presence to listen and engage.” (CRC focus group) | Accrual was successful because there was a person in the hospital/clinic every day. This may require a large, flexible, research team or sharing staff resources with other research programs. | |
| “Relationships matter. Nurses, [Medical Assistants] and other staff helped me know when to approach them.” (CRC focus group) “If they had an ‘information heavy’ day, then I tried not to approach.” (CRC focus group) | Collaboration with clinical teams can help staff time approaches. | |
| “You always have to ask. You have to know your biases. What you think is a bad day to approach might not be a bad day for them.” (CRC focus group) “I had to learn how to navigate the medical chart.” (CRC focus group) | Non-clinical staff may benefit from additional training and support regarding how to interact with ill patients. | |
| Enrollment | “Emphasizing the fact that we wanted to learn from the AYAs, that their voices mattered, was the most important factor [to explain our high enrollment rate].” (CRC focus group) “It helped that they saw the same person each time. It promoted trust.” (CRC focus group) “I said, ‘you know, we really haven’t figured this out yet, and we know we can do better,’ and then the AYA and the parent would start to nod.” (CRC focus group) | Engaging the AYA and family about how their voice and story is valuable, and validating their experience while building rapport, may encourage enrollment. |
| Several parents wanted their AYA children to participate, even when the children declined. (CRC field notes) Parents and AYA patients declined participation for different reasons: parents who declined were commonly concerned about the burden of the intervention; AYAs declined because of disinterest or the stigma of mental health interventions. (CRC field notes) “Having a ‘script’ helped. Like, when they said, ‘I already know how to cope,’ I would say, ‘Great! You are exactly the person we want to learn from!’ And then they would start to listen.” (CRC focus group) | Joint discussions about the study, its research question and rationale, and its requirements may help patients and families make decisions together. When staff take time to explore individual and family concerns, more agree to participate. | |
| AYAs with new cancer were just as likely to enroll within the first few weeks of their diagnosis as later. AYAs with advanced cancer more commonly enrolled >4 weeks after their cancer had progressed. (CRC field notes) | Timing of approach may vary for different patient groups. Hearing of a recurrence may demand more processing time. | |
| Baseline Data Collection | More study participants declined further participation when randomized to the inactive control arm. (CRC field notes) “They were disappointed when they didn’t get PRISM.” (CRC focus group) | Collect baseline data (including surveys) prior to randomization. |
| Consider “active” control arms so participants in both arms “receive” something. | ||
| Study Observation Period | Death and critical illness were prevalent in this population of AYAs with cancer. (CRC field notes) | Study staff must be prepared for uncontrollable attrition and incomplete intervention delivery. Allowing flexibility in study procedures (e.g., allowing surveys to be collected late and/or intervention sessions to be rescheduled) may facilitate retention. |
| Only 1 intervention recipient choose to discontinue participation once they started the intervention. (CRC field notes) | Consider strategies to engage participants early (e.g., trust and rapport building); once they start, they are more likely to finish. | |
| Outcome Data Collection | Most participants who were still alive/well-completed surveys at 6-months. Their preferences for data collection (digital versus paper-pencil) evolved. “They started doing more digital because we started offering them an iPad to do the survey in real time. I think they just wanted to do whatever was most convenient.” (CRC focus group) | Consider patient and staff convenience. Digital survey completion may be facilitated by providing equipment to respond in real time. |
| All Stages | “Screening, coordinating approaches, going back and forth to discuss enrollment, getting to know families, delivering interventions, and monitoring survey collection all took a lot of time. It worked because we could be fully devoted to the project.” (CRC focus group) | Successful conduct of a clinical trial may require at least one CRC to be devoted to the project full-time. |
| “It was much harder for me because I was not working full time with the team. When I started taking on more responsibility, when I became more invested in the impact of the work we do, I also got more enthusiastic.” (CRC focus group) “Connectedness and support within the research team and to the clinical teams helped. We were all working towards the same goal.” (CRC focus group) | Intra-team engagement boosts morale and study success. Consider embedded team-building activities and opportunities to encourage staff ownership of the project. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosenberg, A.R.; Junkins, C.C.; Sherr, N.; Scott, S.; Klein, V.; Barton, K.S.; Yi-Frazier, J.P. Conducting Psychosocial Intervention Research among Adolescents and Young Adults with Cancer: Lessons from the PRISM Randomized Clinical Trial. Children 2019, 6, 117. https://doi.org/10.3390/children6110117
Rosenberg AR, Junkins CC, Sherr N, Scott S, Klein V, Barton KS, Yi-Frazier JP. Conducting Psychosocial Intervention Research among Adolescents and Young Adults with Cancer: Lessons from the PRISM Randomized Clinical Trial. Children. 2019; 6(11):117. https://doi.org/10.3390/children6110117
Chicago/Turabian StyleRosenberg, Abby R., Courtney C. Junkins, Nicole Sherr, Samantha Scott, Victoria Klein, Krysta S. Barton, and Joyce P. Yi-Frazier. 2019. "Conducting Psychosocial Intervention Research among Adolescents and Young Adults with Cancer: Lessons from the PRISM Randomized Clinical Trial" Children 6, no. 11: 117. https://doi.org/10.3390/children6110117
APA StyleRosenberg, A. R., Junkins, C. C., Sherr, N., Scott, S., Klein, V., Barton, K. S., & Yi-Frazier, J. P. (2019). Conducting Psychosocial Intervention Research among Adolescents and Young Adults with Cancer: Lessons from the PRISM Randomized Clinical Trial. Children, 6(11), 117. https://doi.org/10.3390/children6110117





