Biomarkers of Clot Activation and Degradation and Risk of Future Major Cardiovascular Events in Acute Exacerbation of COPD: A Cohort Sub-Study in a Randomized Trial Population
Abstract
:1. Introduction
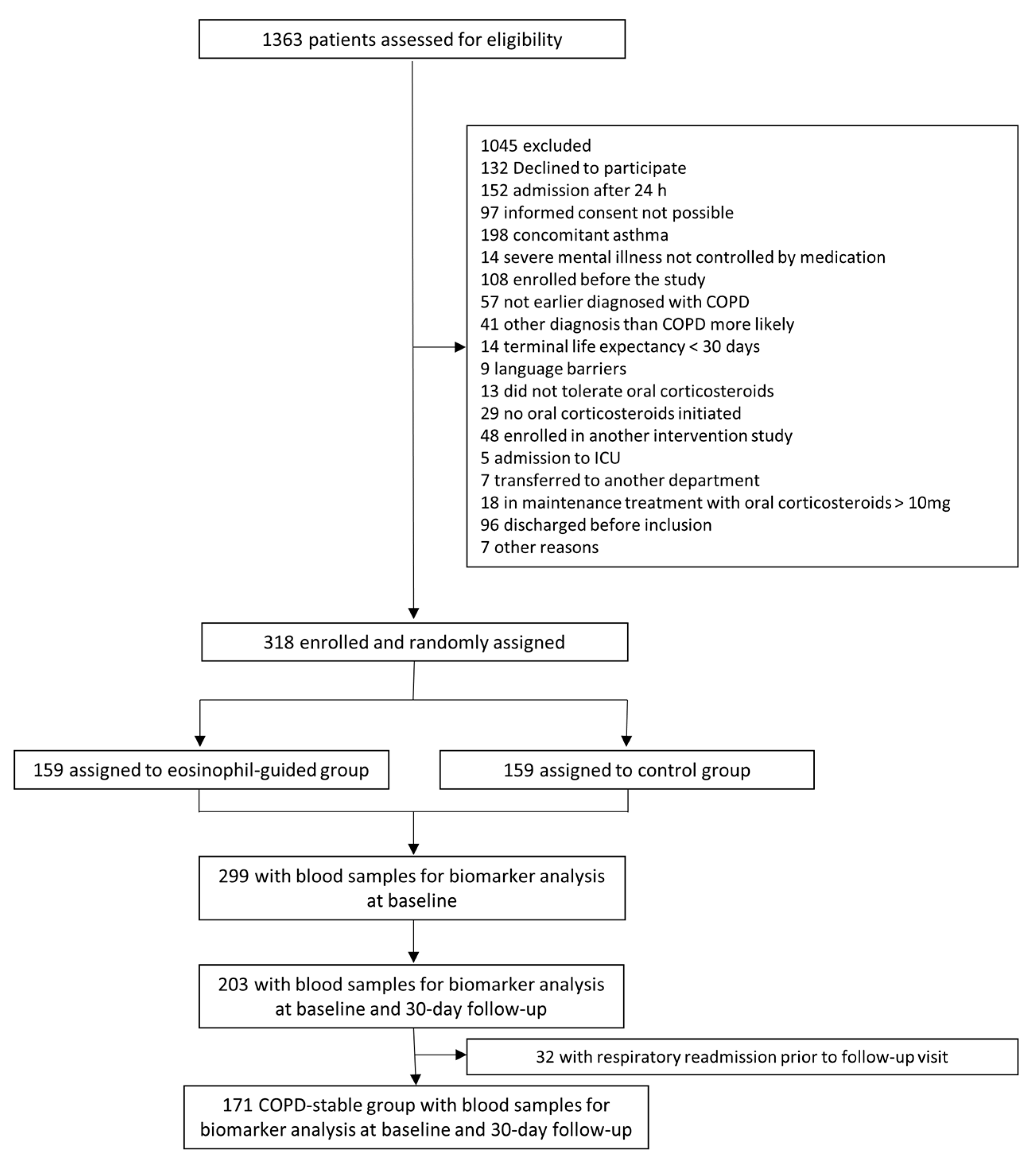
2. Materials and Methods
2.1. Patients
2.2. Measurements
2.3. Confounding Factors
2.4. Outcome
2.5. Reporting
2.6. Statistical Analyses
2.7. Model Control
2.8. Explorative Analyses
3. Results
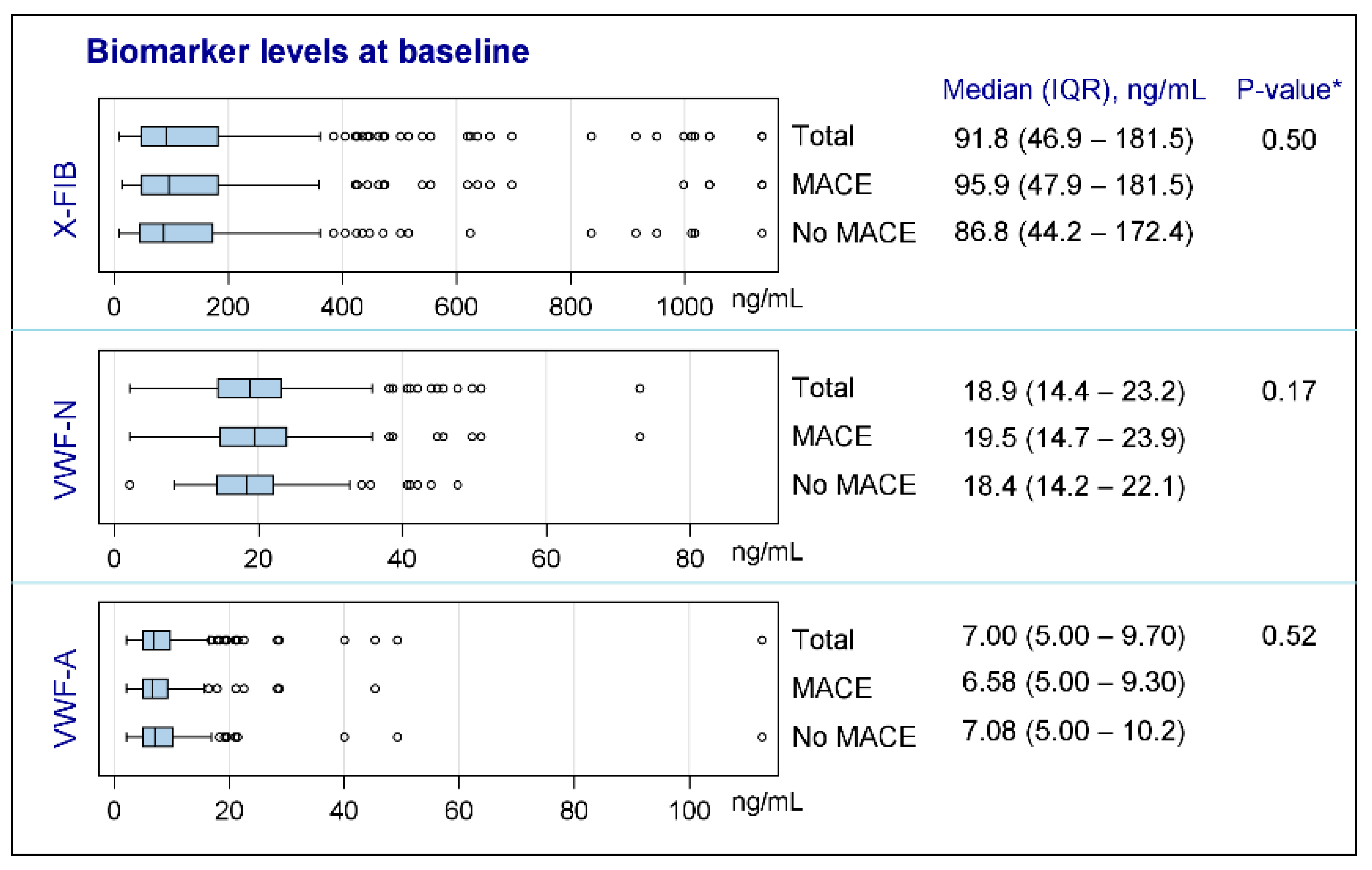
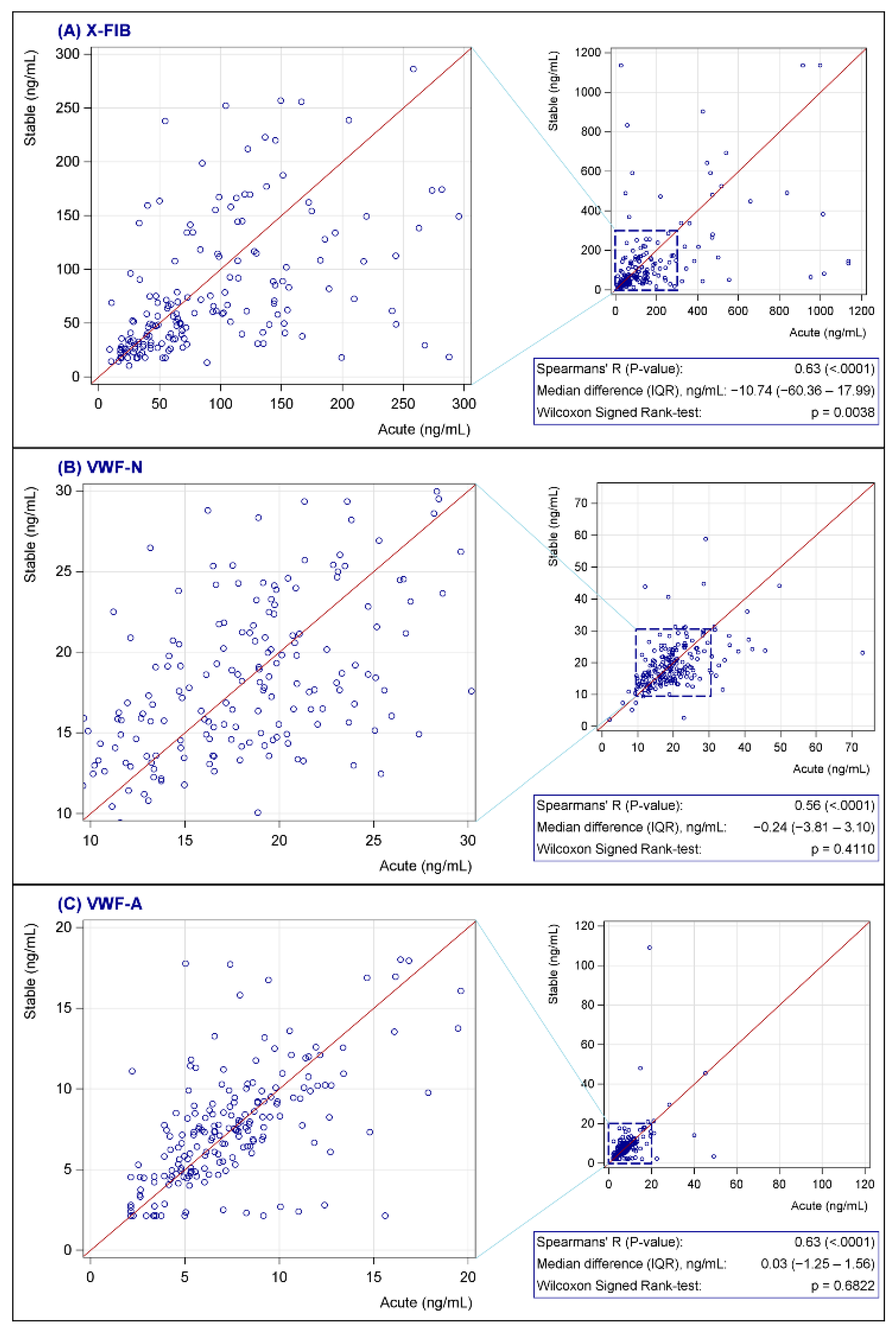
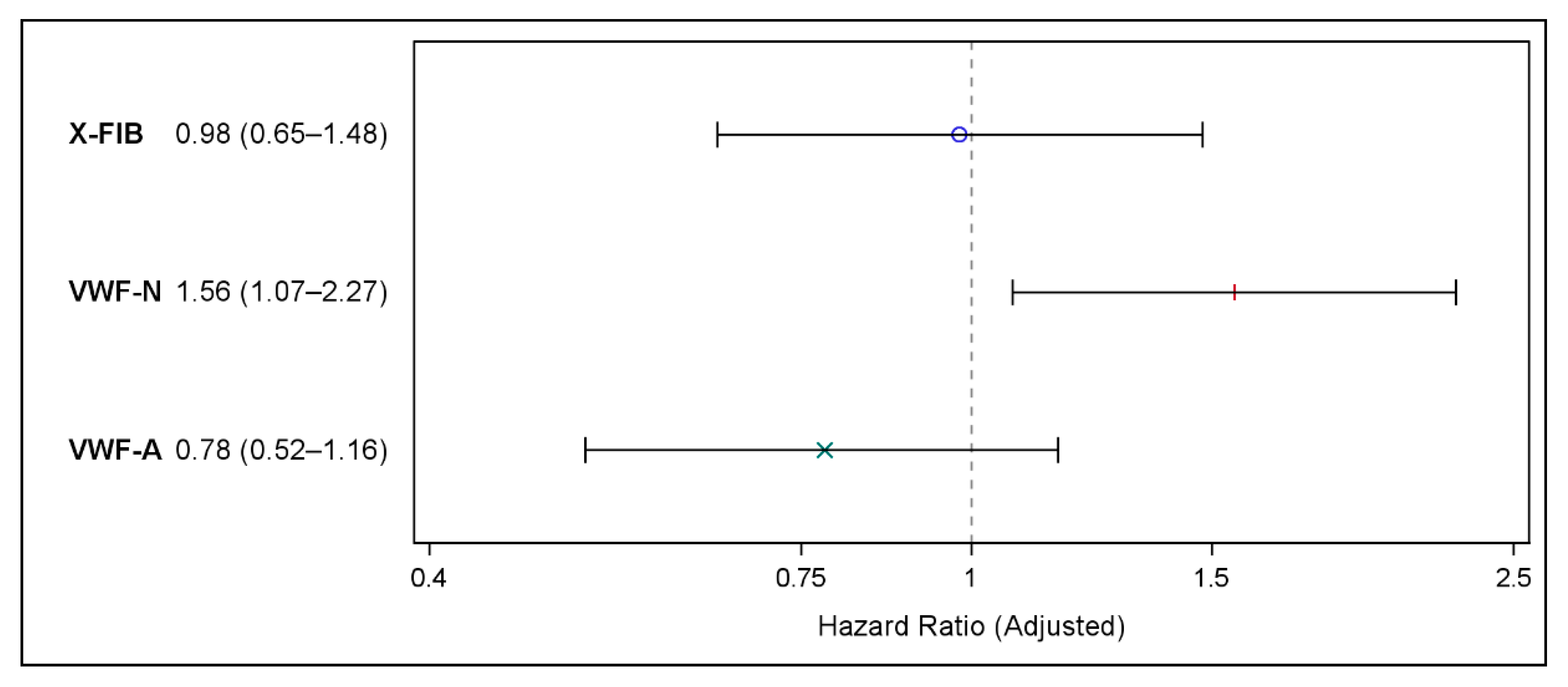
3.1. Primary Outcome
3.2. Secondary Outcome
3.3. Explorative Outcomes
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Diseases, G.B.D.; Injuries, C. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222. [Google Scholar] [CrossRef]
- Müllerova, H.; Agusti, A.; Erqou, S.; Mapel, D.W. Cardiovascular comorbidity in COPD: Systematic literature review. Chest 2013, 144, 1163–1178. [Google Scholar] [CrossRef] [PubMed]
- Papaioannou, A.I.; Bartziokas, K.; Loukides, S.; Tsikrika, S.; Karakontaki, F.; Haniotou, A.; Papiris, S.; Stolz, D.; Kostikas, K. Cardiovascular comorbidities in hospitalised COPD patients: A determinant of future risk? Eur. Respir. J. 2015, 46, 846–849. [Google Scholar] [CrossRef]
- Terzano, C.; Conti, V.; Di Stefano, F.; Petroianni, A.; Ceccarelli, D.; Graziani, E.; Mariotta, S.; Ricci, A.; Vitarelli, A.; Puglisi, G.; et al. Comorbidity, hospitalization, and mortality in COPD: Results from a longitudinal study. Lung 2010, 188, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Agusti, A.; Edwards, L.D.; Rennard, S.I.; MacNee, W.; Tal-Singer, R.; Miller, B.E.; Vestbo, J.; Lomas, D.A.; Calverley, P.M.; Wouters, E.; et al. Persistent systemic inflammation is associated with poor clinical outcomes in COPD: A novel phenotype. PLoS ONE 2012, 7, e37483. [Google Scholar] [CrossRef]
- Goedemans, L.; Bax, J.J.; Delgado, V. COPD and acute myocardial infarction. Eur. Respir. Rev. 2020, 29, 190139. [Google Scholar] [CrossRef] [PubMed]
- Mojzisch, A.; Brehm, M.A. The Manifold Cellular Functions of von Willebrand Factor. Cells 2021, 10, 2351. [Google Scholar] [CrossRef]
- Manon-Jensen, T. Initiation of the wound healing cascade in inflammatory bowel disease: Assessment of Von Willebrand factor ADAMTS-13 processing and formation in crohn’s disease. EC Gastroenterol. Dig. Syst. 2019, 6, 143–154. [Google Scholar]
- Pimanda, J.; Hogg, P. Control of von Willebrand factor multimer size and implications for disease. Blood Rev. 2002, 16, 185–192. [Google Scholar] [CrossRef]
- Reardon, B.; Pasalic, L.; Favaloro, E.J. The Intriguing Relationships of von Willebrand Factor, ADAMTS13 and Cardiac Disease. J. Cardiovasc. Dev. Dis 2021, 8, 115. [Google Scholar] [CrossRef]
- Bartholo, T.P.; Costa, C.H.; Rufino, R. Evaluation of von Willebrand factor in COPD patients. J. Bras. Pneumol. 2014, 40, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Willeit, P.; Thompson, A.; Aspelund, T.; Rumley, A.; Eiriksdottir, G.; Lowe, G.; Gudnason, V.; Di Angelantonio, E. Hemostatic factors and risk of coronary heart disease in general populations: New prospective study and updated meta-analyses. PLoS ONE 2013, 8, e55175. [Google Scholar] [CrossRef] [PubMed]
- Sonneveld, M.A.; Franco, O.H.; Ikram, M.A.; Hofman, A.; Kavousi, M.; de Maat, M.P.; Leebeek, F.W. Von Willebrand Factor, ADAMTS13, and the Risk of Mortality: The Rotterdam Study. Arter. Thromb. Vasc. Biol. 2016, 36, 2446–2451. [Google Scholar] [CrossRef]
- Wieberdink, R.G.; van Schie, M.C.; Koudstaal, P.J.; Hofman, A.; Witteman, J.C.; de Maat, M.P.; Leebeek, F.W.; Breteler, M.M. High von Willebrand factor levels increase the risk of stroke: The Rotterdam study. Stroke 2010, 41, 2151–2156. [Google Scholar] [CrossRef] [PubMed]
- Sonneveld, M.A.; de Maat, M.P.; Portegies, M.L.; Kavousi, M.; Hofman, A.; Turecek, P.L.; Rottensteiner, H.; Scheiflinger, F.; Koudstaal, P.J.; Ikram, M.A.; et al. Low ADAMTS13 activity is associated with an increased risk of ischemic stroke. Blood 2015, 126, 2739–2746. [Google Scholar] [CrossRef] [PubMed]
- Gombos, T.; Mako, V.; Cervenak, L.; Papassotiriou, J.; Kunde, J.; Harsfalvi, J.; Forhecz, Z.; Pozsonyi, Z.; Borgulya, G.; Janoskuti, L.; et al. Levels of von Willebrand factor antigen and von Willebrand factor cleaving protease (ADAMTS13) activity predict clinical events in chronic heart failure. Thromb. Haemost. 2009, 102, 573–580. [Google Scholar] [CrossRef]
- Langholm, L.L.; Ronnow, S.R.; Sand, J.M.B.; Leeming, D.J.; Tal-Singer, R.; Miller, B.E.; Vestbo, J.; Karsdal, M.A.; Manon-Jensen, T. Increased von Willebrand Factor Processing in COPD, Reflecting Lung Epithelium Damage, Is Associated with Emphysema, Exacerbations and Elevated Mortality Risk. Int. J. Chronic Obs. Pulm. Dis. 2020, 15, 543–552. [Google Scholar] [CrossRef]
- Mosesson, M.W. Fibrinogen and fibrin structure and functions. J. Thromb. Haemost. 2005, 3, 1894–1904. [Google Scholar] [CrossRef]
- Manon-Jensen, T.; Langholm, L.L.; Rønnow, S.R.; Karsdal, M.A.; Tal-Singer, R.; Vestbo, J.; Leeming, D.J.; Miller, B.E.; Bülow Sand, J.M. End-product of fibrinogen is elevated in emphysematous chronic obstructive pulmonary disease and is predictive of mortality in the ECLIPSE cohort. Respir. Med. 2019, 160, 105814. [Google Scholar] [CrossRef]
- Kovacevic, K.D.; Mayer, F.J.; Jilma, B.; Buchtele, N.; Obermayer, G.; Binder, C.J.; Blann, A.D.; Minar, E.; Schillinger, M.; Hoke, M. Von Willebrand factor antigen levels predict major adverse cardiovascular events in patients with carotid stenosis of the ICARAS study. Atherosclerosis 2019, 290, 31–36. [Google Scholar] [CrossRef]
- Cushman, M.; Folsom, A.R.; Wang, L.; Aleksic, N.; Rosamond, W.D.; Tracy, R.P.; Heckbert, S.R. Fibrin fragment D-dimer and the risk of future venous thrombosis. Blood 2003, 101, 1243–1248. [Google Scholar] [CrossRef] [PubMed]
- Di Castelnuovo, A.; de Curtis, A.; Costanzo, S.; Persichillo, M.; Olivieri, M.; Zito, F.; Donati, M.B.; de Gaetano, G.; Iacoviello, L.; Investigators, M.-S.P. Association of D-dimer levels with all-cause mortality in a healthy adult population: Findings from the MOLI-SANI study. Haematologica 2013, 98, 1476–1480. [Google Scholar] [CrossRef] [PubMed]
- Hu, G.; Wu, Y.; Zhou, Y.; Wu, Z.; Wei, L.; Li, Y.; Peng, G.; Liang, W.; Ran, P. Prognostic role of D-dimer for in-hospital and 1-year mortality in exacerbations of COPD. Int. J. Chronic Obs. Pulm. Dis. 2016, 11, 2729–2736. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food & Drug Administration. Biomarkers Qualification Review for Plasma Fibrinogen. Available online: https://www.fda.gov/media/92567/download (accessed on 30 May 2022).
- Sivapalan, P.; Lapperre, T.S.; Janner, J.; Laub, R.R.; Moberg, M.; Bech, C.S.; Eklof, J.; Holm, F.S.; Armbruster, K.; Sivapalan, P.; et al. Eosinophil-guided corticosteroid therapy in patients admitted to hospital with COPD exacerbation (CORTICO-COP): A multicentre, randomised, controlled, open-label, non-inferiority trial. Lancet Respir. Med. 2019, 7, 699–709. [Google Scholar] [CrossRef]
- Sun, S.; Karsdal, M.A.; Mortensen, J.H.; Luo, Y.; Kjeldsen, J.; Krag, A.; Jensen, M.D.; Bay-Jensen, A.C.; Manon-Jensen, T. Serological Assessment of the Quality of Wound Healing Processes in Crohn’s Disease. J. Gastrointest. Liver Dis. 2019, 28, 175–182. [Google Scholar] [CrossRef]
- Von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gotzsche, P.C.; Vandenbroucke, J.P.; Initiative, S. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. J. Clin. Epidemiol. 2008, 61, 344–349. [Google Scholar] [CrossRef]
- Peter Kamstrup, J.M.B.S.; Ulrik, C.; Janner, J.; Rønnow, S.R.; Leeming, D.J.; Jensen, S.G.; Wilcke, T.; Miravitlles, M.; Mathioudakis, A.; Lapperre, T.; et al. Protocol: Biomarkers of Endothelial Damage in Patients with COPD. A Substudy of the CORTICO-COP Randomized Controlled Trial. Available online: http://coptrin.dk/wp-content/uploads/2022/01/cortico-endotheldamage-sign-protokol.pdf (accessed on 10 June 2022).
- Bartlett, J.W.; Seaman, S.R.; White, I.R.; Carpenter, J.R. Multiple imputation of covariates by fully conditional specification: Accommodating the substantive model. Stat. Methods Med. Res. 2015, 24, 462–487. [Google Scholar] [CrossRef]
| Total (n = 299) | MACE during Study (n = 149) | No MACE during Study (n = 150) | Missing Data | p-Value | ||
|---|---|---|---|---|---|---|
| MACE | No MACE | |||||
| Sex (male) sex, n (%) | 141 (45) | 68 (46) | 67 (45) | 0 | 0 | 0.87 |
| Age (years), mean (SD) | 75 (9) | 77 (9)) | 73 (9) | 0 | 0 | <0.01 |
| BMI (kg/m2), median (IQR) | 24 (20–27) | 23 (20–26) | 25 (22–28) | 1 | 2 | <0.01 |
| FEV1 (% of predicted), median (IQR) | 31 (23–40) | 29 (23–37) | 34 (24–46) | 9 | 10 | <0.01 |
| MRC, median (IQR) | 4 (3–5) | 4 (3–5) | 4 (3–4) | 2 | 3 | <0.01 |
| CRP (mg/L), median (IQR) | 10.0 (26.0–86.0) | 22.5 (11.0–68.0) | 32.0 (7.0–97.0) | 3 | 3 | 0.45 |
| Smoking status | ||||||
| Current smoker, n (%) | 100 (33) | 56 (19) | 44 (15) | 0 | 0 | 0.28 |
| Former smoker, n (%) | 194 (65) | 91 (30) | 103 (34) | 0 | 0 | |
| Never smoked, n (%) | 5 (2) | <5 (<2) | <5 (<2) | 0 | 0 | |
| Pack years (years) *, median (IQR) | 47 (30–57) | 50 (30–60) | 45 (30–56) | 5 | 2 | 0.51 |
| Medication | ||||||
| Inhaled long-acting muscarinic antagonist, n (%) | 234 (78) | 120 (81) | 114 (76) | 0 | 0 | 0.34 |
| Inhaled long-acting β2-agonist, n (%) | 238 (80) | 121 (79) | 117 (81) | 0 | 0 | 0.64 |
| Inhaled corticosteroid use, n (%) | 169 (55) | 83 (56) | 81 (54) | 0 | 0 | 0.77 |
| Long-term oral corticosteroid, n (%) | 20 (7) | 14 (9) | 6 (4) | 0 | 0 | 0.06 |
| Comorbidities | ||||||
| Previous MACE, n (%) | 129 (43) | 80 (54) | 49 (33) | 0 | 0 | <0.01 |
| Heart failure, n (%) | 81 (27) | 53 (36) | 28 (19) | 0 | 0 | <0.01 |
| Ischemic heart disease incl. stroke, n (%) | 88 (29) | 52 (35) | 36 (24) | 0 | 0 | 0.04 |
| Peripheral vascular disease, n (%) | 41 (14) | 27 (18) | 14 (9) | 0 | 0 | 0.03 |
| Type 2 diabetes mellitus, n (%) | 38 (13) | 24 (16) | 14 (9) | 0 | 0 | 0.08 |
| Hypercholesterolemia, n (%) | 37 (12) | 17 (11) | 20 (13) | 0 | 0 | 0.61 |
| Hypertension, n (%) | 117 (39) | 54 (36) | 63 (42) | 0 | 0 | 0.31 |
| Kidney failure, n (%) | 21 (7) | <21 (<11) | <5 (<2) | 0 | 0 | <0.01 |
| Atrial fibrillation or flutter, n (%) | 55 (18) | 38 (26) | 17 (11) | 0 | 0 | <0.01 |
| Outcome | X-FIB | VWF-N | VWF-A | ||||||
|---|---|---|---|---|---|---|---|---|---|
| High | Low | p-Value | High | Low | p-Value | High | Low | p-Value | |
| MACE, n (%) | 38 (51) | 111 (50) | 0.87 | 45 (60) | 104 (46) | 0.04 | 35 (47) | 114 (51) | 0.53 |
| Myocardial infarction, n (%) | 0 (0) | 7 (3) | 0.19 | 2 (3) | 5 (2) | 1.00 | 3 (4) | 4 (2) | 0.37 |
| Stroke, n (%) | 0 (0) | 1 (<1) | 1.00 | 1 (1) | 0 (0) | 0.25 | 1 (1) | 0 (0) | 0.25 |
| Worsening of heart failure, n (%) | 8 (11) | 13 (6) | 0.19 | 6 (8) | 15 (7) | 0.79 | 1 (1) | 20 (9) | 0.03 |
| Death, n (%) | 30 (40) | 90 (40) | 1.00 | 36 (48) | 84 (38) | 0.13 | 30 (40) | 90 (40) | 1.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kamstrup, P.; Sand, J.M.B.; Ulrik, C.S.; Janner, J.; Rønn, C.P.; Rønnow, S.R.; Leeming, D.J.; Jensen, S.G.; Wilcke, T.; Mathioudakis, A.G.; et al. Biomarkers of Clot Activation and Degradation and Risk of Future Major Cardiovascular Events in Acute Exacerbation of COPD: A Cohort Sub-Study in a Randomized Trial Population. Biomedicines 2022, 10, 2011. https://doi.org/10.3390/biomedicines10082011
Kamstrup P, Sand JMB, Ulrik CS, Janner J, Rønn CP, Rønnow SR, Leeming DJ, Jensen SG, Wilcke T, Mathioudakis AG, et al. Biomarkers of Clot Activation and Degradation and Risk of Future Major Cardiovascular Events in Acute Exacerbation of COPD: A Cohort Sub-Study in a Randomized Trial Population. Biomedicines. 2022; 10(8):2011. https://doi.org/10.3390/biomedicines10082011
Chicago/Turabian StyleKamstrup, Peter, Jannie Marie Bülow Sand, Charlotte Suppli Ulrik, Julie Janner, Christian Philip Rønn, Sarah Rank Rønnow, Diana Julie Leeming, Sidse Graff Jensen, Torgny Wilcke, Alexander G. Mathioudakis, and et al. 2022. "Biomarkers of Clot Activation and Degradation and Risk of Future Major Cardiovascular Events in Acute Exacerbation of COPD: A Cohort Sub-Study in a Randomized Trial Population" Biomedicines 10, no. 8: 2011. https://doi.org/10.3390/biomedicines10082011
APA StyleKamstrup, P., Sand, J. M. B., Ulrik, C. S., Janner, J., Rønn, C. P., Rønnow, S. R., Leeming, D. J., Jensen, S. G., Wilcke, T., Mathioudakis, A. G., Miravitlles, M., Lapperre, T., Bendstrup, E., Frikke-Schmidt, R., Murray, D. D., Itenov, T., Bossios, A., Nielsen, S. D., Vestbo, J., ... Sivapalan, P. (2022). Biomarkers of Clot Activation and Degradation and Risk of Future Major Cardiovascular Events in Acute Exacerbation of COPD: A Cohort Sub-Study in a Randomized Trial Population. Biomedicines, 10(8), 2011. https://doi.org/10.3390/biomedicines10082011










