Natural Photosensitizers in Antimicrobial Photodynamic Therapy
Abstract
1. Introduction
2. Key Factors in Antimicrobial Photodynamic Therapy (APDT)
2.1. Light Sources
2.2. Oxygen
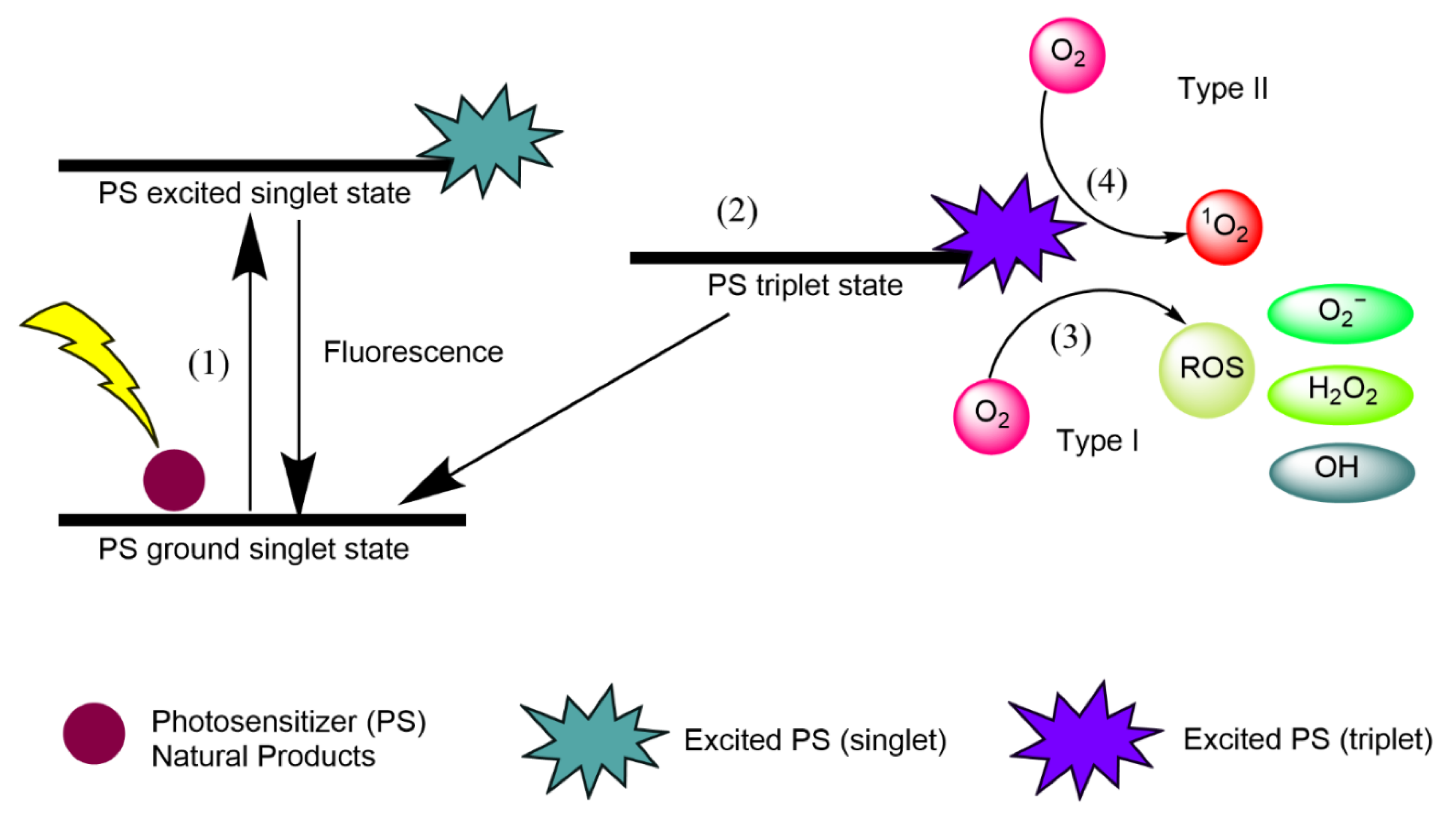
- (1)
- Natural product PSs are converted from the ground singlet state into the excited singlet state when exposed to a specific wavelength of light.
- (2)
- If the PS in the excited singlet state does not return to the initial ground state, it can be subjected to intersystem crossing into the triplet excited state.
- (3)
- A type I reaction comprises transferring a hydrogen atom from PS to an organic molecule to form radicals, and the reduced PS interacts with oxygen through a redox reaction, forming ROS and a superoxide anion radical (O2•−) [20].
- (4)
- The type II reaction comprises direct energy transfer from the activated PS to molecular oxygen to form singlet oxygen (1O2) and is simpler than the type I reaction.
2.3. Photosensitizers (PSs)
- Have a strong absorption peak in the red to near-infrared spectral region (between 650 and 800 nm) [15];
- Possess a substantial triplet quantum yield leading to good ROS production upon irradiation [21];
- Have high tissue selectivity [22];
- Exhibit no dark toxicity [23];
- Have ideal solubility to maintain lipophilic ability to cross the phospholipid membrane and prevent self-aggregation [24];
- Exhibit high stability under storage conditions [25];
- Kill microorganisms sufficiently without damaging eukaryotic host cells [26];
- Display optimal absorption, distribution, metabolism, and excretion (ADME) [24];
- Have a small size to enable microbial membrane permeation [2]; and
- Have low manufacturing costs [23].
3. APDT Targeting Diseases and Organisms
3.1. Target Components of Pathogenic Cells
3.2. Gram-Positive Bacteria
3.3. Gram-Negative Bacteria
3.4. Fungi and Oomycetes
3.5. Viruses
3.6. Mosquitoes
3.7. Plants
3.8. Parasitic Protozoa
4. Preclinical and Clinical APDT Studies
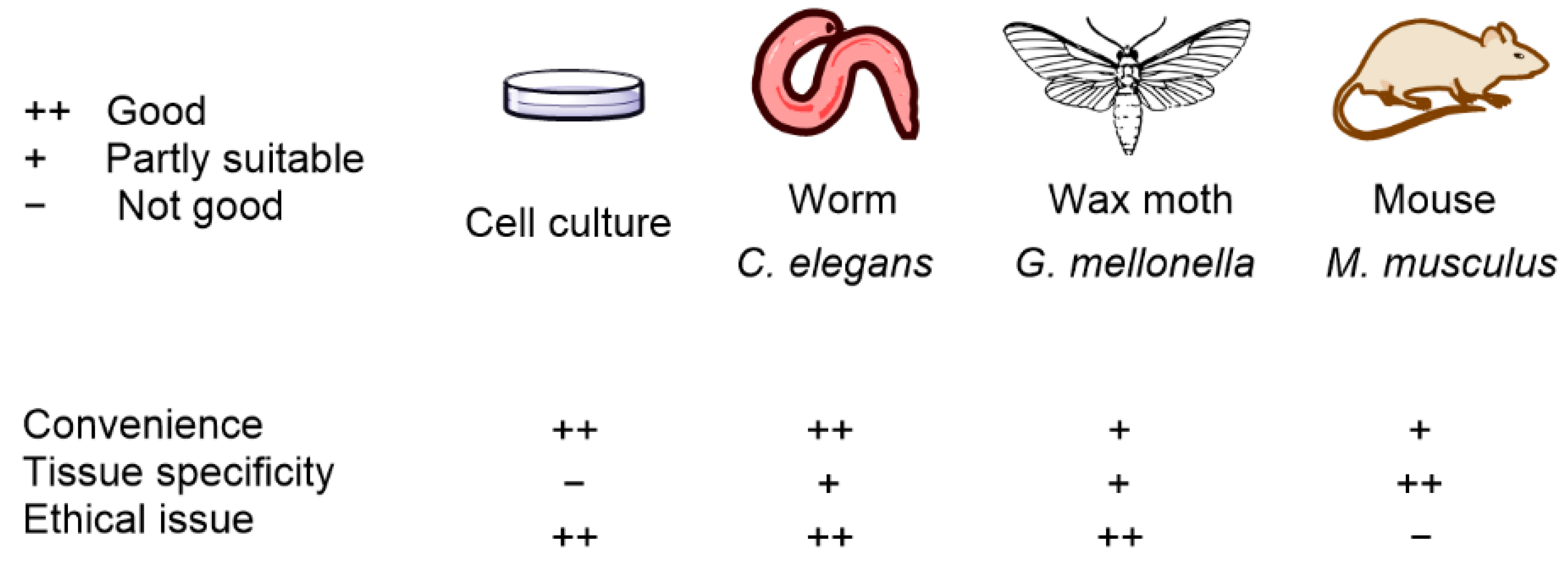
4.1. Preclinical Animal APDT Studies
4.2. Clinical APDT Trials
5. Natural PSs in APDT
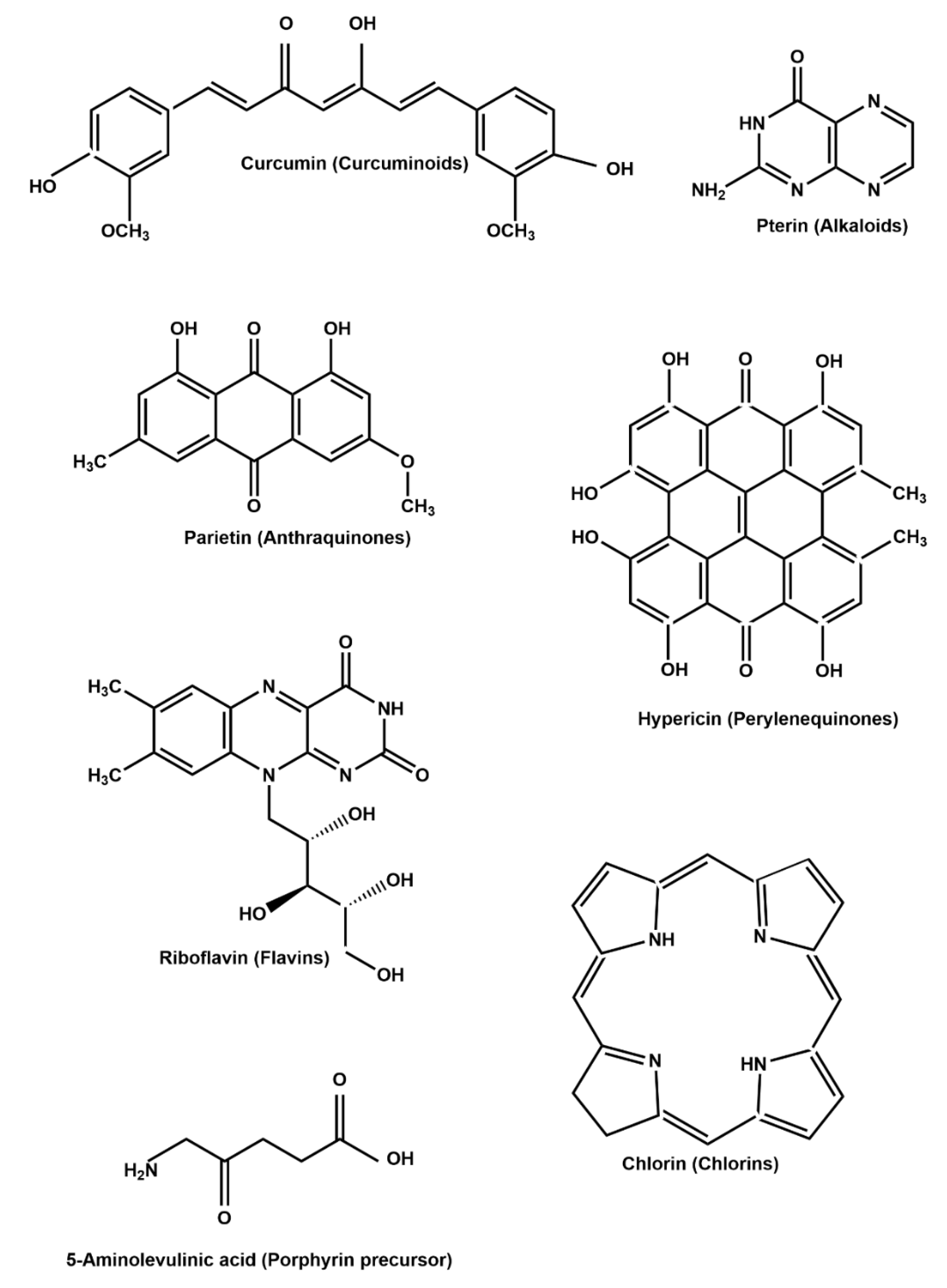
5.1. Curcuminoids
5.2. Alkaloids
5.3. Anthraquinones
5.4. Perylenequinones
5.5. Flavins
5.6. Porphyrins, Chlorins, and Bacteriochlorins
5.7. Natural Extracts
6. Synthetic Derivatives of Natural PSs
- Semisynthesis of chlorin-based PSs from natural chlorophylls [130]; and
7. Current Limitations of APDT
7.1. Limitation of APDT against Gram-Negative Bacteria
7.2. Limitation of Selectivity (Human Cells and Good Bacteria)
7.3. Limitations of Solubility and Light Penetration
7.4. Limitation of Economic Efficiency and Quality Control
8. Emerging Technologies and Solutions to Current APDT Limitations
8.1. Cell Surface Engineering for Enhanced Delivery and Solubility of PSs
8.2. Protein Engineering for Increased Specificity
8.3. Enhanced PS Uptake Strategies via Electroporation and Chemicals
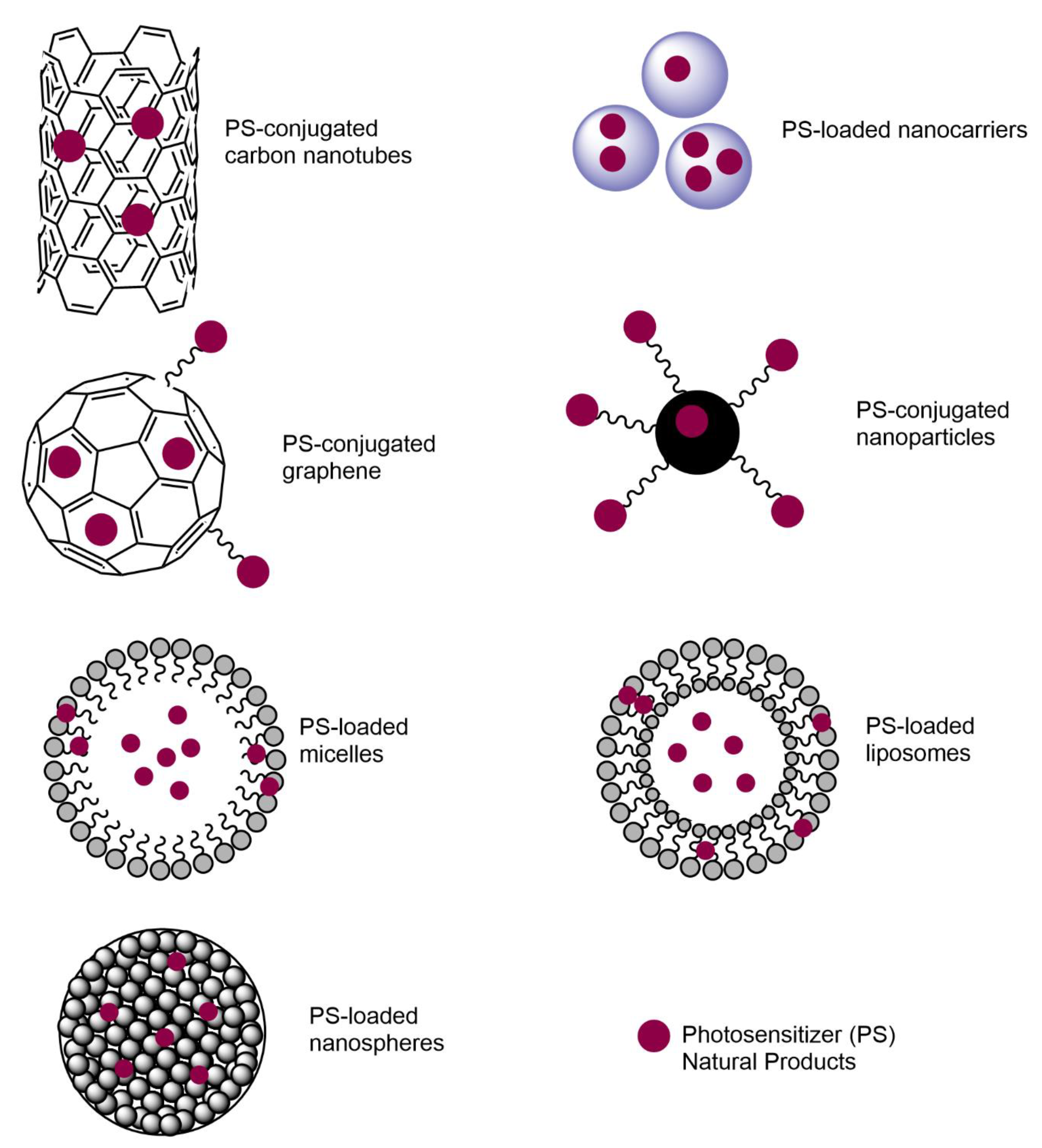
8.4. Nanotechnology for Increased Solubility, Specificity, and Cost
8.5. Computational Simulations of APDT
8.6. Sonodynamic Therapy (SDT) for Enhanced Efficiency
9. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- St. Denis, T.G.; Dai, T.; Izikson, L.; Astrakas, C.; Anderson, R.R.; Hamblin, M.R.; Tegos, G.P. All you need is light, antimicrobial photoinactivation as an evolving and emerging discovery strategy against infectious disease. Virulence 2011, 2, 509–520. [Google Scholar] [CrossRef] [PubMed]
- Maldonado-Carmona, N.; Ouk, T.S.; Calvete, M.J.F.; Pereira, M.M.; Villandier, N.; Leroy-Lhez, S. Conjugating biomaterials with photosensitizers: Advances and perspectives for photodynamic antimicrobial chemotherapy. Photochem. Photobiol. Sci. 2020, 19, 445–461. [Google Scholar] [CrossRef] [PubMed]
- Yagnik, D.; Serafin, V.; Shah, A.J. Antimicrobial activity of apple cider vinegar against Escherichia coli, Staphylococcus aureus and Candida albicans; downregulating cytokine and microbial protein expression. Sci. Rep. 2018, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Turker, H.; Yıldırım, A.B. Screening for antibacterial activity of some turkish plants against fish pathogens: A possible alternative in the treatment of bacterial infections. Biotechnol. Biotechnol. Equip. 2015, 29, 281–288. [Google Scholar] [CrossRef]
- Pang, Z.; Raudonis, R.; Glick, B.R.; Lin, T.J.; Cheng, Z. Antibiotic resistance in Pseudomonas aeruginosa: Mechanisms and alternative therapeutic strategies. Biotechnol. Adv. 2019, 37, 177–192. [Google Scholar] [CrossRef] [PubMed]
- Harbarth, S.; Balkhy, H.H.; Goossens, H.; Jarlier, V.; Kluytmans, J.; Laxminarayan, R.; Saam, M.; Van Belkum, A.; Pittet, D. Antimicrobial resistance: One world, one fight! Antimicrob. Resist. Infect. Control. 2015, 4, 1–15. [Google Scholar] [CrossRef]
- Sowa, A.; Voskuhl, J. Host-guest complexes—Boosting the performance of photosensitizers. Int. J. Pharm. 2020, 586, 119595. [Google Scholar] [CrossRef] [PubMed]
- Corrêa, T.Q.; Blanco, K.C.; Soares, J.M.; Inada, N.M.; Kurachi, C.; Golim, M.d.A.; Deffune, E.; Bagnato, V.S. Photodynamic inactivation for in vitro decontamination of Staphylococcus aureus in whole blood. Photodiagnosis Photodyn. Ther. 2019, 28, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Vieira, C.; Santos, A.; Mesquita, M.Q.; Gomes, A.T.P.C.; Neves, M.G.P.M.S.; Faustino, M.A.F.; Almeida, A. Advances in aPDT based on the combination of a porphyrinic formulation with potassium iodide: Effectiveness on bacteria and fungi planktonic/biofilm forms and viruses. J. Porphyr. Phthalocyanines 2019, 23, 534–545. [Google Scholar] [CrossRef]
- Park, D.; Kim, M.; Choi, J.W.; Baek, J.H.; Lee, S.H.; Baek, K. Antimicrobial photodynamic therapy efficacy against specific pathogenic periodontitis bacterial species. Photodiagnosis Photodyn. Ther. 2020, 30, 101688. [Google Scholar] [CrossRef] [PubMed]
- Simões, J.C.S.; Sarpaki, S.; Papadimitroulas, P.; Therrien, B.; Loudos, G. Conjugated Photosensitizers for Imaging and PDT in Cancer Research. J. Med. Chem. 2020, 63, 14119–14150. [Google Scholar] [CrossRef]
- Ailioaie, L.M.; Litscher, G. Curcumin and photobiomodulation in chronic viral hepatitis and hepatocellular carcinoma. Int. J. Mol. Sci. 2020, 21, 7150. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Liu, T.; Li, Z.; Zhang, X. Progress of photodynamic therapy applications in the treatment of musculoskeletal sarcoma (Review). Oncol. Lett. 2014, 8, 1403–1408. [Google Scholar] [CrossRef]
- Kashef, N.; Huang, Y.Y.; Hamblin, M.R. Advances in antimicrobial photodynamic inactivation at the nanoscale. Nanophotonics 2017, 6, 853–879. [Google Scholar] [CrossRef]
- Babu, B.; Mack, J.; Nyokong, T. Sn(iv) N-confused porphyrins as photosensitizer dyes for photodynamic therapy in the near IR region. Dalton Trans. 2020, 49, 15180–15183. [Google Scholar] [CrossRef] [PubMed]
- Luksiene, Z.; Brovko, L. Antibacterial Photosensitization-Based Treatment for Food Safety. Food Eng. Rev. 2013, 5, 185–199. [Google Scholar] [CrossRef]
- Zhuang, J.; Yang, H.; Li, Y.; Wang, B.; Li, N.; Zhao, N. Efficient photosensitizers with aggregation-induced emission characteristics for lysosome- And Gram-positive bacteria-targeted photodynamic therapy. Chem. Commun. 2020, 56, 2630–2633. [Google Scholar] [CrossRef] [PubMed]
- Memar, M.Y.; Ghotaslou, R.; Samiei, M.; Adibkia, K. Antimicrobial use of reactive oxygen therapy: Current insights. Infect. Drug Resist. 2018, 11, 567–576. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Xuan, Y.; Koide, Y.; Zhiyentayev, T.; Tanaka, M.; Hamblin, M.R. Type I and Type II mechanisms of antimicrobial photodynamic therapy: An in vitro study on gram-negative and gram-positive bacteria. Lasers Surg. Med. 2012, 44, 490–499. [Google Scholar] [CrossRef] [PubMed]
- Villacorta, R.B.; Roque, K.F.J.; Tapang, G.A.; Jacinto, S.D. Plant extracts as natural photosensitizers in photodynamic therapy: In vitro activity against human mammary adenocarcinoma MCF-7 cells. Asian Pac. J. Trop. Biomed. 2017, 7, 358–366. [Google Scholar] [CrossRef]
- Wang, Z.; Zhao, J. Bodipy–Anthracene Dyads as Triplet Photosensitizers: Effect of Chromophore Orientation on Triplet-State Formation Efficiency and Application in Triplet–Triplet Annihilation Upconversion. Org. Lett. 2017, 19, 4492–4495. [Google Scholar] [CrossRef]
- Kwiatkowski, S.; Knap, B.; Przystupski, D.; Saczko, J.; Kędzierska, E.; Knap-Czop, K.; Kotlińska, J.; Michel, O.; Kotowski, K.; Kulbacka, J. Photodynamic therapy—Mechanisms, photosensitizers and combinations. Biomed. Pharmacother. 2018, 106, 1098–1107. [Google Scholar] [CrossRef]
- Abrahamse, H.; Hamblin, M.R. New photossensitizersfot photodynamic therapy. Biochem. J. 2017, 473, 347–364. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Q.; Wu, J.; Pang, X.; Jiang, Y.; Wang, P.; Leung, A.W.; Gao, L.; Jiang, S.; Xu, C. Discovery and Development of Natural Products and their Derivatives as Photosensitizers for Photodynamic Therapy. Curr. Med. Chem. 2017, 25, 839–860. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Hernández, F.; Granada-Ramírez, D.A.; Arias-Cerón, J.S.; Rodriguez-Fragoso, P.; Mendoza-Álvarez, J.G.; Ramón-Gallegos, E.; Cruz-Orea, A.; Luna-Arias, J.P. Use of nanostructured materials in drug delivery. In Nanobiomaterials; Narayan, R., Ed.; Woodhead Publishing: Sawston, UK, 2018; pp. 503–549. [Google Scholar]
- Cieplik, F.; Deng, D.; Crielaard, W.; Buchalla, W.; Hellwig, E.; Al-Ahmad, A.; Maisch, T. Antimicrobial photodynamic therapy–what we know and what we don’t. Crit. Rev. Microbiol. 2018, 44, 571–589. [Google Scholar] [CrossRef] [PubMed]
- Paramanantham, P.; Siddhardha, B.; Lal, S.B.S.; Sharan, A.; Alyousef, A.A.; Al Dosary, M.S.; Arshad, M.; Syed, A. Antimicrobial photodynamic therapy on Staphylococcus aureus and Escherichia coli using malachite green encapsulated mesoporous silica nanoparticles: An in vitro study. PeerJ 2019. [Google Scholar] [CrossRef]
- Hamblin, M.R.; Hasan, T. Photodynamic therapy: A new antimicrobial approach to infectious disease? Photochem. Photobiol. Sci. 2004, 3, 436–450. [Google Scholar] [CrossRef]
- Hamblin, M.R.; Abrahamse, H. Oxygen-Independent Antimicrobial Photoinactivation: Type III Photochemical Mechanism? Antibiotics 2020, 9, 53. [Google Scholar] [CrossRef] [PubMed]
- Rubin, J.E.; Ball, K.R.; Chirino-Trejo, M. Antimicrobial susceptibility of Staphylococcus aureus and Staphylococcus pseudintermedius isolated from various animals. Can. Vet. J. 2011, 52, 162–164. [Google Scholar]
- Liang, X.Y.; Zou, Z.H.; Zou, Z.; Li, C.Y.; Dong, X.X.; Yin, H.J.; Yan, G.H. Effect of antibacterial photodynamic therapy on Streptococcus mutans plaque biofilm in vitro. J. Innov. Opt. Health Sci. 2020, 13, 2050022. [Google Scholar] [CrossRef]
- de Regt, M.J.A.; van Schaik, W.; van Luit-Asbroek, M.; Dekker, H.A.T.; van Duijkeren, E.; Koning, C.J.M.; Bonten, M.J.M.; Willems, R.J.L. Hospital and community ampicillin-resistant Enterococcus faecium are evolutionarily closely linked but have diversified through niche adaptation. PLoS ONE 2012, 7. [Google Scholar] [CrossRef]
- Woźniak, A.; Kruszewska, B.; Pierański, M.K.; Rychłowski, M.; Grinholc, M. Antimicrobial Photodynamic Inactivation Affects the Antibiotic Susceptibility of Enterococcus spp. Clinical Isolates in Biofilm and Planktonic Cultures. Biomolecules 2021, 11, 693. [Google Scholar] [CrossRef]
- Fila, G.; Krychowiak, M.; Rychlowski, M.; Bielawski, K.P.; Grinholc, M. Antimicrobial blue light photoinactivation of Pseudomonas aeruginosa: Quorum sensing signaling molecules, biofilm formation and pathogenicity. J. Biophotonics 2018, 11, 1–12. [Google Scholar] [CrossRef]
- Abdulrahman, H.; Misba, L.; Ahmad, S.; Khan, A.U. Curcumin induced photodynamic therapy mediated suppression of quorum sensing pathway of Pseudomonas aeruginosa: An approach to inhibit biofilm in vitro. Photodiagnosis Photodyn. Ther. 2020, 30, 101645. [Google Scholar] [CrossRef] [PubMed]
- Alam, S.T.; Le, T.A.N.; Park, J.S.; Kwon, H.C.; Kang, K. Antimicrobial biophotonic treatment of ampicillin-resistant Pseudomonas aeruginosa with hypericin and ampicillin cotreatment followed by orange light. Pharmaceutics 2019, 641. [Google Scholar] [CrossRef]
- Faraoni, P.; Gnerucci, A.; Ranaldi, F.; Orsini, B.; Romano, G.; Fusi, F. Side effects of intra-gastric photodynamic therapy: An in vitro study. J. Photochem. Photobiol. B Biol. 2018, 186, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Ganz, R.A.; Viveiros, J.; Ahmad, A.; Ahmadi, A.; Khalil, A.; Tolkoff, M.J.; Nishioka, N.S.; Hamblin, M.R. Helicobacter pylori in patients can be killed by visible light. Lasers Surg. Med. 2005, 36, 260–265. [Google Scholar] [CrossRef] [PubMed]
- Morici, P.; Battisti, A.; Tortora, G.; Menciassi, A.; Checcucci, G.; Ghetti, F.; Sgarbossa, A. The in vitro Photoinactivation of Helicobacter pylori by a Novel LED-Based Device. Front. Microbiol. 2020, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Laguna, V.; Gilaberte, Y.; Millán-Lou, M.I.; Agut, M.; Nonell, S.; Rezusta, A.; Hamblin, M.R. A combination of photodynamic therapy and antimicrobial compounds to treat skin and mucosal infections: A systematic review. Photochem. Photobiol. Sci. 2019, 18, 1020–1029. [Google Scholar] [CrossRef]
- Sung, N.; Back, S.; Jung, J.H.; Kim, K.H.; Kim, J.K.; Lee, J.H.; Ra, Y.; Yang, H.C.; Lim, C.; Cho, S.; et al. Inactivation of multidrug resistant (MDR)- and extensively drug resistant (XDR)-Mycobacterium tuberculosis by photodynamic therapy. Photodiagnosis Photodyn. Ther. 2013, 10, 694–702. [Google Scholar] [CrossRef]
- Zambounis, A.; Sytar, O.; Valasiadis, D.; Hilioti, Z. Effect of photosensitisers on growth and morphology of phytophthora citrophthora coupled with leaf bioassays in pear seedlings. Plant Prot. Sci. 2020, 56, 74–82. [Google Scholar] [CrossRef]
- Hampton, T. New Flu Antiviral Candidate May Thwart Drug Resistance. JAMA 2020, 323, 17. [Google Scholar] [CrossRef] [PubMed]
- Monjo, A.L.A.; Pringle, E.S.; Thornbury, M.; Duguay, B.A.; Monro, S.M.A.; Hetu, M.; Knight, D.; Cameron, C.G.; McFarland, S.A.; McCormick, C. Photodynamic inactivation of herpes simplex viruses. Viruses 2018, 10, 532. [Google Scholar] [CrossRef] [PubMed]
- Almeida, A.; Faustino, M.A.F.; Neves, M.G.P.M.S. Antimicrobial photodynamic therapy in the control of COVID-19. Antibiotics 2020, 9, 320. [Google Scholar] [CrossRef] [PubMed]
- Younis, M.; Khater, H.h.; Hussein, A.; farag, S.; Aboelela, H.; Rashed, G. The Potential Role of Photosensitizers in Fight against Mosquitoes: Phototoxicity of Rose Bengal against Culex Pipiens Larvae. Benha Med. J. 2020, 38. [Google Scholar] [CrossRef]
- Siewert, B.; Stuppner, H. The photoactivity of natural products—An overlooked potential of phytomedicines? Phytomedicine 2019, 60, 152985. [Google Scholar] [CrossRef]
- Issawi, M.; Sol, V.; Riou, C. Plant photodynamic stress: What’s new? Front. Plant Sci. 2018, 9, 681. [Google Scholar] [CrossRef] [PubMed]
- Miranda, N.; Gerola, A.P.; Novello, C.R.; Ueda-Nakamura, T.; de Oliveira Silva, S.; Dias-Filho, B.P.; Hioka, N.; de Mello, J.C.P.; Nakamura, C.V. Pheophorbide a, a compound isolated from the leaves of Arrabidaea chica, induces photodynamic inactivation of Trypanosoma cruzi. Photodiagnosis Photodyn. Ther. 2017, 19, 256–265. [Google Scholar] [CrossRef]
- Dimmer, J.; Cabral, F.V.; Sabino, C.P.; Silva, C.R.; Núñez-Montoya, S.C.; Cabrera, J.L.; Ribeiro, M.S. Natural anthraquinones as novel photosentizers for antiparasitic photodynamic inactivation. Phytomedicine 2019, 61, 152894. [Google Scholar] [CrossRef] [PubMed]
- Galo, Í.D.C.; Carvalho, J.A.; Santos, J.L.M.C.; Braoios, A.; Prado, R.P. The ineffectiveness of antimicrobial photodynamic therapy in the absence of preincubation of the microorganisms in the photosensitizer. Fisioter. Mov. 2020, 33, 1–9. [Google Scholar] [CrossRef]
- Rupel, K.; Zupin, L.; Brich, S.; Mardirossian, M.; Ottaviani, G.; Gobbo, M.; Di Lenarda, R.; Pricl, S.; Crovella, S.; Zacchigna, S.; et al. Antimicrobial activity of amphiphilic nanomicelles loaded with curcumin against Pseudomonas aeruginosa alone and activated by blue laser light. J. Biophotonics 2021, 14, e202000350. [Google Scholar] [CrossRef] [PubMed]
- Sahu, K.; Sharma, M.; Dube, A.; Gupta, P.K. Topical antimicrobial photodynamic therapy improves angiogenesis in wounds of diabetic mice. Lasers Med. Sci. 2015, 30, 1923–1929. [Google Scholar] [CrossRef] [PubMed]
- Mai, B.; Gao, Y.; Li, M.; Wang, X.; Zhang, K.; Liu, Q.; Xu, C.; Wang, P. Photodynamic antimicrobial chemotherapy for staphylococcus aureus and multidrug-resistant bacterial burn infection in vitro and in vivo. Int. J. Nanomed. 2017, 12, 5915–5931. [Google Scholar] [CrossRef] [PubMed]
- Sakima, V.T.; Barbugli, P.A.; Cerri, P.S.; Chorilli, M.; Carmello, J.C.; Pavarina, A.C.; De Oliveira Mima, E.G. Antimicrobial photodynamic therapy mediated by curcumin-loaded polymeric nanoparticles in a murine model of oral candidiasis. Molecules 2018, 2075. [Google Scholar] [CrossRef] [PubMed]
- Alam, S.T.; Hwang, H.; Son, J.D.; Nguyen, U.T.T.; Park, J.S.; Kwon, H.C.; Kwon, J.; Kang, K. Natural photosensitizers from Tripterygium wilfordii and their antimicrobial photodynamic therapeutic effects in a Caenorhabditis elegans model. J. Photochem. Photobiol. B Biol. 2021, 218, 112184. [Google Scholar] [CrossRef]
- Kim, J.Y.; Le, T.A.N.; Lee, S.Y.; Song, D.G.; Hong, S.C.; Cha, K.H.; Lee, J.W.; Pan, C.H.; Kang, K. 3,3′-Diindolylmethane Improves Intestinal Permeability Dysfunction in Cultured Human Intestinal Cells and the Model Animal Caenorhabditis elegans. J. Agric. Food Chem. 2019, 67, 9277–9285. [Google Scholar] [CrossRef] [PubMed]
- Le, T.A.N.; Selvaraj, B.; Lee, J.W.; Kang, K. Measuring the Effects of Bacteria and Chemicals on the Intestinal Permeability of Caenorhabditis elegans. J. Vis. Exp. 2019. [Google Scholar] [CrossRef]
- Lee, S.Y.; Kang, K. Measuring the Effect of Chemicals on the Growth and Reproduction of Caenorhabditis elegans. J. Vis. Exp. 2017. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Kim, J.Y.; Jung, Y.J.; Kang, K. Toxicological evaluation of the topoisomerase inhibitor, etoposide, in the model animal Caenorhabditis elegans and 3T3-L1 normal murine cells. Environ. Toxicol. 2017, 32, 1836–1843. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Xu, M.; Pan, W.; Wang, M.; Wu, X.; Dai, S.; Li, L.; Zeng, K. Antimicrobial and immunomodulatory responses of photodynamic therapy in Galleria mellonella model. BMC Microbiol. 2020, 20, 4–11. [Google Scholar] [CrossRef]
- Figueiredo-Godoi, L.M.A.; Menezes, R.T.; Carvalho, J.S.; Garcia, M.T.; Segundo, A.G.; Jorge, A.O.C.; Junqueira, J.C. Exploring the Galleria mellonella model to study antifungal photodynamic therapy. Photodiagnosis Photodyn. Ther. 2019, 27, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Huang, Y.Y.; Wang, Y.; Wang, X.; Hamblin, M.R. Antimicrobial photodynamic therapy to control clinically relevant biofilm infections. Front. Microbiol. 2018, 9, 1–24. [Google Scholar] [CrossRef]
- Panhóca, V.H.; Florez, F.L.E.; Corrêa, T.Q.; Paolillo, F.R.; De Souza, C.W.O.; Bagnato, V.S. Oral decontamination of orthodontic patients using photodynamic therapy mediated by blue-light irradiation and curcumin associated with sodium dodecyl sulfate. Photomed. Laser Surg. 2016, 34, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Ivanaga, C.A.; Miessi, D.M.J.; Nuernberg, M.A.A.; Claudio, M.M.; Garcia, V.G.; Theodoro, L.H. Antimicrobial photodynamic therapy (aPDT) with curcumin and LED, as an enhancement to scaling and root planing in the treatment of residual pockets in diabetic patients: A randomized and controlled split-mouth clinical trial. Photodiagnosis Photodyn. Ther. 2019, 27, 388–395. [Google Scholar] [CrossRef]
- Cristina Da Silva, F.; Rosa, L.P.; Santos, G.P.D.O.; Inada, N.M.; Blanco, K.C.; Araújo, T.S.D.; Bagnato, V.S. Total mouth photodynamic therapy mediated by blue led and curcumin in individuals with AIDS. Expert Rev. Anti Infect. Ther. 2020, 18, 689–696. [Google Scholar] [CrossRef]
- Song, B.H.; Lee, D.H.; Kim, B.C.; Ku, S.H.; Park, E.J.; Kwon, I.H.; Kim, K.H.; Kim, K.J. Photodynamic therapy using chlorophyll-a in the treatment of acne vulgaris: A randomized, single-blind, split-face study. J. Am. Acad. Dermatol. 2014, 71, 764–771. [Google Scholar] [CrossRef] [PubMed]
- Nicklas, C.; Rubio, R.; Cárdenas, C.; Hasson, A. Comparison of efficacy of aminolaevulinic acid photodynamic therapy vs. adapalene gel plus oral doxycycline for treatment of moderate acne vulgaris–A simple, blind, randomized, and controlled trial. Photodermatol. Photoimmunol. Photomed. 2019, 35, 3–10. [Google Scholar] [CrossRef]
- Zangirolami, A.C.; Dias, L.D.; Blanco, K.C.; Vinagreiro, C.S.; Inada, N.M.; Arnaut, L.G.; Pereira, M.M.; Bagnato, V.S. Avoiding ventilator-associated pneumonia: Curcumin-functionalized endotracheal tube and photodynamic action. Proc. Natl. Acad. Sci. USA 2020, 117, 22967. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Zhang, X.; Ma, C.; Yu, N.; Wang, J.; Xia, L.; Ge, X.; Liu, M.; Duan, A. Clinical analysis of five methods used to treat condylomata acuminata. Dermatology 2014, 227, 338–345. [Google Scholar] [CrossRef]
- Alves, F.; Alonso, G.C.; Carmello, J.C.; Mima, E.G.d.O.; Bagnato, V.S.; Pavarina, A.C. Antimicrobial Photodynamic Therapy mediated by Photodithazine® in the treatment of denture stomatitis: A case report. Photodiagnosis Photodyn. Ther. 2018, 21, 168–171. [Google Scholar] [CrossRef]
- Alves, F.; Carmello, J.C.; Mima, E.G.D.; Costa, C.A.D.; Bagnato, V.S.; Pavarina, A.C. Photodithazine-mediated antimicrobial photodynamic therapy against fluconazole-resistant Candida albicans in vivo. Med. Mycol. 2019, 57, 609–617. [Google Scholar] [CrossRef]
- Wiegell, S.R.; Wulf, H.C. Photodynamic therapy of acne vulgaris using 5-aminolevulinic acid versus methyl aminolevulinate. J. Am. Acad. Dermatol. 2006, 54, 647–651. [Google Scholar] [CrossRef]
- da Silva Souza Campanholi, K.; Jaski, J.M.; da Silva Junior, R.C.; Zanqui, A.B.; Lazarin-Bidóia, D.; da Silva, C.M.; da Silva, E.A.; Hioka, N.; Nakamura, C.V.; Cardozo-Filho, L.; et al. Photodamage on Staphylococcus aureus by natural extract from Tetragonia tetragonoides (Pall.) Kuntze: Clean method of extraction, characterization and photophysical studies. J. Photochem. Photobiol. B Biol. 2020, 203, 111763. [Google Scholar] [CrossRef]
- Bonifácio, D.; Martins, C.; David, B.; Lemos, C.; Neves, M.G.P.M.S.; Almeida, A.; Pinto, D.C.G.A.; Faustino, M.A.F.; Cunha, Â. Photodynamic inactivation of Listeria innocua biofilms with food-grade photosensitizers: A curcumin-rich extract of Curcuma longa vs. commercial curcumin. J. Appl. Microbiol. 2018, 125, 282–294. [Google Scholar] [CrossRef]
- Tyagi, P.; Singh, M.; Kumari, H.; Kumari, A.; Mukhopadhyay, K. Bactericidal activity of curcumin I is associated with damaging of bacterial membrane. PLoS ONE 2015, 10, 1–15. [Google Scholar] [CrossRef]
- Chang, K.C.; Cheng, Y.Y.; Lai, M.J.; Hu, A. Identification of carbonylated proteins in a bactericidal process induced by curcumin with blue light irradiation on imipenem-resistant Acinetobacter baumannii. Rapid Commun. Mass Spectrom. 2020, 34. [Google Scholar] [CrossRef]
- Gao, J.; Matthews, K.R. Effects of the photosensitizer curcumin in inactivating foodborne pathogens on chicken skin. Food Control. 2020, 109, 106959. [Google Scholar] [CrossRef]
- Huang, J.; Chen, B.; Li, H.; Zeng, Q.H.; Wang, J.J.; Liu, H.; Pan, Y.; Zhao, Y. Enhanced antibacterial and antibiofilm functions of the curcumin-mediated photodynamic inactivation against Listeria monocytogenes. Food Control 2020, 108, 106886. [Google Scholar] [CrossRef]
- Chen, B.; Huang, J.; Li, H.; Zeng, Q.H.; Wang, J.J.; Liu, H.; Pan, Y.; Zhao, Y. Eradication of planktonic Vibrio parahaemolyticus and its sessile biofilm by curcumin-mediated photodynamic inactivation. Food Control 2020, 113, 107181. [Google Scholar] [CrossRef]
- Gong, C.; Li, Y.; Gao, R.; Xiao, F.; Zhou, X.; Wang, H.; Xu, H.; Wang, R.; Huang, P.; Zhao, Y. Inactivation of specific spoilage organism (Pseudomonas) of sturgeon by curcumin-mediated photodynamic inactivation. Photodiagnosis Photodyn. Ther. 2020, 31. [Google Scholar] [CrossRef]
- Pan, H.; Wang, D.; Li, H.; Zhang, F. In vitro antimicrobial effect of curcumin-based photodynamic therapy on Porphyromonas gingivalis and Aggregatibacter actinomycetemcomitans. Photodiagnosis Photodyn. Ther. 2020, 32, 102055. [Google Scholar] [CrossRef]
- Paolillo, F.R.; Rodrigues, P.G.S.; Bagnato, V.S.; Alves, F.; Pires, L.; Corazza, A.V. The effect of combined curcumin-mediated photodynamic therapy and artificial skin on Staphylococcus aureus–infected wounds in rats. Lasers Med. Sci. 2020. [Google Scholar] [CrossRef] [PubMed]
- Soares, J.M.; Inada, N.M.; Bagnato, V.S.; Blanco, K.C. Evolution of surviving Streptoccocus pyogenes from pharyngotonsillitis patients submit to multiple cycles of antimicrobial photodynamic therapy. J. Photochem. Photobiol. B Biol. 2020, 210, 111985. [Google Scholar] [CrossRef] [PubMed]
- Tosato, M.G.; Schilardi, P.; de Mele, M.F.L.; Thomas, A.H.; Lorente, C.; Miñán, A. Synergistic effect of carboxypterin and methylene blue applied to antimicrobial photodynamic therapy against mature biofilm of Klebsiella pneumoniae. Heliyon 2020, 6, e03522. [Google Scholar] [CrossRef]
- Tao, R.; Zhang, F.; Tang, Q.-j.; Xu, C.-s.; Ni, Z.-J.; Meng, X.-h. Effects of curcumin-based photodynamic treatment on the storage quality of fresh-cut apples. Food Chem. 2019, 274, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Le, T.D.; Phasupan, P.; Nguyen, L.T. Antimicrobial photodynamic efficacy of selected natural photosensitizers against food pathogens: Impacts and interrelationship of process parameters. Photodiagnosis Photodyn. Ther. 2020, 32, 102024. [Google Scholar] [CrossRef]
- Freitas, M.A.; Pereira, A.H.; Pinto, J.G.; Casas, A.; Ferreira-Strixino, J. Bacterial viability after antimicrobial photodynamic therapy with curcumin on multiresistant Staphylococcus aureus. Future Microbiol. 2019, 14, 739–748. [Google Scholar] [CrossRef] [PubMed]
- Rocha, M.P.; Santos, M.S.; Rodrigues, P.L.F.; Araújo, T.S.D.; de Oliveira, J.M.; Rosa, L.P.; Bagnato, V.S.; da Silva, F.C. Photodynamic therapry with curcumin in the reduction of enterococcus faecalis biofilm in bone cavity: rMicrobiological and spectral fluorescense analysis. Photodiagnosis Photodyn. Ther. 2021, 33, 102084. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wu, J.; Xu, C.; Lu, N.; Gao, Y.; Xue, Y.; Li, Z.; Xue, C.; Tang, Q. Inactivation of microbes on fruit surfaces using photodynamic therapy and its influence on the postharvest shelf-life of fruits. Food Sci. Technol. Int. 2020, 26, 696–705. [Google Scholar] [CrossRef]
- Araújo, B.P.; Silva, E.A.; Rosa, L.P.; Inada, N.M.; Iermak, I.; Romano, R.A.; Mezzacappo, N.F.; Melo, F.F.; Silva, F.C.; Rocha, M.P.; et al. Morpho-molecular mechanisms study of photodynamic therapy and curcumin larvicide action on wild mosquitoes larvae of genus Aedes. bioRxiv 2020. [Google Scholar] [CrossRef]
- Lan, Y.; Lu, S.; Zheng, B.; Tang, Z.; Li, J.; Zhang, J. Combinatory Effect of ALA-PDT and Itraconazole Treatment for Trichosporon asahii. Lasers Surg. Med. 2020, 1–9. [Google Scholar] [CrossRef]
- Barroso, R.A.; Navarro, R.; Tim, C.R.; de Paula Ramos, L.; de Oliveira, L.D.; Araki, Â.T.; Fernandes, K.G.C.; Macedo, D.; Assis, L. Antimicrobial photodynamic therapy against Propionibacterium acnes biofilms using hypericin (Hypericum perforatum) photosensitizer: In vitro study. Lasers Med. Sci. 2020. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.-N.; Zhang, F.; Tang, Q.-J.; Xu, C.-S.; Meng, X.-H. Effect of photodynamic inactivation of Escherichia coli by hypericin. World J. Microbiol. Biotechnol. 2018, 34, 1–8. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, C.; Zhuge, Y.; Zhang, J.; Xu, K.; Zhang, Q.; Zhang, H.; Chen, H.; Chu, M.; Jia, C. Photodynamic antifungal activity of hypocrellin a against candida albicans. Front. Microbiol. 2019, 10, 1–13. [Google Scholar] [CrossRef]
- Comini, L.R.; Morán Vieyra, F.E.; Mignone, R.A.; Páez, P.L.; Laura Mugas, M.; Konigheim, B.S.; Cabrera, J.L.; Núñez Montoya, S.C.; Borsarelli, C.D. Parietin: An efficient photo-screening pigment in vivo with good photosensitizing and photodynamic antibacterial effects in vitro. Photochem. Photobiol. Sci. 2017, 16, 201–210. [Google Scholar] [CrossRef]
- Walter, A.B.; Simpson, J.; Jenkins, J.L.; Skaar, E.P.; Jansen, E.D. Optimization of optical parameters for improved photodynamic therapy of Staphylococcus aureus using endogenous coproporphyrin III. Photodiagnosis Photodyn. Ther. 2020, 29, 101624. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, J.; Geng, S.; Wang, X.; Cui, Z.; Ma, W.; Yuan, M.; Liu, C.; Ji, Y. Aloe-emodin-mediated antimicrobial photodynamic therapy against multidrug-resistant Acinetobacter baumannii: An in vivo study. Photodiagnosis Photodyn. Ther. 2021, 102311. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.P.; Liu, C.C.; Li, J.; Hao, M.; Ji, Y.H.; Zeng, X.Y. The effects of aloe emodin-mediated antimicrobial photodynamic therapy on drug-sensitive and resistant Candida albicans. Photochem. Photobiol. Sci. 2020, 19, 485–494. [Google Scholar] [CrossRef]
- Josewin, S.W.; Ghate, V.; Kim, M.J.; Yuk, H.G. Antibacterial effect of 460 nm light-emitting diode in combination with riboflavin against Listeria monocytogenes on smoked salmon. Food Control. 2018, 84, 354–361. [Google Scholar] [CrossRef]
- Žudyte, B.; Lukšiene, Ž. Toward better microbial safety of wheat sprouts: Chlorophyllin-based photosensitization of seeds. Photochem. Photobiol. Sci. 2019, 18, 2521–2530. [Google Scholar] [CrossRef] [PubMed]
- Glueck, M.; Hamminger, C.; Fefer, M.; Liu, J.; Plaetzer, K. Save the crop: Photodynamic Inactivation of plant pathogens I: Bacteria. Photochem. Photobiol. Sci. 2019, 18, 1700–1708. [Google Scholar] [CrossRef] [PubMed]
- Cogno, I.S.; Gilardi, P.; Comini, L.; Núñez-Montoya, S.C.; Cabrera, J.L.; Rivarola, V.A. Natural photosensitizers in photodynamic therapy: In vitro activity against monolayers and spheroids of human colorectal adenocarcinoma SW480 cells. Photodiagnosis Photodyn. Ther. 2020, 31, 101852. [Google Scholar] [CrossRef] [PubMed]
- Kashef, N.; Karami, S.; Djavid, G.E. Phototoxic effect of hypericin alone and in combination with acetylcysteine on Staphylococcus aureus biofilms. Photodiagnosis Photodyn. Ther. 2015, 12, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Maisch, T. Photoantimicrobials—An update. Transl. Biophotonics 2020, 2. [Google Scholar] [CrossRef]
- Dias, L.D.; Mfouo-Tynga, I.S. Learning from nature: Bioinspired chlorin-based photosensitizers immobilized on carbon materials for combined photodynamic and photothermal therapy. Biomimetics 2020, 5, 53. [Google Scholar] [CrossRef] [PubMed]
- Pucelik, B.; Sułek, A.; Dąbrowski, J.M. Bacteriochlorins and their metal complexes as NIR-absorbing photosensitizers: Properties, mechanisms, and applications. Coord. Chem. Rev. 2020, 416. [Google Scholar] [CrossRef]
- Seeger, M.G.; Ries, A.S.; Gressler, L.T.; Botton, S.A.; Iglesias, B.A.; Cargnelutti, J.F. In vitro antimicrobial photodynamic therapy using tetra-cationic porphyrins against multidrug-resistant bacteria isolated from canine otitis. Photodiagnosis Photodyn. Ther. 2020, 32, 101982. [Google Scholar] [CrossRef] [PubMed]
- Lan, M.; Zhao, S.; Liu, W.; Lee, C.S.; Zhang, W.; Wang, P. Photosensitizers for Photodynamic Therapy. Adv. Healthc. Mater. 2019, 8, 1–37. [Google Scholar] [CrossRef] [PubMed]
- Andreazza, N.L.; de Lourenço, C.C.; Stefanello, M.É.A.; Atvars, T.D.Z.; Salvador, M.J. Photodynamic antimicrobial effects of bis-indole alkaloid indigo from Indigofera truxillensis Kunth (Leguminosae). Lasers Med. Sci. 2015, 30, 1315–1324. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, M.L.L.; da Mota, A.C.C.; Deana, A.M.; Cavalcante, L.A.d.S.; Horliana, A.C.R.T.; Pavani, C.; Motta, L.J.; Fernandes, K.P.S.; Mesquita-Ferrari, R.A.; da Silva, D.F.T.; et al. Antimicrobial photodynamic therapy with Bixa orellana extract and blue LED in the reduction of halitosis—A randomized, controlled clinical trial. Photodiagnosis Photodyn. Ther. 2020, 30, 101751. [Google Scholar] [CrossRef]
- Dascalu, L.M.; Moldovan, M.; Prodan, D.; Ciotlaus, I.; Popescu, V.; Baldea, I.; Carpa, R.; Sava, S.; Chifor, R.; Badea, M.E. Assessment and characterization of some new photosensitizers for antimicrobial photodynamic therapy (aPDT). Materials 2020, 13, 3012. [Google Scholar] [CrossRef] [PubMed]
- Saitawee, D.; Teerakapong, A.; Morales, N.P.; Jitprasertwong, P.; Hormdee, D. Photodynamic therapy of Curcuma longa extract stimulated with blue light against Aggregatibacter actinomycetemcomitans. Photodiagnosis Photodyn. Ther. 2018, 22, 101–105. [Google Scholar] [CrossRef]
- Hormdee, D.; Rinsathorn, W.; Puasiri, S.; Jitprasertwong, P. Anti-Early Stage of Bacterial Recolonization Effect of Curcuma longa Extract as Photodynamic Adjunctive Treatment. Int. J. Dent. 2020. [Google Scholar] [CrossRef]
- Lee, H.J.; Kang, S.M.; Jeong, S.H.; Chung, K.H.; Kim, B.I. Antibacterial photodynamic therapy with curcumin and Curcuma xanthorrhiza extract against Streptococcus mutans. Photodiagnosis Photodyn. Ther. 2017, 20, 116–119. [Google Scholar] [CrossRef]
- Vollmer, A.; Al-Ahmad, A.; Argyropoulou, A.; Thurnheer, T.; Hellwig, E.; Attin, T.; Vach, K.; Wittmer, A.; Ferguson, K.; Skaltsounis, A.L.; et al. Antimicrobial Photoinactivation Using Visible Light Plus Water-Filtered Infrared-A (VIS + wIRA) and Hypericum Perforatum Modifies In Situ Oral Biofilms. Sci. Rep. 2019, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Ryberg, E.C.; Knight, J.; Kim, J.-H. Farm-to-Tap Water Treatment: Naturally-Sourced Photosensitizers for Enhanced Solar Disinfection of Drinking Water. ACS EST Eng. 2020. [Google Scholar] [CrossRef]
- Giacone, L.; Cordisco, E.; Garrido, M.C.; Petenatti, E.; Sortino, M. Photodynamic activity of Tagetes minuta extracts against superficial fungal infections. Med. Mycol. 2020, 58, 797–809. [Google Scholar] [CrossRef] [PubMed]
- Majiya, H.; Galstyan, A. Dye extract of calyces of Hibiscus sabdariffa has photodynamic antibacterial activity: A prospect for sunlight-driven fresh produce sanitation. Food Sci. Nutr. 2020, 8, 3200–3211. [Google Scholar] [CrossRef]
- Postigo, A.; Funes, M.; Petenatti, E.; Bottai, H.; Pacciaroni, A.; Sortino, M. Antifungal photosensitive activity of Porophyllum obscurum (Spreng.) DC.: Correlation of the chemical composition of the hexane extract with the bioactivity. Photodiagnosis Photodyn. Ther. 2017, 20, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Arfao, A.T.; Ewoti, O.V.N.; Onana, M.F.; Djimeli, C.L.; Tchakonté, S.; Dama, N.K.; Nola, M. Combined effect of eucalyptus microcorys aqueous extract and light on pathogenic escherichia coli survival in aquatic microcosm. J. Microbiol. Biotechnol. Food Sci. 2020, 9, 1003–1008. [Google Scholar] [CrossRef]
- Gonçalves, M.L.L.; Da Mota, A.C.C.; Deana, A.M.; Guedes, G.H.; Cavalcante, L.A.D.S.; Prates, R.A.; Horliana, A.C.R.T.; Pavani, C.; Motta, L.J.; Bitencourt, G.D.B.; et al. Photodynamic therapy with Bixa orellana extract and LED for the reduction of halitosis: Study protocol for a randomized, microbiological and clinical trial. Trials 2018, 19, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Andreazza, N.L.; Caramano De Lourenço, C.; Hernandez-Tasco, Á.J.; Pinheiro, M.L.B.; Alves Stefanello, M.É.; Vilaça Costa, E.; Salvador, M.J. Antimicrobial photodynamic effect of extracts and oxoaporphine alkaloid isomoschatoline from Guatteria blepharophylla. J. Photochem. Photobiol. B Biol. 2016, 160, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Xodo, L.E.; Rapozzi, V.; Zacchigna, M.; Drioli, S.; Zorzet, S. The Chlorophyll Catabolite Pheophorbide a as a Photosensitizer for the Photodynamic Therapy. Curr. Med. Chem. 2012, 19, 799–807. [Google Scholar] [CrossRef] [PubMed]
- Quinn, J.C.; Kessell, A.; Weston, L.A. Secondary Plant Products Causing Photosensitization in Grazing Herbivores: Their Structure, Activity and Regulation. Int. J. Mol. Sci. 2014, 15, 1441. [Google Scholar] [CrossRef] [PubMed]
- Brückner, C. The Breaking and Mending of meso-Tetraarylporphyrins: Transmuting the Pyrrolic Building Blocks. Acc. Chem. Res. 2016, 49, 1080–1092. [Google Scholar] [CrossRef]
- Dudkin, S.V.; Makarova, E.A.; Lukyanets, E.A. Synthesis of chlorins, bacteriochlorins and their tetraaza analogues. Russ. Chem. Rev. 2016, 85, 700–730. [Google Scholar] [CrossRef]
- Christian, B. Tweaking the Pigments of Life: Conversion of Porphyrins to Pyrrole-Modified Porphyrins. Curr. Org. Synth. 2018, 15, 724–728. [Google Scholar] [CrossRef]
- Tomé, A.C.; Neves, M.G.P.M.S.; Cavaleiro, J.A.S. Porphyrins and other pyrrolic macrocycles in cycloaddition reactions. J. Porphyr. Phthalocyanines 2009, 13, 408–414. [Google Scholar] [CrossRef]
- Taniguchi, M.; Lindsey, J.S. Synthetic Chlorins, Possible Surrogates for Chlorophylls, Prepared by Derivatization of Porphyrins. Chem. Rev. 2017, 117, 344–535. [Google Scholar] [CrossRef] [PubMed]
- Lindsey, J.S. De Novo Synthesis of Gem-Dialkyl Chlorophyll Analogues for Probing and Emulating Our Green World. Chem. Rev. 2015, 115, 6534–6620. [Google Scholar] [CrossRef] [PubMed]
- Macedo, P.D.; Corbi, S.T.; de Oliveira, G.J.P.L.; Perussi, J.R.; Ribeiro, A.O.; Marcantonio, R.A.C. Hypericin-glucamine antimicrobial photodynamic therapy in the progression of experimentally induced periodontal disease in rats. Photodiagnosis Photodyn. Ther. 2019, 25, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Bresolí-Obach, R.; Gispert, I.; Peña, D.G.; Boga, S.; Gulias, Ó.; Agut, M.; Vázquez, M.E.; Nonell, S. Triphenylphosphonium cation: A valuable functional group for antimicrobial photodynamic therapy. J. Biophotonics 2018, 11, 1–6. [Google Scholar] [CrossRef]
- Basso, G.; Cargnelutti, J.F.; Oliveira, A.L.; Acunha, T.V.; Weiblen, R.; Flores, E.F.; Iglesias, B.A. Photodynamic inactivation of selected bovine viruses by isomeric cationic tetra-platinated porphyrins. J. Porphyr. Phthalocyanines 2019, 23, 1041–1046. [Google Scholar] [CrossRef]
- Guterres, K.B.; Rossi, G.G.; Menezes, L.B.; Anraku de Campos, M.M.; Iglesias, B.A. Preliminary evaluation of the positively and negatively charge effects of tetra-substituted porphyrins on photoinactivation of rapidly growing mycobacteria. Tuberculosis 2019, 117, 45–51. [Google Scholar] [CrossRef]
- Taslı, H.; Akbıyık, A.; Topaloğlu, N.; Alptüzün, V.; Parlar, S. Photodynamic antimicrobial activity of new porphyrin derivatives against methicillin resistant Staphylococcus aureus. J. Microbiol. 2018, 56, 828–837. [Google Scholar] [CrossRef] [PubMed]
- Biyiklioglu, Z.; Ozturk, I.; Arslan, T.; Tunçel, A.; Ocakoglu, K.; Hosgor-Limoncu, M.; Yurt, F. Synthesis and antimicrobial photodynamic activities of axially {4-[(1E)-3-oxo-3-(2-thienyl)prop-1-en-1-yl]phenoxy} groups substituted silicon phthalocyanine, subphthalocyanine on Gram-positive and Gram-negative bacteria. Dye. Pigment. 2019, 166, 149–158. [Google Scholar] [CrossRef]
- Ozturk, I.; Tunçel, A.; Yurt, F.; Biyiklioglu, Z.; Ince, M.; Ocakoglu, K. Antifungal photodynamic activities of phthalocyanine derivatives on Candida albicans. Photodiagnosis Photodyn. Ther. 2020, 30, 101715. [Google Scholar] [CrossRef]
- Meerovich, G.A.; Akhlyustina, E.V.; Tiganova, I.G.; Makarova, E.A.; Tolordava, E.R.; Zhizhimova, Y.S.; Philipova, N.I.; Romanishkin, I.D.; Lukyanets, E.A.; Romanova, Y.M.; et al. Photodynamic inactivation of Pseudomonas aeruginosa bacterial biofilms using photosensitizers based on octacationic derivatives of phthalocyanines and bacteriochlorins. J. Phys. Conf. Ser. 2020, 1439. [Google Scholar] [CrossRef]
- Oliveira, H.; Correia, P.; Pereira, A.R.; Araújo, P.; Mateus, N.; de Freitas, V.; Oliveira, J.; Fernandes, I. Exploring the applications of the photoprotective properties of anthocyanins in biological systems. Int. J. Mol. Sci. 2020, 21, 7464. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Wang, M.; Huang, Y.Y.; El-Hussein, A.; Wolf, L.M.; Chiang, L.Y.; Hamblin, M.R. Progressive cationic functionalization of chlorin derivatives for antimicrobial photodynamic inactivation and related vancomycin conjugates. Photochem. Photobiol. Sci. 2018, 17, 638–651. [Google Scholar] [CrossRef]
- Vassena, C.; Fenu, S.; Giuliani, F.; Fantetti, L.; Roncucci, G.; Simonutti, G.; Romanò, C.L.; De Francesco, R.; Drago, L. Photodynamic antibacterial and antibiofilm activity of RLP068/Cl against Staphylococcus aureus and Pseudomonas aeruginosa forming biofilms on prosthetic material. Int. J. Antimicrob. Agents 2014, 44, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Teófilo, M.Í.S.; de Carvalho Russi, T.M.A.Z.; de Barros Silva, P.G.; Balhaddad, A.A.; Melo, M.A.S.; Rolim, J.P.M.L. The Impact of Photosensitizers Selection on Bactericidal Efficacy Of PDT against Cariogenic Biofilms: A Systematic Review and Meta-Analysis. Photodiagnosis Photodyn. Ther. 2020, 33, 102046. [Google Scholar] [CrossRef]
- Wang, Q.; Zhang, D.; Feng, J.; Sun, T.; Li, C.; Xie, X.; Shi, Q. Enhanced photodynamic inactivation for Gram-negative bacteria by branched polyethylenimine-containing nanoparticles under visible light irradiation. J. Colloid Interface Sci. 2021, 584, 539–550. [Google Scholar] [CrossRef] [PubMed]
- Ghai, I.; Ghai, S. Understanding antibiotic resistance via outer membrane permeability. Infect. Drug Resist. 2018, 11, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Klausen, M.; Ucuncu, M.; Bradley, M. Design of Photosensitizing Agents for Targeted Antimicrobial Photodynamic Therapy. Molecules 2020, 25, 5239. [Google Scholar] [CrossRef] [PubMed]
- Azizullah, A.; Murad, W. Chlorophyll derivatives for pest and disease control: Are they safe? Environ. Impact Assess. Rev. 2015, 50, 156–157. [Google Scholar] [CrossRef]
- Jia, H.R.; Zhu, Y.X.; Chen, Z.; Wu, F.G. Cholesterol-Assisted Bacterial Cell Surface Engineering for Photodynamic Inactivation of Gram-Positive and Gram-Negative Bacteria. ACS Appl. Mater. Interfaces 2017, 9, 15943–15951. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Lee, S.; Yoon, J. Supramolecular photosensitizers rejuvenate photodynamic therapy. Chem. Soc. Rev. 2018, 47, 1174–1188. [Google Scholar] [CrossRef] [PubMed]
- Ferro, S.; Jori, G.; Sortino, S.; Stancanelli, R.; Nikolov, P.; Tognon, G.; Ricchelli, F.; Mazzaglia, A. Inclusion of 5-[4-(1-dodecanoylpyridinium)]-10,15,20-triphenylporphine in supramolecular aggregates of cationic amphiphilic cyclodextrins: Physicochemical characterization of the complexes and strengthening of the antimicrobial photosensitizing activity. Biomacromolecules 2009, 10, 2592–2600. [Google Scholar] [CrossRef] [PubMed]
- Castriciano, M.A.; Zagami, R.; Casaletto, M.P.; Martel, B.; Trapani, M.; Romeo, A.; Villari, V.; Sciortino, M.T.; Grasso, L.; Guglielmino, S.; et al. Poly(carboxylic acid)-Cyclodextrin/Anionic Porphyrin Finished Fabrics as Photosensitizer Releasers for Antimicrobial Photodynamic Therapy. Biomacromolecules 2017, 18, 1134–1144. [Google Scholar] [CrossRef]
- Sharma, M.; Dube, A.; Majumder, S.K. Antibacterial photodynamic activity of photosensitizer-embedded alginate-pectin-carboxymethyl cellulose composite biopolymer films. Lasers Med. Sci. 2020. [Google Scholar] [CrossRef]
- Contreras, A.; Raxworthy, M.J.; Wood, S.; Schiffman, J.D.; Tronci, G. Photodynamically Active Electrospun Fibers for Antibiotic-Free Infection Control. Acs Appl. Bio Mater. 2019, 2, 4258–4270. [Google Scholar] [CrossRef]
- Chandna, S.; Thakur, N.S.; Kaur, R.; Bhaumik, J. Lignin-Bimetallic Nanoconjugate Doped pH-Responsive Hydrogels for Laser-Assisted Antimicrobial Photodynamic Therapy. Biomacromolecules 2020, 21, 3216–3230. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ma, K.; Jiao, T.; Xing, R.; Shen, G.; Yan, X. Water-insoluble photosensitizer nanocolloids stabilized by supramolecular interfacial assembly towards photodynamic therapy. Sci. Rep. 2017, 7, 1–8. [Google Scholar] [CrossRef]
- De Freitas, L.M.; Lorenzón, E.N.; Santos-Filho, N.A.; Zago, L.H.D.P.; Uliana, M.P.; De Oliveira, K.T.; Cilli, E.M.; Fontana, C.R. Antimicrobial Photodynamic therapy enhanced by the peptide aurein. Sci. Rep. 2018, 8, 1–15. [Google Scholar] [CrossRef]
- Hilgers, F.; Bitzenhofer, N.L.; Ackermann, Y.; Burmeister, A.; Grünberger, A.; Jaeger, K.E.; Drepper, T. Genetically encoded photosensitizers as light-triggered antimicrobial agents. Int. J. Mol. Sci. 2019, 20, 4608. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-González, R.; White, J.H.; Agut, M.; Nonell, S.; Flors, C. A genetically-encoded photosensitiser demonstrates killing of bacteria by purely endogenous singlet oxygen. Photochem. Photobiol. Sci. 2012, 11, 1411–1413. [Google Scholar] [CrossRef]
- Hally, C.; Delcanale, P.; Nonell, S.; Viappiani, C.; Abbruzzetti, S. Photosensitizing proteins for antibacterial photodynamic inactivation. Transl. Biophotonics 2020, 2, e201900031. [Google Scholar] [CrossRef]
- Torra, J.; Burgos-Caminal, A.; Endres, S.; Wingen, M.; Drepper, T.; Gensch, T.; Ruiz-González, R.; Nonell, S. Singlet oxygen photosensitisation by the fluorescent protein Pp2FbFP L30M, a novel derivative of Pseudomonas putida flavin-binding Pp2FbFP. Photochem. Photobiol. Sci. 2015, 14, 280–287. [Google Scholar] [CrossRef] [PubMed]
- de Melo, W.d.C.M.A.; Lee, A.N.; Perussi, J.R.; Hamblin, M.R. Electroporation enhances antimicrobial photodynamic therapy mediated by the hydrophobic photosensitizer, hypericin. Photodiagnosis Photodyn. Ther. 2013, 10, 647–650. [Google Scholar] [CrossRef] [PubMed]
- Winter, S.; Tortik, N.; Kubin, A.; Krammer, B.; Plaetzer, K. Back to the roots: Photodynamic inactivation of bacteria based on water-soluble curcumin bound to polyvinylpyrrolidone as a photosensitizer. Photochem. Photobiol. Sci. 2013, 12, 1795–1802. [Google Scholar] [CrossRef]
- Tennert, C.; Drews, A.M.; Walther, V.; Altenburger, M.J.; Karygianni, L.; Wrbas, K.T.; Hellwig, E.; Al-Ahmad, A. Ultrasonic activation and chemical modification of photosensitizers enhances the effects of photodynamic therapy against Enterococcus faecalis root-canal isolates. Photodiagnosis Photodyn. Ther. 2015, 12, 244–251. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Laguna, V.; Pérez-Artiaga, L.; Lampaya-Pérez, V.; García-Luque, I.; Ballesta, S.; Nonell, S.; Paz-Cristobal, M.P.; Gilaberte, Y.; Rezusta, A. Bactericidal effect of photodynamic therapy, alone or in combination with mupirocin or linezolid, on Staphylococcus aureus. Front. Microbiol. 2017, 8, 1–9. [Google Scholar] [CrossRef]
- Mesquita, M.Q.; Dias, C.J.; Gamelas, S.; Fardilha, M.; Neves, M.G.P.M.S.; Faustino, M.A.F. An insight on the role of photosensitizer nanocarriers for photodynamic therapy. An. Acad. Bras. Cienc. 2018, 90, 1101–1130. [Google Scholar] [CrossRef] [PubMed]
- Deda, D.K.; Araki, K. Nanotechnology, light and chemical action: An effective combination to kill cancer cells. J. Braz. Chem. Soc. 2015, 26, 2448–2470. [Google Scholar] [CrossRef]
- Raschpichler, M.; Agel, M.R.; Pinnapireddy, S.R.; Duse, L.; Baghdan, E.; Schäfer, J.; Bakowsky, U. In situ intravenous photodynamic therapy for the systemic eradication of blood stream infections. Photochem. Photobiol. Sci. 2019, 18, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Plenagl, N.; Seitz, B.S.; Duse, L.; Pinnapireddy, S.R.; Jedelska, J.; Brüßler, J.; Bakowsky, U. Hypericin inclusion complexes encapsulated in liposomes for antimicrobial photodynamic therapy. Int. J. Pharm. 2019, 570, 118666. [Google Scholar] [CrossRef] [PubMed]
- Pezzuoli, D.; Cozzolino, M.; Montali, C.; Brancaleon, L.; Bianchini, P.; Zantedeschi, M.; Bonardi, S.; Viappiani, C.; Abbruzzetti, S. Serum albumins are efficient delivery systems for the photosensitizer hypericin in photosensitization-based treatments against Staphylococcus aureus. Food Control. 2018, 94, 254–262. [Google Scholar] [CrossRef]
- Wang, J.; Yang, Y.; Xu, Y.; Zhao, L.; Wang, L.; Yin, Z.; Li, H.; Tan, H.; Liu, K. A dual enhanced anti-bacterial strategy based on high chlorin e6-loaded polyethyleneimine functionalized graphene. RSC Adv. 2021, 11, 739–744. [Google Scholar] [CrossRef]
- Guo, L.Y.; Yan, S.Z.; Tao, X.; Yang, Q.; Li, Q.; Wang, T.S.; Yu, S.Q.; Chen, S.L. Evaluation of hypocrellin A-loaded lipase sensitive polymer micelles for intervening methicillin-resistant Staphylococcus Aureus antibiotic-resistant bacterial infection. Mater. Sci. Eng. C 2020, 106, 110230. [Google Scholar] [CrossRef] [PubMed]
- Mirzahosseinipour, M.; Khorsandi, K.; Hosseinzadeh, R.; Ghazaeian, M.; Shahidi, F.K. Antimicrobial photodynamic and wound healing activity of curcumin encapsulated in silica nanoparticles. Photodiagnosis Photodyn. Ther. 2020, 29, 101639. [Google Scholar] [CrossRef] [PubMed]
- Oda, D.F.; Duarte, M.A.H.; Andrade, F.B.; Moriyama, L.T.; Bagnato, V.S.; de Moraes, I.G. Antimicrobial action of photodynamic therapy in root canals using LED curing light, curcumin and carbopol gel. Int. Endod. J. 2019, 52, 1010–1019. [Google Scholar] [CrossRef]
- Pourhajibagher, M.; Plotino, G.; Chiniforush, N.; Bahador, A. Dual wavelength irradiation antimicrobial photodynamic therapy using indocyanine green and metformin doped with nano-curcumin as an efficient adjunctive endodontic treatment modality. Photodiagnosis Photodyn. Ther. 2020, 29, 101628. [Google Scholar] [CrossRef] [PubMed]
- Vögeling, H.; Plenagl, N.; Seitz, B.S.; Pinnapireddy, S.R.; Dayyoub, E.; Jedelska, J.; Brüßler, J.; Bakowsky, U. Synergistic effects of ultrasound and photodynamic therapy leading to biofilm eradication on polyurethane catheter surfaces modified with hypericin nanoformulations. Mater. Sci. Eng. C 2019, 103, 109749. [Google Scholar] [CrossRef] [PubMed]
- Urrutia, M.N.; Sosa, M.J.; Pissinis, D.E.; Cánneva, A.; Miñán, A.G.; Vignoni, M.; Calvo, A.; Thomas, A.H.; Schilardi, P.L. Immobilization of alkyl-pterin photosensitizer on silicon surfaces through in situ SN2 reaction as suitable approach for photodynamic inactivation of Staphylococcus aureus. Colloids Surf. B Biointerfaces 2020. [Google Scholar] [CrossRef]
- Costley, D.; Nesbitt, H.; Ternan, N.; Dooley, J.; Huang, Y.Y.; Hamblin, M.R.; McHale, A.P.; Callan, J.F. Sonodynamic inactivation of Gram-positive and Gram-negative bacteria using a Rose Bengal–antimicrobial peptide conjugate. Int. J. Antimicrob. Agents 2017, 49, 31–36. [Google Scholar] [CrossRef]
- Alves, F.; Pavarina, A.C.; Mima, E.G.d.O.; McHale, A.P.; Callan, J.F. Antimicrobial sonodynamic and photodynamic therapies against Candida albicans. Biofouling 2018, 34, 357–367. [Google Scholar] [CrossRef] [PubMed]
- Alves, F.; Mayumi Inada, N.; Salvador Bagnato, V.; Kurachi, C. Sonophotodynamic Therapy for the inactivation of Staphylococcus aureus biofilm. In Proceedings of the 17th International Photodynamic Association World Congress, Cambridge, MA, USA, 29 June 2019. [Google Scholar] [CrossRef]
| Chemical Class | Name | Wavelength (nm) | Concentrations of PS | Exposure Time (min) | Light Intensity (W/m2) | Target Organisms | Aim | Reference |
|---|---|---|---|---|---|---|---|---|
| Curcuminoids | Curcumin | 430 | 814 µM | 1–5 | 107 | L. monocytogenes, Salmonella sp. | Poultry | [78] |
| 455–660 | 0–5 µM | 0–60 | 18 | L. monocytogenes | Food-related diseases | [79] | ||
| 450–470 | 5–40 µM | 0.34–1 | 12 | P. gingivalis, A. actinomycetemcomitans | Periodontitis | [82] | ||
| 455–460 | 1.0 µM | 5–60 | 38 | V. parahaemolyticus | Seafood pathogens | [80] | ||
| 450 | 6.1 mM | 4 | 400 | S. pyogenes | Pharyngotonsillitis | [84] | ||
| 420 | 300 µM | 5–25 | 0.3–0.6 | E. coli | Fresh-cut food | [87] | ||
| 470 | 30 µM | 1.5 | 1327 | Pseudomonas sp. | Inactivation of spoilage organism | [81] | ||
| 420 | 0.5–50 µM | 5–30 | 2980 | E. coli | Storage quality of fresh-cut apples | [86] | ||
| 405 | 6.75 mM | 0.22–044 | 3846 | P. aeruginosa | Biofilm inhibition | [35] | ||
| 450 | 1.5% | 12.5 | 800 | Staphylococcus sp. | Rat model of wound healing | [83] | ||
| Blue light | 25–200 µM | 30 | 30 | A. baumannii | Multidrug resistance | [77] | ||
| 450 | 100 µg/mL | 3.4–8.4 | 1500 | S. aureus | Multidrug resistance | [88] | ||
| 470 | 0.5 µM | N.A. | N.A. | E. coli | Shelf life of fruits | [90] | ||
| 450 | 4.1 mM | 5 | 670 | E. faecalis, C. albicans | Endodontic treatment | [89] | ||
| 460 | 10–50% | 120 | 220 | Mosquito larvae | Mosquito larva control | [91] | ||
| Alkaloids | Pterin-6-carboxylic acid | 350–750 | 100 µM | 80 | 31 | K. pneumoniae | Multidrug resistance | [85] |
| Perylenequinones | Hypericin | 660 | 5–15 µg/mL | 0.5–0.84 | 30,180 | C. acnes | Acne vulgaris | [93] |
| 590 | 36 µM | 68 | 164 | E. coli | Food-borne pathogens | [94] | ||
| 590 | 10 µM | 60 | 150 | P. aeruginosa | Pathogen control in a worm model | [36] | ||
| Hypocrellin | 400–780 | 1.0 µg/mL | 30 | 1590 | C. albicans | Antifungal effect | [95] | |
| Anthraquinones | Parietin | 420 | 0.125–250 µg/mL | 30 | 1000 | S. aureus, S. epidermidis, E. coli, P. aeruginosa | Local infections | [96] |
| Anthraquinone | 410 | 2.5–40 µM | 12 | 37 | L. amazonensis | Cutaneous leishmaniasis | [50] | |
| Aloe emodin | 460 | 300–500 µM | 7.2 | 0.3–0.6 | S. aureus, E. coli | Food-related diseases | [97] | |
| 435 | 0.5–100 µM | 10–40 | 800 | A. baumannii | Multidrug resistant A. baumannii | [98] | ||
| 400–780 | 5–100 µM | 20 | 800 | C. albicans | Drug-resistant C. albicans | [99] | ||
| Flavins | Riboflavin | 460 | 100 µM | 90 | 150–580 | L. monocytogenes | Listeriosis | [100] |
| Chlorin-type compounds | Chlorophyllin sodium salt | 405 | 15 mM | 10–50 | 96 | F. oxysporum, E. coli, T. aestivum | Microbial control of contaminated wheat sprouts and seeds | [101] |
| 395 | 1–100 µM | 30 | 148 | R. fascians, E. amylovora, X. axonopodis | Plant pathogens and diseases | [102] | ||
| Porphyrin precursor | 5-aminolevulinic acid | 635 | 50–300 mM | 10–30 | 600 | C. parapsilosis | Trichosporonosis | [92] |
| Extract Name | Wavelength (nm) | Concentration of PS | Exposure Time (min) | Light Intensity (W/m2) | Target Organisms | Aim | Reference |
|---|---|---|---|---|---|---|---|
| B. orellana extract | 395−480 | 20% w/v | 0.34 | 1530 | Clinical trial | Halitosis treatment | [111] |
| 20% spray | 0.34 | 1530 | Biofilms of Gram-negative bacteria | Halitosis treatment | [122] | ||
| C. longa extract | 420–480 | 0.78 µg/mL | 1 | 2800 | A. actinomycetemcomitans | Aggressive periodontitis | [113] |
| 25 µg/mg gel | 2 | 12,000 | F. nucleatum, P. intermedia | Adjunct treatment | [114] | ||
| C. xanthorrhiza extract | 405 | 10–104 ng/mL | 5 | 845 | S. mutans | Dental caries | [115] |
| G. blepharophylla extract | 660 | 50% v/v | 5 | 920 | S. aureus, S. epidermidis, E. coli, C. albicans, C. dubliniensis | Antibacterial and antifungal effect | [123] |
| H. sabdariffa extract | 420 | 0.0625–1 mg/mL | 2–20 | 100 | E. coli, B. subtilis | Photodynamic sanitation of foods | [119] |
| I. truxillensis extract | 660 | 12.5 mg/mL | 5 | 920 | S. aureus, S. epidermidis, E. coli, P. vulgaris | Multidrug-resistant microbial infections | [110] |
| Mixed essential oils | 660 | N.A. | 1 | 400 | Natural saliva S. aureus, E. coli | Oral cavity microbial films | [112] |
| P. obscurum extract | 315–400 | 0.98–1.95 µg/mL | 60 | 34 | 25 clinical strains of Candida sp. | Oropharyngeal candidiasis | [120] |
| T. minuta extract | 315–400 | 50 µg/mL | 60 | 34 | C. albicans, C. krusei, C. parapsilosis, C. tropicalis, C. glabrata, T. rubrum, T. interdigitale | Inhibition of virulence factors for Candida sp. | [118] |
| E. microcorys leaf extract | tungsten filament lamps | 0.05–2% | 0–1440 | 15 | E. coli | Bacterial inhibition | [121] |
| H. perforatum extract (St. John’s wort extract) | 570–1400 | 32 mg/mL | 5 | 2000 | 31 different cultivable species | Oral biofilms | [116] |
| sunlight | 5 g/L | 90 | 1000 | Bacteriophage MS2 | Human enteric virus | [117] | |
| T. wilfordii extract and fraction | 660 | 20 µg/mL | 10–30 | 120,600 | S. aureus, MRSA, S. epidermidis, S. pyogenes, C. albicans | Skin pathogens | [56] |
| Chemical Class | Abbreviation | Wavelength (nm) | Concentration of PS | Exposure Time (min) | Light Intensity (W/m2) | Target Organisms | Aim | Reference |
|---|---|---|---|---|---|---|---|---|
| Perylenequinone derivatives | Hy-g | 590 | 0.2 mg/mL | 6 | 950 | N.A. | Periodontal disease | [132] |
| Phenalenone derivatives | PNPPh3 + | 463 | 0.5–50 µM | 3.63–12 | 167 | S. aureus, E. faecalis, E. coli | Selective killing | [133] |
| Triphenylphosphonium derivatives | Perylene derivatives | 463 | 0.5–50 µM | 3.63–12 | 167 | S. aureus, E. faecalis, E. coli | Selective killing | [133] |
| Porphyrin derivatives | 4-PtTPyP | 400–800 | 0.91 µM | 15/30 | 1000/500 | Bovine viruses | Pharmaceutical contamination | [134] |
| H2TMeP, ZnTMeP | 380–700 | 5 µM | 0–90 | 250 | MRSA | Canine otitis | [108] | |
| TMePyP+ | 370–800 | 50 µM | 90 | 500 | M. massiliense, M. fortuitum | Mycobacteriosis | [135] | |
| PM, PE, PPN, PPL | 655 | 12.50 µM | 15 | 1667 | MRSA | Wound infections | [136] | |
| Phthalocyanine derivatives | Es-SiPc | 390–700 | 32–64 µg/mL | 10–40 | 125 | S. aureus, E. coli | Infectious diseases | [137] |
| RLP068/Cl | 600–700 | 1.2–36 µg/mL | 10 | 500 | P. aeruginosa, S. aureus, C. albicans | Localized infections | [142] | |
| ZnPc | 390–700 | 64 µg/mL | 120 | 125 | C. albicans | HIV-infected patients susceptible to fungal infections | [138] | |
| ZnPcChol8 | 680 | 250 µM | 60 | 278 | 32 clinical P. aeruginosa isolates | Infectious lesions | [139] | |
| Chlorin derivatives | Zn2+-chlorin, Zn2+-mesochlorin | 425 | 100 nM | 30 | 56 | MRSA, E. coli, C. albicans | APDT efficiency is related to the cationic charges of PSs | [141] |
| Bacteriochlorin derivatives | (3-PyEPy) 4BCBr8 | 680 | 250 µM | 60 | 278 | 32 clinical P. aeruginosa isolate | Infectious lesions | [139] |
| Natural PS | Nanostructure | Wavelength (nm) | Concentration of PS | Exposure Time (min) | Light Intensity (W/m2) | Target Organisms | Aim | Property | Reference |
|---|---|---|---|---|---|---|---|---|---|
| Curcumin | PLGA NPs | 447 | 0.2 mg/mL | 0–180 | 1000 | S. saprophyticus | Bloodstream infections | Hemocompatible APDT treatment | [167] |
| PLA dextran NPs | 455 | 260 µM | 1–20 | 360 | C. albicans | Oral candidiasis | Improved water solubility of curcumin | [55] | |
| Silica NPs | 465 | 50, 1000 µg/mL | 10 | 334 | P. aeruginosa, S. aureus | Wound healing | Decreased hydrophobicity | [172] | |
| Carbogel | 430 | 20 µM | 5 | 12,000 | E. faecalis | Root canals | Decreased the bacterial inactivation | [173] | |
| N-CUR@ICG-Met | 450, 810 | 10 µL | 5 | 2000 | E. faecalis | Adjunct endodontic treatment | Improved antibiofilm activity | [174] | |
| Hypericin | Liposomes | 589 | 0.005–0.01 µg/µL | 6 | 617 | S. saprophyticus | Liposomes as carriers | Improved APDT | [168] |
| Hyp-HPβCD-inclusion complex | 589 | 8 µg | 12 | 127 | S. saprophyticus | Catheter surfaces | Deeper penetration into the biofilm | [175] | |
| Serum albumin nanocarriers | 515 | 10 µM | 30 | 164 | S. aureus | Food packaging | Quick delivery to bacteria | [169] | |
| Hypocrellin A | mPEG-PCL micelles | 470 | 250–500 mg/L | 60 | 900 | MRSA | MRSA infections | Effective without any water solubility problem | [171] |
| Tetragonia tetragonoides extract | Nanocarrier micelle structures | 632 | 0.06–7.9 mg/mL | 20 | 34 | S. aureus | Drug resistance in pneumonia | Increased biocompatibility | [74] |
| Chlorin e6 | Graphene | 650 | 5 mg/mL | 30 | 22 | S. aureus | Drug-resistant bacteria | Improved antibacterial efficiency | [170] |
| Pterin | Silicon surfaces | 365 | N.A. | 20 | 63.7 | S. aureus | Microbial biofilms | 84.3% reduction in viable cells | [176] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Polat, E.; Kang, K. Natural Photosensitizers in Antimicrobial Photodynamic Therapy. Biomedicines 2021, 9, 584. https://doi.org/10.3390/biomedicines9060584
Polat E, Kang K. Natural Photosensitizers in Antimicrobial Photodynamic Therapy. Biomedicines. 2021; 9(6):584. https://doi.org/10.3390/biomedicines9060584
Chicago/Turabian StylePolat, Ece, and Kyungsu Kang. 2021. "Natural Photosensitizers in Antimicrobial Photodynamic Therapy" Biomedicines 9, no. 6: 584. https://doi.org/10.3390/biomedicines9060584
APA StylePolat, E., & Kang, K. (2021). Natural Photosensitizers in Antimicrobial Photodynamic Therapy. Biomedicines, 9(6), 584. https://doi.org/10.3390/biomedicines9060584







