A Cellulose Nanofiber Capacitive Humidity Sensor with High Sensitivity and Fast Recovery Characteristics
Abstract
1. Introduction
2. Experimental Section
2.1. Materials
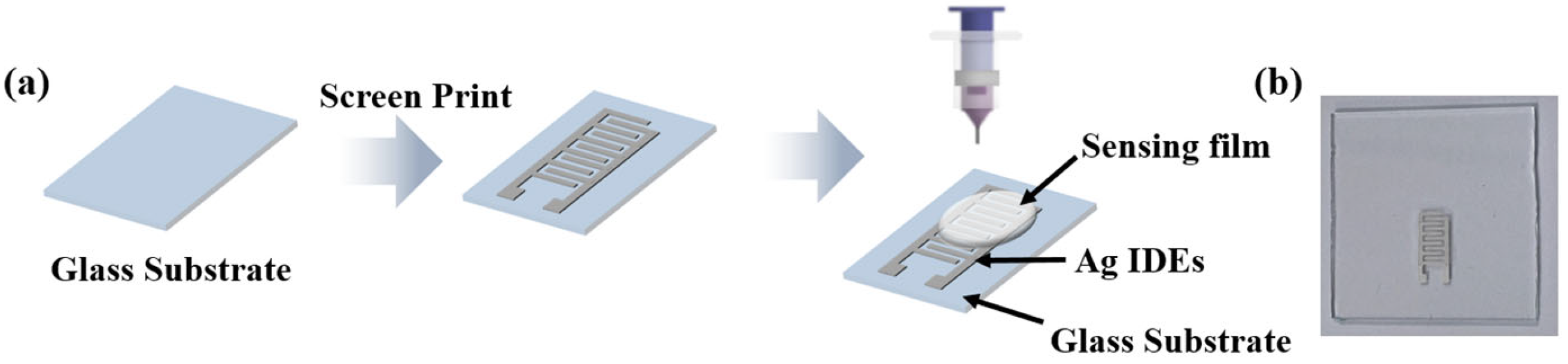
2.2. Preparation of Humidity Sensors
2.3. Preparation of QCM Sensors
2.4. Measurements
3. Results and Discussion
3.1. Materials Characterization
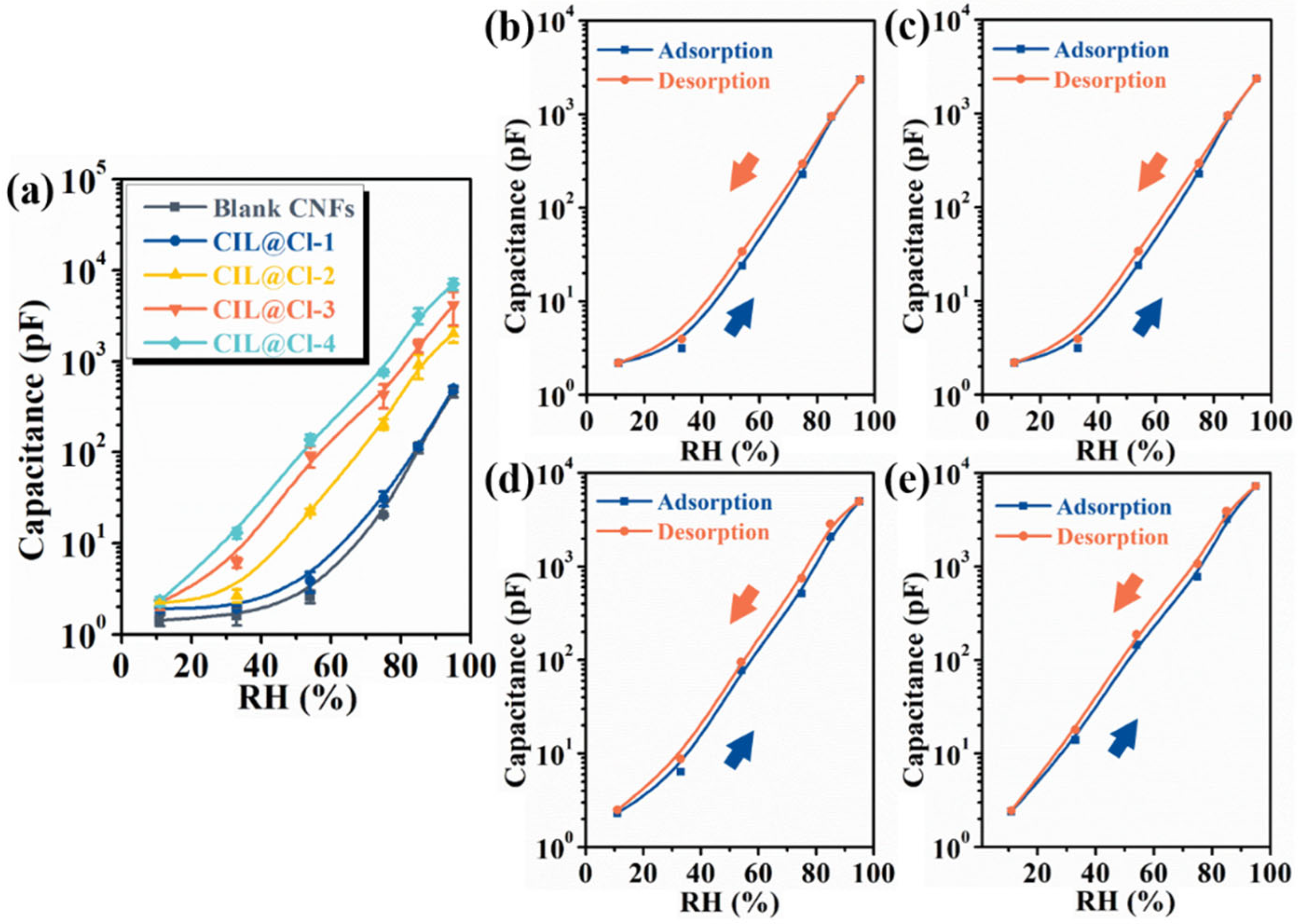
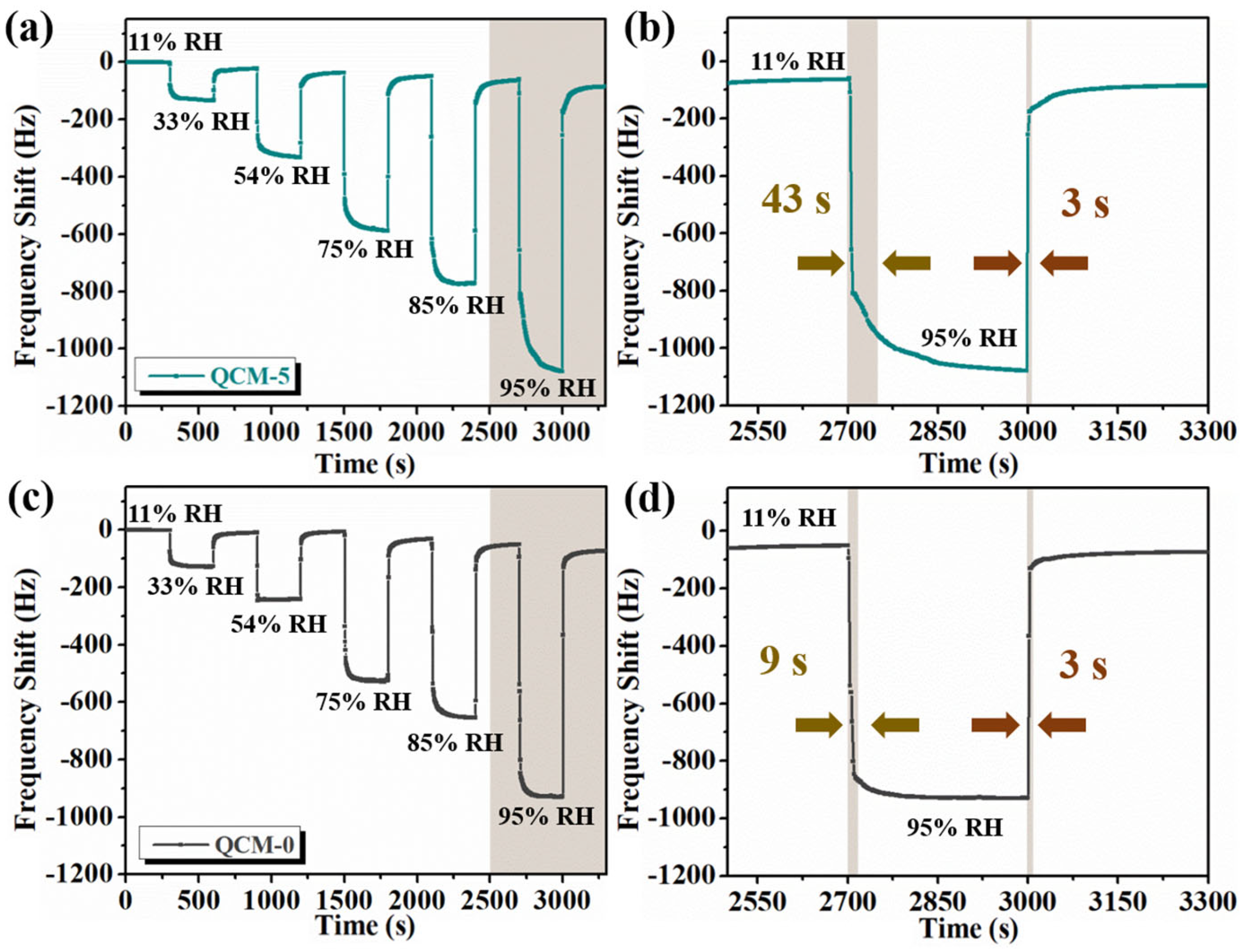
3.2. Humidity-Sensing Properties
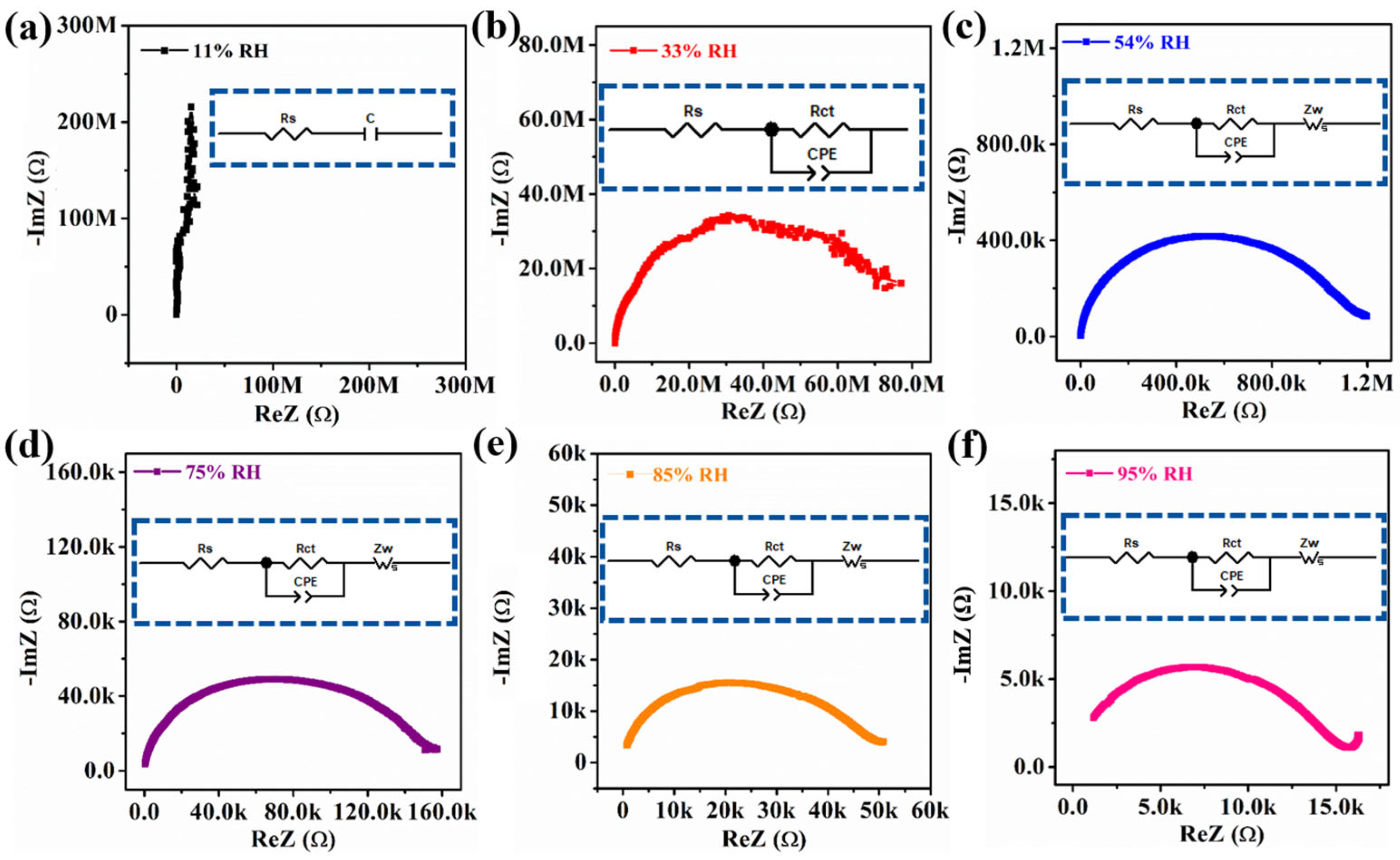
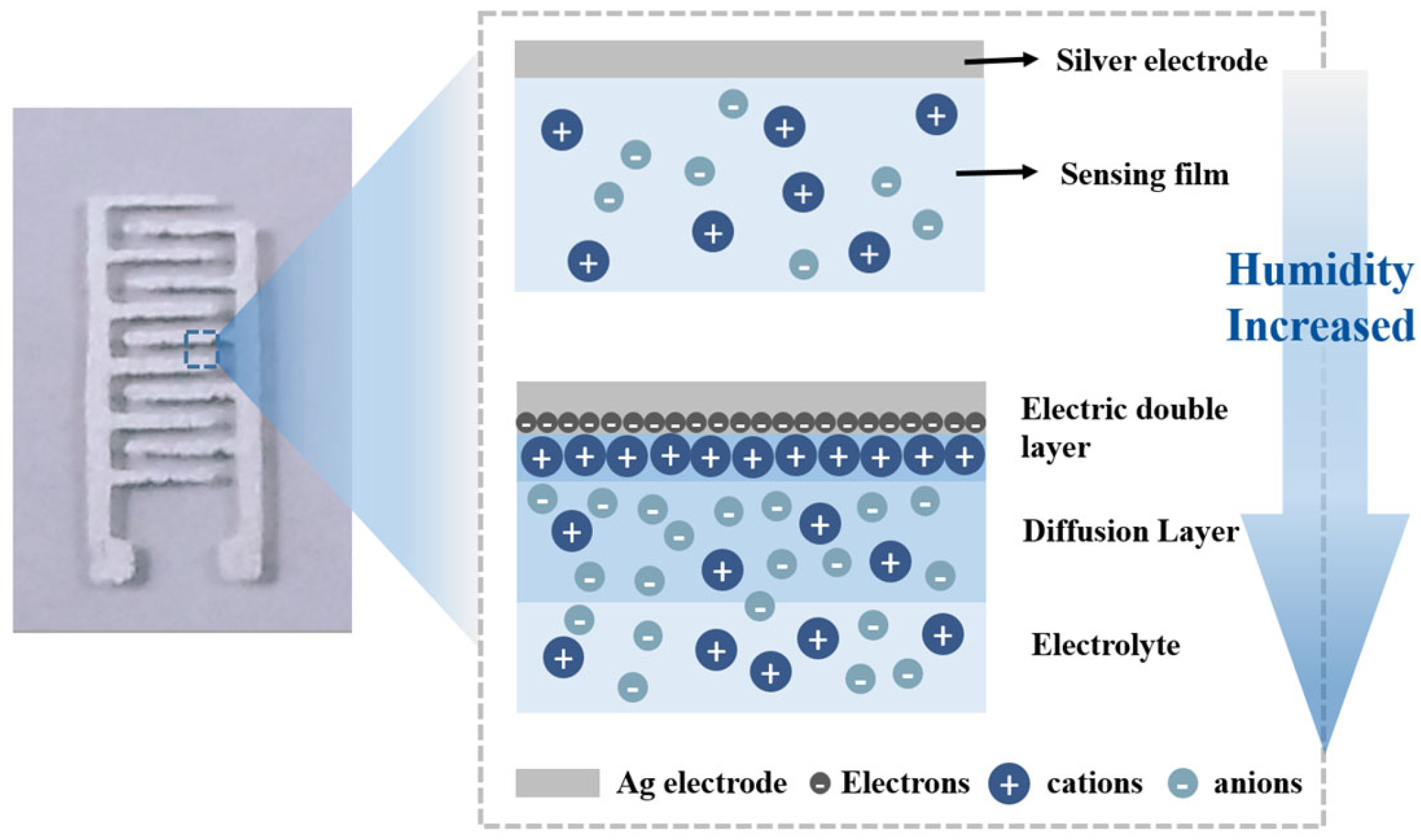
3.3. Humidity-Sensing Mechanism
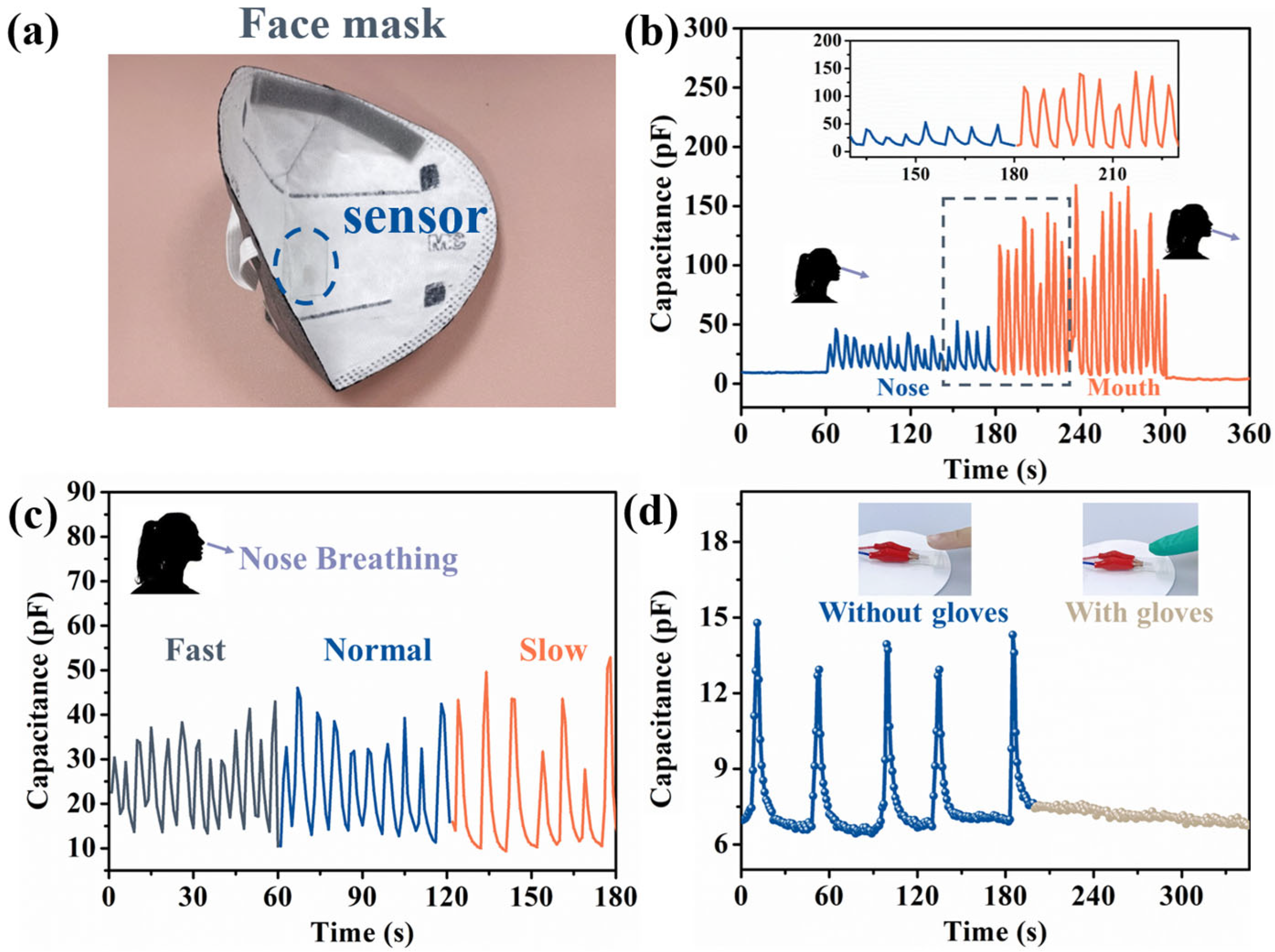
3.4. Application of the Sensor
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dai, J.; Zhao, H.; Lin, X.Z.; Liu, S.; Fei, T.; Zhang, T. Design strategy for ultrafast response humidity sensors based on gel polymer electrolytes and application for detecting respiration. Sens. Actuators B 2020, 304, 127270. [Google Scholar] [CrossRef]
- Wang, G.; Zhang, Y.; Yang, H.; Wang, W.; Dai, Y.; Niu, L.; Lv, C.; Xia, H.; Liu, T. Fast response humidity sensor based on laser printing for respiration monitoring. RSC Adv. 2020, 10, 8910–8916. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Shi, R.; Lou, Z.; Chai, R.; Jiang, K.; Shen, G. Flexible smart noncontact control systems with ultrasensitive humidity sensors. Small 2019, 15, 1902801. [Google Scholar] [CrossRef] [PubMed]
- Cho, M.-Y.; Kim, I.-S.; Kim, S.-H.; Park, C.; Kim, N.-Y.; Kim, S.-W.; Kim, S.; Oh, J.-M. Unique noncontact monitoring of human respiration and sweat evaporation using a CsPb2Br5 based sensor. ACS Appl. Mater. Interfaces 2021, 13, 5602–5613. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Fu, X.; Zhu, H.; Qu, Z.; Qi, Y.; Zhang, Z.; Zhang, Z.; Cheng, G.; Zhang, C.; Ding, J. Self-powered non-contact motion vector sensor for multifunctional human-machine interface. Small Methods 2022, 6, 2200588. [Google Scholar] [CrossRef]
- Guan, X.; Hou, Z.; Wu, K.; Zhao, H.; Liu, S.; Fei, T.; Zhang, T. Flexible humidity sensor based on modified cellulose paper. Sens. Actuators B 2021, 339, 129879. [Google Scholar] [CrossRef]
- Wang, Y.; Sekine, T.; Takeda, Y.; Yokosawa, K.; Matsui, H.; Kumaki, D.; Shiba, T.; Nishikawa, T.; Tokito, S. Fully printed PEDOT: PSS-based temperature sensor with high humidity stability for wireless healthcare monitoring. Sci. Rep. 2020, 10, 2467. [Google Scholar] [CrossRef]
- Luo, J.; Yao, Y.; Duan, X.; Liu, T. Force and humidity dual sensors fabricated by laser writing on polyimide/paper bilayer structure for pulse and respiration monitoring. J. Mater. Chem. C 2018, 6, 4727–4736. [Google Scholar] [CrossRef]
- Li, M.; Yang, L.; Fang, S.; Dong, S.; Hirano, S.; Tachibana, K. Polymer electrolytes containing guanidinium-based polymeric ionic liquids for rechargeable lithium batteries. J. Power Sources 2011, 196, 8662–8668. [Google Scholar] [CrossRef]
- Yuan, Q.; Geng, W.; Li, N.; Tu, J.; Wang, R.; Zhang, T.; Li, X. Study on humidity sensitive property of K2CO3-SBA-15 composites. Appl. Surf. Sci. 2009, 256, 280–283. [Google Scholar] [CrossRef]
- Mahadeva, S.K.; Walus, K.; Stoeber, B. Paper as a platform for sensing applications and other sensors: A review. ACS Appl. Mater. Interfaces 2015, 7, 8345–8362. [Google Scholar] [CrossRef] [PubMed]
- Maxwell, E.J.; Mazzeo, A.D.; Whitesides, G.M. Paper-based electroanalytical sensors for accessible diagnostic testing. MRS Bull. 2013, 38, 309–314. [Google Scholar] [CrossRef]
- Yang, Q.; Saito, T.; Isogai, A. Facile fabrication of transparent cellulose films with high water repellency and gas barrier properties. Cellulose 2012, 19, 1913–1921. [Google Scholar] [CrossRef]
- Koga, H.; Nagashima, K.; Huang, Y.; Zhang, G.; Wang, C.; Takahashi, T.; Inoue, A.; Yan, H.; Kanai, M.; He, Y.; et al. Paper-based disposable molecular sensor constructed from oxide nanowires, cellulose nanofibers, and pencil-drawn electrodes. ACS Appl. Mater. Interfaces 2019, 11, 15044–15050. [Google Scholar] [CrossRef] [PubMed]
- Lu, T.; Pan, H.; Ma, J.; Li, Y.; Bokhari, S.W.; Jiang, X.; Zhu, S.; Zhang, D. Cellulose nanocrystals/polyacrylamide composites of high sensitivity and cycling performance to gauge humidity. ACS Appl. Mater. Interfaces 2017, 9, 18231–18237. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, L.; Zhou, J.; Lu, A. Flexible and transparent cellulose-based ionic film as a humidity sensor. ACS Appl. Mater. Interfaces 2020, 12, 7631–7638. [Google Scholar] [CrossRef]
- Tachibana, S.; Wang, Y.-F.; Sekine, T.; Takeda, Y.; Hong, J.; Yoshida, A.; Abe, M.; Miura, R.; Watanabe, Y.; Kumaki, D.; et al. A printed flexible humidity sensor with high sensitivity and fast response using a cellulose nanofiber/carbon black composite. ACS Appl. Mater. Interfaces 2022, 14, 5721–5728. [Google Scholar] [CrossRef]
- Wang, Y.; Hou, S.; Li, T.; Jin, S.; Shao, Y.; Yang, H.; Wu, D.; Dai, S.; Lu, Y.; Chen, S.; et al. Flexible capacitive humidity sensors based on ionic conductive wood-derived cellulose nanopapers. ACS Appl. Mater. Interfaces 2020, 12, 41896–41904. [Google Scholar] [CrossRef]
- Mahadeva, S.K.; Yun, S.; Kim, J. Flexible humidity and temperature sensor based on cellulose–polypyrrole nanocomposite. Sens. Actuators A 2011, 165, 194–199. [Google Scholar] [CrossRef]
- Huang, L.; Yang, Y.; Ti, P.; Su, G.; Yuan, Q. Graphene oxide quantum dots attached on wood-derived nanocellulose to fabricate a highly sensitive humidity sensor. Carbohydr. Polym. 2022, 288, 119312. [Google Scholar] [CrossRef]
- Molina-Lopez, F.; Briand, D.; de Rooij, N.F. All additive inkjet printed humidity sensors on plastic substrate. Sens. Actuators B 2012, 166–167, 212–222. [Google Scholar] [CrossRef]
- Kafy, A.; Akther, A.; Shishir, M.I.R.; Kim, H.C.; Yun, Y.; Kim, J. Cellulose nanocrystal/graphene oxide composite film as humidity sensor. Sens. Actuators A 2016, 247, 221–226. [Google Scholar] [CrossRef]
- Gaspar, C.; Olkkonen, J.; Passoja, S.; Smolander, M. Paper as active layer in inkjet-printed capacitive humidity sensors. Sensors 2017, 17, 1464. [Google Scholar] [CrossRef]
- Anju, V.P.; Jithesh, P.R.; Narayanankutty, S.K. A novel humidity and ammonia sensor based on nanofibers/polyaniline/polyvinyl alcohol. Sens. Actuators A 2019, 285, 35–44. [Google Scholar] [CrossRef]
- Yao, Y.; Huang, X.; Zhang, B.; Zhang, Z.; Hou, D.; Zhou, Z. Facile fabrication of high sensitivity cellulose nanocrystals based QCM humidity sensors with asymmetric electrode structure. Sens. Actuators B 2020, 302, 12719. [Google Scholar] [CrossRef]
- Zhang, S.; Zhang, Z.; Chen, S.; Zhu, R. Measurement of electric double layer capacitance using dielectrophoresis-based particle manipulation. Anal. Chem. 2021, 93, 5882–5889. [Google Scholar] [CrossRef] [PubMed]



| Sensitive Material | Sensitivity | Range (% RH) | Response/Recovery Time | Ref. |
|---|---|---|---|---|
| wood-derived cellulose nanopapers | - | 7–94 | ~20 s/~2 s | [18] |
| cellulose/PPy | - | 27.8–94.2 | ~418 s/~418 s | [19] |
| cellulose nanofiber/graphene oxide | - | 11–97 | 30 s/11 s | [20] |
| CAB | 2.36 ± 0.08 fF/% RH [a] | 10–70 | 24 ± 3 s/22 ± 4 s | [21] |
| CNC/GO | - | 25–90 | 90 min/35 min | [22] |
| cellulosic paper | 2 pF/% RH [a] | 40–100 | 4–5 min/- | [23] |
| CNF/PANI/PVA | 290–6570% [b] | 30–100 | 41 s/46 s | [24] |
| cellulose nanofiber/[BMIM]Cl | 27.95 pF/% RH [a] | 11–95 | 43 s/~1 s | this work |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hou, Z.; Ma, Z.; Guan, X.; Zhao, H.; Liu, S.; Fei, T.; Zhang, T. A Cellulose Nanofiber Capacitive Humidity Sensor with High Sensitivity and Fast Recovery Characteristics. Chemosensors 2022, 10, 464. https://doi.org/10.3390/chemosensors10110464
Hou Z, Ma Z, Guan X, Zhao H, Liu S, Fei T, Zhang T. A Cellulose Nanofiber Capacitive Humidity Sensor with High Sensitivity and Fast Recovery Characteristics. Chemosensors. 2022; 10(11):464. https://doi.org/10.3390/chemosensors10110464
Chicago/Turabian StyleHou, Zhaonan, Zhiyan Ma, Xin Guan, Hongran Zhao, Sen Liu, Teng Fei, and Tong Zhang. 2022. "A Cellulose Nanofiber Capacitive Humidity Sensor with High Sensitivity and Fast Recovery Characteristics" Chemosensors 10, no. 11: 464. https://doi.org/10.3390/chemosensors10110464
APA StyleHou, Z., Ma, Z., Guan, X., Zhao, H., Liu, S., Fei, T., & Zhang, T. (2022). A Cellulose Nanofiber Capacitive Humidity Sensor with High Sensitivity and Fast Recovery Characteristics. Chemosensors, 10(11), 464. https://doi.org/10.3390/chemosensors10110464





