Functionalization of Bulk SiO2 Surface with Biomolecules for Sensing Applications: Structural and Functional Characterizations
Abstract
:1. Introduction
2. Biomolecule Immobilization
2.1. SiO2 Preparation
2.2. Quality Check of Immobilization Process
2.3. SiO2 Biointerfaces
2.3.1. Protein Immobilization
2.3.2. DNA Immobilization
3. Bioactivity Test
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
Appendix A.1. APTES Protocol for Biomolecule Immobilization
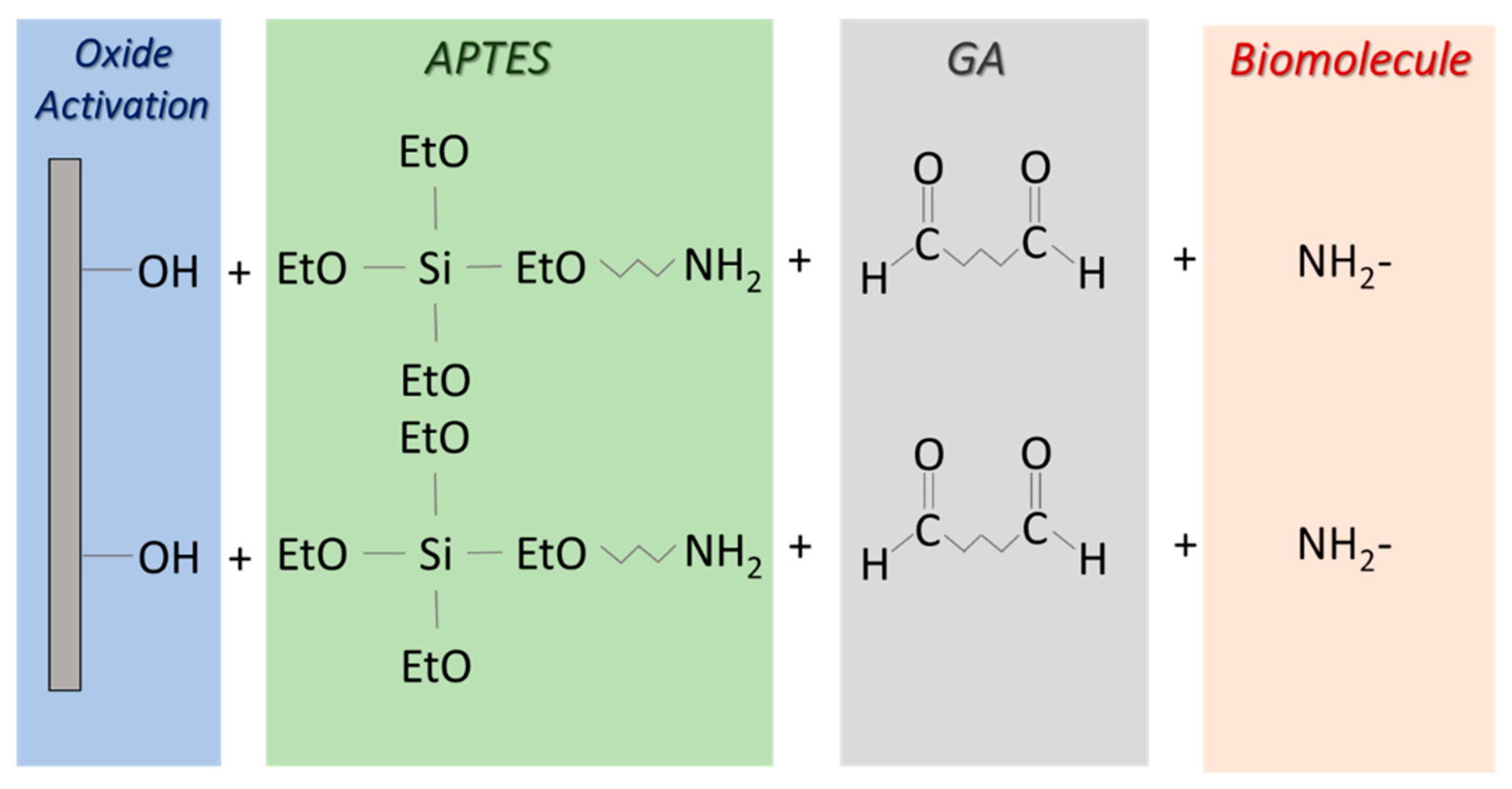
Appendix A.2. GOPS Protocol for Biomolecule Immobilization
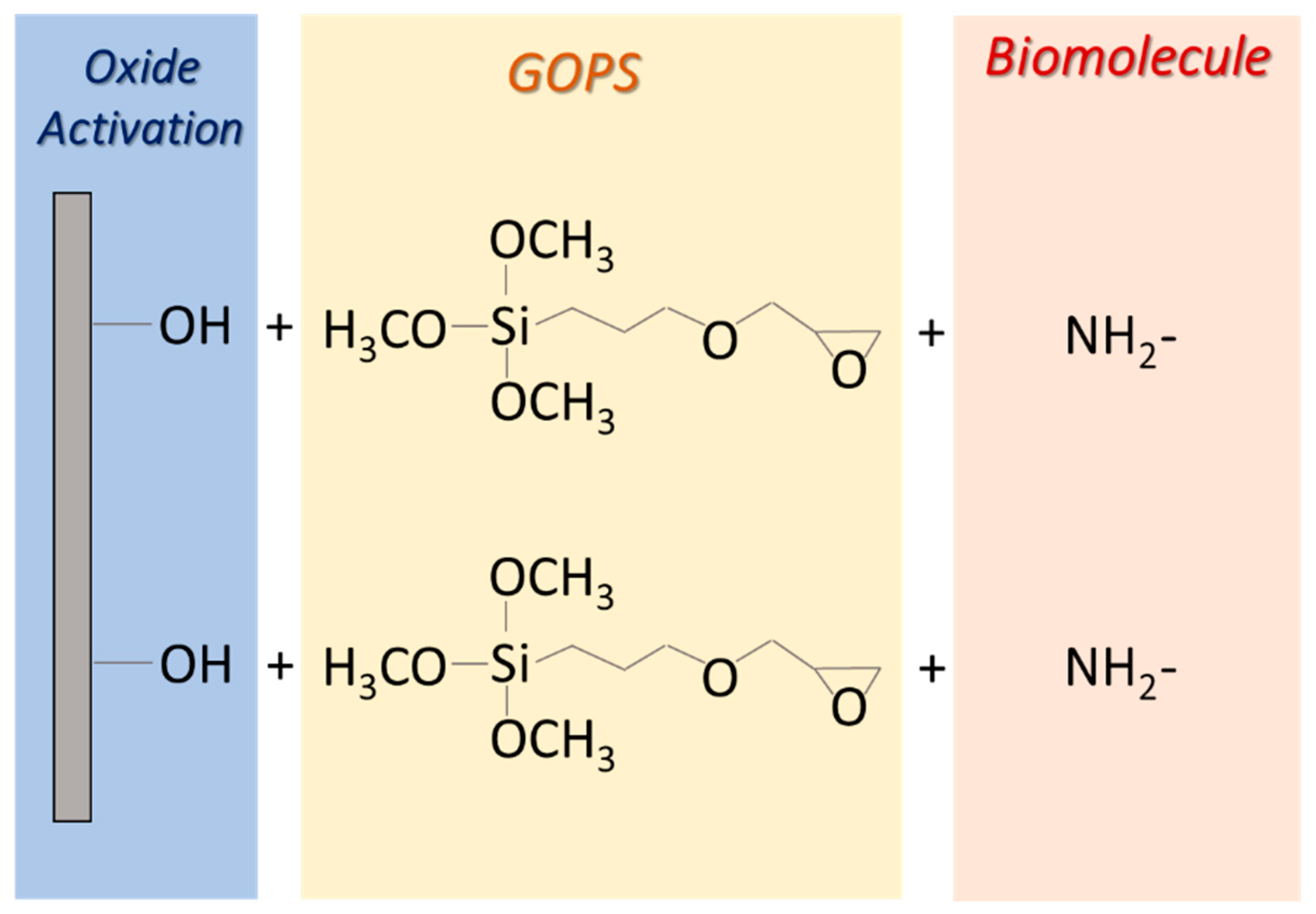
Appendix A.3. Microarray Preparation
References
- Chen, D.; Wang, G.; Li, J. Interfacial Bioelectrochemistry: Fabrication, Properties and Applications of Functional Nanostructured Biointerfaces. J. Phys. Chem. C 2007, 111, 2351–2367. [Google Scholar] [CrossRef]
- Xu, X.; Kratz, K.; Wang, W.; Li, Z.; Roch, T.; Jung, F.; Lendlein, A.; Ma, N. Cultivation and spontaneous differentiation of rat bone marrow-derived mesenchymal stem cells on polymeric surfaces. Clin. Hemorheol. Microcirc. 2013, 55, 143–156. [Google Scholar] [CrossRef] [PubMed]
- Dreaden, E.C.; Austin, L.A.; Mackey, M.A.; El-Sayed, M.A. Size Matters: Gold Nanoparticles in Targeted Cancer Drug Delivery. Ther. Deliv. 2017, 3, 457–478. [Google Scholar] [CrossRef]
- Coffer, J.L. Overview of Semiconducting Silicon Nanowires for Biomedical Applications; Woodhead Publishing Series in Biomaterials; Elsevier: New York, NY, USA, 2014; pp. 3–7. [Google Scholar] [CrossRef]
- Zhang, G.J.; Ning, Y. Silicon nanowire biosensor and its applications in disease diagnostics: A review. Anal. Chim. Acta 2012, 749, 1–15. [Google Scholar] [CrossRef]
- Schena, M.; Shalon, D.; Davis, R.W.; Brown, P.O. Quantitative monitoring of gene expression patterns with a complementary DNA microarray. Science 1995, 270, 467–470. [Google Scholar] [CrossRef]
- Zhu, H.; Sneider, M. Protein arrays and microarrays. Curr. Opin. Chern. Biol. 2001, 5, 40–50. [Google Scholar] [CrossRef]
- Freeman, W.M.; Robertson, D.J.; Vrana, K.E. Fundamentals of DNA hybridization arrays for gene expression analysis. Biotechniques 2000, 29, 1042–1055. [Google Scholar] [CrossRef]
- Santangelo, M.F.; Sciuto, E.L.; Lombardo, S.; Busacca, A.C.; Petralia, S.; Conoci, S.; Libertino, S. Si photomultipliers for bio-sensing applications. J. Sel. Top. Quant. Electr. 2016, 22. [Google Scholar] [CrossRef]
- Favetta, M.; Valletta, A.; Fortunato, G.; Castagna, M.E.; Conoci, S.; Sciuto, E.L.; Cosentino, T.; Sinatra, F.; Libertino, S. Development of Si-based Electrical Biosensors: Simulations and first experimental results. Sens. Bio-Sens. Res. 2015, 6, 72–78. [Google Scholar] [CrossRef]
- Petralia, S.; Sciuto, E.L.; Mirabella, S.; Di Pietro, M.L.; Zimbone, M.; Conoci, S. An Innovative Chemical Strategy for PCR-free Genetic Detection of Pathogens by an Integrated Electrochemical Biosensor. Analyst 2017, 142, 2090–2093. [Google Scholar] [CrossRef]
- Messina, M.A.; Meli, C.; Conoci, S.; Petralia, S. A facile method for urinary phenylalanine measurement on paper-based lab-on-chip for PKU therapy monitoring. Analyst 2017, 142, 4629. [Google Scholar] [CrossRef] [PubMed]
- Saby, C.; Jaffrezic-Renault, N.; Martelet, C.; Colin, B.; Charles, M.H.; Dehair, T.; Mandrand, B. Immobilization of antibodies onto a capacitance silicon-based transducer. Sens. Actuators B 1993, 15–16, 458–463. [Google Scholar] [CrossRef]
- Subramanian, A.; Kennel, S.J.; Oden, P.I.; Jacobson, K.B.; Woodward, J.; Doktycz, M.J. Comparison of techniques for enzyme immobilization on silicon supports. Enzyme Microb. Technol. 1999, 24, 26–34. [Google Scholar] [CrossRef]
- Lobert, P.E.; Bourgeois, D.; Pampin, R.; Akheyar, A.; Hagelsieb, L.M.; Flandre, D.; Remacle, J. Immobilization of DNA on CMOS compatible materials. Sens. Actuators B 2003, 92, 90–97. [Google Scholar] [CrossRef]
- Cai, W.; Peck, J.R.; van der Weide, D.W.; Hamers, R.J. Direct electrical detection of hybridization at DNA-modified silicon surfaces. Biosens. Bioelectr. 2004, 19, 1013–1019. [Google Scholar] [CrossRef] [PubMed]
- Iler, R.K. The Chemistry of Silica; Plenum Press: New York, NY, USA, 1979; ISBN 0-471-02404-X. [Google Scholar]
- Jutzi, P.; Schubert, U. Silicon Chemistry: From the Atom to Extended Systems; Wiley-VCH: Hoboken, NJ, USA, 2003; ISBN 3-527-30647-1. [Google Scholar]
- Herne, T.M.; Tarlov, M.J. Characterization of DNA probes immobilized on gold surfaces. J. Am. Chem. Soc. 1997, 119, 8916–8920. [Google Scholar] [CrossRef]
- Piunno, P.A.E.; Krull, U.J. Fiber-optic DNA sensor for fluorimetric nucleic acid determination. Analyt. Chem. 1995, 67, 2635–2643. [Google Scholar] [CrossRef]
- Libertino, S.; Fichera, M.; Aiello, V.; Statello, G.; Fiorenza, P.; Sinatra, F. Experimental characterization of proteins immobilized on Si-based materials. Microelectr. Eng. 2007, 84, 468–473. [Google Scholar] [CrossRef]
- Libertino, S.; Scandurra, A.; Aiello, V.; Giannazzo, F.; Sinatra, F.; Renis, M.; Fichera, M. Layer uniformity in glucose oxidase immobilization on SiO2 surfaces. Applied Surface. Science 2007, 253, 9116–9123. [Google Scholar]
- Libertino, S.; Giannazzo, F.; Aiello, V.; Scandurra, A.; Sinatra, F.; Renis, M.; Fichera, M. XPS and AFM characterization of the enzyme glucose oxidase immobilized on SiO2 surfaces. Langmuir 2008, 24, 1965–1972. [Google Scholar] [CrossRef]
- Libertino, S.; Fichera, M.; Aiello, V.; Lombardo, S.; Scandurra, A.; Renis, M.; Sinatra, F. Electrical characterization of biological molecules deposition in MOS capacitors. Sens. Lett. 2008, 6, 531–536. [Google Scholar] [CrossRef]
- Hu, S.; Zhao, Y.; Qin, K.; Retterer, S.T.; Kravchenko, I.I.; Weiss, S.M. Enhancing the sensitivity of label-free silicon photonic biosensors through increased probe molecule density. ACS Phot. 2014, 1, 590–597. [Google Scholar] [CrossRef]
- Zhao, Y.; Lawrie, J.L.; Beavers, K.R.; Laibinis, P.E.; and Weiss, S.M. Effect of DNA-induced corrosion on passivated porous silicon biosensors. ACS Appl. Mat. Interface 2014, 6, 13510–13519. [Google Scholar] [CrossRef]
- Chen, E.Y.; Liu, W.F. Biomolecule immobilization and delivery strategies for controlling immune response. Nanotechnol. Prev. Regen. Med. 2018, 207–225. [Google Scholar] [CrossRef]
- Davidsson, R.; Genin, F.; Bengtsson, M.; Laurell, T.; Emneus, J. Microfluidic biosensing systems Part I. Development and optimisation of enzymatic chemiluminescent µ-biosensors based on silicon microchips. Lab Chip 2004, 4, 481–487. [Google Scholar] [CrossRef] [PubMed]
- Shen, M.-Y.; Li, B.-R.; Li, Y.-K. Silicon nanowire field-effect-transistor based biosensors: From sensitive to ultra-sensitive. Biosens. Bioelectron. 2014, 60, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Beniwal, A.; Saini, P.; Kokkiligadda, A.; Vij, S. Use of silicon dioxide nanoparticles for b-galactosidase immobilization and modulated ethanol production by co-immobilized K. marxianus and S. cerevisiae in deproteinized cheese whey. Food Sci. Technol. 2018, 87, 553–561. [Google Scholar] [CrossRef]
- Arreola, J.; Oberländer, J.; Mätzkow, M.; Keusgen, M.; Schöning, M.J. Surface functionalization for spore-based biosensors with organosilanes. Electroch. Acta 2017, 241, 237–243. [Google Scholar] [CrossRef]
- Libertino, S.; Conoci, S.; Scandurra, A.; Spinella, C. Biosensor integration on Si-based devices: Feasibility studies and examples. Sens. Actuators B 2013, 179, 240–251. [Google Scholar] [CrossRef]
- Petralia, S.; Cosentino, T.; Sinatra, F.; Favetta, M.; Fiorenza, P.; Bongiorno, C.; Sciuto, E.L.; Conoci, S.; Libertino, S. Silicon nitride surfaces as active substrate for electrical DNAbiosensors. Sens. Actuators B 2017, 252, 492–502. [Google Scholar] [CrossRef]
- Foglieni, B.; Brisci, A.; San Biagio, F.; Di Pietro, P.; Petralia, S.; Conoci, S.; Ferrari, M.; Cremonesi, L. Integrated PCR amplification and detection processes on a Lab-on-Chip platform: A new advanced solution for molecular diagnostics. Clin. Chem. Lab. Med. 2010, 48, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.; Smith, S.; Windle, B.; Guiseppi-Elie, A. Impact of surface chemistry and blocking strategies on DNA microarrays. Nucleic Acids Res. 2003, 31, e87. [Google Scholar] [CrossRef] [PubMed]
- Daniels, M.W.; Sefcik, J.; Francis, L.F.; McCormick, A.V. Reactions of a trifunctional silane coupling agent in the presence of colloidal silica sols in polar media. J. Colloid Interface Sci. 1999, 219, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Park, O.-H.; Eo, Y.-J.; Choi, Y.-K.; Bae, B.-S. Preparation and optical properties of silica-poly(ethylene oxide) hybrid materials. J. Sol Gel Sci. Technol. 1999, 16, 235–241. [Google Scholar] [CrossRef]
- Mittal, K.L. (Ed.) Contact Angle, Wettability and Adhesion; VSP International Science Publishers: Leiden, The Netherlands, 1993. [Google Scholar]
- Integrated DNA Technologies, Inc. Available online: https://eu.idtdna.com/site/account/login?returnurl=//calc/analyzer (accessed on 15 March 2017).
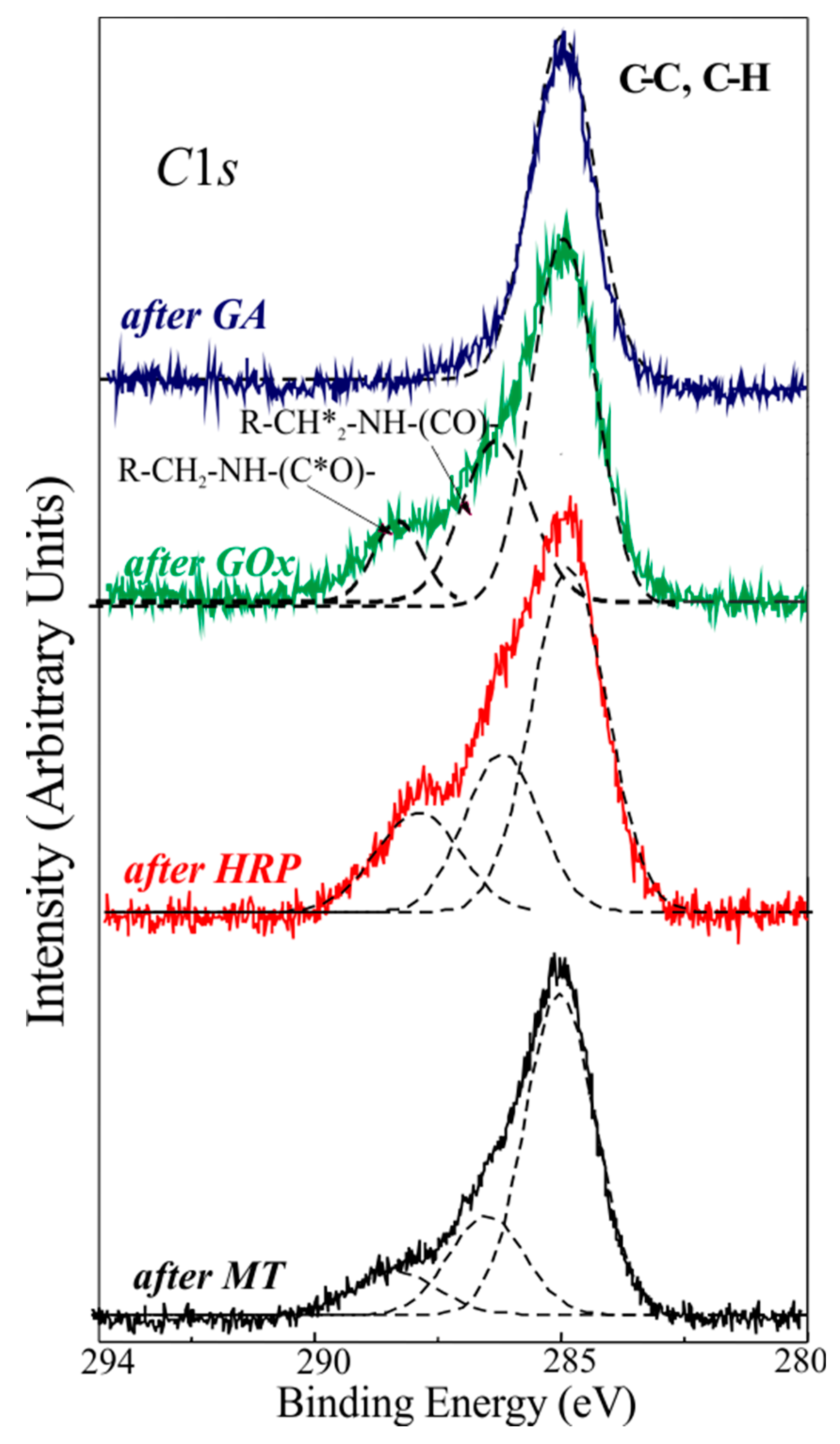
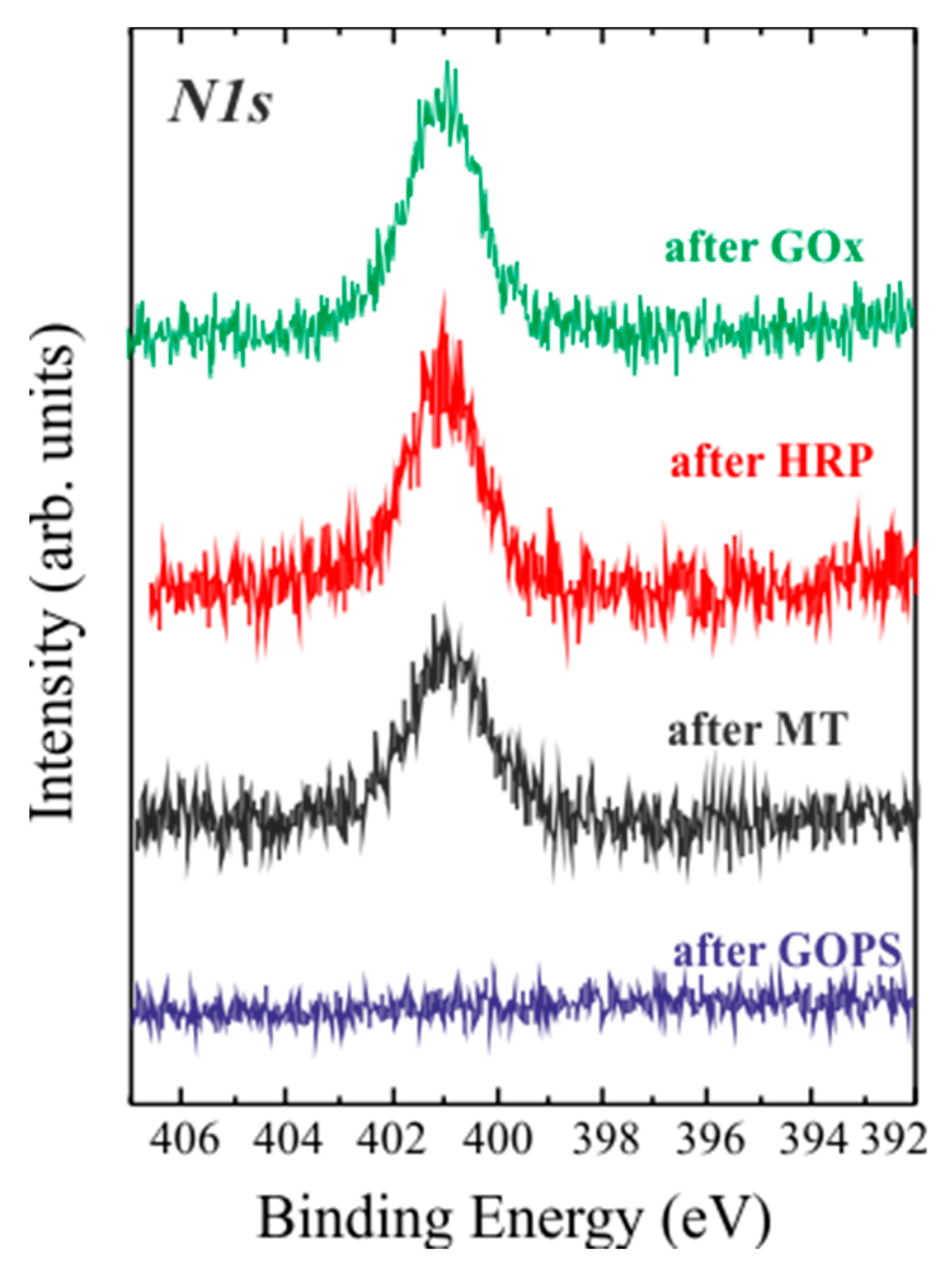

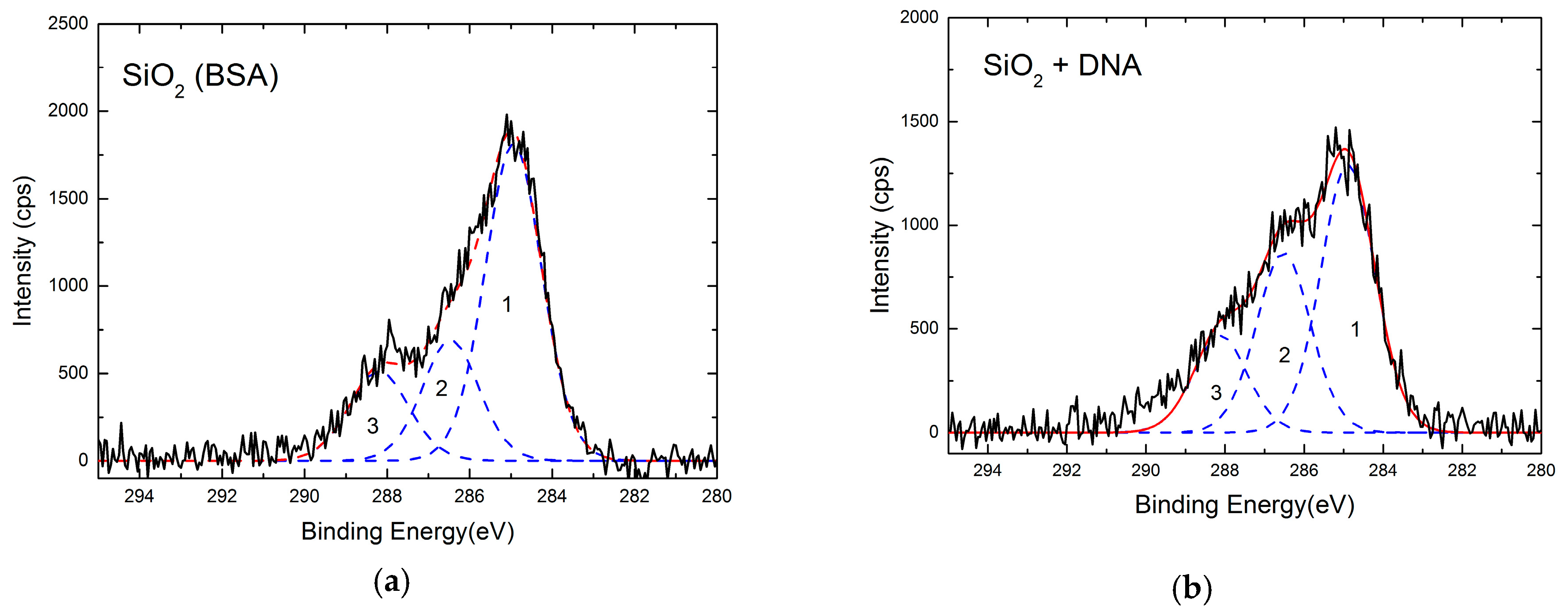


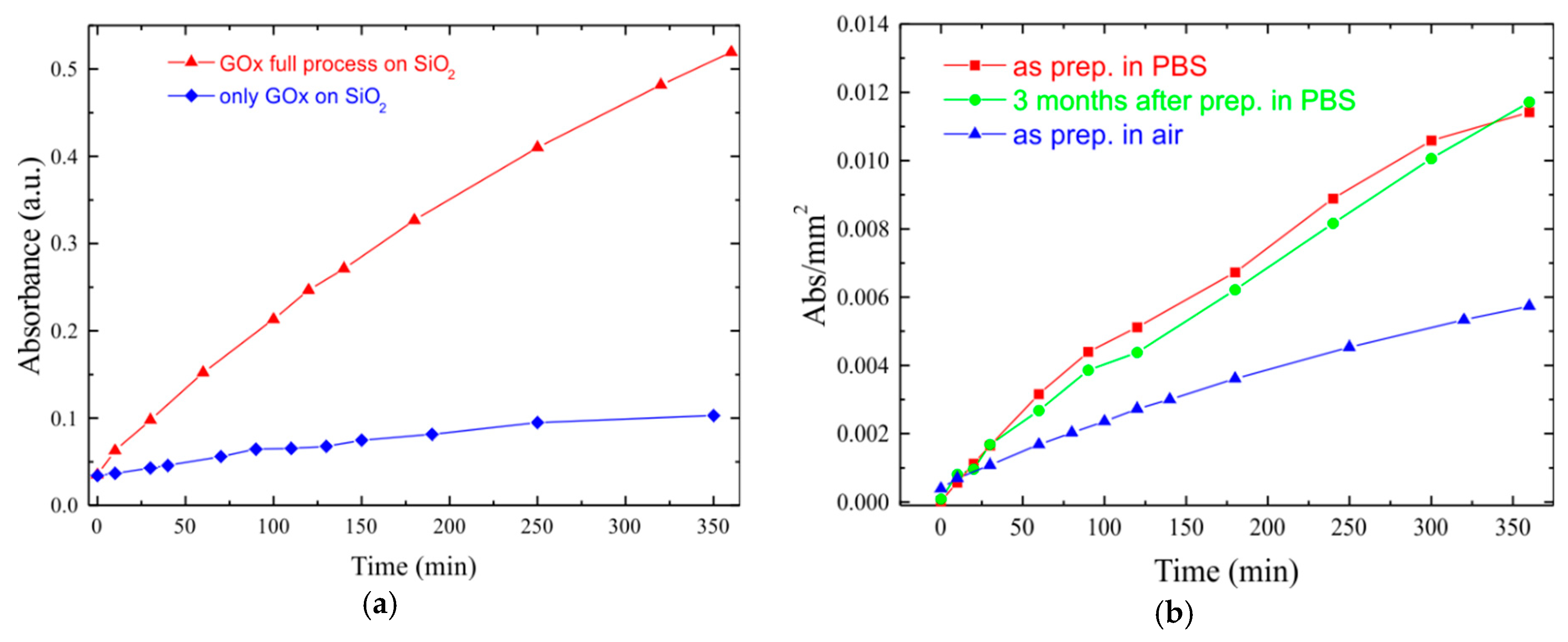
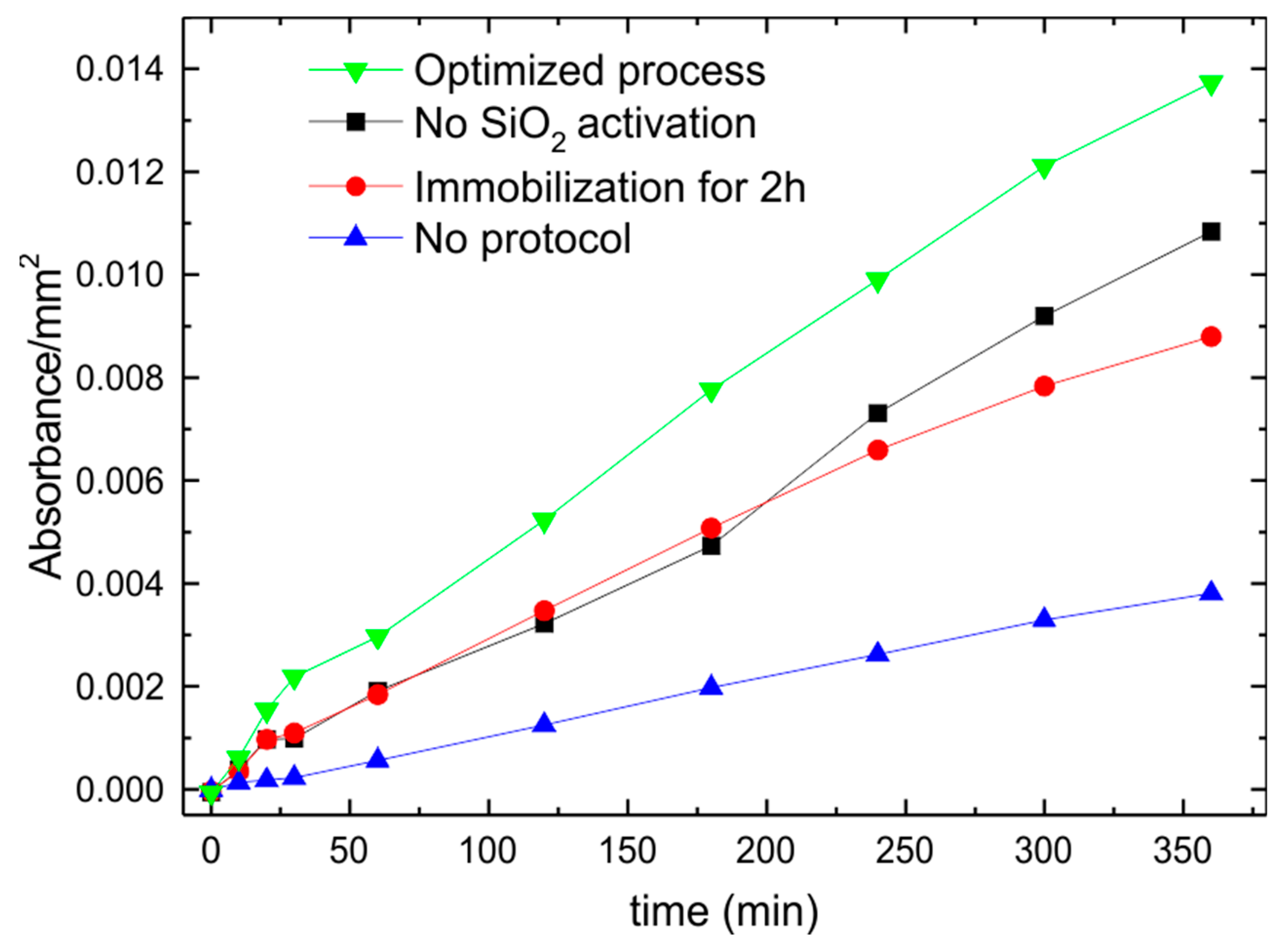
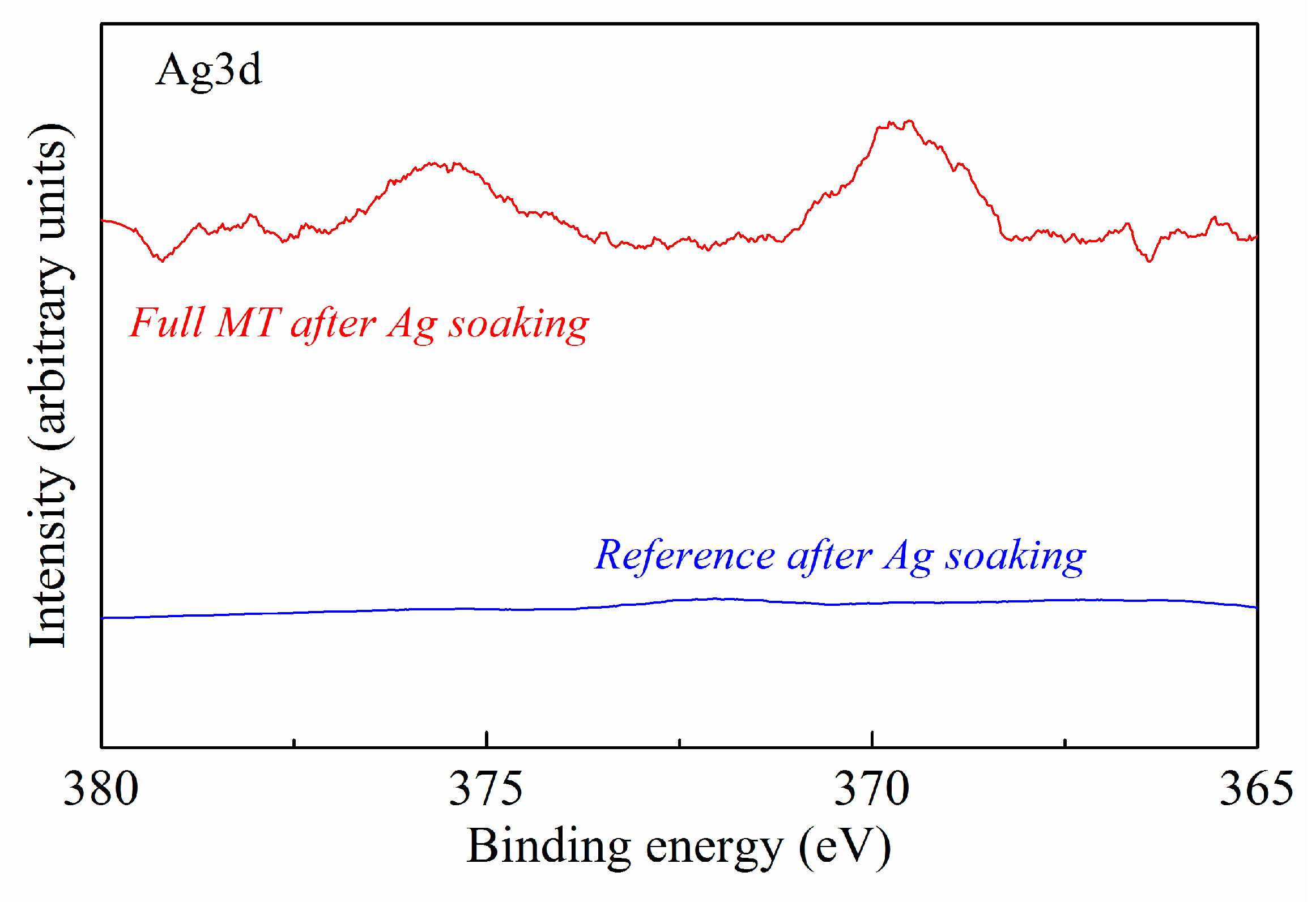
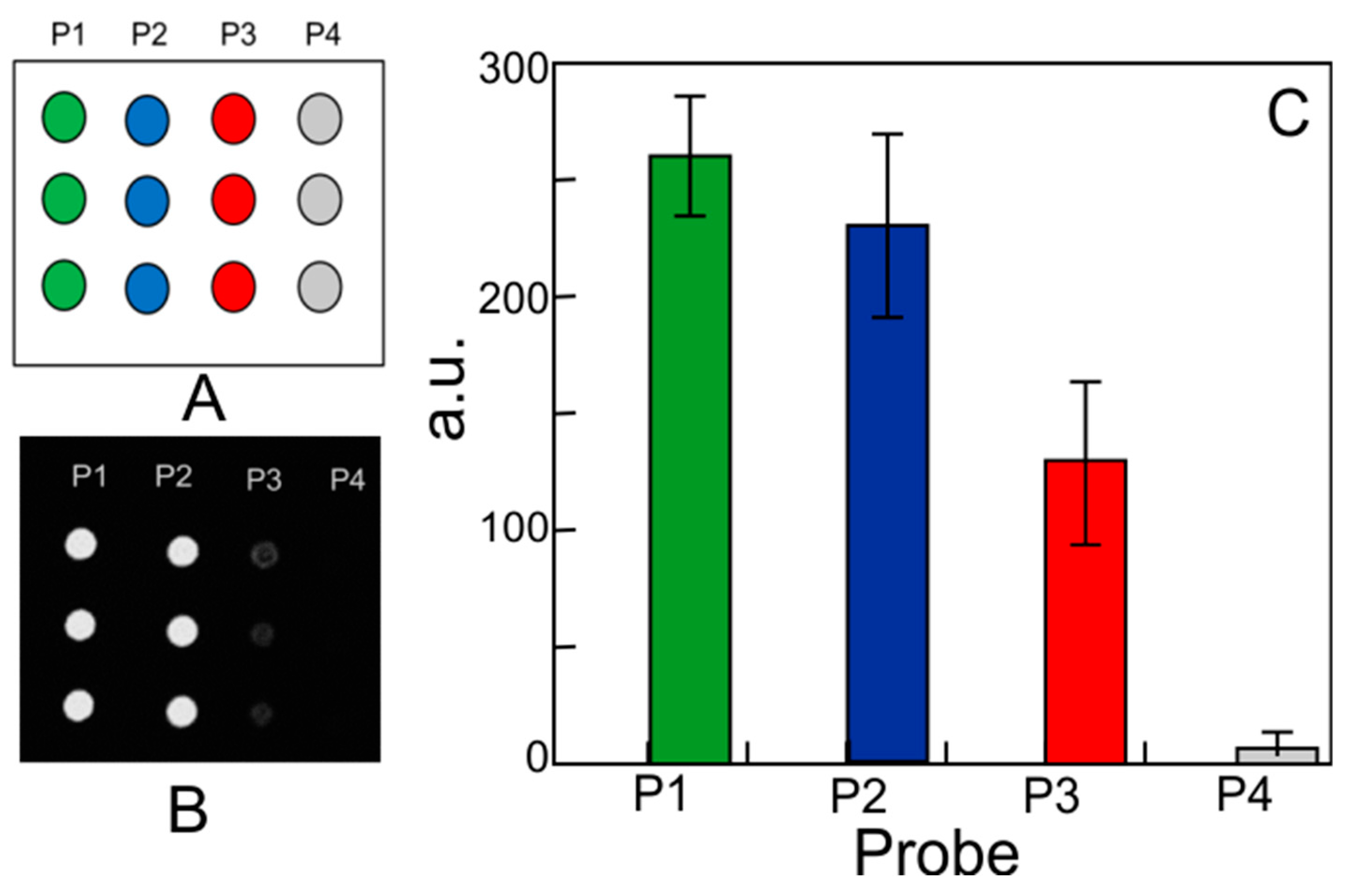
| Sample | C | O | N | Si |
|---|---|---|---|---|
| Reference | 7.7 | 63 | - | 29.3 |
| After GA | 51.2 | 26.5 | - | 22.3 |
| After GOx | 34.9 | 40.4 | 5.9 | 18.8 |
| After HRP | 29.1 | 50.7 | 4.5 | 15.7 |
| After MT | 35.5 | 41.5 | 4.3 | 18.7 |
| Sample | C | O | N | Si |
|---|---|---|---|---|
| Reference | 7 | 64.2 | - | 28.8 |
| After DNA | 35.6 | 40 | 3.1 | 21.3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sciuto, E.L.; Bongiorno, C.; Scandurra, A.; Petralia, S.; Cosentino, T.; Conoci, S.; Sinatra, F.; Libertino, S. Functionalization of Bulk SiO2 Surface with Biomolecules for Sensing Applications: Structural and Functional Characterizations. Chemosensors 2018, 6, 59. https://doi.org/10.3390/chemosensors6040059
Sciuto EL, Bongiorno C, Scandurra A, Petralia S, Cosentino T, Conoci S, Sinatra F, Libertino S. Functionalization of Bulk SiO2 Surface with Biomolecules for Sensing Applications: Structural and Functional Characterizations. Chemosensors. 2018; 6(4):59. https://doi.org/10.3390/chemosensors6040059
Chicago/Turabian StyleSciuto, Emanuele Luigi, Corrado Bongiorno, Antonino Scandurra, Salvatore Petralia, Tiziana Cosentino, Sabrina Conoci, Fulvia Sinatra, and Sebania Libertino. 2018. "Functionalization of Bulk SiO2 Surface with Biomolecules for Sensing Applications: Structural and Functional Characterizations" Chemosensors 6, no. 4: 59. https://doi.org/10.3390/chemosensors6040059
APA StyleSciuto, E. L., Bongiorno, C., Scandurra, A., Petralia, S., Cosentino, T., Conoci, S., Sinatra, F., & Libertino, S. (2018). Functionalization of Bulk SiO2 Surface with Biomolecules for Sensing Applications: Structural and Functional Characterizations. Chemosensors, 6(4), 59. https://doi.org/10.3390/chemosensors6040059







