Simultaneous Analysis of Sensor Data for Breath Control in Respiratory Air
Abstract
:1. Introduction
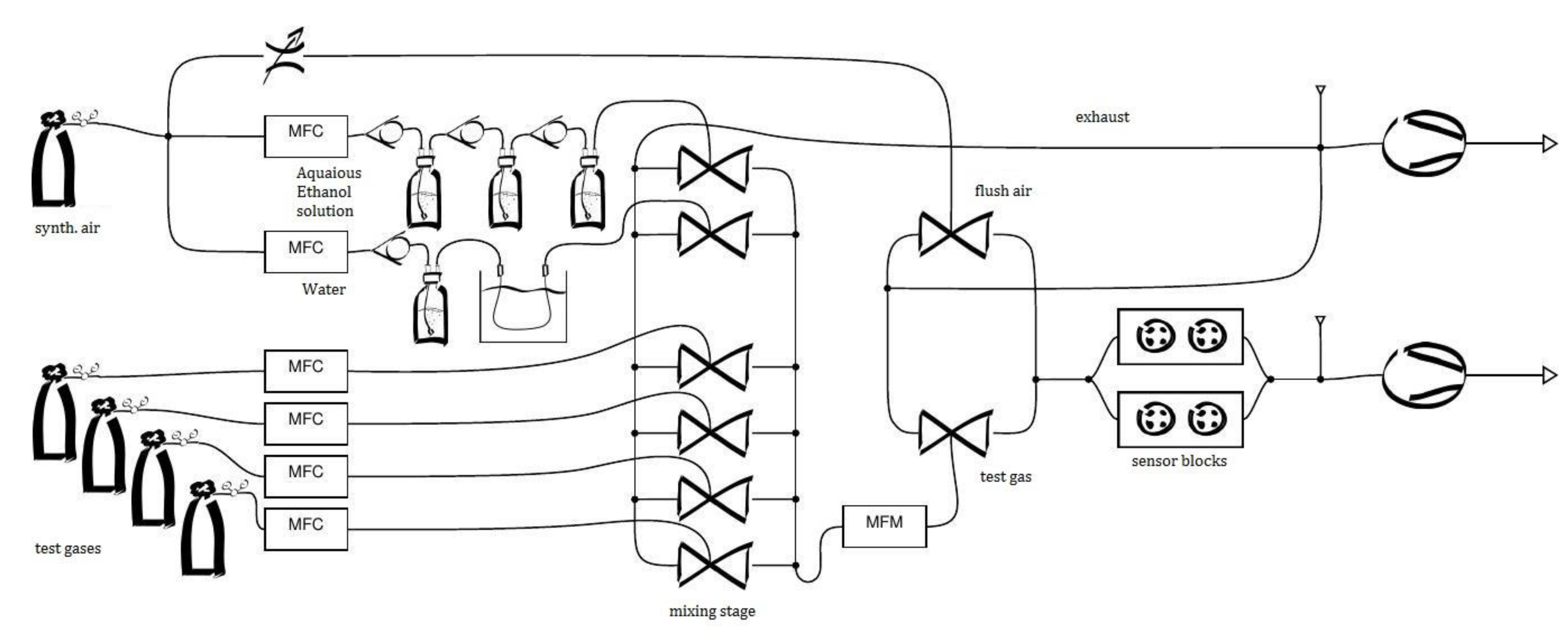
2. Mobile Sensor System AGaMon
3. Concept of SimPlus
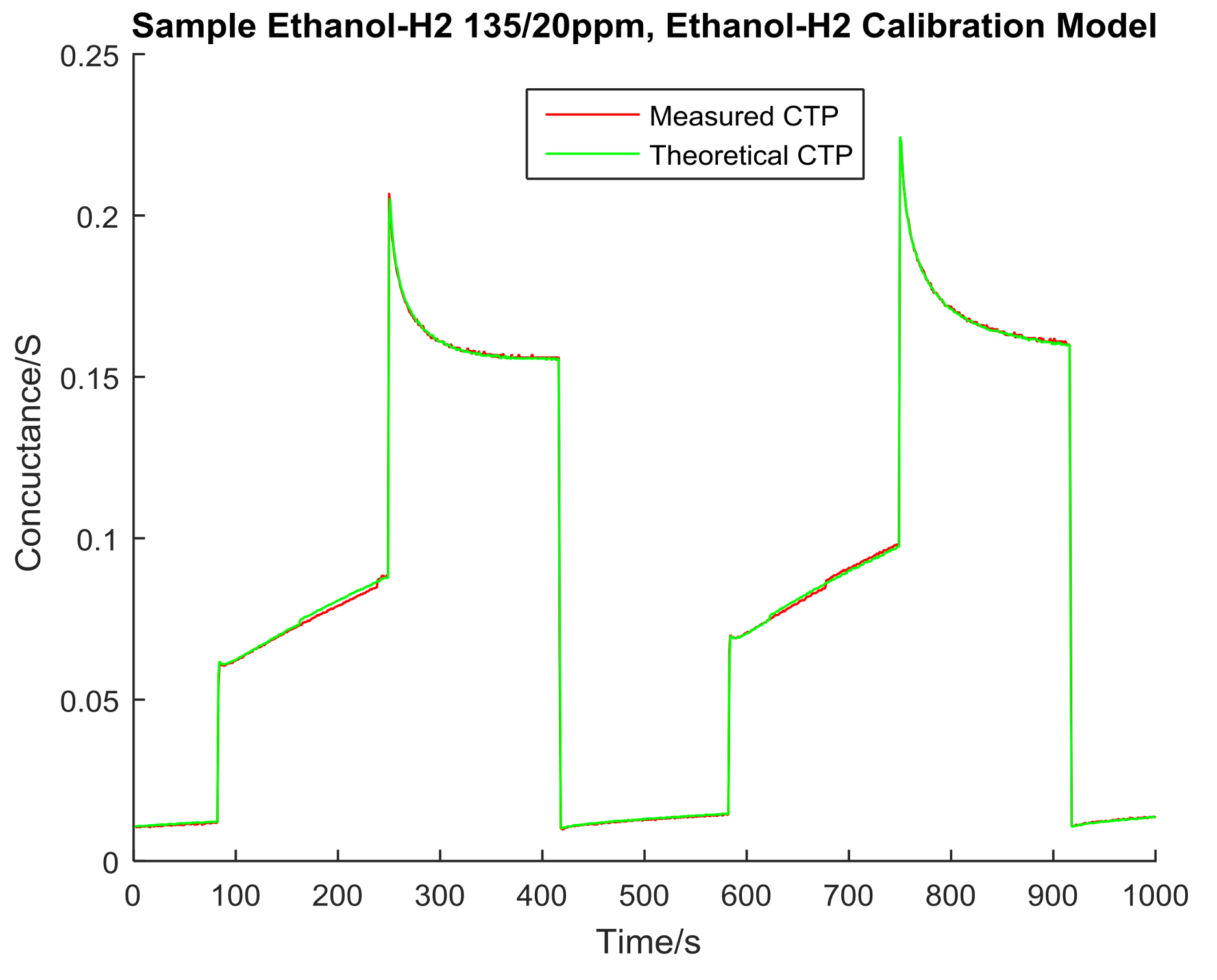
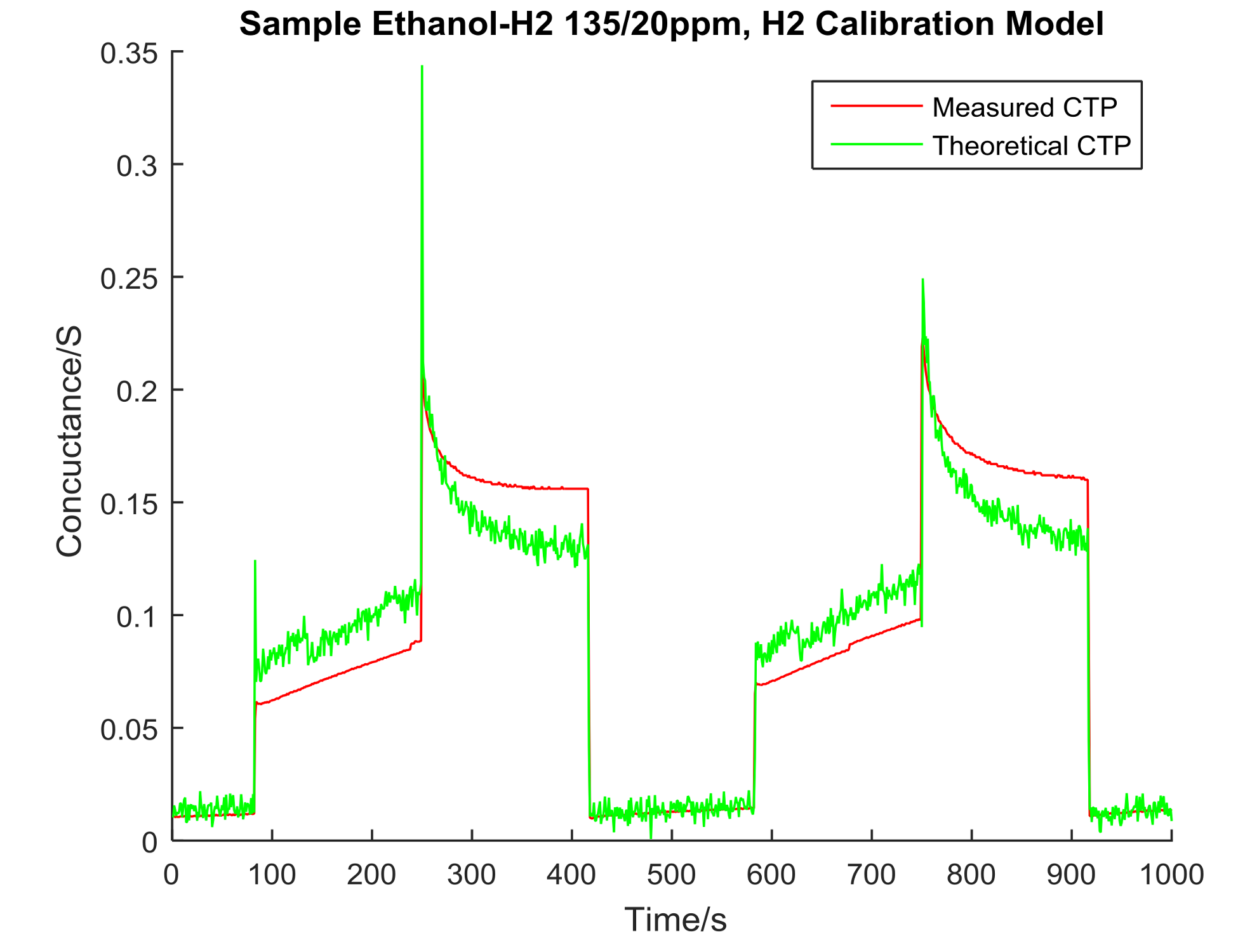
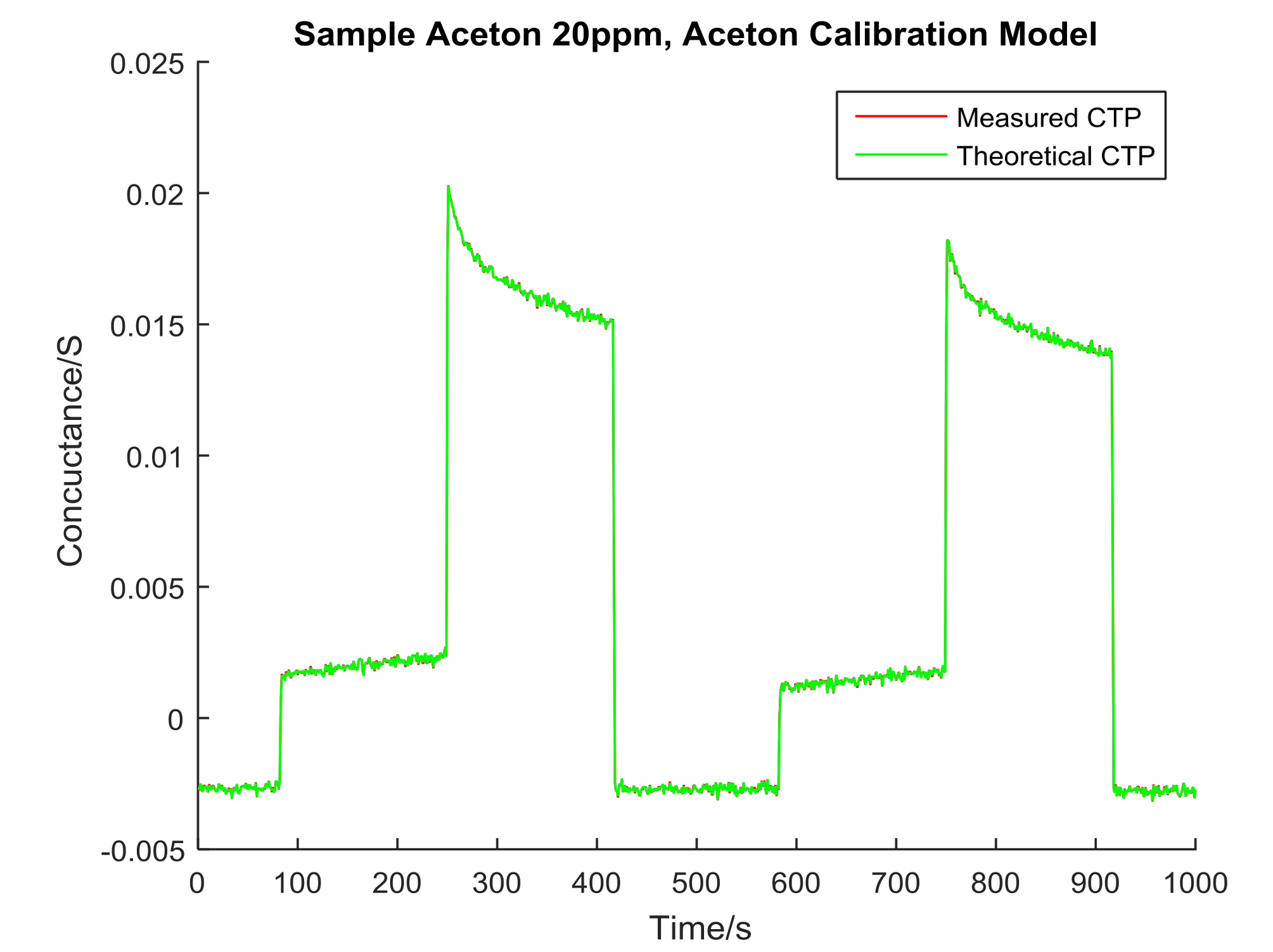
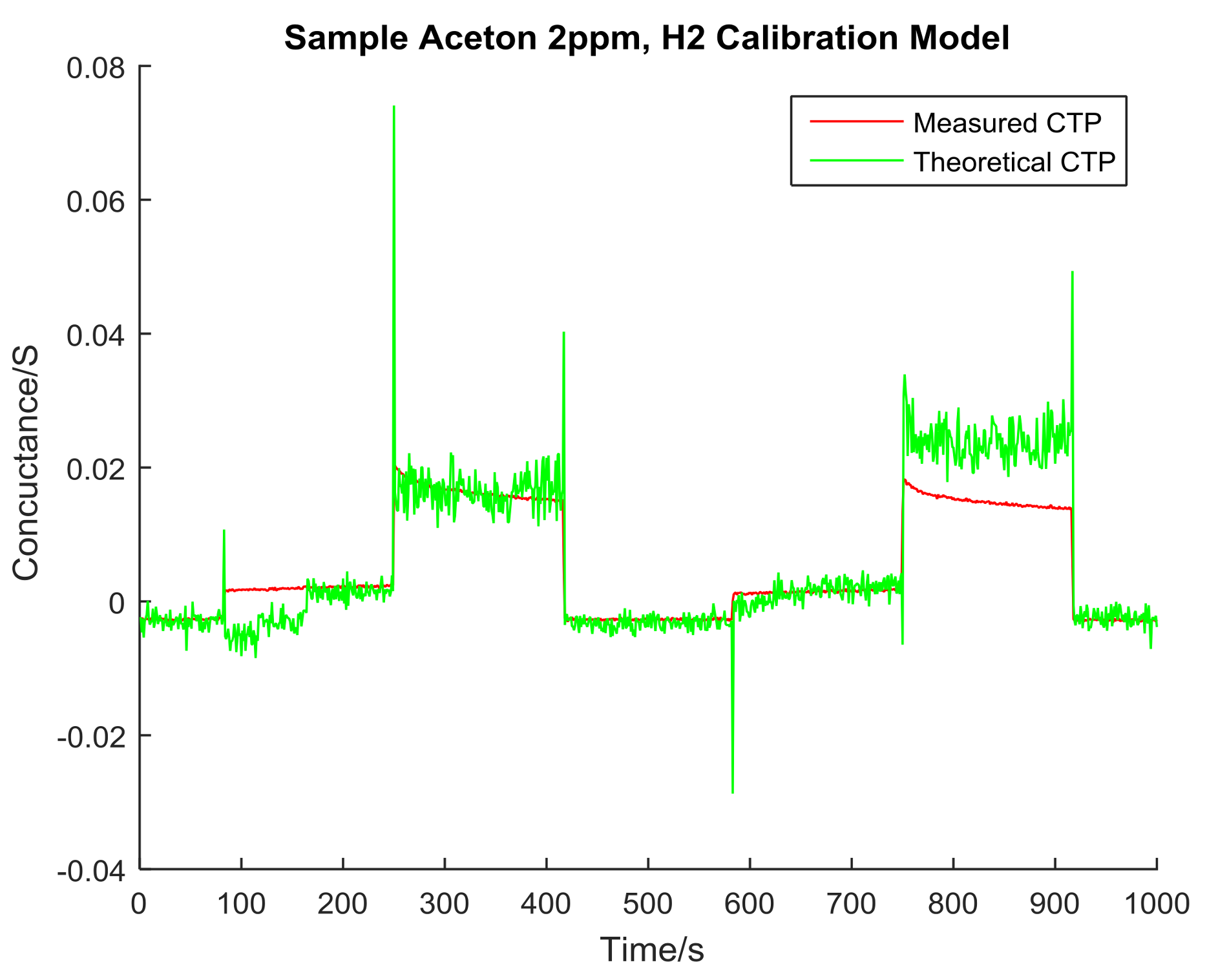
4. Data Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Althainz, P.; Goschnick, J.; Ehrmann, S.; Ache, H.J. Multisensor Microsystem for Contaminants in Air. Sens. Actuators B Chem. 1996, 33, 72–76. [Google Scholar] [CrossRef]
- Sysoev, V.V.; Kiselev, I.; Frietsch, M.; Goschnick, J. Discrimination Power of a Metal-Oxide Thin-Film Sensor Microarray. Sensors 2004, 4, 37–46. [Google Scholar] [CrossRef]
- Frank, K.; Hetznecker, A.; Kohler, H.; Schindler, V.; Schönauer, U.; Arnanthigo, Y.; Falk, D.; Keller, H.B.; Seifert, R. Metal oxide gas sensors for field analysis: Novel SnO2/La2O3 sensor element for analysis of dissolved toluene/ethanol binary mixtures. Sensors 2005, 2, 207–209. [Google Scholar]
- Frank, K.; Magapu, V.; Schindler, V.; Arnanthigo, Y.; Kohler, H.; Keller, H.B.; Seifert, R. Improving the analysis capability of tin oxide gas sensors by dynamic operation, appropriate additives and an advanced evaluation procedure. Sensors 2007, 1, 139–144. [Google Scholar]
- Frank, K.; Magapu, V.; Schindler, V.; Kohler, H.; Keller, H.B.; Seifert, R. Chemical Analysis with Tin Oxide Gas Sensors: Choice of Additives, Method of Operation and Analysis of Numerical Signal. Sens. Lett. 2008, 6, 908–911. [Google Scholar] [CrossRef]
- Seifert, R.; Conrad, T.; Peter, J.; Keller, H.B. Mobile Sensor System AGaMon for Breath Control: Thermo-cyclic Operation and Numerical Signal Analysis of Ternary Gas Mixtures. In Proceedings of the 8th International Conference on Sensor Device Technologies and Applications, Rome, Italy, 10–14 September 2017; pp. 109–114. [Google Scholar]
- Seifert, R.; Keller, H.B.; Frank, K.; Kohler, H. ProSens—An efficient mathematical procedure for calibration and evaluation of tin oxide gas sensor data. Sens. Lett. 2011, 9, 7–10. [Google Scholar] [CrossRef]
- Gardner, J.W. Detection of vapours and odours from a multi sensor array using pat-tern recognition. Part I, Principal component and cluster analysis. Sens. Actuators B Chem. 1991, 4, 109–115. [Google Scholar] [CrossRef]
- Ng, K.T.; Boussaid, F.; Bermak, A. A frequency-based signature gas identification circuit for SnO2 gas sensors. In Proceedings of the 2010 IEEE International Symposium on Circuits and Systems, Paris, France, 30 May–2 June 2010; pp. 2275–2278. [Google Scholar]
- Keller, H.B.; Seifert, R.; Kohler, H. SimSens—A New Mathematical Procedure for Simultaneous Analysis of Gases with Resistive Gas Sensors. Sens. Actuators B. Chem. 2015, 203–207. [Google Scholar] [CrossRef]
- AS-MLV-P2MEMS. MOS Gas Sensor Component for VOC Detection. Available online: http://ams.com/eng/Products/Environmental-Sensors/Gas-Sensors/AS-MLV-P2 (accessed on 11 June 2016).

| Ethanol | H2 | Acetone | Ethanol/H2 | Ethanol/H2 |
|---|---|---|---|---|
| 50 ppm | 10 ppm | 0.5 ppm | 50 ppm/10 ppm | 100 ppm/30 ppm |
| 100 ppm | 20 ppm | 1 ppm | 50 ppm/20 ppm | 175 ppm/10 ppm |
| 175 ppm | 30 ppm | 2 ppm | 50 ppm/30 ppm | 175 ppm/20 ppm |
| - | - | 100 ppm/10 ppm | 175 ppm/30 ppm |
| Ethanol | Ethanol/H2 | Ethanol/H2 | Ethanol/H2 |
|---|---|---|---|
| 135 ppm | 135 ppm/10 ppm | 135 ppm/20 ppm | 1135 ppm/30 ppm |
| Sample/Model | Ethanol | H2 | Acetone | Ethanol/H2 |
|---|---|---|---|---|
| Ethanol 50 ppm | 0.0000 | 0.0092 | 0.1982 | 0.0003 |
| Ethanol 100 ppm | 0.0000 | 0.0353 | 1.1977 | 0.0003 |
| Ethanol 135 ppm | 0.0009 | 0.0511 | 2.1785 | 0.0004 |
| Ethanol 175 ppm | 0.0000 | 0.0807 | 3.3276 | 0.0002 |
| H2 10 ppm | 0.0098 | 0.0000 | 0.2327 | 0.0109 |
| H2 20 ppm | 0.0311 | 0.0000 | 1.0826 | 0.0175 |
| H2 30 ppm | 0.0572 | 0.0000 | 2.4299 | 0.0181 |
| Acetone 0.5 ppm | 0.0043 | 0.0027 | 0.000 | 0.0089 |
| Acetone 1 ppm | 0.0040 | 0.0023 | 0.000 | 0.0088 |
| Acetone 2 ppm | 0.0038 | 0.0024 | 0.000 | 0.0086 |
| Ethanol/H2 50 ppm/10 ppm | 0.0056 | 0.0236 | 1.6922 | 0.0000 |
| Ethanol/H2 50 ppm/20 ppm | 0.0228 | 0.0335 | 3.8462 | 0.0000 |
| Ethanol/H2 50 ppm/30 ppm | 0.0708 | 0.0803 | 7.1130 | 0.0000 |
| Ethanol/H2 100 ppm/10 ppm | 0.0111 | 0.0665 | 4.5184 | 0.0000 |
| Ethanol/H2 100 ppm/20 ppm | 0.0709 | 0.1689 | 8.6767 | 0.0000 |
| Ethanol/H2 100 ppm/30 ppm | 0.3115 | 0.3884 | 14.3375 | 0.0000 |
| Ethanol/H2 135 ppm/10 ppm | 0.0274 | 0.1268 | 6.4551 | 0.0001 |
| Ethanol/H2 135 ppm/20 ppm | 0.1603 | 0.3103 | 11.6890 | 0.0000 |
| Ethanol/H2 135 ppm/30 ppm | 0.5304 | 0.7165 | 18.0694 | 0.0000 |
| Ethanol/H2 175 ppm/10 ppm | 0.0577 | 0.2252 | 8.7091 | 0.0000 |
| Ethanol/H2 175 ppm/20 ppm | 0.3307 | 0.5422 | 15.3622 | 0.0000 |
| Ethanol/H2 175 ppm/30 ppm | 0.8789 | 1.1849 | 22.0615 | 0.0000 |
| Sample | Ethanol | H2 | Acetone |
|---|---|---|---|
| Ethanol 50 ppm | 6.6% | ||
| Ethanol 100 ppm | 6.2% | ||
| Ethanol 135 ppm | 4.6% | ||
| Ethanol 175 ppm | 1.7% | ||
| H2 10 ppm | 1.0% | ||
| H2 20 ppm | 1.5% | ||
| H2 30 ppm | 0.1% | ||
| Acetone 0.5 ppm | 8.0% | ||
| Acetone 1 ppm | 5.0% | ||
| Acetone 2 ppm | 1.0% | ||
| Ethanol/H2 50 ppm/10 ppm | 4.2% | 6.0% | |
| Ethanol/H2 50 ppm/20 ppm | 6.0% | 2.5% | |
| Ethanol/H2 50 ppm/30 ppm | 2.0% | 6.3% | |
| Ethanol/H2 100 ppm/10 ppm | 4.4% | 6.0% | |
| Ethanol/H2 100 ppm/20 ppm | 8.6% | 3.5% | |
| Ethanol/H2 100 ppm/30 ppm | 2.6% | 9.0% | |
| Ethanol/H2 135 ppm/10 ppm | 5.2% | 9.0% | |
| Ethanol/H2 135 ppm/20 ppm | 0.1% | 5.0% | |
| Ethanol/H2 135 ppm/30 ppm | 1.2% | 3.7% | |
| Ethanol/H2 175 ppm/10 ppm | 0.0% | 2.0% | |
| Ethanol/H2 175 ppm/20 ppm | 2.3% | 2.5% | |
| Ethanol/H2 175 ppm/30 ppm | 1.0% | 4.3% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seifert, R.; Conrad, T.; Peter, J.; Keller, H. Simultaneous Analysis of Sensor Data for Breath Control in Respiratory Air. Chemosensors 2018, 6, 15. https://doi.org/10.3390/chemosensors6020015
Seifert R, Conrad T, Peter J, Keller H. Simultaneous Analysis of Sensor Data for Breath Control in Respiratory Air. Chemosensors. 2018; 6(2):15. https://doi.org/10.3390/chemosensors6020015
Chicago/Turabian StyleSeifert, Rolf, Thorsten Conrad, Jens Peter, and Hubert Keller. 2018. "Simultaneous Analysis of Sensor Data for Breath Control in Respiratory Air" Chemosensors 6, no. 2: 15. https://doi.org/10.3390/chemosensors6020015
APA StyleSeifert, R., Conrad, T., Peter, J., & Keller, H. (2018). Simultaneous Analysis of Sensor Data for Breath Control in Respiratory Air. Chemosensors, 6(2), 15. https://doi.org/10.3390/chemosensors6020015





