Platelet-Activating Factor Acetylhydrolase Expression in BRCA1 Mutant Ovarian Cancer as a Protective Factor and Potential Negative Regulator of the Wnt Signaling Pathway
Abstract
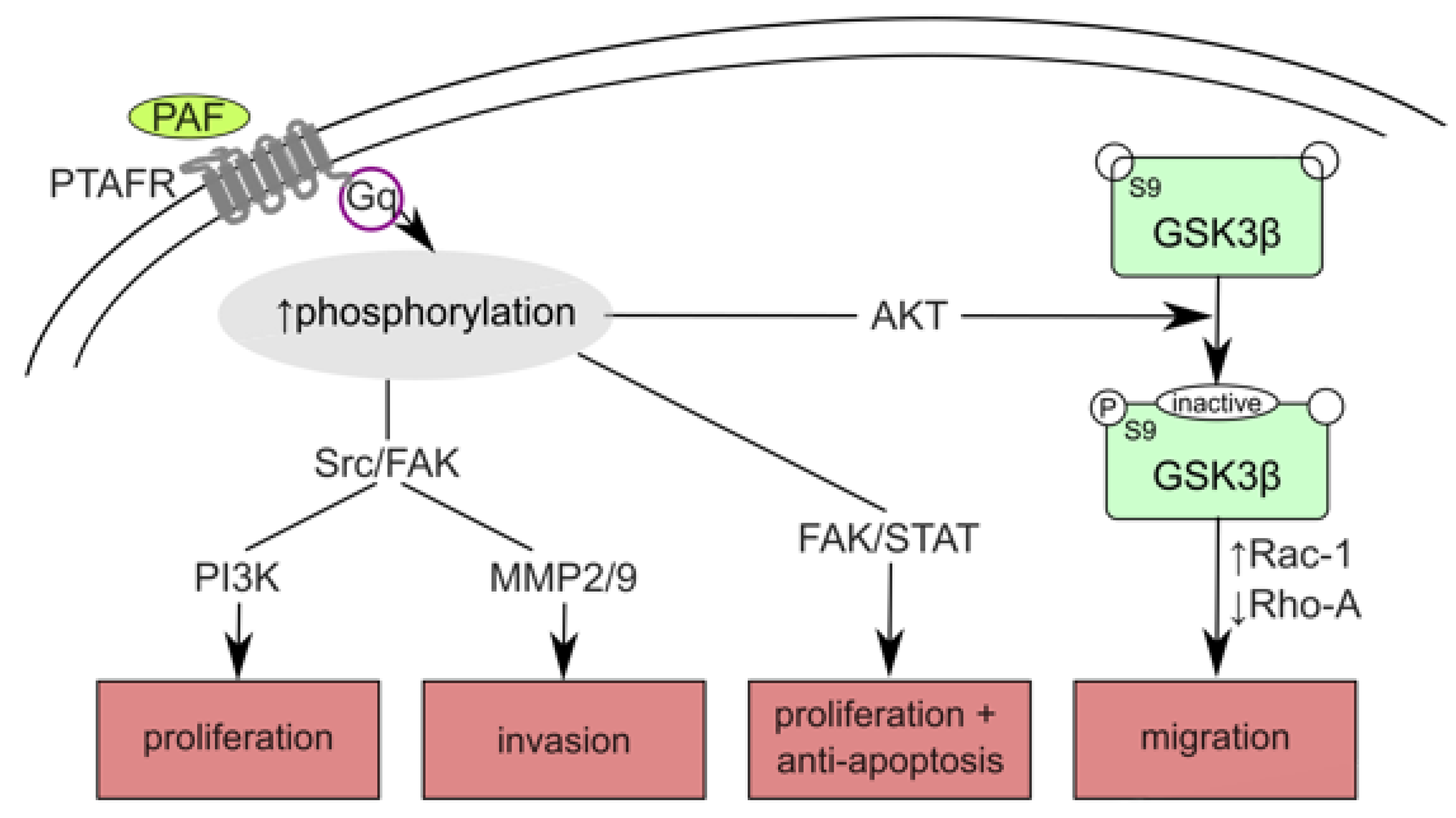
1. Introduction
2. Materials and Methods
2.1. Ethical Approval
2.2. Patients and Specimens
2.3. Immunohistochemistry and Immunocytochemistry
2.4. Staining Evaluation and Statistical Analysis
2.5. PAF-AH ELISA
2.6. Cell Lines
2.7. qPCR
2.8. siRNA Knockdown
2.9. Western Blot
2.10. Cell Viability Assay and Proliferation Assay
2.11. Wound Healing Assay
3. Results
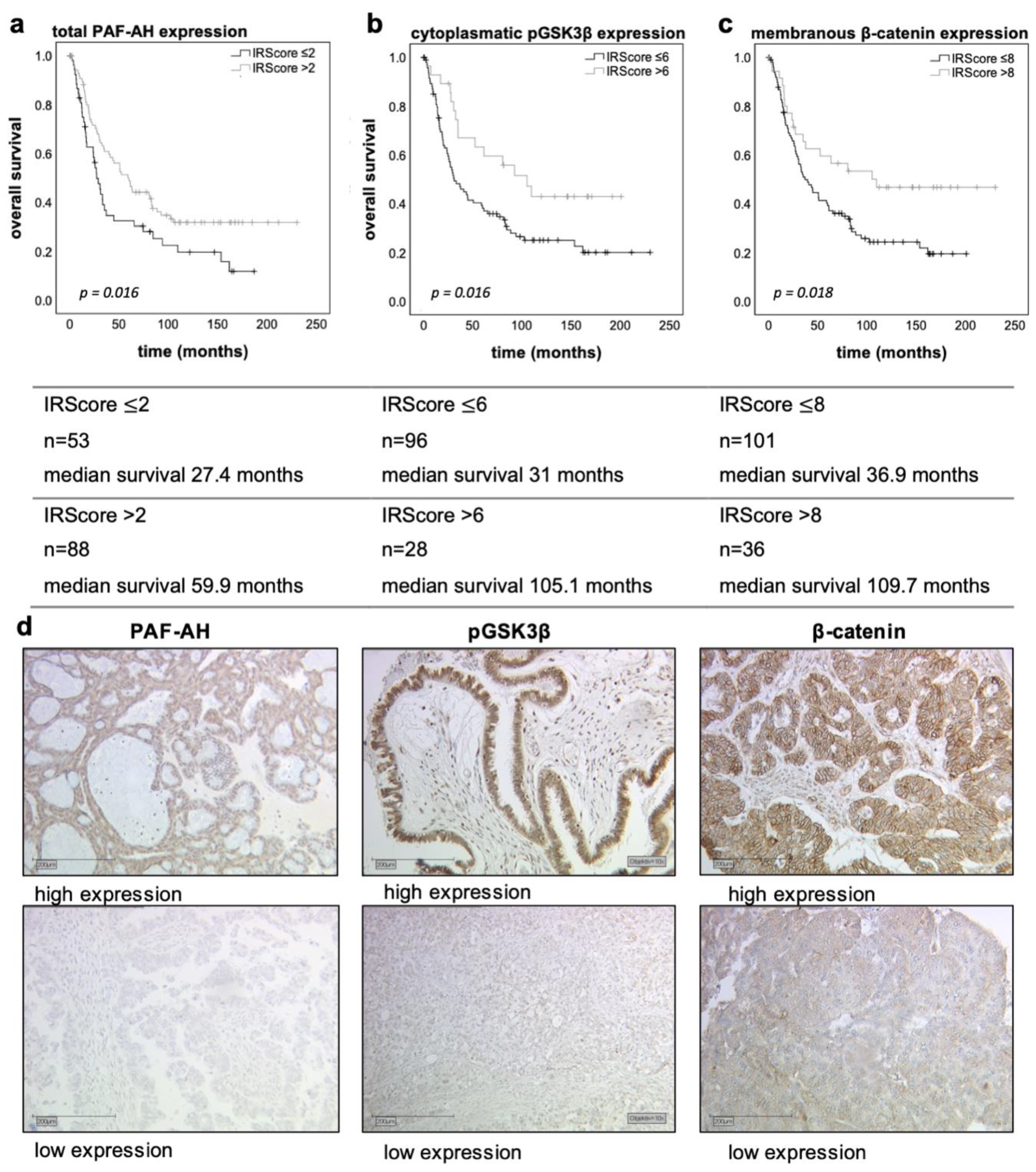
3.1. PAF-AH Is an Independent Positive Prognostic Factor in EOC and Correlated with the Wnt Signaling Proteins pGSK3β and β-Catenin
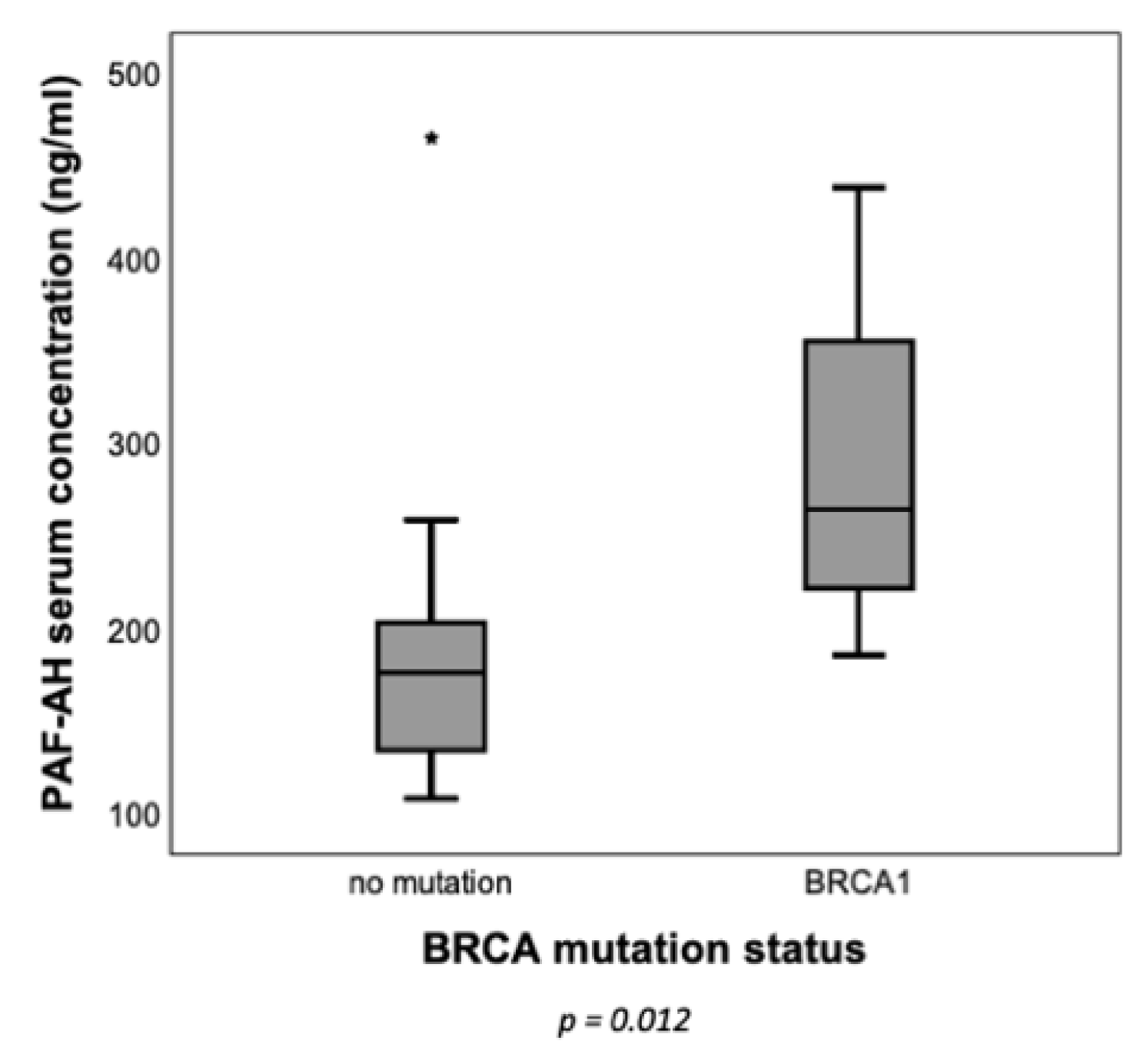
3.2. BRCA1 Mutant Patients Had Higher PAF-AH Levels in Tumor Tissue and in Serum
3.3. Only BRCA1-Negative UWB1.289 Cell Line Showed Relevant Expression of PLA2G7/PAF-AH
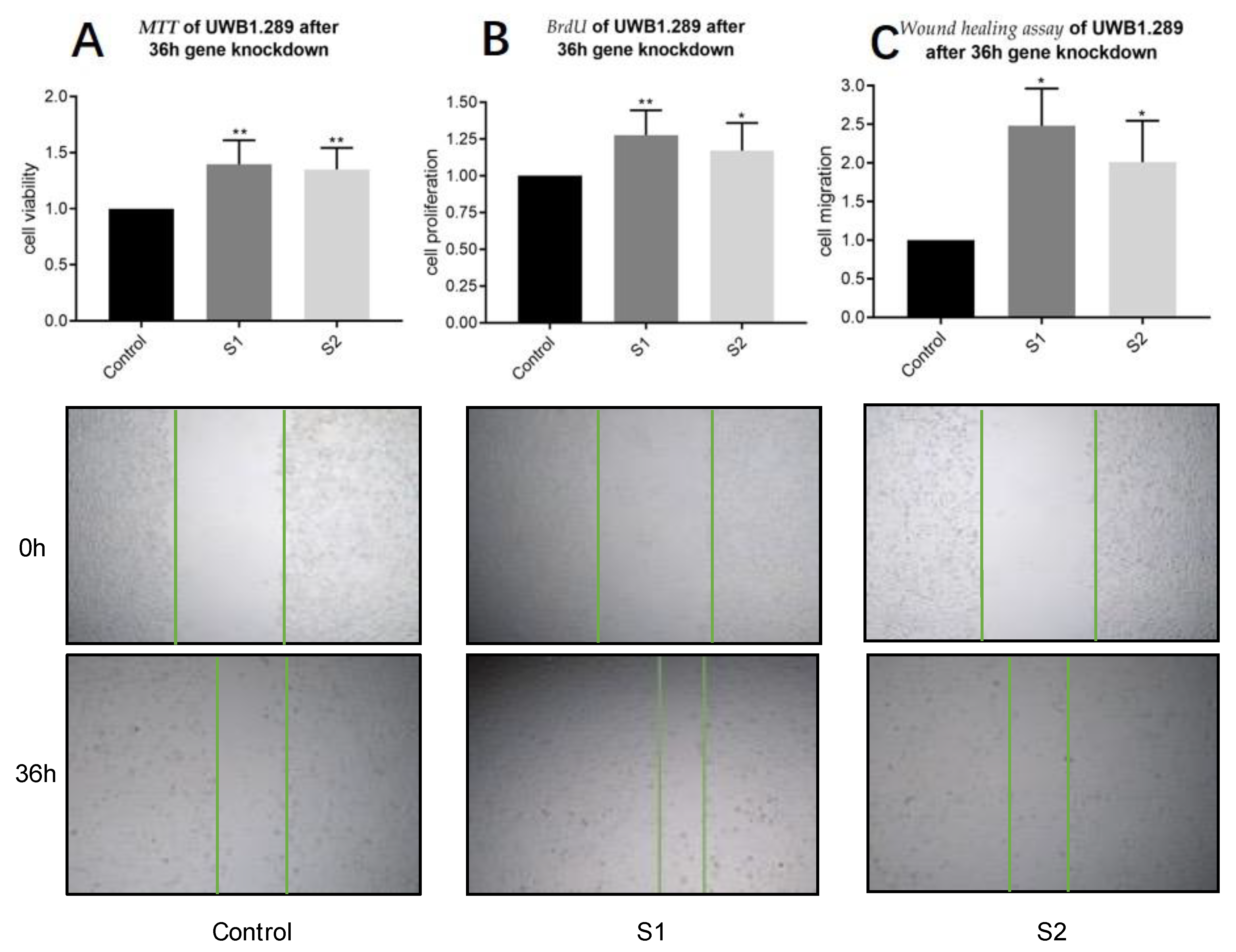
3.4. PLA2G7 Knockdown Enhanced Viability, Proliferation, and Motility of UWB1.289 Cells
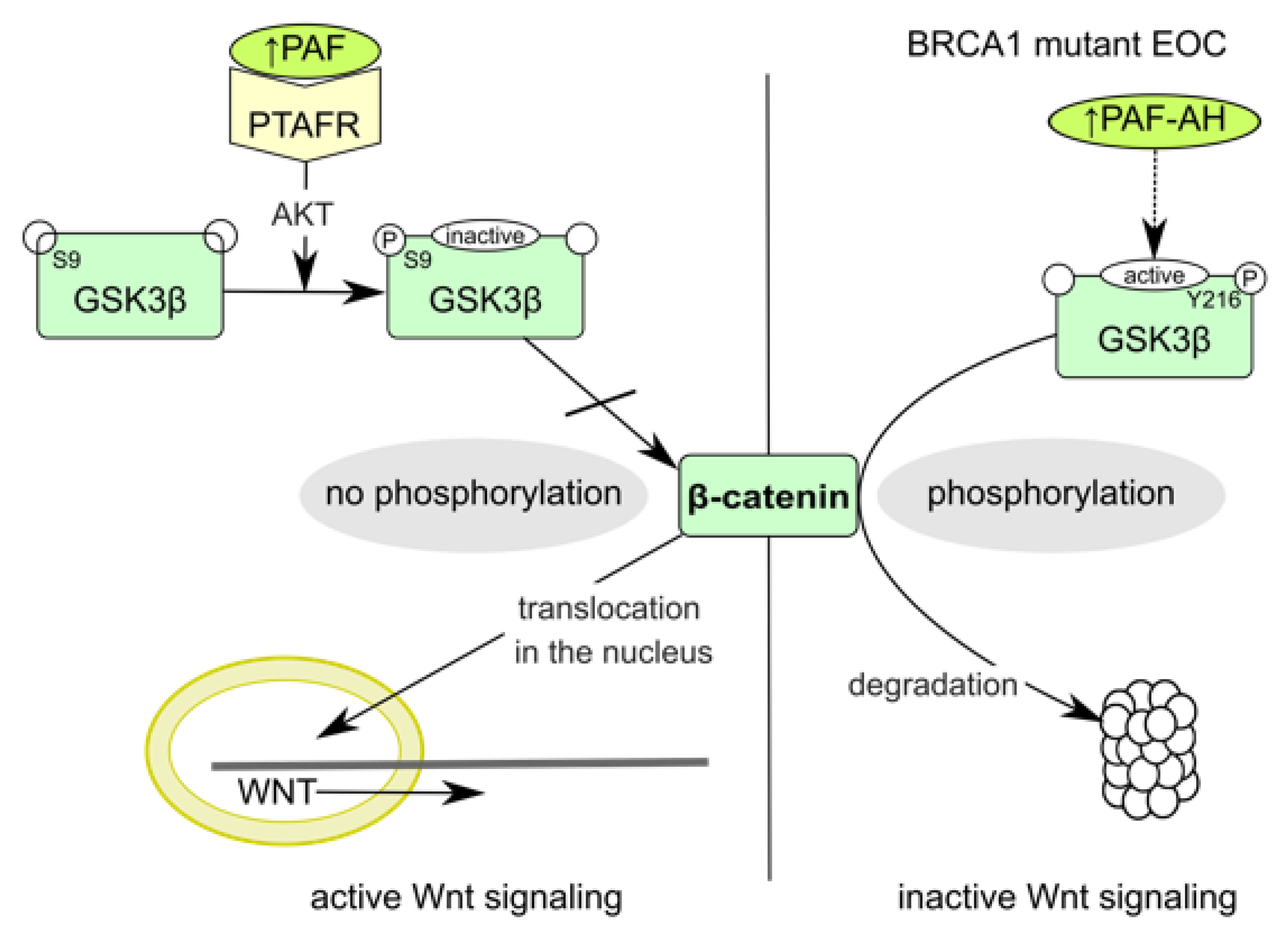
3.5. The Cellular Distribution Pattern of β-Catenin Changed by PLA2G7 Knockdown from the Membrane to Nucleus
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADP | adenosine diphosphate |
| AKT | protein kinase B |
| BRCA | breast cancer gene |
| BrDU | 5-bromo-2-deoxyuridine |
| Cc | correlation coefficient |
| CI | confidence interval |
| DNA | deoxyribonucleic acid |
| ELISA | enzyme-linked immunosorbent assay |
| EMT | epithelial–mesenchymal transition |
| EOC | epithelial ovarian cancer |
| FBS | fetal bovine serum |
| FIGO | International Federation of Gynecology and Obstetrics |
| GAPDH | glycerinaldehyd-3-phosphat-dehydrogenase |
| GSK3β | glycogen synthase kinase-3β |
| ICC | immunocytochemistry |
| IHC | immunohistochemistry |
| IRScore | immunoreactive score |
| MMP | matrix metalloproteinase |
| mRNA | messenger ribonucleic acid |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| OS | overall survival |
| PAF | platelet-activating factor |
| PBS | phosphate-buffered saline |
| PI3K | phosphatidylinositol 3-kinase |
| PLA2G7/PAF-AH | platelet-activating factor acetylhydrolase |
| PTAFR | platelet-activating factor receptor |
| qPCR | quantitative polymerase chain reaction |
| Rac-1 | ras-related C3 botulinum toxin substrate 1 |
| Rho-A | ras homolog family member A |
| RIPA | radioimmunoprecipitation assay |
| ROC | receiver operating characteristic |
| RT | room temperature |
| siRNA | small interfering ribonucleic acid |
| Src/FAK | steroid receptor coactivator/focal adhesion kinase |
| STAT | signal transducer and activator of transcription |
| Tcf/Lef | transcription factor/lymphoid enhancer-binding factor |
| TLE | transducin-like enhancer |
| WHO | World Health Organization |
| WT | wildtype |
References
- Oberaigner, W.; Minicozzi, P.; Bielska-Lasota, M.; Allemani, C.; de Angelis, R.; Mangone, L.; Sant, M. Eurocare Working G: Survival for ovarian cancer in Europe: The across-country variation did not shrink in the past decade. Acta Oncol. 2012, 51, 441–453. [Google Scholar] [CrossRef]
- Torre, L.A.; Trabert, B.; DeSantis, C.E.; Miller, K.D.; Samimi, G.; Runowicz, C.D.; Gaudet, M.M.; Jemal, A.; Siegel, R.L. Ovarian cancer statistics. 2018. CA Cancer J. Clin. 2018, 68, 284–296. [Google Scholar] [CrossRef]
- Aletti, G.D.; Gostout, B.S.; Podratz, K.C.; Cliby, W.A. Ovarian cancer surgical resectability: Relative impact of disease, patient status, and surgeon. Gynecol. Oncol. 2006, 100, 33–37. [Google Scholar] [CrossRef]
- Dembo, A.J.; Davy, M.; Stenwig, A.E.; Berle, E.J.; Bush, R.S.; Kjorstad, K. Prognostic factors in patients with stage I epithelial ovarian cancer. Obstet. Gynecol. 1990, 75, 263–273. [Google Scholar]
- Vergote, I.; De Brabanter, J.; Fyles, A.; Bertelsen, K.; Einhorn, N.; Sevelda, P.; Gore, M.E.; Kaern, J.; Verrelst, H.; Sjovall, K.; et al. Prognostic importance of degree of differentiation and cyst rupture in stage I invasive epithelial ovarian carcinoma. Lancet 2001, 357, 176–182. [Google Scholar] [CrossRef]
- Nelson, H.D.; Pappas, M.; Cantor, A.; Haney, E.; Holmes, R. Risk Assessment, Genetic Counseling, and Genetic Testing for BRCA-Related Cancer in Women: Updated Evidence Report and Systematic Review for the US Preventive Services Task Force. JAMA 2019, 322, 666–685. [Google Scholar] [CrossRef]
- Madariaga, A.; Lheureux, S.; Oza, A.M. Tailoring Ovarian Cancer Treatment: Implications of BRCA1/2 Mutations. Cancers 2019, 11, 416. [Google Scholar] [CrossRef]
- Li, H.; Sekine, M.; Tung, N.; Avraham, H.K. Wild-type BRCA1, but not mutated BRCA1, regulates the expression of the nuclear form of beta-catenin. Mol. Cancer Res. 2010, 8, 407–420. [Google Scholar] [CrossRef]
- Yu, V. Caretaker Brca1: Keeping the genome in the straight and narrow. Breast Cancer Res. 2000, 2, 82–85. [Google Scholar] [CrossRef][Green Version]
- Kuchenbaecker, K.B.; Hopper, J.L.; Barnes, D.R.; Phillips, K.A.; Mooij, T.M.; Roos-Blom, M.J.; Jervis, S.; van Leeuwen, F.E.; Milne, R.L.; Andrieu, N.; et al. Risks of Breast, Ovarian, and Contralateral Breast Cancer for BRCA1 and BRCA2 Mutation Carriers. JAMA 2017, 317, 2402–2416. [Google Scholar] [CrossRef] [PubMed]
- Tsoupras, A.B.; Iatrou, C.; Frangia, C.; Demopoulos, C.A. The implication of platelet activating factor in cancer growth and metastasis: Potent beneficial role of PAF-inhibitors and antioxidants. Infect. Disord. Drug Targets 2009, 9, 390–399. [Google Scholar] [CrossRef]
- Melnikova, V.; Bar-Eli, M. Inflammation and melanoma growth and metastasis: The role of platelet-activating factor (PAF) and its receptor. Cancer Metastasis Rev. 2007, 26, 359–371. [Google Scholar] [CrossRef]
- Chen, J.; Lan, T.; Zhang, W.; Dong, L.; Kang, N.; Zhang, S.; Fu, M.; Liu, B.; Liu, K.; Zhan, Q. Feed-Forward Reciprocal Activation of PAFR and STAT3 Regulates Epithelial-Mesenchymal Transition in Non-Small Cell Lung Cancer. Cancer Res. 2015, 75, 4198–4210. [Google Scholar] [CrossRef]
- Gao, T.; Yu, Y.; Cong, Q.; Wang, Y.; Sun, M.; Yao, L.; Xu, C.; Jiang, W. Human mesenchymal stem cells in the tumour microenvironment promote ovarian cancer progression: The role of platelet-activating factor. BMC Cancer 2018, 18, 999. [Google Scholar] [CrossRef] [PubMed]
- Aponte, M.; Jiang, W.; Lakkis, M.; Li, M.J.; Edwards, D.; Albitar, L.; Vitonis, A.; Mok, S.C.; Cramer, D.W.; Ye, B. Activation of platelet-activating factor receptor and pleiotropic effects on tyrosine phospho-EGFR/Src/FAK/paxillin in ovarian cancer. Cancer Res. 2008, 68, 5839–5848. [Google Scholar] [CrossRef]
- Boccellino, M.; Biancone, L.; Cantaluppi, V.; Ye, R.D.; Camussi, G. Effect of platelet-activating factor receptor expression on CHO cell motility. J. Cell. Physiol. 2000, 183, 254–264. [Google Scholar] [CrossRef]
- Holmes, C.E.; Levis, J.E.; Ornstein, D.L. Activated platelets enhance ovarian cancer cell invasion in a cellular model of metastasis. Clin. Exp. Metastasis 2009, 26, 653–661. [Google Scholar] [CrossRef]
- Yu, Y.; Zhang, X.; Hong, S.; Zhang, M.; Cai, Q.; Jiang, W.; Xu, C. Epidermal growth factor induces platelet-activating factor production through receptors transactivation and cytosolic phospholipase A2 in ovarian cancer cells. J. Ovarian Res. 2014, 7, 39. [Google Scholar] [CrossRef]
- Yu, Y.; Zhang, X.; Hong, S.; Zhang, M.; Cai, Q.; Jiang, W.; Xu, C. The expression of platelet-activating factor receptor modulates the cisplatin sensitivity of ovarian cancer cells: A novel target for combination therapy. Br. J. Cancer 2014, 111, 515–524. [Google Scholar] [CrossRef]
- Stafforini, D.M. Biology of platelet-activating factor acetylhydrolase (PAF-AH, lipoprotein associated phospholipase A2). Cardiovasc. Drugs Ther. 2009, 23, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Arai, H. Platelet-activating factor acetylhydrolase. Prostaglandins Other Lipid Mediat. 2002, 68–69, 83–94. [Google Scholar] [CrossRef]
- Bonin, F.; Ryan, S.D.; Migahed, L.; Mo, F.; Lallier, J.; Franks, D.J.; Arai, H.; Bennett, S.A. Anti-apoptotic actions of the platelet-activating factor acetylhydrolase I alpha2 catalytic subunit. J. Biol. Chem. 2004, 279, 52425–52436. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Guo, Y.; Zhang, Y.; Duo, A.; Jia, Y.; Liu, C.; Li, B. PAFAH1B2 is a HIF1a target gene and promotes metastasis in pancreatic cancer. Biochem. Biophys. Res. Commun. 2018, 501, 654–660. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Zang, Y.; Cao, S.; Lei, D.; Pan, X. Aberrant expression of PAFAH1B3 associates with poor prognosis and affects proliferation and aggressiveness in hypopharyngeal squamous cell carcinoma. Onco Targets Ther. 2019, 12, 2799–2808. [Google Scholar] [CrossRef]
- Mulvihill, M.M.; Benjamin, D.I.; Ji, X.; Le Scolan, E.; Louie, S.M.; Shieh, A.; Green, M.; Narasimhalu, T.; Morris, P.; Luo, K.; et al. Metabolic profiling reveals PAFAH1B3 as a critical driver of breast cancer pathogenicity. Chem. Biol. 2014, 21, 831–840. [Google Scholar] [CrossRef] [PubMed]
- Vainio, P.; Lehtinen, L.; Mirtti, T.; Hilvo, M.; Seppänen-Laakso, T.; Virtanen, J.; Sankila, A.; Nordling, S.; Lundin, J.; Rannikko, A.; et al. Phospholipase PLA2G7, associated with aggressive prostate cancer, promotes prostate cancer cell migration and invasion and is inhibited by statins. Oncotarget 2011, 2, 1176–1190. [Google Scholar] [CrossRef]
- Lehtinen, L.; Vainio, P.; Wikman, H.; Huhtala, H.; Mueller, V.; Kallioniemi, A.; Pantel, K.; Kronqvist, P.; Kallioniemi, O.; Carpèn, O.; et al. PLA2G7 associates with hormone receptor negativity in clinical breast cancer samples and regulates epithelial-mesenchymal transition in cultured breast cancer cells. J. Pathol. Clin. Res. 2017, 3, 123–138. [Google Scholar] [CrossRef]
- Biancone, L.; Cantaluppi, V.; Del Sorbo, L.; Russo, S.; Tjoelker, L.W.; Camussi, G. Platelet-activating factor inactivation by local expression of platelet-activating factor acetyl-hydrolase modifies tumor vascularization and growth. Clin. Cancer Res. 2003, 9, 4214–4220. [Google Scholar]
- Arend, R.C.; Londono-Joshi, A.I.; Straughn, J.M., Jr.; Buchsbaum, D.J. The Wnt/beta-catenin pathway in ovarian cancer: A review. Gynecol. Oncol. 2013, 131, 772–779. [Google Scholar] [CrossRef]
- Nagaraj, A.B.; Joseph, P.; Kovalenko, O.; Singh, S.; Armstrong, A.; Redline, R.; Resnick, K.; Zanotti, K.; Waggoner, S.; DiFeo, A. Critical role of Wnt/beta-catenin signaling in driving epithelial ovarian cancer platinum resistance. Oncotarget 2015, 6, 23720–23734. [Google Scholar] [CrossRef]
- Yamamoto, T.M.; McMellen, A.; Watson, Z.L.; Aguilera, J.; Ferguson, R.; Nurmemmedov, E.; Thakar, T.; Moldovan, G.L.; Kim, H.; Cittelly, D.M.; et al. Activation of Wnt signaling promotes olaparib resistant ovarian cancer. Mol. Carcinog. 2019, 58, 1770–1782. [Google Scholar] [CrossRef]
- Moon, R.T.; Kohn, A.D.; De Ferrari, G.V.; Kaykas, A. WNT and beta-catenin signalling: Diseases and therapies. Nat. Rev. Genet. 2004, 5, 691–701. [Google Scholar] [CrossRef] [PubMed]
- Clevers, H. Wnt/beta-catenin signaling in development and disease. Cell 2006, 127, 469–480. [Google Scholar] [CrossRef]
- Taketo, M.M. Shutting down Wnt signal-activated cancer. Nat. Genet. 2004, 36, 320–322. [Google Scholar] [CrossRef]
- Giles, R.H.; van Es, J.H.; Clevers, H. Caught up in a Wnt storm: Wnt signaling in cancer. Biochim. Biophys. Acta 2003, 1653, 1–24. [Google Scholar] [CrossRef]
- Teeuwssen, M.; Fodde, R. Wnt Signaling in Ovarian Cancer Stemness, EMT, and Therapy Resistance. J. Clin. Med. 2019, 8, 1658. [Google Scholar] [CrossRef]
- Marchion, D.C.; Xiong, Y.; Chon, H.S.; Al Sawah, E.; Bou Zgheib, N.; Ramirez, I.J.; Abbasi, F.; Stickles, X.B.; Judson, P.L.; Hakam, A.; et al. Gene expression data reveal common pathways that characterize the unifocal nature of ovarian cancer. Am. J. Obstet. Gynecol. 2013, 209, 576.e1–576.e16. [Google Scholar] [CrossRef] [PubMed]
- Reinartz, S.; Finkernagel, F.; Adhikary, T.; Rohnalter, V.; Schumann, T.; Schober, Y.; Nockher, W.A.; Nist, A.; Stiewe, T.; Jansen, J.M.; et al. A transcriptome-based global map of signaling pathways in the ovarian cancer microenvironment associated with clinical outcome. Genome Biol. 2016, 17, 108. [Google Scholar] [CrossRef]
- Domoto, T.; Pyko, I.V.; Furuta, T.; Miyashita, K.; Uehara, M.; Shimasaki, T.; Nakada, M.; Minamoto, T. Glycogen synthase kinase-3beta is a pivotal mediator of cancer invasion and resistance to therapy. Cancer Sci. 2016, 107, 1363–1372. [Google Scholar] [CrossRef]
- Wu, Z.Q.; Li, X.Y.; Hu, C.Y.; Ford, M.; Kleer, C.G.; Weiss, S.J. Canonical Wnt signaling regulates Slug activity and links epithelial-mesenchymal transition with epigenetic Breast Cancer 1, Early Onset (BRCA1) repression. Proc. Natl. Acad. Sci. USA 2012, 109, 16654–16659. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Reichert, E.C.; Nakano, T.; Lohse, M.; Gardner, A.A.; Revelo, M.P.; Topham, M.K.; Stafforini, D.M. Deficiency of phospholipase A2 group 7 decreases intestinal polyposis and colon tumorigenesis in Apc(Min/+) mice. Cancer Res. 2013, 73, 2806–2816. [Google Scholar] [CrossRef]
- Scholz, C.; Heublein, S.; Lenhard, M.; Friese, K.; Mayr, D.; Jeschke, U. Glycodelin A is a prognostic marker to predict poor outcome in advanced stage ovarian cancer patients. BMC Res. Notes 2012, 5, 551. [Google Scholar] [CrossRef]
- Remmele, W.; Stegner, H.E. Recommendation for uniform definition of an immunoreactive score (IRS) for immunohistochemical estrogen receptor detection (ER-ICA) in breast cancer tissue. Pathologe 1987, 8, 138–140. [Google Scholar] [PubMed]
- Whitney, J. Testing for differences with the nonparametric Mann-Whitney U test. J. Wound Ostomy Cont. Nurs. 1997, 24, 12. [Google Scholar] [CrossRef]
- Spearman, C. The proof and measurement of association between two things; By, C. Spearman, 1904. Am. J. Psychol. 1987, 100, 441–471. [Google Scholar] [CrossRef]
- Kaplan, E.L.; Meier, P. Nonparametric Estimation from Incomplete Observations. J. Am. Stat. Assoc. 1958, 53, 457–481. [Google Scholar] [CrossRef]
- Youden, W.J. Index for rating diagnostic tests. Cancer 1950, 3, 32–35. [Google Scholar] [CrossRef]
- Perkins, N.J.; Schisterman, E.F. The Inconsistency of “Optimal” Cut-points Using Two ROC Based Criteria. Am. J. Epidemiol. 2006, 163, 670–675. [Google Scholar] [CrossRef]
- Fluss, R.; Faraggi, D.; Reiser, B. Estimation of the Youden Index and its Associated Cutoff Point. Biom. J. 2005, 47, 458–472. [Google Scholar] [CrossRef]
- Cox, D.R. Regression Models and Life-Tables. J. R. Stat. Soc. Ser. B 1972, 34, 187–220. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Tremmel, E.; Hofmann, S.; Kuhn, C.; Heidegger, H.; Heublein, S.; Hermelink, K.; Wuerstlein, R.; Harbeck, N.; Mayr, D.; Mahner, S.; et al. Thyronamine regulation of TAAR1 expression in breast cancer cells and investigation of its influence on viability and migration. Breast Cancer 2019, 11, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Nandy, D.; Asmann, Y.W.; Mukhopadhyay, D.; Basu, A. Role of AKT-glycogen synthase kinase axis in monocyte activation in human beings with and without type 2 diabetes. J. Cell. Mol. Med. 2010, 14, 1396–1407. [Google Scholar] [CrossRef] [PubMed]
- Daniels, D.L.; Weis, W.I. Beta-catenin directly displaces Groucho/TLE repressors from Tcf/Lef in Wnt-mediated transcription activation. Nat. Struct. Mol. Biol. 2005, 12, 364–371. [Google Scholar] [CrossRef]
- Furihata, T.; Kawamata, H.; Kubota, K.; Fujimori, T. Evaluation of the malignant potential of aberrant crypt foci by immunohistochemical staining for beta-catenin in inflammation-induced rat colon carcinogenesis. Int. J. Mol. Med. 2002, 9, 353–358. [Google Scholar]
- Boccellino, M.; Camussi, G.; Giovane, A.; Ferro, L.; Calderaro, V.; Balestrieri, C.; Quagliuolo, L. Platelet-activating factor regulates cadherin-catenin adhesion system expression and beta-catenin phosphorylation during Kaposi’s sarcoma cell motility. Am. J. Pathol. 2005, 166, 1515–1522. [Google Scholar] [CrossRef]
- Tjoelker, L.W.; Wilder, C.; Eberhardt, C.; Stafforinit, D.M.; Dietsch, G.; Schimpf, B.; Hooper, S.; Le Trong, H.; Cousens, L.S.; Zimmerman, G.A.; et al. Anti-inflammatory properties of a platelet-activating factor acetylhydrolase. Nature 1995, 374, 549–553. [Google Scholar] [CrossRef]
- Yu, Y.; Zhang, M.; Zhang, X.; Cai, Q.; Hong, S.; Jiang, W.; Xu, C. Synergistic effects of combined platelet-activating factor receptor and epidermal growth factor receptor targeting in ovarian cancer cells. J. Hematol. Oncol. 2014, 7, 39. [Google Scholar] [CrossRef]
- Rozengurt, E. Mitogenic signaling pathways induced by G protein-coupled receptors. J. Cell. Physiol. 2007, 213, 589–602. [Google Scholar] [CrossRef]
- Penna, C.; Mognetti, B.; Tullio, F.; Gattullo, D.; Mancardi, D.; Moro, F.; Pagliaro, P.; Alloatti, G. Post-ischaemic activation of kinases in the pre-conditioning-like cardioprotective effect of the platelet-activating factor. Acta Physiol. 2009, 197, 175–185. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, D.; Jiang, W.; Edwards, D.; Qiu, W.; Barroilhet, L.M.; Rho, J.H.; Jin, L.; Seethappan, V.; Vitonis, A.; et al. Activated networking of platelet activating factor receptor and FAK/STAT1 induces malignant potential in BRCA1-mutant at-risk ovarian epithelium. Reprod. Biol. Endocrinol. 2010, 8, 74. [Google Scholar] [CrossRef] [PubMed]
- Livnat, I.; Finkelshtein, D.; Ghosh, I.; Arai, H.; Reiner, O. PAF-AH Catalytic Subunits Modulate the Wnt Pathway in Developing GABAergic Neurons. Front. Cell. Neurosci. 2010, 4, 4. [Google Scholar] [CrossRef] [PubMed]
- Axelrad, T.W.; Deo, D.D.; Ottino, P.; Van Kirk, J.; Bazan, N.G.; Bazan, H.E.; Hunt, J.D. Platelet-activating factor (PAF) induces activation of matrix metalloproteinase 2 activity and vascular endothelial cell invasion and migration. FASEB J. 2004, 18, 568–570. [Google Scholar] [CrossRef] [PubMed]



| BRCA Mutation Status | n | Percentage (%) |
|---|---|---|
| Mutation unknown | 141 | 56.9 |
| BRCA1 mutation | 107 | 43.1 |
| BRCA1 | 15 | 6 |
| BRCA1 + 2 | 92 | 37 |
| BRCA Mutation Status | n | Percentage (%) | Overall Survival (Months; Median) | Progression-Free Survival (Months; Median) |
|---|---|---|---|---|
| No mutation | 17 | 73.9 | 25.0 | 17.0 |
| BRCA1 mutation | 6 | 26.1 | 34.5 | 28.0 |
| Clinicopathological Parameters | PAF-AH Total | PAF-AH Nucleus | PAF-AH Cytoplasm | ||||||
|---|---|---|---|---|---|---|---|---|---|
| n | Median IRScore | p | n | Median IRScore | p | n | Median IRScore | p | |
| Histology | <0.001 * | <0.001 * | <0.001 * | ||||||
| Serous | 102 | 3 | 102 | 3 | 102 | 3 | |||
| Clear cell | 11 | 1 | 11 | 1 | 11 | 1 | |||
| Endometrioid | 19 | 3 | 19 | 2 | 19 | 3 | |||
| Mucinous | 10 | 0.5 | 10 | 0.5 | 10 | 1 | |||
| Lymph node | NS | NS | NS | ||||||
| pN0/X | 94 | 3 | 94 | 2 | 94 | 3 | |||
| pN1 | 48 | 3 | 48 | 2 | 48 | 3 | |||
| Distant Metastasis | NS | NS | NS | ||||||
| pM0/X | 137 | 3 | 137 | 2 | 137 | 3 | |||
| pM1 | 5 | 4 | 5 | 2 | 5 | 3 | |||
| FIGO | NS | NS | NS | ||||||
| I/II | 39 | 3 | 39 | 2 | 39 | 3 | |||
| III/IV | 98 | 3 | 98 | 2 | 98 | 3 | |||
| Age | 0.047 * | 0.022 * | 0.047 * | ||||||
| ≤60 years | 75 | 3 | 75 | 2 | 75 | 3 | |||
| >60 years | 67 | 3 | 67 | 3 | 67 | 3 | |||
| Serous Grading | NS | NS | NS | ||||||
| Low | 22 | 3 | 22 | 3 | 22 | 3 | |||
| High | 74 | 3.5 | 74 | 3 | 74 | 3 | |||
| Clear cell, endometrioid, mucinous grading | NS | NS | NS | ||||||
| G1 | 10 | 2.5 | 10 | 1 | 10 | 2.5 | |||
| G2 | 10 | 2 | 10 | 1 | 10 | 2.5 | |||
| G3 | 15 | 2 | 15 | 1 | 15 | 2 | |||
| PAF-AH Nucleus | PAF-AH Cytoplasm | pGSK3β Cytoplasm | β-Catenin Membrane | |
|---|---|---|---|---|
| PAF-AH nucleus | ||||
| Cc | 1 | 0.469 | 0.494 | 0.267 |
| p | - | <0.001 * | <0.001 * | 0.001 * |
| n | 141 | 141 | 135 | 140 |
| PAF-AH cytoplasm | ||||
| 0.469 | 1 | 0.448 | 0.291 | |
| <0.001 * | - | <0.001 * | <0.001 * | |
| 141 | 141 | 135 | 140 | |
| pGSK3β cytoplasm | ||||
| Cc | 0.494 | 0.448 | 1 | 0.224 |
| p | <0.001 * | <0.001 * | - | 0.008 * |
| n | 135 | 135 | 140 | 139 |
| β-Catenin membrane | ||||
| Cc | 0.267 | 0.291 | 0.224 | 1 |
| p | 0.001 * | <0.001 * | 0.008 * | - |
| n | 140 | 140 | 139 | 147 |
| Covariate | p | Hazard Ratio (95% CI) |
|---|---|---|
| Age > 60 vs. 60 | 0.039 * | 1.637 (1.026–2.612) |
| FIGO III/IV vs. I/II | 0.004 * | 2.585 (1.366–4.891) |
| Grading high/G2-3 vs. low/G1 | 0.002 * | 2.797 (1.436–5.449) |
| Total PAF-AH expression high (2) vs. low (2) | 0.021 * | 0.583 (0.369–0.921) |
| Cytoplasmatic pGSK3β (Y216) expression high (>6) vs. low (6) | 0.645 | 0.877 (0.501–1.535) |
| Membranous β-catenin expression high (>8) vs. low (8) | 0.745 | 0.736 (0.545–1.544) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liao, Y.; Badmann, S.; Kaltofen, T.; Mayr, D.; Schmoeckel, E.; Deuster, E.; Mannewitz, M.; Landgrebe, S.; Kolben, T.; Hester, A.; et al. Platelet-Activating Factor Acetylhydrolase Expression in BRCA1 Mutant Ovarian Cancer as a Protective Factor and Potential Negative Regulator of the Wnt Signaling Pathway. Biomedicines 2021, 9, 706. https://doi.org/10.3390/biomedicines9070706
Liao Y, Badmann S, Kaltofen T, Mayr D, Schmoeckel E, Deuster E, Mannewitz M, Landgrebe S, Kolben T, Hester A, et al. Platelet-Activating Factor Acetylhydrolase Expression in BRCA1 Mutant Ovarian Cancer as a Protective Factor and Potential Negative Regulator of the Wnt Signaling Pathway. Biomedicines. 2021; 9(7):706. https://doi.org/10.3390/biomedicines9070706
Chicago/Turabian StyleLiao, Yue, Susann Badmann, Till Kaltofen, Doris Mayr, Elisa Schmoeckel, Eileen Deuster, Mareike Mannewitz, Sarah Landgrebe, Thomas Kolben, Anna Hester, and et al. 2021. "Platelet-Activating Factor Acetylhydrolase Expression in BRCA1 Mutant Ovarian Cancer as a Protective Factor and Potential Negative Regulator of the Wnt Signaling Pathway" Biomedicines 9, no. 7: 706. https://doi.org/10.3390/biomedicines9070706
APA StyleLiao, Y., Badmann, S., Kaltofen, T., Mayr, D., Schmoeckel, E., Deuster, E., Mannewitz, M., Landgrebe, S., Kolben, T., Hester, A., Beyer, S., Burges, A., Mahner, S., Jeschke, U., Trillsch, F., & Czogalla, B. (2021). Platelet-Activating Factor Acetylhydrolase Expression in BRCA1 Mutant Ovarian Cancer as a Protective Factor and Potential Negative Regulator of the Wnt Signaling Pathway. Biomedicines, 9(7), 706. https://doi.org/10.3390/biomedicines9070706







