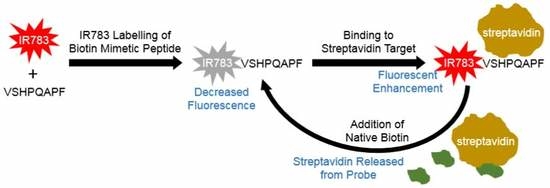
IR-783 Labeling of a Peptide Receptor for ‘Turn-On’ Fluorescence Based Sensing
Abstract
1. Introduction
2. Materials and Methods
3. Results
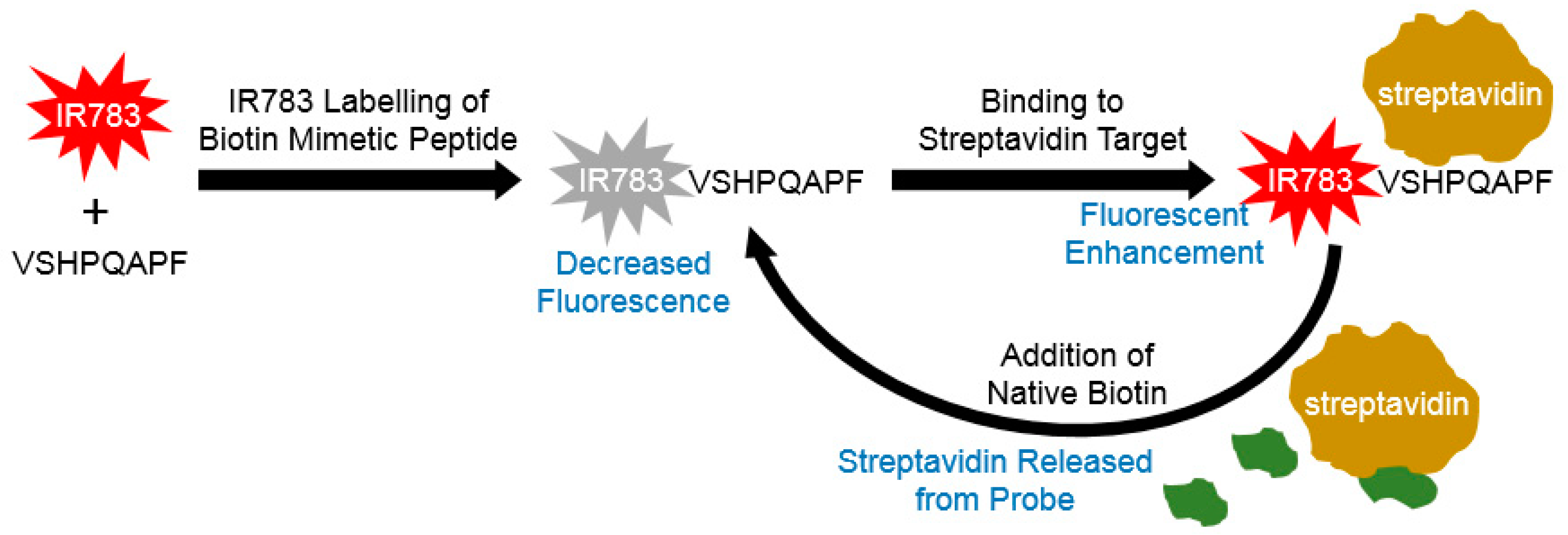
3.1. Spectra of IR-783 Conjugated VSHPQAPF
3.2. Properties of IR-783 Conjugated VSHPQAPF
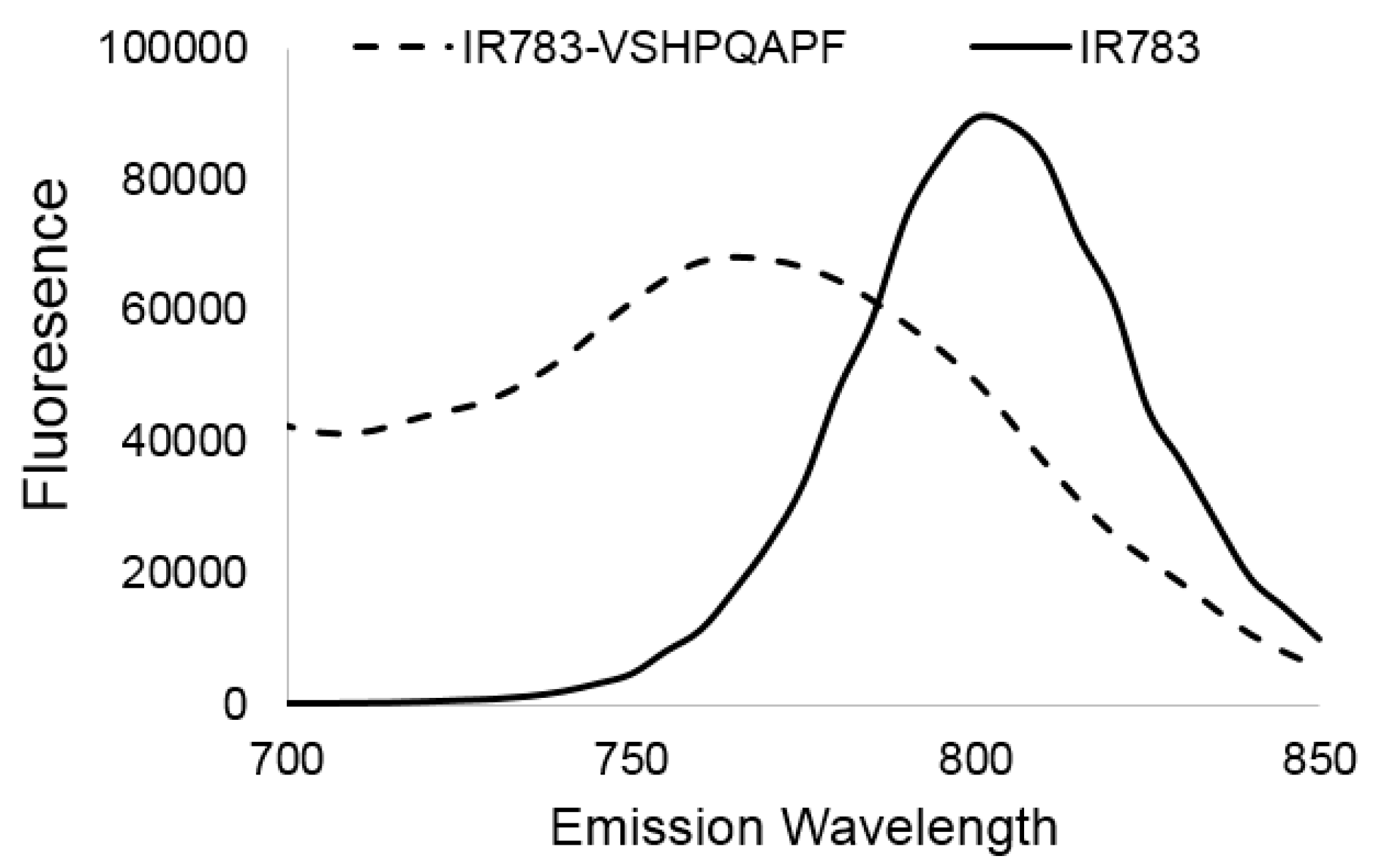
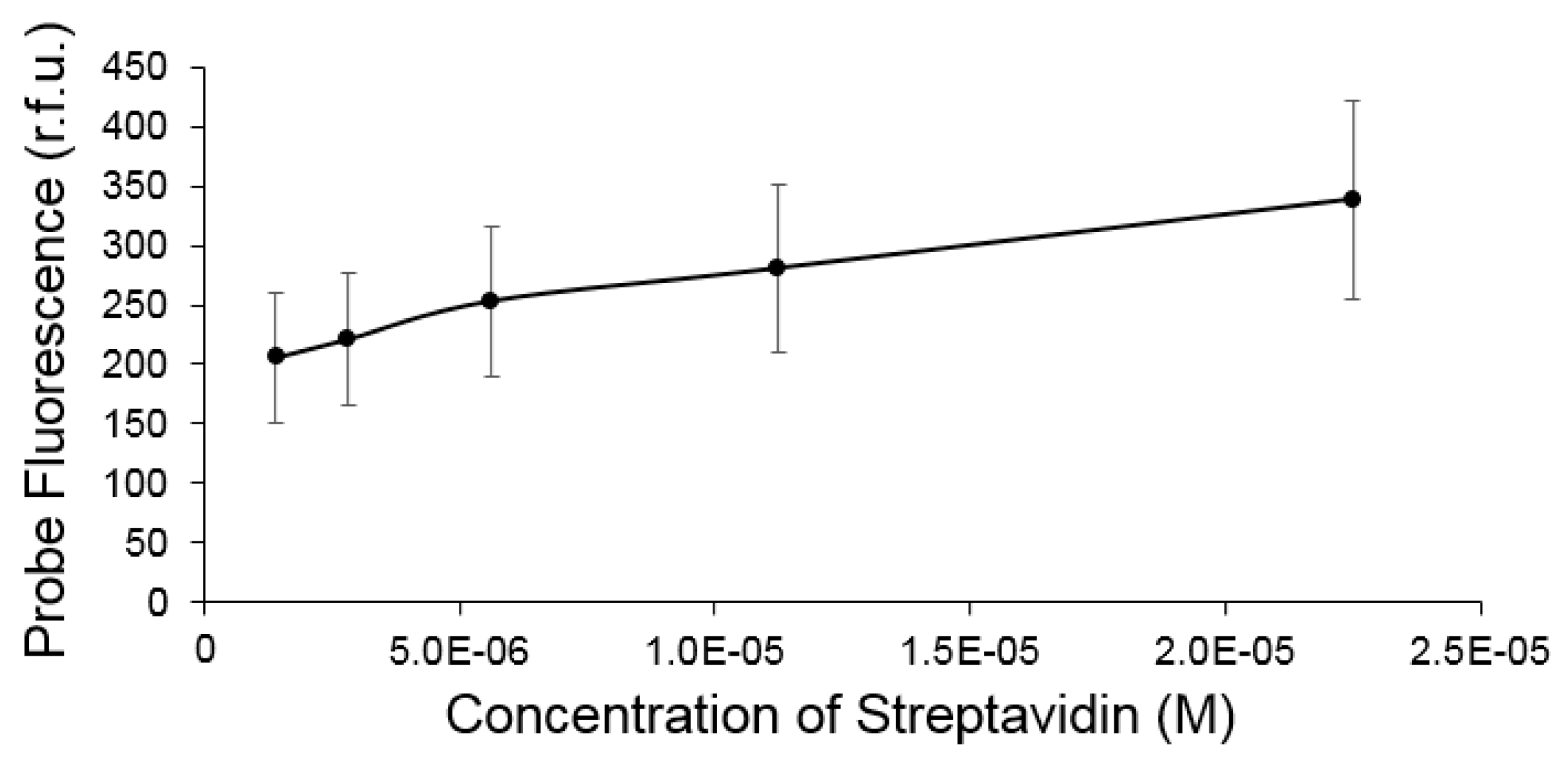
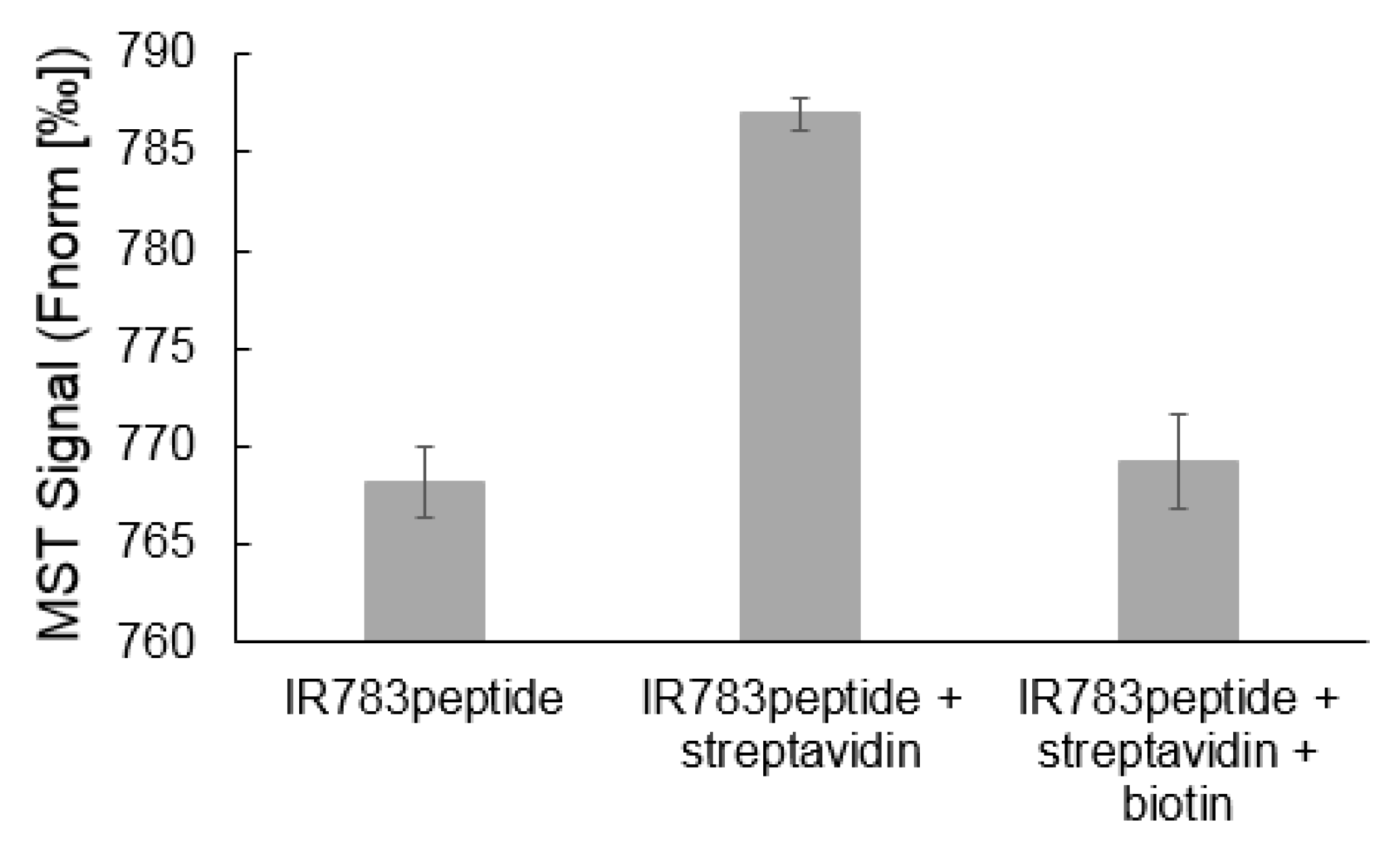
3.2.1. Target Binding Ability
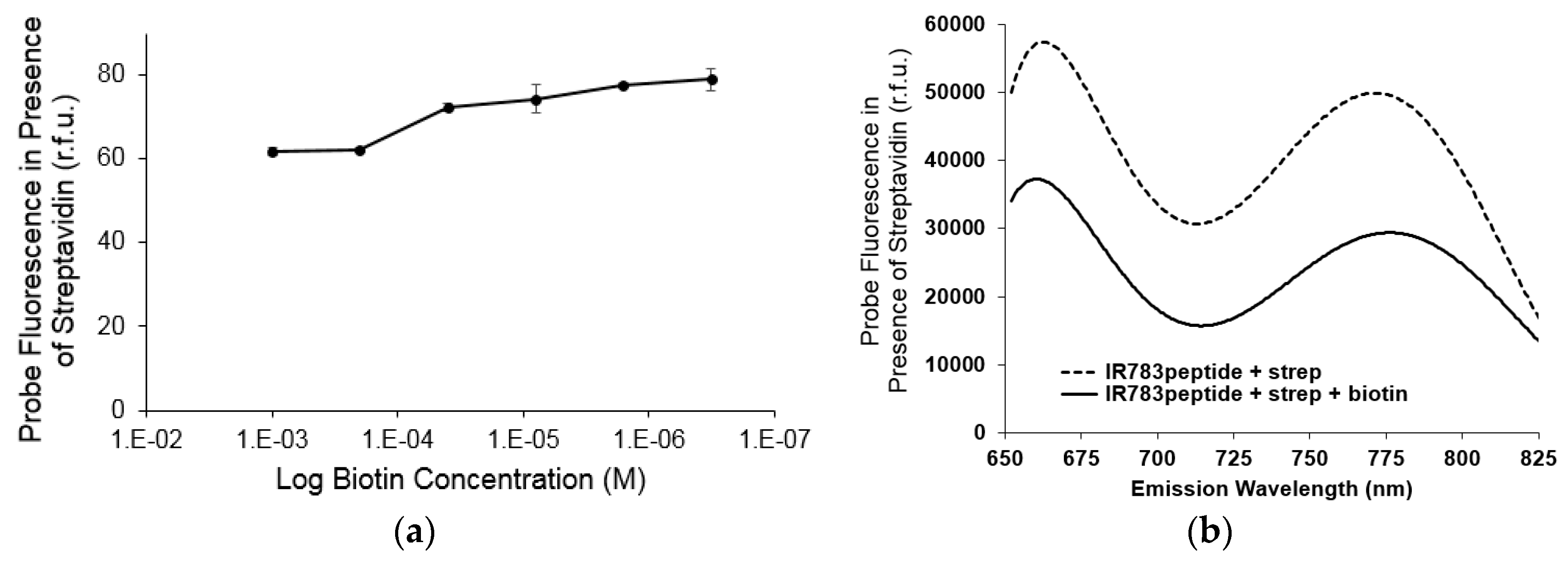
3.2.2. Fluorescence Enhancement
3.2.3. Reversibility of Probe Enhancement
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Zhang, E.; Luo, S.; Tan, X.; Shi, C. Mechanistic study of IR-780 dye as a potential tumor targeting and drug delivery agent. Biomaterials 2014, 35, 771–778. [Google Scholar] [CrossRef] [PubMed]
- Kiyose, K.; Aizawa, S.; Sasaki, E.; Kojima, H.; Hanaoka, K.; Terai, T.; Urano, Y.; Nagano, T. Molecular design strategies for near-infrared ratiometric fluorescent probes based on the unique spectral properties of aminocyanines. Chem. A Eur. J. 2009, 15, 9191–9200. [Google Scholar] [CrossRef] [PubMed]
- Hilderbrand, S.A.; Kelly, K.A.; Weissleder, R.; Tung, C.-H. Monofunctional near-infrared fluorochromes for imaging applications. Bioconj. Chem. 2005, 16, 1275–1281. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Shi, C.; Tong, R.; Qian, W.; Zhau, H.E.; Wang, R.; Zhu, G.; Cheng, J.; Yang, V.W.; Cheng, T.; et al. Near IR heptamethine cyanine dye–mediated cancer imaging. Clin. Cancer Res. 2010, 16, 2833–2844. [Google Scholar] [CrossRef] [PubMed]
- Licha, K.; Riefke, B.; Ebert, B.; Grötzinger, C. Cyanine dyes as contrast agents in biomedical optical imaging. Acad. Radiol. 2002, 9, 320S–322S. [Google Scholar] [CrossRef]
- Li, C.; Greenwood, T.R.; Bhujwalla, Z.M.; Glunde, K. Synthesis and characterization of glucosamine-bound near-infrared probes for optical imaging. Org. Lett. 2006, 8, 3623–3626. [Google Scholar] [CrossRef] [PubMed]
- Kuang, Y.; Zhang, K.; Cao, Y.; Chen, X.; Wang, K.; Liu, M.; Pei, R. Hydrophobic IR-780 dye encapsulated in cRGD-conjugated solid lipid nanoparticles for NIR imaging-guided photothermal therapy. ACS Appl. Mater. Interfaces 2017, 9, 12217–12226. [Google Scholar] [CrossRef] [PubMed]
- Cheng, G.; Fan, J.; Sun, W.; Cao, J.; Hu, C.; Peng, X. A near-infrared fluorescent probe for selective detection of HClO based on Se-sensitized aggregation of heptamethine cyanine dye. Chem. Commun. 2014, 50, 1018–1020. [Google Scholar] [CrossRef] [PubMed]
- Butte, P.V.; Mamelak, A.; Parrish-Novak, J.; Drazin, D.; Shweikeh, F.; Gangalum, P.R.; Chesnokova, A.; Ljubimova, J.Y.; Black, K. Near-infrared imaging of brain tumors using the Tumor Paint BLZ-100 to achieve near-complete resection of brain tumors. Neurosurg. Focus 2014, 36, E1. [Google Scholar] [CrossRef] [PubMed]
- Kiyose, K.; Kojima, H.; Urano, Y.; Nagano, T. Development of a ratiometric fluorescent zinc ion probe in near-infrared region, based on tricarbocyanine chromophore. J. Am. Chem. Soc. 2006, 128, 6548–6549. [Google Scholar] [CrossRef] [PubMed]
- Mujumdar, R.B.; Ernst, L.A.; Mujumdar, S.R.; Waggoner, A.S. Cyanine dye labeling reagents containing isothiocyanate groups. Cytom. J. Int. Soc. Anal. Cytol. 1989, 10, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Song, F.; Lu, E.; Wang, Y.; Zhou, W.; Fan, J.; Gao, Y. Heptamethine cyanine dyes with a large stokes shift and strong fluorescence: A paradigm for excited-state intramolecular charge transfer. J. Am. Chem. Soc. 2005, 127, 4170–4171. [Google Scholar] [CrossRef] [PubMed]
- Ellis, A.L.; Mason, J.C.; Lee, H.-W.; Strekowski, L.; Patonay, G.; Choi, H.; Yang, J.J. Design, synthesis, and characterization of a calcium-sensitive near infrared dye. Talanta 2002, 56, 1099–1107. [Google Scholar] [CrossRef]
- Górecki, T.; Patonay, G.; Strekowski, L.; Chin, R.; Salazar, N. Synthesis of novel near-infrared cyanine dyes for metal ion determination. J. Heterocycl. Chem. 1996, 33, 1871–1876. [Google Scholar] [CrossRef]
- Harrison, V.S.; Carney, C.E.; MacRenaris, K.W.; Waters, E.A.; Meade, T.J. Multimeric near IR–MR contrast agent for multimodal in vivo imaging. J. Am. Chem. Soc. 2015, 137, 9108–9116. [Google Scholar] [CrossRef] [PubMed]
- Lv, W.; Shen, Y.; Yang, H.; Yang, R.; Cai, W.; Zhang, J.; Yuan, L.; Duan, Y.; Zhang, L. A Novel Bimodal Imaging Agent Targeting HER2 Molecule of Breast Cancer. J. Immunol. Res. 2018, 2018. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Yu, F.; Lv, C.; Choo, J.; Chen, L. Fluorescent chemical probes for accurate tumor diagnosis and targeting therapy. Chem. Soc. Rev. 2017, 46, 2237–2271. [Google Scholar] [CrossRef] [PubMed]
- Tung, C.H. Fluorescent peptide probes for in vivo diagnostic imaging. Peptide Sci. Orig. Res. Biomol. 2004, 76, 391–403. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Wu, J.; Di, C.; Zhou, R.; Zhang, H.; Su, P.; Xu, C.; Zhou, P.; Ge, Y.; Liu, D.; et al. A novel peptide-based fluorescence chemosensor for selective imaging of hydrogen sulfide both in living cells and zebrafish. Biosens. Bioelectron. 2017, 92, 602–609. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Wang, J.; Boyd, B.J. Peptide-based biosensors. Talanta 2015, 136, 114–127. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Liu, L.; Zhou, P.; Wu, W.; Wu, J.; Liu, W.; Tang, Y. A peptide-based fluorescent chemosensor for multianalyte detection. Biosens. Bioelectron. 2015, 72, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Pazos, E.; Vazquez, O.; Mascarenas, J.L.; Vazquez, M.E. Peptide-based fluorescent biosensors. Chem. Soc. Rev. 2009, 38, 3348–3359. [Google Scholar] [CrossRef] [PubMed]
- Rajarao, G.K.; Nekhotiaeva, N.; Good, L. Peptide-mediated delivery of green fluorescent protein into yeasts and bacteria. FEMS Microbiol. Lett. 2002, 215, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Hermanson, G.T. Bioconjugate Techniques; Academic Press: Cambridge, MA, USA, 2013. [Google Scholar]
- Bouteiller, C.; Clavé, G.; Bernardin, A.; Chipon, B.; Massonneau, M.; Renard, P.-Y.; Romieu, A. Novel water-soluble near-infrared cyanine dyes: Synthesis, spectral properties, and use in the preparation of internally quenched fluorescent probes. Bioconj. Chem. 2007, 18, 1303–1317. [Google Scholar] [CrossRef] [PubMed]
- Hong, V.; Kislukhin, A.A.; Finn, M. Thiol-selective fluorogenic probes for labeling and release. J. Am. Chem. Soc. 2009, 131, 9986–9994. [Google Scholar] [CrossRef] [PubMed]
- Bartczak, D.; Kanaras, A.G. Preparation of peptide-functionalized gold nanoparticles using one pot EDC/sulfo-NHS coupling. Langmuir 2011, 27, 10119–10123. [Google Scholar] [CrossRef] [PubMed]
- De León-Rodríguez, L.M.; Kovacs, Z. The synthesis and chelation chemistry of DOTA—Peptide conjugates. Bioconj. Chem. 2007, 19, 391–402. [Google Scholar] [CrossRef] [PubMed]
- Pap, E.H.; Dansen, T.B.; van Summeren, R.; Wirtz, K.W. Peptide-based targeting of fluorophores to organelles in living cells. Exp. Cell Res. 2001, 265, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Pap, E.H.; Dansen, T.B.; Wirtz, K.W. Peptide-based targeting of fluorophores to peroxisomes in living cells. Trends Cell Biol. 2001, 11, 10–12. [Google Scholar] [CrossRef]
- Liu, S.; Edwards, D.S. Bifunctional chelators for therapeutic lanthanide radiopharmaceuticals. Bioconj. Chem. 2001, 12, 7–34. [Google Scholar] [CrossRef]
- Li, G.; Xing, Y.; Wang, J.; Conti, P.S.; Chen, K. Near-infrared fluorescence imaging of CD13 receptor expression using a novel Cy5. 5-labeled dimeric NGR peptide. Amino Acids 2014, 46, 1547–1556. [Google Scholar] [CrossRef] [PubMed]
- Schmuck, C.; Heil, M.; Scheiber, J.; Baumann, K. Charge interactions do the job: A combined statistical and combinatorial approach to finding artificial receptors for binding tetrapeptides in water. Angew. Chem. Int. Ed. 2005, 44, 7208–7212. [Google Scholar] [CrossRef] [PubMed]
- Skerra, A.; Schmidt, T.G. Applications of a peptide ligand for streptavidin: The Strep-tag. Biomol. Eng. 1999, 16, 79–86. [Google Scholar] [CrossRef]
- Hundsberger, H.; Önder, K.; Schuller-Götzburg, P.; Virok, D.P.; Herzog, J.; Rid, R. Assembly and use of high-density recombinant peptide chips for large-scale ligand screening is a practical alternative to synthetic peptide libraries. BMC Genom. 2017, 18, 450. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, T.G.; Koepke, J.; Frank, R.; Skerra, A. Molecular interaction between the Strep-tag affinity peptide and its cognate target, streptavidin. J. Mol. Biol. 1996, 255, 753–766. [Google Scholar] [CrossRef] [PubMed]
- Jerabek-Willemsen, M.; Wienken, C.J.; Braun, D.; Baaske, P.; Duhr, S. Molecular interaction studies using microscale thermophoresis. Assay Drug Dev. Technol. 2011, 9, 342–353. [Google Scholar] [CrossRef] [PubMed]
- Awasthi, K.; Nishimura, G. Modification of near-infrared cyanine dyes by serum albumin protein. Photochem. Photobiol. Sci. 2011, 10, 461–463. [Google Scholar] [CrossRef] [PubMed]
- Lou, Z.; Li, P.; Song, P.; Han, K. Ratiometric fluorescence imaging of cellular hypochlorous acid based on heptamethine cyanine dyes. Analyst 2013, 138, 6291–6295. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Yan, M.; Fan, X.-X.; Sun, D.; Yang, S.-Y.; Yang, L.-J.; Li, J.-D.; Jiang, Y.-B. A heptamethine cyanine-based colorimetric and ratiometric fluorescent chemosensor for the selective detection of Ag+ in an aqueous medium. Chem. Commun. 2012, 48, 2243–2245. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Duan, X.; Chen, Z.; Liu, Y.; Xie, T.; Fang, L.; Li, X.; Yin, M.; Tang, B. A near-infrared fluorescent probe for detecting copper (II) with high selectivity and sensitivity and its biological imaging applications. Chem. Commun. 2011, 47, 7755–7757. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Ahsan, S.S.; Santiago-Berrios, M.E.B.; Abruña, H.D.; Webb, W.W. Mechanisms of quenching of Alexa fluorophores by natural amino acids. J. Am. Chem. Soc. 2010, 132, 7244–7245. [Google Scholar] [CrossRef] [PubMed]
- Götz, M.; Hess, S.; Beste, G.; Skerra, A.; Michel-Beyerle, M. Ultrafast electron transfer in the complex between fluorescein and a cognate engineered lipocalin protein, a so-called anticalin. Biochemistry 2002, 41, 4156–4164. [Google Scholar] [CrossRef] [PubMed]
- Vaiana, A.C.; Neuweiler, H.; Schulz, A.; Wolfrum, J.; Sauer, M.; Smith, J.C. Fluorescence quenching of dyes by tryptophan: Interactions at atomic detail from combination of experiment and computer simulation. J. Am. Chem. Soc. 2003, 125, 14564–14572. [Google Scholar] [CrossRef] [PubMed]
- Song, F.; Peng, X.; Lu, E.; Wang, Y.; Zhou, W.; Fan, J. Tuning the photoinduced electron transfer in near-infrared heptamethine cyanine dyes. Tetrahedron Lett. 2005, 46, 4817–4820. [Google Scholar] [CrossRef]
- Rodriguez, P.L.; Harada, T.; Christian, D.A.; Pantano, D.A.; Tsai, R.K.; Discher, D.E. Minimal “Self” peptides that inhibit phagocytic clearance and enhance delivery of nanoparticles. Science 2013, 339, 971–975. [Google Scholar] [CrossRef] [PubMed]
- Sparano, B.A.; Koide, K. A strategy for the development of small-molecule-based sensors that strongly fluoresce when bound to a specific RNA. J. Am. Chem. Soc. 2005, 127, 14954–14955. [Google Scholar] [CrossRef] [PubMed]
- Katz, B.A. Binding to protein targets of peptidic leads discovered by phage display: Crystal structures of streptavidin-bound linear and cyclic peptide ligands containing the HPQ sequence. Biochemistry 1995, 34, 15421–15429. [Google Scholar] [CrossRef] [PubMed]
- Sepunaru, L.; Refaely-Abramson, S.; Lovrinčić, R.; Gavrilov, Y.; Agrawal, P.; Levy, Y.; Kronik, L.; Pecht, I.; Sheves, M.; Cahen, D. Electronic transport via homopeptides: The role of side chains and secondary structure. J. Am. Chem. Soc. 2015, 137, 9617–9626. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwak, E.-A.; Kydd, L.; Lim, B.; Jaworski, J. IR-783 Labeling of a Peptide Receptor for ‘Turn-On’ Fluorescence Based Sensing. Chemosensors 2018, 6, 47. https://doi.org/10.3390/chemosensors6040047
Kwak E-A, Kydd L, Lim B, Jaworski J. IR-783 Labeling of a Peptide Receptor for ‘Turn-On’ Fluorescence Based Sensing. Chemosensors. 2018; 6(4):47. https://doi.org/10.3390/chemosensors6040047
Chicago/Turabian StyleKwak, Eun-A, LeNaiya Kydd, Butaek Lim, and Justyn Jaworski. 2018. "IR-783 Labeling of a Peptide Receptor for ‘Turn-On’ Fluorescence Based Sensing" Chemosensors 6, no. 4: 47. https://doi.org/10.3390/chemosensors6040047
APA StyleKwak, E.-A., Kydd, L., Lim, B., & Jaworski, J. (2018). IR-783 Labeling of a Peptide Receptor for ‘Turn-On’ Fluorescence Based Sensing. Chemosensors, 6(4), 47. https://doi.org/10.3390/chemosensors6040047