Monoclonal Antibodies Targeting Alarmins: A New Perspective for Biological Therapies of Severe Asthma
Abstract
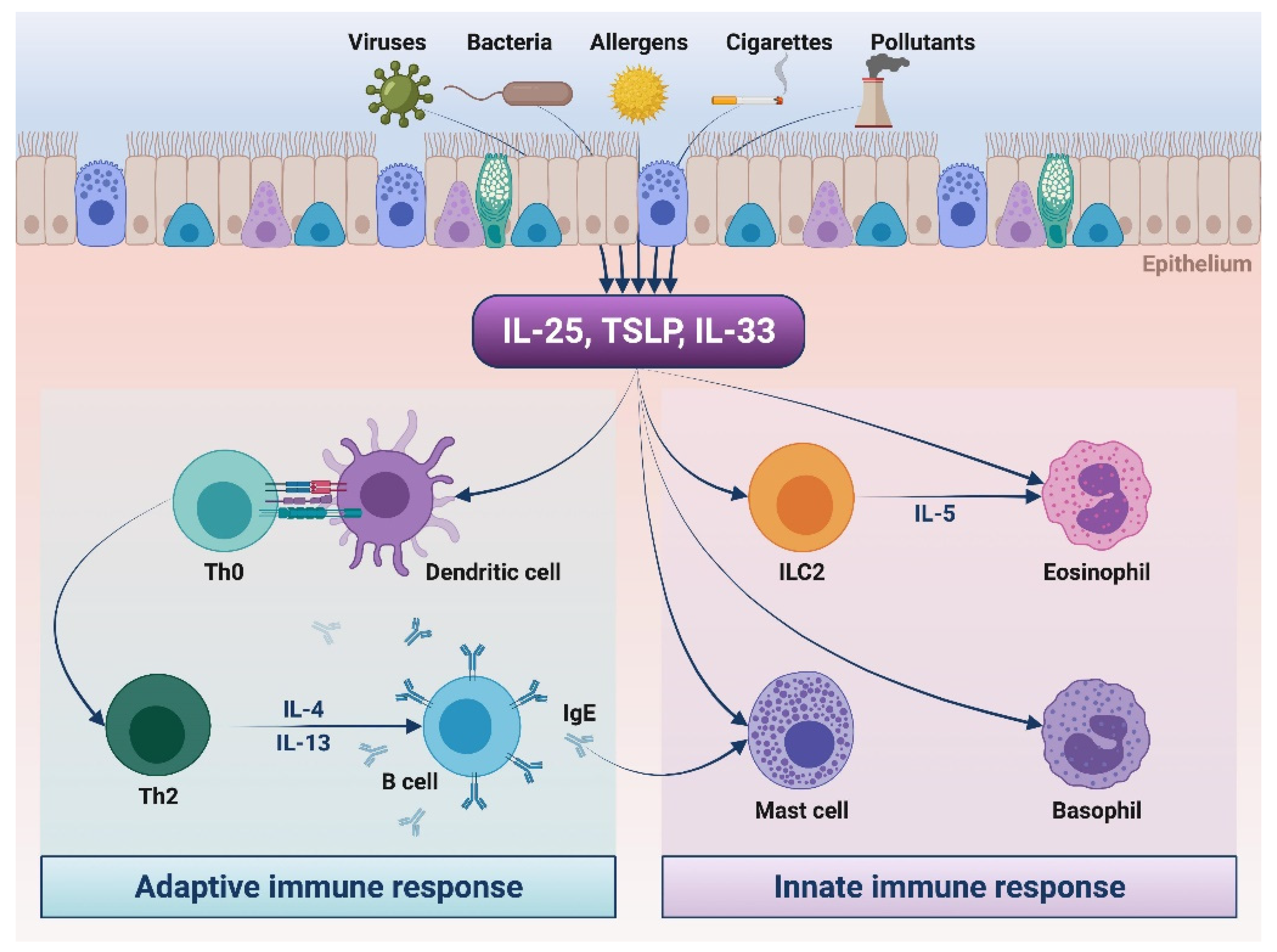
:1. Introduction
2. Pathogenic Roles of Alarmins in Asthma
2.1. TSLP
2.2. IL-33
2.3. IL-25
3. Anti-Alarmins
3.1. Anti-TSLP
3.2. Anti-IL-33
3.3. Anti-IL-25
4. Potential Therapeutic Advantages of Anti-Alarmins with Respect to Currently Available Biologics
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Khalaf, K.; Paoletti, G.; Puggioni, F.; Racca, F.; De Luca, F.; Giorgis, V.; Canonica, G.W.; Heffler, E. Asthma from immune pathogenesis to precision medicine. Semin. Immunol. 2019, 46, 101294. [Google Scholar] [CrossRef] [PubMed]
- Stern, J.; Pier, J.; Litonjua, A.A. Asthma epidemiology and risk factors. Semin. Immunopathol. 2020, 42, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Carr, T.F.; Zeki, A.A.; Kraft, M. Eosinophilic and noneosinophilic asthma. Am. J. Respir. Crit. Care Med. 2018, 197, 22–37. [Google Scholar] [CrossRef]
- Suraya, R.; Nagano, T.; Katsurada, M.; Sekiya, R.; Kobayashi, K.; Nishimura, Y. Molecular mechanism of asthma and its novel molecular target therapeutic agent. Respir. Investig. 2021, 59, 291–301. [Google Scholar] [CrossRef] [PubMed]
- Tliba, O.; Panettieri, R.A., Jr. Paucigranulocytic asthma: Uncoupling of airway obstruction from inflammation. J. Allergy Clin. Immunol. 2019, 143, 1287–1294. [Google Scholar] [CrossRef] [Green Version]
- Nelson, R.K.; Bush, A.; Stokes, J.; Nair, P.; Akuthota, P. Eosinophilic asthma. J. Allergy Clin. Immunol. Pract. 2020, 8, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Kuruvilla, M.E.; Lee, F.E.; Lee, G.B. Understanding asthma phenotypes, endotypes, and mechanisms of disease. Clin. Rev. Allergy Immunol. 2019, 56, 219–233. [Google Scholar] [CrossRef]
- Rodriguez-Rodriguez, N.; Gogoi, M.; McKenzie, A.N.J. Group 2 innate lymphoid cells: Team players in regulating asthma. Annu. Rev. Immunol. 2021, 39, 167–198. [Google Scholar] [CrossRef]
- Roan, F.; Obata-Ninomiya, K.; Ziegler, S.F. Epithelial cell-derived cytokines: More than just signaling the alarm. J. Clin. Investig. 2019, 129, 1441–1451. [Google Scholar] [CrossRef] [Green Version]
- Hong, H.; Liao, S.; Chen, F.; Yang, Q.; Wang, D.Y. Role of IL-25, IL-33, and TSLP in triggering united airway diseases toward type 2 inflammation. Allergy 2020, 75, 2794–2804. [Google Scholar] [CrossRef]
- Adhikary, P.P.; Tan, Z.; Page, B.D.G.; Hedtrich, S. TSLP as druggable target–A silver-lining for atopic diseases? Pharmacol. Ther. 2021, 217, 107648. [Google Scholar] [CrossRef]
- Calven, J.; Ax, E.; Radinger, M. The airway epithelium—A central player in asthma pathogenesis. Int. J. Mol. Sci. 2020, 21, 8907. [Google Scholar] [CrossRef]
- Hammad, H.; Lambrecht, B.N. The basic immunology of asthma. Cell 2021, 184, 1469–1485. [Google Scholar] [CrossRef]
- Tanaka, J.; Watanabe, N.; Kido, M.; Saga, K.; Akamatsu, T.; Nishio, A.; Chiba, T. Human TSLP and TLR3 ligands promote differentiation of Th17 cells with a central memory phenotype under Th2-polarizing conditions. Clin. Exp. Allergy 2009, 39, 89–100. [Google Scholar] [CrossRef]
- Gauvreau, G.M.; White, L.; Davis, B.E. Anti-alarmins entering clinical trials. Curr. Opin. Pulm. Med. 2020, 26, 69–76. [Google Scholar] [CrossRef]
- Porsbjerg, C.M.; Sverrild, A.; Lloyd, C.M.; Menzies-Gow, A.N.; Bel, E.H. Anti-alarmins in asthma: Targeting the airway epithelium with next-generation biologics. Eur Respir. J. 2020, 56, 2000260. [Google Scholar] [CrossRef] [PubMed]
- Pelaia, C.; Crimi, C.; Vatrella, A.; Tinello, C.; Terracciano, R.; Pelaia, G. Molecular targets for biological therapies of severe asthma. Front. Immunol. 2020, 11, 603312. [Google Scholar] [CrossRef]
- Albrecht, M. Turning off the alarm—Targeting alarmins and other epithelial mediators of allergic inflammation with biologics. Allergol. Select 2021, 5, 82–88. [Google Scholar] [CrossRef]
- Pelaia, C.; Pelaia, G.; Crimi, C.; Longhini, F.; Lombardo, N.; Savino, R.; Sciacqua, A.; Vatrella, A. Biologics in severe asthma. Minerva Med. 2021, in press. [Google Scholar]
- Pelaia, C.; Pelaia, G.; Crimi, C.; Maglio, A.; Gallelli, L.; Terracciano, R.; Vatrella, A. Tezepelumab: A potential new biological therapy for severe refractory asthma. Int. J. Mol. Sci. 2021, 22, 4369. [Google Scholar] [CrossRef] [PubMed]
- Cusack, R.P.; Whetstone, C.E.; Xie, Y.; Ranjbar, M.; Gauvreau, G.M. Regulation of eosinophilia in asthma–New therapeutic approaches for asthma treatment. Cells 2021, 10, 817. [Google Scholar] [CrossRef] [PubMed]
- Dorey-Stein, Z.L.; Shenoy, K.V. Tezepelumab as an emerging therapeutic option for the treatment of severe asthma: Evidence to date. Drug Des. Dev. 2021, 15, 331–338. [Google Scholar]
- Mullard, A. Tezepelumab prepares to enter the asthma antibody fray. Nat. Rev. Drug Discov. 2021, 20, 91. [Google Scholar] [PubMed]
- Corren, J.; Zieglerm, S.F. TSLP: From allergy to cancer. Nat. Immunol. 2019, 20, 1603–1609. [Google Scholar] [CrossRef]
- Tsilingiri, K.; Fornasa, G.; Rescigno, M. Thymic stromal lymphopoietin: To cut a long story short. Cell Mol. Gastroenterol. Hepatol. 2017, 3, 174–182. [Google Scholar] [CrossRef] [Green Version]
- Matera, M.G.; Rogliani, P.; Calzetta, L.; Cazzola, M. TSLP inhibitors for asthma: Current status and future prospects. Drugs 2020, 80, 449–458. [Google Scholar] [CrossRef]
- Varricchi, G.; Pecoraro, A.; Marone, G.; Criscuolo, G.; Spadaro, G.; Genovese, A.; Marone, G. Thymic stromal lymphopoietin isoforms, inflammatory disorders, and cancer. Front. Immunol. 2018, 9, 1595. [Google Scholar] [CrossRef] [Green Version]
- Markovic, I.; Savvides, S.N. Modulation of signaling mediated by TSLP and IL-7 in inflammation, autoimmune diseases, and cancer. Front. Immunol. 2020, 11, 1557. [Google Scholar] [CrossRef] [PubMed]
- Gauvreau, G.M.; Sehmi, R.; Ambrose, C.S.; Griffiths, J.M. Thymic stromal lymphopoietin: Its role and potential as a therapeutic target in asthma. Expert Opin. Ther. Targets 2020, 24, 777–792. [Google Scholar] [CrossRef] [PubMed]
- Berraies, A.; Hamdi, B.; Ammar, J.; Hamzaoui, K.; Hamzaoui, A. Increased expression of thymic stromal lymphopoietin in induced sputum from asthmatic children. Immunol. Lett. 2016, 178, 85–91. [Google Scholar] [CrossRef]
- Gluck, J.; Rymarczyk, B.; Kasprzak, M.; Rogala, B. Increased levels of interleukin-33 and thymic stromal lymphopoietin in exhaled breath condensate in chronic bronchial asthma. Int. Arch. Allergy Immunol. 2016, 169, 51–56. [Google Scholar] [CrossRef]
- Li, Y.; Wang, W.; Lv, Z.; Li, Y.; Chen, Y.; Huang, K.; Corrigan, C.J.; Ying, S. Elevated expression of IL-33 and TSLP in the airways of human asthmatics in vivo: A potential biomarker of severe refractory disease. J. Immunol. 2018, 200, 2253–2262. [Google Scholar] [CrossRef] [Green Version]
- Torgerson, D.G.; Ampleford, E.J.; Chiu, G.Y.; Gauderman, W.J.; Gignoux, C.R.; Graves, P.E.; Himes, B.E.; Levin, A.M.; Mathias, R.A.; Hancock, D.B.; et al. Meta-analysis of genome-wide association studies of asthma in ethnically diverse North American populations. Nat. Genet. 2011, 43, 887–892. [Google Scholar] [PubMed]
- Hirota, T.; Takahashi, A.; Kubo, M.; Tsunoda, T.; Tomita, K.; Doi, S.; Fujita, K.; Miyatake, A.; Enomoto, T.; Miyagawa, T.; et al. Genome-wide association study identifies three new susceptibility loci for adult asthma in the Japanese population. Nat. Genet. 2011, 43, 893–896. [Google Scholar] [CrossRef]
- Buchheit, K.M.; Cahill, K.N.; Katz, H.R.; Murphy, K.C.; Feng, C.; Lee-Sarwar, K.; Lai, J.; Bhattacharyya, N.; Israel, E.; Boyce, J.; et al. Thymic stromal lymphopoietin controls prostaglandin D2 generation in patients with aspirin-exacerbated respiratory disease. J. Allergy Clin. Immunol. 2016, 137, 1566–1576.e5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lambrecht, B.N.; Hammad, H.; Fahy, J.V. The cytokines of asthma. Immunity 2019, 50, 975–991. [Google Scholar] [CrossRef] [PubMed]
- Klose, C.S.N.; Artis, D. Innate lymphoid cells as regulators of immunity, inflammation and tissue homeostasis. Nat. Immunol. 2016, 17, 765–774. [Google Scholar] [CrossRef]
- Camelo, A.; Rosignoli, G.; Ohne, Y.; Stewart, R.A.; Overed-Sayer, C.; Sleeman, M.A.; May, R.D. IL-33, IL-25, and TSLP induce a distinct phenotypic and activation profile in human type 2 innate lymphoid cells. Blood Adv. 2017, 1, 577–589. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pelaia, C.; Paoletti, G.; Puggioni, F.; Racca, F.; Pelaia, G.; Canonica, G.W.; Heffler, E. Interleukin-5 in the pathophysiology of severe asthma. Front. Physiol. 2019, 10, 1514. [Google Scholar] [CrossRef] [PubMed]
- Shikotra, A.; Choy, D.F.; Ohri, C.M.; Doran, E.; Butler, C.; Hargadon, B.; Shelley, M.; Abbas, A.R.; Austin, C.D.; Jackman, J.; et al. Increased expression of immunoreactive thymic stromal lymphopoietin in patients with severe asthma. J. Allergy Clin. Immunol. 2012, 129, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.J.; Fu, C.H.; Wang, C.H.; Huang, C.C.; Huang, C.C.; Chang, P.H.; Chen, Y.W.; Wu, C.C.; Wu, C.L.; Kuo, H.P. Impact of chronic rhinosinusitis on severe asthma patients. PLoS ONE 2017, 12, e0171047. [Google Scholar] [CrossRef]
- Liu, S.; Verma, M.; Michalec, L.; Liu, W.; Sripada, A.; Rollins, D.; Good, J.; Ito, Y.; Chu, H.; Gorska, M.M.; et al. Steroid resistance of airway type 2 innate lymphoid cells from patients with severe asthma: The role of thymic stromal lymphopoietin. J. Allergy Clin. Immunol. 2018, 141, 257–268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, C.K.; Hu, S.; Cheung, P.F.; Lam, C.W. Thymic stromal lymphopoietin induces chemotactic and prosurvival effects in eosinophils: Implications in allergic inflammation. Am. J. Respir. Cell Mol. Biol. 2010, 43, 305–315. [Google Scholar] [CrossRef] [PubMed]
- Cook, E.B.; Stahl, J.L.; Schwantes, E.A.; Fox, K.E.; Mathur, S.K. IL-3 and TNF-α increase thymic stromal lymphopoietin receptor (TSLPR) expression on eosinophils and enhance TSLP-stimulated degranulation. Clin. Mol. Allergy 2012, 10, 8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Sajee, D.; Sehmi, R.; Hawke, T.J.; El-Gammal, A.; Howie, K.J.; Watson, R.M.; Londei, M.; Gauvreau, G.M.; O’Byrne, P.M. Expression of IL-33 and TSLP and their receptors in asthmatic airways after inhaled allergen challenge. Am. J. Respir. Crit. Care Med. 2018, 198, 805–807. [Google Scholar] [CrossRef]
- Morshed, M.; Yousefi, S.; Stockle, C.; Simon, H.U.; Simon, D. Thymic stromal lymphopoietin stimulates the formation of eosinophil extracellular traps. Allergy 2012, 67, 1127–1137. [Google Scholar] [CrossRef]
- Salter, B.M.A.; Smith, S.G.; Mukherjee, M.; Plante, S.; Krisna, S.; Nusca, G.; Oliveria, J.P.; Irshad, A.; Gauvreau, G.M.; Chakir, J.; et al. Human bronchial epithelial cell-derived factors from severe asthmatic subjects stimulate eosinophil differentiation. Am. J. Respir. Cell Mol. Biol 2018, 58, 99–106. [Google Scholar] [CrossRef]
- Smith, S.G.; Gugilla, A.; Mukherjee, M.; Merim, K.; Irshad, A.; Tang, W.; Kinoshita, T.; Watson, B.; Oliveria, J.P.; Comeau, M.; et al. Thymic stromal lymphopoietin and IL-33 modulate migration of hematopoietic progenitor cells in patients with allergic asthma. J. Allergy Clin. Immunol. 2015, 135, 1594–1602. [Google Scholar] [CrossRef] [PubMed]
- Salter, B.M.; Oliveria, J.P.; Nusca, G.; Smith, S.G.; Watson, R.M.; Comeau, M.; Sehmi, R.; Gauvreau, G.M. Thymic stromal lymphopoietin activation of basophils in patients with allergic asthma is IL-3 dependent. J. Allergy Clin. Immunol. 2015, 136, 1636–1644. [Google Scholar] [CrossRef] [PubMed]
- Salter, B.M.; Oliveria, J.P.; Nusca, G.; Smith, S.G.; Tworek, D.; Mitchell, P.D.; Watson, R.M.; Sehmi, R.; Gauvreau, G.M. IL-25 and IL-33 induce type 2 inflammation in basophils from subjects with allergic asthma. Respir. Res. 2016, 17, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagarkar, D.R.; Poposki, J.A.; Comeau, M.R.; Biyasheva, A.; Avila, P.C.; Schleimer, R.P.; Kato, A. Airway epithelial cells activate Th2 cytokine production in mast cells through IL-1 and thymic stromal lymphopoietin. J. Allergy Clin. Immunol. 2012, 130, 225–232.e4. [Google Scholar] [CrossRef] [Green Version]
- Pelaia, C.; Crimi, C.; Vatrella, A.; Busceti, M.T.; Gaudio, A.; Garofalo, E.; Bruni, A.; Terracciano, R.; Pelaia, G. New treatments for asthma: From the pathogenic role of prostaglandin D2 to the therapeutic effects of fevipiprant. Pharmacol. Res. 2020, 155, 104490. [Google Scholar] [CrossRef]
- Soumelis, V.; Reche, P.A.; Kanzler, H.; Yuan, W.; Edward, G.; Homey, B.; Gilliet, M.; Ho, S.; Antonenko, S.; Lauerma, A.; et al. Human epithelial cells trigger dendritic cell-mediated allergic inflammation by producing TSLP. Nat. Immunol. 2002, 3, 673–680. [Google Scholar] [CrossRef]
- Barnes, P.J. The cytokine network in asthma and chronic obstructive pulmonary disease. J. Clin. Investig. 2008, 118, 3546–3556. [Google Scholar] [CrossRef] [Green Version]
- Watanabe, N.; Hanabuchi, S.; Soumelis, V.; Yuan, W.; Ho, S.; de Waal Malefyt, R.; Liu, Y.J. Human thymic stromal lymphopoietin promotes dendritic cell-mediated CD4+ T cell homeostatic expansion. Nat. Immunol. 2004, 5, 426–434. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Wang, Y.H.; Duramad, O.; Hori, T.; Delespesse, G.J.; Watanabe, N.; Qin, F.X.; Yao, Z.; Cao, W.; Liu, Y.J. TSLP-activated dendritic cells induce an inflammatory T helper type 2 cell response through OX40 ligand. J. Exp. Med. 2005, 202, 1213–1223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pattarini, L.; Trichot, C.; Bogiatzi, S.; Grandclaudon, M.; Meller, S.; Keuylian, Z.; Durand, M.; Volpe, E.; Madonna, S.; Cavani, A.; et al. TSLP-activated dendritic cells induce human T follicular helper cell differentiation through OX40-ligand. J. Exp. Med. 2017, 214, 1529–1546. [Google Scholar] [CrossRef] [Green Version]
- Lai, J.F.; Thompson, L.J.; Ziegler, S.F. TSLP drives acute TH2-cell differentiation in lungs. J. Allergy Clin. Immunol. 2020, 146, 1406–1418.e7. [Google Scholar] [CrossRef] [PubMed]
- Rochman, Y.; Dienger-Stambaugh, K.; Richgels, P.K.; Lewkowich, I.P.; Kartashov, A.V.; Barski, A.; Khurana Hershey, G.K.; Leonard, W.J.; Singh, H. TSLP signaling in CD4(+) T cells programs a pathogenic T helper 2 cell state. Sci. Signal. 2018, 11, 521. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, K.D.; Vanichsarn, C.; Nadeau, K.C. TSLP directly impairs pulmonary Treg function: Association with aberrant tolerogenic immunity in asthmatic airway. Allergy Asthma Clin. Immunol. 2010, 6, 4. [Google Scholar] [CrossRef] [Green Version]
- Hinks, T.S.C.; Levine, S.J.; Brusselle, G.G. Treatment options in type-2 low asthma. Eur. Respir. J. 2021, 57, 2000528. [Google Scholar] [CrossRef]
- Liang, Y.; Bihui, Y.; Chen, J.; Wu, H.; Xu, Y.; Yang, B.; Lu, Q. Thymic stromal lymphopoietin epigenetically upregulates Fc receptor γ subunit-related receptors on antigen-presenting cells and induces TH2/TH17 polarization through dectin-2. J. Allergy Clin. Immunol. 2019, 144, 1025–1035.e7. [Google Scholar] [CrossRef]
- Semlali, A.; Jacques, E.; Koussih, L.; Gounni, A.S.; Chakir, J. Thymic stromal lymphopoietin-induced human asthmatic airway epithelial cell proliferation through an IL-13-dependent pathway. J. Allergy Clin. Immunol. 2010, 125, 844–850. [Google Scholar] [CrossRef]
- Redhu, N.S.; Gounni, A.S. Function and mechanisms of TSLP/TSLPR complex in asthma and COPD. Clin. Exp. Allergy 2012, 42, 994–1005. [Google Scholar] [CrossRef]
- Shan, L.; Redhu, N.S.; Saleh, A.; Halayko, A.J.; Chakir, J.; Gounni, A.S. Thymic stromal lymphopoietin receptor-mediated IL-6 and CC/CXC chemokines expression in human airway smoot muscle: Role of MAPKs (ERK1/2, p38, and JNK) and STAT3 pathways. J. Immunol. 2010, 184, 7134–7143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Allakhverdi, Z.; Comeau, M.R.; Jessup, H.K.; Delespesse, G. Thymic stromal lymphopoietin as a mediator of crosstalk between bronchial smooth muscles and mast cells. J. Allergy Clin. Immunol. 2009, 123, 958–960.e2. [Google Scholar] [CrossRef]
- Cao, L.; Liu, F.; Liu, Y.; Liu, T.; Wu, J.; Zhao, J.; Wang, J.; Li, S.; Xu, J.; Dong, L. TSLP promotes asthmatic airway remodeling via p38-STAT3 signaling pathway in human lung fibroblasts. Exp. Lung Res. 2018, 44, 288–301. [Google Scholar] [CrossRef] [PubMed]
- Wieczfinska, J.; Pawliczak, R. Thymic stromal lymphopoietin and apocynin alter the expression of airway remodeling factors in human rhinovirus-infected cells. Immunobiology 2017, 222, 892–899. [Google Scholar] [CrossRef] [PubMed]
- Pinto, S.M.; Subbannayya, Y.; Rex, D.A.B.; Raju, R.; Chatterjee, O.; Advani, J.; Radhakrishnan, A.; Keshava Prasad, T.S.; Wani, M.R.; Pandey, A. A network map of IL-33 signaling pathway. J. Cell Commun. Signal. 2018, 12, 615–624. [Google Scholar] [CrossRef] [PubMed]
- Momen, T.; Ahanchian, H.; Reisi, M.; Shamsdin, S.A.; Shahsanai, A.; Keivanfar, M. Comparison of interleukin-33 serum levels in asthmatic patients with a control group and relation with the severity of the disease. Int. J. Prev. Med. 2017, 8, 65. [Google Scholar] [PubMed]
- Prefontaine, D.; Lajoie-Kadoch, S.; Foley, S.; Audusseau, S.; Olivenstein, R.; Halayko, A.J.; Lemière, C.; Martin, J.G.; Hamid, Q. Increased expression of IL-33 in severe asthma: Evidence of expression by airway smooth muscle cells. J. Immunol. 2009, 183, 5094–5103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaur, D.; Gomez, E.; Doe, C.; Berair, R.; Woodman, L.; Saunders, R.; Hollins, F.; Rose, F.R.; Amrani, Y.; May, R.; et al. IL-33 drives airway hyper-responsiveness through IL-13-mediated mast cell: Airway smooth muscle crosstalk. Allergy 2015, 70, 556–567. [Google Scholar] [CrossRef] [Green Version]
- Watanabe, M.; Nakamoto, K.; Inui, T.; Sada, M.; Honda, K.; Tamura, M.; Ogawa, Y.; Yokoyama, T.; Saraya, T.; Kurai, D.; et al. Serum sST2 levels predict severe exacerbation of asthma. Respir. Res. 2018, 19, 169. [Google Scholar] [CrossRef] [PubMed]
- Gasiuniene, E.; Janulaityte, I.; Zemeckiene, Z.; Barkauskiene, D.; Sitkauskiene, B. Elevated levels of IL-33 are associated with allergic and eosinophilic asthma. Scand. J. Immunol. 2019, 89, e12724. [Google Scholar] [CrossRef]
- Wang, W.; Li, Y.; Lv, Z.; Chen, Y.; Li, Y.; Huang, K.; Corrigan, C.J.; Ying, S. Bronchial allergen challenge of patients with atopic asthma triggers an alarmin (IL-33, TSLP, and IL-25) response in the airways epithelium and submucosa. J. Immunol. 2018, 201, 2221–2231. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitchell, P.D.; Salter, B.M.; Oliveria, J.P.; El-Gammal, A.; Tworek, D.; Smith, S.G.; Sehmi, R.; Gauvreau, G.M.; Apos Byrne, P.M.O. IL-33 and its receptor ST2 after inhaled allergen challenge in allergic asthmatics. Int. Arch. Allergy Immunol. 2018, 176, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Chan, B.C.L.; Lam, C.W.K.; Tam, L.S.; Wong, C.K. IL33: Roles in allergic inflammation and therapeutic perspectives. Front. Immunol. 2019, 10, 364. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, C.C.; Kobayashi, T.; Iijima, K.; Hsu, F.C.; Kita, H. IL-33 dysregulates regulatory T cells and impairs established immunologic tolerance in the lungs. J. Allergy Clin. Immunol. 2017, 140, 1351–1363.e7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gabryelska, A.; Kuna, P.; Antczak, A.; Bialasiewicz, P.; Panek, M. IL-33 mediated inflammation in chronic respiratory diseases–Understanding the role of the member of IL-1 superfamily. Front. Immunol. 2019, 10, 692. [Google Scholar] [CrossRef]
- Suzukawa, M.; Koketsu, R.; Iikura, M.; Nakae, S.; Matsumoto, K.; Nagase, H.; Saito, H.; Matsushima, K.; Ohta, K.; Yamamoto, K.; et al. Interleukin-33 enhances adhesion, CD11b expression and survival in human eosinophils. Lab. Investig. 2008, 88, 1245–1253. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.X.; Kaieda, S.; Ameri, S.; Fishgal, N.; Dwyer, D.; Dellinger, A.; Kepley, C.L.; Gurish, M.F.; Nigrovic, P.A. IL-33/ST2 axis promotes mast cell survival via BCLXL. Proc. Natl. Acad. Sci. USA 2014, 111, 10281–10286. [Google Scholar] [CrossRef] [Green Version]
- Kroeger, K.M.; Sullivan, B.M.; Locksley, R.M. IL-18 and IL-33 elicit Th2 cytokines from basophils via a MyD88- and p38α-dependent pathway. J. Leukoc. Biol. 2009, 86, 769–778. [Google Scholar] [CrossRef]
- Rothenberg, M.E.; Saito, H.; Peebles, R.S. Advances in mechanisms of allergic disease in 2016. J. Allergy Clin. Immunol. 2017, 140, 1622–1631. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morimoto, Y.; Hirahara, K.; Kiuchi, M.; Wada, T.; Ichikawa, T.; Kanno, T.; Okano, M.; Kokubo, K.; Onodera, A.; Sakurai, D.; et al. Amphiregulin-producing pathogenic memory T helper 2 cells instruct eosinophils to secrete osteopontin and facilitate airway fibrosis. Immunity 2018, 49, 134–150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, Z.; Wu, J.; Zhao, J.; Liu, F.; Chen, Y.; Bi, L.; Liu, S.; Dong, L. IL-33 promotes airway remodeling and is a marker of asthma disease severity. J. Asthma 2014, 51, 863–869. [Google Scholar] [CrossRef]
- Xu, M.; Dong, C. IL-25 in allergic inflammation. Immunol. Rev. 2017, 278, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Borowczyk, J.; Shutova, M.; Brembilla, N.C.; Boehncke, W.H. IL-25 (IL-17E) in epithelial immunology and pathophysiology. J. Allergy Clin. Immunol. 2021, 148, 40–52. [Google Scholar] [CrossRef]
- Swaidani, S.; Liu, C.; Zhao, J.; Bulek, K.; Li, X. TRAF regulation of IL-17 cytokine signaling. Front. Immunol. 2019, 10, 1293. [Google Scholar] [CrossRef] [Green Version]
- Kouzaki, H.; Tojima, I.; Kita, H.; Shimizu, T. Transcription of interleukin-25 and extracellular release of the protein is regulated by allergen proteases in airway epithelial cells. Am. J. Respir. Cell Mol. Biol. 2013, 49, 741–750. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beale, J.; Jayaraman, A.; Jackson, D.J.; Macintyre, J.D.R.; Edwards, M.R.; Walton, R.P.; Zhu, J.; Man Ching, Y.; Shamji, B.; Edwards, M.; et al. Rhinovirus-induced IL-25 in asthma exacerbation drives type 2 immunity and allergic pulmonary inflammation. Sci. Transl. Med. 2014, 6, 256ra134. [Google Scholar] [CrossRef] [Green Version]
- Cheng, D.; Xue, Z.; Yi, L.; Shi, H.; Zhang, K.; Huo, X.; Bonser, L.R.; Zhao, J.; Xu, Y.; Erle, D.J.; et al. Epithelial interleukin-25 is a key mediator in Th2-high, corticosteroid-responsive asthma. Am. J. Respir. Crit. Care Med. 2014, 190, 639–648. [Google Scholar] [CrossRef] [Green Version]
- Su, J.; Chen, T.; Ji, X.Y.; Liu, C.; Yadav, P.K.; Wu, R.; Yang, P.; Liu, Z. IL-25 downregulates Th1/Th17 immune response in an IL-10-dependent manner in inflammatory bowel disease. Inflamm. Bowel Dis. 2013, 19, 720–728. [Google Scholar] [CrossRef]
- Cheung, P.F.; Wong, C.K.; Ip, W.K.; Lam, C.W. IL-25 regulates the expression of adhesion molecules on eosinophils: Mechanisms of eosinophilia in allergic inflammation. Allergy 2006, 61, 878–885. [Google Scholar] [CrossRef]
- Hong, H.Y.; Chen, F.H.; Sun, Y.Q.; Hu, X.T.; Wei, Y.; Fan, Y.P.; Zhang, J.; Wang, D.H.; Xu, R.; Li, H.B.; et al. Local IL-25 contributes to Th2-biased inflammatory profiles in nasal polyps. Allergy 2018, 73, 459–469. [Google Scholar] [CrossRef] [PubMed]
- Verstraete, K.; Peelman, F.; Braun, H.; Lopez, J.; Van Rompaey, D.; Dansercoer, A.; Vandenberghe, I.; Pauwels, K.; Tavernier, J.; Lambrecht, B.N.; et al. Structure and antagonism of the receptor complex mediated by human TSLP in allergy and asthma. Nat. Commun. 2017, 8, 14937. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gauvreau, G.M.; O’Byrne, P.M.; Boulet, L.P.; Wang, Y.; Cockcroft, D.; Bigler, J.; FitzGerald, J.M.; Boedigheimer, M.; Davis, B.E.; Dias, C.; et al. Effects of an anti-TSLP antibody on allergen-induced asthmatic responses. N. Engl. J. Med. 2014, 370, 2102–2110. [Google Scholar] [CrossRef] [PubMed]
- Corren, J.; Parnes, J.R.; Wang, L.; Mo, M.; Roseti, S.L.; Griffiths, J.M.; van der Merwe, R. Tezepelumab in adults with uncontrolled asthma. N. Engl. J. Med. 2017, 377, 936–946. [Google Scholar] [CrossRef] [PubMed]
- Emson, C.; Corren, J.; Salapa, K.; Hellqvist, Å.; Parnes, J.R.; Colice, G. Efficacy of tezepelumab in patients with severe, uncontrolled asthma with and without nasal polyposis: A post hoc analysis of the phase 2b study PATHWAY. J. Asthma Allergy 2021, 14, 91–99. [Google Scholar] [CrossRef]
- Corren, J.; Karpefors, M.; Hellqvist, Å.; Parnes, J.R.; Colice, G. Tezepelumab reduces exacerbations across all seasons in patients with severe, uncontrolled asthma: A post hoc analysis of the PATHWAY phase 2b study. J. Asthma Allergy 2021, 14, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Menzies-Gow, A.; Corren, J.; Bourdin, A.; Chupp, G.; Israel, E.; Wechsler, M.E.; Brightling, C.E.; Griffiths, J.M.; Hellqvist, Å.; Bowen, K.; et al. Tezepelumab in adults and adolescents with severe, uncontrolled asthma. N. Engl. J. Med. 2021, 384, 1800–1809. [Google Scholar] [CrossRef]
- Wechsler, M.E.; Colice, G.; Griffiths, J.M.; Almqvist, G.; Skärby, T.; Piechowiak, T.; Kaur, P.; Bowen, K.; Hellqvist, Å.; Mo, M.; et al. SOURCE: A phase 3, multicentre, randomized, double-blind, placebo-controlled, parallel-group trial to evaluate the efficacy and safety of tezepelumab in reducing oral corticosteroid use in adults with oral corticosteroid dependent asthma. Respir. Res. 2020, 21, 264. [Google Scholar] [CrossRef]
- Canonica, G.W.; Colombo, G.L.; Bruno, G.M.; Di Matteo, S.; Martinotti, C.; Blasi, F.; Bucca, C.; Crimi, N.; Paggiaro, P.; Pelaia, G.; et al. Shadow cost of oral corticosteroids-related adverse events: A pharmacoeconomic evaluation applied to real-life data from the Severe Asthma Network in Italy (SANI) registry. World Allergy Organ. J. 2019, 12, 100007. [Google Scholar] [CrossRef] [Green Version]
- Menzies-Gow, A.; Ponnarambil, S.; Downie, J.; Bowen, K.; Hellqvist, Å.; Colice, G. DESTINATION: A phase 3, multicentre, randomized, double-blind, placebo-controlled, parallel-group trial to evaluate the long-term safety and tolerability of tezepelumab in adults and adolescents with severe, uncontrolled asthma. Respir. Res. 2020, 21, 279. [Google Scholar] [CrossRef] [PubMed]
- Emson, C.; Diver, S.; Chachi, L.; Megally, A.; Small, C.; Downie, J.; Parnes, J.R.; Bowen, K.; Colice, G.; Brightling, C.E. CASCADE: A phase 2, randomized, double-blind, placebo-controlled, parallel-group trial to evaluate the effect of tezepelumab on airway inflammation in patients with uncontrolled asthma. Respir. Res. 2020, 21, 265. [Google Scholar] [CrossRef] [PubMed]
- Diver, S.; Khalfaoui, L.; Emson, C.; Wenzel, S.E.; Menzies-Gow, A.; Wechsler, M.E.; Johnston, J.; Molfino, N.; Parnes, J.R.; Megally, A.; et al. Effect of tezepelumab on airway inflammatory cells, remodelling, and hyperresponsiveness in patients with moderate-to-severe asthma (CASCADE): A double-blind, randomised, placebo-controlled, phase 2 trial. Lancet Respir. Med. 2021, S2213-2600(21)00226-5. [Google Scholar]
- Nakajima, S.; Kabata, H.; Kabashima, K.; Asano, K. Anti-TSLP antibodies: Targeting a master regulator of type 2 immune responses. Allergol. Int. 2020, 69, 197–203. [Google Scholar] [CrossRef]
- Kelsen, S.G.; Agache, I.O.; Soong, W.; Israel, E.; Chupp, G.L.; Cheung, D.S.; Theess, W.; Yang, X.; Staton, T.L.; Choy, D.F.; et al. Astegolimab (anti-ST2) efficacy and safety in adults with severe asthma: A randomized clinical trial. J. Allergy Clin. Immunol. 2021, S0091-6749(21)00609-6. [Google Scholar]
- Holgado, A.; Braun, H.; Van Nuffel, E.; Detry, S.; Schuijs, M.J.; Deswarte, K.; Vergote, K.; Haegman, M.; Baudelet, G.; Haustraete, J.; et al. IL-33 trap is a novel IL-33-neutralizing biologic that inhibits allergic airway inflammation. J. Allergy Clin. Immunol. 2019, 144, 204–215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bartlett, N.; Girkin, J.; Williams, T.; Vincent, T.; Jackson, C.; Alton, K.; Shimkets, R. ABM125 anti-IL-25 antibody pre-clinical development for viral asthma exacerbations identifies IL-25 mediated regulation of type-2- and anti-viral immunity. Am. J. Respir. Crit. Care Med. 2018, 197, A7759. [Google Scholar]
- Castro, M.; Corren, J.; Pavord, I.D.; Maspero, J.; Wenzel, S.; Rabe, K.F.; Busse, W.W.; Ford, L.; Sher, L.; FitzGerald, J.M.; et al. Dupilumab efficacy and safety in moderate-to-severe uncontrolled asthma. N. Engl. J. Med. 2018, 378, 2486–2496. [Google Scholar] [CrossRef] [PubMed]
- Rabe, K.F.; Nair, P.; Brusselle, G.; Maspero, J.F.; Castro, M.; Sher, L.; Zhu, H.; Hamilton, J.D.; Swanson, B.N.; Khan, A.; et al. Efficacy and safety of dupilumab in glucocorticoid-dependent severe asthma. N. Engl. J. Med. 2018, 378, 2475–2485. [Google Scholar] [CrossRef] [PubMed]
- Tamari, M.; Trier, A.M.; Kim, B.S. Emerging targeted therapeutics underscore immunologic heterogeneity of asthma. J. Allergy Clin. Immunol. 2021, S0091-6749(21)01127-1. [Google Scholar]
- Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention. 2021. Available online: www.ginasthma.org (accessed on 10 August 2021).
| Drug—Trial Name | Target | Duration | Main Results or Endpoints |
|---|---|---|---|
| Tezepelumab—PATHWAY [97] | TSLP | 52 weeks | Fewer asthma exacerbations, higher ACQ-6 score, FEV1 increase. |
| Tezepelumab—NAVIGATOR [100] | TSLP | 52 weeks | Fewer asthma exacerbations, higher ACQ-6 score, FEV1 increase. |
| Tezepelumab—SOURCE [101] | TSLP | 48 weeks | OCS-sparing effect. |
| Tezepelumab—DESTINATION [103] | TSLP | 52 weeks | Safety profile, impact on asthma exacerbations. |
| Tezepelumab—CASCADE [105] | TSLP | 28 weeks | Suppression of airway eosinophilic inflammation. |
| REGN3500 [16] | IL-33 | 36 weeks | Improvement of asthma control. |
| AMG282 [16] | IL-33 | 20 weeks | Impact on asthma exacerbations. |
| GSK3772847 [16] | IL-33 | 16 weeks | Improvement of asthma control. |
| Etokimab [16] | IL-33 | 9 weeks | Reduction of blood eosinophil count. |
| Astegolimab—ZENYATTA [107] | ST2 | 54 weeks | Fewer asthma exacerbations. |
| ABM125 [109] | IL-25 | - | Suppression of type 2 cytokine response induced by rhinovirus. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pelaia, C.; Pelaia, G.; Longhini, F.; Crimi, C.; Calabrese, C.; Gallelli, L.; Sciacqua, A.; Vatrella, A. Monoclonal Antibodies Targeting Alarmins: A New Perspective for Biological Therapies of Severe Asthma. Biomedicines 2021, 9, 1108. https://doi.org/10.3390/biomedicines9091108
Pelaia C, Pelaia G, Longhini F, Crimi C, Calabrese C, Gallelli L, Sciacqua A, Vatrella A. Monoclonal Antibodies Targeting Alarmins: A New Perspective for Biological Therapies of Severe Asthma. Biomedicines. 2021; 9(9):1108. https://doi.org/10.3390/biomedicines9091108
Chicago/Turabian StylePelaia, Corrado, Giulia Pelaia, Federico Longhini, Claudia Crimi, Cecilia Calabrese, Luca Gallelli, Angela Sciacqua, and Alessandro Vatrella. 2021. "Monoclonal Antibodies Targeting Alarmins: A New Perspective for Biological Therapies of Severe Asthma" Biomedicines 9, no. 9: 1108. https://doi.org/10.3390/biomedicines9091108
APA StylePelaia, C., Pelaia, G., Longhini, F., Crimi, C., Calabrese, C., Gallelli, L., Sciacqua, A., & Vatrella, A. (2021). Monoclonal Antibodies Targeting Alarmins: A New Perspective for Biological Therapies of Severe Asthma. Biomedicines, 9(9), 1108. https://doi.org/10.3390/biomedicines9091108











