Urinary Exosomal Cystatin C and Lipopolysaccharide Binding Protein as Biomarkers for Antibody−Mediated Rejection after Kidney Transplantation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Histopathology
2.3. Urine Processing and Exosome Extraction
2.4. Sample Preparation for Proteome Analysis
2.5. Nano-LC-ESI-MS/MS Analysis
2.6. Database Searching and Label Free Quantitation
2.7. Biomarker Candidate Selection
2.8. Evaluation of Exosomes
2.9. Western Blot and Immunohistochemistry
2.10. Statistical Analysis
3. Results
3.1. Patient Characteristics
3.2. Urine Exosome Characterization and Exosomal Proteins
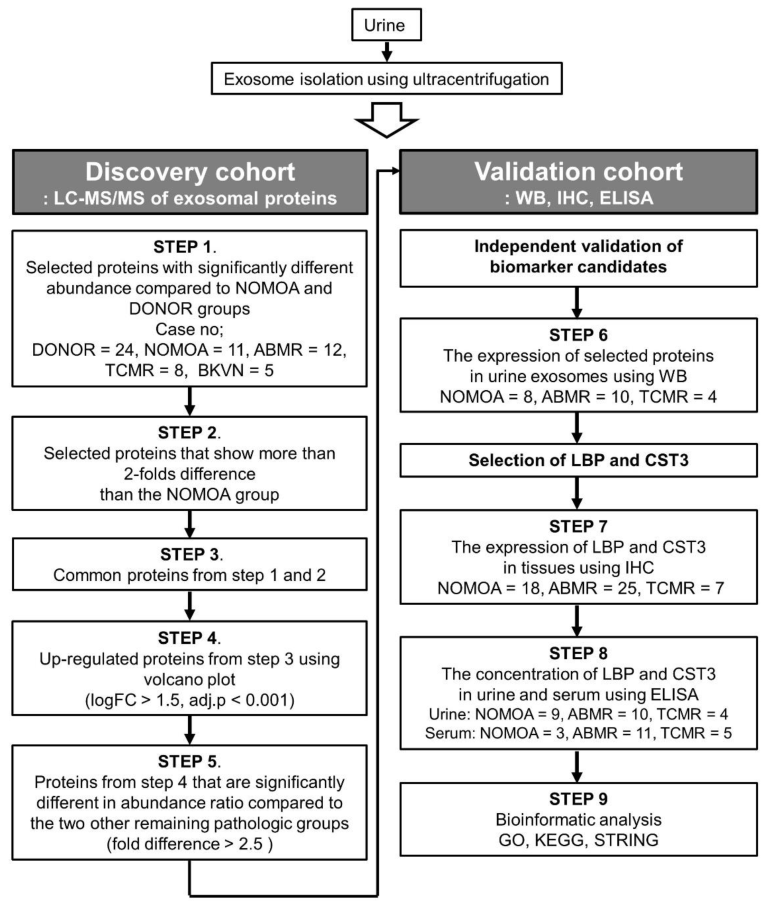
3.3. Discovery of Four Biomarkers for Discriminating Antibody-Mediated Rejection
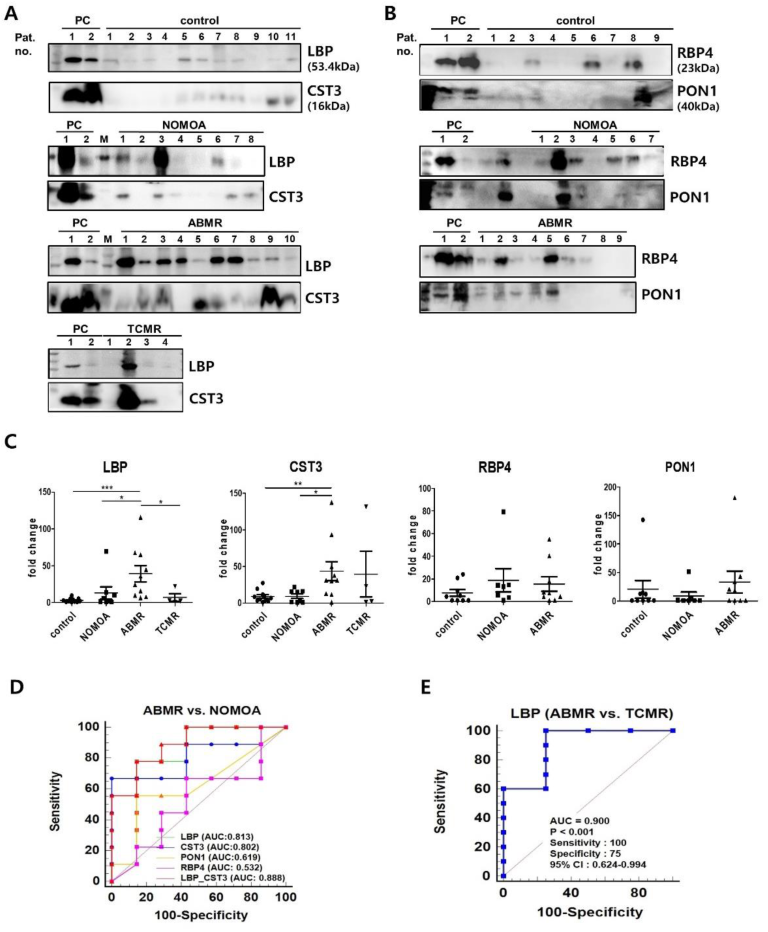
3.4. Validation of the Four Biomarkers for Discriminating Antibody−Mediated Rejection
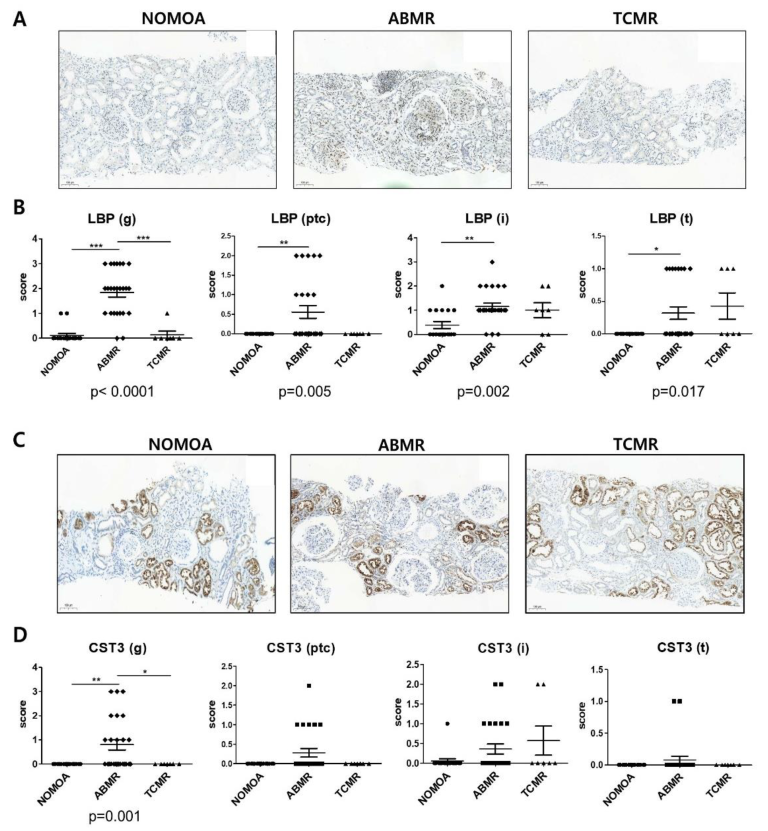
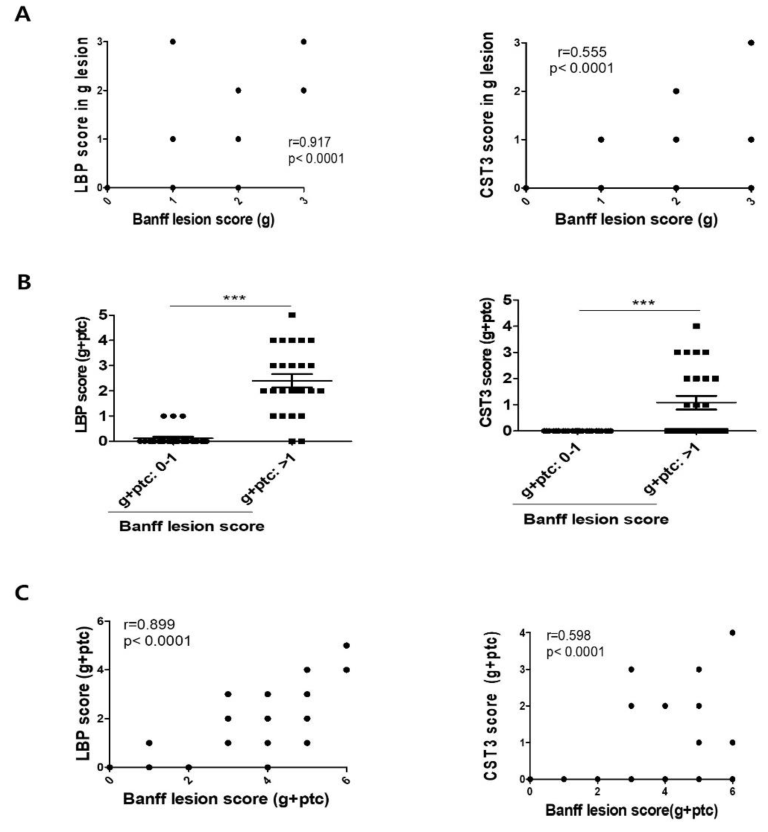
3.5. Comparison of LBP and CST3 Expression in Kidney Allograft Tissue According to Pathologic Groups
3.6. Levels of LBP, CST3, and RBP4 in the Urine and Serum
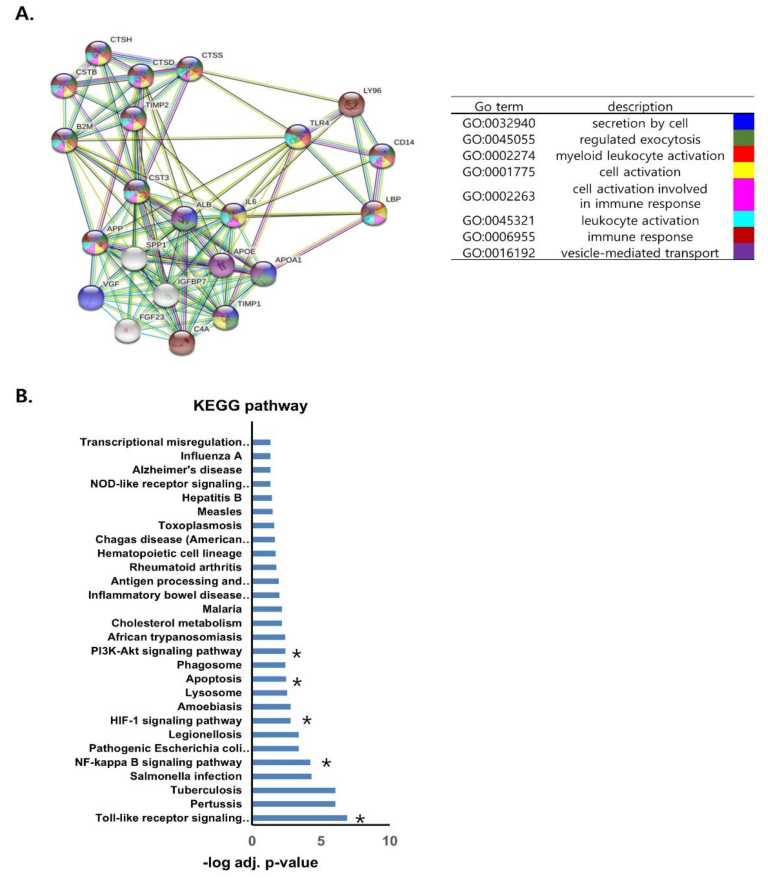
3.7. Protein–Protein Interaction Analysis of LBP and CST3
3.8. Combined Use of LBP and CST3 as Biomarkers for ABMR
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Patents
References
- Treatment of end-stage organ failure in Canada. Healthc. Q. 2012, 15, 84. [CrossRef]
- Halloran, P.F. Immunosuppressive drugs for kidney transplantation. N. Engl. J. Med. 2004, 351, 2715–2729. [Google Scholar] [CrossRef]
- Keown, P.A. New immunosuppressive strategies. Curr. Opin. Nephrol. Hypertens. 1998, 7, 659–663. [Google Scholar] [CrossRef]
- Wu, O.; Levy, A.R.; Briggs, A.; Lewis, G.; Jardine, A. Acute rejection and chronic nephropathy: A systematic review of the literature. Transplantation 2009, 87, 1330–1339. [Google Scholar] [CrossRef] [PubMed]
- Anglicheau, D.; Suthanthiran, M. Noninvasive prediction of organ graft rejection and outcome using gene expression patterns. Transplantation 2008, 86, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Einecke, G.; Sis, B.; Reeve, J.; Mengel, M.; Campbell, P.M.; Hidalgo, L.G.; Kaplan, B.; Halloran, P.F. Antibody-mediated microcirculation injury is the major cause of late kidney transplant failure. Am. J. Transplant. 2009, 9, 2520–2531. [Google Scholar] [CrossRef] [PubMed]
- Sellares, J.; de Freitas, D.G.; Mengel, M.; Reeve, J.; Einecke, G.; Sis, B.; Hidalgo, L.G.; Famulski, K.; Matas, A.; Halloran, P.F. Understanding the causes of kidney transplant failure: The dominant role of antibody-mediated rejection and nonadherence. Am. J. Transplant. 2012, 12, 388–399. [Google Scholar] [CrossRef] [PubMed]
- El-Zoghby, Z.M.; Stegall, M.D.; Lager, D.J.; Kremers, W.K.; Amer, H.; Gloor, J.M.; Cosio, F.G. Identifying specific causes of kidney allograft loss. Am. J. Transplant. 2009, 9, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Naesens, M.; Kuypers, D.R.; De Vusser, K.; Evenepoel, P.; Claes, K.; Bammens, B.; Meijers, B.; Sprangers, B.; Pirenne, J.; Monbaliu, D.; et al. The histology of kidney transplant failure: A long-term follow-up study. Transplantation 2014, 98, 427–435. [Google Scholar] [CrossRef]
- Habal, M.V.; Farr, M.; Restaino, S.; Chong, A. Desensitization in the era of precision medicine: Moving from the bench to bedside. Transplantation 2019, 103, 1574–1581. [Google Scholar] [CrossRef]
- Alvarez, M.L.; Khosroheidari, M.; Kanchi Ravi, R.; DiStefano, J.K. Comparison of protein, microRNA, and mRNA yields using different methods of urinary exosome isolation for the discovery of kidney disease biomarkers. Kidney Int. 2012, 82, 1024–1032. [Google Scholar] [CrossRef]
- Gurung, S.; Perocheau, D.; Touramanidou, L.; Baruteau, J. The exosome journey: From biogenesis to uptake and intracellular signaling. Cell Commun. Signal. 2021, 19, 47. [Google Scholar] [CrossRef]
- Pegtel, D.M.; Gould, S.J. Exosomes. Annu. Rev. Biochem. 2019, 88, 487–514. [Google Scholar] [CrossRef]
- El Fekih, R.; Hurley, J.; Tadigotla, V.; Alghamdi, A.; Srivastava, A.; Coticchia, C.; Choi, J.; Allos, H.; Yatim, K.; Alhaddad, J.; et al. Discovery and validation of a urinary exosome mRNA signature for the diagnosis of human kidney transplant rejection. J. Am. Soc. Nephrol. 2021, 32, 994–1004. [Google Scholar] [CrossRef]
- Golebiewska, J.E.; Wardowska, A.; Pietrowska, M.; Wojakowska, A.; Dębska-Ślizień, A. Small Extracellular Vesicles in Transplant Rejection. Cells 2021, 10, 2989. [Google Scholar] [CrossRef]
- Marino, J.; Paster, J.; Benichou, G. Allorecognition by T Lymphocytes and Allograft Rejection. Front. Immunol. 2016, 7, 582. [Google Scholar] [CrossRef]
- Mastoridis, S.; Martinez-Llordella, M.; Sanchez-Fueyo, A. Extracellular vesicles as mediators of alloimmunity and their therapeutic potential in liver transplantation. World J. Transplant. 2020, 10, 330–344. [Google Scholar] [CrossRef]
- Sigdel, T.K.; Ng, Y.W.; Lee, S.; Nicora, C.D.; Qian, W.-J.; Smith, R.D.; Camp, D.G.; Sarwal, M.M. Perturbations in the urinary exosome in transplant rejection. Front. Med. 2015, 1, 57. [Google Scholar] [CrossRef]
- Go, H.; Shin, S.; Kim, Y.H.; Han, D.J.; Cho, Y.M. Refinement of the criteria for ultrastructural peritubular capillary basement mem brane multilayering in the diagnosis of chronic active/acute antibody-mediated rejection. Transpl. Int. 2017, 30, 398–409. [Google Scholar] [CrossRef]
- Loupy, A.; Haas, M.; Solez, K.; Racusen, L.; Glotz, D.; Seron, D.; Nankivell, B.J.; Colvin, R.B.; Afrouzian, M.; Akalin, E.; et al. The Banff 2015 kidney meeting report: Current challenges in rejection classifica tion and prospects for adopting molecular pathology. Am. J. Transplant. 2017, 17, 28–41. [Google Scholar] [CrossRef]
- Pisitkun, T.; Johnstone, R.; Knepper, M.A. Discovery of urinary biomarkers. Mol. Cell Proteom. 2006, 5, 1760–1771. [Google Scholar] [CrossRef] [PubMed]
- Pellerin, D.; Gagnon, H.; Dubé, J.; Corbin, F. Amicon-adapted enhanced FASP: An in-solution digestion-based alternative sample preparation method to FASP. F1000 Res. 2015, 4, 140. [Google Scholar] [CrossRef]
- Kim, E.Y.; Ahn, H.S.; Lee, M.Y.; Yu, J.; Yeom, J.; Jeong, H.; Min, H.; Lee, H.J.; Kim, K.; Ahn, Y.M. An exploratory pilot study with plasma protein signatures associated with response of patients with depression to antidepressant treatment for 10 weeks. Biomedicines 2020, 8, 455. [Google Scholar] [CrossRef] [PubMed]
- Ahn, H.S.; Yeom, J.; Yu, J.; Kwon, Y.-I.; Kim, J.-H.; Kim, K. Convergence of Plasma Metabolomics and Proteomics Analysis to Discover Signatures of High-Grade Serous Ovarian Cancer. Cancers 2020, 12, 3447. [Google Scholar] [CrossRef]
- Kall, L.; Canterbury, J.D.; Weston, J.; Noble, W.S.; MacCoss, M.J. Semi-supervised learning for peptide identification from shotgun proteomics datasets. Nat. Methods 2007, 4, 923–925. [Google Scholar] [CrossRef]
- Jung, M.K.; Mun, J.Y. Sample preparation and imaging of exosomes by transmission electron microscopy. J. Vis. Exp. 2018, 131, e56482. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, X.; Zheng, X.; Lan, Z.; Shi, J.; Jiang, J.; Zwiep, T.; Li, Q.; Quan, D.; Zhang, Z.-X.; et al. Prevention of allograft rejection in heart transplantation through concurrent gene silencing of TLR and Kinase signaling pathways. Sci. Rep. 2016, 23, 33869. [Google Scholar] [CrossRef]
- Molinero, L.L.; Alegre, M.L. Role of T cell-nuclear factor kappaB in transplantation. Transpl. Rev. 2012, 26, 189–200. [Google Scholar] [CrossRef]
- Yu, T.; Wen, M.; Li, C.; Cheng, C.-H.; Wu, M.-J.; Chen, C.-H.; Shu, K.-H. Expression of hypoxia-inducible factor-1α (HIF-1α) in infiltrating inflammatory cells is associated with chronic allograft dysfunction and predicts long-term graft survival. Nephrol. Dial. Transplant. 2013, 28, 659–670. [Google Scholar] [CrossRef]
- Cardinal, H.; Dieudé, M.; Hébert, M.J. Endothelial Dysfunction in Kidney Transplantation. Front. Immunol. 2018, 9, 1130. [Google Scholar] [CrossRef] [Green Version]
- Yang, C.; Chen, X.; Wei, Z. Targeting the class IA PI3K isoforms p110alpha/delta attenuates heart allograft rejection in mice by suppressing the CD4(+) T lymphocyte response. Am. J. Transl. Res. 2018, 10, 1387–1399. [Google Scholar]
- Nankivell, B.J.; Agrawal, N.; Sharma, A.; Taverniti, A.; P’Ng, C.H.; Shingde, M.; Wong, G.; Chapman, J.R. The clinical and pathological significance of borderline T cell-mediated rejection. Am. J. Transplant. 2019, 19, 1452–1463. [Google Scholar] [CrossRef]
- Meng, L.; Song, Z.; Liu, A.; Dahmen, U.; Yang, X.; Fang, H. Effects of lipopolysaccharide-binding protein (LBP) single nucleotide polymorphism (SNP) in infections, inflammatory diseases, metabolic disorders and cancers. Front. Immunol. 2021, 12, 681810. [Google Scholar] [CrossRef]
- Ryu, J.K.; Kim, S.J.; Rah, S.H.; Kang, J.I.; Jung, H.E.; Lee, D.; Lee, H.K.; Lee, J.-O.; Park, B.S.; Yoon, T.-Y.; et al. Reconstruction of LPS transfer cascade reveals structural determinants within LBP, CD14, and TLR4-MD2 for efficient LPS recognition and transfer. Immunity 2017, 46, 38–50. [Google Scholar] [CrossRef]
- Guha, M.; Mackman, N. LPS induction of gene expression in human monocytes. Cell Signal. 2001, 13, 85–94. [Google Scholar] [CrossRef]
- Gerlach, U.A.; Klopfel, M.; Atanasov, G.; Polenz, D.; Vogt, K.; Ahrlich, S.; Marksteiner, M.; Jurisch, A.; Loddenkemper, C.; Reutzel-Selke, A.; et al. Intragraft and systemic immune parameters discriminating between rejection and long-term graft function in a preclinical model of intestinal transplantation. Transplantation 2017, 101, 1036–1045. [Google Scholar] [CrossRef]
- Freue, G.V.; Sasaki, M.; Meredith, A.; Günther, O.P.; Bergman, A.; Takhar, M.; Mui, A.; Balshaw, R.F.; Ng, R.T.; Opushneva, N.; et al. Proteomic signatures in plasma during early acute renal allograft rejection. Mol. Cell. Proteom. 2010, 9, 1954–1967. [Google Scholar] [CrossRef]
- Grubb, A.O. Cystatin C: Properties and use as diagnostic marker. Adv. Clin. Chem. 2000, 35, 63–99. [Google Scholar]
- Hall, I.E.; Koyner, J.L.; Doshi, M.D.; Marcus, R.J.; Parikh, C.R. Urine cystatin C as a biomarker of proximal tubular function immediately after kidney transplantation. Am. J. Nephrol. 2011, 33, 407–413. [Google Scholar] [CrossRef]
- Spanu, S.; van Roeyen, C.R.; Denecke, B.; Floege, J.; Mühlfeld, A.S. Urinary exosomes: A novel means to non-invasively assess changes in renal gene and protein expression. PLoS ONE 2014, 9, e109631. [Google Scholar] [CrossRef]
- Gwak, H.; Park, S.; Yu, H.; Hyun, K.-A.; Jung, H.-I. A modular microfluidic platform for serial enrichment and harvest of pure extracellular vesicles. Analyst 2022, 147, 1117–1127. [Google Scholar] [CrossRef]


| Discovery Cohort | Validation Cohort | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| ABMR | BKVN | TCMR | NOMOA | DONOR | p | ABMR | TCMR | NOMOA | Control | p | |
| (N = 12) | (N = 5) | (N = 8) | (N = 10) | (N = 24) | (N = 25) | (N = 10) | (N = 19) | (N = 25) | |||
| Age (years) | 52.2 ± 13.6 | 47.2 ± 8.6 | 53.8 ± 12.7 | 48.4 ± 10.2 | 38.5 ± 7.9 | 0.002 | 46.0 ± 10.8 | 51.0 ± 10.9 | 51.0 ± 12.6 | 50.6 ± 9.7 | 0.37 |
| Female sex | 5 (41.7) | 1 (20.0) | 1 (12.5) | 4 (40.0) | 13 (54.2) | 0.326 | 7 (28.0) | 3 (30.0) | 4 (21.1) | 13 (52.0) | 0.011 |
| BMI (kg/m2) | 22.2 ± 2.6 | 23.7 ± 2.2 | 25.4 ± 4.3 | 21.6 ± 3.4 | 25.9 ± 3.8 | 0.011 | 22.0 ± 2.7 | 24.7 ± 2.7 | 22.7 ± 2.8 | 21.9 ± 3.0 | 0.351 |
| Hypertension | 1 (8.3) | 0 | 2 (25.0) | 0 | 2 (8.0) | 0 | 1 (5.3) | 4 (16.0) | |||
| Diabetes | 1 (8.3) | 1 (20.0) | 2 (25.0) | 4 (40.0) | 4 (16.0) | 5 (50.0) | 6 (31.6) | 3 (12.0) | |||
| Glomerulonephritis | 1 (8.3) | 0 | 0 | 1 (10.0) | 5 (20.0) | 0 | 3 (15.8) | 6 (24.0) | |||
| IgA nephropathy | 0 | 1 (20.0) | 1 (12.5) | 1 (10.0) | 3 (12.0) | 2 (20.0) | 1 (5.3) | 2 (8.0) | |||
| FSGS | 0 | 0 | 0 | 1 (10.0) | 0 | 0 | 1 (5.3) | 0 | |||
| PCKD | 0 | 0 | 1 (12.5) | 0 | 0 | 0 | 2 (10.5) | 1 (4.0) | |||
| Unknown | 6 (50.0) | 3 (60.0) | 1 (12.5) | 3 (30.0) | 9 (36.0) | 2 (20.0) | 5 (26.3) | 8 (32.0) | |||
| Others | 3 (25.0) | 1 (20.0) | 0 | 2 (20.0) | 2 (8.0) | 1 (10.0) | 0 | 1 (4.0) | |||
| Serum creatinine (mg/dL) | 3.25 ± 1.41 | 4.37 ± 2.80 | 1.61 ± 0.38 | 2.33 ± 1.12 | 0.005 | 2.01 ± 1.08 | 2.45 ± 1.31 | 1.979 ± 0.75 | 0.508 | ||
| Albumin: creatinine ratio | 1597 ± 1548 | 426.3 ± 643.3 | 290.7 ± 329.4 | 2816 ± 2170 | 0.059 | 1635 ± 1436 | 1422 ± 1737 | 732.6 ± 1027 | 0.236 | ||
| Pre-emptive KT | 2 (16.7) | 1 (20.0) | 0 | 2 (20.0) | 0.594 | 2 (8.0) | 1 (10.0) | 1 (5.3) | 7 (28.0) | 0.05 | |
| Pre-transplant dialysis (month) | 33.1 ± 40.2 | 30.0 ± 20.3 | 62.8 ± 71.6 | 48.4 ± 52.8 | 0.972 | 38.2 ± 54.0 | 49.4 ± 44.7 | 37.3 ± 44.3 | 36.2 ± 55.5 | 0.559 | |
| KT to biopsy (month) | 182.8 ± 45.2 | 37.4 ± 62.1 | 64.3 ± 80.5 | 76.2 ± 67.9 | 0.003 | 105.3 ± 60.9 | 51.5 ± 59.1 | 68.9 ± 77.0 | 42.0 ± 36.5 | 0.002 | |
| Induction | 0.025 | 0.046 | |||||||||
| None | 10 (83.3) | 0 | 2 (25.0) | 3 (30.0) | 6 (24.0) | 2 (20.0) | 3 (15.8) | 1 (4.0) | |||
| ATG | 1 (8.3) | 0 | 1 (12.5) | 2 (20.0) | 3 (12.0) | 1 (10.0) | 0 | 1 (4.0) | |||
| Simulect | 1 (8.3) | 5 (100.0) | 5 (62.5) | 5 (50.0) | 13 (52.0) | 7 (70.0) | 15 (78.9) | 23 (92.0) | |||
| Calcineurin inhibitor | 0.318 | 0.112 | |||||||||
| Tacrolimus | 2 (16.7) | 5 (100.0) | 2 (25.0) | 5 (50.0) | 17 (68.0) | 4 (40.0) | 13 (68.4) | 19 (76.0) | |||
| Cyclosporine | 10 (83.3) | 0 | 6 (75.0) | 5 (50.0) | 8 (32.0) | 6 (60.0) | 6 (31.6) | 6 (24.0) | |||
| Steroid maintenance | 12 (100.0) | 5 (100.0) | 7 (87.5) | 8 (80.0) | 0.078 | 25 (100.0) | 10 (100.0) | 19 (100.0) | 24 (96.0) | 0.08 | |
| A/B/DR HLA mismatch | 3.3 ± 1.5 | 3.2 ± 1.9 | 2.9 ± 2.0 | 3.1 ± 1.5 | 0.903 | 3.0 ± 1.5 | 3.7 ± 2.0 | 2.9 ± 1.6 | 3.0 ± 1.8 | 0.912 | |
| ABO-incompatible KT | 1 (8.3) | 1 (20.0) | 0 | 0 | 0.241 | 3 (12.0) | 3 (30.0) | 5 (26.3) | 0 | 0.009 | |
| DDKT | 2 (16.7) | 2 (40.0) | 3 (37.5) | 3 (30.0) | 0.077 | 8 (32.0) | 3 (30.0) | 6 (31.6) | 3 (12.0) | 0.41 | |
| DSA detection | 6 (50) | 1 (20) | 3 (37.5) | 2 (25%) | 15 (60) | 4 (40) | 4 (21.05) | ||||
| PRAI | 18.67 ± 29.68 | 1.800 ± 2.68 | 11.00 ± 13.85 | 7.625 ± 8.93 | 0.557 | 30.32 ± 31.49 | 30.60 ± 36.98 | 7.84 ± 20.06 | 0.063 | ||
| PRAII | 43.42 ± 35.70 | 26.00 ± 35.80 | 41.63 ± 37.95 | 5.875 ± 15.82 | 0.144 | 65.32 ± 32.99 | 54.20 ± 39.77 | 25.16 ± 31.54 | 0.557 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, M.J.; Lim, S.J.; Ko, Y.; Kwon, H.E.; Jung, J.H.; Kwon, H.; Go, H.; Park, Y.; Kim, T.-K.; Jung, M.; et al. Urinary Exosomal Cystatin C and Lipopolysaccharide Binding Protein as Biomarkers for Antibody−Mediated Rejection after Kidney Transplantation. Biomedicines 2022, 10, 2346. https://doi.org/10.3390/biomedicines10102346
Kim MJ, Lim SJ, Ko Y, Kwon HE, Jung JH, Kwon H, Go H, Park Y, Kim T-K, Jung M, et al. Urinary Exosomal Cystatin C and Lipopolysaccharide Binding Protein as Biomarkers for Antibody−Mediated Rejection after Kidney Transplantation. Biomedicines. 2022; 10(10):2346. https://doi.org/10.3390/biomedicines10102346
Chicago/Turabian StyleKim, Mi Joung, Seong Jun Lim, Youngmin Ko, Hye Eun Kwon, Joo Hee Jung, Hyunwook Kwon, Heounjeong Go, Yangsoon Park, Tae-Keun Kim, MinKyo Jung, and et al. 2022. "Urinary Exosomal Cystatin C and Lipopolysaccharide Binding Protein as Biomarkers for Antibody−Mediated Rejection after Kidney Transplantation" Biomedicines 10, no. 10: 2346. https://doi.org/10.3390/biomedicines10102346







