Small Molecules in the Venom of the Scorpion Hormurus waigiensis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Scorpion Collection
2.2. Venom Extraction and Purification
2.3. Liquid Chromatography/Mass Spectrometry (LC/MS)
2.4. Mass Spectrometry and NMR Analysis
3. Results
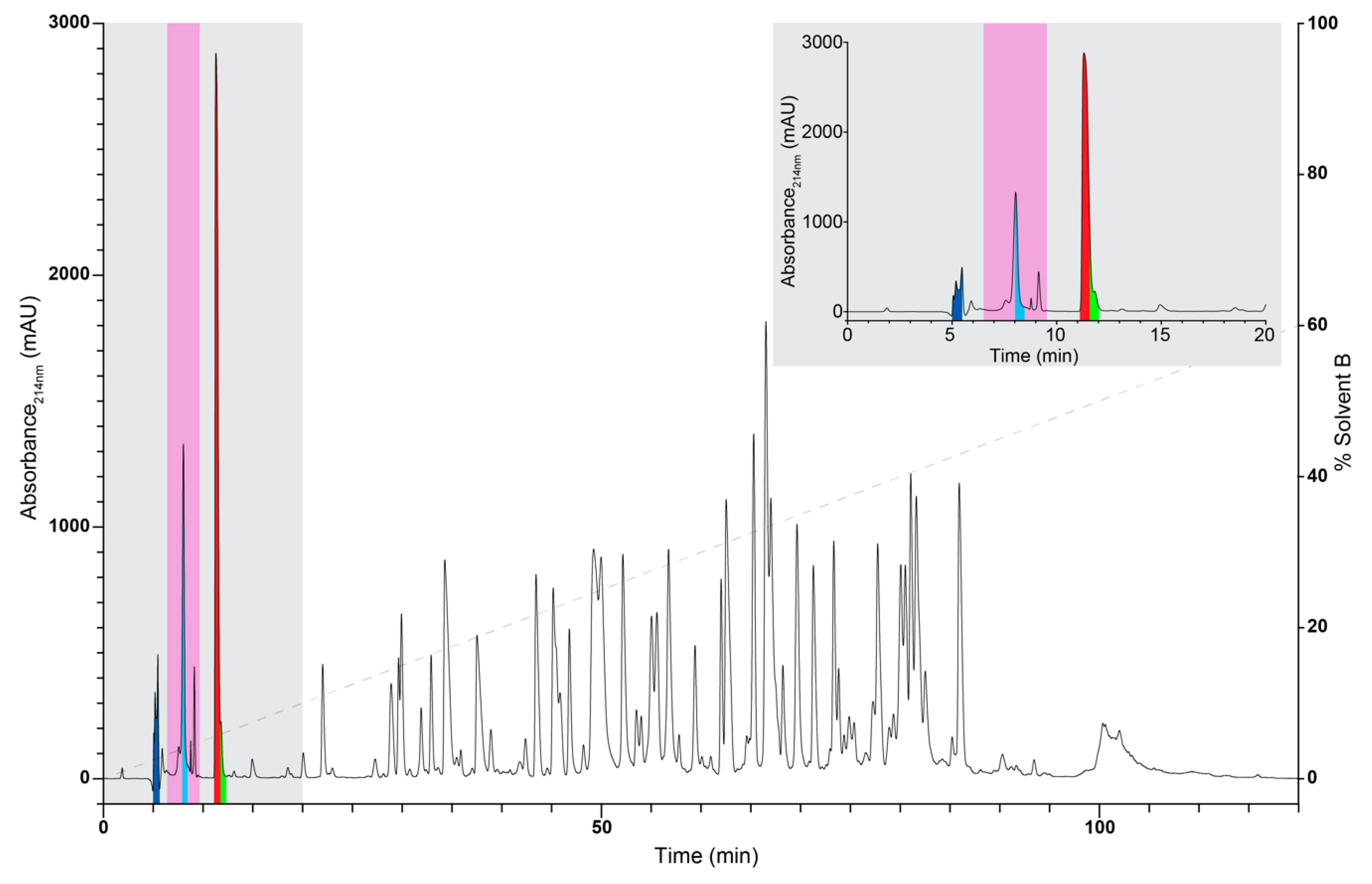
3.1. Venom Collection and Fractionation
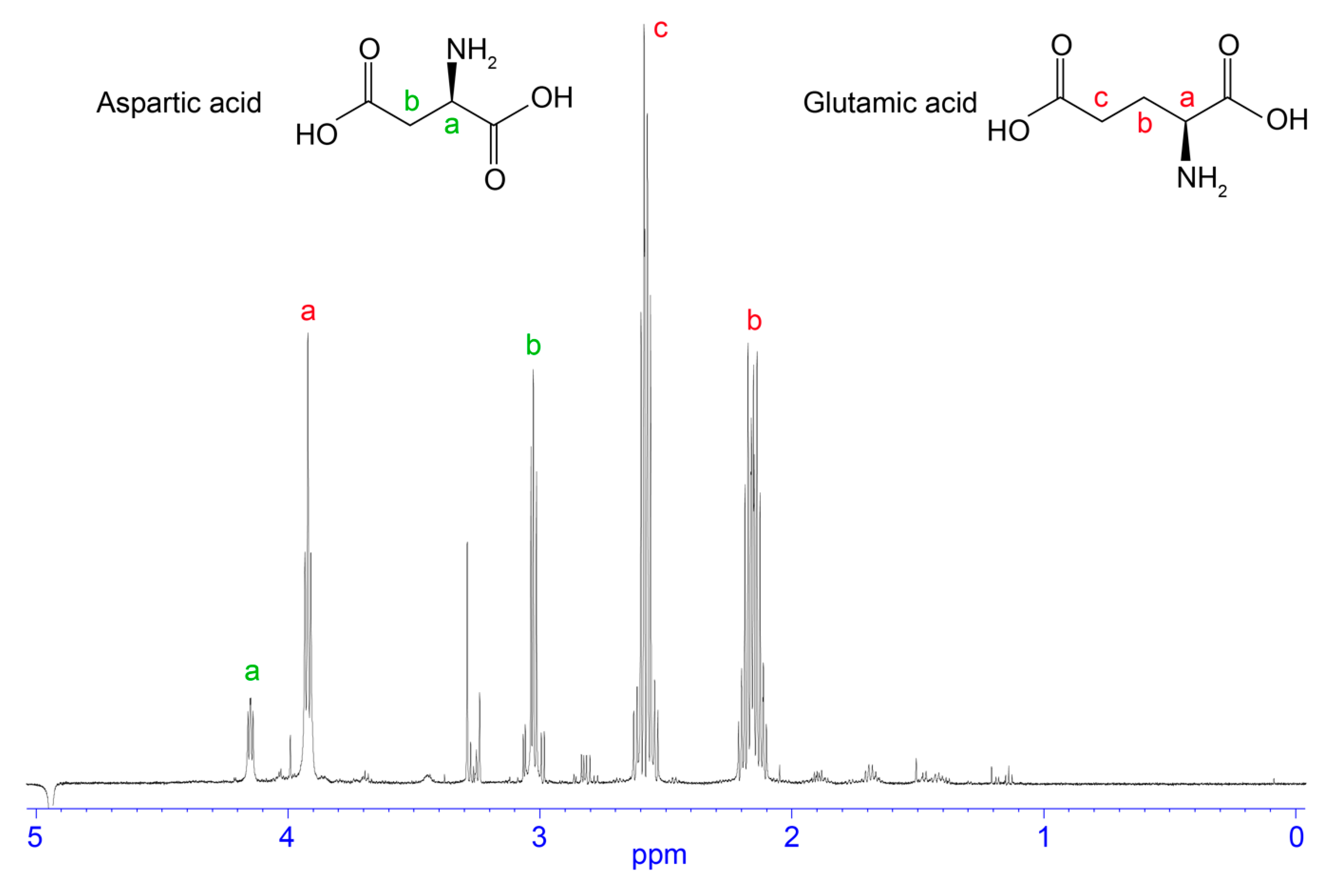
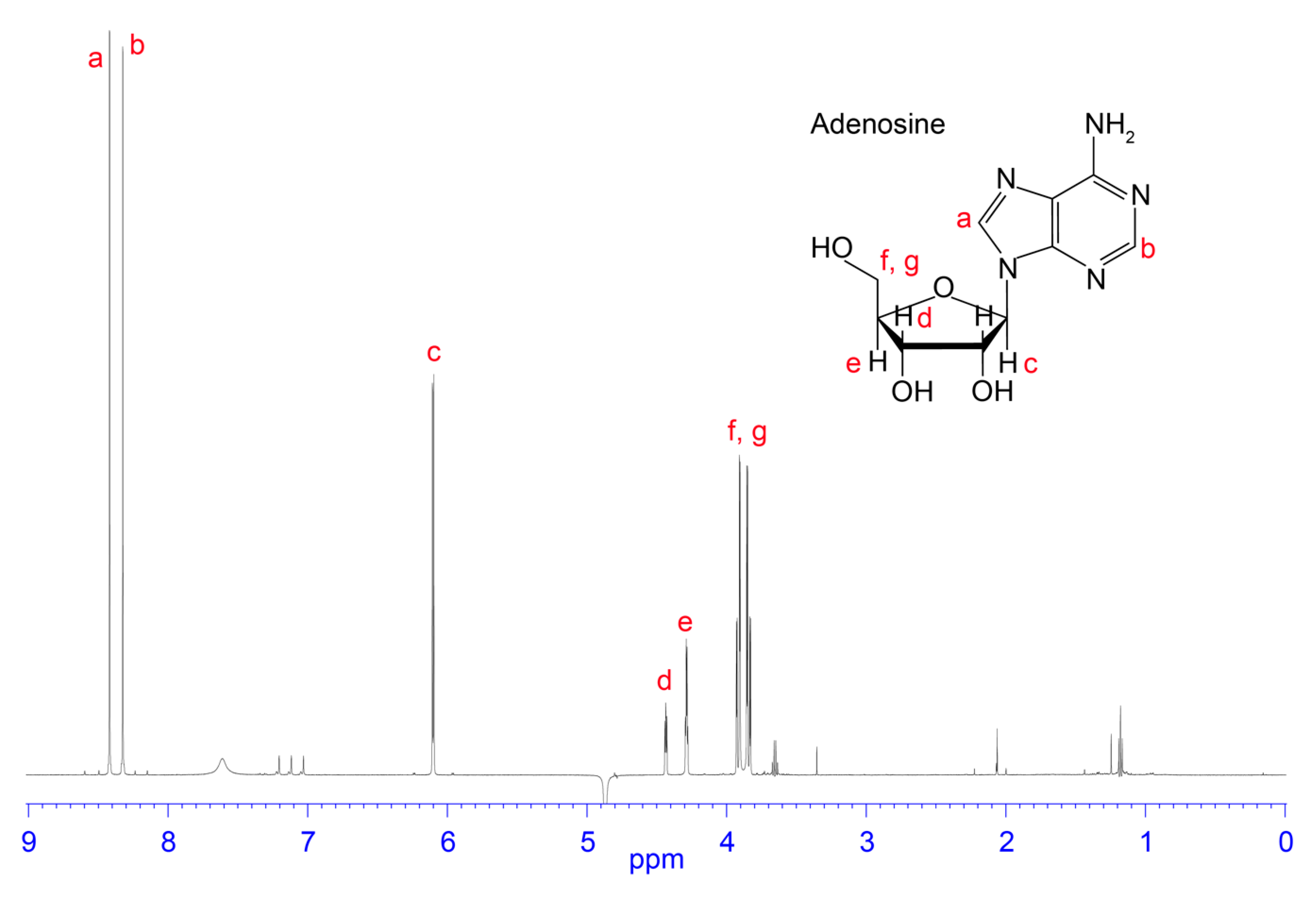
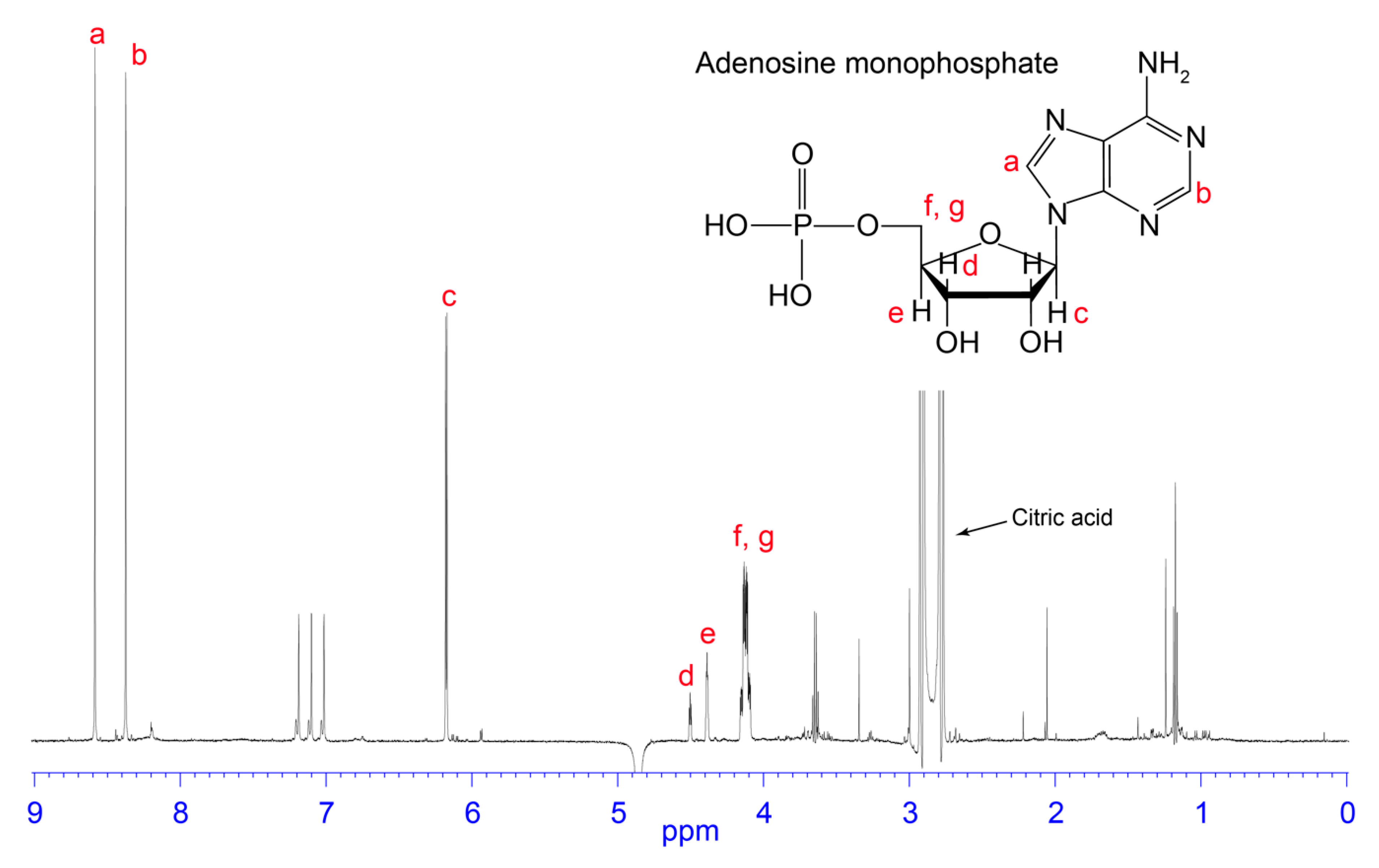
3.2. NMR Analysis of RP-HPLC Fractions
3.3. High-Resolution Mass Spectrometry
3.4. Adenosine Quantitation by LC/MS
4. Discussion
4.1. Adenosine
4.2. AMP
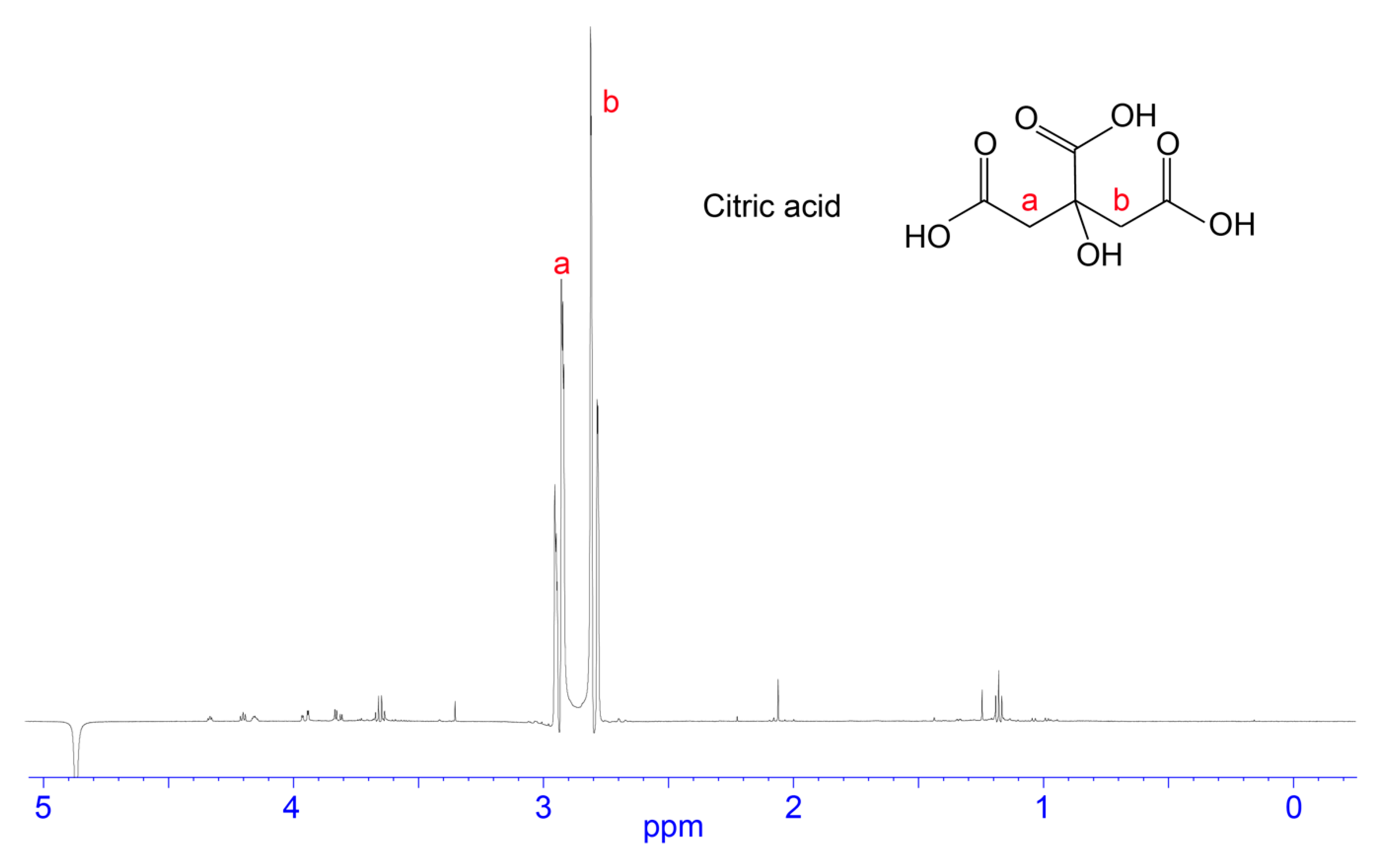
4.3. Citric Acid/Citrate
4.4. Free Amino Acids
4.5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chippaux, J.-P.; Goyffon, M. Epidemiology of scorpionism: A global appraisal. Acta Trop. 2008, 107, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, S.; Knerr, J.M.; Argemi, L.; Bordon, K.C.; Pucca, M.B.; Cerni, F.A.; Arantes, E.C.; Çalışkan, F.; Laustsen, A.H. Scorpion venom: Detriments and benefits. Biomedicines 2020, 8, 118. [Google Scholar] [CrossRef] [PubMed]
- King, G.F. Venoms as a platform for human drugs: Translating toxins into therapeutics. Expert Opin. Biol. Ther. 2011, 11, 1469–1484. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, E.; Gurrola, G.B.; Schwartz, E.F.; Possani, L.D. Scorpion venom components as potential candidates for drug development. Toxicon 2015, 93, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Possani, L.D.; Becerril, B.; Delepierre, M.; Tytgat, J. Scorpion toxins specific for Na+-channels. Eur. J. Biochem. 1999, 264, 287–300. [Google Scholar] [CrossRef] [Green Version]
- Smith, J.J.; Herzig, V.; King, G.F.; Alewood, P.F. The insecticidal potential of venom peptides. Cell. Mol. Life Sci. 2013, 70, 3665–3693. [Google Scholar] [CrossRef]
- Inceoglu, B.; Lango, J.; Jing, J.; Chen, L.; Doymaz, F.; Pessah, I.N.; Hammock, B.D. One scorpion, two venoms: Prevenom of Parabuthus transvaalicus acts as an alternative type of venom with distinct mechanism of action. Proc. Natl. Acad. Sci. USA 2003, 100, 922–927. [Google Scholar] [CrossRef] [Green Version]
- Aird, S.D. Ophidian envenomation strategies and the role of purines. Toxicon 2002, 40, 335–393. [Google Scholar] [CrossRef]
- Daly, N.L.; Wilson, D. Structural diversity of arthropod venom toxins. Toxicon 2018, 152, 46–56. [Google Scholar] [CrossRef]
- Villar-Briones, A.; Aird, S. Organic and peptidyl constituents of snake venoms: The picture is vastly more complex than we imagined. Toxins (Basel) 2018, 10, 392. [Google Scholar] [CrossRef] [Green Version]
- Wilson, D.; Boyle, G.; McIntyre, L.; Nolan, M.; Parsons, P.; Smith, J.; Tribolet, L.; Loukas, A.; Liddell, M.; Rash, L. The aromatic head group of spider toxin polyamines influences toxicity to cancer cells. Toxins (Basel) 2017, 9, 346. [Google Scholar] [CrossRef] [Green Version]
- Banerjee, S.; Gnanamani, E.; Lynch, S.R.; Zuñiga, F.Z.; Jiménez-Vargas, J.M.; Possani, L.D.; Zare, R.N. An alkaloid from scorpion venom: Chemical structure and synthesis. J. Nat. Prod. 2018, 81, 1899–1904. [Google Scholar] [CrossRef] [PubMed]
- Carcamo-Noriega, E.N.; Sathyamoorthi, S.; Banerjee, S.; Gnanamani, E.; Mendoza-Trujillo, M.; Mata-Espinosa, D.; Hernández-Pando, R.; Veytia-Bucheli, J.I.; Possani, L.D.; Zare, R.N. 1,4-Benzoquinone antimicrobial agents against Staphylococcus aureus and Mycobacterium tuberculosis derived from scorpion venom. Proc. Natl. Acad. Sci. USA 2019, 116, 12642–12647. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thien, T.V.; Anh, H.N.; Trang, N.T.T.; Van Trung, P.; Khoa, N.C.; Osipov, A.; Dubovskii, P.; Ivanov, I.; Arseniev, A.; Tsetlin, V. Low-molecular-weight compounds with anticoagulant activity from the scorpion Heterometrus laoticus venom. Dokl. Biochem. Biophys. 2017, 476, 316–319. [Google Scholar] [CrossRef] [PubMed]
- Tran, T.; Hoang, A.; Nguyen, T.; Phung, T.; Nguyen, K.; Osipov, A.; Ivanov, I.; Tsetlin, V.; Utkin, Y. Anticoagulant activity of low-molecular weight compounds from Heterometrus laoticus scorpion venom. Toxins (Basel) 2017, 9, 343. [Google Scholar] [CrossRef] [Green Version]
- Fenton, A.W.; West, P.R.; Odell, G.V.; Hudiburg, S.M.; Ownby, C.L.; Mills, J.N.; Scroggins, B.T.; Shannon, S.B. Arthropod venom citrate inhibits phospholipase A2. Toxicon 1995, 33, 763–770. [Google Scholar] [CrossRef]
- Francke, O.F. Conspectus Genericus Scorpionorum 1758–1985 (Arachnida: Scorpiones) updated through 2018. Zootaxa 2019, 4657, 1–56. [Google Scholar] [CrossRef]
- Arjunwadkar, A.; Reddy, S.R.R. Spermidine in the venom of the scorpion, Palamneus phipsoni. Toxicon 1983, 21, 321–325. [Google Scholar] [CrossRef]
- Adam, K.; Weiss, C. Distribution of 5-hydroxytryptamine in scorpion venoms. Nature 1959, 183, 1398–1399. [Google Scholar] [CrossRef]
- Dai, L.; Corzo, G.; Naoki, H.; Andriantsiferana, M.; Nakajima, T. Purification, structure–function analysis, and molecular characterization of novel linear peptides from scorpion Opisthacanthus madagascariensis. Biochem. Biophys. Res. Commun. 2002, 293, 1514–1522. [Google Scholar] [CrossRef]
- Luna-Ramírez, K.; Quintero-Hernandez, V.; Juárez-González, V.R.; Possani, L.D. Whole transcriptome of the venom gland from Urodacus yaschenkoi scorpion. PLoS ONE 2015, 10, e0127883. [Google Scholar] [CrossRef] [PubMed]
- Hisada, M.; Fujita, T.; Naoki, H.; Itagaki, Y.; Irie, H.; Miyashita, M.; Nakajima, T. Structures of spider toxins: Hydroxyindole-3-acetylpolyamines and a new generalized structure of type-E compounds obtained from the venom of the Joro spider, Nephila clavata. Toxicon 1998, 36, 1115–1125. [Google Scholar] [CrossRef]
- Itagaki, Y.; Fujita, T.; Naoki, H.; Yasuhara, T.; Andriantsiferana, M.; Nakajima, T. Detection of new spider toxins from a Nephilengys borbonica venom gland using on-line μ-column HPLC continuous flow (FRIT) FAB LC/MS and MS/MS. Nat. Toxins 1997, 5, 1–13. [Google Scholar] [CrossRef]
- McCormick, K.D.; Meinwald, J. Neurotoxic acylpolyamines from spider venoms. J. Chem. Ecol. 1993, 19, 2411–2451. [Google Scholar] [CrossRef]
- Palma, M.S.; Nakajima, T. A natural combinatorial chemistry strategy in acylpolyamine toxins from nephilinae orb-web spiders. Toxin Rev. 2005, 24, 209–234. [Google Scholar] [CrossRef]
- Skinner, W.S.; Dennis, P.A.; Lui, A.; Carney, R.L.; Quistad, G.B. Chemical characterization of acylpolyamine toxins from venom of a trap-door spider and two tarantulas. Toxicon 1990, 28, 541–546. [Google Scholar] [CrossRef]
- Cesar, L.M.; Mendes, M.A.; Tormena, C.F.; Marques, M.R.; De Souza, B.M.; Saidemberg, D.M.; Bittencourt, J.C.; Palma, M.S. Isolation and chemical characterization of PwTx-II: A novel alkaloid toxin from the venom of the spider Parawixia bistriata (Araneidae, Araneae). Toxicon 2005, 46, 786–796. [Google Scholar] [CrossRef]
- Marques, M.R.; Mendes, M.A.; Tormena, C.F.; Souza, B.M.; Marcondes Cesar, L.M.; Rittner, R.; Palma, M.S. Structure determination of a tetrahydro-β-carboline of arthropod origin: A novel alkaloid-toxin subclass from the web of spider Nephila clavipes. Chem. Biodivers. 2005, 2, 525–534. [Google Scholar] [CrossRef]
- Saidemberg, D.M.; Ferreira, M.A.; Takahashi, T.N.; Gomes, P.C.; Cesar-Tognoli, L.M.; da Silva-Filho, L.C.; Tormena, C.F.; da Silva, G.V.; Palma, M.S. Monoamine oxidase inhibitory activities of indolylalkaloid toxins from the venom of the colonial spider Parawixia bistriata: Functional characterization of PwTX-I. Toxicon 2009, 54, 717–724. [Google Scholar] [CrossRef]
- Chan, T.K.; Geren, C.; Howell, D.; Odell, G. Adenosine triphosphate in tarantula spider venoms and its synergistic effect with the venom toxin. Toxicon 1975, 13, 61–66. [Google Scholar] [CrossRef]
- Horni, A.; Weickmann, D.; Hesse, M. The main products of the low molecular mass fraction in the venom of the spider Latrodectus menavodi. Toxicon 2001, 39, 425–428. [Google Scholar] [CrossRef]
- Rodrigues, M.C.A.; Guizzo, R.; Gobbo-Neto, L.; Ward, R.J.; Lopes, N.P.; dos Santos, W.F. The biological activity in mammals and insects of the nucleosidic fraction from the spider Parawixia bistriata. Toxicon 2004, 43, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Savel-Niemann, A. Tarantula (Eurypelma californicum) venom, a multicomponent system. Biol. Chem. Hoppe Seyler 1989, 370, 485–498. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, F.C.; Taggi, A.E.; Gronquist, M.; Malik, R.U.; Grant, J.B.; Eisner, T.; Meinwald, J. NMR-spectroscopic screening of spider venom reveals sulfated nucleosides as major components for the brown recluse and related species. Proc. Natl. Acad. Sci. USA 2008, 105, 14283–14287. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taggi, A.E.; Meinwald, J.; Schroeder, F.C. A new approach to natural products discovery exemplified by the identification of sulfated nucleosides in spider venom. J. Am. Chem. Soc. 2004, 126, 10364–10369. [Google Scholar] [CrossRef]
- Schanbacher, F.; Lee, C.; Hall, J.; Wilson, I.; Howell, D.; Odell, G. Composition and properties of tarantula Dugesiella hentzi (Girard) venom. Toxicon 1973, 11, 21–29. [Google Scholar] [CrossRef]
- Escoubas, P.; Diochot, S.; Corzo, G. Structure and pharmacology of spider venom neurotoxins. Biochimie 2000, 82, 893–907. [Google Scholar] [CrossRef]
- Kuhn-Nentwig, L.; Stöcklin, R.; Nentwig, W. Venom composition and strategies in spiders: Is everything possible? Adv. Insect Physiol. 2011, 40, 1–86. [Google Scholar]
- Vassilevski, A.; Kozlov, S.; Grishin, E. Molecular diversity of spider venom. Biochemistry (Moscow) 2009, 74, 1505–1534. [Google Scholar] [CrossRef]
- Gomes, P.C.; de Souza, B.M.; Dias, N.B.; Cesar-Tognoli, L.M.; Silva-Filho, L.C.; Tormena, C.F.; Rittner, R.; Richardson, M.; Cordeiro, M.N.; Palma, M.S. Nigriventrine: A low molecular mass neuroactive compound from the venom of the spider Phoneutria nigriventer. Toxicon 2011, 57, 266–274. [Google Scholar] [CrossRef] [Green Version]
- Odell, G.; Fenton, A.; Ownby, C.; Doss, M.; Schmidt, J. The role of venom citrate. Toxicon 1999, 37, 407–409. [Google Scholar] [PubMed]
- Yoshioka, M.; Narai, N.; Teshima, T.; Matsumoto, T.; Wakamiya, T.; Shiba, T.; Tokoro, N.; Okauchi, T.; Kono, Y. Characterization of a new insecticide, clavamine, from the venom of a spider, Nephila clavata by use of a synthetic compound. Chem. Pharm. Bull. (Tokyo) 1992, 40, 3005–3008. [Google Scholar] [CrossRef] [Green Version]
- Aird, S.D. Taxonomic distribution and quantitative analysis of free purine and pyrimidine nucleosides in snake venoms. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2005, 140, 109–126. [Google Scholar] [CrossRef] [PubMed]
- Francis, B.; Seebart, C.; Kaiser, I.I. Citrate is an endogenous inhibitor of snake venom enzymes by metal-ion chelation. Toxicon 1992, 30, 1239–1246. [Google Scholar] [CrossRef]
- Koch, L. The taxonomy, geographic distribution and evolutionary radiation of Australo-Papuan scorpions. Rec. West. Aust. Mus. 1977, 5 Pt 2, 83–367. [Google Scholar]
- Monod, L.; Volschenk, E. Liocheles litodactylus (Scorpiones: Liochelidae): An unusual new Liocheles species from the Australian wet tropics (Queensland). Mem. Qld. Mus. 2004, 49, 675–690. [Google Scholar]
- Isbister, G.; Volschenk, E.; Seymour, J. Scorpion stings in Australia: Five definite stings and a review. Intern. Med. J. 2004, 34, 427–430. [Google Scholar] [CrossRef]
- Smith, J.J.; Hill, J.M.; Little, M.J.; Nicholson, G.M.; King, G.F.; Alewood, P.F. Unique scorpion toxin with a putative ancestral fold provides insight into evolution of the inhibitor cystine knot motif. Proc. Natl. Acad. Sci. USA 2011, 108, 10478–10483. [Google Scholar] [CrossRef] [Green Version]
- Smith, J.J.; Vetter, I.; Lewis, R.J.; Peigneur, S.; Tytgat, J.; Lam, A.; Gallant, E.M.; Beard, N.A.; Alewood, P.F.; Dulhunty, A.F. Multiple actions of φ-LITX-Lw1a on ryanodine receptors reveal a functional link between scorpion DDH and ICK toxins. Proc. Natl. Acad. Sci. USA 2013, 110, 8906–8911. [Google Scholar] [CrossRef] [Green Version]
- Santos, M.S.; Silva, C.G.; Neto, B.S.; Júnior, C.R.G.; Lopes, V.H.; Júnior, A.G.T.; Bezerra, D.A.; Luna, J.V.; Cordeiro, J.B.; Júnior, J.G. Clinical and epidemiological aspects of scorpionism in the world: A systematic review. Wilderness Environ. Med. 2016, 27, 504–518. [Google Scholar] [CrossRef] [Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Moore, S.; Smyth, W.F.; Gault, V.A.; O’Kane, E.; McClean, S. Mass spectrometric characterisation and quantitation of selected low molecular mass compounds from the venom of Haplopelma lividum (Theraphosidae). Rapid Commun. Mass Spectrom. 2009, 23, 1747–1755. [Google Scholar] [CrossRef] [PubMed]
- Konno, K.; Hisada, M.; Naoki, H.; Itagaki, Y.; Yasuhara, T.; Juliano, M.A.; Juliano, L.; Palma, M.S.; Yamane, T.; Nakajima, T. Isolation and sequence determination of peptides in the venom of the spider wasp (Cyphononyx dorsalis) guided by matrix-assisted laser desorption/ionization time of flight (MALDI-TOF) mass spectrometry. Toxicon 2001, 39, 1257–1260. [Google Scholar] [CrossRef]
- Hink, W.; Romstedt, K.; Burke, J.; Doskotch, R.; Feller, D. Inhibition of human platelet aggregation and secretion by ant venom and a compound isolated from venom. Inflammation 1989, 13, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Yimin, S.; Bingkun, X.; Songping, L.; Shunyi, L. Separation and identification of inosine in the Selenocosmia huwena spider. Chin. J. Anal. Chem. 1995, 9, 25. [Google Scholar]
- Geren, C.R.; Chan, T.K.; Howell, D.E.; Odell, G.V. Partial characterization of the low molecular weight fractions of the extract of the venom apparatus of the brown recluse spider and of its hemolymph. Toxicon 1975, 13, 233–238. [Google Scholar] [CrossRef]
- venoMS. Available online: https://www.venoms.ch/ (accessed on 30 June 2020).
- Laustsen, A.H. Toxin synergism in snake venoms. Toxin Rev. 2016, 35, 165–170. [Google Scholar] [CrossRef] [Green Version]
- Tilley, S.L.; Wagoner, V.A.; Salvatore, C.A.; Jacobson, M.A.; Koller, B.H. Adenosine and inosine increase cutaneous vasopermeability by activating A3 receptors on mast cells. J. Clin. Investig. 2000, 105, 361–367. [Google Scholar] [CrossRef] [Green Version]
- Juhasz-Nagy, A.; Aviado, D.M. Inosine as a cardiotonic agent that reverses adrenergic beta blockade. J. Pharmacol. Exp. Ther. 1977, 202, 683–695. [Google Scholar]
- Evans, E.R.J.; Northfield, T.D.; Daly, N.L.; Wilson, D.T. Venom costs and optimization in scorpions. Front. Ecol. Evol. 2019, 7, 196. [Google Scholar] [CrossRef] [Green Version]
- McCormick, S.; Polis, G.A. Arthropods that prey on vertebrates. Biol. Rev. Camb. Philos. Soc. 1982, 57, 29–58. [Google Scholar] [CrossRef]
- Burnstock, G. Purinoceptors: Ontogeny and phylogeny. Drug Dev. Res. 1996, 39, 204–242. [Google Scholar] [CrossRef]
- Gangur, A.N.; Smout, M.; Liddell, M.J.; Seymour, J.E.; Wilson, D.; Northfield, T.D. Changes in predator exposure, but not in diet, induce phenotypic plasticity in scorpion venom. Proc. R. Soc. Lond. B Biol. Sci. 2017, 284, 20171364. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gillespie, J. The biological significance of the linkages in adenosine triphosphoric acid. J. Physiol. 1934, 80, 345. [Google Scholar] [CrossRef] [PubMed]
- Horta, C.; Rezende, B.; Oliveira-Mendes, B.; Carmo, A.; Capettini, L.; Silva, J.; Gomes, M.; Chávez-Olórtegui, C.; Bravo, C.; Lemos, V. ADP is a vasodilator component from Lasiodora sp. mygalomorph spider venom. Toxicon 2013, 72, 102–112. [Google Scholar] [CrossRef]
- Nentwig, W. The species referred to as Eurypelma californicum (Theraphosidae) in more than 100 publications is likely to be Aphonopelma hentzi. J. Arachnol. 2012, 40, 128–131. [Google Scholar] [CrossRef]
- Burnstock, G. Purine and pyrimidine receptors. Cell. Mol. Life Sci. 2007, 64, 1471. [Google Scholar] [CrossRef]
- Gawlowski, D.M.; Duran, W.N. Dose-related effects of adenosine and bradykinin on microvascular permselectivity to macromolecules in the hamster cheek pouch. Circ. Res. 1986, 58, 348–355. [Google Scholar] [CrossRef] [Green Version]
- Krayem, N.; Gargouri, Y. Scorpion venom phospholipases A2: A minireview. Toxicon 2020, 184, 48–54. [Google Scholar] [CrossRef]
- Ortiz, E.; Rendón-Anaya, M.; Rego, S.C.; Schwartz, E.F.; Possani, L.D. Antarease-like Zn-metalloproteases are ubiquitous in the venom of different scorpion genera. Biochim. Biophys. Acta 2014, 1840, 1738–1746. [Google Scholar] [CrossRef]
- Lee, Y.L.; Thrupp, L.; Owens, J.; Cesario, T.; Shanbrom, E. Bactericidal activity of citrate against Gram-positive cocci. Lett. Appl. Microbiol. 2001, 33, 349–351. [Google Scholar] [CrossRef]
- Gao, B.; Xu, J.; del Carmen Rodriguez, M.; Lanz-Mendoza, H.; Hernández-Rivas, R.; Du, W.; Zhu, S. Characterization of two linear cationic antimalarial peptides in the scorpion Mesobuthus eupeus. Biochimie 2010, 92, 350–359. [Google Scholar] [CrossRef] [PubMed]
- Moerman, L.; Bosteels, S.; Noppe, W.; Willems, J.; Clynen, E.; Schoofs, L.; Thevissen, K.; Tytgat, J.; Van Eldere, J.; Van Der Walt, J. Antibacterial and antifungal properties of α-helical, cationic peptides in the venom of scorpions from southern Africa. Eur. J. Biochem. 2002, 269, 4799–4810. [Google Scholar] [CrossRef] [PubMed]
- Willems, J.; Noppe, W.; Moerman, L.; van der Walt, J.; Verdonck, F. Cationic peptides from scorpion venom can stimulate and inhibit polymorphonuclear granulocytes. Toxicon 2002, 40, 1679–1683. [Google Scholar] [CrossRef]
- Lee, S.-C.; Guan, H.-H.; Wang, C.-H.; Huang, W.-N.; Tjong, S.-C.; Chen, C.-J.; Wu, W.-G. Structural basis of citrate-dependent and heparan sulfate-mediated cell surface retention of cobra cardiotoxin A3. J. Biol. Chem. 2005, 280, 9567–9577. [Google Scholar] [CrossRef] [Green Version]
- Bieber, A. Metal and nonprotein constituents in snake venoms. In Snake Venoms; Springer: Berlin, Germany, 1979; pp. 295–306. [Google Scholar]
- Kuhn-Nentwig, L.; Schaller, J.; Nentwig, W. Biochemistry, toxicology and ecology of the venom of the spider Cupiennius salei (Ctenidae). Toxicon 2004, 43, 543–553. [Google Scholar] [CrossRef]
- Olive, M.F.; Powell, G.; McClure, E.; Gipson, C.D. Neurotransmitter Systems: Glutamate. In The Therapeutic Use of N-Acetylcysteine (NAC) in Medicine; Springer: Berlin, Germany, 2019; pp. 19–28. [Google Scholar]
- Monod, L.; Prendini, L. Evidence for Eurogondwana: The roles of dispersal, extinction and vicariance in the evolution and biogeography of Indo-Pacific Hormuridae (Scorpiones: Scorpionoidea). Cladistics 2015, 31, 71–111. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Evans, E.R.J.; McIntyre, L.; Northfield, T.D.; Daly, N.L.; Wilson, D.T. Small Molecules in the Venom of the Scorpion Hormurus waigiensis. Biomedicines 2020, 8, 259. https://doi.org/10.3390/biomedicines8080259
Evans ERJ, McIntyre L, Northfield TD, Daly NL, Wilson DT. Small Molecules in the Venom of the Scorpion Hormurus waigiensis. Biomedicines. 2020; 8(8):259. https://doi.org/10.3390/biomedicines8080259
Chicago/Turabian StyleEvans, Edward R. J., Lachlan McIntyre, Tobin D. Northfield, Norelle L. Daly, and David T. Wilson. 2020. "Small Molecules in the Venom of the Scorpion Hormurus waigiensis" Biomedicines 8, no. 8: 259. https://doi.org/10.3390/biomedicines8080259
APA StyleEvans, E. R. J., McIntyre, L., Northfield, T. D., Daly, N. L., & Wilson, D. T. (2020). Small Molecules in the Venom of the Scorpion Hormurus waigiensis. Biomedicines, 8(8), 259. https://doi.org/10.3390/biomedicines8080259




