Intranasal Fentanyl in Preterm Infants Undergoing Peripherally Inserted Central Catheter Placement (INFENT PICC): A Feasibility Randomized Controlled Trial
Abstract
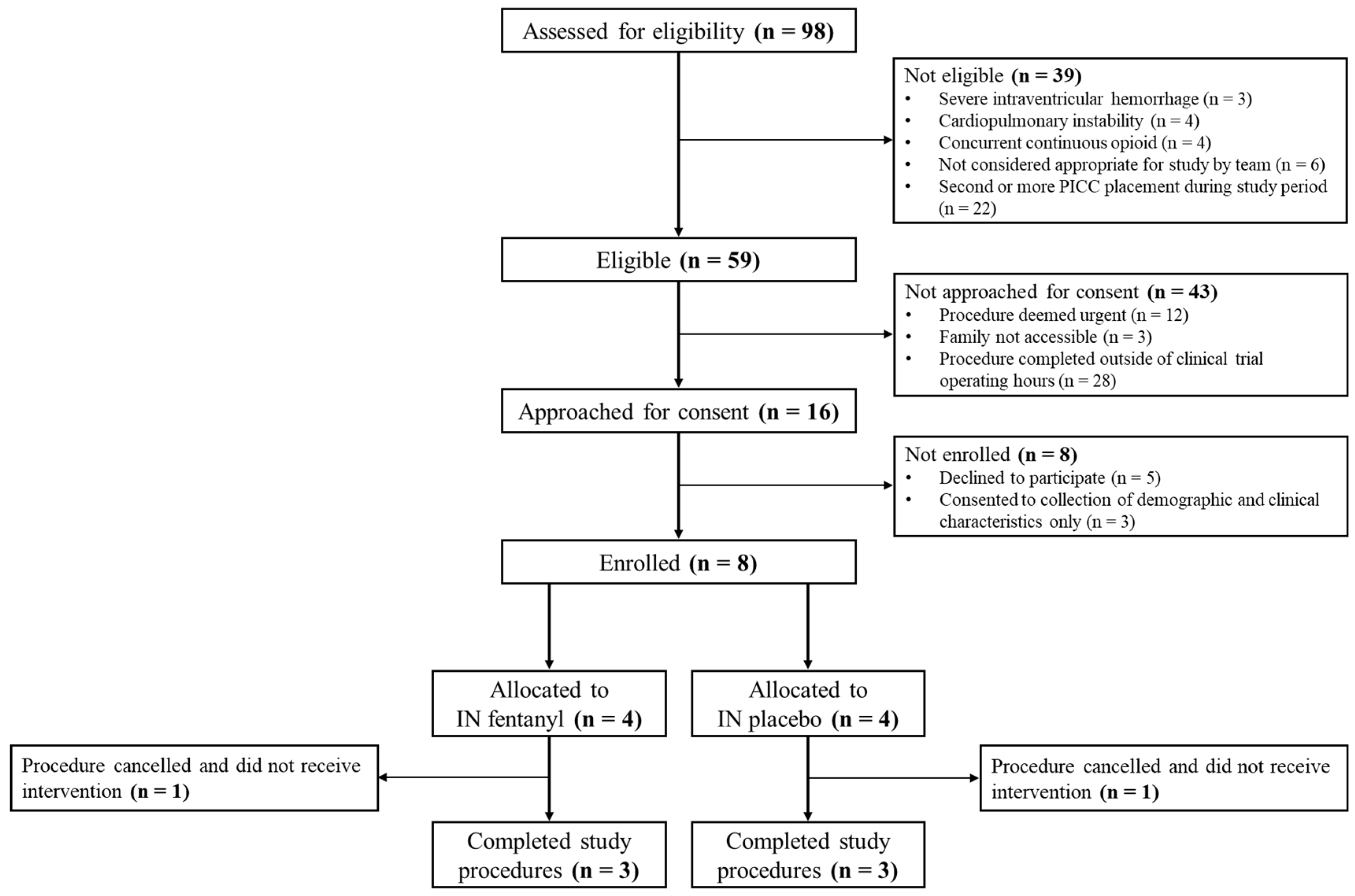
1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
2.2. Participants
2.3. Randomization
2.4. Interventions
2.4.1. Standard of Care
2.4.2. IN Fentanyl or Placebo
2.4.3. Co-Interventions
2.5. Outcomes
2.5.1. Recruitment
2.5.2. Completeness of Data Collection for Pain Score Assessment
- A consent rate of 50% and recruitment of four infants per month over a 6-month period: in 2022, there was an average of 10 PICC placements per month in infants with a GA at birth < 32 weeks at Mount Sinai Hospital based on data captured in the Canadian Neonatal Network database (personal communication). Assuming eight infants meet eligibility criteria and a 50% consent rate, the target of four infants per month was derived.
- At least 80% of video-recordings suitable for pain score assessment at the needle insertion phase of the PICC placement by blinded outcome assessors: this target was chosen as equipment failures preventing pain score assessment have been reported to be as high as 20% in previous RCTs of procedural analgesia for PICC placement [21,22,23,24,47].
2.5.3. Adverse Events
2.5.4. Acceptability and Adoption of IN Fentanyl
2.6. Sample Size
2.7. Blinding
2.8. Analytic Methods
3. Results
3.1. Study Infants
3.2. Feasibility Outcomes
3.3. Pain Assessment
3.4. Healthcare Provider Survey
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CI | Confidence interval |
| GA | Gestational age |
| ICC | Intraclass correlation coefficient |
| IQR | Interquartile range |
| IN | Intranasal |
| NICU | Neonatal intensive care unit |
| PICC | Peripherally inserted central catheter |
| PIPP-R | Premature infant pain profile-revised |
| RCT | Randomized controlled trial |
References
- Cruz, M.D.; Fernandes, A.M.; Oliveira, C.R. Epidemiology of painful procedures performed in neonates: A systematic review of observational studies. Eur. J. Pain 2016, 20, 489–498. [Google Scholar] [CrossRef] [PubMed]
- Hall, R.W.; Anand, K.J.S. Short-and long-term impact of neonatal pain and stress. Neoreviews 2005, 6, e69–e75. [Google Scholar] [CrossRef]
- Boggini, T.; Pozzoli, S.; Schiavolin, P.; Erario, R.; Mosca, F.; Brambilla, P.; Fumagalli, M. Cumulative procedural pain and brain development in very preterm infants: A systematic review of clinical and preclinical studies. Neurosci. Biobehav. Rev. 2021, 123, 320–336. [Google Scholar] [CrossRef]
- Vinall, J.; Grunau, R.E. Impact of repeated procedural pain-related stress in infants born very preterm. Pediatr. Res. 2014, 75, 584–587. [Google Scholar] [CrossRef]
- Lago, P.; Frigo, A.C.; Baraldi, E.; Pozzato, R.; Courtois, E.; Rambaud, J.; Anand, K.J.S.; Carbajal, R. Sedation and analgesia practices at Italian neonatal intensive care units: Results from the EUROPAIN study. Ital. J. Pediatr. 2017, 43, 26. [Google Scholar] [CrossRef]
- Johnston, C.; Barrington, K.J.; Taddio, A.; Carbajal, R.; Filion, F. Pain in Canadian NICUs: Have we improved over the past 12 years? Clin. J. Pain 2011, 27, 225–232. [Google Scholar] [CrossRef]
- Carbajal, R.; Rousset, A.; Danan, C.; Coquery, S.; Nolent, P.; Ducrocq, S.; Saizou, C.; Lapillonne, A.; Granier, M.; Durand, P.; et al. Epidemiology and treatment of painful procedures in neonates in intensive care units. JAMA 2008, 300, 60–70. [Google Scholar] [CrossRef]
- Harrison, D.; Loughnan, P.; Johnston, L. Pain assessment and procedural pain management practices in neonatal units in Australia. J Paediatr. Child Health 2006, 42, 6–9. [Google Scholar] [CrossRef]
- Esmaeilizand, R.; Shah, V.; Sorokan, T.; Ye, X.; Dow, K. Utilization of central lines in Canadian neonatal intensive care units. Paediatr. Child Health 2015, 20, e57. [Google Scholar] [CrossRef][Green Version]
- McCay, A.S.; Elliott, E.C.; Walden, M. PICC placement in the neonate. N. Engl. J. Med. 2014, 370, e17. [Google Scholar] [CrossRef] [PubMed]
- Costa, P.; de Camargo PP Bueno, M.; Kimura, A.F. Measuring pain in neonates during placement of central line catheter via peripheral insertion. Acta Paul. De Enferm. 2010, 23, 35–40. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, L.; Lei, X.; Dong, W.; Zhang, L. Pain-related changes in crSO2 among premature infants undergoing PICC insertion. J. Matern.-Fetal Neonatal Med. 2023, 36, 2241976. [Google Scholar] [CrossRef]
- Pillai Riddell, R.R.; Bucsea, O.; Shiff, I.; Chow, C.; Gennis, H.G.; Badovinac, S.; DiLorenzo-Klas, M.; Racine, N.M.; Kohut, S.A.; Lisi, D.; et al. Non-pharmacological management of infant and young child procedural pain. Cochrane Database Syst. Rev. 2023, 6, CD006275. [Google Scholar] [CrossRef]
- Shen, Q.; Huang, Z.; Leng, H.; Luo, X.; Zheng, X. Efficacy and safety of non-pharmacological interventions for neonatal pain: An overview of systematic reviews. BMJ Open 2022, 12, e062296. [Google Scholar] [CrossRef]
- McNair, C.; Campbell-Yeo, M.; Johnston, C.; Taddio, A. Nonpharmacologic management of pain during common needle puncture procedures in infants. Clin. Perinatol. 2019, 46, 709–730. [Google Scholar] [CrossRef] [PubMed]
- Stevens, B.; Yamada, J.; Ohlsson, A.; Haliburton, S.; Shorkey, A. Sucrose for analgesia in newborn infants undergoing painful procedures. Cochrane Database Syst. Rev. 2016, 7, CD001069. [Google Scholar] [CrossRef] [PubMed]
- Witt, N.; Coynor, S.; Edwards, C.; Bradshaw, H. A guide to pain assessment and management in the neonate. Curr. Emerg. Hosp. Med. Rep. 2016, 4, 1–10. [Google Scholar] [CrossRef]
- Gao, H.; Xu, G.; Li, F.; Lv, H.; Rong, H.; Mi, Y.; Li, M. Effect of combined pharmacological, behavioral, and physical interventions for procedural pain on salivary cortisol and neurobehavioral development in preterm infants: A randomized controlled trial. Pain 2021, 162, 253–262. [Google Scholar] [CrossRef]
- Foster, J.P.; Taylor, C.; Spence, K. Topical anaesthesia for needle-related pain in newborn infants. Cochrane Database Syst. Rev. 2017, 2, CD010331. [Google Scholar] [CrossRef]
- de Marcatto, J.O.; Vasconcelos, P.C.B.; Araujo, C.M.; Tavares, E.C.; Silva, Y.P.E. EMLA versus glucose for PICC insertion: A randomised triple-masked controlled study. Arch. Dis. Child. Fetal Neonatal Ed. 2011, 96, F467–F468. [Google Scholar] [CrossRef] [PubMed]
- Taddio, A.; Lee, C.; Yip, A.; Parvez, B.; McNamara, P.J.; Shah, V. Intravenous morphine and topical tetracaine for treatment of pain in neonates undergoing central line placement. JAMA 2006, 295, 793–800. [Google Scholar] [CrossRef]
- Lemyre, B.; Sherlock, R.; Hogan, D.; Gaboury, I.; Blanchard, C.; Moher, D. How effective is tetracaine 4% gel, before a peripherally inserted central catheter, in reducing procedural pain in infants: A randomized double-blind placebo controlled trial [ISRCTN75884221]. BMC Med. 2006, 4, 11. [Google Scholar] [CrossRef] [PubMed]
- Ballantyne, M.; McNair, C.; Ung, E.; Gibbins, S.; Stevens, B. A randomized controlled trial evaluating the efficacy of tetracaine gel for pain relief from peripherally inserted central catheters in infants. Adv. Neonatal Care 2003, 3, 297–307. [Google Scholar] [CrossRef]
- Roofthooft, D.W.E.; Simons, S.H.P.; van Lingen, R.A.; Tibboel, D.; Anker, J.N.v.D.; Reiss, I.K.; van Dijk, M. Randomized controlled trial comparing different single doses of intravenous paracetamol for placement of peripherally inserted central catheters in preterm infants. Neonatology 2017, 112, 150–158. [Google Scholar] [CrossRef]
- Lago, P.; Tiozzo, C.; Boccuzzo, G.; Allegro, A.; Zacchelo, F. Remifentanil for percutaneous intravenous central catheter placement in preterm infant: A randomized controlled trial. Pediatr. Anesth. 2008, 18, 736–744. [Google Scholar] [CrossRef]
- Nilsson, S.; Tokariev, A.; Vehviläinen, T.; Fellman, V.; Vanhatalo, S.; Norman, E. Depression of cortical neuronal activity after a low-dose fentanyl in preterm infants. Acta Paediatr. 2025, 114, 109–115. [Google Scholar] [CrossRef]
- Snyers, D.; Tribolet, S.; Rigo, V. Intranasal analgosedation for infants in the neonatal intensive care unit: A systematic review. Neonatology 2022, 119, 273–284. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; Tabbara, N.; Cheng, C.; Shah, V. Intranasal fentanyl for procedural analgesia in preterm infants. Front. Pain Res. 2022, 2, 815014. [Google Scholar] [CrossRef] [PubMed]
- McNair, C.; Graydon, B.; Taddio, A. A cohort study of intranasal fentanyl for procedural pain management in neonates. Paediatr. Child Health 2018, 23, e170–e175. [Google Scholar] [CrossRef]
- Kara, N.; Arman, D.; Seymen, Z.; Gül, A.; Cömert, S. Effects of fentanyl and sucrose on pain in retinopathy examinations with pain scale, near-infrared spectroscopy, and ultrasonography: A randomized trial. World J. Pediatr. 2023, 19, 873–882. [Google Scholar] [CrossRef]
- De Lemus-Varela, M.L.; Villaseñor-Chávez, D.A.; Gómez-Meda, B.C.; Martínez-Ruezgas, M.E.; Gallegos-Arreola, M.P.; Torres-Mendoza, B.M. Efficacy and safety of multimodal analgesic therapy to prevent pain during screening for retinopathy of prematurity. Cardiol. Vasc. Res. 2022, 4, 1–6. [Google Scholar]
- Sindhur, M.; Balasubramanian, H.; Srinivasan, L.; Kabra, N.S.; Agashe, P.; Doshi, A. Intranasal fentanyl for pain management during screening for retinopathy of prematurity in preterm infants: A randomized controlled trial. J. Perinatol. 2020, 40, 881–887. [Google Scholar] [CrossRef]
- Kaushal, S.; Placencia, J.L.; Maffei, S.R.; Chumpitazi, C.E. Intranasal fentanyl use in neonates. Hosp. Pharm. 2020, 55, 126–129. [Google Scholar] [CrossRef] [PubMed]
- Ku, L.C.; Simmons, C.; Smith, P.B.; Greenberg, R.; Fisher, K.; Hornik, C.; Cotten, C.M.; Goldberg, R.N.; Bidegain, M. Intranasal midazolam and fentanyl for procedural sedation and analgesia in infants in the neonatal intensive care unit. J. Neonatal-Perinat. Med. 2019, 12, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Thabane, L.; Ma, J.; Chu, R.; Cheng, J.; Ismaila, A.; Rios, L.P.; Robson, R.; Thabane, M.; Giangregorio, L.; Goldsmith, C.H. A tutorial on pilot studies: The what, why and how. BMC Med. Res. Methodol. 2010, 10, 1. [Google Scholar] [CrossRef] [PubMed]
- Eldridge, S.M.; Lancaster, G.A.; Campbell, M.J.; Thabane, L.; Hopewell, S.; Coleman, C.L.; Bond, C.M. Defining feasibility and pilot studies in preparation for randomised controlled trials: Development of a conceptual framework. PLoS ONE 2016, 11, e0150205. [Google Scholar] [CrossRef]
- Lancaster, G.A.; Dodd, S.; Williamson, P.R. Design and analysis of pilot studies: Recommendations for good practice. J. Eval. Clin. Pract. 2004, 10, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Turner, M.A. Clinical trials of medicines in neonates: The influence of ethical and practical issues on design and conduct. Br. J. Clin. Pharmacol. 2015, 79, 370–378. [Google Scholar] [CrossRef]
- Shaikh, H.; Lyle, A.N.J.; Oslin, E.; Gray, M.M.; Weiss, E.M. Eligible infants included in neonatal clinical trials and reasons for noninclusion: A systematic review. JAMA Netw. Open 2024, 7, e2441372. [Google Scholar] [CrossRef]
- Wilman, E.; Megone, C.; Oliver, S.; Duley, L.; Gyte, G.; Wright, J.M. The ethical issues regarding consent to clinical trials with pre-term or sick neonates: A systematic review (framework synthesis) of the empirical research. Trials 2015, 16, 502. [Google Scholar] [CrossRef]
- Megone, C.; Wilman, E.; Oliver, S.; Duley, L.; Gyte, G.; Wright, J. The ethical issues regarding consent to clinical trials with pre-term or sick neonates: A systematic review (framework synthesis) of the analytical (theoretical/philosophical) research. Trials 2016, 17, 443. [Google Scholar] [CrossRef]
- Proctor, E.; Silmere, H.; Raghavan, R.; Hovmand, P.; Aarons, G.; Bunger, A.; Griffey, R.; Hensley, M. Outcomes for implementation research: Conceptual distinctions, measurement challenges, and research agenda. Adm. Policy Ment. Health Ment. Health Serv. Res. 2011, 38, 65–76. [Google Scholar] [CrossRef] [PubMed]
- Prescott, M.G.; Iakovleva, E.; Simpson, M.R.; Pedersen, S.A.; Munblit, D.; Vallersnes, O.M.; Austad, B. Intranasal analgesia for acute moderate to severe pain in children—A systematic review and meta-analysis. BMC Pediatr. 2023, 23, 405. [Google Scholar] [CrossRef]
- Murphy, A.; O’Sullivan, R.; Wakai, A.; Grant, T.S.; Barrett, M.J.; Cronin, J.; McCoy, S.C.; Hom, J.; Kandamany, N. Intranasal fentanyl for the management of acute pain in children. Cochrane Database Syst. Rev. 2014, 10, CD009942. [Google Scholar] [CrossRef]
- Stevens, B.J.; Gibbins, S.; Yamada, J.; Dionne, K.; Lee, G.; Johnston, C.; Taddio, A. The premature infant pain profile-revised (PIPP-R): Initial validation and feasibility. Clin. J. Pain 2014, 30, 238–243. [Google Scholar] [CrossRef]
- Campbell-Yeo, M.; Carrier, L.; Benoit, B.; Kim, T.; Bueno, M.; Rao, M.; Riahi, S.; Stevens, B. Evaluation of the premature infant pain profile-revised (PIPP-R) e-learning module: Immediate and sustained competency. Adv. Neonatal Care 2022, 22, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.H.; Kim, H.S.; Lee, J.; Choi, K.Y.; Lee, J.H.; Kim, E.-K.; Park, M.S.; Choi, J.-H. A comparative study of two remifentanil doses for procedural pain in ventilated preterm infants: A randomized, controlled study. Pediatr. Crit. Care Med. 2014, 15, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Julious, S.A. Sample size of 12 per group rule of thumb for a pilot study. Pharm. Stat. 2005, 4, 287–291. [Google Scholar] [CrossRef]
- Whitehead, A.L.; Julious, S.A.; Cooper, C.L.; Campbell, M.J. Estimating the sample size for a pilot randomised trial to minimise the overall trial sample size for the external pilot and main trial for a continuous outcome variable. Stat. Methods Med. Res. 2016, 25, 1057–1073. [Google Scholar] [CrossRef]
- Brown, L.D.; Cai, T.T.; DasGupta, A. Interval estimation for a binomial proportion. Stat. Sci. 2001, 16, 101–133. [Google Scholar] [CrossRef]
- Koo, T.K.; Li, M.Y. A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J. Chiropr. Med. 2016, 15, 155–163. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. 2024. Available online: www.R-project.org (accessed on 27 August 2025).
- Canadian Paediatric Society Fetus and Newborn Committee. Managing Pain in Newborns: A Multidimensional Approach 2024. Available online: https://cps.ca/documents/position/managing-pain-in-newborns (accessed on 1 January 2025).
- Feeley, N.; Cossette, S.; Côté, J.; Héon, M.; Stremler, R.; Martorella, G.; Purden, M. The importance of piloting an RCT intervention. Can. J. Nurs. Res. 2009, 41, 85–99. [Google Scholar]
- Stevens, B.; Bueno, M.; Barwick, M.; Campbell-Yeo, M.; Chambers, C.; Estabrooks, C.; Flynn, R.; Gibbins, S.; Harrison, D.; Isaranuwatchai, W.; et al. The implementation of infant pain practice change resource to improve infant procedural pain practices: A hybrid type 1 effectiveness-implementation study. Pain 2024, 166, 1587–1596. [Google Scholar] [CrossRef] [PubMed]
- Yousef, N.; Moreau, R.; Soghier, L. Simulation in neonatal care: Towards a change in traditional training? Eur. J. Pediatr. 2022, 181, 1429–1436. [Google Scholar] [CrossRef] [PubMed]
- Franck, L.S.; Cox, S.; Allen, A.; Winter, I. Parental concern and distress about infant pain. Arch. Dis. Child. Fetal Neonatal Ed. 2004, 89, F71–F75. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Fentanyl Group (n = 4) | Control Group (n = 4) |
|---|---|---|
| Gestational age at birth, weeks | 25.5 (24.7, 26.3) | 23.4 (23, 25.3) |
| Birth weight, kg | 0.79 (0.69, 0.96) | 0.64 (0.52, 0.82) |
| Female sex | 1 (25) | 2 (50) |
| Apgar score (1 min) | 7 (5, 8) | 2 (2, 4) |
| Apgar score (5 min) | 9 (8, 9) | 9 (8, 9) |
| Postnatal age, days | 6 (5, 6) | 5 (4, 6) |
| Postmenstrual age, weeks | 26.2 (25.5, 27) | 23.9 (23.6, 25.8) |
| Weight at PICC placement, kg | 0.79 (0.63, 0.98) | 0.55 (0.52, 0.7) |
| Invasive ventilatory support | 2 (50) | 3 (75) |
| PIPP-R Score by PICC Placement Phase | Fentanyl Group (n = 3) | Control Group (n = 3) |
|---|---|---|
| Intranasal solution administration | 3 (1.5, 5) | 9 (4.5, 10) |
| Oral sucrose administration | 3 (1.5, 3) | 4 (2, 6) * |
| Skin preparation | 0 (0, 3.5) | 7 (3.5, 10) |
| Needle insertion | 0 (0, 1.5) | 9 (7.5, 11.5) |
| Second needle insertion | 3 † | 8.5 (8, 9) ‡ |
| Catheter insertion and threading | 0 (0, 1.5) | 9 (4.5, 9.5) |
| Recovery | 0 (0, 0) | 3 (1.5, 3) |
| Questions | Responses (n = 15) |
|---|---|
| Giving/observing the IN medication was stressful for me | |
| Strongly disagree | 11 (73%) |
| Disagree | 2 (13%) |
| Neither agree nor disagree | 0 |
| Agree | 1 (7%) |
| Strongly agree | 1 (7%) |
| Receiving the IN medication was stressful for the baby | |
| Strongly disagree | 8 (53%) |
| Disagree | 6 (40%) |
| Neither agree nor disagree | 1 (7%) |
| Agree | 0 |
| Strongly agree | 0 |
| What are your barriers to IN fentanyl use in preterm infants? Check all that apply | |
| Lack of comfort with IN administration route | 6 (40%) |
| Concerns about efficacy | 3 (20%) |
| Concerns about adverse events | 1 (7%) |
| What are your enablers to IN fentanyl use in preterm infants? Check all that apply | |
| Institution-specific IN fentanyl guideline | 13 (87%) |
| Additional education and training on IN administration | 7 (47%) |
| Clinical experience with IN administration | 11 (73%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tabbara, N.; McLeod, S.L.; Taddio, A.; Shah, V. Intranasal Fentanyl in Preterm Infants Undergoing Peripherally Inserted Central Catheter Placement (INFENT PICC): A Feasibility Randomized Controlled Trial. Children 2025, 12, 1156. https://doi.org/10.3390/children12091156
Tabbara N, McLeod SL, Taddio A, Shah V. Intranasal Fentanyl in Preterm Infants Undergoing Peripherally Inserted Central Catheter Placement (INFENT PICC): A Feasibility Randomized Controlled Trial. Children. 2025; 12(9):1156. https://doi.org/10.3390/children12091156
Chicago/Turabian StyleTabbara, Najla, Shelley L. McLeod, Anna Taddio, and Vibhuti Shah. 2025. "Intranasal Fentanyl in Preterm Infants Undergoing Peripherally Inserted Central Catheter Placement (INFENT PICC): A Feasibility Randomized Controlled Trial" Children 12, no. 9: 1156. https://doi.org/10.3390/children12091156
APA StyleTabbara, N., McLeod, S. L., Taddio, A., & Shah, V. (2025). Intranasal Fentanyl in Preterm Infants Undergoing Peripherally Inserted Central Catheter Placement (INFENT PICC): A Feasibility Randomized Controlled Trial. Children, 12(9), 1156. https://doi.org/10.3390/children12091156





