Sleep Stage Transitions and Sleep-Dependent Memory Consolidation in Children with Narcolepsy–Cataplexy
Abstract
:1. Introduction
Objective and Hypothesis
2. Materials and Methods
2.1. Participants
2.2. Questionnaires
2.3. Neuropsychological Assessment
2.4. Polysomnographic Recording
2.5. Stage Transitions
2.6. Sleep-Dependent Memory Consolidation Task
2.7. Statistical Analysis
3. Results
3.1. Questionnaires and Neuropsychological Assessment
3.2. Sleep Architecture
3.3. Sleep Transition
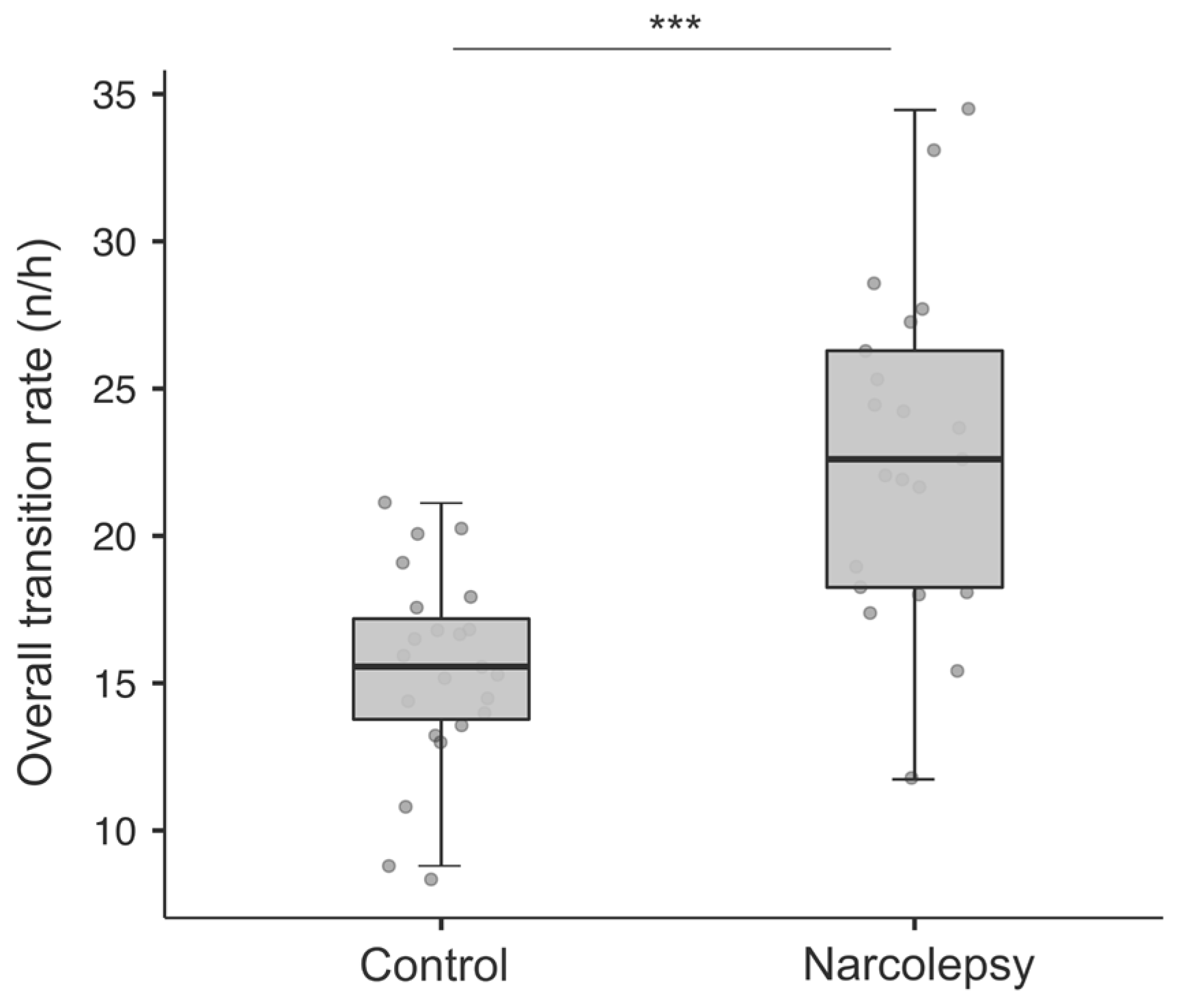
3.3.1. Overall and Stage-Specific Transition Rate (Number/Hour)
3.3.2. Global Relative Transition Frequency
3.3.3. Overnight REM Sleep Transition
3.3.4. Altered First REM Sleep Period Transition
3.3.5. Sleep-Dependent Memory Consolidation
3.3.6. Sleep Transitions and Sleep-Dependent Memory Performance
4. Discussion
4.1. Altered First REM Period Sleep Transition
4.2. Overnight REM Sleep Transitions
4.3. Memory and Sleep Transition
4.4. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Berry, R.B.; Brooks, R.; Gamaldo, C.; Harding, S.M.; Lloyd, R.M.; Quan, S.F.; Troester, M.T.; Vaughn, B.V. Aasm Scoring Manual Updates for 2017 (Version 2.4). J. Clin. Sleep Med. 2017, 13, 665–666. [Google Scholar] [CrossRef]
- Ito, E.; Inoue, Y. The International Classification of Sleep Disorders, Third Edition. American Academy of Sleep Medicine. Includes Bibliographies and Index. Nihon Rinsho 2015, 73, 916–923. [Google Scholar]
- Dye, T.J.; Gurbani, N.; Simakajornboon, N. Epidemiology and Pathophysiology of Childhood Narcolepsy. Paediatr. Respir. Rev. 2018, 25, 14–18. [Google Scholar] [CrossRef]
- Scheer, D.; Schwartz, S.W.; Parr, M.; Zgibor, J.; Sanchez-Anguiano, A.; Rajaram, L. Prevalence and Incidence of Narcolepsy in a Us Health Care Claims Database, 2008–2010. Sleep 2019, 42, zsz091. [Google Scholar] [CrossRef] [PubMed]
- Plazzi, G.; Clawges, H.M.; Owens, J.A. Clinical Characteristics and Burden of Illness in Pediatric Patients with Narcolepsy. Pediatr. Neurol. 2018, 85, 21–32. [Google Scholar] [CrossRef]
- Morse, A.M. Narcolepsy in Children and Adults: A Guide to Improved Recognition, Diagnosis and Management. Med. Sci. 2019, 7, 106. [Google Scholar] [CrossRef]
- Peyron, C.; Faraco, J.; Rogers, W.; Ripley, B.; Overeem, S.; Charnay, Y.; Nevsimalova, S.; Aldrich, M.; Reynolds, D.; Albin, R.; et al. A mutation in a case of early onset narcolepsy and a generalized absence of hypocretin peptides in human narcoleptic brains. Nat. Med. 2000, 6, 991–997. [Google Scholar] [CrossRef]
- Cvetkovic-Lopes, V.; Bayer, L.; Dorsaz, S.; Maret, S.; Pradervand, S.; Dauvilliers, Y.; Lecendreux, M.; Lammers, G.-J.; Donjacour, C.E.; Du Pasquier, R.A.; et al. Elevated Tribbles homolog 2-specific antibody levels in narcolepsy patients. J. Clin. Investig. 2010, 120, 713–719. [Google Scholar] [CrossRef]
- Mignot, E.; Lin, L.; Rogers, W.; Honda, Y.; Qiu, X.; Lin, X.; Okun, M.; Hohjoh, H.; Miki, T.; Hsu, S.H.; et al. Complex Hla-Dr and -Dq Interactions Confer Risk of Narcolepsy-Cataplexy in Three Ethnic Groups. Am. J. Hum. Genet. 2001, 68, 686–699. [Google Scholar] [CrossRef]
- Rocca, F.L.; Pizza, F.; Ricci, E.; Plazzi, G. Narcolepsy During Childhood: An Update. Neuropediatrics 2015, 46, 181–198. [Google Scholar] [CrossRef]
- Blackwell, J.E.; Alammar, H.A.; Weighall, A.R.; Kellar, I.; Nash, H.M. A Systematic Review of Cognitive Function and Psychosocial Well-Being in School-Age Children with Narcolepsy. Sleep Med. Rev. 2017, 34, 82–93. [Google Scholar] [CrossRef]
- Inocente, C.O.; Gustin, M.; Lavault, S.; Guignard-Perret, A.; Raoux, A.; Christol, N.; Gerard, D.; Dauvilliers, Y.; Reimão, R.; Bat-Pitault, F.; et al. Quality of life in children with narcolepsy. CNS Neurosci. Ther. 2014, 20, 763–771. [Google Scholar] [CrossRef] [PubMed]
- White, M.; Charbotel, B.; Fort, E.; Bastuji, H.; Franco, P.; Putois, B.; Mazza, S.; Peter-Derex, L. Academic and Professional Paths of Narcoleptic Patients: The Narcowork Study. Sleep Med. 2020, 65, 96–104. [Google Scholar] [CrossRef]
- Mignot, E.; Lammers, G.J.; Ripley, B.; Okun, M.; Nevsimalova, S.; Overeem, S.; Vankova, J.; Black, J.; Harsh, J.; Bassetti, C.; et al. The Role of Cerebrospinal Fluid Hypocretin Measurement in the Diagnosis of Narcolepsy and Other Hypersomnias. Arch. Neurol. 2002, 59, 1553–1562. [Google Scholar] [CrossRef]
- Luca, G.; Haba-Rubio, J.; Dauvilliers, Y.; Lammers, G.J.; Overeem, S.; Donjacour, C.E.; Mayer, G.; Javidi, S.; Iranzo, A.; Santamaria, J.; et al. Clinical, Polysomnographic and Genome-Wide Association Analyses of Narcolepsy with Cataplexy: A European Narcolepsy Network Study. J. Sleep Res. 2013, 22, 482–495. [Google Scholar] [CrossRef]
- Johns, M.W. Sensitivity and Specificity of the Multiple Sleep Latency Test (Mslt), the Maintenance of Wakefulness Test and the Epworth Sleepiness Scale: Failure of the Mslt as a Gold Standard. J. Sleep Res. 2000, 9, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Reiter, J.; Katz, E.; Scammell, T.E.; Maski, K. Usefulness of a Nocturnal Soremp for Diagnosing Narcolepsy with Cataplexy in a Pediatric Population. Sleep 2015, 38, 859–865. [Google Scholar] [CrossRef]
- Sorensen, G.L.; Knudsen, S.; Jennum, P. Sleep Transitions in Hypocretin-Deficient Narcolepsy. Sleep 2013, 36, 1173–1177. [Google Scholar] [CrossRef]
- Diniz Behn, C.G.; Klerman, E.B.; Mochizuki, T.; Lin, S.C.; Scammell, T.E. Abnormal Sleep/Wake Dynamics in Orexin Knockout Mice. Sleep 2010, 33, 297–306. [Google Scholar] [CrossRef]
- Pizza, F.; Vandi, S.; Iloti, M.; Franceschini, C.; Liguori, R.; Mignot, E.; Plazzi, G. Nocturnal Sleep Dynamics Identify Narcolepsy Type 1. Sleep 2015, 38, 1277–1284. [Google Scholar] [CrossRef]
- Drakatos, P.; Kosky, C.A.; Higgins, S.E.; Muza, R.T.; Williams, A.J.; Leschziner, G.D. First Rapid Eye Movement Sleep Periods and Sleep-Onset Rapid Eye Movement Periods in Sleep-Stage Sequencing of Hypersomnias. Sleep Med. 2013, 14, 897–901. [Google Scholar] [CrossRef]
- Drakatos, P.; Suri, A.; Higgins, S.E.; Ebrahim, I.O.; Muza, R.T.; Kosky, C.A.; Williams, A.J.; Leschziner, G.D. Sleep Stage Sequence Analysis of Sleep Onset Rem Periods in the Hypersomnias. J. Neurol. Neurosurg. Psychiatry 2013, 84, 223–227. [Google Scholar] [CrossRef]
- Marti, I.; Valko, P.O.; Khatami, R.; Bassetti, C.L.; Baumann, C.R. Multiple Sleep Latency Measures in Narcolepsy and Behaviourally Induced Insufficient Sleep Syndrome. Sleep Med. 2009, 10, 1146–1150. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, J.; Lam, V.; Ho, C.K.; Zhou, J.; Li, S.X.; Lam, S.P.; Yu, M.W.; Tang, X.; Wing, Y.K. Altered Sleep Stage Transitions of Rem Sleep: A Novel and Stable Biomarker of Narcolepsy. J. Clin. Sleep Med. 2015, 11, 885–894. [Google Scholar] [CrossRef]
- Thieux, M.; Zhang, M.; Marcastel, A.; Herbillon, V.; Guignard-Perret, A.; Seugnet, L.; Lin, J.S.; Guyon, A.; Plancoulaine, S.; Franco, P. Intellectual Abilities of Children with Narcolepsy. J. Clin. Med. 2020, 9, 4075. [Google Scholar] [CrossRef] [PubMed]
- Filardi, M.; D’Anselmo, A.; Agnoli, S.; Rubaltelli, E.; Mastria, S.; Mangiaruga, A.; Franceschini, C.; Pizza, F.; Corazza, G.E.; Plazzi, G. Cognitive Dysfunction in Central Disorders of Hypersomnolence: A Systematic Review. Sleep Med. Rev. 2021, 59, 101510. [Google Scholar] [CrossRef] [PubMed]
- Ingram, D.G.; Jesteadt, L.; Crisp, C.; Simon, S.L. Perceived Challenges in Pediatric Narcolepsy: A Survey of Parents, Youth, and Sleep Physicians. J. Clin. Sleep Med. 2021, 17, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Aguirre, M.; Broughton, R.; Stuss, D. Does Memory Impairment Exist in Narcolepsy-Cataplexy? J. Clin. Exp. Neuropsychol. 1985, 7, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Rogers, A.E.; Rosenberg, R.S. Tests of Memory in Narcoleptics. Sleep 1990, 13, 42–52. [Google Scholar]
- Cipolli, C.; Mazzetti, M.; Plazzi, G. Sleep-Dependent Memory Consolidation in Patients with Sleep Disorders. Sleep Med. Rev. 2013, 17, 91–103. [Google Scholar] [CrossRef]
- Mazzetti, M.; Plazzi, G.; Campi, C.; Cicchella, A.; Mattarozzi, K.; Tuozzi, G.; Vandi, S.; Vignatelli, L.; Cipolli, C. Sleep-Dependent Consolidation of Motor Skills in Patients with Narcolepsy-Cataplexy. Arch. Ital. Biol. 2012, 150, 185–193. [Google Scholar] [PubMed]
- Cipolli, C.; Campana, G.; Campi, C.; Mattarozzi, K.; Mazzetti, M.; Tuozzi, G.; Vandi, S.; Vignatelli, L.; Plazzi, G. Sleep and Time Course of Consolidation of Visual Discrimination Skills in Patients with Narcolepsy-Cataplexy. J. Sleep Res. 2009, 18, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Asp, A.; Lund, F.; Benedict, C.; Wasling, P. Impaired Procedural Memory in Narcolepsy Type 1. Acta Neurol. Scand. 2022, 146, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Strauss, M.; Griffon, L.; Van Beers, P.; Elbaz, M.; Bouziotis, J.; Sauvet, F.; Chennaoui, M.; Léger, D.; Peigneux, P. Order matters: Sleep spindles contribute to memory consolidation only when followed by rapid-eye-movement sleep. Sleep J. 2022, 45, 1–9. [Google Scholar] [CrossRef]
- Iber, C. The Aasm Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications; American Academy of Sleep Medicine: Westchester, IL, USA, 2007; Volume 1. [Google Scholar]
- Janssen, K.C.; Phillipson, S.; O’Connor, J.; Johns, M.W. Validation of the Epworth Sleepiness Scale for Children and Adolescents Using Rasch Analysis. Sleep Med. 2017, 33, 30–35. [Google Scholar] [CrossRef]
- Snow, A.; Gozal, E.; Malhotra, A.; Tiosano, D.; Perlman, R.; Vega, C.; Shahar, E.; Gozal, D.; Hochberg, Z.; Pillar, G. Severe Hypersomnolence after Pituitary/Hypothalamic Surgery in Adolescents: Clinical Characteristics and Potential Mechanisms. Pediatrics 2002, 110, e74. [Google Scholar] [CrossRef]
- Denis, I.; Turcotte, S.; Morin, C.M.; Belleville, G.; Foldes-Busque, G. A Preliminary Validation of the Pediatric Adaptation of the Insomnia Severity Index. Encephale 2023, 49, 474–480. [Google Scholar] [CrossRef]
- Helsel, W.J.; Matson, J.L. The Assessment of Depression in Children: The Internal Structure of the Child Depression Inventory (Cdi). Behav. Res. Ther. 1984, 22, 289–298. [Google Scholar] [CrossRef]
- Margalit, M. Cultural Differences in the Hyperactive Syndrome Rated in the Conners Abbreviated Scale. J. Learn. Disabil. 1981, 14, 330–331. [Google Scholar] [CrossRef]
- Wechsler, D. Wechsler Intelligence Scale for Children, 4th ed.; PsychCorp.: San Antonio, TX, USA, 2003. [Google Scholar]
- Cohen, M.J. Children’s Memory Scale; The Psychological Corporation: San Antonio, TX, USA, 1997. [Google Scholar]
- Laffan, A.; Caffo, B.; Swihart, B.J.; Punjabi, N.M. Utility of Sleep Stage Transitions in Assessing Sleep Continuity. Sleep 2010, 33, 1681–1686. [Google Scholar]
- Kishi, A.; Struzik, Z.R.; Natelson, B.H.; Togo, F.; Yamamoto, Y. Dynamics of Sleep Stage Transitions in Healthy Humans and Patients with Chronic Fatigue Syndrome. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 294, R1980-7. [Google Scholar] [CrossRef]
- Rasch, B.; Buchel, C.; Gais, S.; Born, J. Odor Cues During Slow-Wave Sleep Prompt Declarative Memory Consolidation. Science 2007, 315, 1426–1429. [Google Scholar] [CrossRef]
- Diekelmann, S.; Buchel, C.; Born, J.; Rasch, B. Labile or Stable: Opposing Consequences for Memory When Reactivated During Waking and Sleep. Nat. Neurosci. 2011, 14, 381–386. [Google Scholar] [CrossRef] [PubMed]
- de Lecea, L.; Kilduff, T.S.; Peyron, C.; Gao, X.; Foye, P.E.; Danielson, P.E.; Fukuhara, C.; Battenberg, E.L.; Gautvik, V.T.; Bartlett, F.S., 2nd; et al. The Hypocretins: Hypothalamus-Specific Peptides with Neuroexcitatory Activity. Proc. Natl. Acad. Sci. USA 1998, 95, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Saper, C.B.; Scammell, T.E.; Lu, J. Hypothalamic Regulation of Sleep and Circadian Rhythms. Nature 2005, 437, 1257–1263. [Google Scholar] [CrossRef]
- Ferri, R.; Franceschini, C.; Zucconi, M.; Drago, V.; Manconi, M.; Vandi, S.; Poli, F.; Bruni, O.; Plazzi, G. Sleep Polygraphic Study of Children and Adolescents with Narcolepsy/Cataplexy. Dev. Neuropsychol. 2009, 34, 523–538. [Google Scholar] [CrossRef]
- Ferri, R.; Miano, S.; Bruni, O.; Vankova, J.; Nevsimalova, S.; Vandi, S.; Montagna, P.; Ferini-Strambi, L.; Plazzi, G. Nrem Sleep Alterations in Narcolepsy/Cataplexy. Clin. Neurophysiol. 2005, 116, 2675–2684. [Google Scholar] [CrossRef]
- Miano, S.; Rizzoli, A.; Evangelisti, M.; Bruni, O.; Ferri, R.; Pagani, J.; Villa, M.P. Nrem Sleep Instability Changes Following Rapid Maxillary Expansion in Children with Obstructive Apnea Sleep Syndrome. Sleep Med. 2009, 10, 471–478. [Google Scholar] [CrossRef]
- Maski, K.P.; Colclasure, A.; Little, E.; Steinhart, E.; Scammell, T.E.; Navidi, W.; Behn, C.D. Stability of Nocturnal Wake and Sleep Stages Defines Central Nervous System Disorders of Hypersomnolence. Sleep 2021, 44, zsab021. [Google Scholar] [CrossRef] [PubMed]
- Pizza, F.; Moghadam, K.K.; Vandi, S.; Detto, S.; Poli, F.; Mignot, E.; Ferri, R.; Plazzi, G. Daytime Continuous Polysomnography Predicts Mslt Results in Hypersomnias of Central Origin. J. Sleep Res. 2013, 22, 32–40. [Google Scholar] [CrossRef]
- Mukai, J.; Uchida, S.; Miyazaki, S.; Nishihara, K.; Honda, Y. Spectral Analysis of All-Night Human Sleep EEG in Narcoleptic Patients and Normal Subjects. J. Sleep Res. 2003, 12, 63–71. [Google Scholar] [CrossRef]
- Christensen, J.A.; Carrillo, O.; Leary, E.B.; Peppard, P.E.; Young, T.; Sorensen, H.B.; Jennum, P.; Mignot, E. Sleep-Stage Transitions During Polysomnographic Recordings as Diagnostic Features of Type 1 Narcolepsy. Sleep Med. 2015, 16, 1558–1566. [Google Scholar] [CrossRef]
- Wada, M.; Mimura, M.; Noda, Y.; Takasu, S.; Plitman, E.; Honda, M.; Natsubori, A.; Ogyu, K.; Tarumi, R.; Graff-Guerrero, A.; et al. Neuroimaging Correlates of Narcolepsy with Cataplexy: A Systematic Review. Neurosci. Res. 2019, 142, 16–29. [Google Scholar] [CrossRef]
- Medrano-Martínez, P.; Gómez-Sacristan, Á.; Peraita-Adrados, R. Is Memory Impaired in Narcolepsy Type 1? J. Sleep Res. 2022, 31, e13593. [Google Scholar] [CrossRef]
- Giuditta, A.; Ambrosini, M.V.; Montagnese, P.; Mandile, P.; Cotugno, M.; Grassi Zucconi, G.; Vescia, S. The sequential hypothesis of the function of sleep. Behav. Brain Res. 1995, 69, 157–166. [Google Scholar] [CrossRef]
- Hahn, M.A.; Heib, D.; Schabus, M.; Hoedlmoser, K.; Helfrich, R.F. Slow Oscillation-Spindle Coupling Predicts Enhanced Memory Formation from Childhood to Adolescence. Elife 2020, 9, e53730. [Google Scholar] [CrossRef]
- Gorgoni, M.; D’Atri, A.; Scarpelli, S.; Reda, F.; De Gennaro, L. Sleep Electroencephalography and Brain Maturation: Developmental Trajectories and the Relation with Cognitive Functioning. Sleep Med. 2020, 66, 33–50. [Google Scholar] [CrossRef]
- Joechner, A.K.; Wehmeier, S.; Werkle-Bergner, M. Electrophysiological Indicators of Sleep-Associated Memory Consolidation in 5- to 6-Year-Old Children. Psychophysiology 2021, 58, e13829. [Google Scholar] [CrossRef]

| Control Children | Narcoleptic Children | |||||
|---|---|---|---|---|---|---|
| Variables | N | Score | N | Score | t/X2 Values | p Values |
| Questionnaires | ||||||
| AESS | 21 | 2.86 ± 2.76 | 19 | 15.74 ± 3.45 | −13.10 | <0.001 ** |
| ISI | 20 | 4.45 ± 2.95 | 18 | 11.72 ± 4.2 | −6.23 | <0.001 ** |
| CDI | 19 | 11.81 ± 9.46 | 18 | 10.11 ± 8.22 | 0.59 | 0.57 |
| Conners | 20 | 5.53 ± 6.20 | 18 | 9.06 ± 7.85 | −1.52 | 0.14 |
| Neuropsychological assessment | ||||||
| WISC-IV scores | ||||||
| IQ | 23 | 111.87 ± 12.74 | 16 | 115.88 ± 19.60 | −0.77 | 0.44 |
| VCI | 23 | 116.78 ± 12.68 | 19 | 126.84 ± 16.40 | −2.24 | 0.031 * |
| PRI | 23 | 109.39 ± 8.86 | 19 | 109.16 ± 16.40 | 0.06 | 0.953 |
| WMI | 23 | 102.61 ± 12.58 | 19 | 106.74 ± 12.30 | −1.07 | 0.292 |
| PSI | 23 | 103.04 ± 19.88 | 18 | 99.61 ± 2.10 | 0.55 | 0.589 |
| Daytime verbal memory: Stories | ||||||
| Immediate recall | 15 | 12.67 ± 2.32 | 11 | 11.82 ± 2.34 | 0.766 | 0.451 |
| Delayed recall | 15 | 12.47 ± 2.47 | 11 | 12 ± 3.19 | 0.42 | 0.678 |
| Delayed recognition | 14 | 13.21 ± 2.46 | 9 | 11.33 ± 2.65 | 1.741 | 0.096 |
| Daytime visual memory: Dots localization | ||||||
| Learning | 14 | 11.79 ± 1.89 | 12 | 10.25 ± 3.05 | 1.569 | 0.13 |
| Learning and immediate recall | 14 | 12.00 ± 0.39 | 12 | 10.00 ± 3.10 | 2.397 | 0.025 * |
| Delayed recall | 15 | 12.53 ± 2.07 | 12 | 10.25 ± 2.45 | 2.626 | 0.015 * |
| Control Children | Narcoleptic Children | |||||
|---|---|---|---|---|---|---|
| Variables | N | Score | N | Score | t/X2 Values | p Values |
| TST (min) | 23 | 531.57 ± 45.68 | 21 | 481.38 ± 45.68 | 2.85 | 0.007 ** |
| LAT (min) | 23 | 29.98 ± 30.73 | 21 | 5.14 ± 5.61 | 3.67 | 0.001 ** |
| Latency REM (min) | 23 | 145.7 ± 59.06 | 21 | 43.19 ± 64.1 | 5.48 | <0.001 ** |
| Sleep efficiency (%) | 23 | 92.62 ± 5.48 | 21 | 83.83 ± 5.18 | 5.46 | <0.001 ** |
| N1 (min) | 23 | 63.87 ± 24.34 | 21 | 74.68 ± 31.69 | −1.28 | 0.209 |
| N1 (%) | 23 | 11.97 ± 3.89 | 21 | 15.61 ± 6.57 | −2.26 | 0.029 * |
| N2 (min) | 23 | 238.83 ± 45.79 | 21 | 185.07 ± 32.92 | 4.43 | <0.001 ** |
| N2 (%) | 23 | 44.65 ± 5.38 | 21 | 38.43 ± 5.87 | 3.67 | 0.001 ** |
| N3 (min) | 23 | 123.61 ± 28.39 | 21 | 104.48 ± 25.17 | 2.36 | 0.023 * |
| N3 (%) | 23 | 23.54 ± 5.73 | 21 | 21.76 ± 5.24 | 1.08 | 0.288 |
| REM (min) | 23 | 104.26 ± 28.34 | 21 | 117.12 ± 28.53 | −1.5 | 0.141 |
| REM (%) | 23 | 19.67 ± 4.98 | 21 | 24.2 ± 4.81 | −3.06 | 0.004 ** |
| Arousals (nb) | 23 | 10.49 ± 3.76 | 21 | 13.92 ± 5.08 | −2.50 | 0.017 * |
| WASO (min) | 23 | 33.97 ± 28.55 | 21 | 90.64 ± 29.25 | −6.5 | <0.001 ** |
| AHOI, n/h | 21 | 0.62 ± 0.05 | ||||
| Control Children | |||||
|---|---|---|---|---|---|
| (nb/h) | W | N1 | N2 | N3 | REM |
| W | 1.97 ± 0.85 | 0.65 ± 0.41 | 0.08 ± 0.10 | 0.35 ± 0.26 | |
| N1 | 0.75 ± 0.49 | 2.77 ± 0.90 | 0.06 ± 0.08 | 1.13 ± 0.61 | |
| N2 | 1.18 ± 0.61 | 1.65 ± 0.95 | 1.16 ± 0.57 | 0.45 ± 0.20 | |
| N3 | 0.36 ± 0.17 | 0.16 ± 0.31 | 0.78 ± 0.61 | 0.00 ± 0.00 | |
| REM | 0.70 ± 0.43 | 1.06 ± 0.54 | 0.19 ± 0.20 | 0.00 ± 0.00 | |
| Narcoleptic children | |||||
| (nb/h) | W | N1 | N2 | N3 | REM |
| W | 4.40 ± 1.83 *** | 1.09 ± 0.63 ** | 0.18 ± 0.19 * | 1.06 ± 0.60 *** | |
| N1 | 1.99 ± 1.09 *** | 3.98 ± 1.90 ** | 0.03 ± 0.06 | 1.10 ± 0.51 | |
| N2 | 2.61 ± 0.91 ** | 1.83 ± 1.15 | 0.86 ± 0.41 | 0.28 ± 0.23 * | |
| N3 | 0.67 ± 0.29 ** | 0.09 ± 0.16 | 0.31 ± 0.30 ** | 0.00 ± 0.00 | |
| REM | 1.51 ± 0.65 *** | 0.71 ± 0.51 * | 0.21 ± 0.21 | 0.00 ± 0.00 | |
| Control Children | |||||
|---|---|---|---|---|---|
| (%) | Wake | N1 | N2 | N3 | REM |
| W | 12.87 ± 4,83 | 4.17 ± 2.33 | 0.53 ± 0.71 | 2.57 ± 3.32 | |
| N1 | 4.69 ± 2.84 | 17.87 ± 3.74 | 0.35 ± 0.51 | 7.26 ± 3.48 | |
| N2 | 7.86 ± 3.88 | 10.21 ± 4.89 | 7.48 ± 3.43 | 2.97 ± 1.15 | |
| N3 | 2.44 ± 1.43 | 1.03 ± 1.92 | 4.85 ± 3.88 | 0.00 ± 0.00 | |
| REM | 4.77 ± 3.08 | 6.84 ± 3.29 | 1.23 ± 1.27 | 0.00 ± 0.00 | |
| Narcoleptic children | |||||
| (%) | Wake | N1 | N2 | N3 | REM |
| W | 18.64 ± 4.98 *** | 5.36 ± 4.74 | 0.88 ± 1.02 | 5.21 ± 3.97 ** | |
| N1 | 8.53 ± 4.15 *** | 16.41 ± 5.84 | 0.10 ± 0.24 | 4.85 ± 2.04 ** | |
| N2 | 11.70 ± 4.23 ** | 7.48 ± 3.94 * | 3.72 ± 1.30 *** | 1.20 ± 0.97 *** | |
| N3 | 3.05 ± 1.46 | 0.37 ± 0.64 | 1.29 ± 1.02 ** | 0.0 ± 0.0 | |
| REM | 7.05 ± 3.89 * | 3.06 ± 2.31 *** | 1.09 ± 1.25 | 0.0 ± 0.0 | |
| Control Children | Narcoleptic Children | |
|---|---|---|
| Stage preceding REM stage (%) | ||
| W | 18.92 ± 13.71 | 42.09 ± 20.07 *** |
| N1 | 55.75 ± 18.49 | 46.21 ± 20.68 |
| N2 | 25.33 ± 13.08 | 11.71 ± 10.22 *** |
| N3 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| Number of participants entering the first REM sleep period (FREMP) from different stages | ||
| W | 0/23 (0%) | 2/21 (9.52%) |
| N1 | 0/23 (0%) | 17/21 (80.96%) |
| N2 | 20/23 (86.96%) | 1/21 (4.76%) |
| N3 | 0/23 (0%) | 0/21 (0%) |
| Unknown | 3/23 (13.04%) | 1/21 (4.76%) |
| W | 0/23 (0%) | 2/21 (9.52%) |
| MSLT1 | MSLT2 | MSLT3 | MSLT4 | MSLT5 | |
|---|---|---|---|---|---|
| No. of patients with SOREM | |||||
| 20/20 (100%) | 20/20 (100%) | 16/20 (80%) | 13/20 (65%) | 6/7 (85.71%) | |
| Stage preceding SOREM | |||||
| W | 7/20 (35.0%) | 4/20 (20%) | 3/16 (18.75%) | 2/13 (15.38%) | 1/6 (16.67%) |
| N1 | 13/20 (65.0%) | 16/20 (80%) | 13/16 (81.25%) | 11/13 (84.62%) | 5/6 (83.33%) |
| N2 | 0/21 (0%) | 0/20(0%) | 0/16 (0%) | 0/13 (0%) | 0/6 (0%) |
| N3 | 0/21 (0%) | 0/20(0%) | 0/16 (0%) | 0/13 (0%) | 0/6 (0%) |
| Control Children | Narcoleptic Children | |||||
|---|---|---|---|---|---|---|
| Variables | N | Score | N | Score | t/X2 Values | p Values |
| Learning phase (before PSG recording) | ||||||
| Number of trials | 23 | 3.48 ± 1.93 | 21 | 2.86 ± 1.20 | 1.27 | 0.211 |
| Correct answers (%) | 23 | 82.96 ± 8.37 | 21 | 82.05 ± 6.88 | 0.39 | 0.698 |
| Recall session (after PSG recording) | ||||||
| Correct answers (%) | 23 | 84.26 ± 12.84 | 21 | 74.00 ± 13.85 | 2.55 | 0.015 * |
| Memory retention score | 23 | 2.09 ± 16.40 | 21 | −9.10 ± 12.25 | 2.08 | 0.044 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gagnon, K.; Rey, A.E.; Guignard-Perret, A.; Guyon, A.; Reynaud, E.; Herbillon, V.; Lina, J.-M.; Carrier, J.; Franco, P.; Mazza, S. Sleep Stage Transitions and Sleep-Dependent Memory Consolidation in Children with Narcolepsy–Cataplexy. Children 2023, 10, 1702. https://doi.org/10.3390/children10101702
Gagnon K, Rey AE, Guignard-Perret A, Guyon A, Reynaud E, Herbillon V, Lina J-M, Carrier J, Franco P, Mazza S. Sleep Stage Transitions and Sleep-Dependent Memory Consolidation in Children with Narcolepsy–Cataplexy. Children. 2023; 10(10):1702. https://doi.org/10.3390/children10101702
Chicago/Turabian StyleGagnon, Katia, Amandine E. Rey, Anne Guignard-Perret, Aurore Guyon, Eve Reynaud, Vania Herbillon, Jean-Marc Lina, Julie Carrier, Patricia Franco, and Stéphanie Mazza. 2023. "Sleep Stage Transitions and Sleep-Dependent Memory Consolidation in Children with Narcolepsy–Cataplexy" Children 10, no. 10: 1702. https://doi.org/10.3390/children10101702
APA StyleGagnon, K., Rey, A. E., Guignard-Perret, A., Guyon, A., Reynaud, E., Herbillon, V., Lina, J.-M., Carrier, J., Franco, P., & Mazza, S. (2023). Sleep Stage Transitions and Sleep-Dependent Memory Consolidation in Children with Narcolepsy–Cataplexy. Children, 10(10), 1702. https://doi.org/10.3390/children10101702








