Antimicrobial and Antibiofilm Activities of Citrus Water-Extracts Obtained by Microwave-Assisted and Conventional Methods
Abstract
1. Introduction
2. Experimental Section
2.1. Plant Material
2.2. Hot Water Extraction (HWE)
2.3. Microwave-Assisted Extraction (MAE)
2.4. Yield (%) of Peel Extracts and Preparation for Bioassay
2.5. Bacteria and Culture Conditions
2.6. Antimicrobial Assay
2.7. Static Biofilm Formation
2.8. Statistical Analysis
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ABSmax | Maximum absorbance |
| DT | Duplication time |
| HWE | Hot water extraction |
| λABS | Lag time |
| LSD | Least significant difference |
| HWE | Hot water extraction |
| MAE | Microwave-assisted extraction |
| μABSmax | Maximum specific growth rate |
| N.T. | Not treated |
| CFH | Chloramphenicol |
References
- Byrd, A.L.; Belkaid, Y.; Segre, J.A. The human skin microbiome. Nat. Rev. Microbiol. 2018, 16, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, J.A.; Blaser, M.J.; Caporaso, J.G.; Jansson, J.K.; Lynch, S.V.; Knight, R. Current understanding of the human microbiome. Nat. Med. 2018, 24, 392–400. [Google Scholar] [CrossRef] [PubMed]
- Anderson, D.J.; Podgorny, K.; Berríos-Torres, S.I.; Bratzler, D.W.; Dellinger, E.P.; Greene, L.; Nyquist, A.-C.; Saiman, L.; Yokoe, D.S.; Maragakis, L.L.; et al. Strategies to prevent surgical site infections in acute care hospitals: 2014 update. Infect. Control Hosp. Epidemiol. 2014, 35, 605–627. [Google Scholar] [CrossRef] [PubMed]
- Dyar, O.J.; Pagani, L.; Pulcini, C. Strategies and challenges of antimicrobial stewardship in long-term care facilities. Clin. Microbiol. Infect. 2015, 21, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Kanamori, H.; Weber, D.J.; Rutala, W.A. Healthcare outbreaks associated with a water reservoir and infection prevention strategies. Clin. Infect. Dis. 2016, 62, 1423–1435. [Google Scholar] [CrossRef] [PubMed]
- Fontana, C.; Favaro, M. Coagulase-positive and coagulase-negative staphylococci in human disease. In Pet-to-Man Travelling Staphylococci; Savino, V., Ed.; Academic Press: London, UK, 2018; pp. 25–42. [Google Scholar]
- Musthaq, S.; Mazuy, A.; Jakus, J. The microbiome in dermatology. Clin. Dermatol. 2018. [Google Scholar] [CrossRef]
- Flores-Mireles, A.L.; Walker, J.N.; Caparon, M.; Hultgren, S.J. Urinary tract infections: Epidemiology, mechanisms of infection and treatment options. Nat. Rev. Microbiol. 2015, 13, 269–284. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, T.; Hattori, K.; Inoue, A.; Ishii, T.; Yumoto, T.; Tsukahara, K.; Nakao, A.; Ishihara, S.; Nakayama, S. Bacteremia or pseudobacteremia? Review of Pseudomonas fluorescens infections. World J. Emerg. Med. 2017, 8, 151–154. [Google Scholar] [CrossRef] [PubMed]
- O’Toole, G.; Kaplan, H.B.; Kolter, R. Biofilm formation as microbial development. Annu. Rev. Microbiol. 2000, 54, 49–79. [Google Scholar] [CrossRef] [PubMed]
- Parsek, M.R.; Singh, P.K. Bacterial biofilms: An emerging link to disease pathogenesis. Annu. Rev. Microbiol. 2003, 57, 677–701. [Google Scholar] [CrossRef] [PubMed]
- Hall, C.W.; Mah, T.F. Molecular mechanisms of biofilm-based antibiotic resistance and tolerance in pathogenic bacteria. FEMS Microbiol. Rev. 2017, 41, 276–301. [Google Scholar] [CrossRef] [PubMed]
- Negi, S. Exploring plant and agro-industrial wastes for antimicrobial biochemicals. In Biotransformation of Waste Biomass into High Value Biochemicals; Brar, S., Dhillon, G., Soccol, C., Eds.; Springer: New York, NY, USA, 2014; pp. 335–365. [Google Scholar]
- Lanuzza, F.; Mondello, F.; Tripodo, M.M. Studies about the Utilization of Citrus Wastes in View of Environment Protection. In Pathways to Environmental Sustainability; Salomone, R., Saija, G., Eds.; Springer: London, UK, 2014; pp. 147–156. [Google Scholar]
- Chhikara, N.; Kour, R.; Jaglan, S.; Gupta, P.; Gat, Y.; Panghal, A. Citrus medica: Nutritional, phytochemical composition and health benefits–A review. Food Funct. 2018, 9, 1978–1992. [Google Scholar] [CrossRef] [PubMed]
- Braidy, N.; Behzad, S.; Habtemariam, S.; Ahmed, T.; Daglia, M.; Nabavi, S.M.; Sobarzo-Sanchez, E.; Nabavi, S.F. Neuroprotective effects of citrus fruit-derived flavonoids, nobiletin and tangeretin in Alzheimer’s and Parkinson’s disease. CNS Neurol. Disord. Drug Targets 2017, 16, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Gualdani, R.; Cavalluzzi, M.M.; Lentini, G.; Habtemariam, S. The chemistry and pharmacology of citrus limonoids. Molecules 2016, 21, 1530. [Google Scholar] [CrossRef] [PubMed]
- Al-Snafi, A.E. Medicinal plants with antimicrobial activities (part 2): Plant based review. Sch. Acad. J. Pharm. 2016, 5, 208–239. [Google Scholar] [CrossRef]
- Putnik, P.; BursaćKovačević, D.; RežekJambrak, A.; Barba, F.J.; Cravotto, G.; Binello, A.; Shpigelman, A. Innovative “green” and novel strategies for the extraction of bioactive added value compounds from citrus wastes—A review. Molecules 2017, 22, 680. [Google Scholar] [CrossRef] [PubMed]
- Flórez, N.; Conde, E.; Domínguez, H. Microwave assisted water extraction of plant compounds. J. Chem. Technol. Biotechnol. 2015, 90, 590–607. [Google Scholar] [CrossRef]
- Shan, Y. Comprehensive Utilization of Citrus by-Products; Academic Press: London, UK, 2016; pp. 335–365. [Google Scholar]
- Louche, L.M.M.; Gaydou, E.M.; Lesage, J.C. Determination of phlorin as peel marker in orange (Citrus sinensis) fruits and juices. J. Agric. Food Chem. 1998, 46, 4193–4197. [Google Scholar] [CrossRef]
- Miyake, Y.; Hiramitsu, M. Isolation and extraction of antimicrobial substances against oral bacteria from lemon peel. J. Food Sci. Technol. 2011, 48, 635–639. [Google Scholar] [CrossRef] [PubMed]
- Adukwu, E.C.; Allen, S.C.H.; Phillips, C.A. The anti-biofilm activity of lemongrass (Cymbopogon flexuosus) and grapefruit (Citrus paradisi) essential oils against five strains of Staphylococcus aureus. J. Appl. Microbiol. 2012, 113, 1217–1227. [Google Scholar] [CrossRef] [PubMed]
- Vikram, A.; Jayaprakasha, G.K.; Jesudhasan, P.R.; Pillai, S.D.; Patil, B.S. Suppression of bacterial cell–cell signalling, biofilm formation and type III secretion system by citrus flavonoids. J. Appl. Microbiol. 2010, 109, 515–527. [Google Scholar] [CrossRef] [PubMed]
- EUCAST, Disk Diffusion Method for Antimicrobial Susceptibility Testing—Version 6.0, 2017. Available online: www.eucast.org (accessed on 16 May 2018).
- Dalgaard, P.; Koutsoumanis, K. Comparison of maximum specific growth rates and lag times estimated from absorbance and viable count data by different mathematical models. J. Microbiol. Methods 2001, 43, 183–196. [Google Scholar] [CrossRef]
- Wang, L.; Jo, M.J.; Katagiri, R.; Harata, K.; Ohta, M.; Ogawa, A.; Kamegai, M.; Ishida, Y.; Tanoue, S.; Kimura, S.; et al. Antioxidant effects of citrus pomace extracts processed by super-heated steam. LWT 2018, 90, 331–338. [Google Scholar] [CrossRef]
- Hayat, K.; Zhang, X.; Farooq, U.; Abbas, S.; Xia, S.; Jia, C.; Zhong, F.; Zhang, J. Effect of microwave treatment on phenolic content and antioxidant activity of citrus mandarin pomace. Food Chem. 2010, 123, 423–429. [Google Scholar] [CrossRef]
- Xu, G.H.; Chen, J.C.; Liu, D.H.; Zhang, Y.H.; Jiang, P.; Ye, X.Q. Minerals, phenolic compounds, and antioxidant capacity of citrus peel extract by hot water. J. Food Sci. 2008, 73, C11–C18. [Google Scholar] [CrossRef] [PubMed]
- Ferrazzano, G.F.; Roberto, L.; Amato, I.; Cantile, T.; Sangianantoni, G.; Ingenito, A. Antimicrobial properties of green tea extract against cariogenic microflora: An in vivo study. J. Med. Food 2011, 14, 907–911. [Google Scholar] [CrossRef] [PubMed]
- Di Donna, L.; Gallucci, G.; Malaj, N.; Romano, E.; Tagarelli, A.; Sindona, G. Recycling of industrial essential oil waste: Brutieridin and Melitidin, two anticholesterolaemic active principles from bergamot albedo. Food Chem. 2011, 125, 438–441. [Google Scholar] [CrossRef]
- Papoutsis, K.; Vuong, Q.V.; Golding, J.B.; Hasperué, J.H.; Pristijono, P.; Bowyer, M.C.; Scarlett, C.J.; Stathopoulos, C.E. Pretreatment of citrus by-products affects polyphenol recovery: A review. Food Rev. Int. 2018, 1–26. [Google Scholar] [CrossRef]
- Proestos, C.; Komaitis, N. Application of microwave-assisted extraction to the fast extraction of plant phenolic compounds. LWT 2008, 41, 652–659. [Google Scholar] [CrossRef]
- Rahmanian, N.; Jafari, S.M.; Wani, T.A. Bioactive profile, dehydration, extraction and application of the bioactive components of olive leaves. Trends Food Sci. Tech. 2015, 42, 150–172. [Google Scholar] [CrossRef]
- Louche, L.M.M.; Luro, F.; Gaydou, E.M.; Lesage, J.C. Phlorin screening in various citrus species and varieties. J. Agric. Food Chem. 2000, 48, 4728–4733. [Google Scholar] [CrossRef]
- Nipornram, S.; Tochampa, W.; Rattanatraiwong, P.; Singanusong, R. Optimization of low power ultrasound-assisted extraction of phenolic compounds from mandarin (Citrus reticulata Blanco cv. Sainampueng) peel. Food Chem. 2018, 241, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Safdar, M.N.; Kausar, T.; Jabbar, S.; Mumtaz, A.; Ahad, K.; Saddozai, A.A. Extraction and quantification of polyphenols from kinnow (Citrus reticulate L.) peel using ultrasound and maceration techniques. J. Food Drug Anal. 2017, 25, 488–500. [Google Scholar] [CrossRef] [PubMed]
- Trentin, D.S.; Giordani, R.B.; Zimmer, K.R.; da Silva, A.G.; da Silva, M.V.; dos Santos Correia, M.T.; Baumvol, I.J.R.; Macedo, A.J. Potential of medicinal plants from the Brazilian semi-arid region (Caatinga) against Staphylococcus epidermidis planktonic and biofilm lifestyles. J. Ethnopharmacol. 2011, 137, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Trentin, D.S.; Silva, D.B.; Amaral, M.W.; Zimmer, K.R.; Silva, M.V.; Lopes, N.P.; Giordani, R.B.; Macedo, A.J. Tannins possessing bacteriostatic effect impair Pseudomonas aeruginosa adhesion and biofilm formation. PLoS ONE 2013, 8, e66257. [Google Scholar] [CrossRef] [PubMed]
- Kohanski, M.A.; Dwyer, D.J.; Collins, J.J. How antibiotics kill bacteria: From targets to networks. Nat. Rev. Microbiol. 2010, 8, 423–435. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Qiu, Y.; Shi, H.; Yin, H. The importance of lag time extension in determining bacterial resistance to antibiotics. Analyst 2016, 141, 3059–3067. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Xie, S.; Ahmed, S.; Wang, F.; Gu, Y.; Zhang, C.; Chai, X.; Wu, Y.; Cai, J.; Cheng, G. Antimicrobial Activity and Resistance: Influencing Factors. Front. Pharmacol. 2017, 8, 364. [Google Scholar] [CrossRef] [PubMed]
- Theophel, K.; Schacht, V.J.; Schlüter, M.; Schnell, S.; Stingu, C.S.; Schaumann, R.; Bunge, M. The importance of growth kinetic analysis in determining bacterial susceptibility against antibiotics and silver nanoparticles. Front. Microbiol. 2014, 5, 544. [Google Scholar] [CrossRef] [PubMed]
- Belda-Galbis, C.M.; Pina-Pérez, M.C.; Espinosa, J.; Marco-Celdrán, A.; Martínez, A.; Rodrigo, D. Use of the modified Gompertz equation to assess the Stevia rebaudiana Bertoni antilisterial kinetics. Food Microbial. 2014, 38, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Bevilacqua, A.; Corbo, M.R.; Sinigaglia, M. In vitro evaluation of the antimicrobial activity of eugenol, limonene, and citrus extract against bacteria and yeasts, representative of the spoiling microflora of fruit juices. J. Food Prot. 2010, 73, 888–894. [Google Scholar] [CrossRef] [PubMed]
- Deepika, M.S.; Thangam, R.; Sakthidhasan, P.; Arun, S.; Sivasubramanian, S.; Thirumurugan, R. Combined effect of a natural flavonoid rutin from Citrus sinensis and conventional antibiotic gentamicin on Pseudomonas aeruginosa biofilm formation. Food Control 2018, 90, 282–294. [Google Scholar] [CrossRef]
- Alav, I.; Sutton, J.M.; Rahman, K.M. Role of bacterial efflux pumps in biofilm formation. J. Antimicrob. Chemother. 2018, dky042. [Google Scholar] [CrossRef] [PubMed]
- Rasko, D.A.; Sperandio, V. Anti-virulence strategies to combat bacteria-mediated disease. Nat. Rev. Drug Discov. 2010, 9, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Reen, F.J.; Gutiérrez-Barranquero, J.A.; Parages, M.L. Coumarin: A novel player in microbial quorum sensing and biofilm formation inhibition. Appl. Microbiol. Biot. 2018, 102, 2063–2073. [Google Scholar] [CrossRef] [PubMed]
- Villa, F.; Cappitelli, F. Plant-derived bioactive compounds at sub-lethal concentrations: Towards smart biocide-free antibiofilm strategies. Phytochem. Rev. 2013, 12, 245–254. [Google Scholar] [CrossRef]
- Yue, J.; Yang, H.; Liu, S.; Song, F.; Guo, J.; Huang, C. Influence of naringenin on the biofilm formation of Streptococcus mutans. J. Dent. 2018. [Google Scholar] [CrossRef] [PubMed]

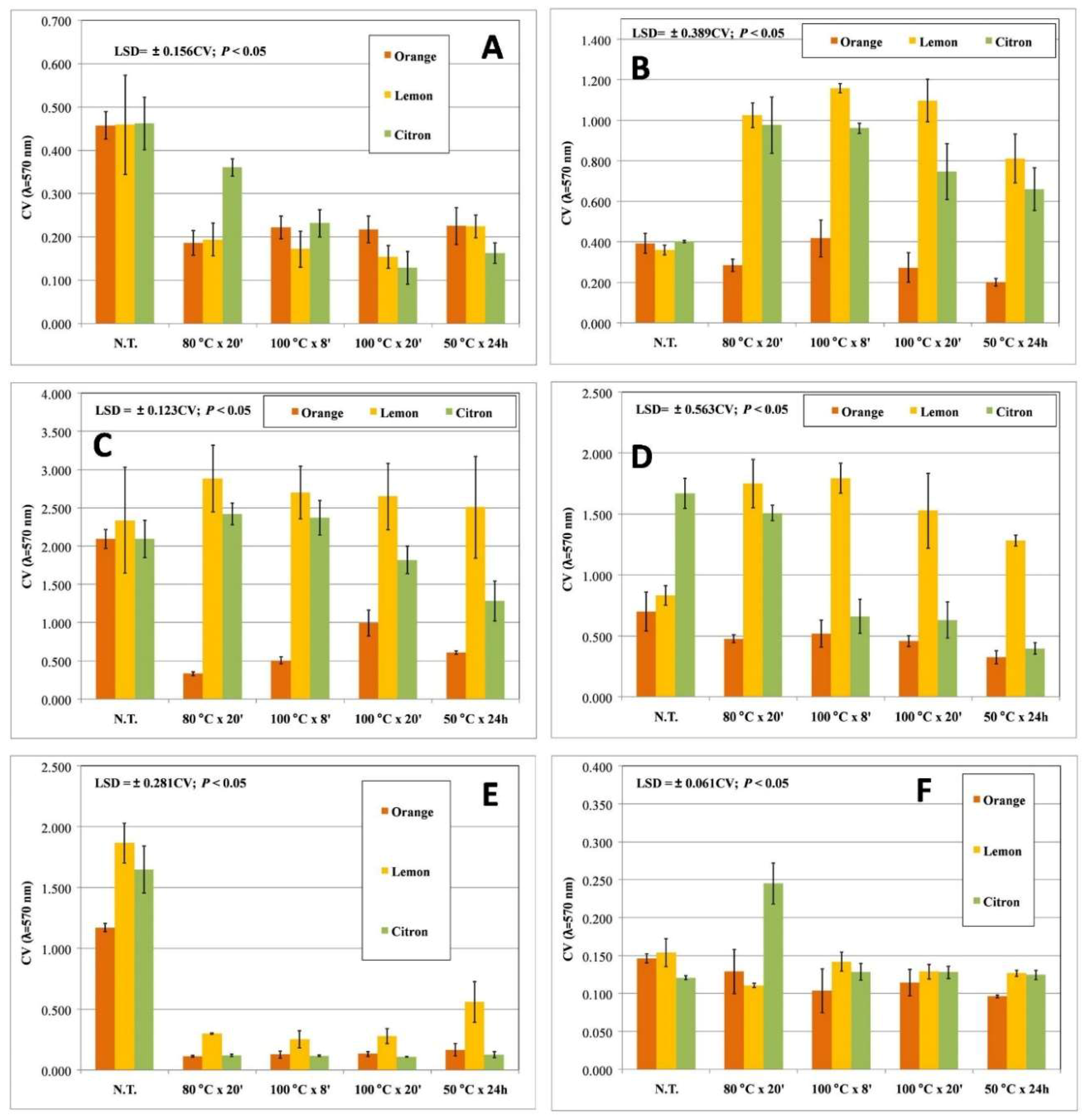
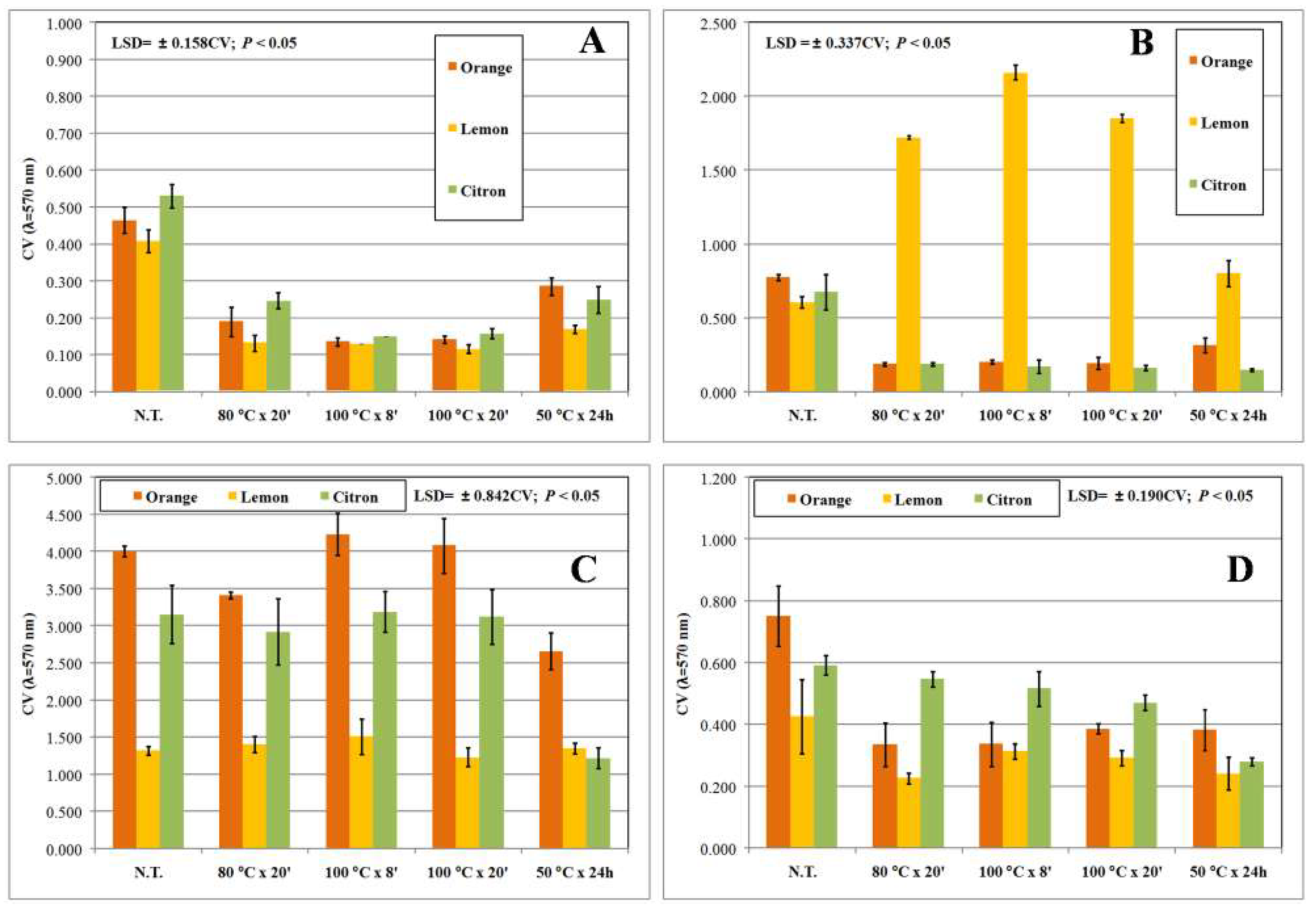
| Target Strain | Growth Conditions |
|---|---|
| Staphylococcus epidermidis UR63 | Tryptic Soy Broth (TSB), 37 °C, 130 rpm |
| Staphylococcus saprophyticus UR18 | |
| Staphylococcus caprae DSM 20608 | |
| Staphylococcus xylosus DSM20266T | |
| Pseudomonas fluorescens NCPPB1964 | Luria Bertani (LB), 30 °C, 130 rpm |
| Pseudomonas fluorescens ITEM 17298 | |
| Pseudomonas fluorescens ITEM 17299 | |
| Pseudomonas fluorescens ITEM 84094 | |
| Pseudomonas putida ITEM 17297 | |
| Escherichia coli K12 | Luria Bertani (LB), 37 °C, 130 rpm |
| Bacterial Strain | Citron | Lemon | Orange | LSD | Citron | Lemon | Orange | LSD | |
|---|---|---|---|---|---|---|---|---|---|
| Extracts | λABS (h) | ABSmax | |||||||
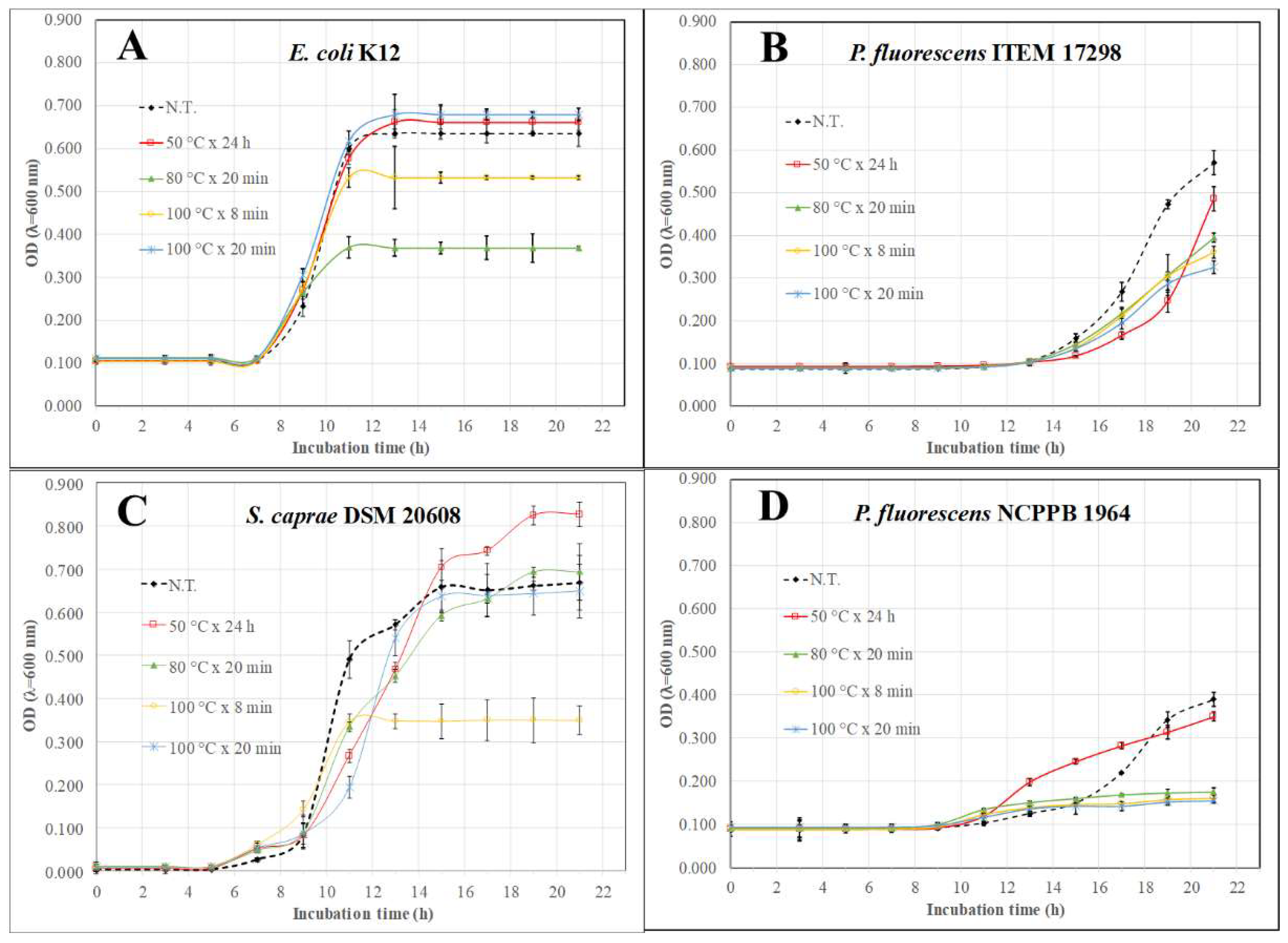
| E. coli K12 | N.T. | 7.4 | 7.3 | 6.9 | n.s. | 0.711 | 0.699 | 0.664 | 0.109 |
| HWE | 9.1 | 8.6 | 7.7 | 0.642 | 0.785 | 1.249 | |||
| MAE1 | 10.1 | 10.8 | 9.5 | 0.368 | 0.733 | 1.186 | |||
| MAE2 | 9.4 | 9.7 | 8.9 | 0.532 | 0.800 | 1.000 | |||
| MAE3 | 9.1 | 11.1 | 13.8 | 0.679 | 0.787 | 1.180 | |||
| P. fluorescens 84094 | N.T. | 12.3 | 12.1 | 11.5 | 1.9 | 0.687 | 0.676 | 0.642 | 0.180 |
| HWE | 13.4 | 7.9 | 13.8 | 0.805 | 0.999 | 0.611 | |||
| MAE1 | 12.0 | 7.6 | 12.1 | 1.141 | 1.036 | 0.929 | |||
| MAE2 | 13.8 | 6.8 | 12.4 | 0.747 | 1.042 | 0.844 | |||
| MAE3 | 14.2 | 8.1 | 12.2 | 0.654 | 0.966 | 0.909 | |||
| P. fluorescens ITEM 17298 | N.T. | 13.5 | 13.2 | 12.6 | 1.5 | 0.541 | 0.532 | 0.506 | 0.057 |
| HWE | 13.8 | 13.0 | 10.8 | 0.678 | 0.475 | 0.549 | |||
| MAE1 | 15.5 | 14.3 | 10.8 | 0.900 | 0.298 | 0.695 | |||
| MAE2 | 15.6 | 15.5 | 10.7 | 0.735 | 0.269 | 0.559 | |||
| MAE3 | 14.9 | 14.7 | 10.7 | 0.800 | 0.291 | 0.523 | |||
| P. fluorescens ITEM 17299 | N.T. | 13.1 | 12.8 | 12.2 | 1.6 | 0.549 | 0.540 | 0.513 | 0.189 |
| HWE | 14.8 | 14.0 | 9.7 | 1.091 | 1.174 | 0.772 | |||
| MAE1 | 14.3 | 14.7 | 9.8 | 1.010 | 0.897 | 0.557 | |||
| MAE2 | 14.3 | 13.7 | 11.1 | 0.956 | 0.982 | 0.619 | |||
| MAE3 | 15.2 | 14.2 | 11.5 | 0.934 | 0.823 | 0.524 | |||
| P. fluorescens NCPPB 1964 | N.T. | 13.5 | 13.2 | 12.6 | 2.0 | 0.41 | 0.41 | 0.39 | 0.034 |
| HWE | 20.0 | 14.0 | 10.3 | 0.304 | 0.516 | 0.350 | |||
| MAE1 | 20.0 | 15.2 | 21.6 | 0.293 | 0.430 | 0.176 | |||
| MAE2 | 19.4 | 14.9 | 25.5 | 0.304 | 0.427 | 0.161 | |||
| MAE3 | 16.9 | 15.0 | 31.5 | 0.475 | 0.420 | 0.155 | |||
| P. putida ITEM 17297 | N.T. | 14.9 | 14.6 | 14.1 | 3.2 | 0.399 | 0.392 | 0.372 | 0.576 |
| HWE | 16.3 | 9.8 | 11.9 | 0.846 | 0.994 | 1.528 | |||
| MAE1 | 16.5 | 9.2 | 13.9 | 1.592 | 0.780 | 1.414 | |||
| MAE2 | 16.4 | 7.8 | 13.6 | 1.569 | 0.877 | 1.426 | |||
| MAE3 | 16.5 | 5.2 | 13.2 | 1.586 | 0.930 | 1.402 | |||
| S. caprae DSM 20608 | N.T. | 14.0 | 13.8 | 13.1 | 1.6 | 0.713 | 0.700 | 0.666 | 0.210 |
| HWE | 13.6 | 7.3 | 2.4 | 1.198 | 1.293 | 0.824 | |||
| MAE1 | 13.3 | 4.9 | 8.1 | 1.108 | 1.153 | 0.693 | |||
| MAE2 | 13.1 | 5.1 | 9.3 | 1.163 | 1.229 | 0.349 | |||
| MAE3 | 13.1 | 5.7 | 8.6 | 1.255 | 1.206 | 0.648 | |||
| S. epidermidis UR63 | N.T. | 9.3 | - | - | 1.0 | 0.902 | - | - | 0.125 |
| HWE | 8.3 | - | - | 1.224 | - | - | |||
| MAE1 | 7.0 | - | - | 1.248 | - | - | |||
| MAE2 | 6.6 | - | - | 1.253 | - | - | |||
| MAE3 | 7.8 | - | - | 1.217 | - | - | |||
| S. saprophyticus UR18 | N.T. | - | 7.0 | 6.9 | 1.3 | - | 0.354 | 0.348 | 0.038 |
| HWE | - | 11.1 | 9.5 | - | 0.362 | 0.342 | |||
| MAE1 | - | 9.3 | 10.8 | - | 0.406 | 0.400 | |||
| MAE2 | - | 10.9 | 10.5 | - | 0.426 | 0.318 | |||
| MAE3 | - | 9.8 | 14.2 | - | 0.420 | 0.414 | |||
| S. xylosus DSM20266T | N.T. | 11.3 | 11.1 | - | 1.2 | 0.901 | 0.885 | - | 0.088 |
| HWE | 13.4 | 3.9 | - | 1.079 | 1.063 | - | |||
| MAE1 | 12.2 | 8.8 | - | 1.108 | 1.041 | - | |||
| MAE2 | 12.2 | 9.0 | - | 1.199 | 0.430 | - | |||
| MAE3 | 12.2 | 8.0 | - | 1.231 | 0.807 | - | |||
| Header | Citron | Lemon | Orange | LSD | Citron | Lemon | Orange | LSD | |
|---|---|---|---|---|---|---|---|---|---|
| Extracts | µABS (h−1) | DT (h) | |||||||
| E. coli K12 | N.T. | 0.231 | 0.227 | 0.216 | 0.081 | 4.3 | 4.2 | 4.0 | 1.9 |
| HWE | 0.420 | 0.123 | 0.206 | 1.6 | 5.8 | 3.5 | |||
| MAE1 | 0.294 | 0.160 | 0.220 | 2.3 | 4.1 | 3.3 | |||
| MAE2 | 0.402 | 0.220 | 0.212 | 1.7 | 3.5 | 3.4 | |||
| MAE3 | 0.426 | 0.173 | 0.394 | 1.6 | 3.9 | 1.8 | |||
| P. fluorescens 84094 | N.T. | 0.234 | 0.230 | 0.219 | 0.023 | 3.3 | 3.2 | 3.0 | 0.5 |
| HWE | 0.230 | 0.166 | 0.250 | 2.9 | 4.3 | 2.9 | |||
| MAE1 | 0.225 | 0.168 | 0.254 | 3.0 | 4.3 | 2.8 | |||
| MAE2 | 0.214 | 0.163 | 0.256 | 3.1 | 4.1 | 2.8 | |||
| MAE3 | 0.202 | 0.165 | 0.261 | 3.3 | 4.0 | 2.7 | |||
| P. fluorescens ITEM 17298 | N.T. | 0.215 | 0.211 | 0.201 | 0.023 | 3.7 | 3.6 | 3.4 | 0.5 |
| HWE | 0.184 | 0.192 | 0.167 | 3.9 | 3.5 | 4.3 | |||
| MAE1 | 0.309 | 0.141 | 0.204 | 2.3 | 4.9 | 3.5 | |||
| MAE2 | 0.277 | 0.135 | 0.174 | 2.6 | 5.0 | 4.1 | |||
| MAE3 | 0.258 | 0.139 | 0.169 | 2.8 | 5.0 | 4.2 | |||
| P. fluorescens ITEM 17299 | N.T. | 0.213 | 0.209 | 0.199 | 0.056 | 3.6 | 3.5 | 3.3 | 0.7 |
| HWE | 0.313 | 0.331 | 0.194 | 2.3 | 2.2 | 3.7 | |||
| MAE1 | 0.289 | 0.314 | 0.155 | 2.5 | 2.3 | 4.6 | |||
| MAE2 | 0.302 | 0.315 | 0.194 | 2.4 | 2.1 | 3.7 | |||
| MAE3 | 0.293 | 0.286 | 0.182 | 2.4 | 2.4 | 3.9 | |||
| P. fluorescens NCPPB 1964 | N.T. | 0.162 | 0.159 | 0.152 | 0.015 | 6.5 | 3.9 | 6.3 | 1.1 |
| HWE | 0.113 | 0.176 | 0.110 | 5.6 | 4.0 | 12.3 | |||
| MAE1 | 0.128 | 0.164 | 0.056 | 7.0 | 4.2 | 14.1 | |||
| MAE2 | 0.106 | 0.161 | 0.049 | 3.3 | 4.2 | 16.4 | |||
| MAE3 | 0.215 | 0.159 | 0.042 | 3.8 | 3.8 | 3.6 | |||
| P. putida ITEM 17297 | N.T. | 0.212 | 0.208 | 0.198 | 0.071 | 2.0 | 3.3 | 2.1 | 1.3 |
| HWE | 0.351 | 0.219 | 0.344 | 1.4 | 4.0 | 1.6 | |||
| MAE1 | 0.514 | 0.172 | 0.438 | 1.4 | 4.2 | 1.7 | |||
| MAE2 | 0.513 | 0.163 | 0.422 | 1.4 | 5.2 | 1.8 | |||
| MAE3 | 0.528 | 0.141 | 0.392 | 2.1 | 2.0 | 1.9 | |||
| S. caprae DSM 20608 | N.T. | 0.378 | 0.372 | 0.353 | 0.060 | 2.1 | 1.9 | 1.9 | 0.3 |
| HWE | 0.334 | 0.375 | 0.349 | 2.4 | 2.4 | 1.7 | |||
| MAE1 | 0.305 | 0.277 | 0.399 | 2.3 | 2.3 | 1.9 | |||
| MAE2 | 0.306 | 0.292 | 0.359 | 2.3 | 2.1 | 1.9 | |||
| MAE3 | 0.315 | 0.315 | 0.402 | 1.3 | - | ||||
| S. epidermidis UR63 | N.T. | 0.549 | - | - | 0.057 | 1.5 | - | - | 0.4 |
| HWE | 0.494 | - | - | 1.7 | - | - | |||
| MAE1 | 0.414 | - | - | 1.7 | - | - | |||
| MAE2 | 0.419 | - | - | 2.0 | - | - | |||
| MAE3 | 0.352 | - | - | - | - | - | |||
| S. saprophyticus UR18 | N.T. | - | 0.155 | 0.153 | 0.025 | - | 4.8 | 4.7 | 0.5 |
| HWE | - | 0.169 | 0.206 | - | 4.2 | 3.5 | |||
| MAE1 | - | 0.164 | 0.220 | - | 4.4 | 3.3 | |||
| MAE2 | - | 0.148 | 0.212 | - | 4.9 | 3.4 | |||
| MAE3 | - | 0.163 | 0.394 | - | 4.4 | 1.8 | |||
| S. xylosus DSM20266T | N.T. | 0.341 | 0.336 | - | 0.032 | 2.1 | 2.0 | - | 0.30.5 |
| HWE | 0.234 | 0.332 | - | 3.1 | 2.0 | - | |||
| MAE1 | 0.203 | 0.407 | - | - | 4.8 | 4.7 | |||
| MAE2 | 0.220 | 0.272 | - | - | 4.2 | 3.5 | |||
| MAE3 | 0.221 | 0.274 | - | - | 4.4 | 3.3 | |||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caputo, L.; Quintieri, L.; Cavalluzzi, M.M.; Lentini, G.; Habtemariam, S. Antimicrobial and Antibiofilm Activities of Citrus Water-Extracts Obtained by Microwave-Assisted and Conventional Methods. Biomedicines 2018, 6, 70. https://doi.org/10.3390/biomedicines6020070
Caputo L, Quintieri L, Cavalluzzi MM, Lentini G, Habtemariam S. Antimicrobial and Antibiofilm Activities of Citrus Water-Extracts Obtained by Microwave-Assisted and Conventional Methods. Biomedicines. 2018; 6(2):70. https://doi.org/10.3390/biomedicines6020070
Chicago/Turabian StyleCaputo, Leonardo, Laura Quintieri, Maria Maddalena Cavalluzzi, Giovanni Lentini, and Solomon Habtemariam. 2018. "Antimicrobial and Antibiofilm Activities of Citrus Water-Extracts Obtained by Microwave-Assisted and Conventional Methods" Biomedicines 6, no. 2: 70. https://doi.org/10.3390/biomedicines6020070
APA StyleCaputo, L., Quintieri, L., Cavalluzzi, M. M., Lentini, G., & Habtemariam, S. (2018). Antimicrobial and Antibiofilm Activities of Citrus Water-Extracts Obtained by Microwave-Assisted and Conventional Methods. Biomedicines, 6(2), 70. https://doi.org/10.3390/biomedicines6020070










