Use of Platelet Rich Fibrin (PRF)-Based Autologous Membranes for Tooth Extraction in Patients under Bisphosphonate Therapy: A Case Report
Abstract
1. Introduction
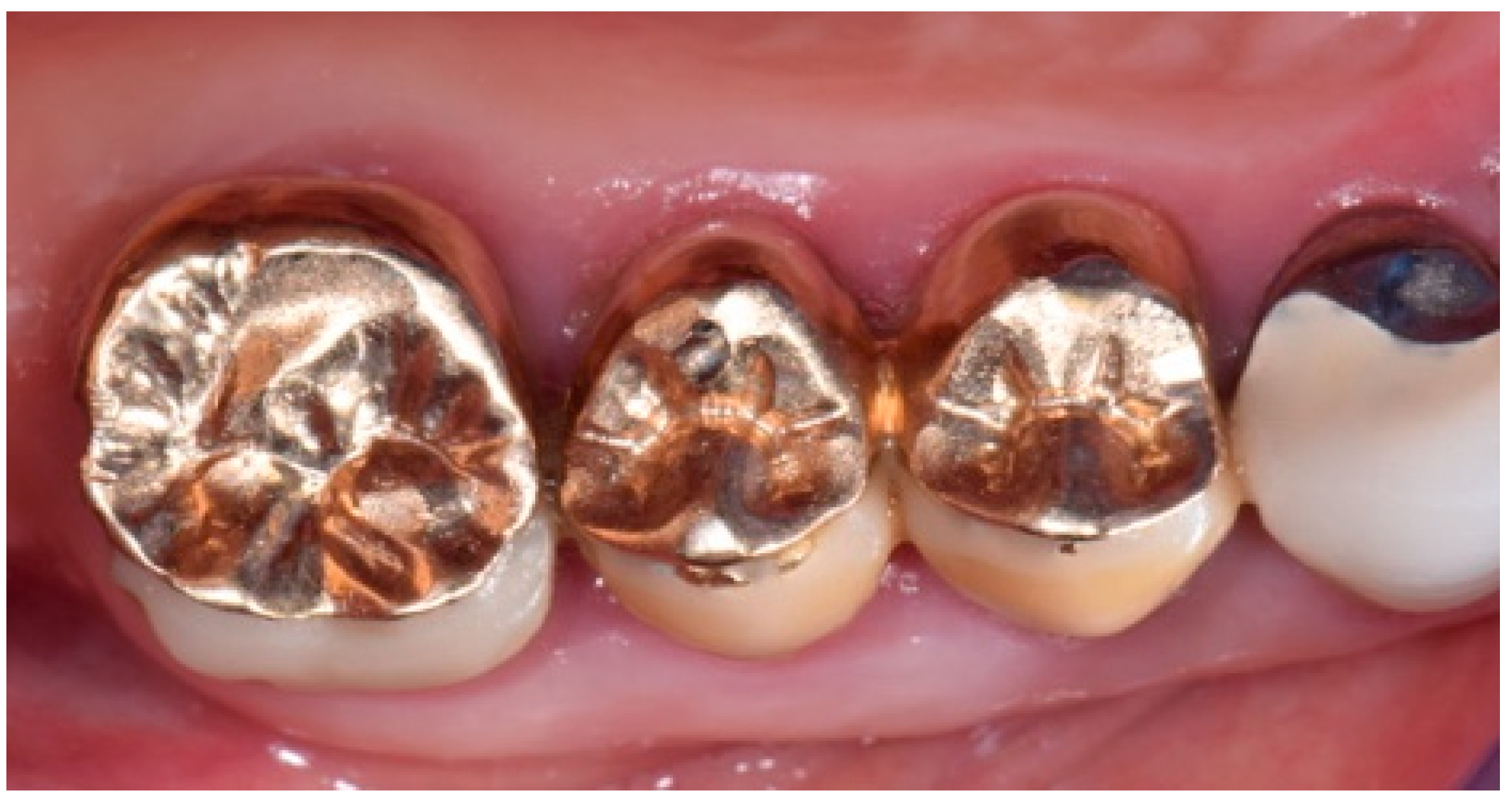
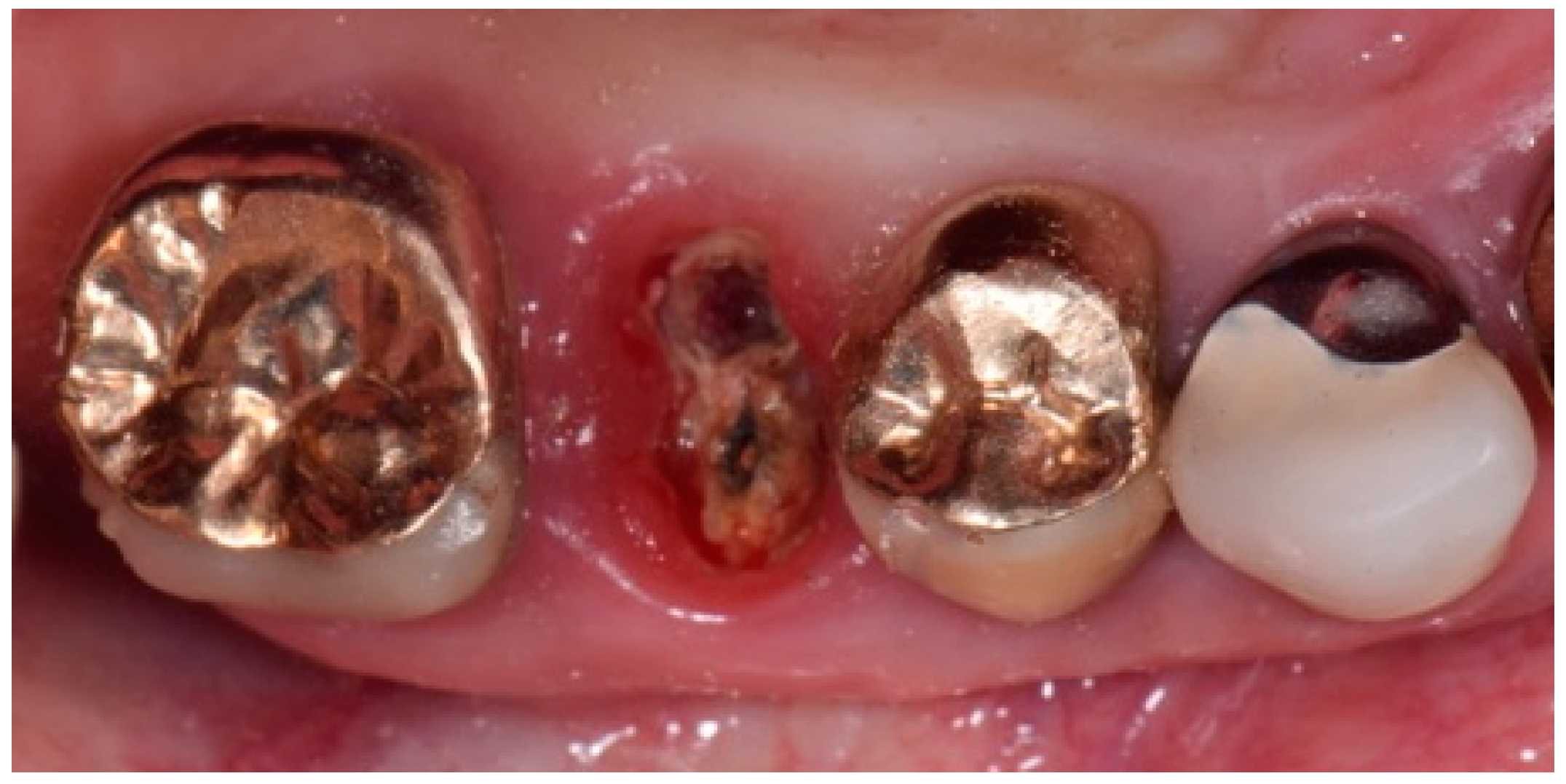
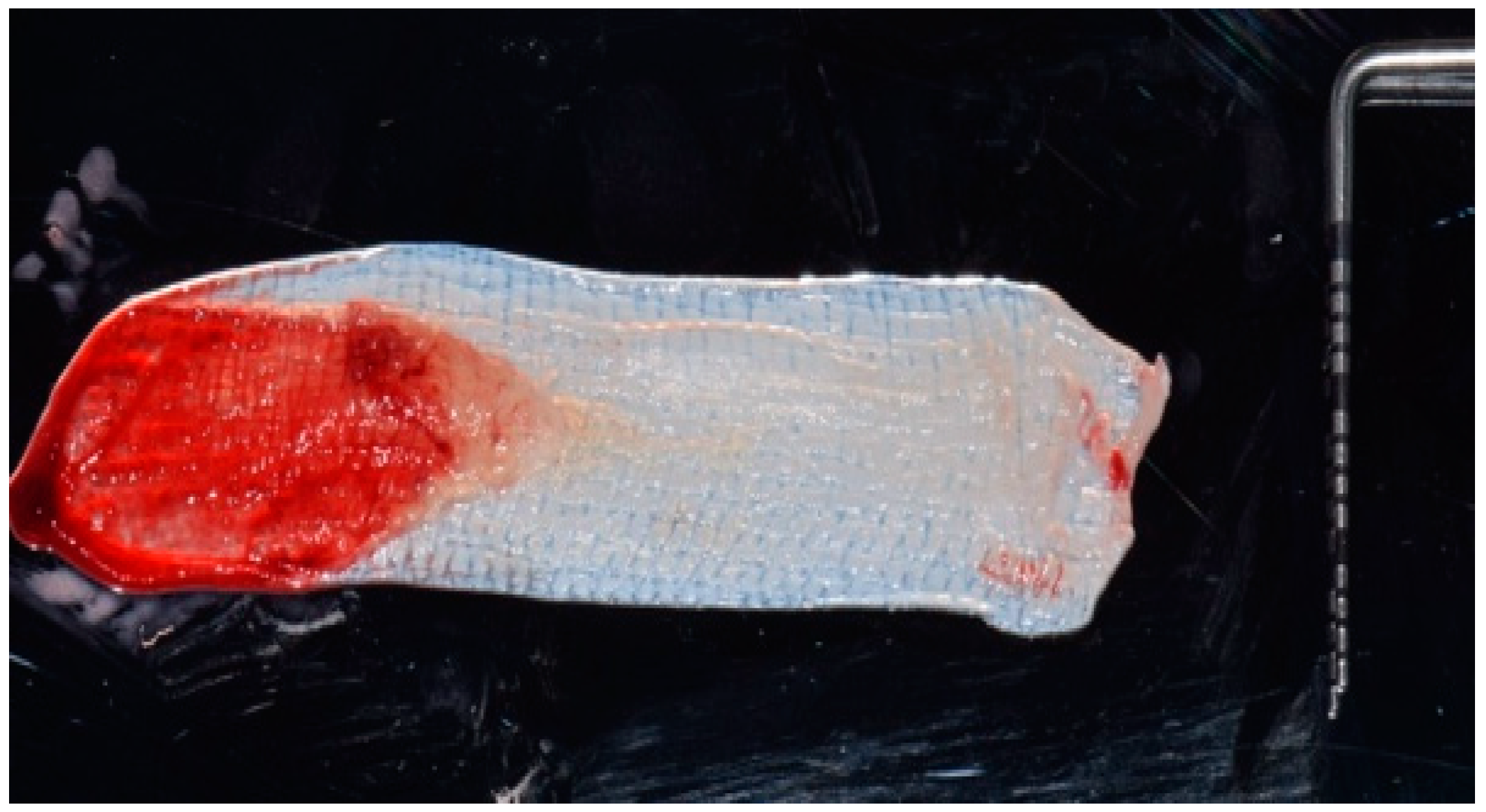
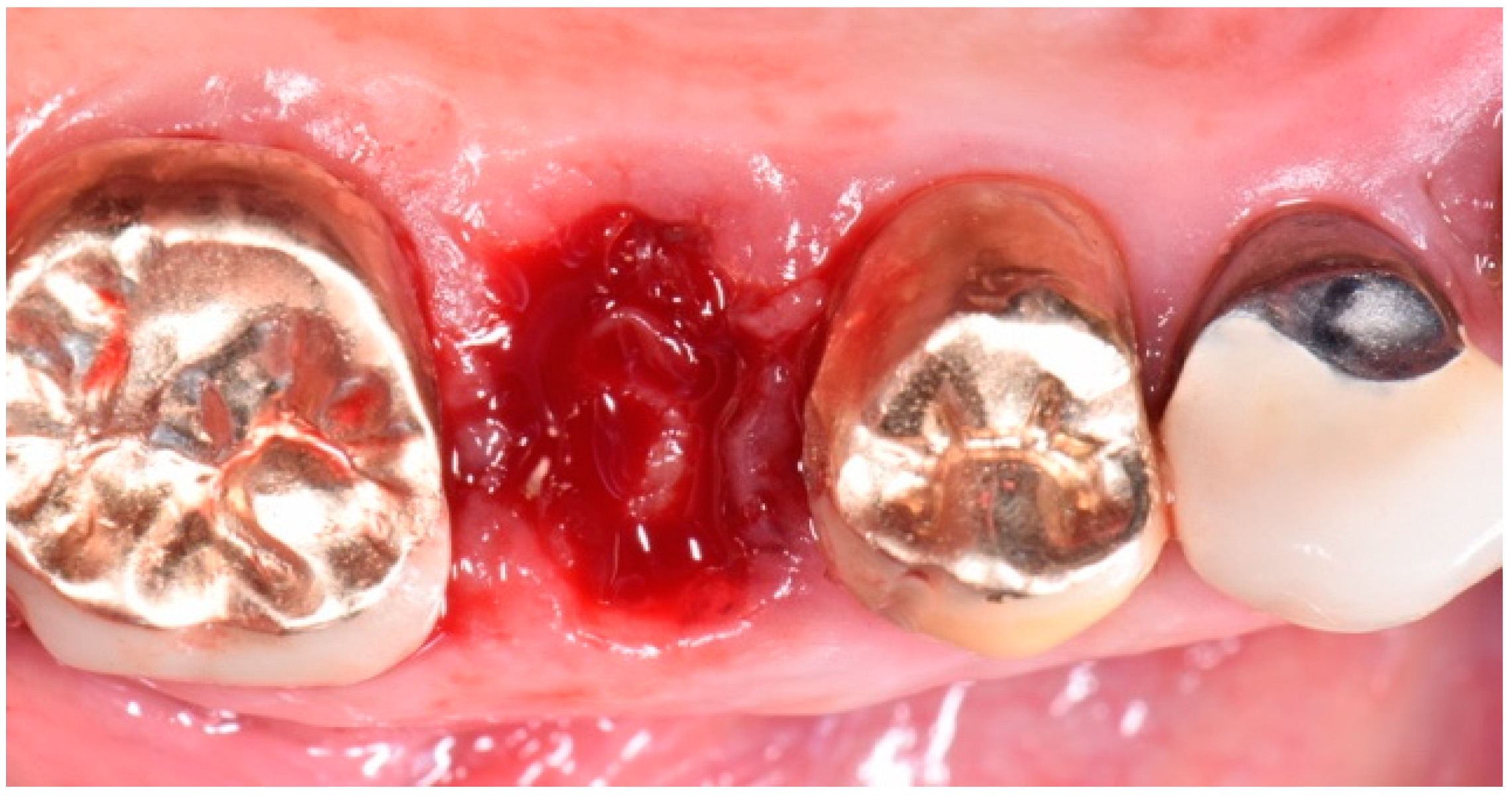
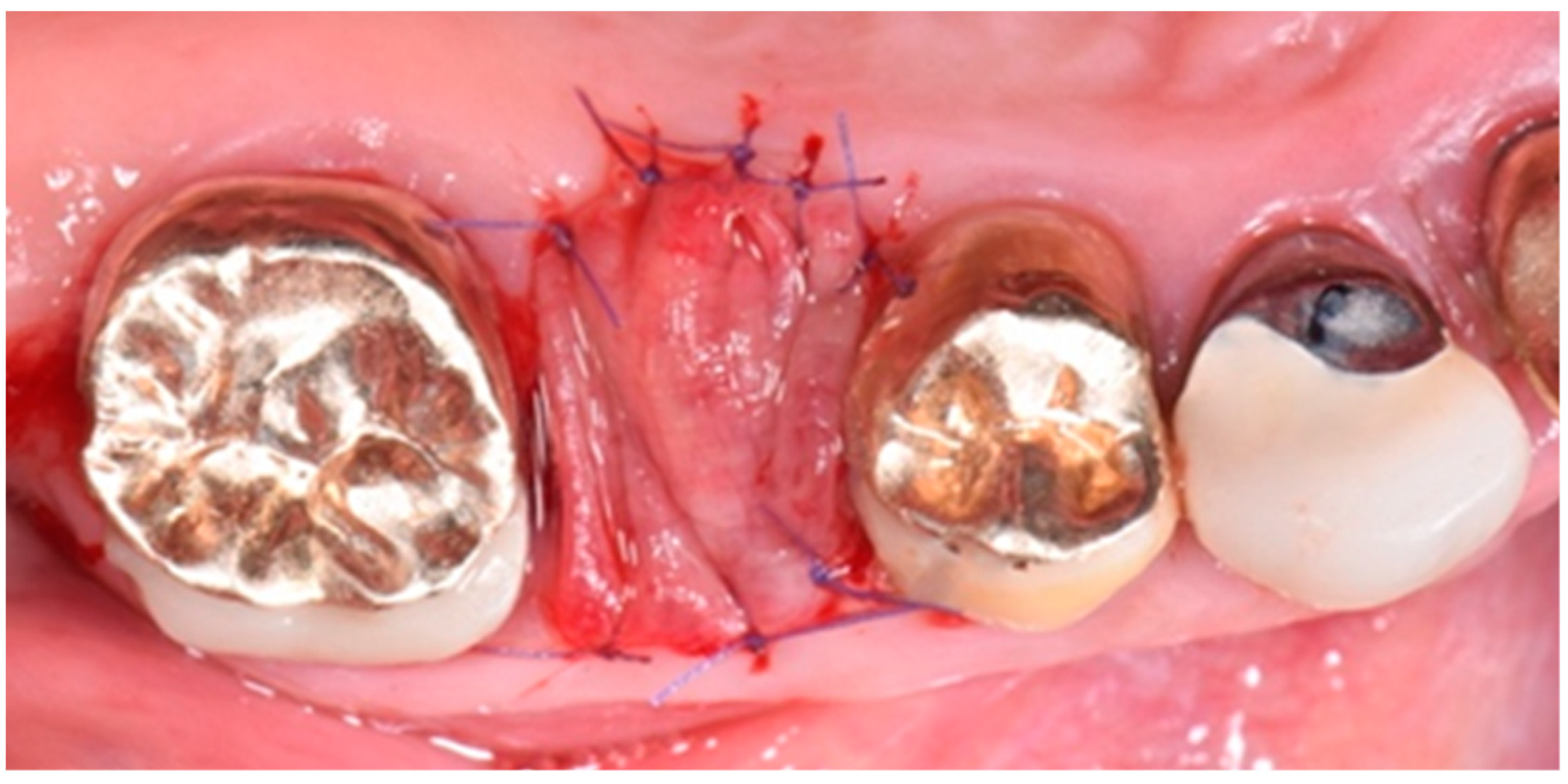
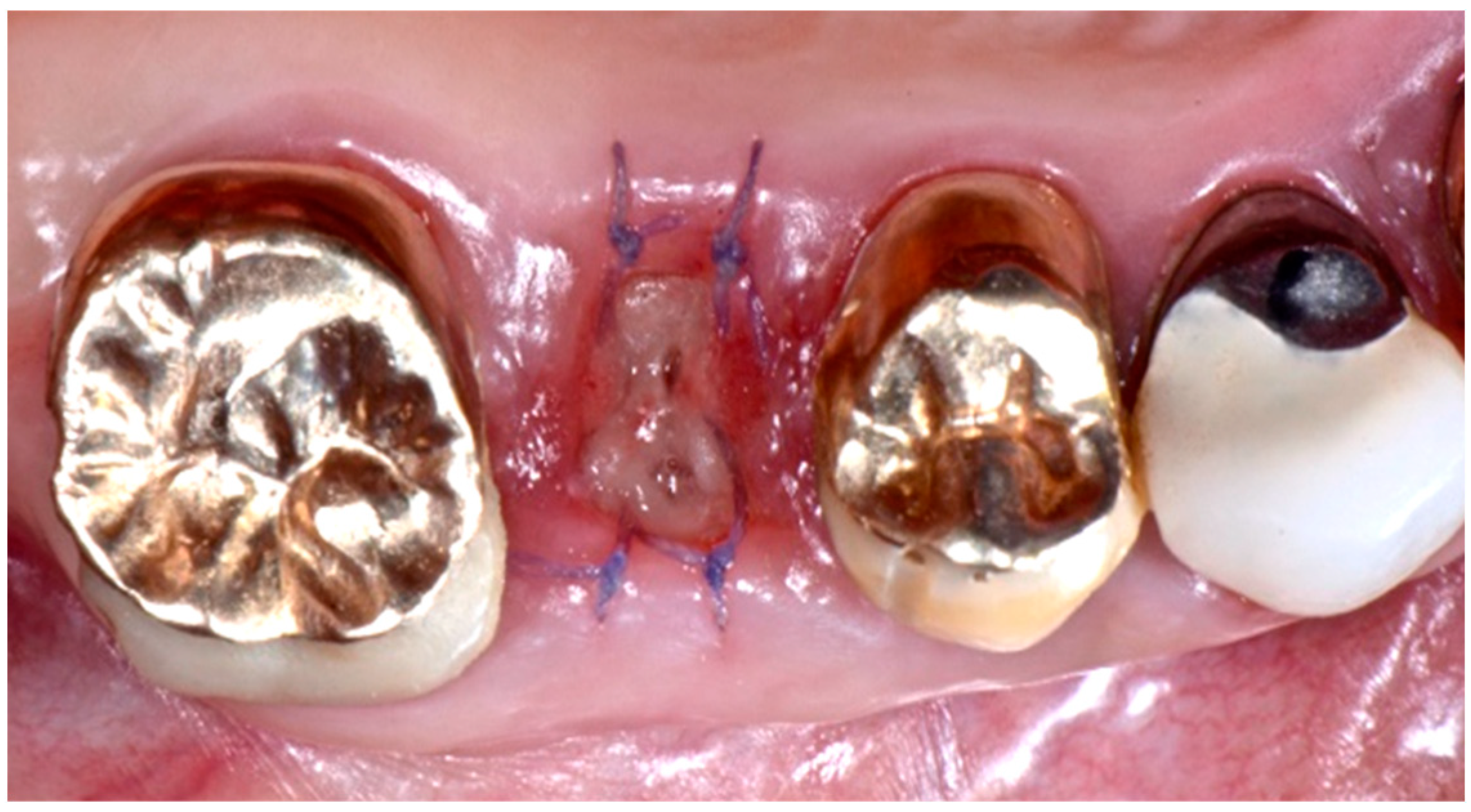
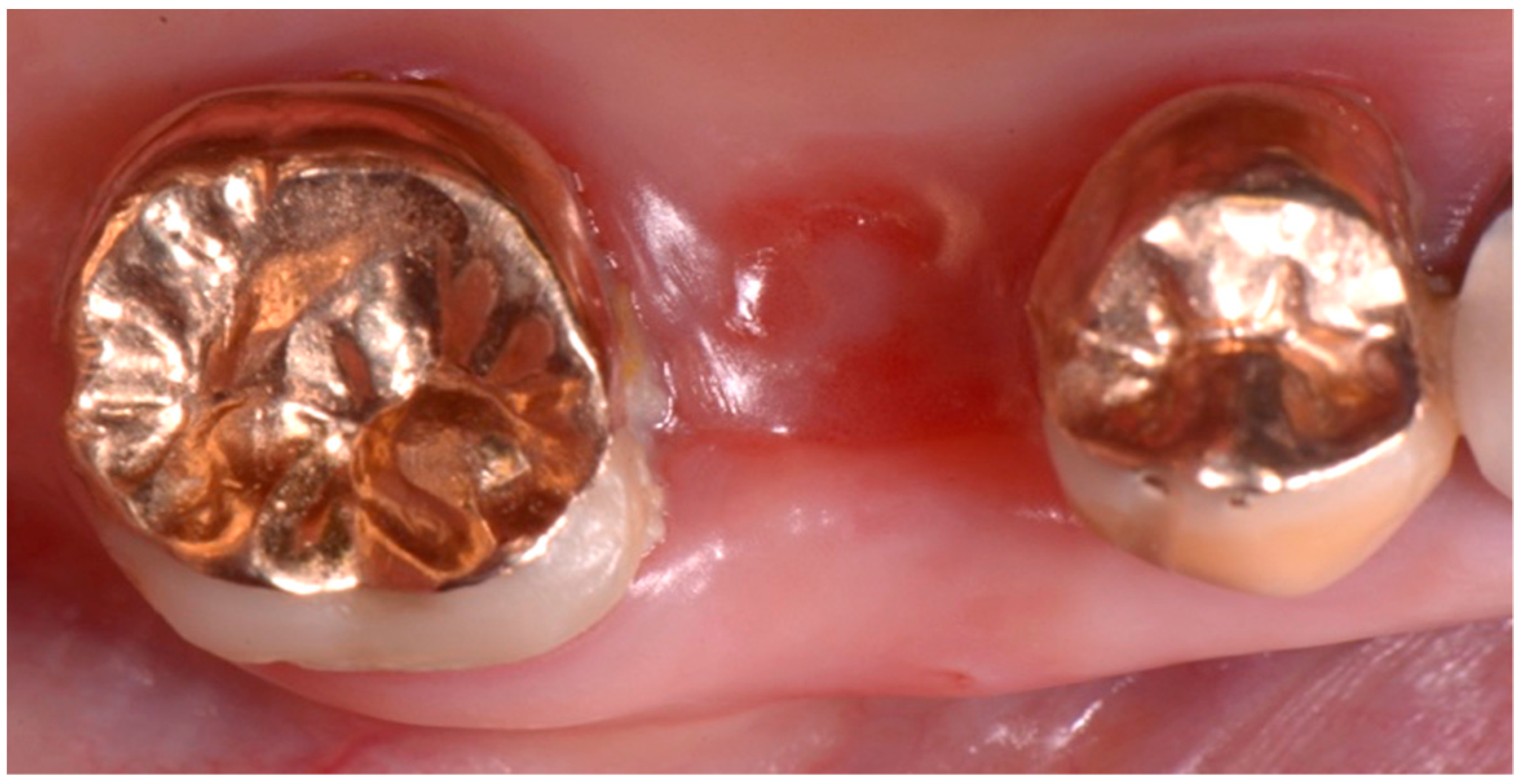
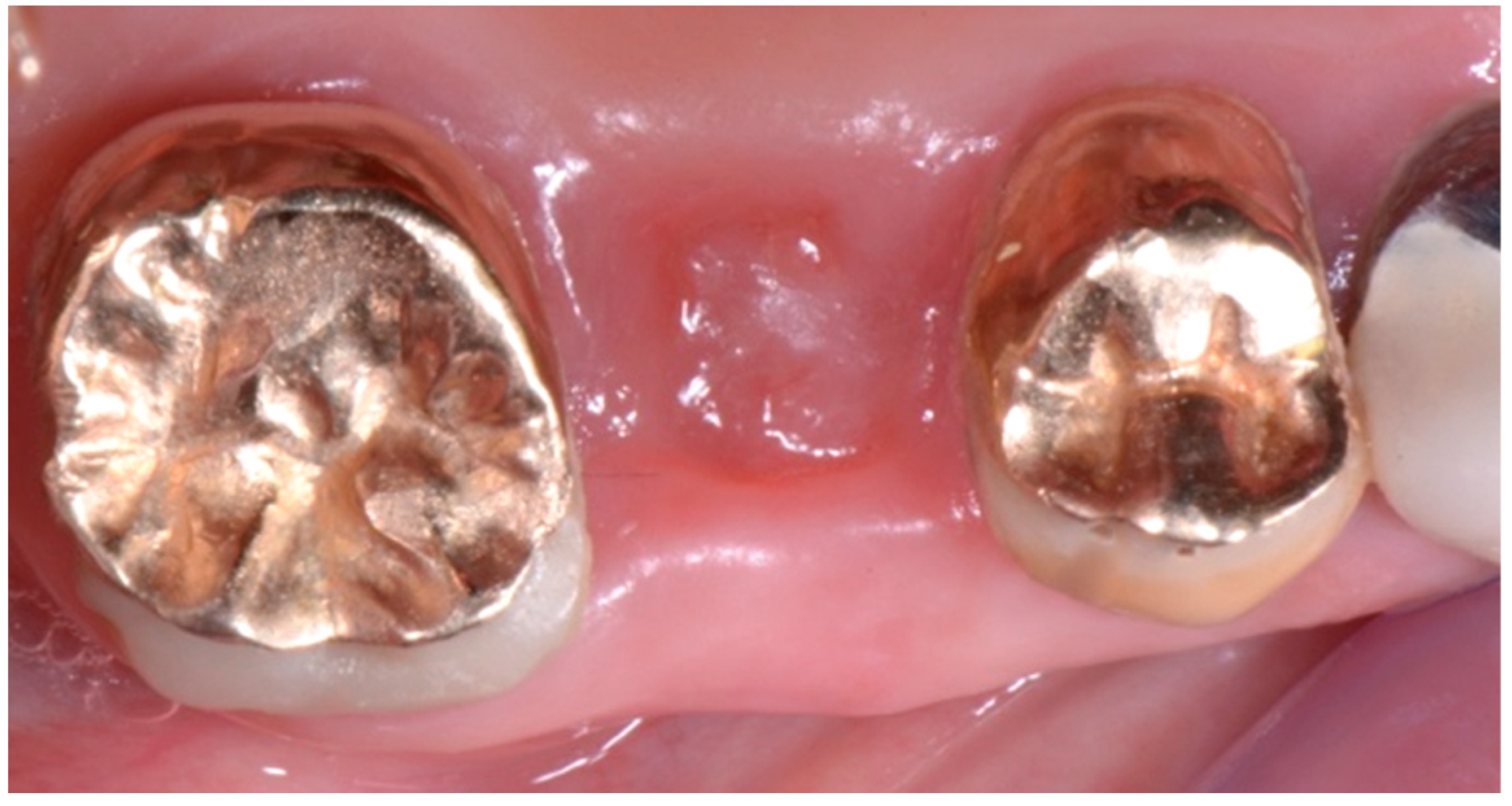
2. Description of the Case
3. Discussion and Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Beth-Tasdogan, N.H.; Mayer, B.; Hussein, H.; Zolk, O. Interventions for managing medication-related osteonecrosis of the jaw. Cochrane Database Syst. Rev. 2017, 10. [Google Scholar] [CrossRef] [PubMed]
- El-Rabbany, M.; Sgro, A.; Lam, D.K.; Shah, P.S.; Azarpazhooh, A. Effectiveness of treatments for medication-related osteonecrosis of the jaw: A systematic review and meta-analysis. J. Am. Dent. Assoc. 2017, 148, 584–594. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, T.; Kawakita, A.; Ueda, N.; Funahara, R.; Tachibana, A.; Kobayashi, M.; Kondou, E.; Takeda, D.; Kojima, Y.; Sato, S.; et al. A multicenter retrospective study of the risk factors associated with medication-related osteonecrosis of the jaw after tooth extraction in patients receiving oral bisphosphonate therapy: Can primary wound closure and a drug holiday really prevent MRONJ? Osteoporos. Int. 2017, 28, 2465–2473. [Google Scholar] [CrossRef] [PubMed]
- Giudice, A.; Barone, S.; Giudice, C.; Bennardo, F.; Fortunato, L. Can platelet-rich fibrin improve healing after surgical treatment of medication-related osteonecrosis of the jaw? A pilot study. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2018, 126, 390–403. [Google Scholar] [CrossRef] [PubMed]
- Thakkar, D.J.; Deshpande, N.C.; Dave, D.H.; Narayankar, S.D. A comparative evaluation of extraction socket preservation with demineralized freeze-dried bone allograft alone and along with platelet-rich fibrin: A clinical and radiographic study. Contemp. Clin. Dent. 2016, 7, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Mourão, C.F.D.A.B.; Calasans-Maia, M.D.; Del Fabbro, M.; Vieira, F.L.D.; de Mello Machado, R.C.; Capella, R.; Miron, R.J.; Alves, G.G. The use of Platelet-rich Fibrin in the management of medication-related osteonecrosis of the jaw: A case series. J. Stomatol. Oral Maxillofac. Surg. 2019. [Google Scholar] [CrossRef]
- Ghanaati, S.; Herrera-Vizcaino, C.; Al-Maawi, S.; Lorenz, J.; Miron, R.J.; Nelson, K.; Schwarz, F.; Choukroun, J.; Sader, R. Fifteen Years of Platelet Rich Fibrin in Dentistry and Oromaxillofacial Surgery: How High is the Level of Scientific Evidence? J. Oral Implantol. 2018, 44, 471–492. [Google Scholar] [CrossRef]
- Park, J.H.; Kim, J.W.; Kim, S.J. Does the Addition of Bone Morphogenetic Protein 2 to Platelet-Rich Fibrin Improve Healing After Treatment for Medication-Related Osteonecrosis of the Jaw? J. Oral Maxillofac. Surg. 2017, 75, 1176–1184. [Google Scholar] [CrossRef]
- Lodi, G.; Sardella, A.; Salis, A.; Demarosi, F.; Tarozzi, M.; Carrassi, A. Tooth Extraction in Patients Taking Intravenous Bisphosphonates: A Preventive Protocol and Case Series. J. Oral Maxillofac. Surg. 2010, 68, 107–110. [Google Scholar] [CrossRef]
- Del Fabbro, M.; Gallesio, G.; Mozzati, M. Autologous platelet concentrates for bisphosphonate-related osteonecrosis of the jaw treatment and prevention—A systematic review of the literature. Eur. J. Cancer 2015, 51, 62–74. [Google Scholar] [CrossRef]
- Inchingolo, F.; Cantore, S.; DiPalma, G.; Georgakopoulos, I.; Almasri, M.; Gheno, E.; Motta, A.; Marrelli, M.; Farronato, D.; Ballini, A.; et al. Platelet rich fibrin in the management of medication-related osteonecrosis of the jaw: A clinical and histopathological evaluation. J. Biol. Regul. Homeost. Agents 2017, 31, 811–816. [Google Scholar] [PubMed]
- Cano-Durán, J.A.; Peña-Cardelles, J.F.; Ortega-Concepción, D.; Paredes-Rodríguez, V.M.; García-Riart, M.; López-Quiles, J. The role of Leucocyte-rich and platelet-rich fibrin (L-PRF) in the treatment of the medication-related osteonecrosis of the jaws (MRONJ). J. Clin. Exp. Dent. 2017, 9, 1051–1059. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Jornet, P.; Perez, A.S.; Mendes, R.A.; Tobias, A.; Arturo, S.P.; Rui, A.M.; Aurelio, T. Medication-related osteonecrosis of the jaw: Is autologous platelet concentrate application effective for prevention and treatment? A systematic review. J. Cranio Maxillofac. Surg. 2016, 44, 1067–1072. [Google Scholar] [CrossRef] [PubMed]
- Kumar, K.R.; Genmorgan, K.; Abdul Rahman, S.M.; Rajan, M.A.; Kumar, T.A.; Prasad, V.S. Role of plasma-rich fibrin in oral surgery. J. Pharm. Bioallied Sci. 2016, 8 (Suppl. S1), 36–38. [Google Scholar]
- Grecu, A.F.; Reclaru, L.; Ardelean, L.C.; Nica, O.; Ciucă, E.M.; Ciurea, M.E. Platelet-Rich Fibrin and its Emerging Therapeutic Benefits for Musculoskeletal Injury Treatment. Medicina 2019, 55, 141. [Google Scholar] [CrossRef]
- Miron, R.J.; Zucchelli, G.; Pikos, M.A.; Salama, M.; Lee, S.; Guillemette, V.; Fujioka-Kobayashi, M.; Bishara, M.; Zhang, Y.; Wang, H.L.; et al. Use of platelet-rich fibrin in regenerative dentistry: A systematic review. Clin. Oral Investig. 2017, 21, 1913–1927. [Google Scholar] [CrossRef]
- Choukroun, J.; Diss, A.; Simonpieri, A.; Girard, M.O.; Schoeffler, C.; Dohan, S.L.; Dohan, A.J.; Mouhyi, J.; Dohan, D.M. Platelet-rich fibrin (PRF): A second-generation platelet concentrate. Part IV: Clinical effects on tissue healing. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2006, 101, 56–60. [Google Scholar] [CrossRef]
- Dohan, D.M.; Choukroun, J.; Diss, A.; Dohan, S.L.; Dohan, A.J.; Mouhyi, J.; Gogly, B. Platelet-rich fibrin (PRF): A second-generation platelet concentrate. Part I: Technological concepts and evolution. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2006, 101, 37–44. [Google Scholar] [CrossRef]
- Asaka, T.; Ohga, N.; Yamazaki, Y.; Sato, J.; Satoh, C.; Kitagawa, Y. Platelet-rich fibrin may reduce the risk delayed recovery in tooth-extracted patients undergoing oral bisphosphonate therapy: A trial study. Clin. Oral Investig. 2017, 21, 2165–2172. [Google Scholar] [CrossRef]
- Dohan, D.M.; Choukroun, J.; Diss, A.; Dohan, S.L.; Dohan, A.J.; Mouhyi, J.; Gogly, B. Platelet-rich fibrin (PRF): A second-generation platelet concentrate. Part II: Platelet-related biologic features. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2006, 101, 45–50. [Google Scholar] [CrossRef]
- Dohan, D.M.; Choukroun, J.; Diss, A.; Dohan, S.L.; Dohan, A.J.; Mouhyi, J.; Gogly, B. Platelet-rich fibrin (PRF): A second-generation platelet concentrate. Part III: Leucocyte activation: A new feature for platelet concentrates? Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2006, 101, 51–55. [Google Scholar] [CrossRef] [PubMed]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pispero, A.; Bancora, I.; Khalil, A.; Scarnò, D.; Varoni, E.M. Use of Platelet Rich Fibrin (PRF)-Based Autologous Membranes for Tooth Extraction in Patients under Bisphosphonate Therapy: A Case Report. Biomedicines 2019, 7, 89. https://doi.org/10.3390/biomedicines7040089
Pispero A, Bancora I, Khalil A, Scarnò D, Varoni EM. Use of Platelet Rich Fibrin (PRF)-Based Autologous Membranes for Tooth Extraction in Patients under Bisphosphonate Therapy: A Case Report. Biomedicines. 2019; 7(4):89. https://doi.org/10.3390/biomedicines7040089
Chicago/Turabian StylePispero, Alberto, Ivan Bancora, Antonious Khalil, Dario Scarnò, and Elena M. Varoni. 2019. "Use of Platelet Rich Fibrin (PRF)-Based Autologous Membranes for Tooth Extraction in Patients under Bisphosphonate Therapy: A Case Report" Biomedicines 7, no. 4: 89. https://doi.org/10.3390/biomedicines7040089
APA StylePispero, A., Bancora, I., Khalil, A., Scarnò, D., & Varoni, E. M. (2019). Use of Platelet Rich Fibrin (PRF)-Based Autologous Membranes for Tooth Extraction in Patients under Bisphosphonate Therapy: A Case Report. Biomedicines, 7(4), 89. https://doi.org/10.3390/biomedicines7040089





