Complete and Incomplete Pentalogy of Cantrell
Abstract
1. Introduction
2. Description
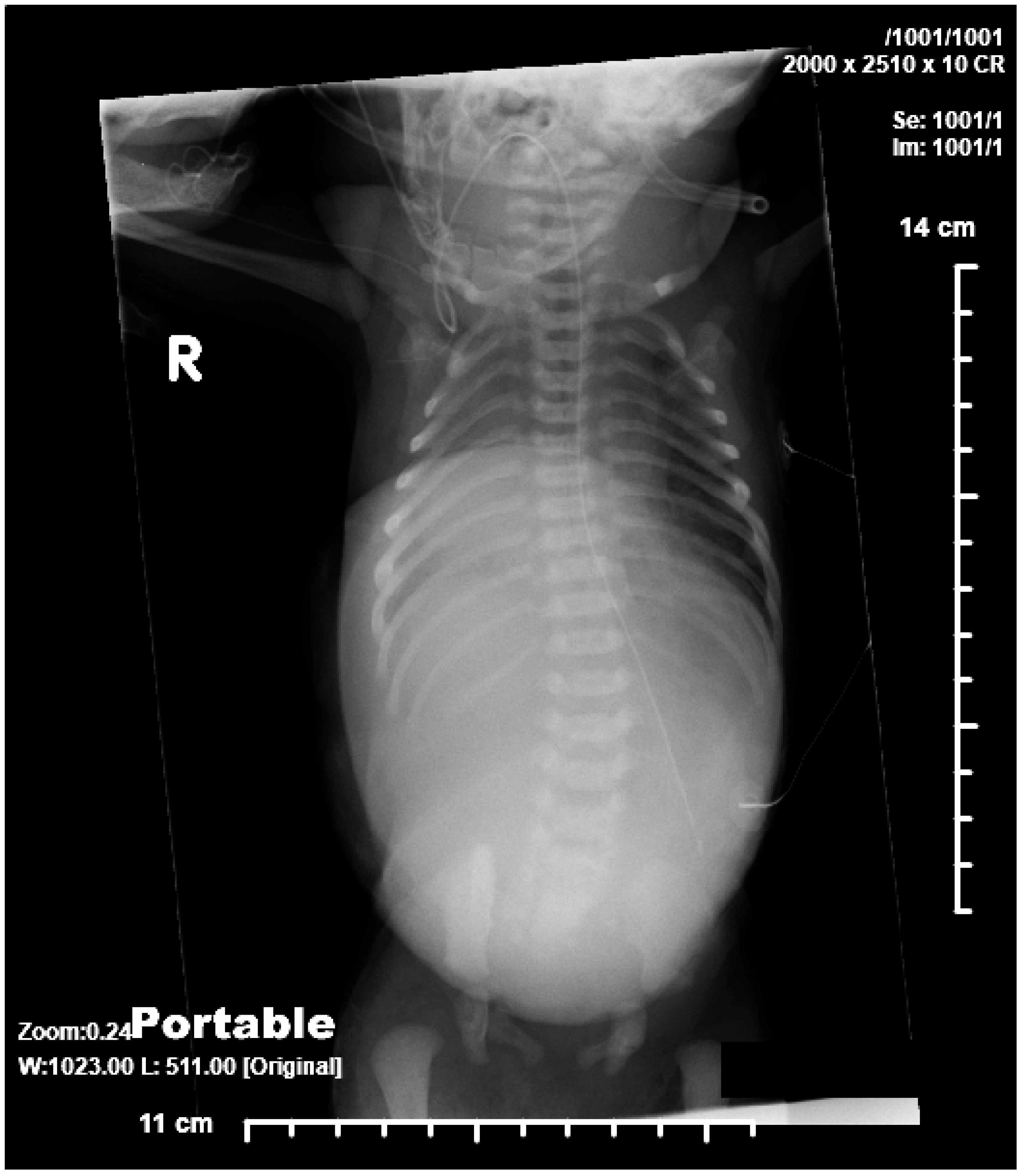
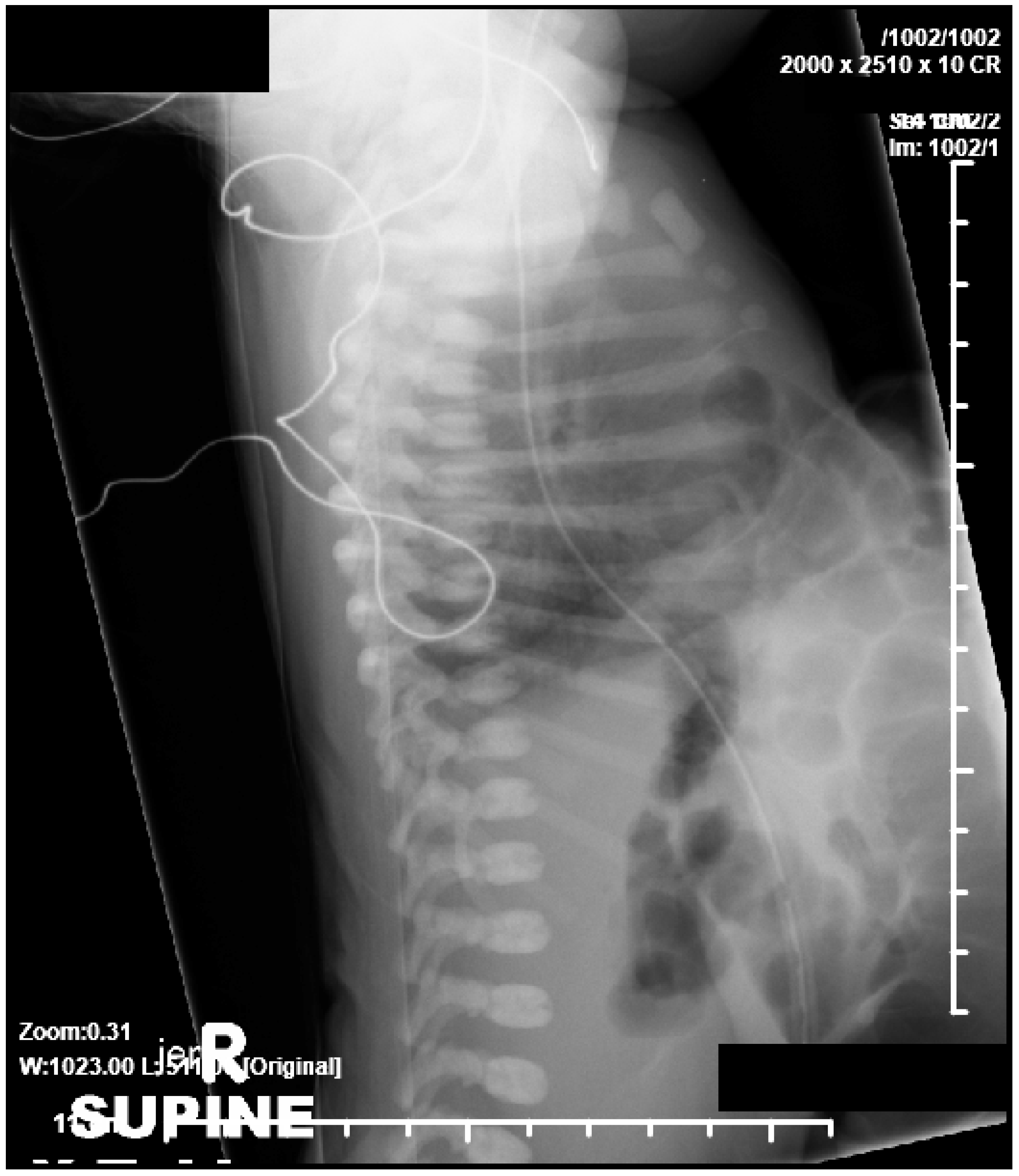
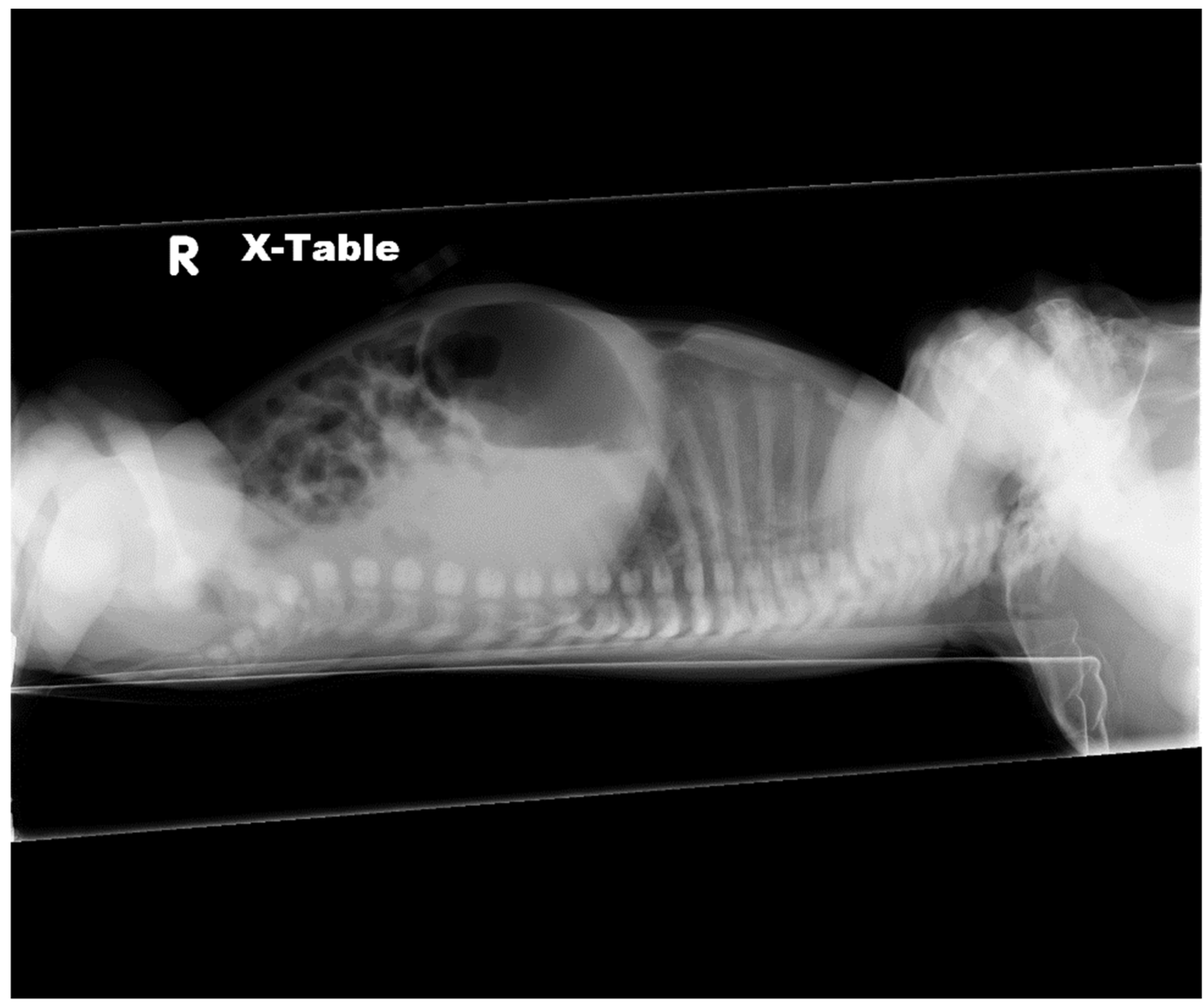
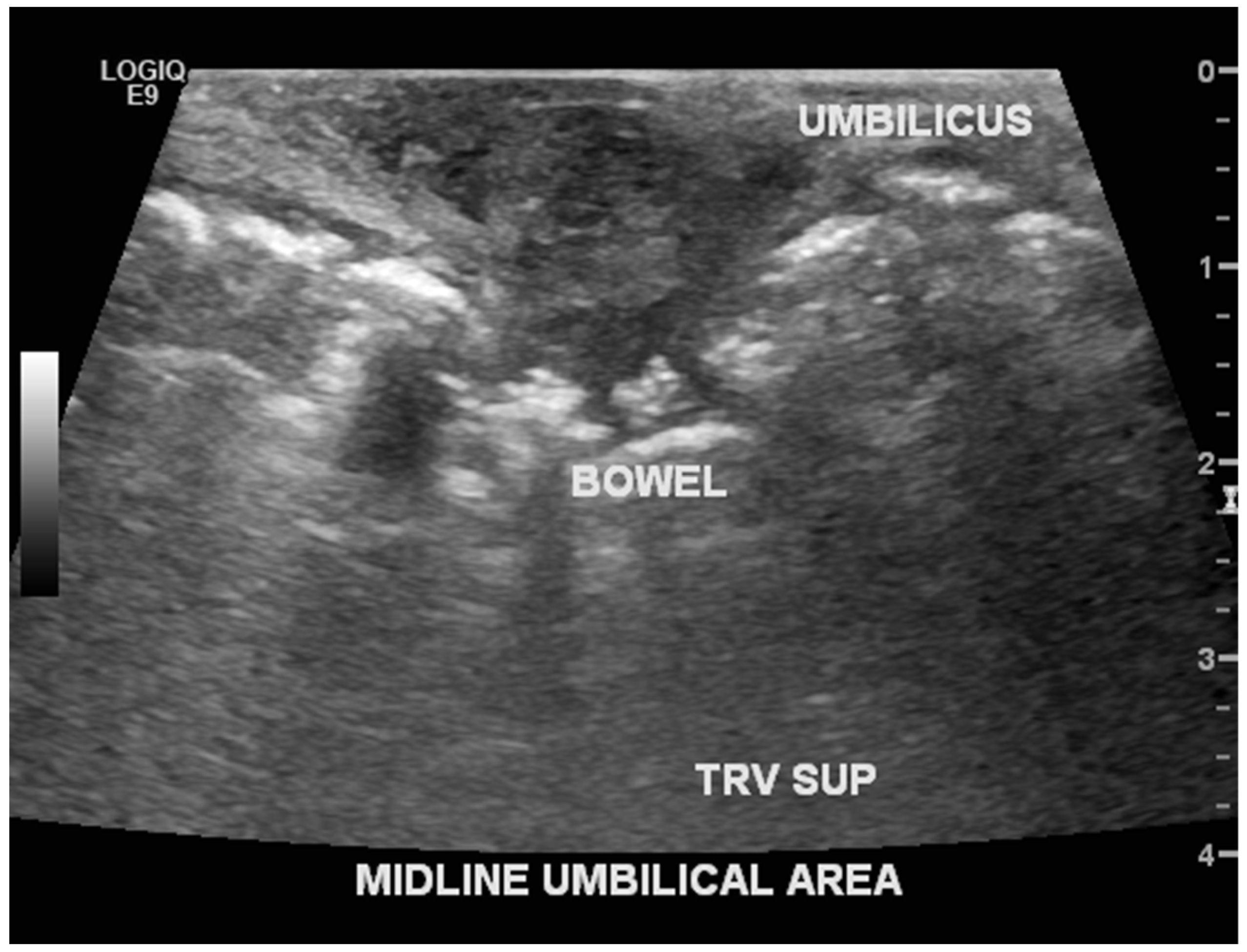
2.1. Case 1
2.2. Case 2
3. Discussion
4. Conclusions
Conflicts of Interest
References
- Carmi, R.; Boughman, J.A. Pentalogy of Cantrell and associated midline anomalies: A possible ventral midline developmental field. Am. J. Med. Genet. 1992, 42, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Cantrell, J.R.; Haller, J.A.; Ravitch, M.M. A syndrome of congenital defects involving the abdominal wall, sternum, diaphragm, pericardium, and heart. Surg. Gynecol. Obstet. 1958, 107, 602–614. [Google Scholar] [PubMed]
- Toyama, W.M. Combined congenital defects of the anterior abdominal wall, sternum, diaphragm, pericardium, and heart: A case report and review of the syndrome. Pediatrics 1972, 50, 778–792. [Google Scholar] [PubMed]
- Mărginean, C.; Mărginean, C.O.; Gozar, L.; Meliţ, L.E.; Suciu, H.; Gozar, H.; Crişan, A.; Cucerea, M. Cantrell syndrome-A rare complex congenital anomaly: A case report and literature review. Front. Pediatr. 2018, 6, 201. [Google Scholar] [CrossRef]
- Torres, U.S.; Portela-Oliveira, E.; Braga Fdel, C.; Werner, H., Jr.; Daltro, P.A.; Souza, A.S. When closure fails: What the radiologist needs to know about the embryology, anatomy, and prenatal imaging of ventral body wall defects. Semin. Ultrasound. CT MR 2015, 36, 522–536. [Google Scholar] [CrossRef]
- Engum, S.A. Embryology, sternal clefts, ectopia cordis, and Cantrell’s pentalogy. Semin. Pediatr. Surg. 2008, 17, 154–160. [Google Scholar] [CrossRef]
- Restrepo, M.S.; Cerqua, A.; Turek, J.W. Pentalogy of Cantrell with ectopia cordis totalis, total anomalous pulmonary venous connection, and tetralogy of Fallot: A case report and review of the literature. Congenit. Heart Dis. 2014, 9, E129–E134. [Google Scholar] [CrossRef]
- Singh, N.; Bera, M.L.; Sachdev, M.S.; Aggarwal, N.; Joshi, R.; Kohli, V. Pentalogy of Cantrell with left ventricular diverticulum: A case report and review of literature. Congenit. Heart Dis. 2010, 5, 454–457. [Google Scholar] [CrossRef]
- Balderrábano-Saucedo, N.; Vizcaíno-Alarcón, A.; Sandoval-Serrano, E.; Segura-Stanford, B.; Arévalo-Salas, L.A.; de la Cruz, L.R.; Espinosa-Islas, G.; Puga-Muñuzuri, F.J. Pentalogy of Cantrell: Forty-two years of experience in the Hospital Infantil de Mexico Federico Gomez. World J. Pediatr. Congenit. Heart Surg. 2011, 2, 211–218. [Google Scholar] [CrossRef]
- Mallula, K.K.; Sosnowski, C.; Awad, S. Spectrum of Cantrell’s pentalogy: Case series from a single tertiary care center and review of the literature. Pediatr. Cardiol. 2013, 34, 1703–1710. [Google Scholar] [CrossRef]
- Ojha, V.; Sh, C.; Ganga, K.P.; Saxena, A.; Gulati, G. Congenital left ventricular diverticulum in Pentalogy of Cantrell: Puzzle solved on dual source CT. Ann. Thorac. Surg. 2019, 108, e205. [Google Scholar] [CrossRef] [PubMed]
- Steiner, M.B.; Vengoechea, J.; Collins, R.T., 2nd. Duplication of the ALDH1A2 gene in association with pentalogy of Cantrell: A case report. J. Med. Case Rep. 2013, 7, 287. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, M.P.; Bulk, S.; Celli, J.; Lampe, A.; Gabbett, M.T.; Ousager, L.B.; van der Smagt, J.J.; Soller, M.; Stattin, E.L.; Mannens, M.A.; et al. Mutation update for the PORCN gene. Hum. Mutat. 2011, 32, 723–728. [Google Scholar] [CrossRef] [PubMed]
- Parvari, R.; Weinstein, Y.; Ehrlich, S.; Steinitz, M.; Carmi, R. Linkage localization of the thoraco-abdominal syndrome (TAS) gene to Xq25-26. Am. J. Med. Genet. 1994, 49, 431–434. [Google Scholar] [CrossRef]
- MacTaggart, B.; Bowen, C.; Markowitz, M.; Chong, J.; Bamshad, M.; Ma, X.; Adelstein, R.S. Using whole-exome sequencing to identify genetic variants in patients diagnosed with pentalogy of Cantrell. In Proceedings of the 30th Annual Showcase of NIH Intramural Research, Bethesda, MD, USA, 14 September 2016. [Google Scholar]
- Atis, A.; Demirayak, G.; Saglam, B.; Aksoy, F.; Sen, C. Craniorachischisis with a variant of pentalogy of Cantrell, with lung extrophy. Fetal Pediatr. Pathol. 2011, 30, 431–436. [Google Scholar] [CrossRef]
- Smigiel, R.; Jakubiak, A.; Lombardi, M.P.; Jaworski, W.; Slezak, R.; Patkowski, D.; Hennekam, R.C. Co-occurrence of severe Goltz-Gorlin syndrome and pentalogy of Cantrell—Case report and review of the literature. Am. J. Med. Genet. A 2011, 155, 1102–1105. [Google Scholar] [CrossRef]
- Madžarac, V.; Matijević, R.; Škrtić, A.; Duić, Ž.; Fistonić, N.; Partl, J.Z. Pentalogy of Cantrell with unilateral kidney evisceration: A case report and review of literature. Fetal Pediatr. Pathol. 2016, 35, 43–49. [Google Scholar] [CrossRef]
- Albu, C.; Staicu, A.; Popa-Stănilă, R.T.; Milaş, A.C.; Chiriac, L.B.; Kovács, T.E.; Stamatian, V.F. Multidisciplinary approach of assessing malformed fetuses exemplified in a rare case of pentalogy of Cantrell associated with craniorachischisis, pulmonary extrophy and right-sided aortic arch with aberrant brachiocephalic artery. Rom. J. Morphol. Embryol. 2018, 59, 911–915. [Google Scholar]
- Victoria, T.; Andronikou, S.; Bowen, D.; Laje, P.; Weiss, D.A.; Johnson, A.M.; Peranteau, W.H.; Canning, D.A.; Adzick, N.S. Fetal anterior abdominal wall defects: Prenatal imaging by magnetic resonance imaging. Pediatr. Radiol. 2018, 48, 499–512. [Google Scholar] [CrossRef]
- Türkyilmaz, G.; Avcı, S.; Sıvrıkoz, T.; Erturk, E.; Altunoglu, U.; Turkyilmazlmaz, S.E.; Kalelioglu, I.H.; Has, R.; Yuksel, A. Prenatal diagnosis and management of ectopia cordis: Varied presentation spectrum. Fetal Pediatr. Pathol. 2019, 38, 127–137. [Google Scholar] [CrossRef]
- Naburi, H.; Assenga, E.; Patel, S.; Massawe, A.; Manji, K. Class II pentalogy of Cantrell. BMC Res. Notes 2015, 29, 318. [Google Scholar] [CrossRef] [PubMed]
- Van Hoorn, J.H.; Moonen, R.M.; Huysentruyt, C.J.; van Heurn, L.W.; Offermans, J.P.; Mulder, A.L. Pentalogy of Cantrell: Two patients and a review to determine prognostic factors for optimal approach. Eur. J. Pediatr. 2008, 167, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Morales, J.M.; Patel, S.G.; Duff, J.A.; Villareal, R.L.; Simpson, J.W. Ectopia cordis and other midline defects. Ann. Thorac. Surg. 2000, 70, 111–114. [Google Scholar] [CrossRef]
- Chia, H.L.; Yeow, V.K. Repair of inferior sternal cleft using bilateral sternal bar turnover flaps in a patient with pentalogy of Cantrell. Arch. Plast. Surg. 2014, 41, 77–80. [Google Scholar] [CrossRef] [PubMed]
- Alshomer, F.; Aldaghri, F.; Alohaideb, N.; Aljehani, R.; Murad, M.A.; Hashem, F. Reconstruction of congenital sternal clefts: Surgical experience and literature review. Plast. Reconstr. Surg. Glob. Open 2017, 5, e1567. [Google Scholar] [CrossRef]

© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kylat, R.I. Complete and Incomplete Pentalogy of Cantrell. Children 2019, 6, 109. https://doi.org/10.3390/children6100109
Kylat RI. Complete and Incomplete Pentalogy of Cantrell. Children. 2019; 6(10):109. https://doi.org/10.3390/children6100109
Chicago/Turabian StyleKylat, Ranjit I. 2019. "Complete and Incomplete Pentalogy of Cantrell" Children 6, no. 10: 109. https://doi.org/10.3390/children6100109
APA StyleKylat, R. I. (2019). Complete and Incomplete Pentalogy of Cantrell. Children, 6(10), 109. https://doi.org/10.3390/children6100109




