First Fifty Years of Chemoresistive Gas Sensors
Abstract
:1. Introduction: Historical Overview
2. Chemoresistive Gas Sensors
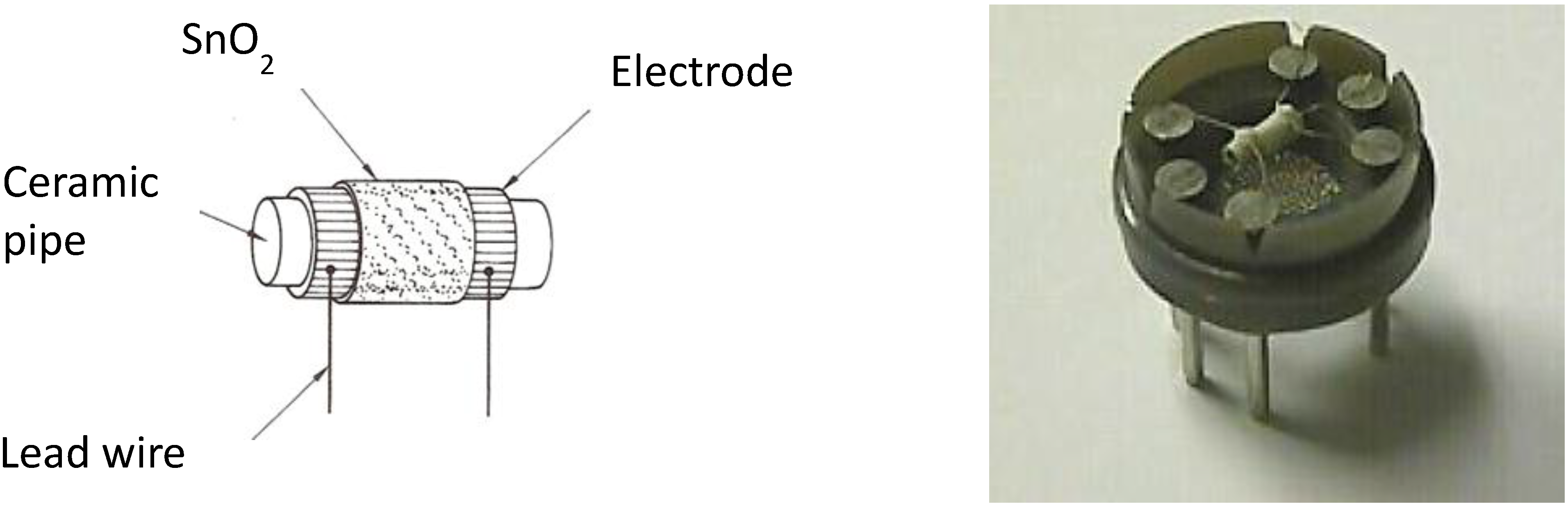
2.1. The Device
| Characteristic | Electrochemical | Optical | Chemoresistive |
|---|---|---|---|
| Cost | low | high | low |
| Lifetime | short | long | long |
| Sensitivity | high | high | high |
| Selectivity | good | excellent | poor |
| Response time | fast | fast | fast |
| Size | medium | large | small |

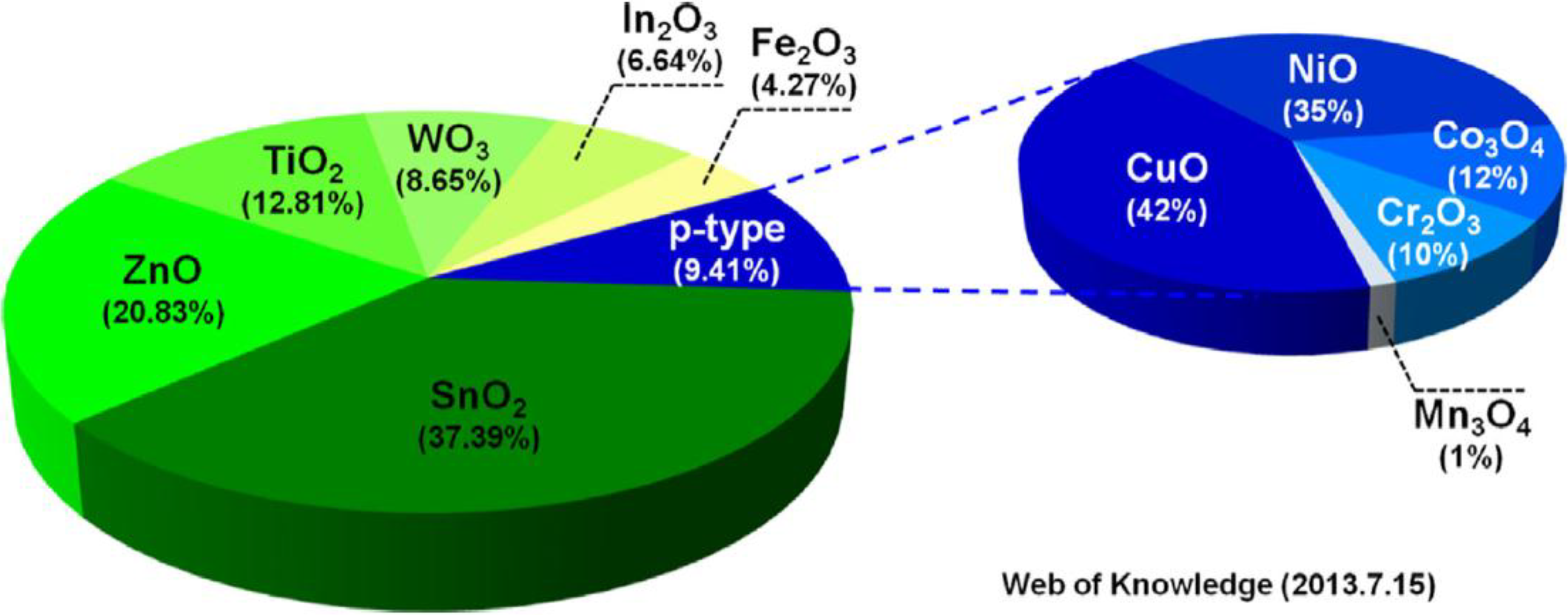
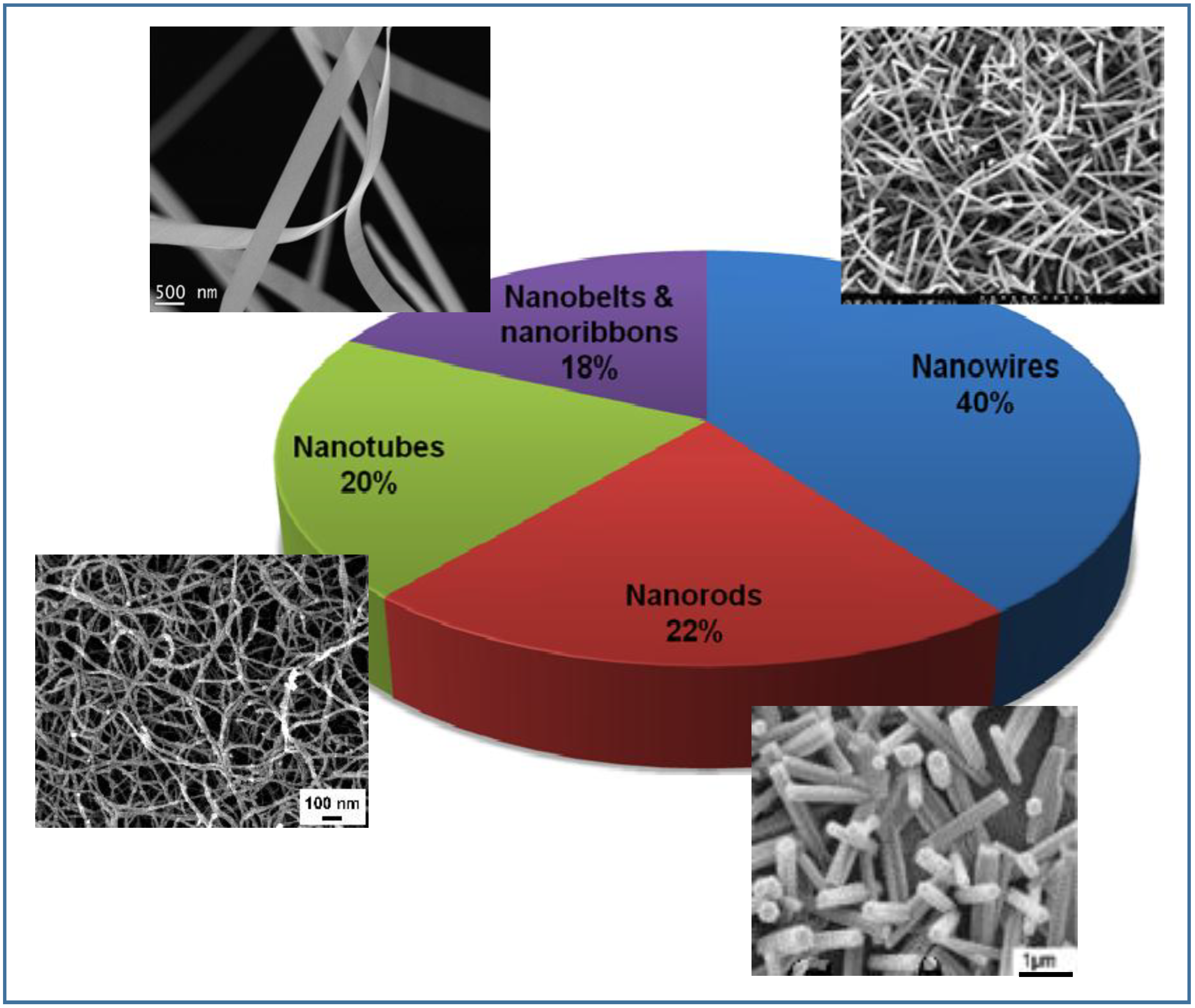
2.2. Sensing Materials
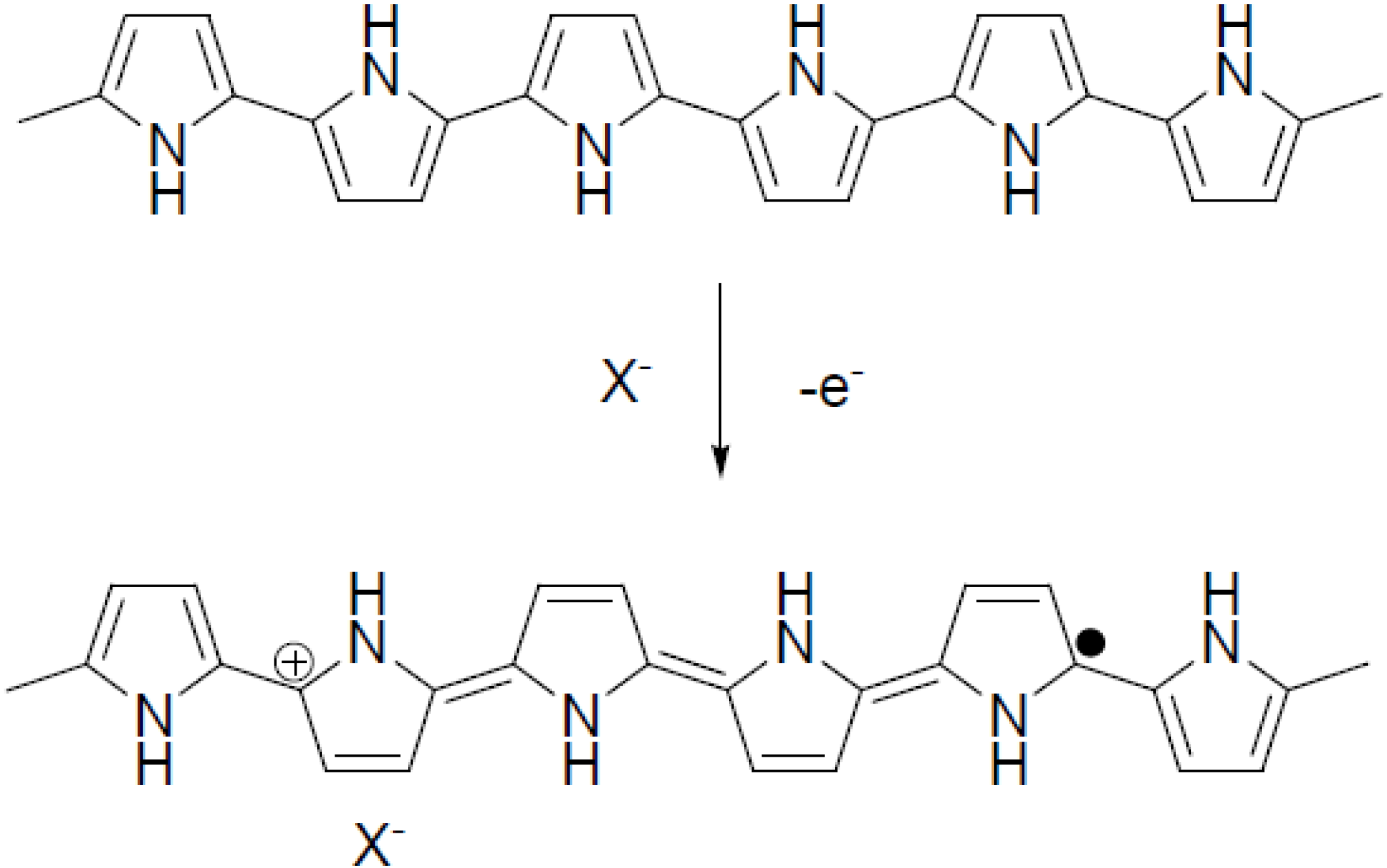
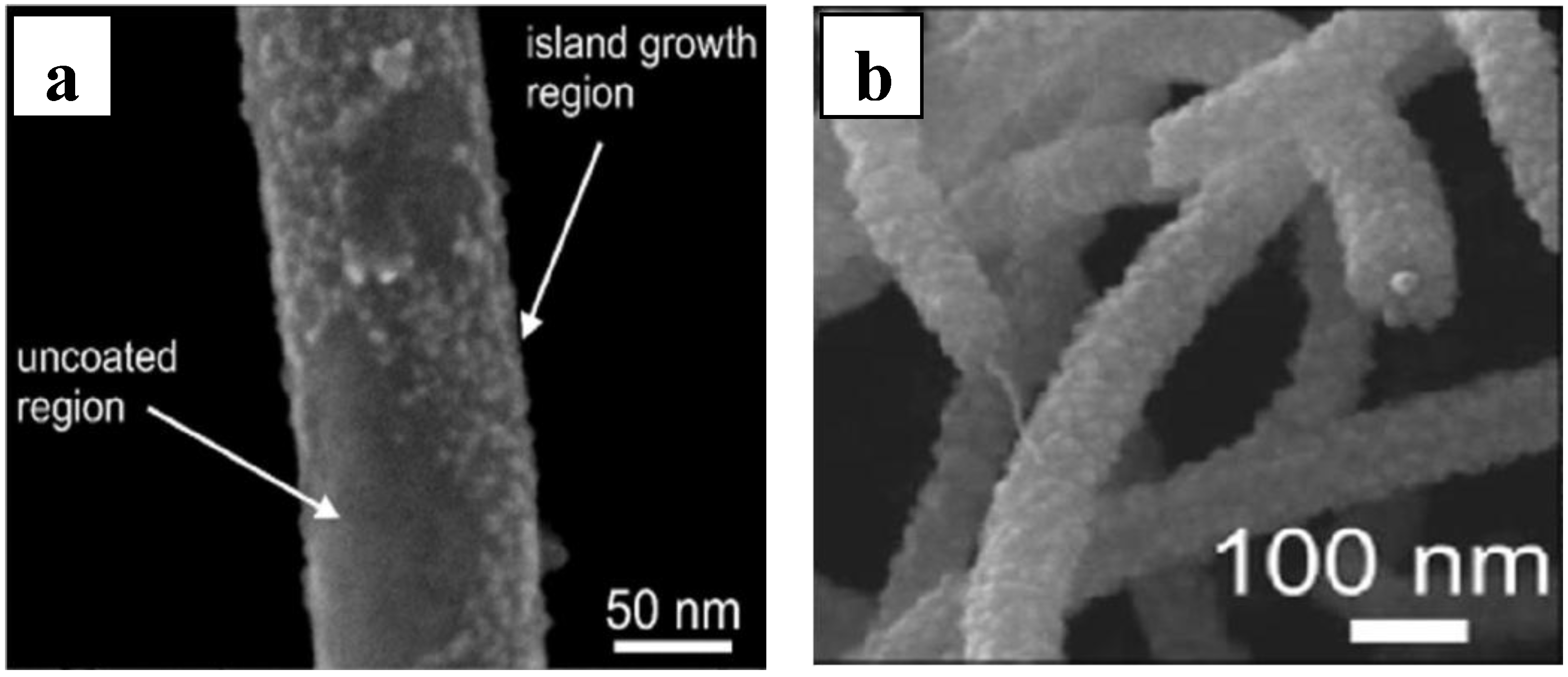

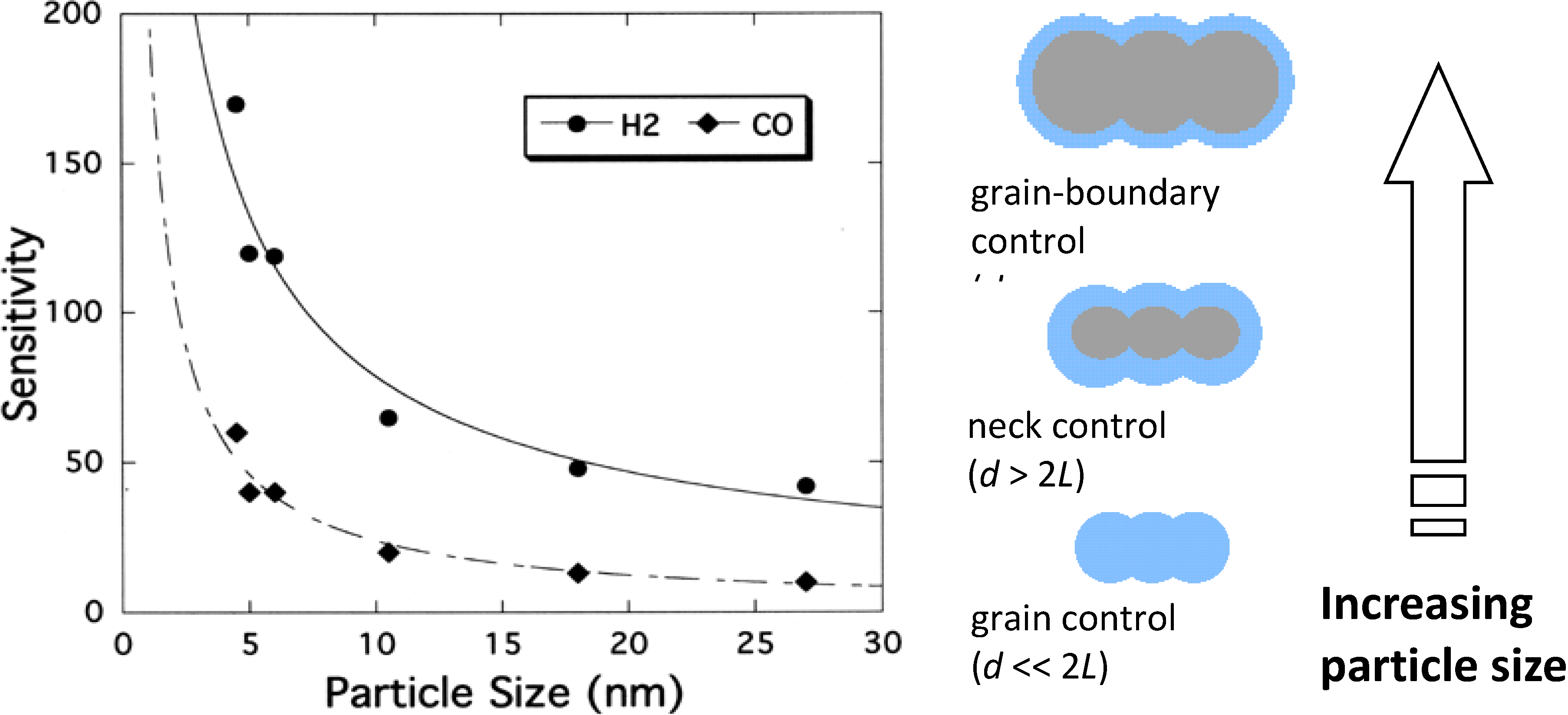
2.3. Working Mechanism
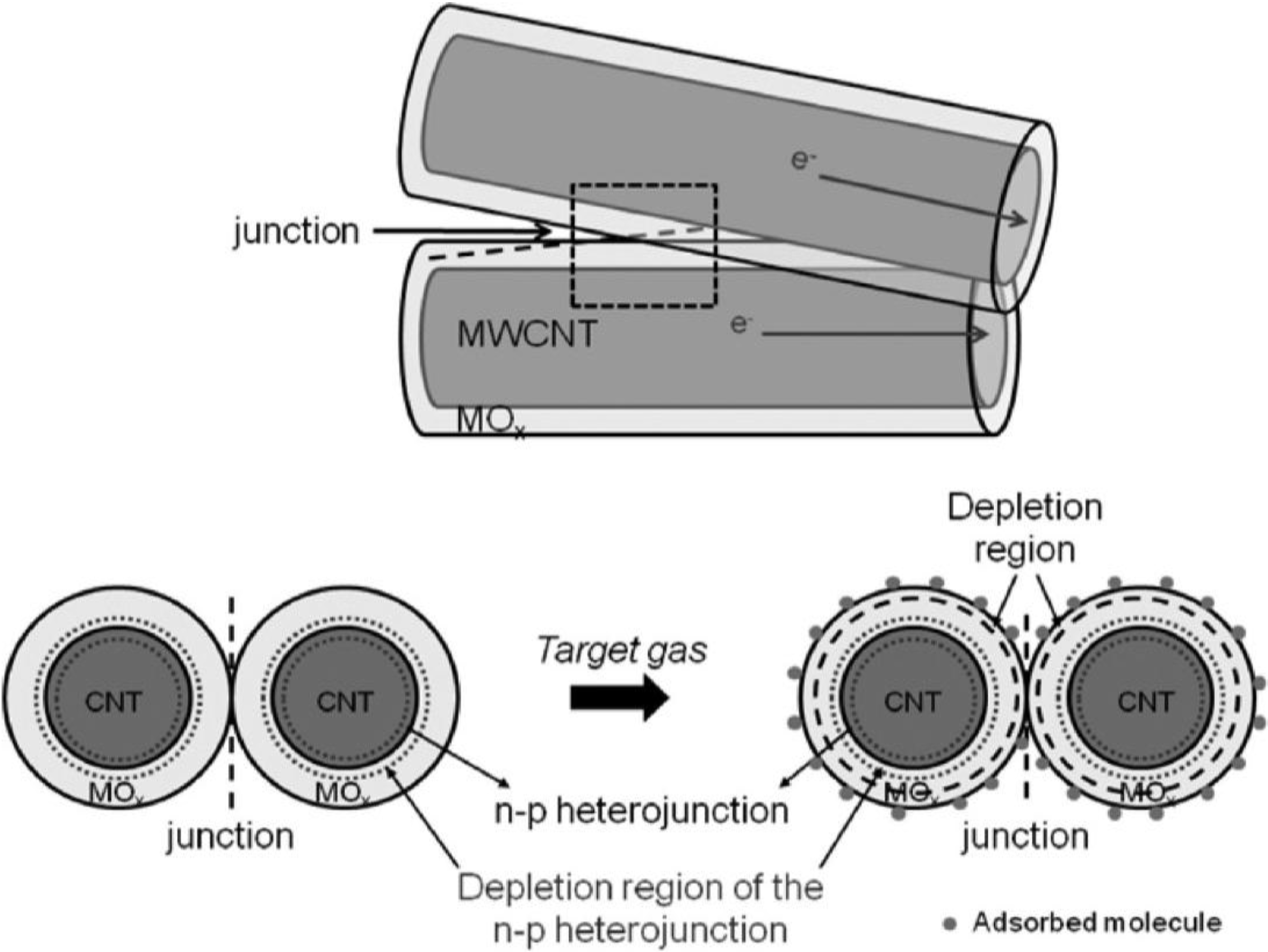
3. Applications of Chemoresistive Gas Sensors
3.1. Environmental Applications
3.2. Automotive Applications
3.3. Biomedical Applications
3.4. Miscellaneous
4. New Opportunities


5. Conclusions
Conflicts of Interest
References
- Seiyama, T.; Kato, A. A new detector for gaseous components using semiconductor thin film. Anal. Chem. 1962, 34, 1502–1503. [Google Scholar] [CrossRef]
- Brattain, W.H.; Bardeen, J. Surface properties of germanium. Bell Syst. Tech. J. 1952, 32, 1–41. [Google Scholar] [CrossRef]
- Heiland, G. Zum einfluss von wasserstoff auf die elektrische leitfähigkeit von ZnO-kristallen. Z. Phys. 1954, 138, 459–464. [Google Scholar] [CrossRef]
- Shaver, P.J. Activated tungsten oxide gas detectors. Appl. Phys. Lett. 1967, 11, 255–257. [Google Scholar] [CrossRef]
- Taguchi, N. Gas Detecting Devices. U.S. Patent 3,631,436, 28 December 1971. [Google Scholar]
- Seiyama, T. Chemical sensors—Current status and future outlook. In Chemical Sensor Technology; Seiyama, T., Ed.; Elsevier: Amsterdam, The Netherlands, 1988; Volume 1. [Google Scholar]
- Yamazoe, N. New approaches for improving semiconductor gas sensors. Sens. Actuators B Chem. 1991, 5, 7–19. [Google Scholar] [CrossRef]
- Shimizu, Y.; Egashira, M. Basic aspects and challenges of semiconductor gas sensors. MRS Bull. 1999, 24, 18–24. [Google Scholar] [CrossRef]
- Yamazoe, N. Toward innovations of gas sensor technology. Sens. Actuators B Chem. 2005, 108, 2–14. [Google Scholar] [CrossRef]
- Williams, D.E. Solid State Gas Sensors; Moseley, P.T., Tofield, B.C., Eds.; Adam Hilger: Bristol, UK, 1987. [Google Scholar]
- Gerblinger, J.; Hardtl, K.H.; Meixner, H.; Aigner, R. High-Temperature Microsensors. In Sensors Set: A Comprehensive Survey; Gopel, W., Ed.; VCH-Verlag: Weinheim, Germany, 1995; p. 181. [Google Scholar]
- Sberveglieri, G. Gas Sensors; Kluwer Academic Publishing: Berlin, Germany, 1992. [Google Scholar]
- Capone, S.; Siciliano, P. Encyclopedia of Nanoscience and Nanotechnology; Nalwa, H.S., Ed.; American Scientific Publishers: Valencia, CA, USA, 2004; Volume 3, p. 769. [Google Scholar]
- Gurlo, A.; Bârsan, N.; Weimar, U. Gas sensors based on semiconductiong metal oxides. In Metal Oxides: Chemistry and Applications; Fierro, J.L.G., Ed.; CRC Press: Boca Raton, FL, USA, 2006; p. 683. [Google Scholar]
- Kim, H.-J.; Lee, J.-H. Highly sensitive and selective gas sensors using p-type oxide semiconductors: Overview. Sens. Actuators B Chem. 2014, 192, 607–627. [Google Scholar] [CrossRef]
- Yamazoe, N.; Shimanoe, K. Receptor function and response of semiconductor gas sensor. J. Sens. 2009, 2009, 875704. [Google Scholar] [CrossRef]
- Aleixandre, M.; Gerboles, M. Review of small commercial sensors for indicative monitoring of ambient gas. Chem. Eng. Trans. 2012, 30, 169–174. [Google Scholar]
- Huang, X.-J.; Choi, Y.-K. Chemical sensors based on nanostructured materials. Sens. Actuators B Chem. 2007, 122, 659–671. [Google Scholar] [CrossRef]
- Jimenez-Cadena, G.; Riu, J.; Xavier Rius, F. Gas sensors based on nanostructured materials. Analyst 2007, 132, 1083–1099. [Google Scholar] [CrossRef]
- Ogawa, H.; Nishikawa, M.; Abe, A. Hall measurement studies and an electrical conduction model of tin oxide ultrafine particle films. J. Appl. Phys. 1982, 53, 4448–4455. [Google Scholar] [CrossRef]
- Korotcenkov, G. Gas response control through structural and chemical modification of metal oxide films: State of the art and approaches. Sens. Actuators B Chem. 2005, 107, 209–232. [Google Scholar] [CrossRef]
- Mahmood, M.R.; Soga, T.; Mamat, M.H.; Khusaimi, Z.; Nor, A.M. A Review on zinc oxide nanostructures: Doping and gas sensing. Adv. Mater. Res. 2013, 667, 329–332. [Google Scholar] [CrossRef]
- Tricoli, A.; Righettoni, M.; Pratsinis, S.E. Minimal cross-sensitivity to humidity during ethanol detection by SnO2-TiO2 solid solutions. Nanotechnology 2009, 20, 315502. [Google Scholar] [CrossRef] [PubMed]
- Ramamoorthy, R.; Dutta, P.K.; Akbar, S.A. Oxygen sensors: Materials, methods, designs and applications. J. Mater. Sci. 2003, 38, 4271–4282. [Google Scholar] [CrossRef]
- Malagù, C.; Guidi, V.; Carotta, M.C.; Martinelli, G. Unpinning of Fermi level in nanocrystalline semiconductors. Appl. Phys. Lett. 2004, 84, 4158–4160. [Google Scholar]
- Neri, G. Encyclopedia of Sensors; Grimes, C.A., Dickey, E.C., Pishko, M.V., Eds.; American Scientific Publishers: Valencia, CA, USA, 2006; Volume 6, p. 1. [Google Scholar]
- Cabot, A.; Arbiol, J.; Morante, J.R.; Weimar, U.; Barsan, N.; Gopel, W. Analysis of the noble metal catalytic additives introduced by impregnation of as obtained SnO2 sol-gel nanocrystals for gas sensors. Sens. Actuators B Chem. 2000, 70, 87–100. [Google Scholar] [CrossRef]
- Baraton, M.-I.; Merhari, L. Influence of the particle size on the surface reactivity and gas sensing properties of SnO2 nanopowders. Mater. Trans. 2001, 42, 1616–1622. [Google Scholar] [CrossRef]
- Xu, C.; Tamaki, J.; Miura, N.; Yamazoe, N. Grain size effects on gas sensitivity of porous SnO2-based elements. Sens. Actuators B Chem. 1991, 3, 147–155. [Google Scholar] [CrossRef]
- Dolbec, R.; el Khakani, M.A.; Serventi, A.M.; Saint-Jacques, R.G. Influence of the nanostructural characteristics on the gas sensing properties of pulsed laser deposited tin oxide thin films. Sens. Actuators B Chem. 2003, 93, 566–571. [Google Scholar] [CrossRef]
- Gurlo, A.; Ivanovskaya, M.; Barsan, N.; Schweizer-Berberich, M.; Weimar, U.; Gopel, W.; Dieguez, A. Grain size control in nanocrystalline In2O3 semiconductor gas sensors. Sens. Actuators B Chem. 1997, 44, 327–333. [Google Scholar] [CrossRef]
- Ansari, S.G.; Boroojerdian, P.; Sainkar, S.R.; Karekar, R.N.; Aiyer, R.C.; Kulkarni, S.K. Grain size effects on H2 gas sensitivity of thick film resistor using SnO2 nanoparticles. Thin Solid Films 1997, 295, 271–276. [Google Scholar] [CrossRef]
- Dieguez, A.; Romano-Rodriguez, A.; Morante, J.R.; Weimar, U.; Schweizer-Berberich, M.; Gopel, W. Morphological analysis of nanocrystalline SnO2 for gas sensor applications. Sens. Actuators B Chem. 1996, 31, 1–8. [Google Scholar] [CrossRef]
- Korotcenkov, G. The role of morphology and crystallographic structure of metal oxides in response of conductometric-type gas sensors. Mater. Sci. Eng. R 2008, 61, 1–39. [Google Scholar] [CrossRef]
- Rothschild, A.; Komem, Y. Metal oxide gas sensors with nanosized grains. J. Electroceram. 2004, 13, 697–701. [Google Scholar] [CrossRef]
- Nylander, C.; Armgarth, M.; Lundstrom, L. An ammonia detector based on a conducting polymer. Anal. Chem. Symp. Ser. 1983, 17, 203–207. [Google Scholar]
- Jiang, T.; Wang, Z.; Li, Z.; Wang, W.; Xu, X.; Liu, X.; Wang, J.; Wang, C. Synergic effect within n-type inorganic–p-type organic nano-hybrids in gas sensors. J. Mater. Chem. C 2013, 1, 3017–3025. [Google Scholar] [CrossRef]
- Kong, J.; Franklin, N.R.; Zhou, C.; Chapline, M.G.; Peng, S.; Cho, K.; Dai, H. Nanotube molecular wires as chemical sensors. Science 2000, 287, 622–625. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.; Cho, K. Ab initio study of doped carbon nanotube sensors. Nano Lett. 2003, 3, 513–517. [Google Scholar] [CrossRef]
- Sin, M.L.Y.; Chow, G.C.T.; Wong, G.M.K.; Li, W.J.; Leong, P.H.W.; Wong, K.W. Ultralow-power alcohol vapor sensors using chemically functionalized multiwalled carbon nanotubes. IEEE Trans. Nanotechnol. 2007, 6, 571–577. [Google Scholar] [CrossRef]
- Sun, Y.P.; Fu, K.F.; Lin, Y.; Huang, W.J. Functionalized carbon nanotubes: Properties and applications. Acc. Chem. Res. 2002, 35, 1096–1104. [Google Scholar] [CrossRef] [PubMed]
- Lu, G.; Ocola, L.E.; Chen, J. Room-temperature gas sensing based on electron transfer between discrete tin oxide nanocrystals and multiwalled carbon nanotubes. Adv. Mater. 2009, 21, 1–5. [Google Scholar]
- Sun, Y.-F.; Liu, S.-B.; Meng, F.-L.; Liu, J.-Y.; Jin, Z.; Kong, L.-T.; Liu, J.-H. Metal oxide nanostructures and their gas sensing properties: A review. Sensors 2012, 12, 2610–2631. [Google Scholar] [CrossRef] [PubMed]
- Willinger, M.G.; Neri, G.; Rauwel, E.; Bonavita, A.; Micali, G.; Pinna, N. Vanadium oxide sensing layer grown on carbon nanotubes by a new atomic layer deposition process. Nano Lett. 2008, 8, 4201–4204. [Google Scholar] [CrossRef] [PubMed]
- Marichy, C.; Pinna, N. Carbon-nanostructures coated/decorated by atomic layer deposition: Growth and applications. Coord. Chem. Rev. 2013, 257, 3232–3253. [Google Scholar] [CrossRef]
- Lu, J.G.; Chang, P.; Fan, Z. Quasi-one-dimensional metal oxide materials—Synthesis, properties and applications. Mater. Sci. Eng. R 2006, 52, 49–91. [Google Scholar] [CrossRef]
- Pan, Z.W.; Dai, Z.R.; Wang, Z.L. Nanobelts of semiconducting oxides. Science 2001, 291, 1947–1949. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Zhang, D.; Liu, X.; Han, S.; Tang, T.; Han, J.; Zhou, C. In2O3 nanowires as chemical sensors. Appl. Phys. Lett. 2003, 82, 1613–1615. [Google Scholar] [CrossRef]
- Cao, G. Nanostructures and Nanomaterials; IC Press: London, UK, 2004. [Google Scholar]
- Comini, E. Metal oxide nano-crystals for gas sensing. Anal. Chim. Acta 2006, 568, 28–40. [Google Scholar] [CrossRef] [PubMed]
- Comini, E.; Faglia, G.; Sberveglieri, G. Stable and highly sensitive gas sensors based on semiconducting oxide nanobelts. Appl. Phys. Lett. 2002, 81, 1869–1871. [Google Scholar] [CrossRef]
- Law, M.; Kind, H.; Messer, B.; Kim, F.; Yang, P.D. Photochemical sensing of NO2 with SnO2 nanoribbon nanosensors at room temperature. Angew. Chem. Int. Ed. 2002, 41, 2405–2408. [Google Scholar] [CrossRef]
- Choi, K.J.; Jang, H.W. One-dimensional oxide nanostructures as gas-sensing materials: Review and issues. Sensors 2010, 10, 4083–4099. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H. Gas sensors using hierarchical and hollow oxide nanostructures: Overview. Sens. Actuators B Chem. 2009, 140, 319–336. [Google Scholar] [CrossRef]
- Schedin, F.; Geim, A.K.; Morozov, S.V.; Hill, E.W.; Blake, P.; Katsnelson, M.I.; Novoselov, K.S. Detection of individual gas molecules adsorbed on graphene. Nat. Mater. 2007, 6, 652–655. [Google Scholar] [CrossRef] [PubMed]
- Wolkenstein, F.F. Effect of ionizing radiation on the adsorptive and catalytic properties of semiconductors. Discuss. Faraday Soc. 1961, 31, 209–218. [Google Scholar] [CrossRef]
- Morrison, R.S. Semiconductor gas sensors. Sens. Actuators 1982, 2, 329–341. [Google Scholar] [CrossRef]
- Yamazoe, N.; Kurokawa, Y.; Seiyama, T. Effects of additives on semiconductor gas sensors. Sens. Actuators B Chem. 1983, 4, 283–289. [Google Scholar] [CrossRef]
- Gopel, W.; Schierbaum, K. SnO2 sensors: Current status and future prospects. Sens. Actuators B Chem. 1995, 26/27, 1–12. [Google Scholar] [CrossRef]
- Azad, A.M.; Akbar, S.A.; Mhaisalkar, S.G.; Birkefeld, L.D.; Goto, K.S. Solid-State Gas Sensors: A Review. J. Electrochem. Soc. 1992, 139, 3690–3704. [Google Scholar] [CrossRef]
- Barsan, N.; Schweizer-Berberich, M.; Göpel, W. Fundamental and practical aspects in the design of nanoscaled SnO2 gas sensors: A status report. Fresenius. J. Anal. Chem. 1999, 365, 287–304. [Google Scholar] [CrossRef]
- Leblanc, E.; Perier-Camby, L.; Thomas, G.; Gibert, R.; Primet, M.; Gelin, P. NOx adsorption onto dehydroxylated or hydroxylated tin dioxide surface. Application to SnO2-based sensors. Sens. Actuators B Chem. 2000, 62, 67–72. [Google Scholar] [CrossRef]
- Li, Y.; Wlodarski, W.; Galatsis, K.; Moslih, S.H.; Cole, J.; Russo, S.; Rockelmann, N. Gas sensing properties of p-type semiconducting Cr-doped TiO2 thin films. Sens. Actuators B Chem. 2002, 83, 160–163. [Google Scholar] [CrossRef]
- Yamazoe, N.; Shimanoe, K. Theory of power laws for semiconductor gas sensors. Sens. Actuators B Chem. 2008, 128, 566–573. [Google Scholar] [CrossRef]
- Rothschild, A.; Komem, Y. The effect of grain size on the sensitivity of nanocrystalline metal-oxide gas sensors. J. Appl. Phys. 2004, 95, 6374–6380. [Google Scholar] [CrossRef]
- Chen, X.; Wong, C.K.Y.; Yuan, C.A.; Zhang, G. Nanowire-Based Gas Sensors. Sens. Actuators B Chem. 2013, 177, 178–195. [Google Scholar] [CrossRef]
- Wei, B.-Y.; Hsu, M.-C.; Su, P.-G.; Lin, H.-M.; Wu, R.-J.; Lai, H.-J. A novel SnO2 gas sensor doped with carbon nanotubes operating at room temperature. Sens. Actuators B Chem. 2004, 101, 81–89. [Google Scholar] [CrossRef]
- Marichy, C.; Russo, P.A.; Latino, M.; Tessonnier, J.-P.; Willinger, M.-G.; Donato, N.; Neri, G.; Pinna, N. Tin dioxide-carbon heterostructures applied to gas sensing: Structure-dependent properties and general sensing mechanism. J. Phys. Chem. C 2013, 117, 19729–19739. [Google Scholar]
- Wilson, A.D.; Baietto, M. Applications and advances in electronic-nose technologies. Sensors 2009, 9, 5099–5148. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.-D.; Lee, A.-S. Environmental gas sensors. IEEE Sens. J. 2001, 1, 214–224. [Google Scholar] [CrossRef]
- Fine, G.F.; Cavanagh, L.M.; Afonja, A.; Binions, R. Metal oxide semi-conductor gas sensors in environmental monitoring. Sensors 2010, 10, 5469–5502. [Google Scholar] [CrossRef] [PubMed]
- Marsal, A.; Dezanneau, G.; Cornet, A.; Morante, J.R. A new CO2 gas sensing material. Sens. Actuators B Chem. 2003, 95, 266–270. [Google Scholar] [CrossRef]
- Pijolat, C.; Pupier, C.; Sauvan, M.; Tournier, G.; Lalauze, R. Gas detection for automotive pollution control. Sens. Actuators B Chem. 1999, 59, 195–202. [Google Scholar] [CrossRef]
- Cederquist, A.; Gibbons, E.; Meitzler, A. Characterization of Zirconia and Titania Engine Exhaust Gas Sensors for Air/Fuel Feedback Control Systems. SAE Tech. Paper 1976. [Google Scholar] [CrossRef]
- Holt, C.T.; Azad, A.-M.; Swartz, S.L.; Rao, R.R.; Dutta, P.K. Carbon monoxide sensor for PEM fuel cell systems. Sens. Actuators B Chem. 2002, 87, 414–420. [Google Scholar] [CrossRef]
- D’Amico, A.; di Natale, C.; Paolesse, R.; Macagnano, A.; Martinelli, E.; Pennazza, G.; Santonico, M.; Bernabei, M.; Roscioni, C.; Galluccio, G.; et al. Olfactory systems for medical applications. Sens. Actuators B Chem. 2008, 130, 458–465. [Google Scholar] [CrossRef]
- Disease Markers in Exhaled Breath; Marczin, N.; Kharitonov, S.A.; Yacoub, M.H.; Barnes, P.J. (Eds.) Marcel Dekker: New York, NY, USA, 2002.
- Righettoni, M.; Amann, A.; Pratsinis, S.E. Breath analysis by nanostructured metal oxides as chemo-resistive gas sensors. Mater. Today 2014. [Google Scholar] [CrossRef]
- Casalinuovo, I.; Pierro, D. Application of electronic noses for disease diagnosis and food spoilage detection. Sensors 2006, 6, 1428–1439. [Google Scholar] [CrossRef]
- Gonzalez-Jimenez, J.; Monroy, J.G.; Blanco, J.L. The multi-chamber electronic nose—An improved olfaction sensor for mobile robotics. Sensors 2011, 11, 6145–6164. [Google Scholar] [CrossRef] [PubMed]
- Neri, G. Better sensors through chemistry: Some selected examples. Lect. Notes Electr. Eng. 2011, 91, 19–30. [Google Scholar]
- Carbone, L.; Cozzoli, P.D. Colloidal heterostructured nanocrystals: Synthesis and growth mechanisms. Nano Today 2010, 5, 449–493. [Google Scholar] [CrossRef]
- Ataman, C.; Kinkeldei, T.; Mattana, G.; Vásquez Quintero, A.; Molina-Lopez, F.; Courbat, J.; Cherenack, K.; Briand, D.; Tröster, G.; de Rooij, N.F. A robust platform for textile integrated gas sensors. Sens. Actuators B Chem. 2013, 177, 1053–1061. [Google Scholar] [CrossRef]
- Gardner, J.W.; Guha, P.K.; Udrea, F.; Covington, J.A. CMOS interfacing for integrated gas sensors: A review. IEEE Sens. J. 2010, 10, 1833–1848. [Google Scholar] [CrossRef]
- MEMS Technical Data. Available online: http://www.memsgassensor.com (accessed on 28 February 2014).
- Oletic, D.; Bilas, V. Empowering smartphone users with sensor node for air quality measurement. J. Phys. Conf. Ser. 2013, 450, 012028. [Google Scholar] [CrossRef]
- Sensorcon. Available online: http://sensorcon.com (accessed on 10 December 2014).
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Neri, G. First Fifty Years of Chemoresistive Gas Sensors. Chemosensors 2015, 3, 1-20. https://doi.org/10.3390/chemosensors3010001
Neri G. First Fifty Years of Chemoresistive Gas Sensors. Chemosensors. 2015; 3(1):1-20. https://doi.org/10.3390/chemosensors3010001
Chicago/Turabian StyleNeri, Giovanni. 2015. "First Fifty Years of Chemoresistive Gas Sensors" Chemosensors 3, no. 1: 1-20. https://doi.org/10.3390/chemosensors3010001
APA StyleNeri, G. (2015). First Fifty Years of Chemoresistive Gas Sensors. Chemosensors, 3(1), 1-20. https://doi.org/10.3390/chemosensors3010001






