Diagnostic Blood Biomarkers in Alzheimer’s Disease
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. MRI Acquisition and Processing
2.3. β- Amyloid PET Imaging and Processing
2.4. CSF ATN Biomarker Analysis
2.5. Plasma NFL and Aβ1–42 Analysis
2.6. Statistical Analyses
2.7. Data Availability
3. Results
3.1. Fluid Biomarker Concentrations and Demographic Data
3.2. Severe Brain Atrophy in Participants with AD Dementia
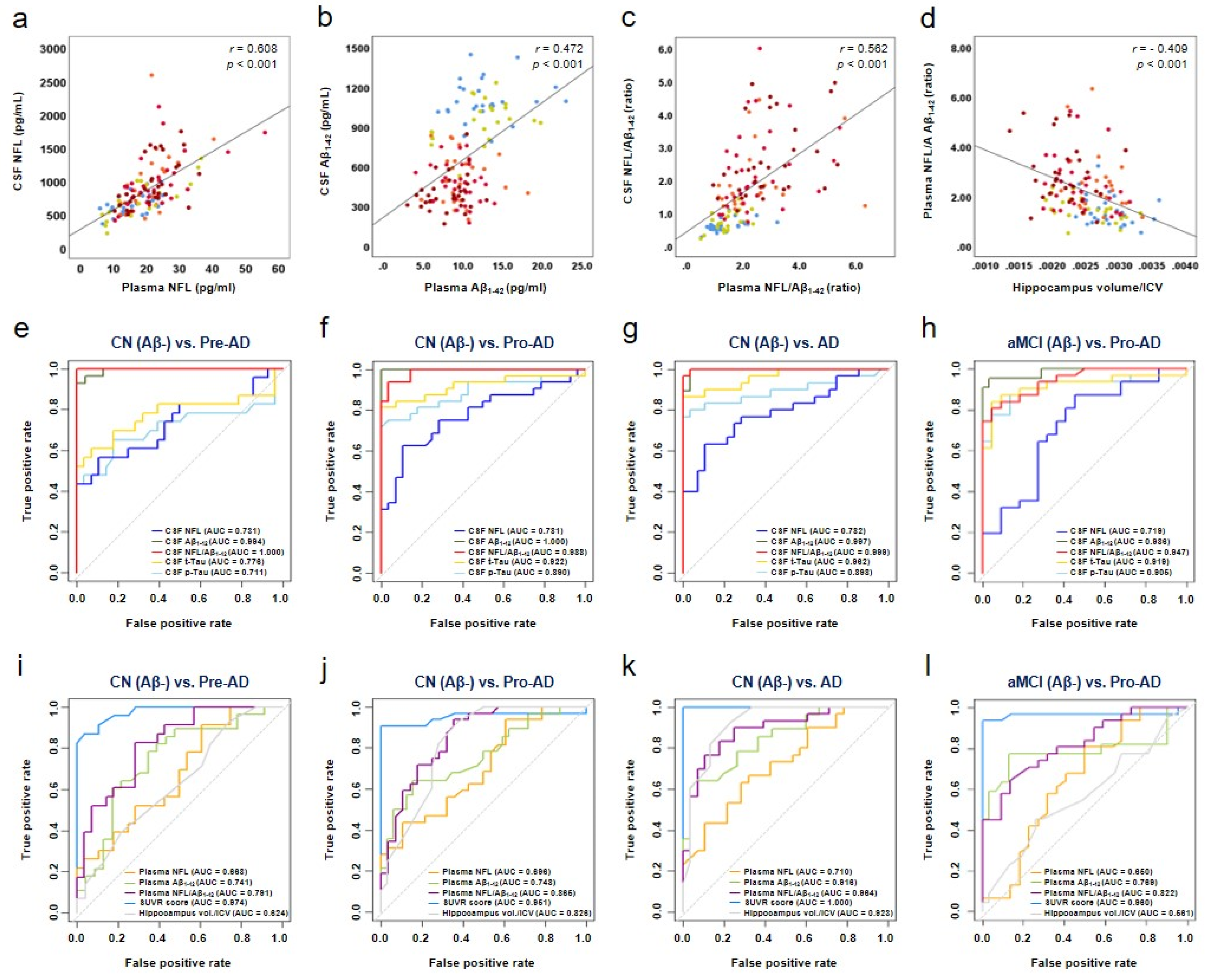
3.3. Association of Plasma NFL/Aβ1–42 with CSF NFL/Aβ1–42 and Hippocampal Volume/ICV in AD
3.4. Diagnostic Accuracy of Plasma NFL/Aβ1–42
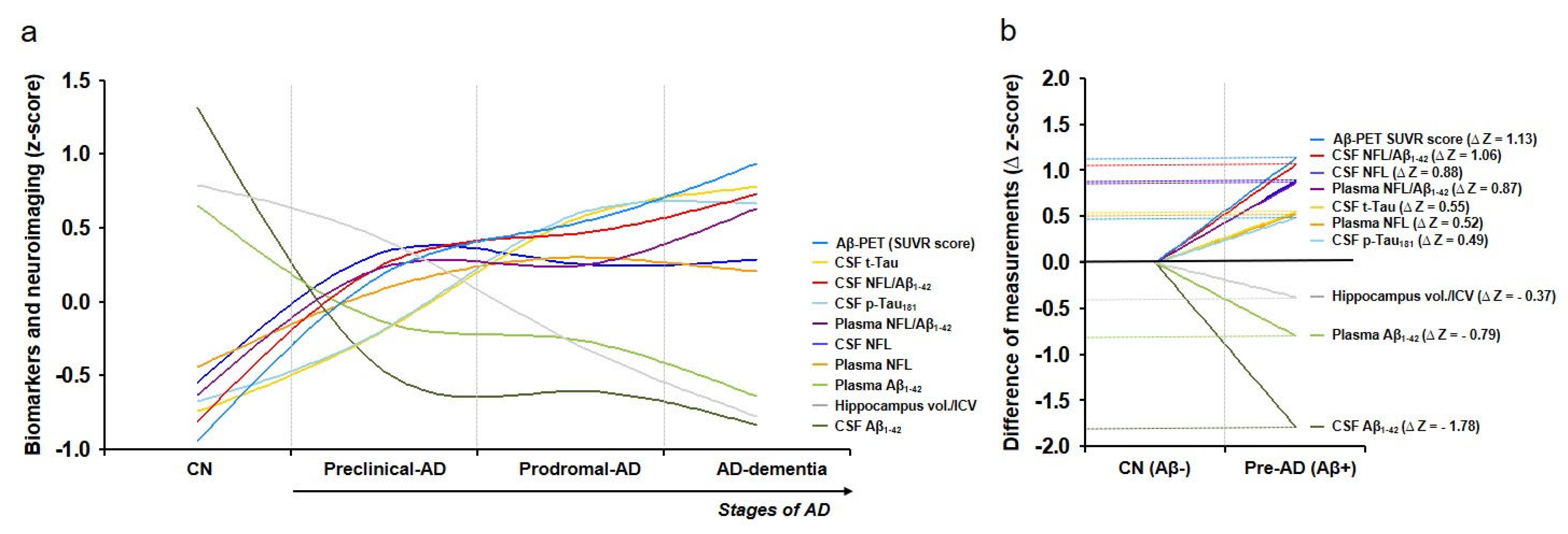
3.5. Dynamics of Biomarkers and Neuroimaging in AD
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bureau USC. An Aging World: 2020. Available online: https://mtgis-portal.geo.census.gov/arcgis/apps/MapSeries/index.html?appid=3d832796999042daae7982ff36835e2e (accessed on 1 March 2021).
- Bronzuoli, M.R.; Iacomino, A.; Steardo, L.; Scuderi, C. Targeting neuroinflammation in Alzheimer’s disease. J. Inflamm. Res. 2016, 9, 199–208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ballard, C.; Gauthier, S.; Corbett, A.; Brayne, C.; Aarsland, D.; Jones, E. Alzheimer’s disease. Lancet 2011, 377, 1019–1031. [Google Scholar] [CrossRef]
- Duong, S.; Patel, T.; Chang, F. Dementia: What pharmacists need to know. Can. Pharm. J. 2017, 150, 118–129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, L.K.; Chao, S.P.; Hu, C.J. Clinical trials of new drugs for Alzheimer disease. J. Biomed. Sci. 2020, 27, 18. [Google Scholar] [CrossRef]
- Van Oostveen, W.M.; de Lange, E.C.M. Imaging techniques in Alzheimer’s disease: A review of applications in early diagnosis and longitudinal monitoring. Int. J. Mol. Sci. 2021, 22, 2110. [Google Scholar]
- Blennow, K. A review of fluid biomarkers for Alzheimer’s disease: Moving from CSF to blood. Neurol. Ther. 2017, 6, 15–24. [Google Scholar] [CrossRef]
- Niemantsverdriet, E.; Valckx, S.; Bjerke, M.; Engelborghs, S. Alzheimer’s disease CSF biomarkers: Clinical indications and rational use. Acta Neurologica Belgica 2017, 117, 591–602. [Google Scholar] [CrossRef] [Green Version]
- Karantali, E.; Kazis, D.; Chatzikonstantinou, S.; Petridis, F.; Mavroudis, I. The role of neurofilament light chain in frontotemporal dementia: A meta-analysis. Aging Clin. Exp. Res. 2021, 33, 869–881. [Google Scholar] [CrossRef]
- Kang, M.S.; Aliaga, A.A.; Shin, M.; Mathotaarachchi, S.; Benedet, A.L.; Pascoal, T.A.; Therriault, J.; Chamoun, M.; Savard, M.; Devenyi, G.A.; et al. Alzheimer’s disease neuroimaging I. Amyloid-beta modulates the association between neurofilament light chain and brain atrophy in Alzheimer’s disease. Mol. Psychiatry 2020, 1–13. [Google Scholar]
- De Wolf, F.; Ghanbari, M.; Licher, S.; McRae-McKee, K.; Gras, L.; Weverling, G.J.; Wermeling, P.; Sedaghat, S.; Ikram, M.K.; Waziry, R.; et al. Plasma tau, neurofilament light chain and amyloid-beta levels and risk of dementia; A population-based cohort study. Brain 2020, 143, 1220–1232. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mielke, M.M.; Syrjanen, J.A.; Blennow, K.; Zetterberg, H.; Vemuri, P.; Skoog, I.; Machulda, M.M.; Kremers, W.K.; Knopman, D.S.; Jack, C.; et al. Plasma and CSF neurofilament light: Relation to longitudinal neuroimaging and cognitive measures. Neurology 2019, 93, e252–e260. [Google Scholar] [CrossRef] [Green Version]
- Jack, C.R., Jr.; Bennett, D.A.; Blennow, K.; Carrillo, M.C.; Feldman, H.H.; Frisoni, G.B.; Hampel, H.; Jagust, W.J.; Johnson, K.A.; Knopman, D.S.; et al. A/T/N: An unbiased descriptive classification scheme for Alzheimer disease biomarkers. Neurology 2016, 87, 539–547. [Google Scholar] [CrossRef]
- Bacioglu, M.; Maia, L.F.; Preische, O.; Schelle, J.; Apel, A.; Kaeser, S.A.; Schweighauser, M.; Eninger, T.; Lambert, M.; Pilotto, A.; et al. Neurofilament light chain in blood and CSF as marker of disease progression in mouse models and in neurodegenerative diseases. Neuron 2016, 91, 56–66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andersson, E.; Janelidze, S.; Lampinen, B.; Nilsson, M.; Leuzy, A.; Stomrud, E.; Blennow, K.; Zetterberg, H.; Hansson, O. Blood and cerebrospinal fluid neurofilament light differentially detect neurodegeneration in early Alzheimer’s disease. Neurobiol. Aging 2020, 95, 143–153. [Google Scholar] [CrossRef]
- Preische, O.; Schultz, S.A.; Apel, A.; Kuhle, J.; Kaeser, S.A.; Barro, C.; Graber, S.; Kuder-Buletta, E.; LaFougere, C.; Laske, C.; et al. Dominantly inherited Alzheimer network. Serum neurofilament dynamics predicts neurodegeneration and clinical progression in presymptomatic Alzheimer’s disease. Nat. Med. 2019, 25, 277–283. [Google Scholar] [CrossRef]
- Gisslen, M.; Price, R.W.; Andreasson, U.; Norgren, N.; Nilsson, S.; Hagberg, L.; Fuchs, D.; Spudich, S.; Blennow, K.; Zetterberg, H. Plasma concentration of the neurofilament light protein (NFL) is a biomarker of CNS injury in HIV infection: A cross-sectional study. EBioMedicine 2016, 3, 135–140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Osborn, K.E.; Khan, O.A.; Kresge, H.A.; Bown, C.W.; Liu, D.; Moore, E.E.; Gifford, K.A.; Acosta, L.M.Y.; Bell, S.P.; Hohman, T.J.; et al. Cerebrospinal fluid and plasma neurofilament light relate to abnormal cognition. Alzheimer’s Dement. 2019, 11, 700–709. [Google Scholar] [CrossRef]
- Mattsson, N.; Cullen, N.C.; Andreasson, U.; Zetterberg, H.; Blennow, K. Association between longitudinal plasma neurofilament light and neurodegeneration in patients with Alzheimer disease. JAMA Neurol. 2019, 76, 791–799. [Google Scholar] [CrossRef]
- Delaby, C.; Alcolea, D.; Carmona-Iragui, M.; Illan-Gala, I.; Morenas-Rodriguez, E.; Barroeta, I.; Altuna, M.; Estelles, T.; Santos-Santos, M.; Turon-Sans, J.; et al. Differential levels of neurofilament light protein in cerebrospinal fluid in patients with a wide range of neurodegenerative disorders. Sci. Rep. 2020, 10, 9161. [Google Scholar] [CrossRef] [PubMed]
- Park, J.E.; Lim, D.S.; Cho, Y.H.; Choi, K.Y.; Lee, J.J.; Kim, B.C.; Lee, K.H.; Lee, J.S. Plasma contact factors as novel biomarkers for diagnosing Alzheimer’s disease. Biomark. Res. 2021, 9, 5. [Google Scholar] [CrossRef]
- Ahmed, S.; Kim, B.C.; Lee, K.H.; Jung, H.Y. Alzheimer’s disease neuroimaging I. Ensemble of ROI-based convolutional neural network classifiers for staging the Alzheimer disease spectrum from magnetic resonance imaging. PLoS ONE 2020, 15, e0242712. [Google Scholar] [CrossRef]
- Choi, Y.Y.; Lee, J.J.; Choi, K.Y.; Seo, E.H.; Choo, I.H.; Kim, H.; Song, M.K.; Choi, S.M.; Cho, S.H.; Kim, B.C.; et al. The aging slopes of brain structures vary by ethnicity and sex: Evidence from a large magnetic resonance imaging dataset from a single Scanner of cognitively healthy elderly people in Korea. Front. Aging Neurosci. 2020, 12, 233. [Google Scholar] [CrossRef]
- Dubois, B.; Feldman, H.H.; Jacova, C.; Hampel, H.; Molinuevo, J.L.; Blennow, K.; DeKosky, S.T.; Gauthier, S.; Selkoe, D.; Bateman, R.; et al. Advancing research diagnostic criteria for Alzheimer’s disease: The IWG-2 criteria. Lancet Neurol. 2014, 13, 614–629. [Google Scholar] [CrossRef]
- Park, J.E.; Choi, K.Y.; Kim, B.C.; Choi, S.M.; Song, M.K.; Lee, J.J.; Kim, J.; Song, H.C.; Kim, H.W.; Ha, J.M.; et al. Cerebrospinal fluid biomarkers for the diagnosis of prodromal Alzheimer’s disease in amnestic mild cognitive impairment. Dement. Geriatr. Cogn. Dis. Extra 2019, 9, 100–113. [Google Scholar] [CrossRef]
- Lim, H.J.; Park, J.E.; Kim, B.C.; Choi, S.M.; Song, M.K.; Cho, S.H.; Seo, H.J.; Kim, J.; Song, H.C.; Choi, K.Y.; et al. Comparison of two analytical platforms in cerebrospinal fluid biomarkers for the classification of Alzheimer’s disease spectrum with amyloid PET imaging. J. Alzheimer’s Dis. 2020, 75, 949–958. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.Y.; Lee, J.J.; Gunasekaran, T.I.; Kang, S.; Lee, W.; Jeong, J.; Lim, H.J.; Zhang, X.; Zhu, C.; Won, S.Y.; et al. Neuroimaging initative AD. APOE promoter polymorphism-219T/G is an effect modifier of the influence of APOE epsilon4 on Alzheimer’s disease risk in a multiracial sample. J. Clin. Med. 2019, 8, 1236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barban, F.; Mancini, M.; Cercignani, M.; Adriano, F.; Perri, R.; Annicchiarico, R.; Carlesimo, G.A.; Ricci, C.; Lombardi, M.G.; Teodonno, V.; et al. A pilot study on brain plasticity of functional connectivity modulated by cognitive training in mild Alzheimer’s disease and mild cognitive impairment. Brain Sci. 2017, 7, 50. [Google Scholar] [CrossRef]
- Gorbach, T.; Pudas, S.; Lundquist, A.; Oradd, G.; Josefsson, M.; Salami, A.; de Luna, X.; Nyberg, L. Longitudinal association between hippocampus atrophy and episodic-memory decline. Neurobiol. Aging 2017, 51, 167–176. [Google Scholar] [CrossRef]
- Sabri, O.; Seibyl, J.; Rowe, C.; Barthel, H. Beta-amyloid imaging with florbetaben. Clin. Transl. Imaging 2015, 3, 13–26. [Google Scholar] [CrossRef] [Green Version]
- Guerin, J.S.; Murray, D.W.; McGrath, M.M.; Yuille, M.A.; McPartlin, J.M.; Doran, P.P. Molecular medicine Ireland guidelines for standardized biobanking. Biopreserv. Biobank 2010, 8, 3–63. [Google Scholar] [CrossRef] [Green Version]
- Kapogiannis, D.; Mattson, M.P. Disrupted energy metabolism and neuronal circuit dysfunction in cognitive impairment and Alzheimer’s disease. Lancet Neurol. 2011, 10, 187–198. [Google Scholar] [CrossRef] [Green Version]
- Crane, P.K.; Walker, R.; Larson, E.B. Glucose levels and risk of dementia. N. Engl. J. Med. 2013, 369, 1863–1864. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gordon, B.A.; Blazey, T.M.; Su, Y.; Hari-Raj, A.; Dincer, A.; Flores, S.; Christensen, J.; McDade, E.; Wang, G.; Xiong, C.; et al. Spatial patterns of neuroimaging biomarker change in individuals from families with autosomal dominant Alzheimer’s disease: A longitudinal study. Lancet Neurol. 2018, 17, 241–250. [Google Scholar] [CrossRef] [Green Version]
- Kim, Y.; Jiang, X.; Giancardo, L.; Pena, D.; Bukhbinder, A.S.; Amran, A.Y.; Schulz, P.E. Alzheimer’s disease neuroimaging I. Multimodal phenotyping of Alzheimer’s disease with longitudinal magnetic resonance imaging and cognitive function data. Sci. Rep. 2020, 10, 5527. [Google Scholar] [CrossRef]
- Anoop, A.; Singh, P.K.; Jacob, R.S.; Maji, S.K. CSF biomarkers for Alzheimer’s disease diagnosis. Int. J. Alzheimer’s Dis. 2010, 2010, 606802. [Google Scholar] [CrossRef] [Green Version]
- Kunal, D.; Veer, B.G.; Victor, L.V.; Dhamidhu, E.; Petra, L.G.; Christopher, F.; Pierrick, B.; Qiao-Xin, L.; Steven, C.; Ashley, I.B.; et al. Cerebrospinal fluid neurofilament light concentration predics brain atrophy and cognition in Alzheimer’s disease. Alzheimer’s Dement. 2020, 12, 12005. [Google Scholar]
- Palmqvist, S.; Insel, P.S.; Stomrud, E.; Janelidze, S.; Zetterberg, H.; Brix, B.; Eichenlaub, U.; Dage, J.L.; Chai, X.; Blennow, K.; et al. Cerebrospinal fluid and plasma biomarker trajectories with increasing amyloid deposition in Alzheimer’s disease. EMBO Mol. Med. 2019, 11, e11170. [Google Scholar] [CrossRef]
- Nakamura, A.; Kaneko, N.; Villemagne, V.L.; Kato, T.; Doecke, J.; Dore, V.; Fowler, C.; Li, Q.X.; Martins, R.; Rowe, C.; et al. High performance plasma amyloid-beta biomarkers for Alzheimer’s disease. Nature 2018, 554, 249–254. [Google Scholar] [CrossRef]
- Bobinski, M.; Wegiel, J.; Wisniewski, H.; Tarnawski, M.; Mlodzik, B.; Reisberg, B.; De Leon, M.; Miller, D. Atrophy of hippocampal formation subdivisions correlates with stage and duration of Alzheimer disease. Dement. Geriatr. Cogn. Disord. 1995, 6, 205–210. [Google Scholar] [CrossRef]
- Juottonen, K.; Laakso, M.; Insausti, R.; Lehtovirta, M.; Pitkänen, A.; Partanen, K.; Soininen, H. Volumes of the entorhinal and perirhinal cortices in Alzheimer’s disease. Neurobiol. Aging 1998, 19, 15–22. [Google Scholar] [CrossRef]
- Stoub, T.; Rogalski, E.; Leurgans, S.; Bennett, D.; de Toledo-Morrell, L. Rate of entorhinal and hippocampal atrophy in incipient and mild AD: Relation to memory function. Neurobiol. Aging 2010, 31, 1089–1098. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Du, A.; Schuff, N.; Kramer, J.; Ganzer, S.; Zhu, X.; Jagust, W.; Miller, B.; Reed, B.R.; Mungas, D.; Yaffe, K. Higher atrophy rate of entorhinal cortex than hippocampus in AD. Neurology 2004, 62, 422–427. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.F.; Shen, X.N.; Li, J.Q.; Suckling, J.; Tan, C.C.; Wang, Y.J.; Feng, L.; Zhang, C.; Tan, L.; Dong, Q. Clinical and biomarker trajectories in sporadic Alzheimer’s disease: A longitudinal study. Alzheimer’s Dement. Diagn. Assess. Dis. Monit. 2020, 12, e12095. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | Total | CN (n = 51) | aMCI (n = 54) | AD (n = 31) | ||
|---|---|---|---|---|---|---|
| Aβ− | Aβ+ (Preclinical AD) | Aβ− | Aβ+ (Prodromal AD) | Aβ+ | ||
| n | 136 | 28 | 23 | 22 | 32 | 31 |
| Age, mean (SD), y | 136 | 69.4 (6.3) | 73.9 (2.5) * | 69.1 (8.6) | 72.7 (8.0) *† | 65.2 (8.7) *† |
| Education, mean (SD), y | 132 | 9.3 (4.2) | 11.2 (5.5) | 10.4 (5.1) | 9.6 (5.1) | 6.4 (3.5) *† |
| Female sex, No. (%) | 136 | 17 (60.7) | 10 (43.5) | 6 (27.3) | 16 (50.0) | 21.0 (67.7) |
| K-MMSE score, mean (SD) | 131 | 26.5 (2.3) | 27.5 (1.9) | 25.7 (3.1) | 24.8 (2.8) * | 18.9 (4.3) *† |
| APOE ε4 carrier, No. (%) | 133 | 3 (10.7) | 16 (69.5) * | 2 (9.1) | 26 (81.3) *† | 24 (77.4) *† |
| CSF biomarkers, mean (SD), pg/mL | ||||||
| NFL concentrations, pg/mL | 136 | 655.7 (150.0) | 989.2 (487.5) * | 693.7 (281.7) | 960.5 (398.3) *† | 970.4 (360.9) *† |
| Aβ1–42 con., pg/mL | 136 | 1089.3 (160.2) | 516.4 (192.3) * | 947.3 (161.5) | 473.0 (147.1) *† | 399.1 (135.1) *† |
| t-Tau con., pg/mL | 136 | 209.7 (54.6) | 322.9 (122.1) * | 206.8 (70.9) | 475.7 (210.8) *† | 522.8 (217.1) *† |
| p-Tau181 con., pg/mL | 136 | 40.0 (8.8) | 52.6 (19.4) * | 38.2 (9.4) | 72.2 (26.8) *† | 74.4 (27.0) *† |
| Plasma biomarkers, mean (SD), pg/mL | ||||||
| NFL con., pg/mL | 136 | 16.7 (6.0) | 20.9 (6.5) * | 18.1 (9.1) | 22.5 (9.3) * | 21.8 (6.6) * |
| Aβ1–42 con., pg/mL | 136 | 12.7 (3.9) | 9.9 (3.0) * | 12.4 (3.8) | 9.5 (2.2) *† | 8.2 (2.4) *† |
| Combination biomarkers, ratio | ||||||
| CSF NFL/Aβ1–42 ratio | 136 | 0.62 (0.17) | 2.02 (1.0) * | 0.74 (0.3) | 2.29 (1.3) *† | 2.62 (1.18) *† |
| Plasma NFL/Aβ1–42 ratio | 136 | 1.46 (0.65) | 2.46 (1.3) * | 1.46 (0.5) | 2.46 (1.1) *† | 2.92 (1.19) *† |
| Neuroimaging | ||||||
| Aβ- PET (SUVR score) | 135 | 1.0 (0.06) | 1.24 (0.13) * | 0.97 (0.06) | 1.30 (0.19) *† | 1.3886 (0.11) *† |
| Hippocampal volume/ICV | 134 | 0.0029 (0.00032) | 0.0027 (0.00031) | 0.0025 (0.00043) | 0.0024 (0.00037) * | 0.0021 (0.00038) *† |
| Entorhinal cortex (mm) | 134 | 3.4213 (0.32244) | 3.3355 (2.28296) | 3.3066 (0.47337) | 3.0681 (0.38707) * | 2.9610 (0.45533) *† |
| Molecules | CSF Biomarkers | Plasma Biomarkers | Combination Biomarkers | Neuroimaging Data | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| NFL | Aβ1–42 | t-Tau | p-Tau181 | NFL | Aβ1–42 | CSFNFL/Aβ1–42 | PlasmaNFL/Aβ1–42 | Aβ− PET (SUVR) | Hippocampal Volume/ICV | Entorhinal Thickness | |
| CSF NFL concentrations | 1 | −0.259 ** | 0.486 * | 0.502 * | 0.608 * | −0.110 | 0.710 * | 0.521 * | 0.334 * | −0.359 ** | −0.194 ** |
| CSF Aβ1–42 concentrations | 1 | −0.410 * | −0.357 * | −0.242 ** | 0.472 * | −0.736 * | −0.462 * | −0.701 * | 0.340 * | 0.245 * | |
| CSF t-Tau concentrations | 1 | 0.923 * | 0.265 ** | −0.305 * | 0.491 * | 0.382 * | 0.617 * | −0.427 * | −0.378 * | ||
| CSF p-Tau181 concentrations | 1 | 0.280 ** | −0.304* | 0.476 * | 0.364 * | 0.555 * | −0.392 * | −0.334 * | |||
| Plasma NFL concentrations | 1 | 0.169 ** | 0.493 * | 0.612 * | 0.218 ** | −0.432 * | −0.221 ** | ||||
| Plasma Aβ1–42 concentrations | 1 | −0.321 * | −0.503 * | −0.374 * | 0.086 | 0.031 | |||||
| CSF NFL/Aβ1–42 ratio | 1 | 0.562 * | 0.580 * | −0.379 * | −0.213 ** | ||||||
| Plasma NFL/Aβ1–42 ratio | 1 | 0.410 * | −0.409 * | −0.132 | |||||||
| Aβ− PET (SUVR score) | 1 | −0.348 * | −0.307 * | ||||||||
| Hippocampal volume/ICV | 1 | 0.622 * | |||||||||
| Entorhinal thickness | 1 | ||||||||||
| CSF Biomarker (pg/mL) | Plasma Biomarker (pg/mL) | Combination (∆, Delta Ratio) | Neuroimaging Data | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NFL | Aβ1–42 | t-Tau | p-Tau181 | NFL | Aβ1–42 | CSF NFL/Aβ1–42 | Plasma NFL/Aβ1–42 | Aβ− PET (SUVR) | Hippocampal Volume/ICV | Entorhinal Cortex | ||
| CN (Aβ−) versus Pre-AD | Cutoff | >696.2 | <817.3 | >241.5 | >43.6 | >17.3 | <10.45 | >0.89 | >1.7 | >1.0695 | <0.0028 | <3.3995 |
| Sen (%) | 65.2 | 96.4 | 76.2 | 66.7 | 69.6 | 67.9 | 100.0 | 69.6 | 91.3 | 57.1 | 57.1 | |
| Spe (%) | 60.7 | 95.2 | 67.9 | 64.3 | 50.0 | 69.6 | 96.4 | 66.7 | 82.1 | 56.5 | 56.5 | |
| AUC (95%CI) | 0.731 (0.59–0.88) | 0.994 (0.98–1.00) | 0.776 (0.63–0.92) | 0.711 (0.55–0.87) | 0.668 (0.52–0.82) | 0.741 (0.60–0.88) | 1.000 (1.00–1.00) | 0.791 (0.67–0.91) | 0.974 (0.94–1.0) | 0.624 (0.47–0.78) | 0.598 (0.44–0.76) | |
| p value | 0.005 | <0.001 | 0.003 | 0.010 | 0.041 | 0.003 | <0.001 | <0.001 | <0.001 | 0.130 | 0.233 | |
| CN (Aβ−) versus Pro-AD | Cut-off | >735.7 | <745.6 | >276.9 | >48.8 | >19.0 | <9.3 | >0.94 | >2.05 | >1.1015 | <0.0026 | <3.2835 |
| Sen (%) | 75.8 | 100.0 | 84.8 | 81.8 | 63.6 | 84.6 | 93.9 | 72.2 | 90.6 | 75.0 | 75.0 | |
| Spe (%) | 71.4 | 93.9 | 85.7 | 82.1 | 57.1 | 61.1 | 96.4 | 76.9 | 92.9 | 75.0 | 74.2 | |
| AUC (95%CI) | 0.781 (0.66–0.90) | 1.000 (1.00–1.00) | 0.922 (0.85–0.99) | 0.890 (0.80–0.98) | 0.696 (0.57–0.83) | 0.748 (0.58–0.92) | 0.988 (0.97–1.00) | 0.865 (0.74–0.99) | 0.951 (0.89–1.00) | 0.826 (0.72–0.93) | 0.793 (0.68–0.91) | |
| p value | <0.001 | <0.001 | <0.001 | <0.001 | 0.009 | 0.02 | <0.001 | 0.001 | <0.001 | <0.001 | <0.001 | |
| CN (Aβ−) versus AD dementia | Cut-off | >752.4 | <620.0 | >284.7 | >52.4 | >20.9 | <8.5 | >1.26 | >2.30 | >1.2075 | <0.0025 | <3.2675 |
| Sen (%) | 71.0 | 100.0 | 87.1 | 83.9 | 64.5 | 84.6 | 96.8 | 93.8 | 100.0 | 85.7 | 75.0 | |
| Spe (%) | 75.0 | 93.5 | 89.3 | 89.3 | 67.9 | 75.0 | 100.0 | 92.3 | 100.0 | 83.3 | 73.3 | |
| AUC (95%CI) | 0.782 (0.67–0.90) | 0.997 (0.99–1.00) | 0.962 (0.92–1.00) | 0.898 (0.81–0.97) | 0.710 (0.58–0.84) | 0.916 (0.82–1.00) | 0.999 (0.99–1.00) | 0.964 (0.90–1.00) | 1.00 (1.00–1.00) | 0.923 (0.85–0.99) | 0.804 (0.69–0.92) | |
| p value | <0.001 | <0.001 | <0.001 | <0.001 | 0.006 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | |
| aMCI (Aβ−) versus Pro-AD | Cut-off | >763.6 | <745.6 | >259.5 | >45.0 | >18.8 | <10.45 | >1.08 | >1.77 | >1.0545 | <0.0024 | <3.2530 |
| Sen (%) | 66.7 | 95.5 | 87.9 | 84.8 | 63.6 | 77.3 | 84.8 | 75.0 | 90.6 | 54.5 | 68.2 | |
| Spe (%) | 68.2 | 93.9 | 86.4 | 86.4 | 63.6 | 68.8 | 86.4 | 72.7 | 90.9 | 53.1 | 67.7 | |
| AUC (95%CI) | 0.719 (0.58–0.86) | 0.986 (0.96–1.00) | 0.919 (0.84–0.99) | 0.905 (0.82–0.99) | 0.650 (0.49–0.81) | 0.769 (0.62–0.92) | 0.947 (0.90–0.99) | 0.822 (0.71–0.93) | 0.960 (0.90–1.00) | 0.561 (0.40–0.72) | 0.717 (0.56–0.87) | |
| p value | 0.006 | <0.001 | <0.001 | <0.001 | 0.061 | 0.001 | <0.001 | <0.001 | <0.001 | 0.449 | 0.009 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, J.E.; Gunasekaran, T.I.; Cho, Y.H.; Choi, S.-M.; Song, M.-K.; Cho, S.H.; Kim, J.; Song, H.-C.; Choi, K.Y.; Lee, J.J.; et al. Diagnostic Blood Biomarkers in Alzheimer’s Disease. Biomedicines 2022, 10, 169. https://doi.org/10.3390/biomedicines10010169
Park JE, Gunasekaran TI, Cho YH, Choi S-M, Song M-K, Cho SH, Kim J, Song H-C, Choi KY, Lee JJ, et al. Diagnostic Blood Biomarkers in Alzheimer’s Disease. Biomedicines. 2022; 10(1):169. https://doi.org/10.3390/biomedicines10010169
Chicago/Turabian StylePark, Jung Eun, Tamil Iniyan Gunasekaran, Yeong Hee Cho, Seong-Min Choi, Min-Kyung Song, Soo Hyun Cho, Jahae Kim, Ho-Chun Song, Kyu Yeong Choi, Jang Jae Lee, and et al. 2022. "Diagnostic Blood Biomarkers in Alzheimer’s Disease" Biomedicines 10, no. 1: 169. https://doi.org/10.3390/biomedicines10010169
APA StylePark, J. E., Gunasekaran, T. I., Cho, Y. H., Choi, S.-M., Song, M.-K., Cho, S. H., Kim, J., Song, H.-C., Choi, K. Y., Lee, J. J., Park, Z.-Y., Song, W. K., Jeong, H.-S., Lee, K. H., Lee, J. S., & Kim, B. C. (2022). Diagnostic Blood Biomarkers in Alzheimer’s Disease. Biomedicines, 10(1), 169. https://doi.org/10.3390/biomedicines10010169






