CuO Thin Films Functionalized with Gold Nanoparticles for Conductometric Carbon Dioxide Gas Sensing
Abstract
:1. Introduction
2. Materials and Methods
2.1. Gas Sensor Fabrication
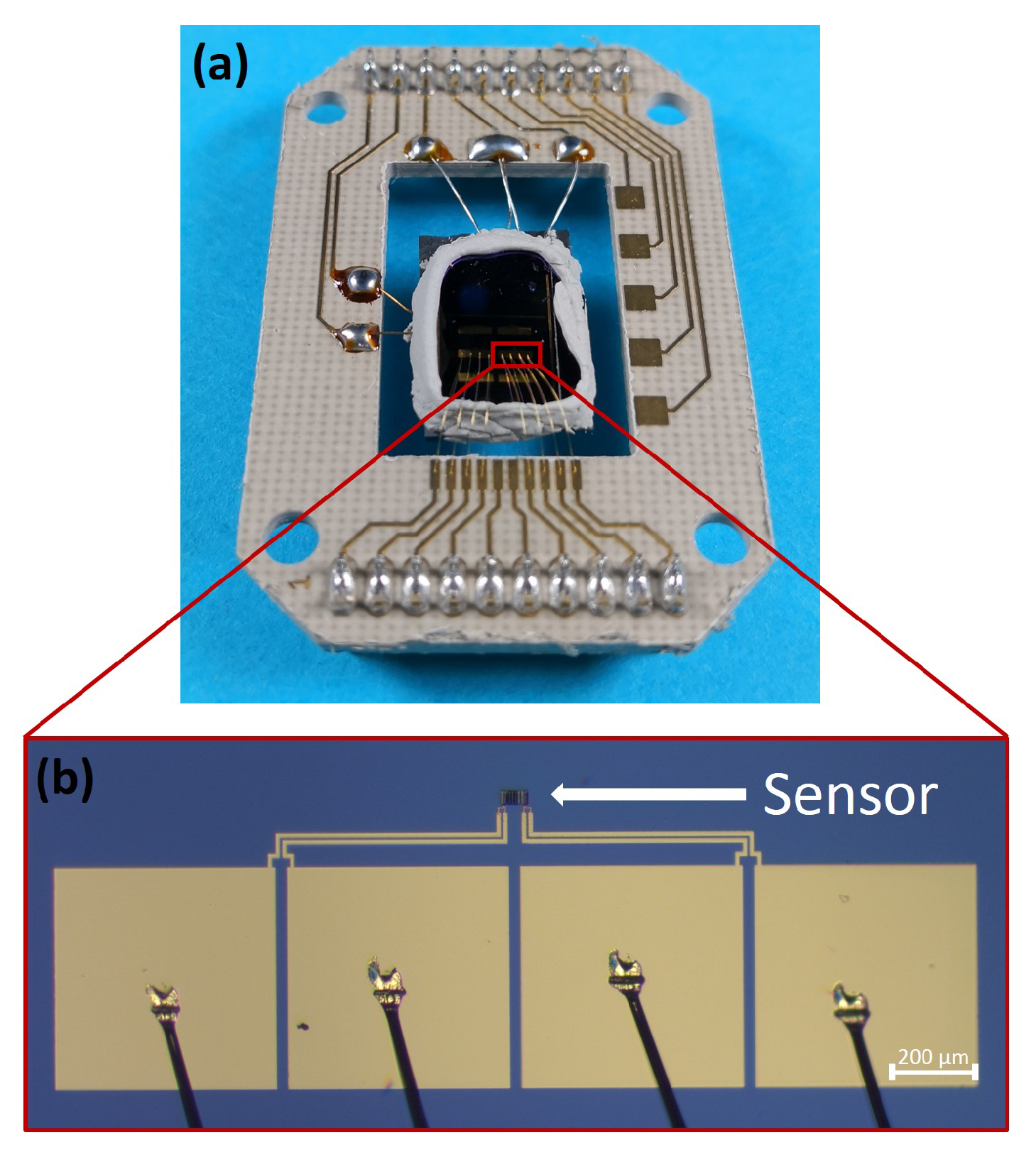
- The base substrate for fabricating the gas sensors is a silicon substrate with a thermal silicon oxide layer of 300 nm on top (SIEGERT WAFER GmbH). Gold electrodes are employed to electrically contact the CuO sensing film. The fabrication of the electrode system is done by photolithography with a positive photoresist (AZ®MIR701, Microchemicals, EVG 620 Mask aligner) and thermal evaporation (Univex Evaporator 450 by Leybold GmbH) of 5 nm thick titanium layer and a gold layer of 200 nm thickness as electrode material. The lift-off process is realized by submerging the substrate in acetone for a duration of 12 h (see Table A2 in appendix for the applied process parameters). The geometry of the electrode system is designed to enable four-point measurement (see Figure 1b) and to ensure precise electrical resistance measurements.
- In the second step the Cu-films are deposited on top of the Ti/Au electrode system. For this a 60 µm by 25 µm structure is exposed by electron beam lithography (Raith eLINE plus, positive electron beam resist AR-P 672.08, ALLRESIST GmbH) into the resist. The structure itself is subdivided into six strips, which are separated by a distance of 2.25 µm. After resist development, a Ti adhesion layer (thickness 5 nm) and a Cu layer (thickness 500 nm) are deposited by thermal evaporation (Univex Evaporator 450, Leybold GmbH). The lift-off process is realized by submerging the entire substrate in acetone for 4 h.
- In the third step the assembly of the gas sensor stack is performed. Metal oxide gas sensors need to be operated at higher temperatures (200 C–400 C). Therefore, the sensor stack, containing the substrate with Ti/Au electrode system and the Cu layer (as precursor for CuO), must be merged with a heater and a thermocouple. The heater consists of two Pt 6.8 elements (Delta-R GmbH) connected in series; a Pt100 element (Delta-R GmbH) is used as the thermocouple to monitor operation temperature. The sensors are glued with both heater elements and thermocouple to a sensor system with thermoconductive adhesive (Aremco Ceramabond 865, Aremco Products Inc.). The final device is soldered, and wire bonded to a custom-made printed circuit board (PCB) as shown in Figure 1a.
- The fourth step addresses the thermal oxidization of the Cu metal layer, which is performed in situ in the gas measurement set-up (GMS). The atmospheric condition in the GMS is provided by synthetic air (Linde Gas GmbH) with relative humidity level (rH) of 50%. For a duration of six hours the gas sensor is heated at a constant temperature of 400 C, which results in oxidation of the Cu layer. The entire Cu metal layer is oxidized to form a continuous CuO film.
- The sensor fabrication is finalized in the fifth step by functionalizing the CuO film with Au nanoparticles (Au-NPs). This is performed by stepwise drop coating a volume of 5 µL of an aqueous Au-NPs solution (CANdot® Series G, c = 100 g Au/mL, Fraunhofer IAP-CAN) onto the sensor surface. To ensure a fast evaporation of the solvent during nanoparticles deposition, the sensor is heated up to 85 C. This is repeated several times; each consecutive step is started once the solvent is evaporated.
2.2. Sensitivity Calculation and Data Evaluation
2.3. Scanning Electron Beam Microscopy
2.4. Raman Spectroscopy
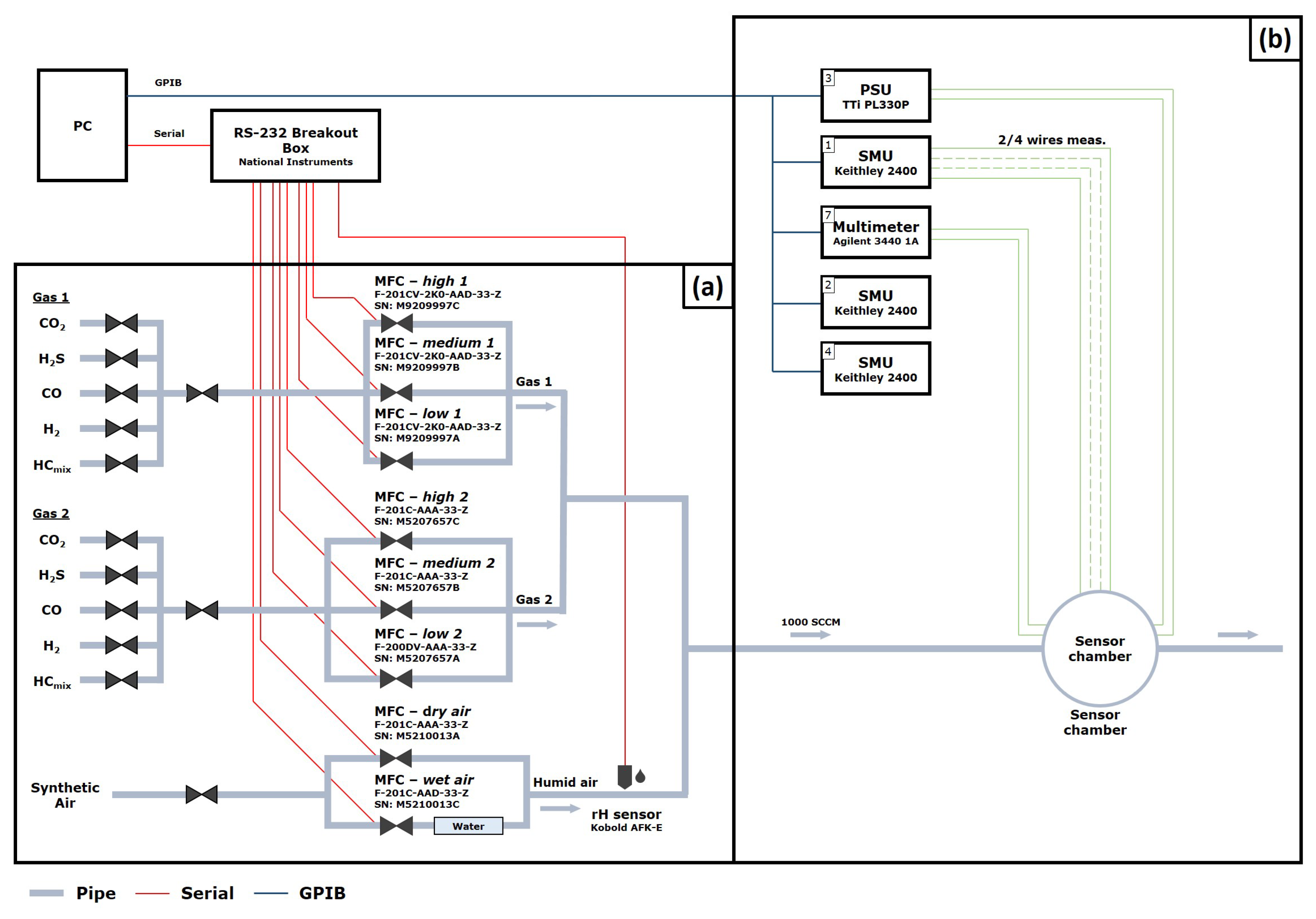
2.5. Gas Measurements Set-Up
3. Results
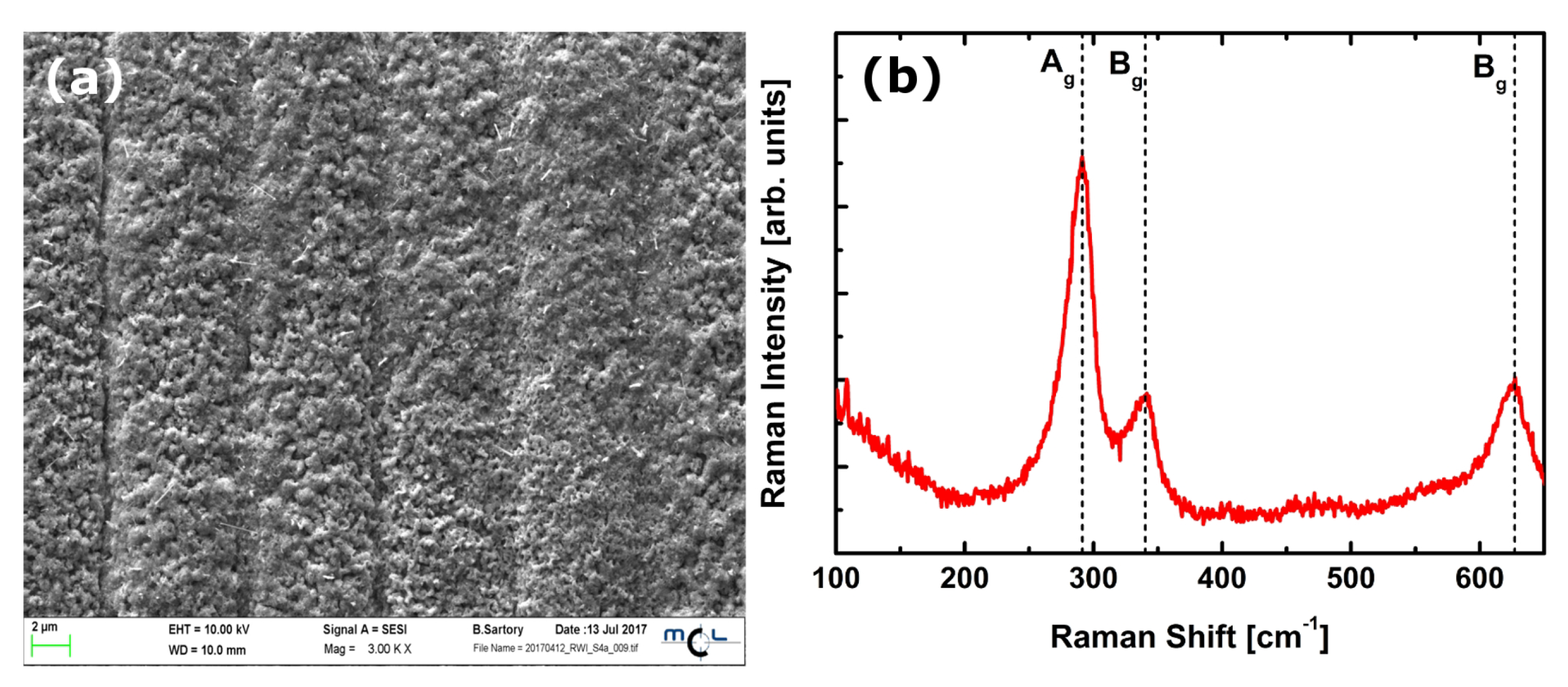
3.1. Copper Oxide Characterization
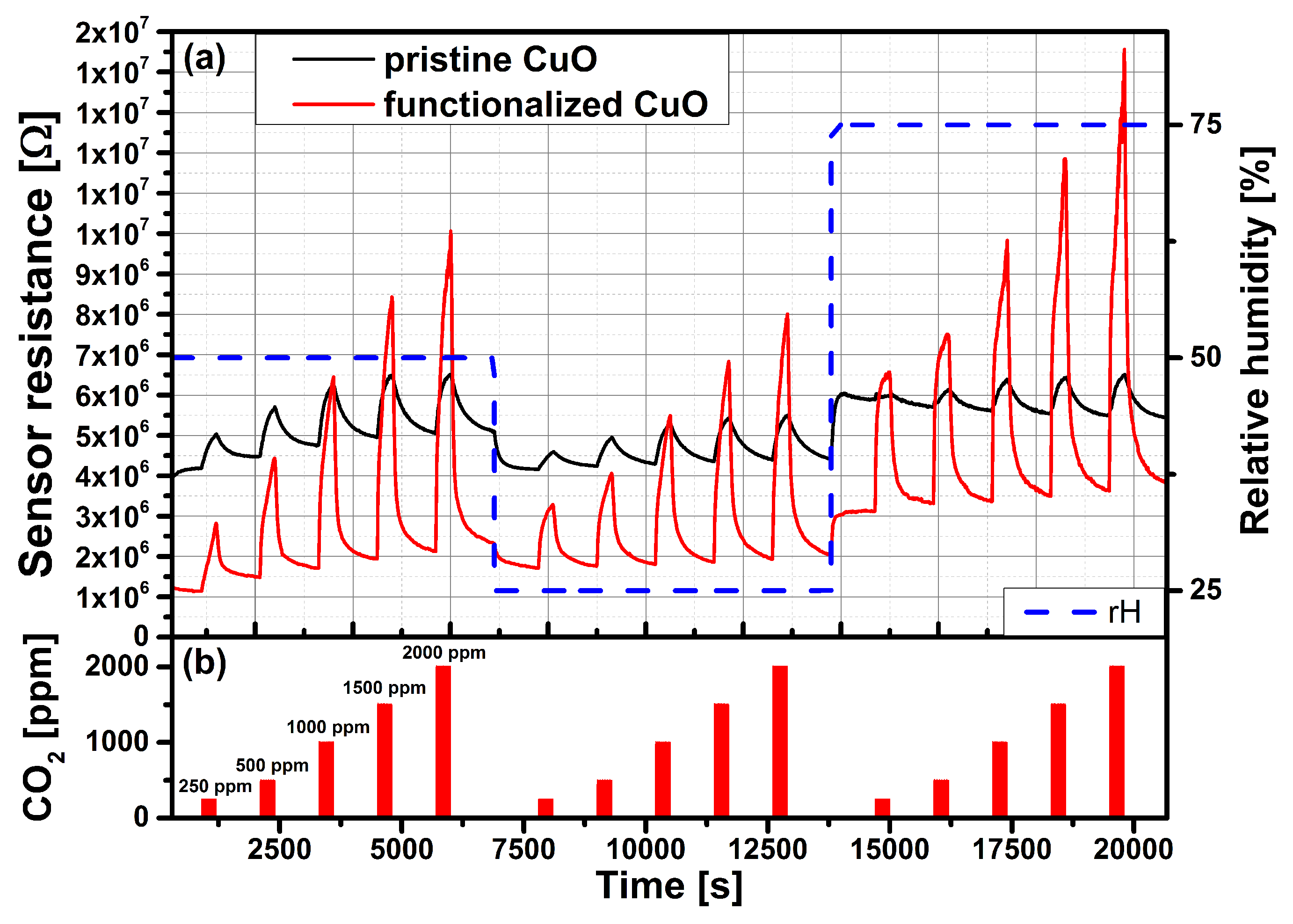
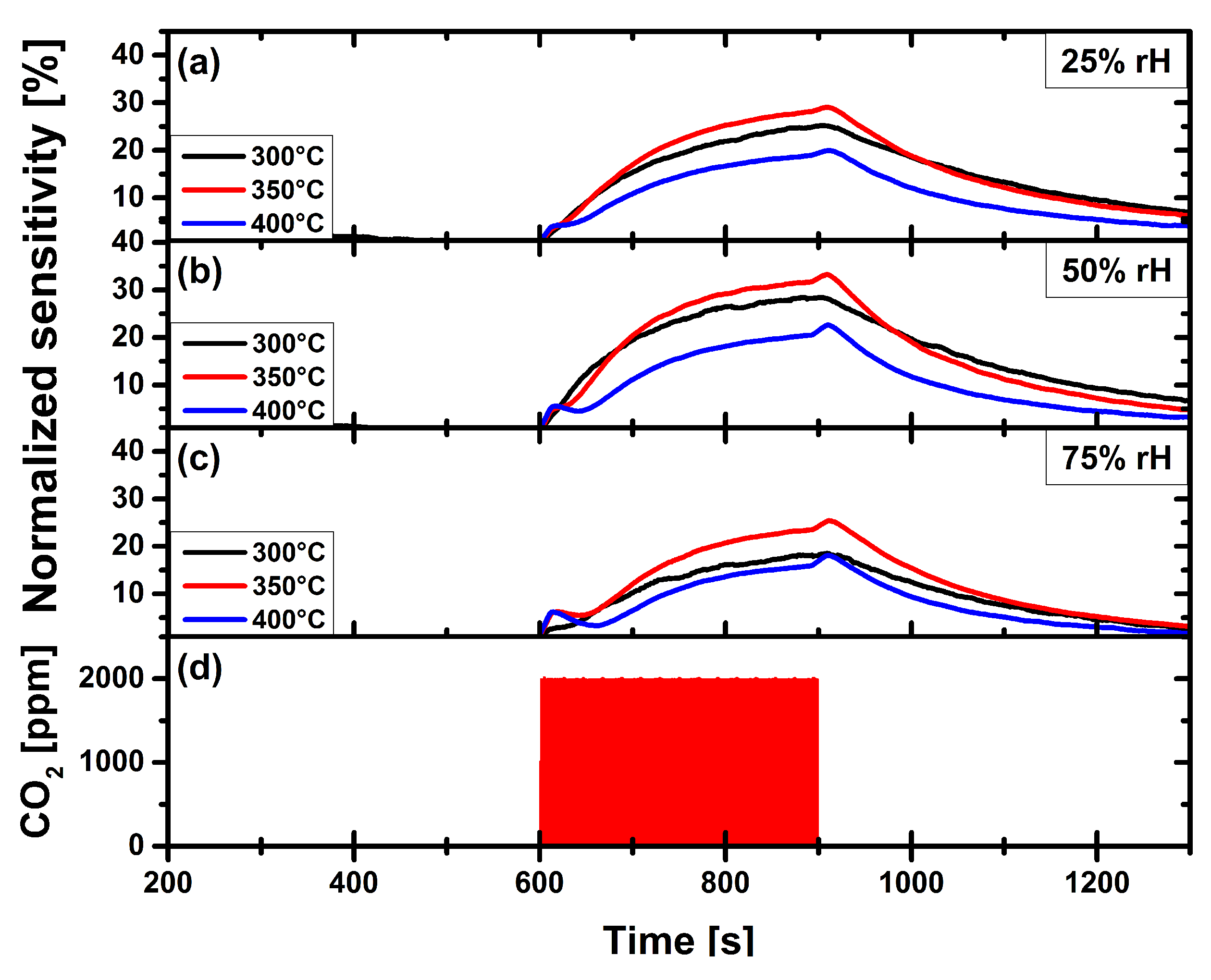
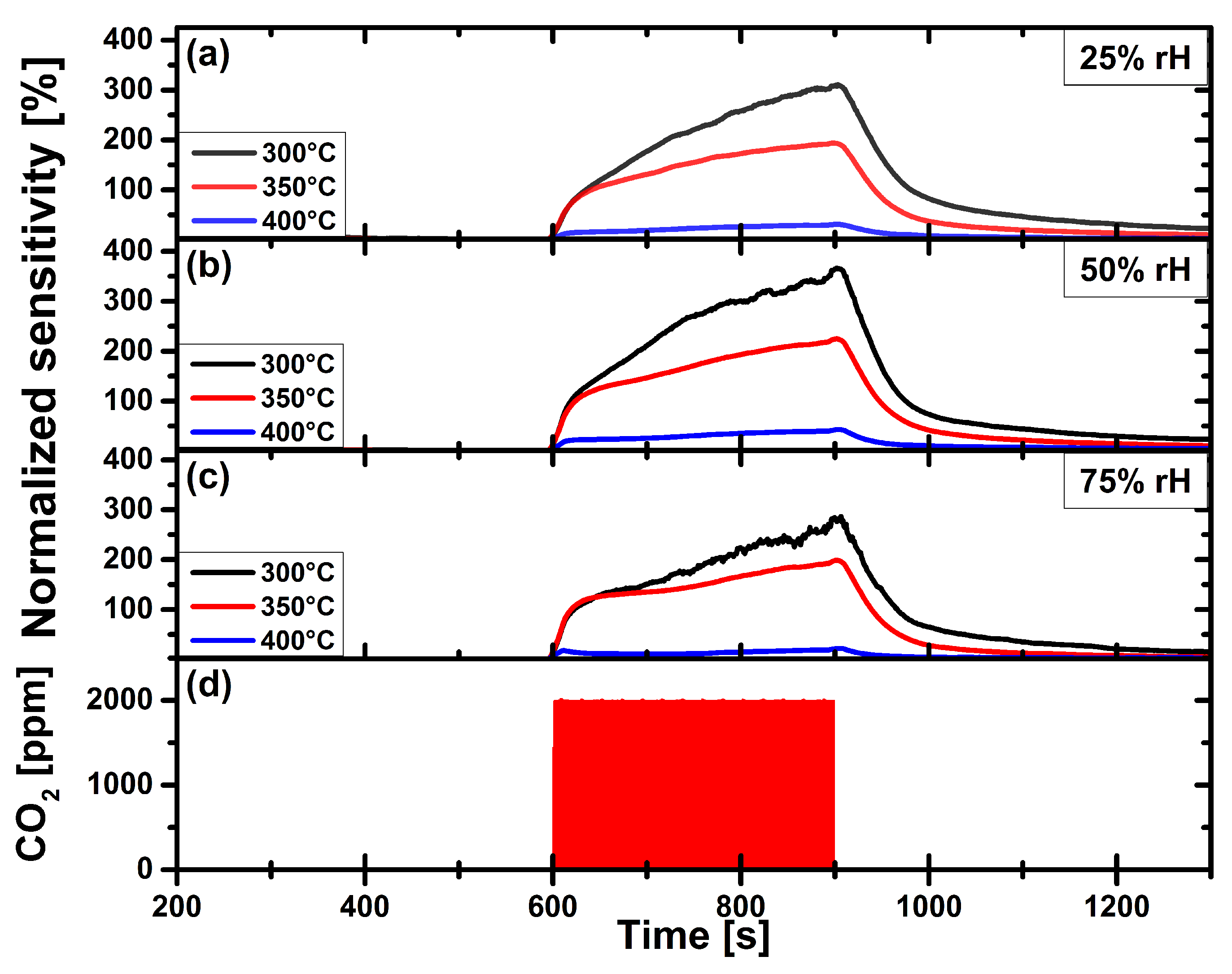
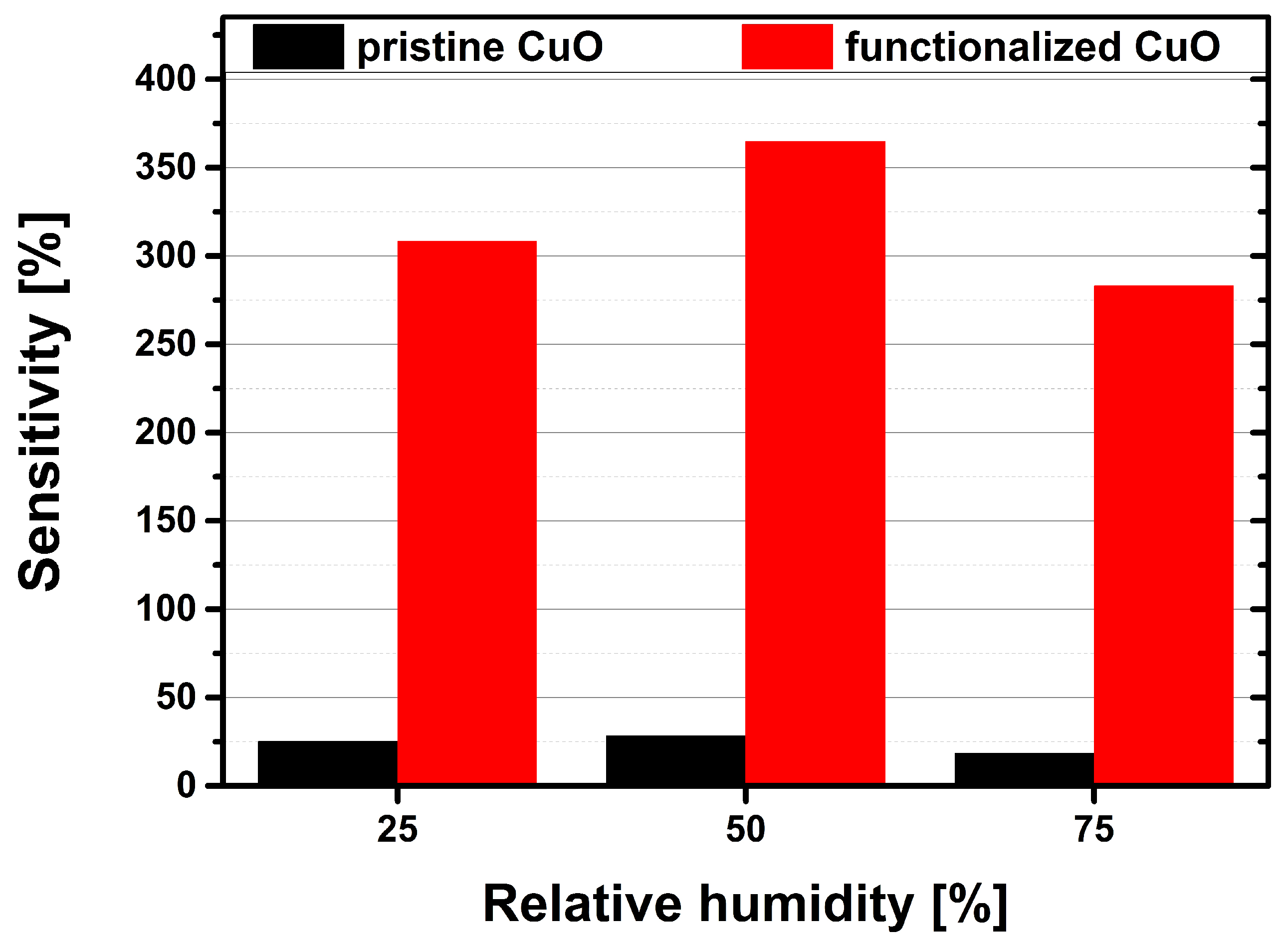
3.2. Carbon Dioxide Measurements of a Pristine and Functionalized Gas Sensor
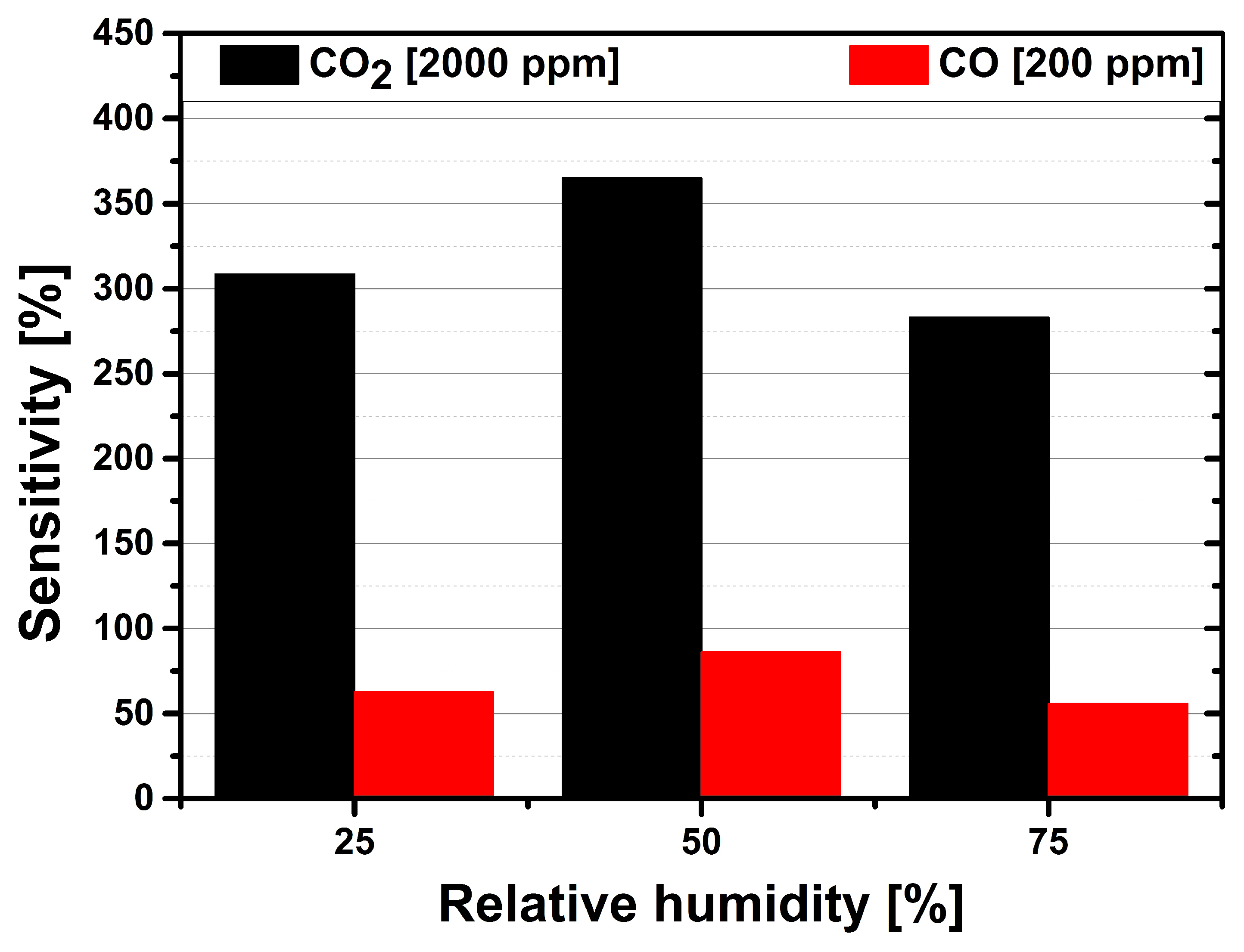
3.3. Carbon Monoxide Measurements
4. Discussion and Conclusions
5. Patents
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| MOx | Metal oxide |
| CO | Carbon dioxide |
| CMOS | Complementary metal oxide semiconductor |
| CuO | Cupric oxide |
| CuO | Cuprous oxide |
| HVAC | Heating, ventilation and air conditioning |
| rH | Relative humidity |
| ppm | Parts per million |
| Au-NP | Gold nanoparticles |
| GMS | Gas measurement set-up |
Appendix A. Supplementary Information
Appendix A.1. Metal Oxide-Based Carbon Dioxide Gas Sensors
Appendix A.2. Photolithography
| (1) Photolithography | |
| Positive photoresist | AZ®MIR701 (Microchemicals GmbH) |
| Spin-coating speed [rpm] | 4000 |
| Spin-coating duration [s] | 35 |
| Annealing temperature [C] | 90 |
| Annealing duration [s] | 60 |
| Developer | AZ®MIF726 (Microchemicals GmbH) |
| Development duration [s] | 30 |
| Stopper | deionized water |
| Stopper duration [s] | 120 |
| (2) Thermal Evaporation | |
| Adhesion layer | titanium |
| Adhesion layer thickness [nm] | 5 |
| Primary layer | gold |
| Primary layer thickness [nm] | 200 |
| (3) Lift-Off | |
| Solvent | acetone |
| Duration [h] | 12 |
Appendix A.3. Electron Beam Lithography
| (1) Electron Beam Lithography | |
| Positive PMMA resist | AR-P 672.08 (ALLRESIST GmbH) |
| Spin-coating speed [rpm] | 2000 |
| Spin-coating duration [s] | 60 |
| Annealing temperature [C] | 180 |
| Annealing duration [s] | 300 |
| Developer | AR600-55 (ALLRESIST GmbH) |
| Development duration [s] | 15 |
| Stopper | AR600-60 (ALLRESIST GmbH) |
| Stopper duration [s] | 60 |
| (2) Thermal Evaporation | |
| Adhesion layer | titanium |
| Adhesion layer thickness [nm] | 5 |
| Primary layer | copper |
| Primary layer thickness [nm] | 500 |
| (3) Lift-Off | |
| Solvent | acetone |
| Duration [h] | 4 |
| (4) Thermal Oxidation | |
| Oxidation temperature [C] | 400 |
| Oxidation duration [h] | 6 |
| Relative humidity [%] | 50 |
| (5) Au Nanoparticles Depositon | |
| Deposition volume [µL] | 40 |
| Deposition temperature [C] | 85 |
Appendix A.4. Equations for Gibbs Free Energy Calculation
- Standard Gibbs free energy for the formation of a compound:G: Change of the standard Gibbs free energy of the examined chemical reaction at standard conditions. Standard conditions refer to a temperature of 298.15 K and a pressure of 1 bar.: Sum over all standard Gibbs free energies of the products.: Sum over all standard Gibbs free energies of the educts.
- Gibbs free energy at varying pressures (malachite):
- Gibbs free energy at varying pressures (azurite):G: Change of Gibbs free energy of formation at a partial pressure varying from standard conditions.G: Change of the standard Gibbs free energy of the examined chemical reaction at standard conditions. Standard conditions refer to a temperature of 298.15 K and a pressure of 1 bar.R: Gas constant. R = 8.314T: Temperature.p: Partial pressure of CO.
- Gibbs free energy of formation for educts and products envolved in the formation of malachite and azurite (standard conditions):Table A4. Gibbs free energy of formation for educts and products of malachite and azurite formation at 298.15 K and 1 bar.
Appendix A.5. Gas Measurement Set-Up
References
- Pachauri, R.K.; Allen, M.R.; Barros, V.R.; Broome, J.; Cramer, W.; Christ, R.; Church, J.A.; Clarke, L.; Dahe, Q.; Dasgupta, P.; et al. Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; IPCC: Geneva, Switzerland, 2014; p. 28. [Google Scholar]
- Ahn, J.; Brook, E.J.; Mitchell, L.; Rosen, J.; McConnell, J.R.; Taylor, K.; Etheridge, D.; Rubino, M. Atmospheric CO2 over the last 1000 years: A high-resolution record from the West Antarctic Ice Sheet (WAIS) Divide ice core. Glob. Biogeochem. Cycles 2011, 26. [Google Scholar] [CrossRef]
- Recent Monthly Average Mauna Loa CO2. 2018. Available online: https://www.esrl.noaa.gov/gmd/ccgg/trends/index.html (accessed on 18 July 2018).
- Seppänen, O.A.; Fisk, W.J.; Mendell, M.J. Association of Ventilation Rates and CO2 Concentrations with Health andOther Responses in Commercial and Institutional Buildings. Indoor Air 1999, 9, 226–252. [Google Scholar] [CrossRef] [PubMed]
- Nassif, N. A robust CO2-based demand-controlled ventilation control strategy for multi-zone HVAC systems. Energy Build. 2012, 45, 72–81. [Google Scholar] [CrossRef]
- Raum-Luftqualitätsfühler QPA10.. QPA20.., 2005. Available online: https://hit.sbt.siemens.com/RWD/DB/DE/de/Assets/39693.pdf (accessed on 18 July 2018).
- Ratterman, M.; Shen, L.; Klotzkin, D.; Papautsky, I. Carbon dioxide luminescent sensor based on a CMOS image array. Sens. Actuators B Chem. 2014, 198, 1–6. [Google Scholar] [CrossRef]
- Wang, J.; Wen, Z.; Yang, B.; Yang, X. Optical carbon dioxide sensor based on fluorescent capillary array. Results Phys. 2017, 7, 323–326. [Google Scholar] [CrossRef]
- Pentyala, V.; Davydovskaya, P.; Ade, M.; Pohle, R.; Urban, G. Carbon dioxide gas detection by open metal site metal organicframeworks and surface functionalized metal organic frameworks. Sens. Actuators B Chem. 2016, 225, 363–368. [Google Scholar] [CrossRef]
- Lorenc, P.; Strzelczyk, A.; Chachulski, B.; Jasinski, G. Properties of Nasicon-based CO2 sensors with Bi8Nb2O17 reference electrode. Solid State Ion. 2015, 271, 48–55. [Google Scholar] [CrossRef]
- Wang, X.; Qin, H.; Sun, L.; Hu, J. CO2 sensing properties and mechanism of nanocrystalline LaFeO3 sensor. Sens. Actuators B Chem. 2013, 188, 965–971. [Google Scholar] [CrossRef]
- Lee, J.; Choi, N.J.; Lee, H.K.; Kim, J.; Lim, S.; Kwon, J.; Lee, S.; Moon, S.; Jong, J.; Yoo, D. Low power consumption solid electrochemical-type micro CO2 gas sensor. Sens. Actuators B Chem. 2017, 248, 957–960. [Google Scholar] [CrossRef]
- Abouda, A.; Al-Keleshb, H.; El Roubyc, W.; Farghalic, A.; Hamdedeinc, A.; Khedrc, M. CO2 responses based on pure and doped CeO2 nano-pellets. J. Mater. Res. Technol. 2018, 7, 14–20. [Google Scholar] [CrossRef]
- Yadav, A.; Lokhande, A.; Kim, J.H.; Lokhande, C. Enhanced sensitivity and selectivity of CO2 gas sensor based on modified La2O3 nanorods. J. Alloys Compd. 2017, 723, 880–886. [Google Scholar] [CrossRef]
- Tanvir, N.; Yurchenko, O.; Laubender, E.; Urban, G. Investigation of low temperature effects on work function based CO2 gas sensing of nanoparticulate CuO films. Sens. Actuators B Chem. 2017, 247, 968–974. [Google Scholar] [CrossRef]
- Tanvir, N.; Yurchenko, O.; Wilbertz, C.; Urban, G. Investigation of CO2 reaction with copper oxide nanoparticles for room temperature gas sensing. J. Mater. Chem. A 2016, 4, 5294–5302. [Google Scholar] [CrossRef]
- Herran, J.; Mandayo, G.; Castano, E. Semiconducting BaTiO3-CuO mixed oxide thin films for CO2 detection. Thin Solids Films 2009, 517, 6192–6197. [Google Scholar] [CrossRef]
- Debbichi, L.; Marco de Lucas, M.; Pierson, J.; Krüger, P. Vibrational Properties of CuO and Cu4O3 from First-Principles Calculations, and Raman and Infrared Spectroscopy. J. Phys. Chem. 2012, 116, 10232–10237. [Google Scholar] [CrossRef]
- Deutsche Forschungsgemeinschaft. MAK– und BAT–Werte–Liste 2013: Maximale Arbeitsplatzkonzentrationen und Biologische Arbeitsstofftoleranzwerte. Mitteilung 49; Wiley VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2013; p. 93. [Google Scholar] [CrossRef]
- Steinhauer, S.; Brunet, E.; Maier, T.; Mutinati, G.; Köck, A. Suspended CuO nanowires for ppb level H2S sensing in dry and humid atmosphere. Sens. Actuators B Chem. 2013, 186, 550–556. [Google Scholar] [CrossRef]
- Chen, M.; Yue, Y.; Ju, Y. Growth of metal and metal oxide nanowires driven by the stress-induced migration. J. Appl. Phys. 2012, 111, 104305. [Google Scholar] [CrossRef]
- Xu, C.H.; Woo, C.H.; Shi, S.Q. Formation of CuO nanowires on Cu foil. Chem. Phys. Lett. 2004, 399, 62–66. [Google Scholar] [CrossRef]
- Adilov, S.R.; Afanaciev, V.P.; Kashkul, I.N.; Kumekov, S.E.; Mukhin, N.V.; Terukov, E.I. Studying the Composition and Structure of Films Obtained by Thermal Oxidation of Copper. Glass Phys. Chem. 2017, 43, 272–275. [Google Scholar] [CrossRef]
- Abdelmounaïm, C.; Amara, Z.; Maha, A.; Mustapha, D. Effects of molarity on structural, optical, morphological and CO2 gas sensing properties of nanostructured copper oxide films deposited by spray pyrolysis. Mater. Sci. Semicond. Process. 2016, 43, 214–221. [Google Scholar] [CrossRef]
- Ishihara, T.; Higuchi, M.; Takagi, T.; Ito, M.; Nishiguchi, H.; Takita, Y. Preparation of CuO thin films on porous BaTiO3 by self-assembled multibilayer film formation and application as a CO2 sensor. J. Mater. Chem. 1998, 8, 2037–2042. [Google Scholar] [CrossRef]
- Seguin, M. Thermogravimetric And Differential Thermal Analysis Of Malachite And Azurite In Inert Atmospheres And In Air. Can. Mineral. 1975, 13, 127–132. [Google Scholar]
- Korotcenkov, G. Handbook of Gas Sensor Materials, 1st ed.; Springer-Verlag New York: New York, NY, USA, 2013; pp. 273–279. [Google Scholar]
- Yang, X.; Kattel, S.; Senanayake, S.; Boscoboinik, J.; Nie, X.; Graciani, J.; Rodriguez, J.; Liu, P.; Stacchiola, D.; Chen, J.G. Low Pressure CO2 Hydrogenation to Methanol over Gold Nanoparticles Activated on a CeOx/TiO2 Interface. J. Am. Chem. Soc. 2015, 137, 10104–10107. [Google Scholar] [CrossRef] [PubMed]
- Iwata, T.; Matsuda, K.; Takahashi, K.; Sawada, K. CO2 Sensing Characteristics of a La2O3/SnO2 Stacked Structure with Micromachined Hotplates. Sensors 2017, 17, 2156. [Google Scholar] [CrossRef] [PubMed]
- Marsal, A.; Dezanneau, G.; Cornet, A.; Morante, J. A new CO2 gas sensing material. Sens. Actuators B Chem. 2003, 95, 266–270. [Google Scholar] [CrossRef]
- Trung, D.; Toan, L.; Hong, H.; Lam, T.; Trung, T.; Hieu, N. Selective detection of carbon dioxide using LaOCl-functionalized SnO2 nanowires for air-quality monitoring. Talanta 2012, 88, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Robie, R.; Waldbaum, D. Thermodynamic properties of minerals and related substances at 298.15 K (25.0 °C) and one atmosphere (1.013 bars) pressure and at higher temperatures. Available online: https://pubs.er.usgs.gov/publication/b1259 (accessed on 22 November 2018).


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wimmer-Teubenbacher, R.; Sosada-Ludwikowska, F.; Zaragoza Travieso, B.; Defregger, S.; Tokmak, O.; Niehaus, J.S.; Deluca, M.; Köck, A. CuO Thin Films Functionalized with Gold Nanoparticles for Conductometric Carbon Dioxide Gas Sensing. Chemosensors 2018, 6, 56. https://doi.org/10.3390/chemosensors6040056
Wimmer-Teubenbacher R, Sosada-Ludwikowska F, Zaragoza Travieso B, Defregger S, Tokmak O, Niehaus JS, Deluca M, Köck A. CuO Thin Films Functionalized with Gold Nanoparticles for Conductometric Carbon Dioxide Gas Sensing. Chemosensors. 2018; 6(4):56. https://doi.org/10.3390/chemosensors6040056
Chicago/Turabian StyleWimmer-Teubenbacher, Robert, Florentyna Sosada-Ludwikowska, Bernat Zaragoza Travieso, Stefan Defregger, Oeznur Tokmak, Jan Steffen Niehaus, Marco Deluca, and Anton Köck. 2018. "CuO Thin Films Functionalized with Gold Nanoparticles for Conductometric Carbon Dioxide Gas Sensing" Chemosensors 6, no. 4: 56. https://doi.org/10.3390/chemosensors6040056
APA StyleWimmer-Teubenbacher, R., Sosada-Ludwikowska, F., Zaragoza Travieso, B., Defregger, S., Tokmak, O., Niehaus, J. S., Deluca, M., & Köck, A. (2018). CuO Thin Films Functionalized with Gold Nanoparticles for Conductometric Carbon Dioxide Gas Sensing. Chemosensors, 6(4), 56. https://doi.org/10.3390/chemosensors6040056






