The Relationship between Sleep and Cognitive Performance in Autism Spectrum Disorder (ASD): A Pilot Study
Abstract
1. Introduction
2. Material ad Methods
2.1. Sleep Measures
2.2. Actigraphy
2.3. CANTAB Testing
2.3.1. Motor Screening Task (MOT)
2.3.2. Intradimensional/Extradimensional (IED) Shift
2.4. Simple Reaction Time (SRT)
2.5. Statistical Analysis
3. Results
3.1. Sleep-Related Measures
3.2. Cognitive Performance
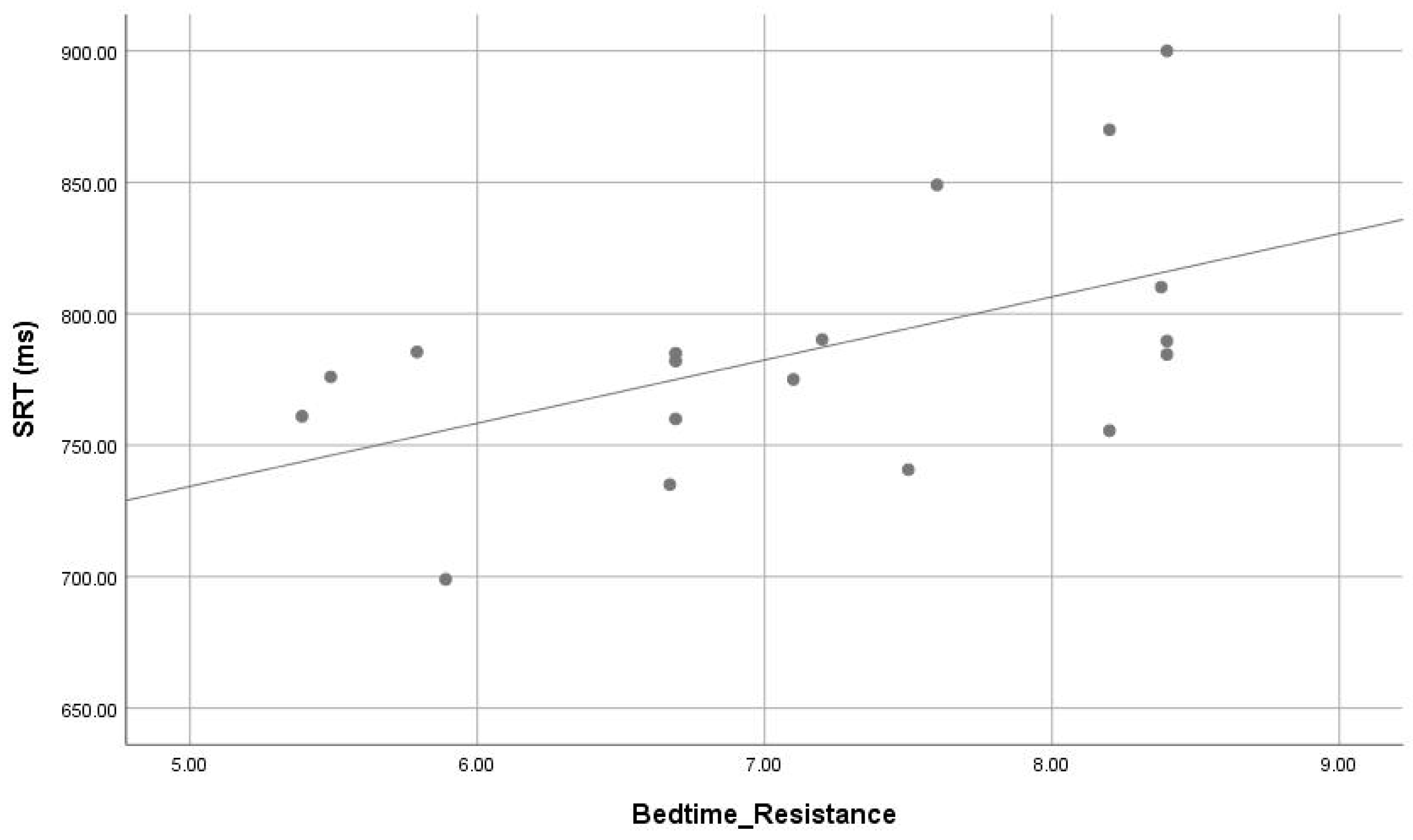
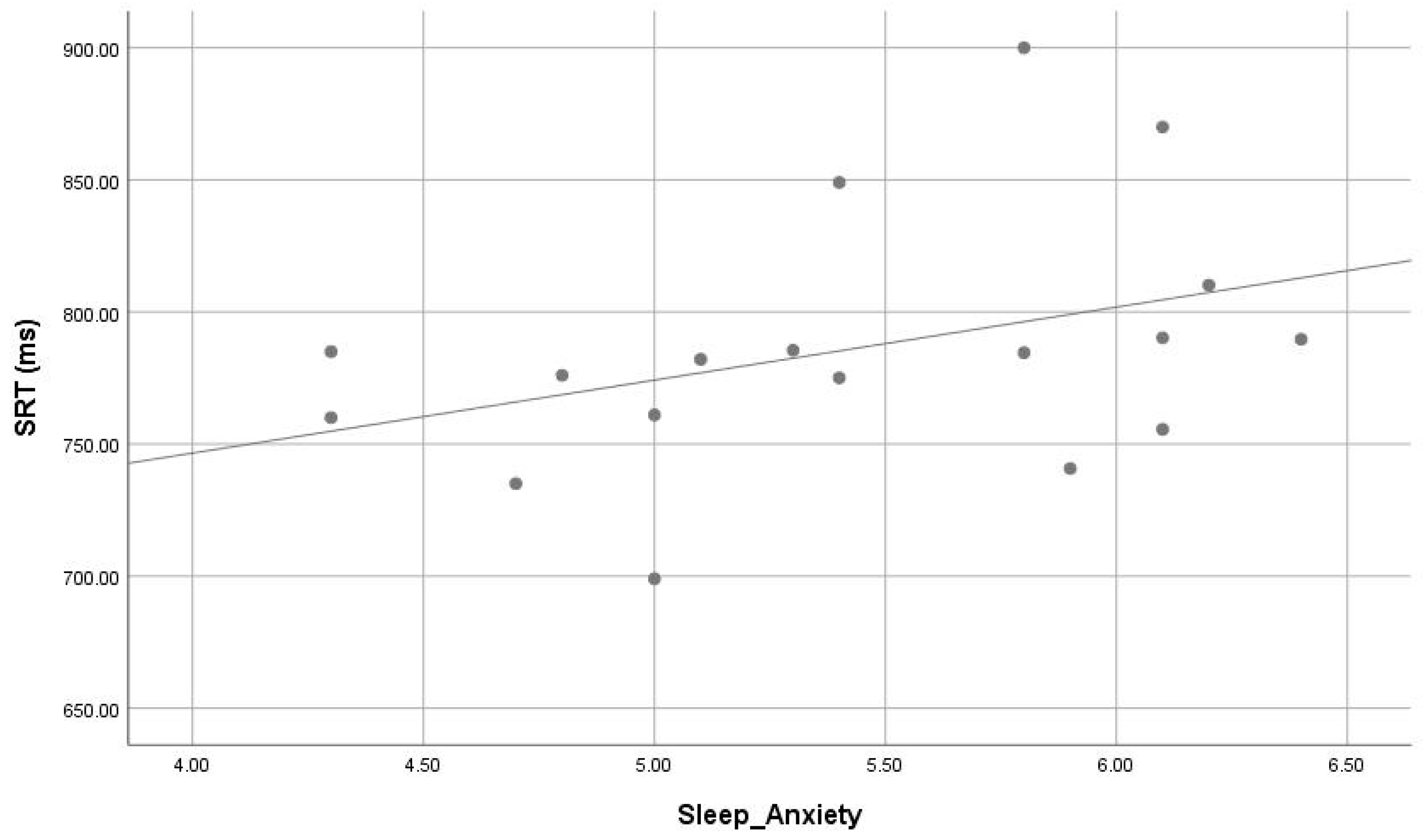
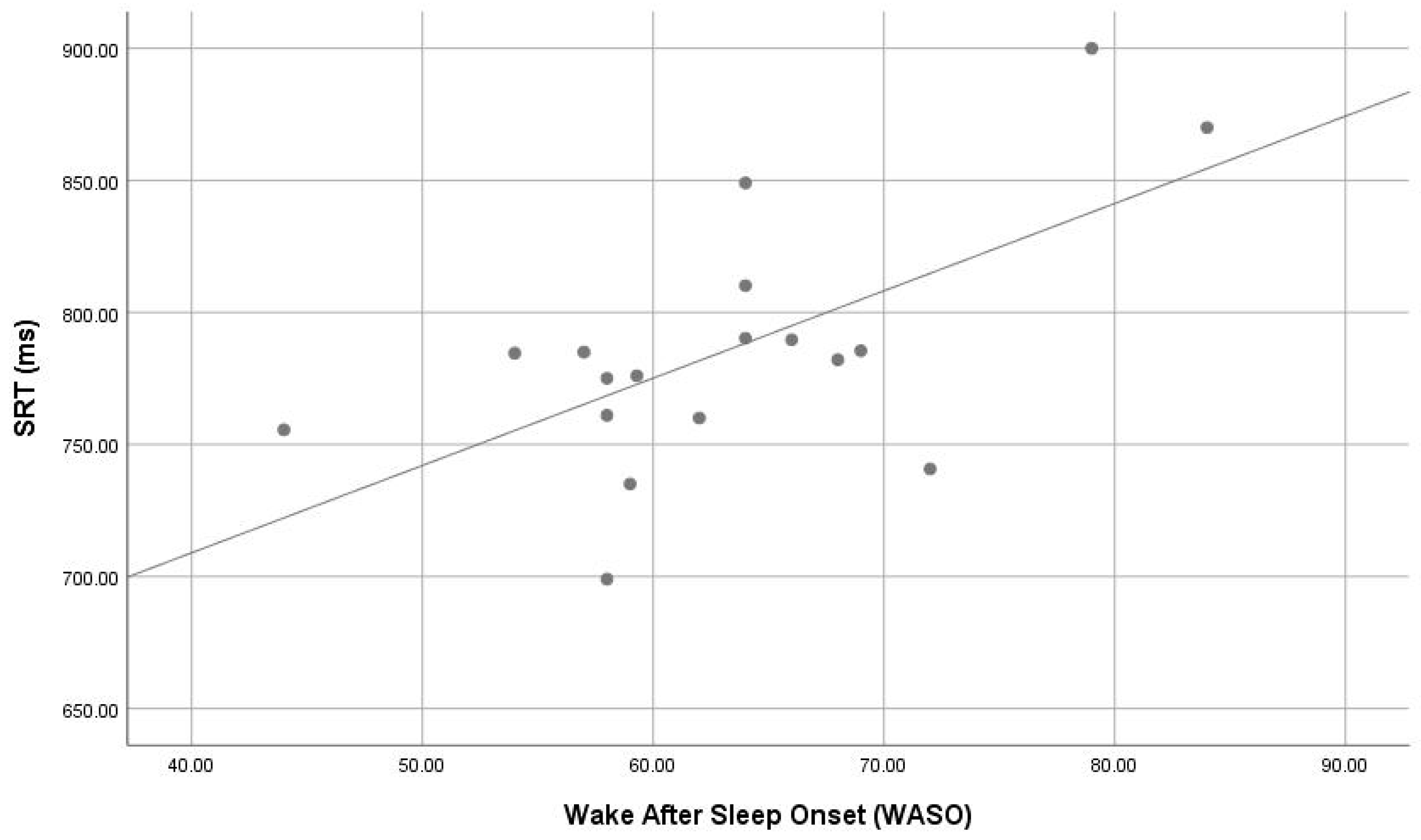
3.3. Correlation between Sleep Parameters and Cognitive Functions
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-V) 5; American Psychiatric Association: Arlington, VA, USA, 2013. [Google Scholar]
- Krakowiak, P.; Goodlin-Jones, B.; Hertz-Picciotto, I.; Croen, L.A.; Hansen, R.L. Sleep problems in children with autism spectrum disorders, developmental delays, and typical development: A population-based study. J. Sleep Res. 2008, 17, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Couturier, J.L.; Speechley, K.N.; Steele, M.; Norman, R.; Stringer, B.; Nicolson, R. Parental perception of sleep problems in children of normal intelligence with pervasive developmental disorders: Prevalence, severity, and pattern. J. Am. Acad. Child Adolesc. Psychiatry 2005, 44, 815–822. [Google Scholar] [CrossRef] [PubMed]
- Owens, J.A.; Spirito, A.; McGuinn, M. The Children’s Sleep Habits Questionnaire (CSHQ): Psychometric properties of a survey instrument for school-aged children. Sleep 2000, 23, 1043–1051. [Google Scholar] [CrossRef] [PubMed]
- Malow, B.A.; Marzec, M.; McGrew, S.G.; Wang, L.; Stone, W. Characterizing sleep in children with autism spectrum disorders: A multidimensional approach. Sleep 2006, 29, 1563–1571. [Google Scholar] [CrossRef] [PubMed]
- Goodlin-Jones, B.L.; Sitnick, S.L.; Tang, K.; Liu, J.; Anders, T.F. The children’s sleep habits questionnaire in toddlers and preschool children. J. Dev. Behav. Pediatr. 2008, 29, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Sitnick, S.L.; Goodlin-Jones, B.L.; Anders, T.F. The use of actigraphy to study sleep disorders in preschoolers: Some concerns about detection of nighttime awakenings. Sleep 2008, 31, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Wiggs, L.; Stores, G. Sleep patterns and sleep disorders in children with autistic spectrum disorders: Insights using parent report and actigraphy. Dev. Med. Child Neurol. 2004, 46, 372–380. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, G.; Johnson, C.R.; Minshew, N.J. Attentional processes in autism. J. Autism Dev. Disord. 2001, 31, 433–440. [Google Scholar] [CrossRef] [PubMed]
- Elia, M.; Ferri, R.; Musumeci, S.A.; Del Gracco, S.; Bottitta, M.; Scuderi, C.; Miano, G.; Panerai, S.; Bertrand, T.; Grubar, J.C. Sleep in subjects with autistic disorder: A neurophysiological study. Brain Dev. 2000, 22, 88–92. [Google Scholar] [CrossRef]
- Miano, S.; Ferri, R. Epidemiology and management of insomnia in children with autistic spectrum disorders. Paediatr. Drugs 2010, 12, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Schreck, K.A.; Mulick, J.A. Parental reports of sleep problems in children with autism. J. Autism Dev. Disord. 2000, 30, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Sadeh, A.; Pergamin, L.; Bar-Haim, Y. Sleep in children with attention deficit hyperactivity disorders: A meta-analysis of polysomnographic studies. Sleep Med. Rev. 2006, 10, 381–398. [Google Scholar] [CrossRef] [PubMed]
- Golombek, D.A.; Casiraghi, L.P.; Agostino, P.V.; Paladino, N.; Duhart, J.M.; Plano, S.A.; Chiesa, J.J. The times they’re a-changing: Effects of circadian desynchronization on physiology disease. J. Physiol. Paris 2013, 107, 310–322. [Google Scholar] [CrossRef] [PubMed]
- Touchette, E.; Côté, S.M.; Petit, D.; Liu, X.; Boivin, M.; Falissard, B.; Tremblay, R.E.; Montplaisir, J.Y. Short nighttime sleep-duration and hyperactivity trajectories in early childhood. Pediatrics 2009, 124, e985–e993. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.P.; Giannotti, F.; Cortesi, F. Sleep patterns in autism spectrum disorders. Child Adolesc. Psychiatr. Clin. N. Am. 2009, 18, 917–928. [Google Scholar] [CrossRef] [PubMed]
- Schwichtenberg, A.J.; Young, G.S.; Hutman, T.; Iosif, A.M.; Sigman, M.; Rogers, S.J.; Ozonoff, S. Behavior and sleep problems in children with a family history of autism. Autism Res. 2013, 6, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.A.; Schreck, K.A.; Mulick, J.A. Sleep disruption as a correlate to cognitive and adaptive behavior problems in autism spectrum disorders. Res. Dev. Disabil. 2012, 33, 1408–1417. [Google Scholar] [CrossRef] [PubMed]
- Pascualvaca, D.M.; Fantie, B.D.; Papageorgiou, M.; Mirsky, A.F. Attentional capacities in children with autism: Is there a general deficit in shifting focus? J. Autism Dev. Disord. 1998, 28, 467–478. [Google Scholar] [CrossRef] [PubMed]
- Belleville, S.; Menard, E.; Mottron, L.; Menard, M.C. Working memory in autism. In Working Memory and Neurodevelopmental Disorders; Allowa, T.P., Gathercole, S., Eds.; Psychology Press: New York, NY, USA, 2006; pp. 213–238. [Google Scholar]
- Geurts, H.M.; Verte, S.; Oosterlaan, J.; Roeyers, H.; Sergeant, J.A. How specific are executive functioning deficits in attention deficit hyperactivity disorder and autism? J. Child Psychol. Psychiatry 2004, 45, 836–854. [Google Scholar] [CrossRef] [PubMed]
- Minshew, N.J.; Luna, B.; Sweeney, J.A. Oculomotor evidence for neocortical systems but not cerebellar dysfunction in autism. Neurology 1999, 52, 917–922. [Google Scholar] [CrossRef] [PubMed]
- Mostofsky, S.H.; Goldberg, M.C.; Landa, R.J.; Denckla, M.B. Evidence for a deficit in procedural learning in children and adolescents with autism: Implications for cerebellar contribution. J. Int. Neuropsychol. Soc. 2000, 6, 752–759. [Google Scholar] [CrossRef] [PubMed]
- Luciana, M. Practitioner review: Computerized assessment of neuropsychological function in children: Clinical and research applications of the Cambridge Neuropsychological Testing Automated Battery (CANTAB). J. Child Psychol. Psychiatry 2003, 44, 649–663. [Google Scholar] [CrossRef] [PubMed]
- Open Science Collaboration. Estimating the reproducibility of psychological science. Science 2015, 349, aac4716. [Google Scholar] [CrossRef] [PubMed]
- Sahakian, B.J.; Owen, A.M. Computerized assessment in neuropsychiatry using CANTAB: Discussion paper. J. R. Soc. Med. 1992, 85, 399–402. [Google Scholar] [PubMed]
- Smith, P.J.; Need, A.C.; Cirulli, E.T.; Chiba-Falek, O.; Attix, D.K. A comparison of the Cambridge Automated Neuropsychological Test Battery (CANTAB) with “traditional” neuropsychological testing instruments. J. Clin. Exp. Neuropsychol. 2013, 35, 319–328. [Google Scholar] [CrossRef] [PubMed]
- De Rover, M.; Pironti, V.A.; McCabe, J.A.; Acosta-Cabronero, J.; Arana, F.S.; Morein-Zamir, S.; Hodges, J.R.; Robbins, T.W.; Fletcher, P.C.; Nestor, P.J.; et al. Hippocampal dysfunction in patients with mild cognitive impairment: A functional neuroimaging study of a visuospatial paired associates learning task. Neuropsychologia 2011, 49, 2060–2070. [Google Scholar] [CrossRef] [PubMed]
- Robbins, T.W.; James, M.; Owen, A.M.; Sahakian, B.J.; Lawrence, A.D.; McInnes, L.; Rabbitt, P.M. A study of performance on tests from the CANTAB battery sensitive to frontal lobe dysfunction in a large sample of normal volunteers: Implications for theories of executive functioning and cognitive aging. Cambridge Neuropsychological Test Automated Battery. J. Int. Neuropsychol. Soc. 1998, 4, 474–490. [Google Scholar] [CrossRef] [PubMed]
- De Luca, C.R.; Wood, S.J.; Anderson, V.; Buchanan, J.A.; Proffitt, T.M.; Mahony, K.; Pantelis, C. Normative data from the CANTAB. I: Development of executive function over the lifespan. J. Clin. Exp. Neuropsychol. 2003, 25, 242–254. [Google Scholar] [CrossRef] [PubMed]
- Brett, Z.H.; Humphreys, K.L.; Fleming, A.S.; Kraemer, G.W.; Drury, S.S. Using crossspecies comparisons and neurobiological framework to understand earlysocial deprivation effects on behavioral development. Dev. Psychopathol. 2015, 27, 347–367. [Google Scholar] [CrossRef] [PubMed]
- Bogaczewicz, A.; Sobow, T.; Kowalski, J.; Ząbek, J.; Woźniacka, A.; Bogaczewicz, J. Cambridge Neuropsychological Test Automated Battery in assessment of cognitive parameters in patients with systemic lupus erythematosus in relation to autoantibody profile. Reumatologia 2015, 53, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Baddam, S.K.R.; Canapari, C.A.; van Noordt, S.J.R.; Crowley, M.J. Sleep disturbances in child and adolescent mental health disorders: a review of the variability of objective sleep markers. Med. Sci. (Basel) 2018, 2, 46. [Google Scholar] [CrossRef] [PubMed]
- Mazzone, L.; Postorino, V.; Siracusano, M.; Riccioni, A.; Curatolo, P. The Relationship between Sleep Problems, Neurobiological Alterations, Core Symptoms of Autism Spectrum Disorder, and Psychiatric Comorbidities. J. Clin. Med. 2018, 5, 102. [Google Scholar] [CrossRef] [PubMed]
- Sadeh, A.; Gruber, R.; Raviv, A. Sleep, neurobehavioral functioning, and behavior problems in school-age children. Child Dev. 2002, 73, 405–417. [Google Scholar] [CrossRef] [PubMed]
- Happe, F.; Frith, U. The neuropsychology of autism. Brain 1996, 119, 1377–1400. [Google Scholar] [CrossRef] [PubMed]
- Belmonte, M.K.; Yurgelun-Todd, D.A. Functional anatomy of impaired selective attention and compensatory processing in autism. Brain Res. Cognit. Brain Res. 2003, 17, 651–664. [Google Scholar] [CrossRef]
- Luna, B.; Minshew, N.J.; Garver, K.E.; Lazar, N.A.; Thulborn, K.R.; Eddy, W.F.; Sweeney, J.A. Neocortical system abnormalities in autism: An fMRI study of spatial working memory. Neurology 2002, 59, 834–840. [Google Scholar] [CrossRef] [PubMed]
- Muller, R.A.; Kleinhans, N.; Kemmotsu, N.; Pierce, K.; Courchesne, E. Abnormal variability and distribution of functional maps in autism: An FMRI study of visuomotor learning. Am. J. Psychiatry 2003, 160, 1847–1862. [Google Scholar] [CrossRef] [PubMed]
- Giuditta, A.; Ambrosini, M.V.; Montagnese, P.; Mandile, P.; Cotugno, M.; Grassi Zucconi, G.; Vescia, S. The sequential hypothesis of the function of sleep. Behav. Brain Res. 1995, 69, 157–166. [Google Scholar] [CrossRef]
- Stickgold, R.; Whidbee, D.; Schirmer, B.; Patel, V.; Hobson, J.A. Visual discrimination task improvement: A multi-step process occurring during sleep. J. Cognit. Neurosci. 2000, 12, 246–254. [Google Scholar] [CrossRef]
| Assessment Tools | Variable | ASD Group | Mean ± Std. D | p Value |
|---|---|---|---|---|
| Age | ASD-PS | 9.4 ± 1.13 | 0.622 | |
| ASD-GS | 9.2 ± 1.05 | |||
| CSHQ | Bedtime_Resistance | ASD-PS | 7.7 ± 0.64 | 0.001 |
| ASD-GS | 6.3 ± 0.92 | |||
| Sleep_Duration | ASD-PS | 4.4 ± 0.67 | 0.021 | |
| ASD-GS | 3.6 ± 0.70 | |||
| Sleep_Anxiety | ASD-PS | 5.7 ± 0.60 | 0.019 | |
| ASD-GS | 5.03 ± 0.52 | |||
| NightWakings | ASD-PS | 5.3 ± 0.51 | 0.007 | |
| ASD-GS | 4.4 ± 0.68 | |||
| Total score | ASD-PS | 47.8 ± 3.0 | 0.001 | |
| ASD-GS | 52.8 ± 2.2 | |||
| Actigraphy | Total Sleep Time (TST) | ASD-PS | 433.9 ± 26.02 | 0.083 |
| ASD-GS | 455.9 ± 24.05 | |||
| Efficiency | ASD-PS | 66.6 ± 9.13 | 0.189 | |
| ASD-GS | 72.6 ± 9.3 | |||
| Wake After Sleep Onset (WASO) | ASD-PS | 66.7 ± 9.18 | 0.077 | |
| ASD-GS | 59.03 ± 7.6 | |||
| Number of Awakenings | ASD-PS | 22.2 ± 5.2 | 0.692 | |
| ASD-GS | 21.4 ± 2.3 |
| CANTAB Variable | ASD Group | Mean ± Std. D | Min-Max | p Value |
|---|---|---|---|---|
| IED | ASD-PS | 44.3 ± 18.8 | 12.00–63.00 | 0.789 |
| ASD-GS | 41.6 ± 22.9 | 11.00–63.00 | ||
| IEDStages | ASD-PS | 8.0 ± 1.0 | 7.00–9.00 | 0.801 |
| ASD-GS | 8.1 ± 0.9 | 7.00–9.00 | ||
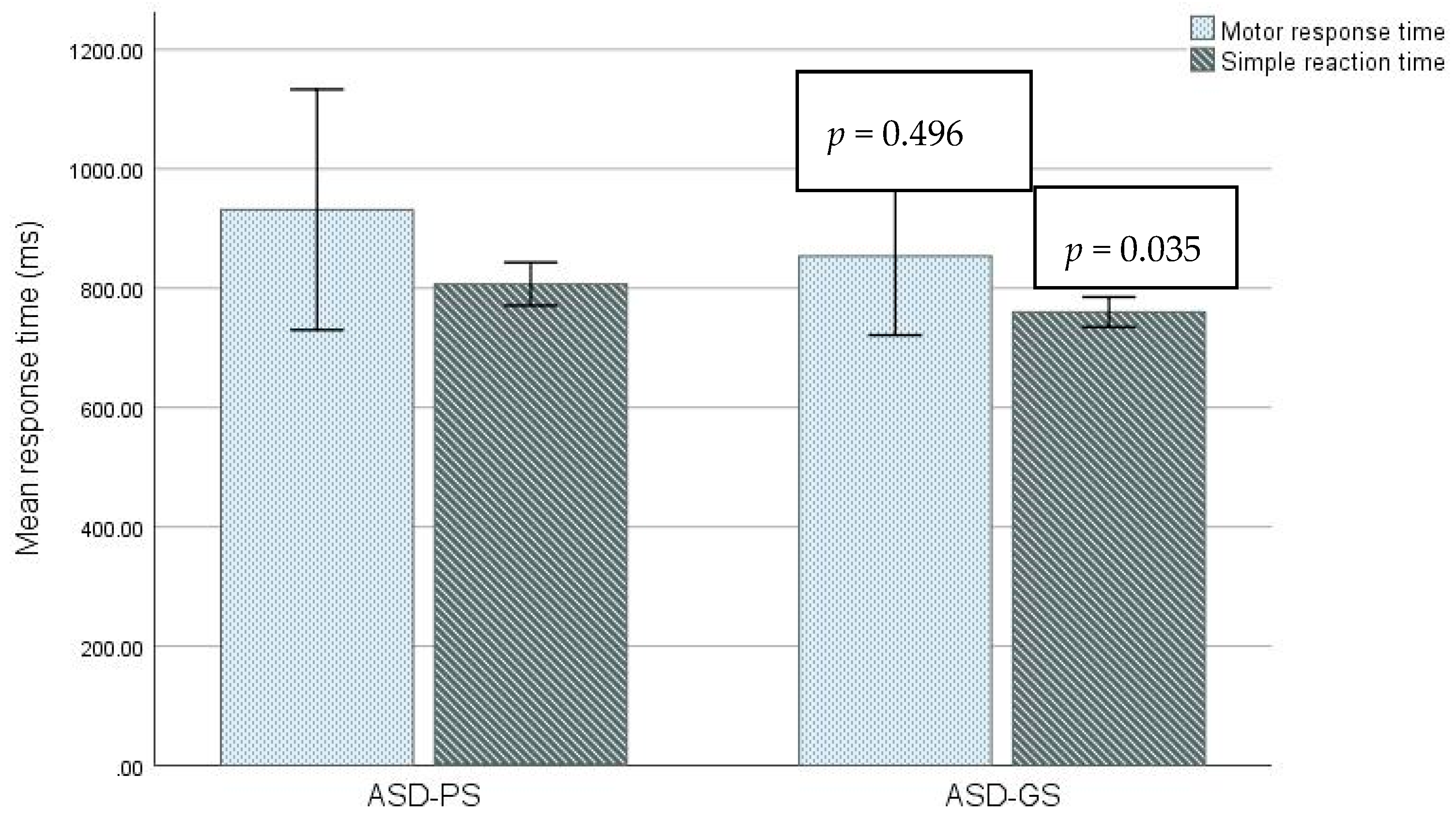
| MOT | ASD-PS | 931.4 ± 281.9 | 559.1–1394.0 | 0.496 |
| ASD-GS | 853.5 ± 158.4 | 703.7–1204.5 | ||
| MOT_Error | ASD-PS | 12.2 ± 2.0 | 9.01–15.7 | 0.193 |
| ASD-GS | 13.4 ± 1.5 | 11.7–15.7 | ||
| SRT | ASD-PS | 806.9 ± 50.6 | 740.7–900.0 | 0.035 |
| ASD-GS | 759.8 ± 30.1 | 699.0–785.4 | ||
| SRTMaximum | ASD-PS | 799.8 ± 106.8 | 698.5–998.0 | 0.130 |
| ASD-GS | 737.6 ± 27.6 | 709.5–782.5 | ||
| SRT_SD | ASD-PS | 464.4 ± 238.4 | 250.5–716.5 | 0.092 |
| ASD-GS | 631.9 ± 125.2 | 553.3–920.4 | ||
| SRT_Percentage | ASD-PS | 96.2 ± 2.3 | 92.0–100 | 0.386 |
| ASD-GS | 97.5 ± 3.5 | 92.0–100 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al Backer, N.B.; Alzawad, M.; Habibullah, H.; Bashir, S. The Relationship between Sleep and Cognitive Performance in Autism Spectrum Disorder (ASD): A Pilot Study. Children 2018, 5, 153. https://doi.org/10.3390/children5110153
Al Backer NB, Alzawad M, Habibullah H, Bashir S. The Relationship between Sleep and Cognitive Performance in Autism Spectrum Disorder (ASD): A Pilot Study. Children. 2018; 5(11):153. https://doi.org/10.3390/children5110153
Chicago/Turabian StyleAl Backer, Nouf Backer, Malak Alzawad, Hafiz Habibullah, and Shahid Bashir. 2018. "The Relationship between Sleep and Cognitive Performance in Autism Spectrum Disorder (ASD): A Pilot Study" Children 5, no. 11: 153. https://doi.org/10.3390/children5110153
APA StyleAl Backer, N. B., Alzawad, M., Habibullah, H., & Bashir, S. (2018). The Relationship between Sleep and Cognitive Performance in Autism Spectrum Disorder (ASD): A Pilot Study. Children, 5(11), 153. https://doi.org/10.3390/children5110153





